Abstract
Conventional light microscopic evaluation of a semen ejaculate does not fully utilize potential indicators of functional impairment in sperm organelles. The technique described here facilitates critical evaluation of morphological features of spermatozoal organelles at an ultrastructural level and helps identify vulnerable targets. Compared with a battery of sperm function assays employed in andrology clinics, this relatively less expensive technique efficiently uses semen as biopsy material and thus serves as a comprehensive means to assess the impact of toxicants on the male reproductive system.
Introduction
A variety of toxicants exposed either in experimental paradigms or contaminated environments are known to adversely affect spermatogenesis quantitatively as well as qualitatively. The impact is usually determined by histological evaluations of seminiferous epithelium or quantification of sperm in tail portion of the epididymis or ejaculated semen. Likewise, normalcy of shape of sperm and the structural integrity and functional parameters such as motility are routinely evaluated using various light microscopic techniques. A mature human spermatozoon consists of the head region (nucleus containing twenty-three chromosomes in a condensed chromatin form surrounded by an acrosomal cap), a mid region (containing axoneme, outer dense fibers, and mitochondria, which provide adenosine triphosphate that supports motility), and a tail region (principal piece consisting of contractile proteins). All of these segments are surrounded by a plasma membrane. Structural integrity of all of these components—plasma membrane, acrosome, nuclear chromatin, and axoneme—are pivotal in accomplishing fertilization. It is beyond the scope of this article to comprehensively review the biological attributes of these individual components.
To the extent that structure reflects function or vice versa, not all structurally normal sperm are functionally normal—for example, not all morphologically normal sperm are motile and not all motile sperm are morphologically normal. In the latter case, while mitochondrial sheath in the sperm flagellum is normal, the other spermatozoal organelles might be defective. In such instances, a battery of biochemical analyses is used to evaluate functional competence of individual spermatozoal organelles, especially in clinical investigations. This elaborate approach has several limitations in experimental investigations as these individual analyses are labor intensive and are restricted by time constraints because of the necessity to obtain a freshly collected semen sample for each test.
The purpose of this article is to present an approach that emphasizes the use of semen sample (in case of rodents, a cauda epididymal luminal fluid) as a biopsy material so as to discern the morphological aberrations of the seminal components and associate the malformations to various phases of spermatogenesis, spermiation, and post-testicular sperm transit and maturation. This relatively simple morphological procedure circumvents functional evaluations and yet facilitates a routine differential analysis as well as a critical evaluation of multiple spermatozoal organelles; the former using a light microscope and the latter using a transmission electron microscope. Furthermore, this procedure is useful in characterizing any pathologic condition in the reproductive tract as it enables identification and characterization of any exfoliated cells along with the germ cells in the ejaculate. In illustrating this method, examples from several reproductive toxicologic studies performed in the author’s laboratory using rabbits as an animal model are presented; subtle lesions in germ cells that are not discernible ordinarily at a light microscopic level are emphasized. In addition, some of these unique lesions induced by toxicants are compared with those manifested in idiopathic cases of subfertile stallions so as to draw attention that these clinical situations may not always be “idiopathic,” as infertility is often dubbed, but might have been cases that were inadvertently/unwittingly subjected to environmental exposures.
Why Use Transmission Electron Microscopy?
Routine light microscopy does not permit characterization of subtle lesions in spermatozoal organelles that are critical to ensure fertilization of ovum. Also, using light microscopic techniques, cellular detritus that may be present in semen consequent to a toxicologic insult or a pathologic condition cannot be discerned to identify the origin of the lesion and is usually discounted as debris. This “debris” may provide useful information pertinent to the underlying pathological condition and it takes transmission electron microscopy (TEM) to optimally utilize semen sample as biopsy material. In other words, utilizing this technique, in addition to identifying subtle lesions in critical spermatozoal organelles, one can identify prematurely desquamated germ cells, denuded fragments of Sertoli cells that support differentiation of germ cells, and any epithelial fragments from excurrent ducts and accessory glands.
Procedures
Processing of Semen Sample
An aliquot of gel-free semen is fixed in 10× the volume of cacodylate-buffered 6% glutaraldehyde (in 0.1 M sodium cacodylate buffer, pH 7.4, with 0.15 M sucrose). The fixed sample is centrifuged at 300× g for 5 min. The supernatant is poured off, and the pellet is suspended for 15 min in 0.1 M sodium cacodylate buffer. This procedure is repeated twice to remove fixative.
The sediment is resuspended in buffer, and two drops (∼100-µl aliquot) of the suspension is added to 1 ml of distilled water in a glass vial. A wet smear is freshly prepared by placing a drop of this suspension on a glass slide and placing a cover slip for phase-contrast or differential interference-contrast (Nomarski) microscopy. Dry smears are also prepared by placing a drop of the suspension on a glass slide and distributing the fluid evenly with a smooth glass rod or the tip of a long Pasteur pipette. The slide is placed on a heating block at 80°C, stained with 0.5% toluidine blue in 1% sodium borate for 45 sec, rinsed with distilled water, and allowed to dry. Cover slips are mounted using Permount and are used for bright-field microscopy.
The remaining suspension is washed once more in sodium cacodylate buffer as already described, the supernatant poured off, and the pellet processed for TEM. The pellet is post-fixed in a solution of 1% osmium tetroxide in 0.1 M cacodylate buffer for 90 min and centrifuged, washed thrice in cacodylate buffer. Osmicated pellets are dehydrated through a graded series of ethanol (15 min each in 50%, 70%, 95% × 3, and 100% × 3), rinsed in propylene oxide (15 min × 3), and embedded in Poly/Bed 812 (Polysciences Inc., Warrington, PA) as follows: 50:50 propylene oxide: Poly/Bed at ambient temperature overnight; 100% Poly/Bed in vacuum for 8 hr, 100% Poly/Bed in vacuum for 16 hr, in 45°C oven for 8 hr, and in 60°C oven for 16 hr. From these plastic tissue blocks, 1-µm-thick sections are cut, placed on glass slides, and stained with toluidine blue for characterization of exfoliated cells using a light microscope. Then, 60- to 80-nm thin sections are cut, mounted on 300-mesh nickel grids, and stained with uranyl acetate (8g/100 ml of 75% ethanol) and lead citrate for ultrastructural evaluation of spermatozoal organelles and other cellular constituents using a transmission electron microscope.
