Abstract
Surgical implantation of radiotelemetric transmitters is a current practice to collect a variety of physiological parameters in unrestrained laboratory animals, and in rodents in particular. In this study, the incidence of peritoneal sarcomatosis arising secondary to surgically implanted telemetry devices (< 15% of implanted Sprague Dawley rats) is considered to represent a significant issue for both animal welfare and data validity in affected animals. Macroscopically, the telemetry-associated fibrosarcomas spread along the visceral and parietal peritoneum and mesentery surrounding abdominal organs. The histologic morphology of these sarcomas was typically an undifferentiated sarcoma, although well-differentiated fibrosarcomas and telangiectatic and pleomorphic variants were noted. Using special stains such as Masson’s Trichrome demonstrated a collagenous extracellular matrix in 50% of these rats, which is consistent with a fibroblastic origin. Immunohistochemical studies clearly delineated the mesenchymal components of the sarcomas (fibroblasts and smooth muscle cells); one case, however, was diagnosed as an osteosarcoma.
Introduction
Rats are the major species used in drug safety assessment because they satisfy regulatory guidelines for both general toxicology and safety pharmacology; there is a large historical database for comparison; they are relatively small, and thus do not require large amounts of drug; and their cardiovascular systems respond to vasoactive substances in a similar way to humans. PhysioTel transmitters are designed for monitoring and collecting data from conscious, freely moving laboratory animals, providing stress-free data collection without the percutaneous infections associated with tether systems. PhysioTel transmitters are available in various sizes to support a variety of research models including mice, rats, dogs, and primates. The shape of DSI transmitters are also designed to accommodate both subcutaneous and intraperitoneal placement (Brockway et al. 1991; Deveney et al. 1998; Kramer et al. 2001).
The scientific literature provides several compelling arguments for the use of implantable radiotelemetry as a more appropriate alternative to traditional methods of monitoring laboratory animals in terms of quality and quantity of the data collected, animal welfare (reduction and refinement alternatives), and to reduce overall animal research costs (Brockway et al. 1991; Deveney et al. 1998; Kramer and Kinter 2003).
Malignant tumors in animals have been linked to the implantation of foreign bodies (Brand, Buoen, Johnson, et al. 1975; Moizhess and Vasiliev 1989), even relatively inert materials such as glass (Brand, Buoen, and Brand 1975; McCarthy et al. 1996). Experimental animals and some humans have been implanted with a variety of biomaterials, orthopaedic hardware, or prosthetic joints, which have shown either a sarcoma or a tissue reaction developed at implantation sites that were considered “proliferative, probably preneoplastic” (Keel et al. 2001; Kirkpatrick et al. 2000; Memoli et al. 1986; Scheon 2004). A smooth surface, mature fibrosis, dormancy of macrophage activity, an inflammatory reaction and close contact of the carcinogenic cell with the foreign body (FB) were noted as preconditions for FB tumorigenesis (Beachy et al. 2004; Mohizhess 2008).
Similar tumors have been observed, albeit infrequently, at microchip implantation sites, and these tumors also are associated with chronic inflammation (Sura et al. 2011; Tillmann et al. 1997; Troyk 1999). Although the use of telemetry devices in rats and other species is not new, the development of malignant neoplasms in the peritoneal cavity following abdominal implantation of telemetry devices in rats has not been reported.
In this report, the authors present the first reported cases of abdominal sarcomas arising in association with implanted telemetry transponders in eight adult male CD rats over a period of one year after implantation. In our facility, two types of radiotelemetry devices were implanted in the abdominal cavity of fifty-five male Sprague Dawley (SD) rats during the period of 2007–2010. Of these animals, eight rats developed peritoneal sarcomatosis adjacent to or surrounding the telemetry implants. Full necropsy, histopathology, and immunohistochemistry were performed on this cohort of rats, and these results form the basis of this assessment of peritoneal sarcomatosis associated with telemetry implants in SD rats.
Materials and Methods
Animals and Surgery
All animal experimentation was licensed under the British Animals (Scientific Procedures) Act 1986. Male SD - Crl:CD (SD) rats were obtained from Charles River, Manston, UK. They were provided with ad libitum RM1 (E) SQC, Special Diet Services pellets, and a bottle of reverse-osmotic treated tap water. The room temperature was 21°C ± 2°C with a relative humidity of 55 ± 10% and a twelve-hour light/dark cycle. For research purposes, they were singly housed in conventional open-top cages (Techniplast, Italy) under a conventional barrier with paperwool, chewing wood stick medium blocks and Maxi Fun Tunnels as enrichments, and on Litaspen Premium wood chip bedding (all by Lillico). Animals weighed between 250 and 350 g at the time of surgery, which correlated to approximately seven to nine weeks old based on Charles River growth curve.
