Abstract
Perfluorinated compounds (PFCs) are environmentally widespread, persistent, and bioaccumulative chemicals with multiple toxicities reported in experimental models and wildlife, including immunomodulation. The two most commonly detected compounds, which also generally occur in the highest concentrations in environmentally exposed organisms, are perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS). PFOA and PFOS have been reported to alter inflammatory responses, production of cytokines, and adaptive and innate immune responses in rodent models, avian models, reptilian models, and mammalian and nonmammalian wildlife. Mounting evidence suggests that immune effects in laboratory animal models occur at serum concentrations below, within the reported range, or just above those reported for highly exposed humans and wildlife. Thus, the risk of immune effects for humans and wildlife exposed to PFCs cannot be discounted, especially when bioaccumulation and exposure to multiple PFCs are considered. This review contains brief descriptions of current and recently published work exploring immunomodulation by PFOA, PFOS, and other PFCs in rodent models, alternative laboratory models, and wildlife.
Keywords
Introduction
Perfluorinated compounds (PFCs) are used to manufacture myriad consumer products, from adhesives and water- and stain-repellent surfaces to nonstick coatings and lubricants. Two major classes are the perfluorinated carboxylates (i.e., perfluorooctanoic acid [PFOA]) and the perfluorinated sulfonates (i.e., perfluorooctane sulfonic acid [PFOS]), which can be grouped as perfluorinated alkyl acids (PFAAs). Certain perfluorinated precursors undergo chemical, microbial, and photolytic degradation to a limited number of extremely stable degradation products that include other PFCs. As a result of their widespread use and the long half-life of their degradation products, PFCs are pervasive in the environment, and several homologues have been reported in the serum and tissues of humans and wildlife (Lau et al. 2007). Concerns about the toxicological effects of PFCs, notably PFOS and PFOA, prompted several actions by the U.S. Environmental Protection Agency (USEPA), including a draft human health risk assessment of PFOA and the creation of significant new use rules for PFOS and related sulfonates. By 2002, PFOS production was voluntarily phased out by its major manufacturer. An additional agreement for PFOA will result in elimination of PFOA from emissions and products produced by six major manufacturers by 2015 (USEPA 2010). Although these voluntary measures by the companies that manufacture PFCs will and have reduced emissions of PFOS and PFOA into the environment, the stability of these compounds ensures that they will have a continued presence in environmental and biological media.
General toxicological findings associated with laboratory animals exposed to PFOA or PFOS include hepatomegaly and hepatic peroxisome proliferation; liver, testicular (Leydig cell), and pancreatic (acinar cell) tumors; reproductive and developmental deficits; neurotoxicity; and immunotoxicity. This review briefly summarizes previously published data and presents new, unpublished findings associated with the immunotoxicity of PFOA, PFOS, and other PFCs of environmental concern. The body of evidence concerning the ability of PFCs to modulate the immune system has grown since the first review of PFC immunotoxicity (DeWitt, et al. 2009), and several studies have reported serum concentrations in rodent models at immune effect levels within the range of human and wildlife exposures, suggesting risk of immunomodulation in humans and wildlife. In addition, we have included data from studies of nontraditional laboratory models and effects detected in wild-living species.
The first investigations specific to the immune effects of PFOA were published in the early 2000s (Yang et al. 2002; Yang, Xie, and Depierre 2000; Yang et al. 2001), and the first report of PFOS immunotoxicity was published in 2007 (Peden-Adams et al. 2007). In each of these studies, reductions in lymphoid organ weights, lymphoid cell numbers, and de novo antibody synthesis were demonstrated after oral (dietary or gavage) exposure. However, these were not the first studies to report the immunotoxicity of PFCs. Before PFCs became contaminants of environmental concern, several homologues were assessed for their ability to serve as artificial oxygen carriers, drug delivery vehicles, and contrast agents. In fact, a 20% emulsion of PFCs (Fluosol-DA, comprised, in part, of perfluorodecalin and perfluorotripolyamine) was approved by the Food and Drug Administration (FDA) in 1989 as an artificial oxygen carrier for intracoronary use but was withdrawn from the market in the mid-1990s because of cumbersome preparation (Spiess 2009). Studies of the immunotoxicity of Fluosol-DA demonstrated that its effect on antibody production was highly variable and dependent on species, dose, route of exposure, and timing of administration; however, one study did report that a single dose given to female Wistar rats or female NIH mice increased lymphoid organ weights and antigen-specific immunoglobulin M (IgM) antibody responses (Lowe and Bollands 1988). Second-generation artificial oxygen carriers contain higher concentrations of PFCs, but they have not yet received FDA approval. One particular study with perflubron, an agent composed of 60% PFCs, reported no statistical changes in delayed hypersensitivity, lymphocyte stimulation, immunoglobulin levels, and the levels of several cytokines in human volunteers given a single dose of the compound (Noveck et al. 2000). While this class of PFCs does not seem to modulate the immune system at anticipated medicinal doses, accumulating evidence suggests that the class of PFCs found in the environment does modulate the immune system at doses comparable with serum concentrations found in humans and wildlife.