Light and Transmission Electron Microscopic Evaluation
At the outset, it should be recognized that the use of light microscopic and TEM techniques is complementary to each other. The scope of this article precludes detailed description of the morphometrics used for evaluation of sperm using light and transmission electron microscopy. The reader may use a variety of analytical/quantification methods to meet individual needs, but some of the procedures used in the author’s laboratory can be found in several publications (Seed et al. 1996; Veeramachaneni 2011; Veeramachaneni et al. 1993, 2006; Veeramachaneni, Moeller, and Sawyer 2006; Veeramachaneni and Sawyer 1996).
It is recommended that ideally a light microscope equipped with planapochromatic as well as phase-contrast objective lenses together with a universal condenser that enables bright-field, phase-contrast, and differential interference-contrast microscopy be used. It is preferable that the microscope is interfaced with a computerized imaging system and a data collection spreadsheet. Utilization of toluidine blue-stained smears (bright field microscopy) as well as unstained wet smears (differential interference-contrast and phase-contrast microscopy) enables compensating deficits inherent to these procedures (e.g., artifacts associated with smear preparation, resolution of subtle defects) and thus enhances critical evaluation.
As for the quality of a transmission electron microscope for this purpose, an instrument with a resolution of ∼1 nm is sufficient, as the best resolution attainable with ultrathin sections of plastic-embedded material is about 2 nm. Although quantification of morphological defects is not practical at an ultrastructural level, TEM does elucidate light microscopic findings in characterizing ultrastructural lesions in plasma membranes and acrosomes as well as intracellular inclusions. It is this detail that facilitates identifying the subcellular targets of a toxicant or even establishing the origin of a tumor. It should be reiterated that a variety of biochemical and immunocytochemical markers are available to assess the functional status of the sperm organelles discussed in the following, but the ultrastructural approach described here provides a more global picture. To illustrate this concept, a few examples including unique lesions that can only be detected using TEM are presented next.
Illustrations
Mitochondrial Lesions