A total of fifty-five rats were chronically instrumented with radiotelemetric implant. Each transmitter contained a temperature and pressure sensor, a pair of flexible leads, a micro-electronics module to process and digitize the information from the sensors, a magnetic switch with built-in battery, and a radio frequency transmitter. Of these animals, forty-three rats were instrumented with an electroencephalogram/electromyogram (EEG/EMG) radiotelemetric implant (PhysioTel-Plus, Model TL11M2-F40-EET, Data Sciences Intl., St. Paul, MN, USA). An EEG cortical electrode was implanted on the right hemisphere. A pair of EMG electrodes was implanted in the dorsal neck muscle. A further twelve rats were implanted with electrocardiogram/ blood pressure (ECG/BP) radiotelemetric implant (PhysioTel-Plus, Model TL11M2-C50-PXT, Data Sciences Intl.). The BP electrode was placed in the abdominal aorta via the femoral and iliac arteries. The positive biopotential ECG lead was sutured to the caudal side of the xiphoid process of the sternum. The negative ECG biopotential lead was tunneled subcutaneously to a midline incision on the ventral side of the neck, passed down toward the manubrium sterni between the sternomastoid and sternohyoid muscles, and secured close to the thoracic inlet. In both surgeries, the transmitter was sutured to the right side of the abdominal wall inside the peritoneal cavity. As postoperative care, animals received carprofen (5 mg/kg SC UID) at the induction and up to four days post-surgery and enrofloxacine (100 mg/l of drinking water) from the day prior the surgery up to five days post-surgery. They also received a mash of food pellets wet with drinking water following the surgery until they regained their presurgery body weight.
Necropsy and Histopathological Examination
The fifty-five animals were divided into two cohorts. In an initial cohort of forty-two rats (thirty implanted with an EEG/BP telemetry transducer and twelve implanted with an ECG/EMG telemetry transducer), a gross necropsy with tissue collection for histopathological examination necropsy was performed on two animals that were found dead and five clinically ill animals that were euthanized because of clinical signs. Of these seven animals, five had been implanted with an EEG/EMG telemetry transducer and two had been implanted with an ECG/BP telemetry transducer. The remaining thirty-five animals remained asymptomatic and were terminally euthanized twelve months following the surgery and discarded. An additional cohort of thirteen terminally euthanized rats implanted with an EEG/EMG telemetry transducer underwent a gross necropsy. Lesions and core tissues were collected from one animal presenting with lesions that were similar to the preterminally euthanized animals in the initial cohort.
Tissues were fixed in 10% neutral buffered formalin for approximately forty-eight hours prior to routine paraffin embedding and histologic processing followed by staining with hematoxylin and eosin. Histologic sections were examined on an Olympus BX-51 optical microscope, and photomicrographs were captured with an Olympus ColorView IIIu digital camera with Cell D software (Soft Imaging Systems GmbH, Munster, Germany). Further analysis with a panel of histologic (Masson’s Trichrome and phosphotungstic acid hematoxylin) and immunohistochemical stains was performed on serial sections.
Immunohistochemistry Method
Immunohistochemistry was performed using the avidin-biotin perioxidase complex (ABC) technique for different antigens to characterize the immunophenotype of the tumor (Makin et al. 1984; Moll et al. 1982). Five antibodies were used: a monoclonal mouse antibody against vimentin (1:200, DAKO, Dako Corporation, Carpinteria, CA), smooth muscle actin (SMA; 2 µg/mL, DAKO), S-100 protein (1:100, DAKO), CD31 (1:100, DAKO), and polyclonal pancytokeratin (1:100, DAKO).
To obtain immunostained preparations, sections (4 µm thick) of the paraffin-embedded tissue were cut from the appropriate blocks onto slides, then slides were deparaffinized by immersion in three changes of xylene and rehydrated in descending alcohol solutions. Slides were washed four times in phosphate-buffered saline (PBS), pH 7.4. Endogenous peroxidase activity was blocked by incubating the slides in 3% hydrogen peroxide for ten minutes at room temperature and then rinsing the slides twice in PBS. Slides were then incubated in normal goat serum (blocking serum) 1:10, for twenty minutes in a moist chamber at room temperature, then excess blocking agent was removed.