Serum Concentrations
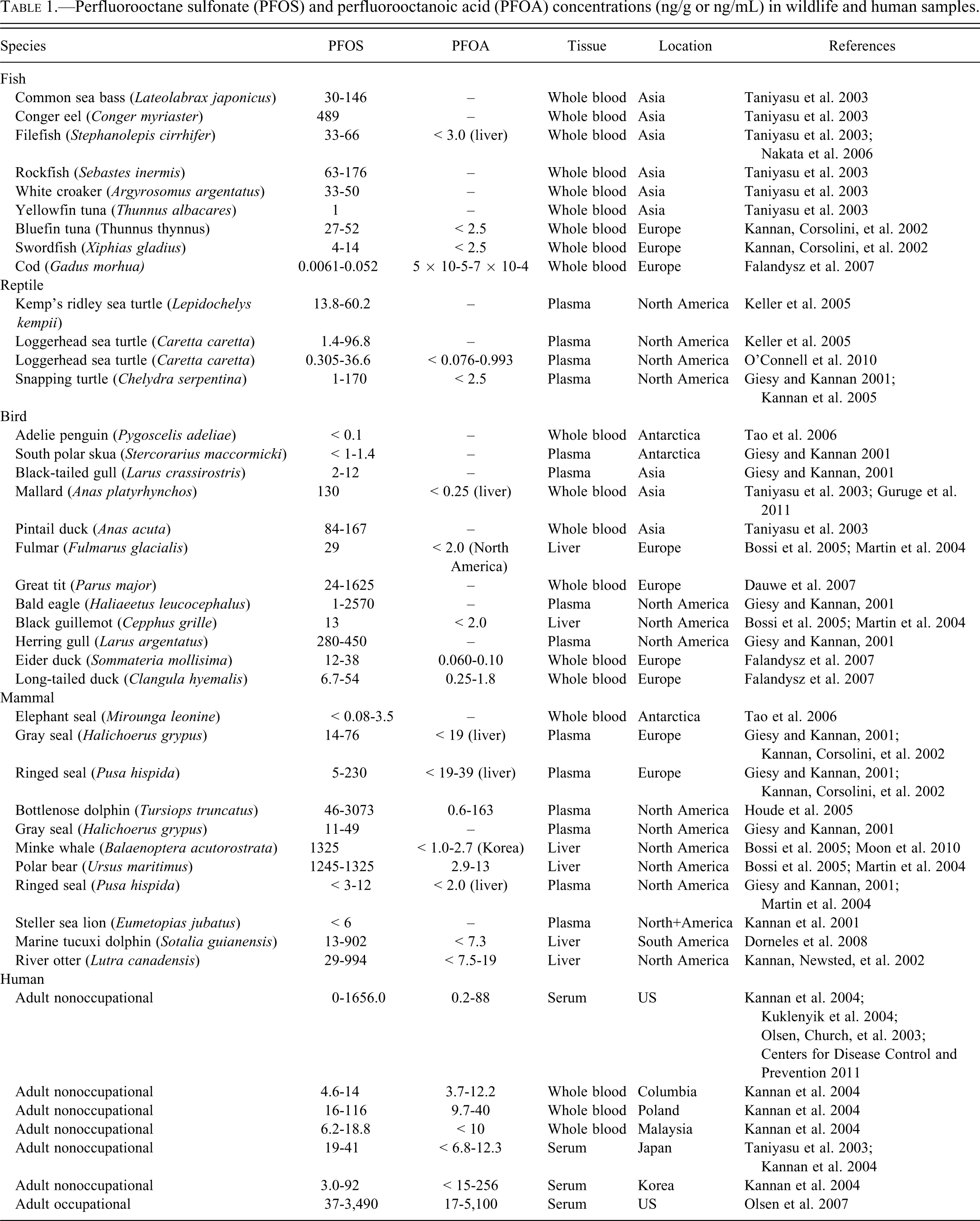
Through the National Health and Nutrition Examination Survey, the Centers for Disease Control and Prevention (2011) measured 219 environmental chemicals in the blood or urine of participants in the survey. For this sample representative of the U.S. population, 12 PFCs were measured. In 2005-2006, the median PFOA serum concentration was 4.2 ng/mL (3.8-4.5 ng/mL 95% confidence interval), and the median PFOS serum concentration was 17.5 ng/mL (16.8-18.6 ng/mL 95% confidence interval), representing the highest PFC concentrations, followed by perfluorohexane sulfonic acid, with a median concentration of 1.8 ng/mL. Perfluorononanoic acid (PFNA), 2-(N-methyl-perfluorooctane sulfonamido) acetic acid (Me-PFOSA-AcOH), and perfluorodecanoic acid each had detectable serum concentrations, and all other measured PFCs were below the limit of detection. Median serum concentrations of PFOA and PFOS in residents from a community exposed to PFCs via contaminated drinking water were 27 ng/mL and 20 ng/mL, respectively (Steenland et al. 2009). Table 1 shows reported concentrations for PFOA and PFOS in humans and wildlife, while maximum serum concentrations for PFOA and PFOS in the general human population, occupationally exposed humans, and select wildlife and animal models are shown in Figures 1 and 2.
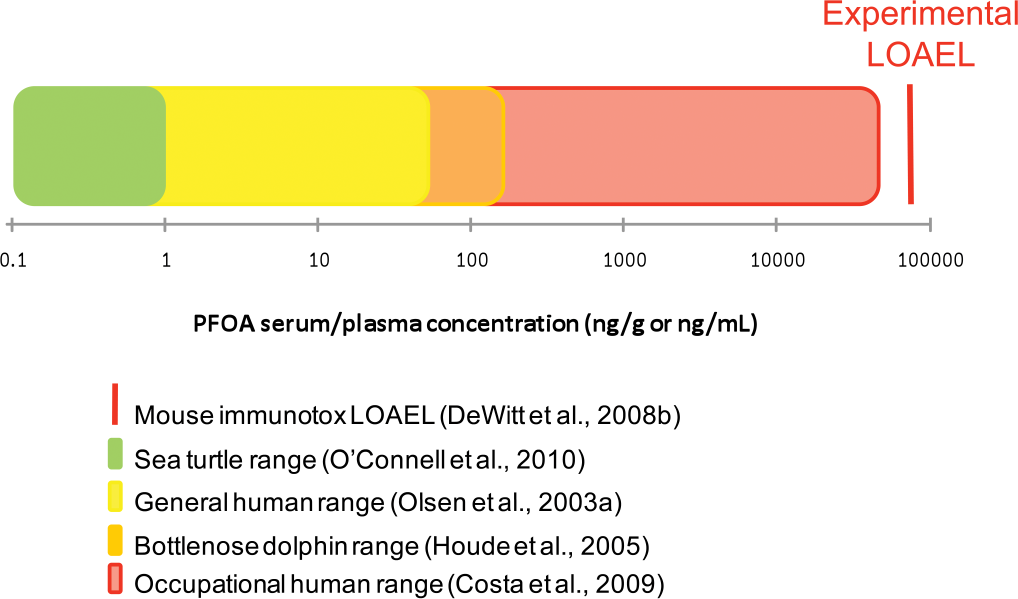
Comparison of human and wildlife serum/plasma concentrations (ng/g or ng/mL) of perfluorooctanoic acid (PFOA) to the resulting serum concentrations measured at the lowest “lowest observed adverse effect level” (LOAEL) for any immune parameter measured in laboratory mammalian models.