Because mitochondria provide adenosine triphosphate as a source of energy for the sliding of axonemal microtubules facilitating movement of sperm flagellum, any disruption of mitochondrial structure and function impairs sperm motility. Oxidative stress resulting from a variety of adverse factors including chemical pollutants can damage mitochondria; for example, polychlorinated biphenyl (PCB) exposure has been shown in numerous studies to compromise sperm quality, namely, sperm motility (Hauser 2006). Similarly increased urinary concentrations of monoesters and oxidative metabolites of phthalates have been associated with lowered sperm motility (Hauser et al. 2006). While it is difficult to associate structural deficits in mitochondria to a single chemical exposure, swelling of mitochondrial matrix and disruption of cristae is a commonly observed ultrastructural lesion that can only be discerned with TEM (Fig. 1). Mitochondria in mid-pieces of spermatozoa. Transmission electron micrographs of normal (A) and abnormal (B) mitochondria in mid-pieces. In panel B, note conspicuous swelling of mitochondrial matrix and disorganized cristae (open arrows) compared with fairly uniform electron-dense mitochondria (black arrow) in the mid-piece in panel A. A segment of mid-piece is seen having no mitochondrial at all (gray arrow). A variety of toxicants can cause mitochondrial lesions and dysfunction. Also seen are swollen acrosomal and plasma membranes disrupting their normal placement (arrow head). Proximal centriole also appears disorganized. It is not uncommon to find multiple defects of organelles in a single sperm. Scale bars = A and B: 1 µm.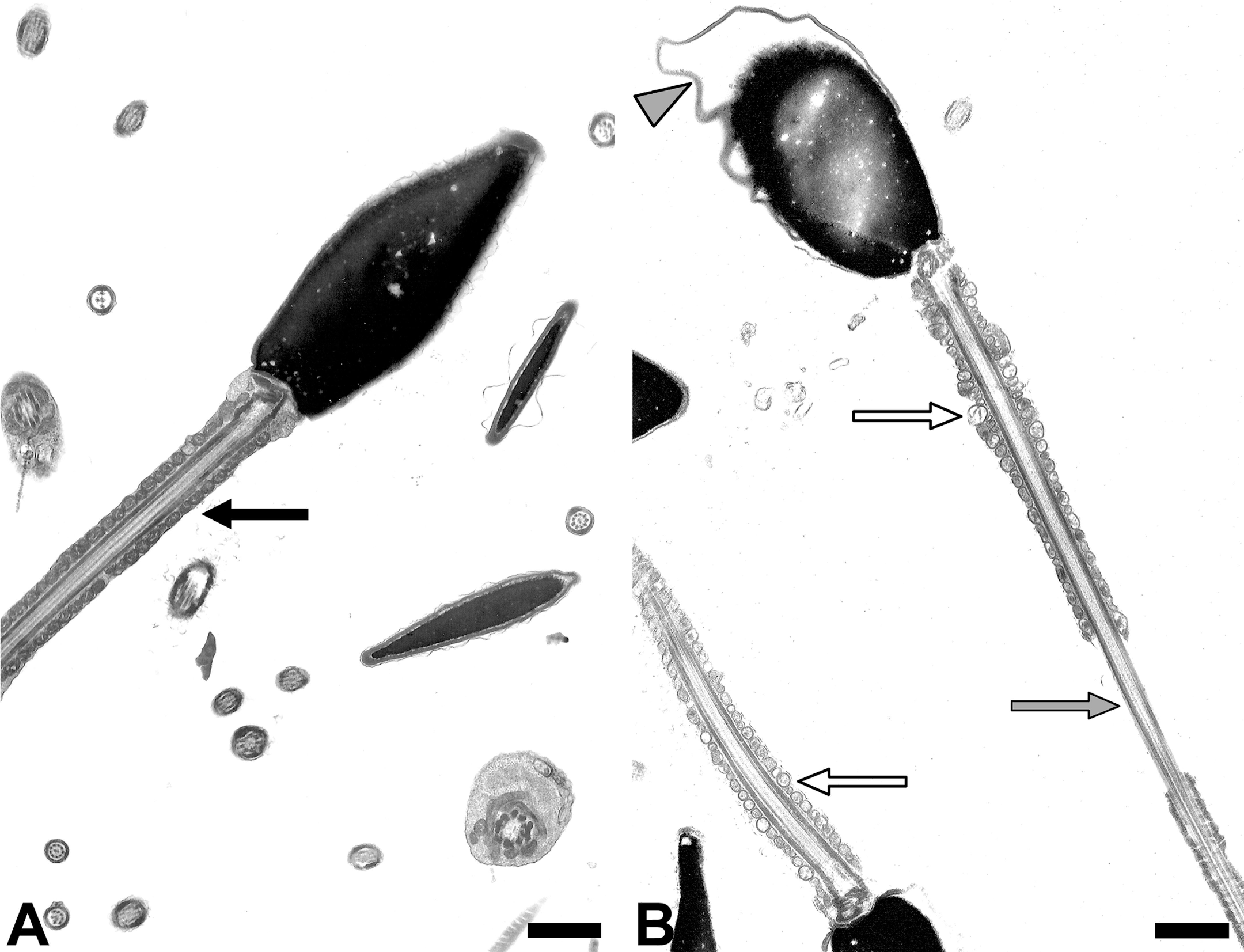
Aberrations in Plasma and Acrosomal Membranes
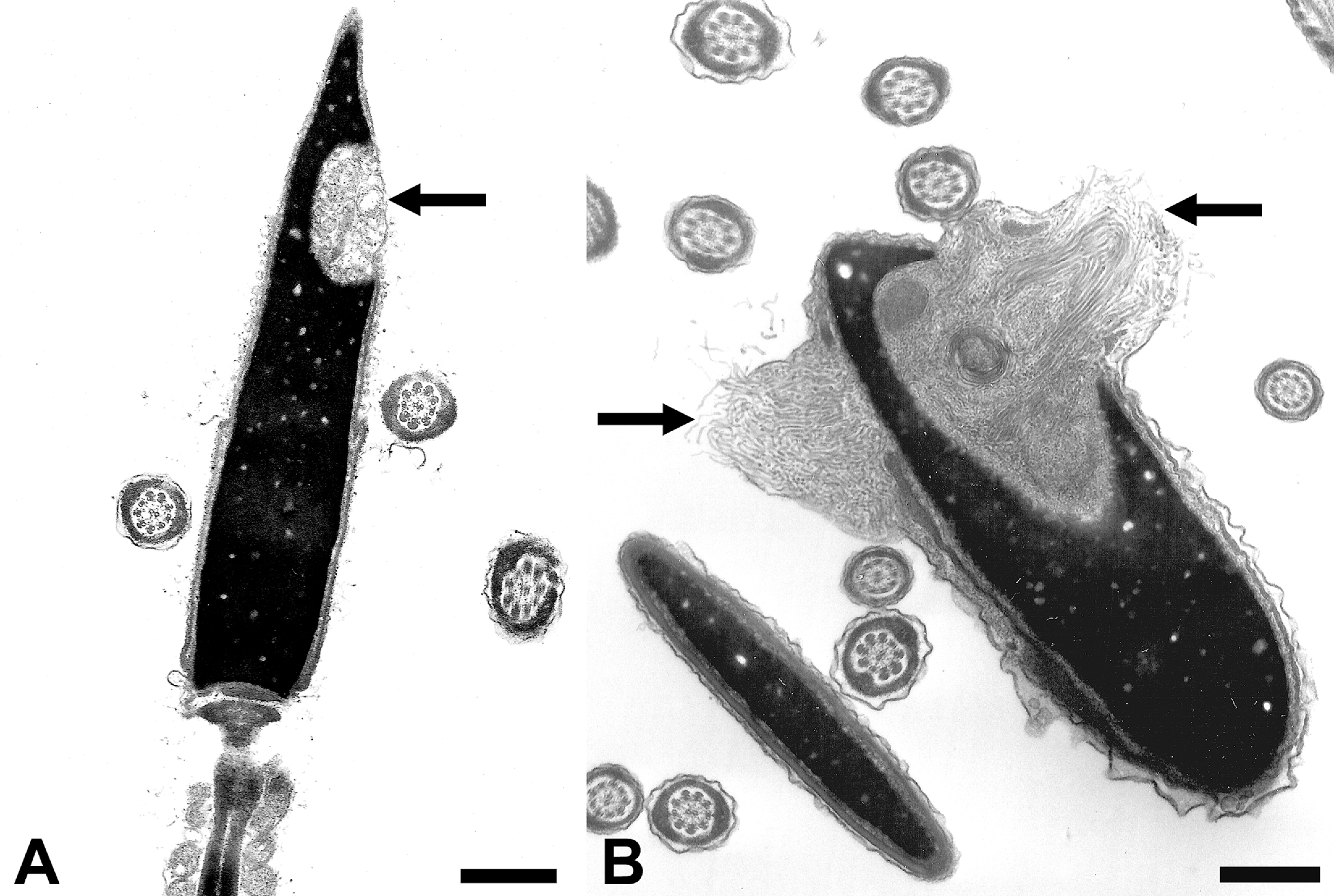
The three sequential events during fertilization are (i) acrosome reaction (fusion of plasma membrane and outer acrosomal membrane releasing the acrosomal enzymes that facilitate penetration of corona radiata of ovulated egg), (ii) sperm binding to zp3 (a glycoprotein of the zona pellucida of the ovum), and (iii) sperm-egg fusion. A variety of pollutants including cigarette smoke have been implicated in premature acrosomal reaction. Normally, acrosome reaction occurs in the female reproductive tract when sperm are in contact with corona radiata. Thus, premature acrosomal reaction in the male reproductive tract itself (Fig. 2) before reaching the vicinity of the egg in the oviduct impairs the ability of sperm to penetrate the vestments of the egg. Likewise, loss of sperm plasma membrane (where receptors for zp3 protein are present) precludes binding of the sperm nucleus to that of the egg and hence fertilization.