Slides were boiled for ten minutes in a 1:10 dilution of Antigen Retrieval Citra solution (BioGenex, San Ramon, CA), using a microwave oven on a power setting of 600 watts. Slides were allowed to cool twenty to thirty minutes and were then rinsed in several changes of distilled water. Tissue sections for vimentin and S100 were treated with 0.1% trypsin at 37°C for twenty minutes.
Slides were incubated for two hours with one of the primary antibodies described previously at appropriate dilutions, or negative control reagent at room temperature. The slides were washed in PBS for five minutes, and the secondary antibody (prediluted biotinylated anti-immunoglobulins in PBS with carrier protein, 0.1% sodium azide, and 1% human serum) was added to slides and incubated in a moist chamber at room temperature for twenty minutes. The slides were rinsed with buffer, incubated with the label (prediluted horseradish peroxidase-labeled streptavidin in PBS with carrier protein and 0.01% thimerosal) for twenty minutes at room temperature, rinsed well with buffer, and incubated for five minutes with 3,3′-diaminobenzidine (DAB). The color change was monitored on positive control slides and stopped by immersing all slides in deionized water. Slides were counterstained by incubating with Mayer’s hematoxylin for forty seconds, rinsed with tap water, immersed into ammonia water for ten seconds, and then washed with running tap water. Slides were dehydrated through ascending alcohol solutions, cleaned in xylene, and coverslipped. Appropriate positive controls for the antibodies used were run concurrently and showed adequate immunostaining. Negative control sections for immunohistochemical staining were stained without the primary antibody.
Results
In the initial cohort of forty-two rats, seven animals died or were euthanized 8.5–11.5 months following the surgery. Five rats were humanly euthanized by a raised concentration of CO2, and two rats were found dead. Three of the five early euthanized animals presented abdominal distension, and a range of nonspecific clinical signs (including anorexia, weight loss, general malaise, hypothermia, pale mucous membranes, and chromodachryorrhea) was noted in all five rats, resulting in the euthanasia. No clinical signs were noted prior to the death of the two animals found dead. Pathologic findings in affected animals were consistent with peritoneal sarcomatosis originating in the tissue surrounding the telemetry implants. The remaining thirty-five asymptomatic animals of this cohort were terminally euthanized twelve months following implantation and were not necropsied.
However, given the high incidence of peritoneal sarcomatosis, a second cohort of twelve asymptomatic rats was terminally euthanized twelve months following implantation. These rats underwent pathologic examination, and peritoneal sarcomatosis was noted in one out of the twelve.
Macroscopically in all animals, numerous nodular neoplastic masses were noted adjacent to or surrounding the telemetry implants, and extending along the mesentery and the visceral surface of the abdominal wall (Figure 1A) and pancreas (Figure 2B); they also extended variable distance within the skeletal muscles of the abdominal wall (Figure 1C) and abdominal surface of the diaphragm. A majority of the telemetry associated sarcomas had morphology typical of undifferentiated sarcomas, with spindle-shaped neoplastic cells arranged in bundles, streams, and fascicles (Figure 2D). Most neoplastic cells had ovoid to oval nuclei with stippled chromatin and one or more nucleoli. There was mild to moderate anisocytosis and anisokaryosis, and the mitotic figures averaged two to six per 400× magnification. Other morphologic variants included a well-differentiated fibrosarcoma with minimal to abundant collagenous matrix, a telangiectatic sarcoma that formed multiple blood-filled channels (Figure 2E), and a pleomorphic tumor with numerous multinucleate cells (Figure 2F). In one rat, the tumor mass was composed of spindled and polygonal cells arranged in bundles, streams, and whorls. Multifocally, neoplastic cells in some areas were separated or surrounded by a homogeneous eosinophilic matrix consistent with osteoid. Some of the osteoid was mineralized, and neoplastic cells were located within osteoid lacunae (Figure 2G).
Figure 1. Sprague Dawley CD rat: Numerous sessile and pedunculated small nodules from a few millimeters to 8 cm distributed on the serosal surface within the abdominal cavity with a moderate amount of hemorrhage; note the presence of the transmitter (arrow).