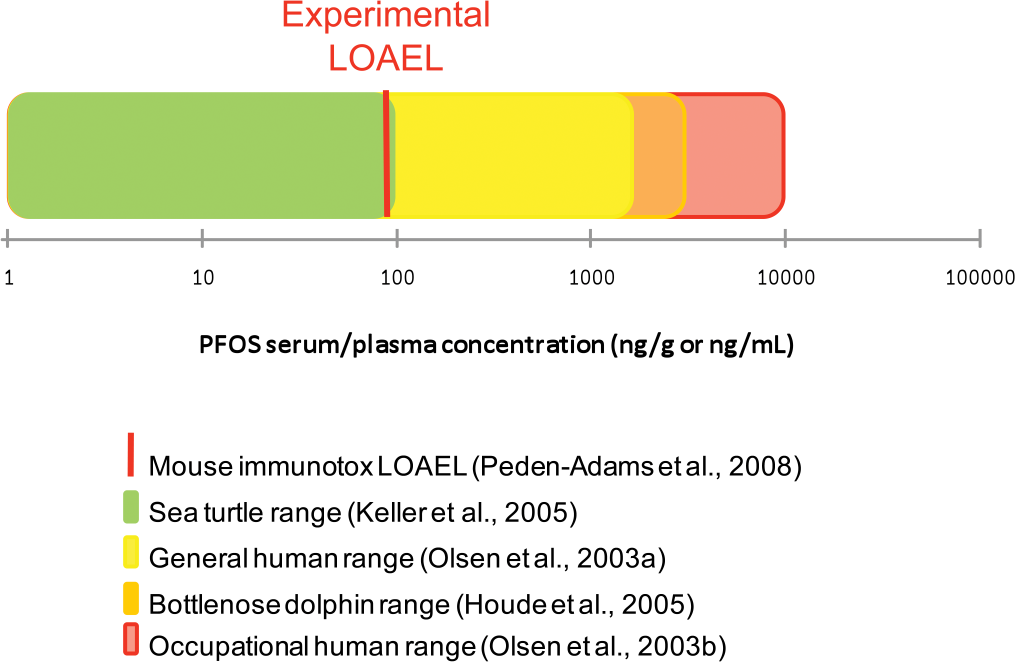
Comparison of human and wildlife serum/plasma concentrations (ng/g or ng/mL) of perfluorooctane sulfonate (PFOS) to the resulting serum concentrations measured at the lowest “lowest observed adverse effect level” (LOAEL) for any immune parameter measured in laboratory mammalian models.
Perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) concentrations (ng/g or ng/mL) in wildlife and human samples.
General Health Effects in Humans
In a comprehensive review, Steenland, Fletcher, and Savitz (2010) evaluated the epidemiologic literature on PFOA and its association with a dozen health outcomes, including immunomodulation. The authors concluded that the most consistent health findings associated with PFOA levels were modest increases in cholesterol and uric acid (Steenland, Fletcher, and Savitz 2010). Although certain chronic diseases with an inflammatory component, including diabetes, heart disease, and stroke, have been reported to be elevated in studies of exposed populations, the evidence was found to be too meager and inconsistent for solid conclusions (Steenland, Fletcher, and Savitz 2010). Similarly, these authors concluded that data on reproductive and developmental outcomes in humans are of limited relevance because the suggested decrements in birth weight associated with PFOA exposure were within the normal birth weight range and would have little clinical significance (Steenland, Fletcher, and Savitz 2010). In addition, slight changes reported in serum immunoglobulin levels (Costa, Sartori, and Consonni 2009) in a small study of male PFOA workers and increases in monocyte counts of residents who lived in a water district contaminated with PFOA (Emmett et al. 2006) are inconclusive. The epidemiological evidence for PFOS and other PFCs is similar to PFOA: some associations with health outcomes exist, such as increased cholesterol levels, but associations are small, and data sets are limited.
