Abstract
Here, we report the effects of estrogen and the selective estrogen receptor modulator (SERM) levormeloxifene on adrenocortical measures in ovariectomized female cynomolgus monkeys (Macaca fascicularis). Animals were randomized into one of five treatment groups, each containing 23 to 26 animals: (1) placebo, (2) 0.016 mg/kg 17β-estradiol (E2), (3) 0.5 mg/kg levormeloxifene (L1), (4) 1.0 mg/kg levormeloxifene (L2), and (5) 5.0 mg/kg levormeloxifene (L3). Treatments were administered orally each day for 18 mo. All doses of levormeloxifene resulted in adrenal weights at least 50% greater than placebo (p < .0001 for all). The target dose of levormeloxifene (L2) resulted in higher serum concentrations of cortisol (+63%), dehydroepiandrosterone-sulfate (+73%), and androstenedione (+37%) compared with the placebo group (p < .05 for all). In contrast, E2 resulted in no significant differences in adrenal weight or adrenocortical steroids. Oral E2 and all SERM doses resulted in similar reductions in serum gonadotropins and at least threefold greater uterine weight versus placebo (p < .0001 for all). Results indicate that the SERM levormeloxifene, in contrast to E2, may have robust stimulatory effects on adrenocortical hormones in a postmenopausal model. These findings warrant further investigation into long-term SERM effects on adrenocortical function.
Adrenocortical hormones influence a variety of chronic diseases in postmenopausal women (Lobo 2001; Shifren 2002; Labrie et al. 2003). For example, age-associated declines in adrenal-derived androgens have been associated with increased risk of osteoporosis, loss of muscle mass and libido, depression, and cognitive decline (Slemenda et al. 1996; Greendale, Edelstein, and Barrett-Connor 1997; Gower and Nyman 2000; Barrett-Connor and Goodman-Gruen 1999), while elevated concentrations of circulating cortisol (Cort) have been implicated in decreased cognitive function and metabolic dysfunction, including insulin resistance and type 2 diabetes (Lupien et al. 1998; Bjorntorp 1997). Despite these observations, determinants of adrenocortical hormone production in postmenopausal women are poorly understood.
Selective estrogen receptor modulators (SERMs) are nonsteroidal agents that bind estrogen receptors (ERs) and elicit tissue-specific estrogen agonist and antagonist effects (Shiau et al. 1998). This versatile class of compounds includes tamoxifen and raloxifene, which are important endocrine therapies and preventive agents for ER-positive breast cancers (Martino et al. 2004; Fisher et al. 2005). Raloxifene and newer generation SERMs also have ER agonist activity in bone and are widely used clinically for prevention of osteoporosis in postmenopausal women (Siris et al. 2005; Silverman et al. 2008; Cummings et al. 2010). Apart from these beneficial applications, SERMs elicit a range of secondary, nontarget effects, including modulation of endocrine hormones (e.g., Rossi et al. 2009). Long-term effects of SERMs on adrenocortical activity are unclear, however, particularly in postmenopausal women.
Levormeloxifene is a benzopyran SERM previously under development for the prevention and treatment of postmenopausal osteoporosis (Alexandersen et al. 2001) but discontinued because of the high incidence of adverse events in the reproductive tract (Goldstein and Nanavati 2002; Warming et al. 2003; Ravn, Nielsen, and Christiansen 2006). Endocrine-related effects of levormeloxifene are poorly documented. The primary goal of this study was to evaluate the adrenocortical effects of this SERM in comparison to oral micronized estradiol (E2) in a postmenopausal primate model.