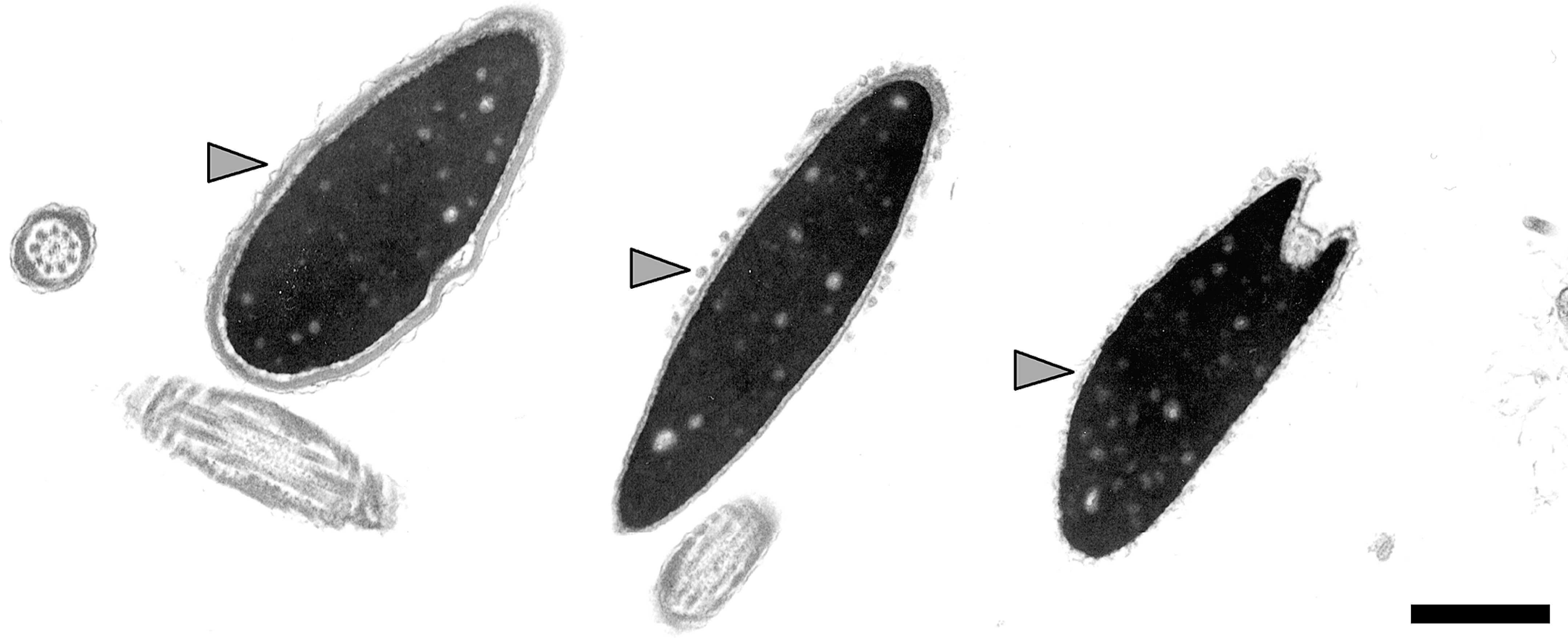
Plasma and acrosomal membranes of sperm nuclei. Transmission electron micrographs of normal, intact plasma and acrosomal membranes (left), acrosome undergoing acrosomal reaction (fusion of the outer acrosomal membrane with plasma membrane forming openings; middle), and acrosomal reaction almost complete (acrosomal vesicles mostly have disappeared; right). These changes in sperm nuclear vestments can only be discerned at an ultrastructural level. Scale bar = 1 µm.
Acrosomal Dysgenesis
Considering the pivotal role that an acrosome plays in accomplishing fertilization, it is imperative that the morphogenesis of acrosome occurs unperturbed. A spectrum of malformations of acrosomes—acrosomal dysplasia, rarefaction, vesiculation, and sharing between two or more sperm—are induced by environmental toxicants. Consequently, it is not uncommon to encounter acrosomal abnormalities such as shared acrosomes in idiopathic cases of subfertile domesticated animals (Fig. 3A). These unique forms of malformations can be induced by a variety of toxicants; for example, common chemical contaminants in drinking water (a mixture of industrial chemicals found in ground water near Superfund sites; Veeramachaneni, Palmer, and Amann 2001), phthalates (dibutyl phthalate; Higuchi et al. 2003), water disinfection by-products (dibromoacetic acid; Veeramachaneni, Palmer, and Klinefelter 2007), and pesticides (anti-androgenic insecticide DDT and the fungicide vinclozolin; Veeramachaneni et al. 2006, 2007). In fact, the morphogenesis of this defect can be charted developmentally following experimental exposure to these toxicants (Fig. 3B and 3C). Interestingly, some of these acrosomal malformations seem to perpetuate, indicating a possible epigenetic effect on stem germ cells (Veeramachaneni 2008).
Acrosomal dysgenesis—shared acrosomes. A: Transmission electron micrograph of two conjoined sperm nuclei in ejaculated equine semen. Note presence of a dysplastic acrosome (arrows) shared between two sperm heads. B and C: Morphogenesis of this unique malformation in rabbits exposed developmentally to anti-androgenic pesticides. As two adjacent round spermatids differentiate, they share a common golgi apparatus (B) and acrosomic vesicle (C), which spreads around both spermatidal nuclei as they condense and elongate resulting in conjoined sperm. These unique defects appear to result from developmental exposure to a variety of environmental pollutants. Scale bars = A, B, and C: 2 µm.
Nuclear Deformities: Defective Chromatin Condensation and Nuclear Inclusions
The third event in fertilization—sperm-egg fusion—and consequent formation of pronuclei and assembly of first mitotic spindle depends upon normally packaged sperm nuclear chromatin and contribution of a normal centriole by the sperm. Defects in chromatin condensation (Fig. 4) and decondensation mechanisms and nuclear shaping can interfere with this event. A variety of physical (e.g., heat) and chemical (e.g., PCBs, DDE) agents can disrupt these processes (Rignell-Hydborn et al. 2005).