Immunomodulation
Antibody Responses
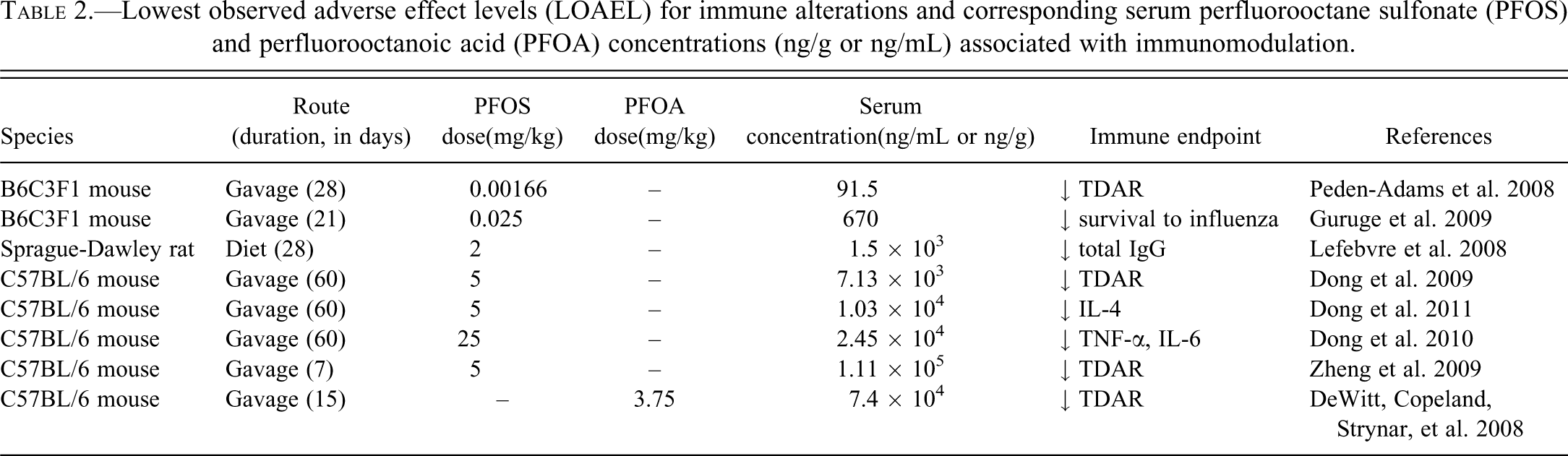
Both PFOA and PFOS suppress the T-cell-dependent IgM antibody responses (TDAR) in adult mice exposed via drinking water or oral gavage. Assessing IgM TDAR to an antigen is one of the most sensitive and predictive measures of immune function currently in use, as it requires T cells, B cells, and antigen-presenting cells to function properly to elicit an antibody response (Luster et al. 1992). The lowest observed adverse effect level (LOAEL) for suppression of IgM TDAR by PFOA was 3.75 mg/kg/d (corresponding to a serum concentration of 7.4 × 104 ng/mL) when given to female C57BL/6 mice by drinking water for 15 days (DeWitt et al. 2008) and 0.00166 mg/kg/d (corresponding to a serum concentration of 91.5 ng/mL) in male B6C3F1 mice given PFOS by oral gavage for 28 days (Peden-Adams et al. 2008). Zheng et al. (2009) also demonstrated PFOS-induced suppression of TDAR in C57BL/6 male mice with a LOAEL of 5 mg/kg via oral gavage for only 7 days (corresponding to a serum concentration of 1.11 × 105 ng/mL). Table 2 presents a list of LOAELs for immunomodulation and associated serum concentrations. At this time, the DeWitt, Copeland, Strynar, et al. (2008) reference is the only immune-related LOAEL reported for PFOA and the Peden-Adams et al. (2008) reference contains the lowest reported immune-related LOAEL for PFOS. The margin of exposure (MOE), which is the ratio of the NOAEL (no observed adverse effect level) to expected human exposure, calculated from these studies in relation to the general human population serum PFC concentrations is approximately 15,000 for PFOA and 0.5 to 10.8 (Fair et al. 2011) for PFOS. For occupational exposures, the calculated MOEs are 12 for highly exposed PFOA workers and approximately 0.005 for workers highly exposed to PFOS. In a separate study by Dong et al. (2009), the calculated MOE associated with suppression of TDAR in male C57BL/6 mice given PFOS via gavage for 60 days was approximately 50 in relation to highly exposed humans. As exposure increases, MOEs decrease and represent a smaller difference between animal data and anticipated human exposures (Aylward et al. 2008). Although MOEs are not values of risks themselves, when an MOE is less than 100, it is suggestive of potential health risks to exposed populations (Faustman and Omenn 2001). In contrast, Qazi et al. (2010) reported that 28 days of dietary exposure to PFOS did not affect IgM TDAR in B6C3F1 male mice at serum concentrations 8- to 85-fold greater than occupationally exposed humans. Similarly, Lefebvre et al. (2008) indicated that when Sprague Dawley rats were given PFOS in their diet for 28 days, IgG TDAR was not statistically suppressed, although they did not measure IgM TDAR. Qazi et al. (2010) proposed that the route of administration has a substantial impact on immunomodulation by PFOS and possibly other PFCs. Regardless of the presence of some conflicting data, the weight of evidence clearly supports the conclusion that IgM TDAR is an endpoint sensitive to modulation by PFCs in adult organisms.
Lowest observed adverse effect levels (LOAEL) for immune alterations and corresponding serum perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) concentrations (ng/g or ng/mL) associated with immunomodulation.
Recent studies have investigated the mode of action of PFOS-induced suppression of TDAR by examining the key events in cell signaling, cell activation, and cell populations that are important in mounting a successful antibody response. These studies indicate that key cell populations are not altered (Peden-Adams et al. 2010) and that modulation of some cell-signaling responses may be involved (Fair et al. 2011; Peden-Adams et al. 2010). B-cell control of interleukin (IL)-6 production through CD40 is critical to IgM secretion and is mediated, in part, by c-Jun and nuclear factor kappa B (NF-κβ) (Baccam et al. 2003; Bishop and Hostager 2003; Delerive et al. 1999). Also, c-Jun and NF-κβ each are negatively regulated by peroxisome proliferator–activated receptor (PPARα) agonists like PFOS (Andersen et al. 2008; Cunard et al. 2002; DeWitt et al. 2009). CD40 ligation on the surface of B cells with CD154 on helper T cells is critical for humoral responses to T-dependent antigens and results in B-cell proliferation, differentiation, and IL-6 stimulation of immunoglobulin production (Contin et al. 2003; Melgosa, Hollenbaugh, and Wilson 1999). Ex vivo production of IL-6, a proinflammatory cytokine necessary for antibody production, was increased in B cells but not T cells from mice not challenged with antigen (Fair et al. 2011; Peden-Adams et al. 2010). However, after 28 days of oral exposure to PFOS and an in vivo antigen challenge, IL-6 production in B cells stimulated with anti-CD40 or soluble CD154 in vitro was suppressed. IL-4, IL-5, and IL-6 production (critical to TDAR) by T cells was not affected in either challenged or unchallenged animals. In addition, nuclear translocation of NF-κβ (p65) and AP-1 (c-Jun) in splenic nuclear extracts was not altered, suggesting a possible role for altered CD40-CD154 signaling in the B cell following antigen challenge that is independent of NF-κβ and AP-1 (Peden-Adams et al. 2010). Although these studies begin to elucidate possible pathways altered in PFOS-induced TDAR suppression, additional studies are needed to clarify the mechanism associated with this effect.