Materials and Methods
Study Design and Treatments
This study was initiated in 1996 as part of the late-stage preclinical evaluation of levormeloxifene (prior to discontinuation). The experiment followed a 5-way parallel-arm design in which 121 adult ovariectomized female cynomolgus monkeys (Macaca fascicularis) with a mean estimated age of 8 to 12 years were randomized into one of the following five treatments with 23 to 26 monkeys per group: (1) placebo (C), (2) micronized 17β-estradiol (E2) at a dose of 0.016 mg/kg, and the SERM levormeloxifene (NNC 46-0020; Novo Nordisk, Målöv, Denmark) at (3) 0.5 mg/kg (L1), (4) 1.0 mg/kg (L2), and (5) 5.0 mg/kg (L3). Dose equivalents approximated a standard estrogen therapy dose of E2 for postmenopausal women and low (L1), target (L2), and high (L3) human-equivalent doses of levormeloxifene. Estradiol treatment resulted in trough serum concentrations of ˜50 to 200 pg/mL (Hotchkiss et al. 2001), comparable to those seen in the midfollicular phase of intact animals (Weinbauer et al. 2008) and premenopausal women (e.g., Smith et al. 2011; Nagata et al. 2011). Serum levormeloxifene concentrations measured by high-performance liquid chromatography (Kiehr and Christensen 1999) averaged 10.9, 18.5, and 51.2 ng/mL for L1, L2, and L3 doses, respectively (Hotchkiss et al. 2001), comparable to those reported in women receiving similar doses (Skrumsager et al. 2002). Ovariectomies were performed by an experienced veterinary surgeon at Wake Forest School of Medicine according to standard procedures. Treatments were administered once daily 7 d/wk between 9 a.m. and 11 a.m. in a Crystal Light vehicle by voluntary oral dosing from a syringe for 18 mo (Hotchkiss et al. 2001). Because the study was also designed to determine treatment effects on atherosclerosis, all animals were fed a diet designed to mimic the amount of fat, cholesterol, and other nutrients consumed in many industrialized nations. Diets contained 10% casein, 10% lactalbumin, 21% dextrin, 20% sucrose, 7% alphacel, 20% butter, 3% soybean oil, 0.13% cholesterol, 2.5% Teklad vitamin mix, and 5% #2 Ausman-Hayes mineral mix. The diet was custom formulated at Wake Forest School of Medicine to minimize soy phytoestrogens, which are present in standard monkey chow. Additional details on the diet are provided elsewhere (Hotchkiss et al. 2001).
Female cynomolgus macaques have a similar reproductive pathophysiology and endocrine profile to women and are commonly used in studies of hormonal agents, including estrogens and SERMs (e.g., Weinbauer et al. 2008; Cline et al. 1998). Unlike rodents, human and nonhuman primates produce large amounts of adrenal dehydroepiandrosterone (DHEA) and DHEA-sulfate (DHEA-S) that serve as androgen precursors and decline with age (Labrie et al. 1997, 2003). In addition, the ovariectomized monkey model isolates the adrenal gland as the sole source of DHEA-S and downstream androgens (as well as Cort), allowing for more direct evaluation of adrenocortical effects.
All animals for this study were originally imported from Bogor, Indonesia, and housed in stable social groups of three to five animals each. All procedures involving these animals were conducted in compliance with state and federal laws, standards of the U.S. Department of Health and Human Services, and guidelines established by the Wake Forest University Animal Care and Use Committee. The facilities and laboratory animal program of Wake Forest University are fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care.
At the end of the treatment period, animals were sedated with ketamine and euthanized using sodium pentobarbital (100 mg/kg, intravenous), as recommended by the Panel on Euthanasia of the American Veterinary Medical Association. Euthanasia was performed for data collection related to cardiovascular endpoints. Adrenal glands, pituitary glands, and uteri were collected and weighed using standard procedures. These tissues were retained by the study sponsor following collection, precluding histologic evaluation.
Serum Hormone Concentrations
Blood was collected by femoral venipuncture under ketamine sedation (30 mg/monkey intramuscularly) between 8:00 a.m. and 10:00 a.m. following an overnight fast and ˜ 22 h after drug administration on the previous day. Serum concentrations of follicle-stimulating hormone (FSH), luteinizing hormone (LH), Cort, DHEA-S, androstenedione (A4), and total testosterone (T) were measured. Cort, DHEA-S, A4, and T were determined by radioimmunoassay (RIA) with commercially available kits (Coat-A-Count, Diagnostic Products Corporation, Los Angeles, CA, for DHEA-S, A4, and T; Diagnostic Systems Laboratories, Webster, TX, for Cort). LH and FSH were measured via bioassay and RIA, respectively, as described previously (Wilson 1998). Hormone assays were modified as needed for use in monkeys, using charcoal-stripped monkey serum as a diluent for low standards. Intraassay coefficients of variation were less than 15% for all assays. All hormone assays were performed at the Yerkes National Primate Research Center Biomarkers Core Laboratory.