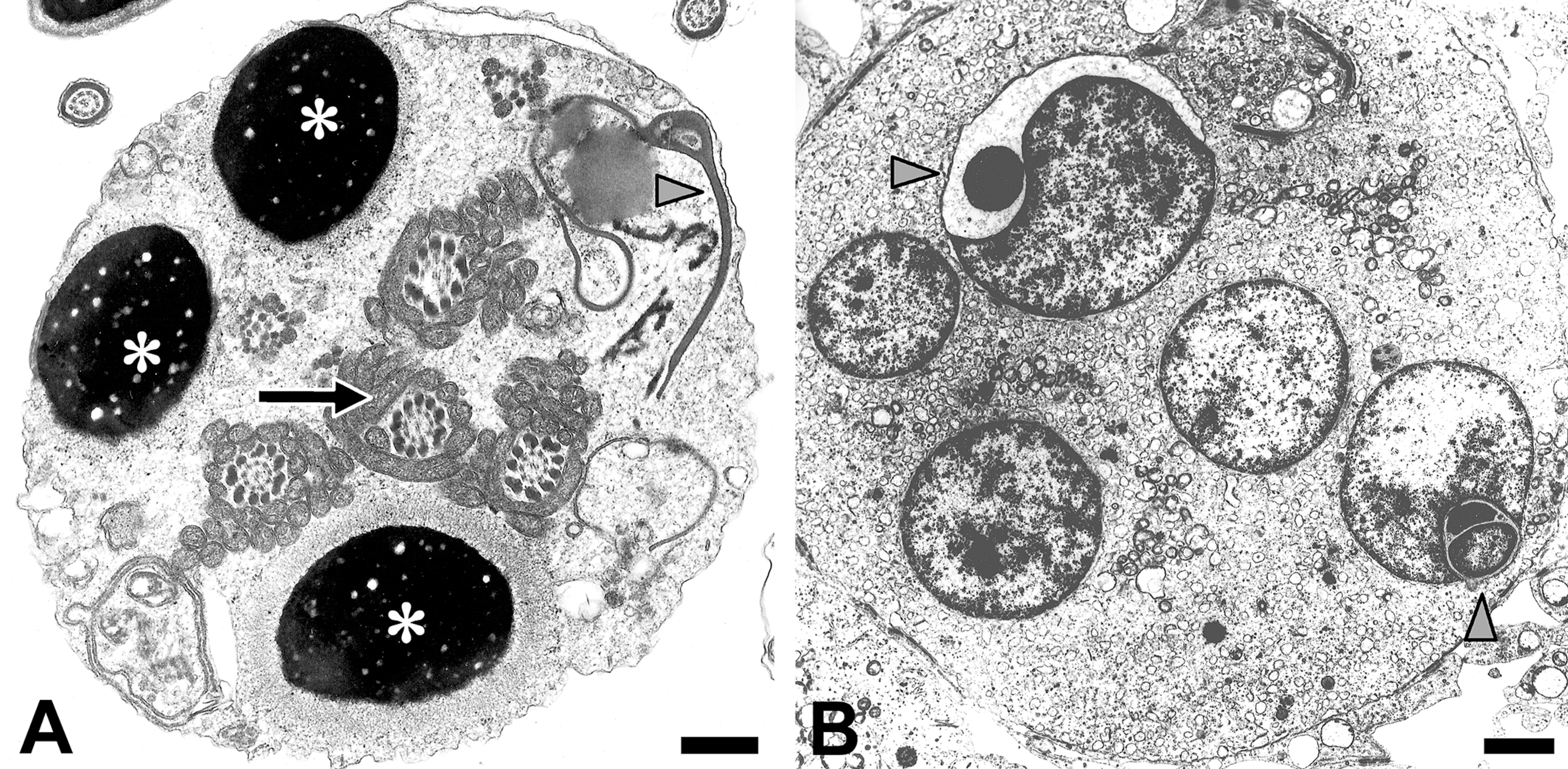
Defective nuclear compaction—incomplete chromatin condensation. Photomicrograph of chromatin clumps resulting from breakage of incompletely condensed equine sperm nuclei. A: Toluidine blue-stained dry smear. Note darkly stained clumps of chromatin (asterisks), which are commonly ignored as debris or stain deposits at a light microscopic level. B: Transmission electron micrograph of four incompletely condensed sperm nuclei with ruptured plasma membranes (arrows). Note flocculent chromatin fragments (asterisk) in the upper right corner. Impaired chromatin condensation and breakage can be induced by exposure to a variety of environmental factors, including physical and chemical agents. Along with fragmented chromatin of sperm nuclei, nuclei of differentiating germ cells are also affected. The identity of these prematurely shed germ cells is difficult to discern at a light microscopic level (panel A; arrow). Scale bars = A:10 µm; B: 1 µm.