While TDAR measures the role of both T and B cells in antibody production, T-cell-independent IgM antibody responses (TIAR) assess the role of the B cell in generating antibodies. Evaluating both TDAR and TIAR helps to determine whether a deficit exists in B cells, T cells, or macrophages, or in the process itself. TIAR also were suppressed after exposure to PFOA or PFOS. Suppression of TIAR was observed in C57BL/6 female mice given 1.88 mg/kg of PFOA in drinking water for 15 days (DeWitt, unpublished data), and Peden-Adams et al. (2008) reported that exposure to PFOS also suppressed TIAR in B6C3F1 mice. The suppression of both TDAR and TIAR suggest that B cells, macrophages, or both may be specific targets of PFCs.
Inflammation and Cytokine Signaling
It is generally accepted that agonists of PPARα are anti-inflammatory and that a major pathway of PFC toxicity is ligation of PPARα. However, relatively few studies have evaluated the ability of PFOA or other PFCs to modulate inflammatory responses, and the role of PPARα in PFC toxicity is still not definitive. Two studies by Taylor et al. (2002, 2005) established that PFOA suppressed inflammation induced by intraplantar injection of carrageenan as a model of cutaneous inflammation. In addition, Griesbacher et al. (2008) demonstrated that PFOA had no effect on inflammatory edema in a rat model of cerulean-induced pancreatitis, although it did suppress synthesis of the proinflammatory prostanoid PGE2 in the pancreas. Conversely, preliminary data in wild bottlenose dolphins have shown positive correlations between PFOS and PFOA plasma levels and clinical indices of inflammation and immunity (Peden-Adams, Keil, et al. 2009). Additionally, mice exposed to PFOA exhibit increased IgE levels that could trigger an increase in inflammation (Fairley et al. 2007). Finally, no alteration in inflammation was observed in the common carp liver following exposure to PFOS (Hoff et al. 2003). Other studies report similar pro- and anti-inflammatory results with other PPARα agonists (Clark 2002; Hill et al. 1999). Thus, there is not a general relationship between PFOA and PFOS and inflammation, and the species or disease model may influence the outcome and therefore contribute to the variability among studies.
TNFα and IL-6 in vivo/ex vivo
TNF-α and IL-6 are markers of inflammation that are part of a tightly controlled process in the body that, when modulated, can result in deleterious health effects. The balance between TNF-α and IL-6 is critical to the control of the inflammatory process during early stages of infection (Copeland et al. 2005; Yimin and Kohanawa 2006). LPS injection, by either the intravenous or intraperitoneal route, is a common model of inflammation (Fan et al. 2007; Fan et al. 2006; Hopkins 2007). Following intraperitoneal injection, LPS is absorbed rapidly and distributed throughout the body quickly (with approximately 10% of the LPS administered appearing in circulation within 15 minutes; Hopkins 2007). When triggered by infection (as mimicked by LPS injection) monocytes and macrophages are the first cell types to respond and TNF-α is the first cytokine released, followed shortly by IL-6 and IL-1 (Hopkins 2007). Increases in basal serum levels of IL-6 and in serum levels of IL-6 and TNFα following LPS injection have been demonstrated following exposure to either PFOA or PFOS at levels ranging from 50 mg/kg to 400 mg/kg total dose (Dong et al. 2010; Qazi et al. 2009). However, one study reported an increase in serum IL-6 and decrease in serum TNFα at 1 mg/kg total dose (Mollenhauer et al. 2011). Differences in results among these studies may be related to gender and or mouse strain used, as Qazi, Bogdanska, et al. (2009) and Dong et al. (2010) used male C57Bl/6 mice, whereas Mollenhauer et al. (2011) used female B6C3F1 mice. Finally, in a study by Son et al. (2009), in male ICR mice given 0.5 mg/kg to 47 mg/kg of PFOA in drinking water for 21 days, levels of IL-6, TNFα, and IL-1β were all increased by the higher doses of PFOA.
Changes in the ex vivo production of TNFα and IL-6 by peritoneal macrophages following in vitro LPS stimulation or in vivo LPS injection have been reported at PFOS concentrations ranging from 25 mg/kg to 400 mg/kg total dose (Dong et al. 2010; Mollenhauer et al. 2011; Qazi, Bogdanska, et al. 2009). As discussed above, IL-6 production was increased in B cells from naive mice, whereas after an in vivo challenge IL-6 production by B cells was decreased (Fair et al. 2011; Peden-Adams et al. 2010). Production of IL-6 by T cells has been reported to remain unchanged with or without an in vivo antigen challenge (Fair et al. 2011; Peden-Adams et al. 2010). Exposure to a PFOA mass fraction of 400 mg/kg (total dose) increased ex vivo TNFα production after both in vitro and in vivo LPS exposure (Qazi, Bogdanska, et al. 2009). Additionally, ex vivo production of TNFα and IL-6 from mixed spleen cell cultures (unstimulated or simulated in vitro with LPS) was increased after exposure to 50 mg/kg and 125 mg/kg PFOS (Dong et al. 2010). These results suggest that TNF-α and IL-6 are modulated by PFOS and PFOA exposure both in vivo and ex vivo; however, the specific effect varies by dose, strain, and exposure route.