Statistical Analyses
Data were analyzed using the SAS statistical package (version 9.1, SAS Institute, Cary, NC). All data were evaluated for normality and homogeneity of variances among groups. Group comparisons were evaluated using a nonparametric Kruskal-Wallis analysis of variance test followed by two-sided Wilcoxon rank sum pairwise analysis and reported as mean ± standard error. All pairwise p values were adjusted for the number of pairwise tests (seven total for each treatment group versus control and each SERM group versus E2) using a Bonferroni correction. A two-tailed significance level of .05 was chosen for all comparisons.
Results
Treatment with E2 and levormeloxifene did not significantly affect total body weight at any time point but resulted in at least threefold greater mean uterine weight at 18 mo compared with placebo (p < .001 for all; Table 1 ). In contrast, left, right, and total adrenal gland weights were significantly greater (>50%) in each levormeloxifene group (p < .001 for all) but not the E2 group compared with control (Table 1). Effects on adrenal weight (adjusted for body weight) were comparable across all SERM doses (+7%, +69%, +56%, and +64% for E2, L1, L2, and L3 groups, respectively) with no evident dose-dependent response.
Treatment effects on body, uterine, and adrenal weights.
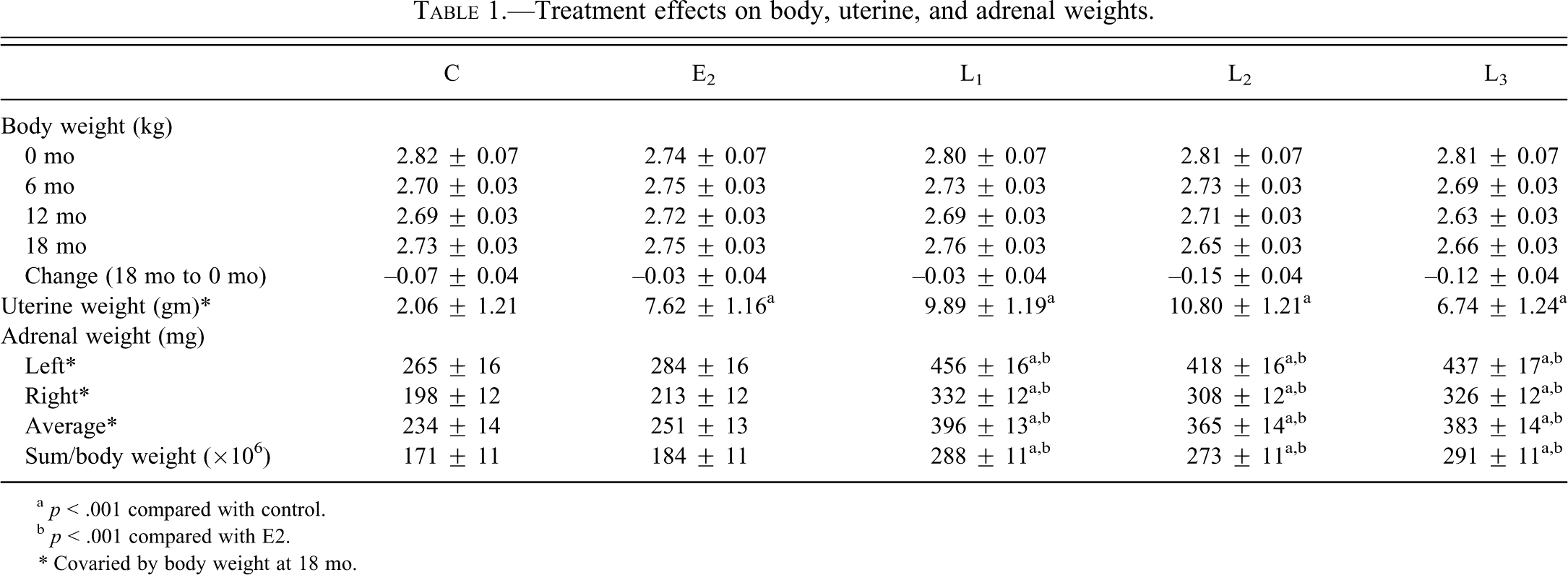
a p < .001 compared with control.
b p < .001 compared with E2.
* Covaried by body weight at 18 mo.
Consistent with adrenal weight effects, serum Cort values were significantly higher for all SERM doses at 3 mo, 6 mo, 9 mo, 12 mo, and 15 mo (p < .01 for all) and for the target L2 dose at 18 mo (p < .001) compared with both control and E2 groups (Table 2 ). No significant group differences were seen at baseline time points (–6 mo and –3 mo). Cort effects averaged across all time points (3 mo to 18 mo) were comparable for all levormeloxifene doses (+9%, +68%, +73%, and +69% for E2, L1, L2, and L3 groups, respectively).