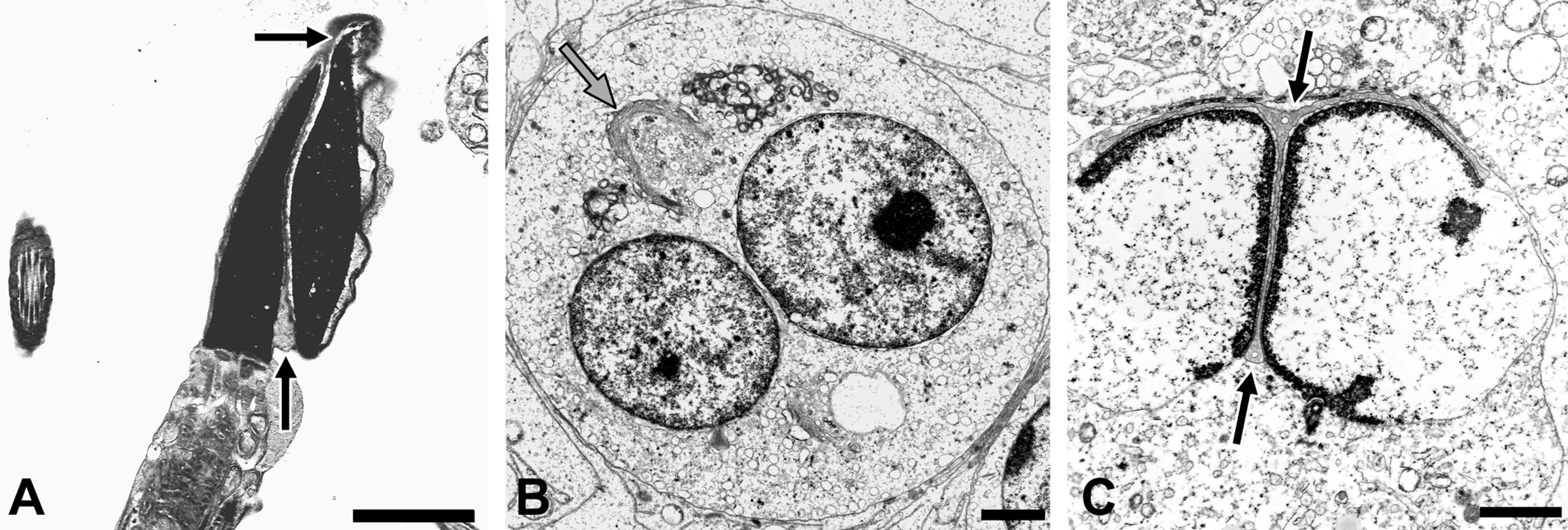
In addition to the defects in chromatin packaging, nuclear deformities can also result from aberrations during spermiogenesis; for example, invasion of the cytoplasm of the supporting somatic cell, the Sertoli cell into the spermatid nucleus (Fig. 5A), or invasion of abnormally proliferated microtubules (Fig. 5B) from manchette, a microtubular organelle important in shaping the spermatozoon. Once found, this microtubular mass defect continues to manifest, suggesting that it is a developmental defect affecting stem cells (Veeramachaneni and Sawyer 1996; Veeramachaneni 2011). Although a clear etiology is not known, this defect is empirically associated with exposure to anabolic steroids.
Nuclear inclusions—cytoplasmic vesicles and microtubule masses. Transmission electron micrographs of equine sperm nuclei containing cytoplasmic vesicles (A) and masses of microtubules (B). A whorl of aberrant microtubule mass is also seen protruding from nuclear and plasma membranes. In contrast to the cytoplasmic inclusion seen in panel A, which might arise from Sertoli cell, the inclusions in panel B involve proliferation of abnormal microtubules possibly originating from sites associated with the manchette, a microtubular organelle important in shaping the spermatozoon. These subtle differences between cytoplasmic and microtubular inclusions cannot be discerned with light microscopy. Scale bars = A and B: 0.5 µm.
Cellular Detritus in Semen: Denuded Cells from Testes and Accessory Sex Organs
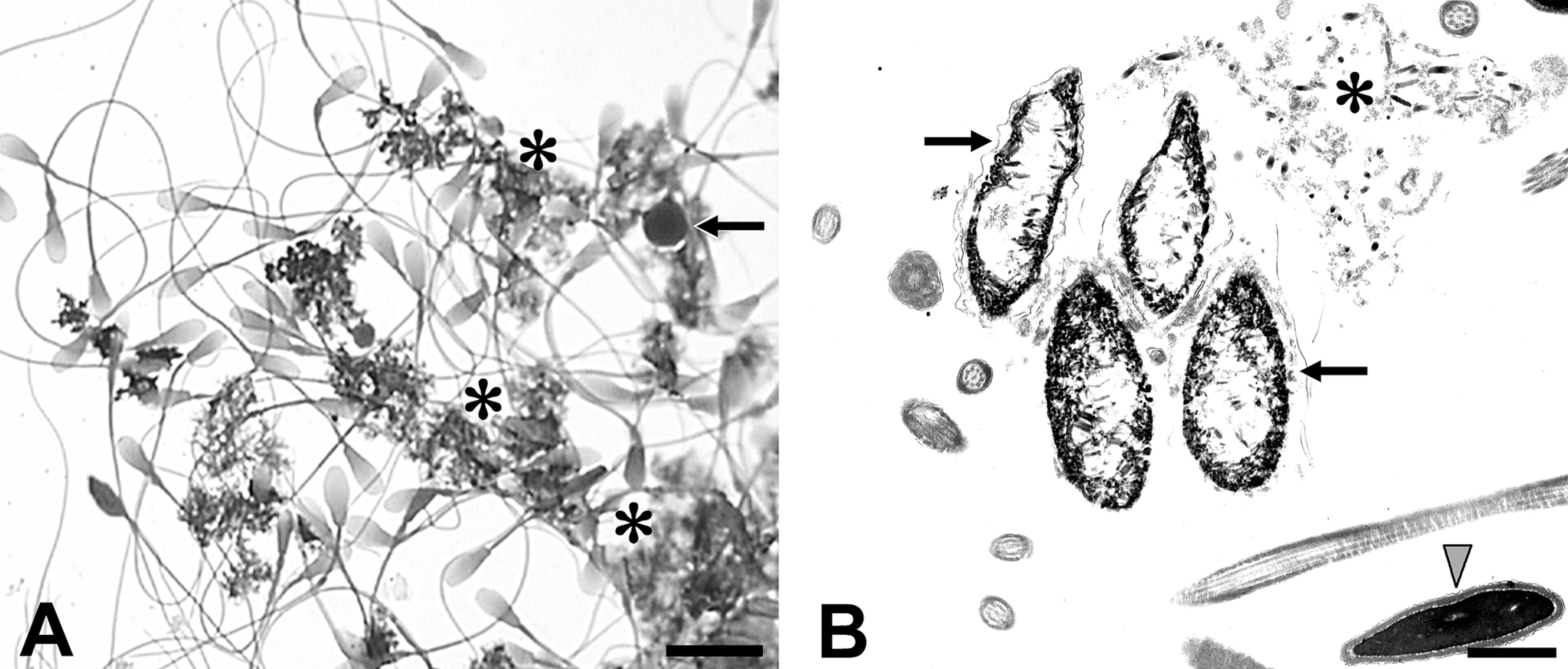
In disease conditions it is not uncommon to find cells and cellular debris in ejaculates other than spermatozoa. Depending upon the nature and severity of the insult, blood elements and even pathogens can be found. Although presence of degenerate Sertoli cell fragments enclosing differentiating germ cells (Fig. 6) are more common in disease conditions, epithelial cells lining the excurrent ducts or accessory organs (Fig. 7) also are found. A wide variety of chemicals such as 2,5-hexanedione, carbendazim, and mono-(2-ethylhexyl) phthalate are known to target the Sertoli cell causing seminiferous epithelial sloughing, vacuolization, spermatid head retention, germ cell apoptosis, and altered microtubule assembly (Moffit et al. 2007). At higher doses, some of these chemicals (e.g., carbendazim and its parent chemical, the fungicide benomyl) cause extensive sloughing of Sertoli cells and germ cells occluding sperm tansit in the proximal excurrent ducts (Hess and Nakai 2000), thereby inducing secondary lesions in these regions resulting in exfoliation of lining epithelial cells. Exposures to this type of chemicals may occur both occupationally and environmentally. During routine light microscopic evaluations, the source of these somatic cell components cannot be discerned and usually are discounted as innocuous debris. When the exfoliation is extensive, the milieu (e.g., pH) of seminal plasma is altered, causing changes in plasma and acrosomal membranes ensuing in premature acrosomal reaction. Such subtle changes in membranes as well as lesions in cytoskeletal components can be resolved only by TEM.