TNF-α and IL-6 in vitro
Brieger et al. (2011) and Corsini et al. (2011) evaluated the effects of PFOA and PFOS on cytokine secretion in vitro and reported that both compounds can alter both pro- and anti-inflammatory pathways to produce a net anti-inflammatory effect. Brieger et al. (2011) collected whole blood from human volunteers and measured TNF-α and IL-6 after incubating blood with LPS and 0.1 µg/mL to 100 µg/mL of PFOA or PFOS. Levels of TNF-α were decreased by PFOS exposure, and IL-6 concentrations were increased by both PFOA and PFOS exposure. Corsini et al. (2011) evaluated the in vitro response of human leukocytes and THP-1 cells (a human promyelocytic cell line) exposed to 0.1 µg/mL to 100 µg/mL of PFOA or PFOS and stimulated with LPS. Both PFOA and PFOS were able to alter pathways involving I-κB, NF-κB, and TNF-α. However, the pattern of cytokine alteration by each compound differed by dose and by the involvement of PPARα. PFOS actions were
TH1/TH2 cytokine profiles
T-helper (TH)-1 and TH2 cells are subsets of T cells that each play specific roles in immunity. TH1 cells promote cell-mediated immunity against intracellular pathogens, and the prototypical cytokines secreted are IFNγ and IL-2. TH2 cells are involved in humoral immunity and secrete IL-4 to promote production of immunoglobulins. If cytokine profiles are shifted toward one subset over another, the risk of immune dysfunction increases. In separate studies, Dong et al. (2011) and Zheng et al. (2011) evaluated the effect of short-term or subchronic PFOS exposure on the balance between TH1 and TH2 cytokines in C57BL/6 male mice. In the short-term model (7 days of exposure to 5 or 20 mg/kg of PFOS), the cytokine balance favored TH2 responses (Zheng et al. 2011). Numbers of T cells producing IL-2 and levels of ex vivo IFNγ production from mixed spleen cell cultures were reduced by 20 mg/kg. IL-4, a TH2 cytokine, was increased by 5 mg/kg and 20 mg/kg; however, at the 20 mg/kg dose, serum corticosterone was increased and body mass was reduced by nearly 10%, suggesting systemic toxicity at this dose. In the subchronic model (60 days of exposure to 0.5-50 mg/kg of PFOS), results were nearly identical to the short-term model, with similarly reported changes in IL-2, IFNγ, and IL-4, and in numbers of cells producing IL-2 (Dong et al. 2011). In addition, numbers of cells producing IL-10, a TH2 cytokine, also were reduced. Serum corticosterone levels did not differ among treatment groups, and body weight reductions were observed only in the 50 mg/kg dose group. The serum concentrations for the subchronic study, where stimulation of IL-4 production occurred, were in the upper range of concentrations reported for occupationally exposed humans.
PPARα
The differential effect of PPARα with regard to the ability of PFOA and PFOS to alter cytokine release is consistent with data that indicate that PFOA’s developmental toxicity is dependent on PPARα (Abbott et al. 2007) whereas PFOS’s is not (Abbott et al. 2009). Further, data by Peden-Adams et al. (2010) suggest that suppression of the IgM TDAR by PFOS also is not dependent on PPARα, based on similar suppression of the plaque-forming cell response in wild-type and PPARα-constitutive knockout mice (PPARα CKO) treated for 28 days via oral gavage of 0 to 5 mg/kg PFOS total dose. The gene encoding the PPARα receptor in the PPARα CKO mice contains a mutation that prevents ligand binding to the receptor (Lee et al. 1995). Additionally, PPARα nuclear translocation was not different from control responses in adult female B6C3F1, C57Bl/6 (WT), or PPARα CKO mice (Peden-Adams et al. 2010). DeWitt, Copeland, and Luebke (2008) demonstrated that suppression of IgM TDAR by PFOA also is not dependent on PPARα activation, as IgM suppression was approximately equivalent in wild-type and PPARα CKO mice. These studies suggest that TDAR suppression by both PFOA and PFOS is not dependent on PPARα activation. Qazi, Xia, et al. (2009) reported that PFOS-induced modulation of lymphocyte numbers was partially dependent on PPARα, as reductions in lymphocytes in the thymus were partially attenuated and reductions in splenic lymphocytes were eliminated following dietary exposure in PPARα CKO mice (0.01-0.02% in feed for 10 days; approximately 400 mg/kg PFOS total administered dose for high-dose group). However, as suppression of IgM TDAR appears to be related to effects on B cells and/or macrophages, PPARα-dependent reductions of thymic lymphocytes numbers are likely not related to changes in TDAR. Recent evidence by Nakamura et al. (2011) indicates that, at least for liver hepatomegaly and induction of PPARα target genes, the mouse PPARα is sensitive to relatively low doses (0.3 mg/kg) of PFOA. It is likely that in addition to strain and concentration differences that may impact receptor occupancy, the toxicity and immunomodulation induced by exposure to PFCs may occur via multiple pathways, and the relative contributions of these pathways and the role of PPARα likely differ among PFCs.