Treatment effects on serum cortisol.
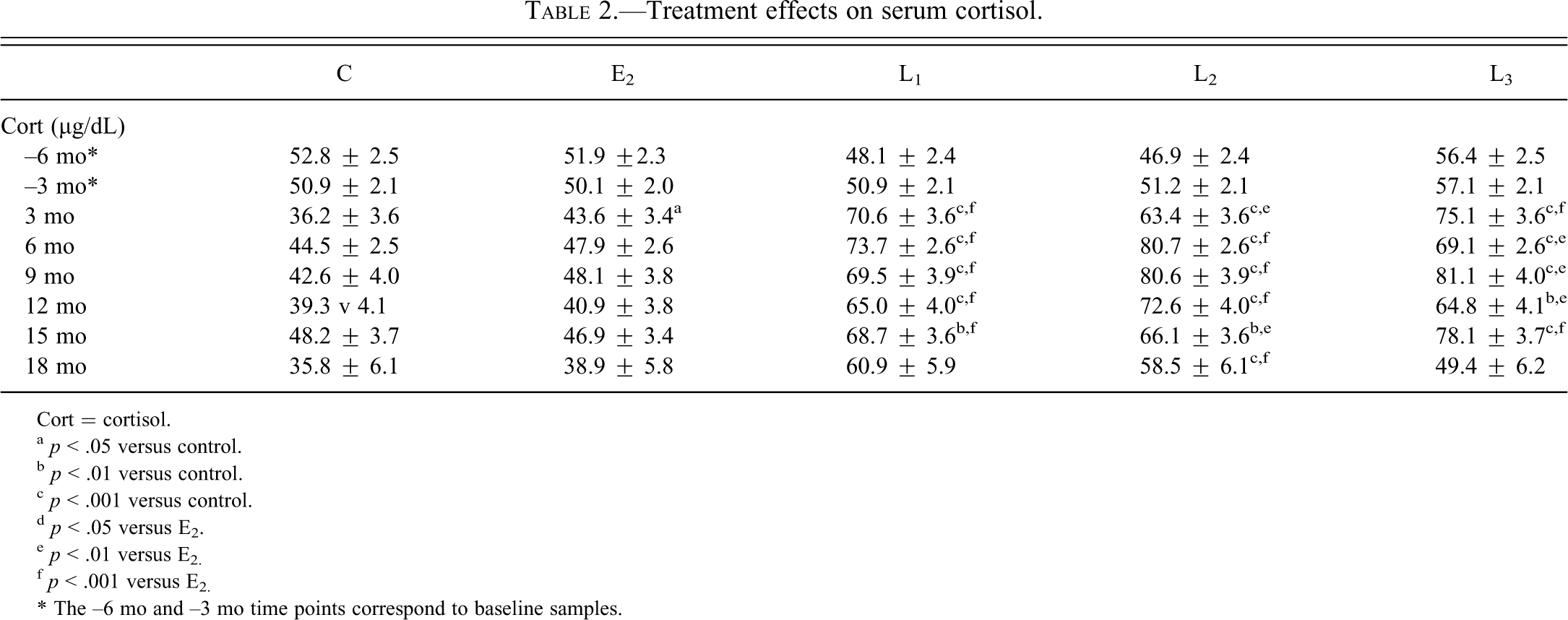
Cort = cortisol.
a p < .05 versus control.
b p < .01 versus control.
c p < .001 versus control.
d p < .05 versus E2.
e p < .01 versus E2.
f p < .001 versus E2.
* The –6 mo and –3 mo time points correspond to baseline samples.
Similar effects were seen for DHEA-S, the primary sex steroid produced by the adrenal cortex. Serum DHEA-S concentrations were higher for L3 at 6 mo and for L1, L2, and L3 at 18 mo compared with control and E2 groups (p < .05 for all; Table 3 ). Treatment effects on A4, the major downstream metabolite of DHEA-S, were also higher for L2 at 6 mo and for L1, L2, and L3 at 18 mo compared with control and E2 groups (p < .05 for all; Table 3), while significant effects on T were observed only for the L2 group at 6 mo and L1 and L2 groups at 18 mo compared with E2 (p < .05 for all; Table 3).