Cohorts of malformed spermatids in seminiferous epithelium. A: Transmission electron micrograph of an exfoliated Sertoli cell fragment in the ejaculate of a stallion. Note presence of vacuolations in three spermatid nuclei indicating incomplete condensation of chromatin (asterisks). Also seen are four mid-pieces with aberrant organizational pattern of mitochondria (arrow) and isolated dysplastic acrosomes (arrow head). B: Transmission electron micrograph of a testicular section from a vinclozolin-exposed rabbit showing acrosomal-nuclear dysgenesis (arrow heads). Several spermatid nuclei are enclosed in the same cell membrane, indicating that they belong to a syncytium. This so-called multinucleated giant cell shows dysplastic acrosomes invading at least two deformed spermatid nuclei (in this plane of section), indicating that such defects originate from a common progenitor spermatogonium. Scale bars = A: 0.5 µm; B: 2 µm.
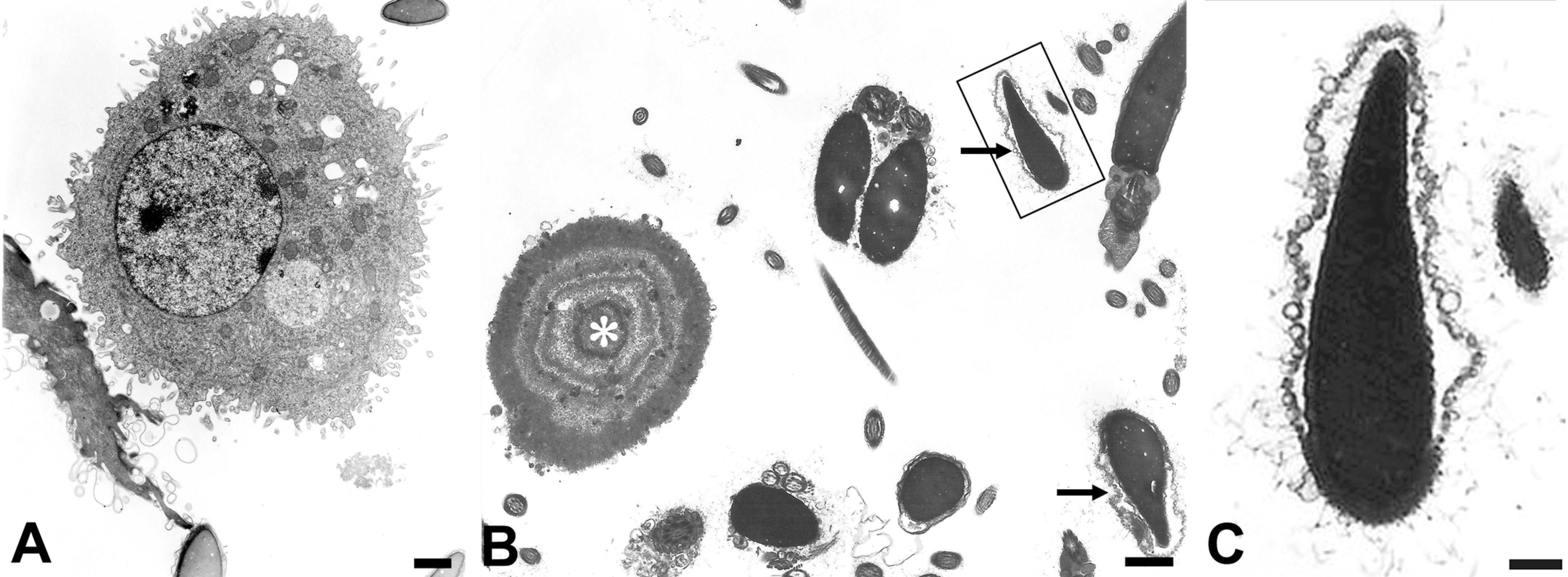
Cellular detritus from accessory organs present in semen. A: Transmission electron micrograph of an exfoliated epididymal epithelial cell. The origin of such denuded cells can be identified only by ultrastructural features of lining epithelium characteristic to the particular region of excurrent ducts. B: Transmission electron micrograph of a prostatic concretion (asterisk) and several profiles of acrosome-reacted sperm. When semen ejaculates contain degenerate material shed from excurrent ducts and accessory sex glands, the seminal plasma milieu is altered, resulting in premature acrosomal reaction (arrows). C: The acrosome-reacted sperm in the boxed profile in panel B is shown at a higher magnification. Definitive identification of prematurely shed germ cells and characterization of somatic cells is possible only by transmission electron microscopy (TEM). Scale bars = A, B, and C: 1 µm.