Developmental Immunotoxicity
To date, only two studies have addressed the developmental immunotoxicity of PFCs. Keil et al. (2008) reported that when given from gestational days 0 through 17, PFOS induced suppression of IgM TDAR in male but not female offspring. In a similar study, Hu, Strynar, and DeWitt (2010) demonstrated that when given PFOA from gestational days 6 through 17, PFOA did not induce suppression of IgM TDAR in female offspring; male offspring were not evaluated. Although this difference may be sex related, it also is likely associated with dose sensitivity of mouse strains to the respective PFCs and the type of PFC administered. The PFOS LOAEL for IgM TDAR reported by the Keil et al. (2008) was 5 mg/kg, which was also the highest dose administered. In the Hu et al. (2010) study of PFOA, 5 mg/kg was fetotoxic, and therefore the highest dose for assessing developmental immunotoxicity was 1 mg/kg. In both studies, 1 mg/kg of PFOA (associated with serum concentrations of 183 ng/mL) or PFOS (no serum concentrations reported) was not sufficient to suppress IgM TDAR.
Effect of Diet
Y. Wang et al. (2011) conducted a study based on the hypothesis that a high-fat diet could attenuate the immunotoxic effects of PFOS, as it is known to alter lipid homeostasis. BALB/c mice were gavaged with 5 mg/kg or 20 mg/kg of PFOS for 14 days and fed regular or high-fat diets. Lymphoid organ atrophy and structural changes in lymphoid tissues, including increased apoptosis, were increased in the 20 mg/kg group given the high-fat diet relative to control groups. The data indicate that a high-fat diet was insufficient to ameliorate the effects of PFOS on certain immune parameters, assuming that the effects of PFOS and lipid metabolism are interrelated. The authors suggested that the effects of PFOS on immune organs were indirect, resulting after altered lipid metabolism; however, the relationship between altered lipid metabolism and immunomodulation by PFOS is not clear.
Host Resistance
The observed shift in the TH1/TH2 balance noted by Dong et al. (2011) and Zheng et al. (2011) suggests that PFOS may reduce cell-mediated immunity and therefore the ability to fight intracellular pathogens and cancerous cells. However, no alteration in delayed-type hypersensitivity, a measure of proinflammatory cell-mediated immunity critical to resistance to intracellular pathogens, was observed by Lefebvre et al. (2008), DeWitt, Copeland et al. (2008), or Dong et al. (2011). In the one study to date that has assessed the effects of a PFC in a host-resistance model, Guruge et al. (2009) observed that emaciation and mortality associated with influenza A infection was increased in B6C3F1 female mice exposed to 0.005 mg/kg or 0.025 mg/kg of PFOS for 21 days. Statistically significant increases in susceptibility to influenza A–induced mortality were noted at 0.525 mg/kg total dose. Decreases in TDAR may be indicative of susceptibility to influenza virus (Burleson and Burleson 2008), and interestingly, in the female B6C3F1 model, Peden-Adams et al. (2008) reported a corresponding effect level of 0.5 mg/kg total dose for reduction in TDAR. Serum PFOS concentrations for both studies were within the range of serum concentrations for occupationally exposed humans, the higher range of background exposures for humans, and for certain types of wildlife. More studies are warranted to determine if exposure to PFOS increases host susceptibility to other types of pathogens.
Immune Effects in Nontraditional Models and Wildlife
In wild-sampled avian species, PFOS occurs at higher concentrations than other PFCs routinely measured (Table 1). To determine if PFOS affected immune parameters in a standard model of avian developmental toxicity, Peden-Adams et al. (2009) injected fertile chicken eggs prior to incubation with PFOS at 1, 2.5, or 5 mg/kg of egg mass. When immune endpoints were evaluated in 14-day-old chickens, the TDAR was decreased and serum lysozyme activity was increased at all doses examined. PFOS serum concentrations associated with these immunological changes (154-1607 ng/mL) were within the range of concentrations reported for several species of wild birds. To our knowledge, no other studies of immune alterations in avian species have been reported in the literature. Considering that PFCs have been detected in a variety of avian species and that the immune effects reported by Peden-Adams, Stuckey, et al. (2009) occurred at environmentally relevant doses, it is possible that changes in immune function and disease resistance may occur as a consequence of PFC exposure in wild birds; however, additional studies, including field sampling, are needed to further investigate the potential health effects in avian species.
PFOS is also the PFC with the highest concentration in many aquatic species, including sea turtles and dolphins (Table 1). Loggerhead sea turtles along the southeast U.S. Atlantic coast had detectable levels of serum PFCs, and these concentrations positively correlated with an indicator of liver damage (plasma aspartate aminotransferase) and an indicator of adaptive immunity (T-cell proliferation) and negatively correlated with an indicator of innate immune function (plasma lysozyme activity; Keller et al. 2008). The maximum PFOS concentration measured in the loggerhead sea turtles (96.8 ng/mL; Keller et al. 2005) was above the serum concentration (91.5 ng/g) measured in mice at the LOAEL for reduced IgM TDAR (Peden-Adams et al. 2008) (see Figure 2). Thus, the concentrations known to cause immunosuppression in rodent models and the exposure concentrations in individual sea turtles are similar. Data collected from wild Atlantic bottlenose dolphins reveal that their blood levels are even greater (mean PFOS = 1315 ng/mL; Houde et al. 2005) (Figure 2). Evidence of altered immune responses were seen in dolphins from South Carolina and Florida sampled in 2003; increased C-reactive protein, increased LPS-induced lymphocyte proliferation, and decreased plasma lysozyme activity were correlated to increasing PFOS and PFOA serum concentrations (Peden-Adams, Keil, et al. 2009). Moreover, a field study found significant associations between infectious diseases and elevated exposures to PFOS in sea otters from coastal California, suggesting possible immune effects of PFOS and related PFCs in this species (Kannan, Perrotta, and Thomas 2006). Data from wild sea turtles, dolphins, and sea otters suggest that coastal marine organisms are exposed to environmental levels of PFCs that may compromise immune function. This is important because altered immune functions may lead to decreased resistance to infectious diseases, with population-level impacts for these species that are already protected because of conservation concerns. Additionally, they serve as sentinels or bioindicators of exposure to environmental contaminants and, more importantly, the adverse health effects from that exposure (Bossart 2006; Keller, Peden-Adams, and Aguirre 2006).