Treatment effects on adrenal-derived androgens.
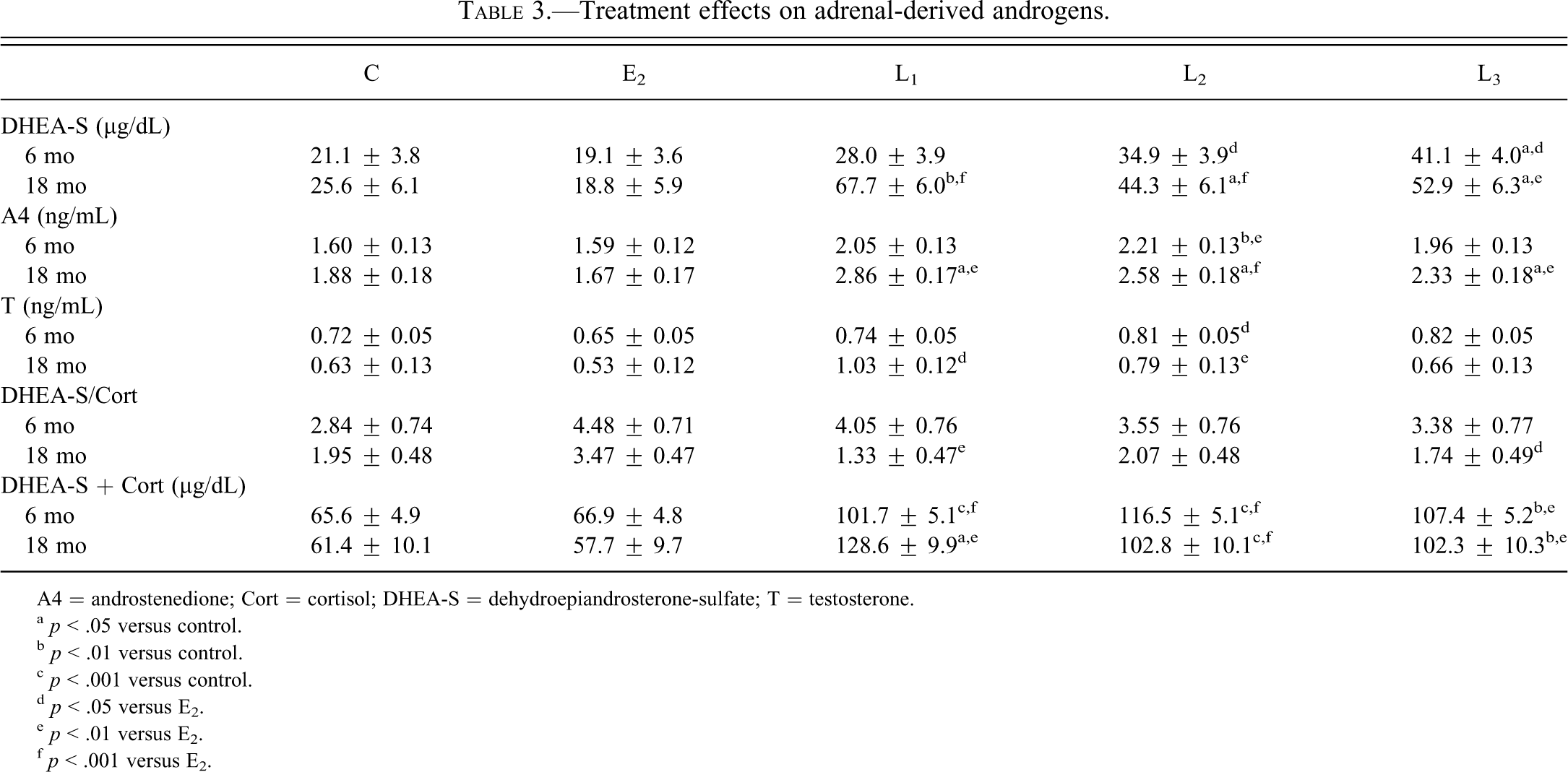
A4 = androstenedione; Cort = cortisol; DHEA-S = dehydroepiandrosterone-sulfate; T = testosterone.
a p < .05 versus control.
b p < .01 versus control.
c p < .001 versus control.
d p < .05 versus E2.
e p < .01 versus E2.
f p < .001 versus E2.