Germ Cell Atypia: Intratubular Germ Cell Neoplasia/Carcinoma in Situ
In addition to disruptive effects on spermiogenesis, some toxicants may also adversely affect differentiation of stem germ cells producing atypical germ cells with features characteristic of carcinoma in situ (CIS) (Veeramachaneni 2008). CIS cells have been demonstrated in adult rabbits that were developmentally exposed to dibutyl phthalate (Higuchi et al. 2003) or DDT either by itself or as a mixture with vinclozolin (Veeramachaneni et al. 2007). CIS, also known as intratubular germ cell neoplasia, is a pre-invasive precursor of testicular germ cell tumors, the most common cancer type of human male adolescents and young adults (Loveland, Rajpert-DeMeyts, and Veeramachaneni 2010). CIS also has been associated with some forms of impaired spermatogenesis in humans. Importantly, presence of exfoliated cells that are positive for CIS-markers have been documented in human semen (Hoei-Hansen et al. 2007), indicating that a seminal sample could be used as biopsy material and that it is possible to detect these atypical cells by their ultrastructural features using TEM (Fig. 8). Thus, ultrastructural evaluation of semen not only offers a valuable tool to assess the impact of exposure to toxicants but also serves as a noninvasive method for detection of testicular neoplasia.
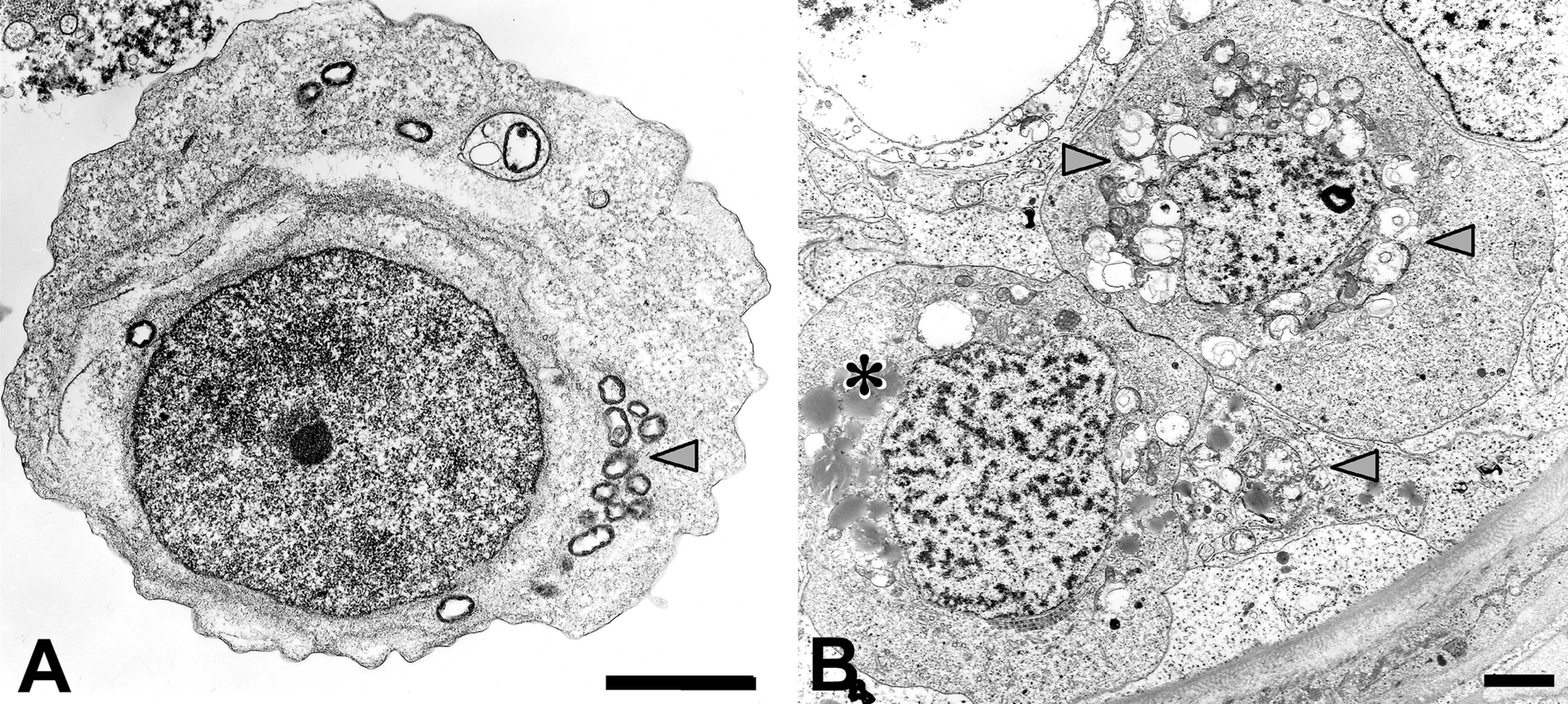
Germ cell atypia: Carcinoma in situ or intratubular germ cell neoplasia. A: Transmission electron micrograph of a prematurely shed abnormal germ cell in ejaculate. Note swollen mitochondria (arrow head) and glycogen aggregates in the cytoplasm. It is not uncommon to find atypical germ cells resembling neoplastic germ cells (called carcinoma in situ; CIS) or intratubular germ cell neoplasia, the precursor cells of testicular germ cell tumors, in semen. B: Transmission electron micrograph of two CIS cells in the testis of a rabbit exposed to DDT. Note irregular nuclear contours and swollen mitochondria (arrow heads) in addition to accumulation of lipid (asterisk) and glycogen in the cytoplasm. These ultrastructural features are not typical of normally differentiating germ cells and have been attributed to neoplastic germ cells in humans. Scale bars = A and B: 2 µm.
Take-Home Message
Conventional light microscopic evaluation of a semen ejaculate does not fully utilize potential indicators of functional impairment in sperm organelles. The technique described here facilitates critical evaluation of morphological features of spermatozoal organelles at an ultrastructural level and helps identify vulnerable targets. Compared with a battery of sperm function assays employed in andrology clinics, this relatively less expensive technique efficiently uses semen as biopsy material and thus serves as a comprehensive means to assess the impact of toxicants on the male reproductive system.
Footnotes
Acknowledgments
The author gratefully acknowledges technical assistance of Carol Moeller and Jennifer Palmer and thanks Dr. Karamjeet Pandher for his efforts in organizing this symposium.
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIEHS Superfund P42-ES-05949 and R21-ES-014607; USEPA STAR Grants R826131 and R829429 and Co-op RFA#R826465; and Colorado State University College of Veterinary Medicine and Biomedical Sciences Research Council.