Evaluating adaptive immune function in wild species can be particularly difficult, as many traditional tests to evaluate humoral immunity require challenging the organism to elicit an immune response, and then measuring the strength of the response at a later time. Ex vivo assays, such as lymphocyte proliferation, have been used as surrogate measures (Keller et al. 2006; Keller, Peden-Adams, and Aguirre 2006). However, the myriad substances these animals are exposed to may confound any assessment of toxicity in wild species, and the results obtained from these studies are correlative at best. Similarly, assays for innate immune function, such as serum lysozyme activity, which measures antibacterial responses, also reflect whole-body and whole-life exposures. It is therefore critical that observations in wild species be confirmed by surrogate models under controlled laboratory conditions to decrease uncertainties (Peden-Adams et al. 2009). As a laboratory model for evaluating health effects in reptilian species, Western fence lizards were exposed (28 days via gavage) to 0.01, 0.1, or 1.0 mg/kg as a total administered dose of PFOS, to approximate the levels of PFOS reported in sea turtles (Keller et al. 2005). Plasma lysozyme mean activities appeared to be reduced by PFOS, and T-cell proliferation was slightly increased (Keller et al. 2008; Peden-Adams et al. 2009), adding to the weight of evidence suggesting that the immune systems of nonmammalian species can be affected at current environmental levels of PFCs.
Immune Effects in Humans
Only limited data are available on the effects of PFCs on the immune system in occupationally or environmentally exposed human populations. A detailed assessment of immune measures in the population living within close proximity to a PFOA production facility in West Virginia has been conducted by the C8 Science Panel (www.c8sciencepanel.org), but the data from this particular study have not yet been published. In a brief status report by the C8 Science Panel (Fletcher, Steenland, and Savitz 2009), levels of IgA, IgE (females only), and C-reactive protein decreased with increasing PFOA serum concentration, and antinuclear antibodies, a marker of autoimmunity, increased with increasing PFOA serum concentration. In a publication resulting from the Taiwan Birth Panel cohort study, prenatal exposure to PFOA and PFOS was positively correlated with cord blood IgE levels, particularly in male children (I. J. Wang et al. 2011). Although there was no association with atopic dermatitis in this study, these data suggest the potential for elevated risk of allergic disease.
Other PFC Compounds and Immunotoxicity
Fang et al. (2008) exposed male BALB/c mice to 1, 3, or 5 mg/kg of PFNA for 14 days and measured a variety of immune endpoints, including lymphoid organ mass, numbers of lymphocytes in the spleen and thymus, PPARα expression, and various immune signaling pathways. Similar to findings associated with PFOA and PFOS, PFNA reduced body and lymphoid organ weights and altered numbers of splenic and thymic lymphocytes. All doses of PFNA reduced production of IL-4 by splenic lymphocytes, and the two highest doses were associated with increases in serum corticosterone and messenger RNA (mRNA) of PPARα and PPARγ. Fang et al. (2008) hypothesized that activation of PPARs and the hypothalamic-pituitary-adrenal axis produced toxic effects on immune homeostasis. In a related study using male Sprague-Dawley rats given the same PFNA doses for the same duration, similar effects were observed on body and thymus weights and mRNA of PPARα; however, serum corticosterone was elevated only in the highest-dose group, as was IL-4 (Fang et al. 2009). In addition, IL-1 was elevated and IL-2 was reduced in the two highest dose groups, phosphorylated JNK (p-JNK) was elevated in all dose groups, and p38 was elevated in the two lowest dose groups. Fang et al. (2009) concluded that activation of p-JNK and p38 kinases by cytokine release and increases in IL-1 contributed to the observed reduction in thymus weight via apoptosis of thymic lymphocytes.
Conclusions
Accumulated evidence indicates that exposure to PFCs suppresses antigen-specific antibody responses in a number of mouse strains and that this suppression occurs at serum concentrations below, within the reported range, or just above those reported for highly exposed humans and wildlife. As demonstrated in Figure 1, the serum levels associated with PFOA-induced immune effects is higher than the concentrations reported for humans and wildlife. For PFOS (Figure 2), some human and wildlife species have serum concentrations that exceed the threshold for PFOS-induced immune effects, suggesting a potential risk for altered disease resistance. Additional studies have demonstrated that other immune measures, including cytokine expression and signaling related to inflammation and TH cell responses, are altered by exposure to PFCs. Immune effects reported in laboratory animals appear to be somewhat independent from PPARα activation. However, this varies with the type of PFC and endpoint being evaluated and may change under situations where exposure includes multiple PFCs that may alter receptor occupancy and therefore activation. In addition, changes in immune parameters that occur at doses where systemic toxicity-associated body mass changes do not occur do not appear to be stress dependent. DeWitt, Copeland, and Luebke (2009) demonstrated that IgM TDAR was suppressed in adrenalectomized mice given 15 mg/kg of PFOA for 10 days at levels equivalent to intact mice. In exposed humans, elevated levels of PFOA and PFOS have been associated with significant changes in clinical markers of immune and inflammatory responses. The strong weight of evidence for health effects in loggerhead sea turtles and dolphins, the correlation in responses between in vitro and in vivo data, and the general concordance of findings reported in laboratory animal studies indicate that exposure to PFCs can adversely affect immune function.
Footnotes
Acknowledgments
We would like to thank Drs. Robert Luebke, Kristina Thayer, and Katherine Sharpless for their review and helpful suggestions. We thank Dr. Meagan Mollenhauer and Mr. Kenny Lee for assistance with the tables.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article. This research was supported in part by the Intramural Research Program of the National Institute of Environmental Health Sciences, National Institutes of Health. This article may be the work product of an employee or group of employees of the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH); however, the statements, opinions or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NIEHS, NIH, or the U.S. government.