Effects on DHEA-S and Cort were relatively balanced, as indicated by the lack of significant treatment effects on the Cort/DHEA-S ratio compared with placebo (Table 3). In contrast, the sum of Cort and DHEA-S was higher for all SERM doses at 6 mo and 18 mo compared with control and E2 groups (p < .05 for all; Table 3). Overall effects across the 6-mo and 18-mo time points were –2%, +83%, +73%, and +65% for the E2, L1, L2, and L3 groups, respectively. Total adrenal weight correlated strongly with the sum of Cort and DHEA-S at both 6 mo (r = 0.65, p < 10–15) and 18 mo (r = 0.67, p < 10–16; Figs. 1A, B). Among individual hormones, adrenal weight correlated most strongly with DHEA-S (r = 0.63, p < 10–14), followed by A4 (r = 0.53, p < 10–9), T (r = 0.45, p < 10–6), and Cort (r = 0.39, p < 10–5) at 18 mo.
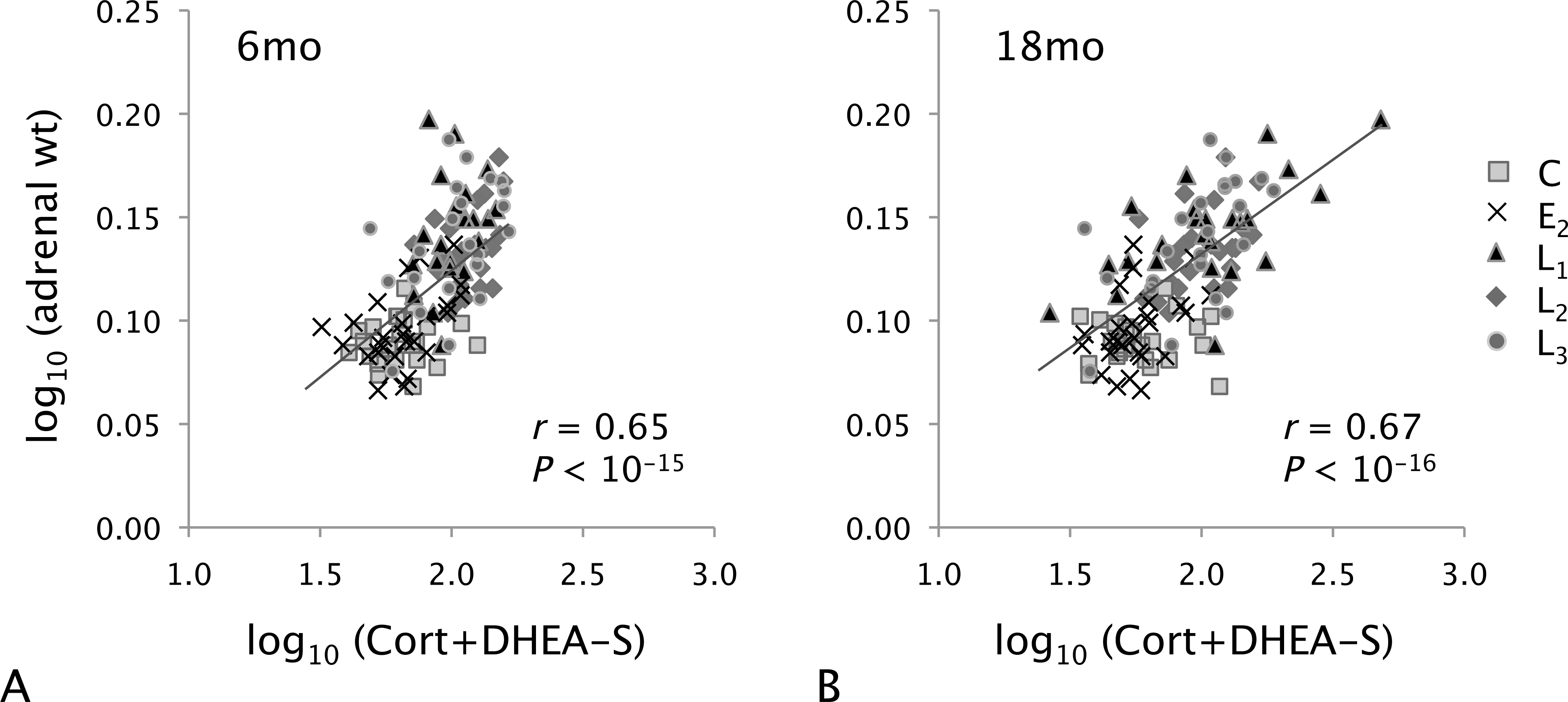
Regression analysis of total adrenal weight and serum concentrations of Cort + DHEA-S at (A) 6 mo and (B) 18 mo of treatment.
In contrast to the adrenal gland, E2 and levormeloxifene had similar effects on pituitary gland measures. Mean serum FSH and LH concentrations increased from ˜ 1 ng/mL to 8 to 12 ng/mL following ovariectomy in the control group, while subsequent treatment with E2 and all SERM doses resulted in at least 50% lower FSH and LH compared with placebo at 6 mo and 12 mo of treatment (p < .001 for all; Table 4). Similar changes have been reported in postmenopausal women following levormeloxifene treatment (Skrumsager et al. 2002). No significant group differences were seen for pituitary weight (Table 4).
Treatment effects on serum gonadotropins and pituitary weight.
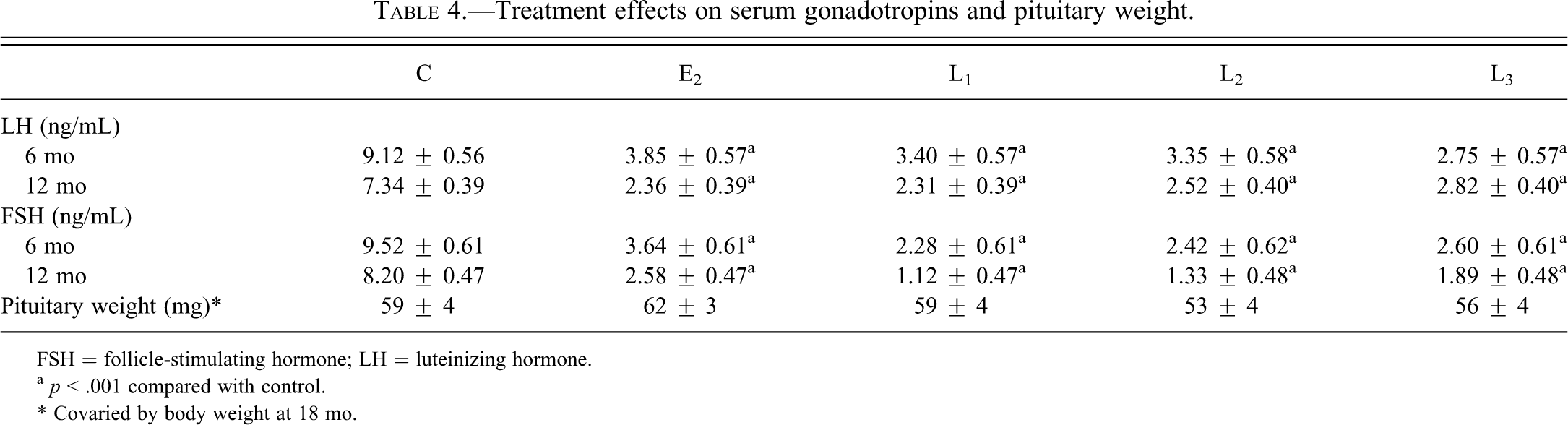
FSH = follicle-stimulating hormone; LH = luteinizing hormone.
a p < .001 compared with control.
* Covaried by body weight at 18 mo.
Discussion
Data presented here demonstrate that the adrenal cortex is a potential target of SERM activity. The experimental SERM levormeloxifene increased adrenal weight and serum concentrations of Cort and DHEA-S at all doses given. In contrast, E2 had no significant effects on adrenal weight or adrenocortical steroids. Both levormeloxifene and E2 reduced serum gonadotropins to a similar degree, suggesting that adrenocortical effects of levormeloxifene were not directly mediated by changes in these pituitary hormones.
Prior data related to adrenocortical effects of SERMs are limited. In a previous small macaque study, tamoxifen but not raloxifene resulted in 50% higher adrenal weights (Sikoski et al. 2007). Histologically, this effect corresponded to thickening of the zona fasciculata (the primary site of adrenal Cort production), with cellular hypertrophy and vacuolation. Similar results were observed in a larger macaque study (described in Cline et al. 1998) in which ovariectomized female macaques treated with a standard human equivalent dose of tamoxifen for 2.5 y had mean adrenal weights 74% higher than the placebo group (p = .001; unpublished data). A small short-term study of ovariectomized female macaques also reported estrogen antagonist properties of tamoxifen on the hypothalamic-pituitary-adrenal axis, including diminished adrenocorticotropic hormone (ACTH) and Cort response to ACTH across a 20-h period following treatment (Wilson et al. 2003). It is unclear how or if these acute effects relate to the observed changes in adrenal weight following more chronic exposure. In studies of postmenopausal women with breast cancer, tamoxifen has been reported to increase Cort and DHEA-S (Rossi et al. 2009) and to increase Cort and decrease DHEA-S (Löfgren et al. 2004). In contrast, raloxifene treatment was shown to have no effect on the androgen profile in a cross-sectional study (Christodoulakos et al. 2005) and to reduce Cort, DHEA-S, and A4 in a 12-mo trial (Genazzani et al. 2003), suggesting that adrenal effects may vary for different SERMs.
Adrenocortical cells express ERs and respond to changes in ER activity (de Cremoux et al. 1998), suggesting that SERMs may directly affect adrenal function via ERs. Several lines of evidence suggest that ER activity may negatively regulate certain androgenic pathways and that ER antagonism may facilitate androgenesis. For example, estrogen treatment inhibits androgen production in Leydig and ovarian thecal cells by decreasing expression and activity of cytochrome P450 17α-hydroxylase/17,20-lyase (CYP17), a key enzyme in both androgen and glucocorticoid production (Nozu, Dufau, and Catt 1981; Johnson, Martin, and Tsai-Morris 1984). Conversely, male ER-α knockout mice display elevated androgen synthesis and CYP17 activity in Leydig cells (Akingbemi et al. 2003), and CYP17 upregulation in the ovary has been reported in rats treated with various SERMs (Harris et al. 2008). The observed SERM effects could also be mediated indirectly through increased ACTH, as reported in one prior study of postmenopausal women receiving raloxifene (Genazzani et al. 2003). However, this latter study, as in the macaque tamoxifen study (Wilson et al. 2003), found decreased adrenal responsiveness to ACTH challenge, supporting the idea that SERM effects are due at least in part to direct alteration of adrenocortical steroidogenesis.
Although not well documented, SERMs and other estrogen ablative agents are popular off-label supplements used by male body builders to increase endogenous androgen production (Basaria 2010). These effects are thought to be mediated by increased gonadal T production secondary to increased LH (as reported in premenopausal but not postmenopausal women). The degree of androgen increase observed here (40%–70%) was similar to that reported in men taking estrogen antagonists (Leder et al. 2004; Taxel et al. 2001), only in the absence of gonadal influence. These findings raise the possibility that androgenic SERM effects in men may be due in part to adrenocortical as well as gonadal effects. Such androgenic effects may have clinical relevance given recent interest in the use of SERMs to treat benign prostatic hyperplasia and to support bone strength in prostate cancer patients receiving anti-androgen therapy (Ho and Habib 2011; Smith et al. 2010).
Adrenocortical steroids are associated with risk for a variety of postmenopausal health conditions, including osteoporosis, cognitive decline, and diabetes. Findings from this study identify the adrenal cortex as a potential target of SERM action and suggest that extended use of certain types of SERMs may contribute to higher circulating Cort and androgens. Potential downstream effects of these hormonal changes on other body systems are not clear at this time. Further studies are needed to determine mechanisms underlying the observed SERM effects, including changes in ACTH and response to ACTH challenge, direct effects on specific adrenocortical cell enzymes, and concurrent alterations in steroid hormone and Cort-binding globulins, which help regulate bioactive serum androgens and Cort, respectively. This information may be useful in the safety evaluation of future SERM therapies and contribute to a better understanding of hormonal regulation of adrenocortical activity.
Footnotes
Acknowledgments
The authors thank Dewayne Cairnes, Melissa Ayers, and Jean Gardin for technical assistance.
Financial disclosure: This work was supported by the National Institutes of Health (NIH) National Heart, Lung, and Blood Institute (NHLBI; HL 45666 and HL 40962) and by Novo Nordisk. The contents are solely the responsibility of the authors and do not necessarily represent the views of the NIH, NHLBI, or Novo Nordisk. The authors declare that there are no other potential conflicts of interest that could be perceived as biasing the impartiality of the research reported.