Figure 2. Abdominal masses, Sprague Dawley CD rats. Invasion of the liver (A), exocrine and endocrine (arrow) pancreas (B), adjacent skeletal muscles (SK) of the abdominal wall (C), by the neoplastic cells which are often arranged in bundles, streams, and fascicles (D) with telangiectatic (E), pleomorphic variants with multinucleation (F), and focal production of osteoid (arrows) and small bony trabeculae (arrow head) (G). H&E stain; 5×.
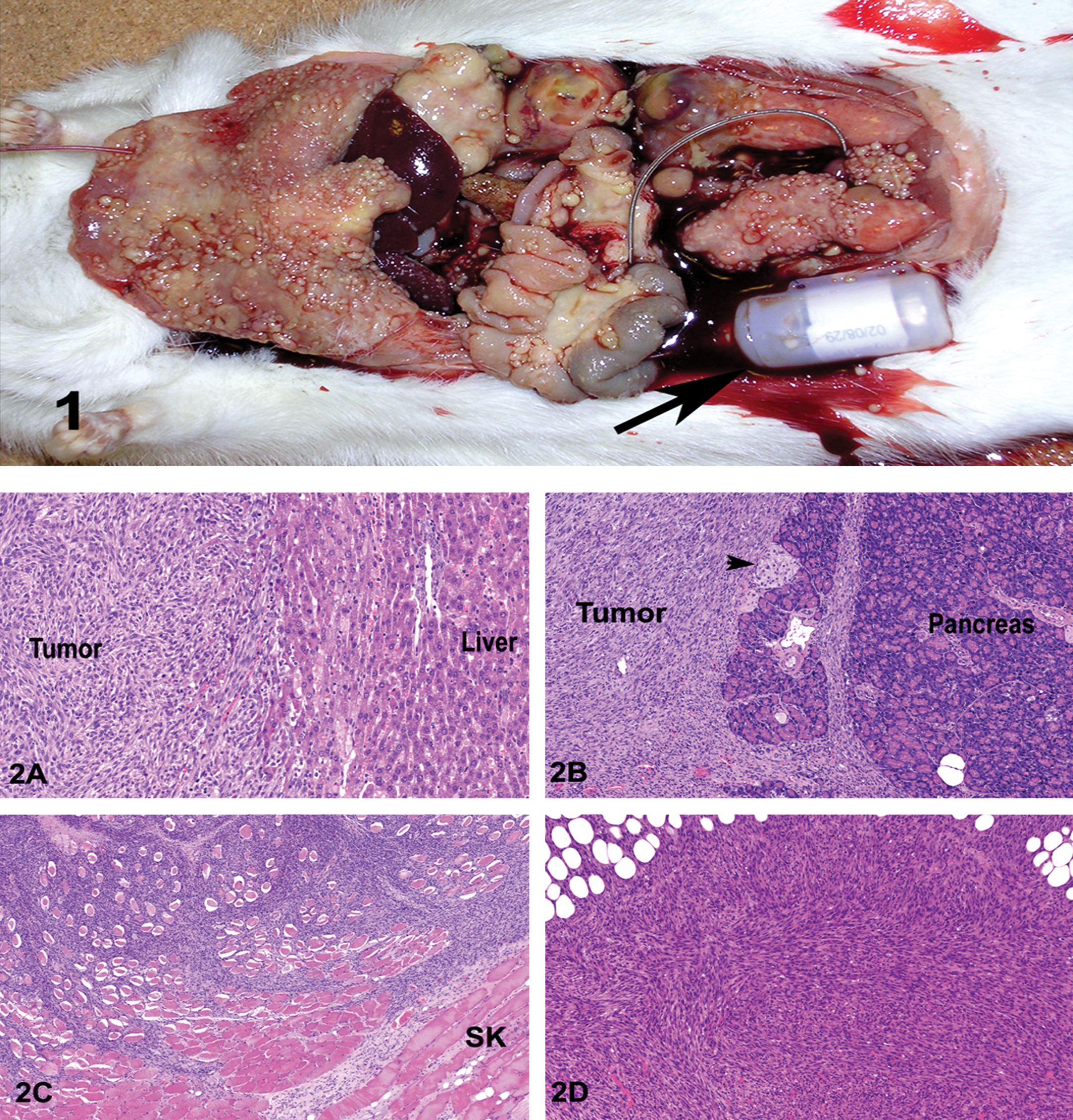
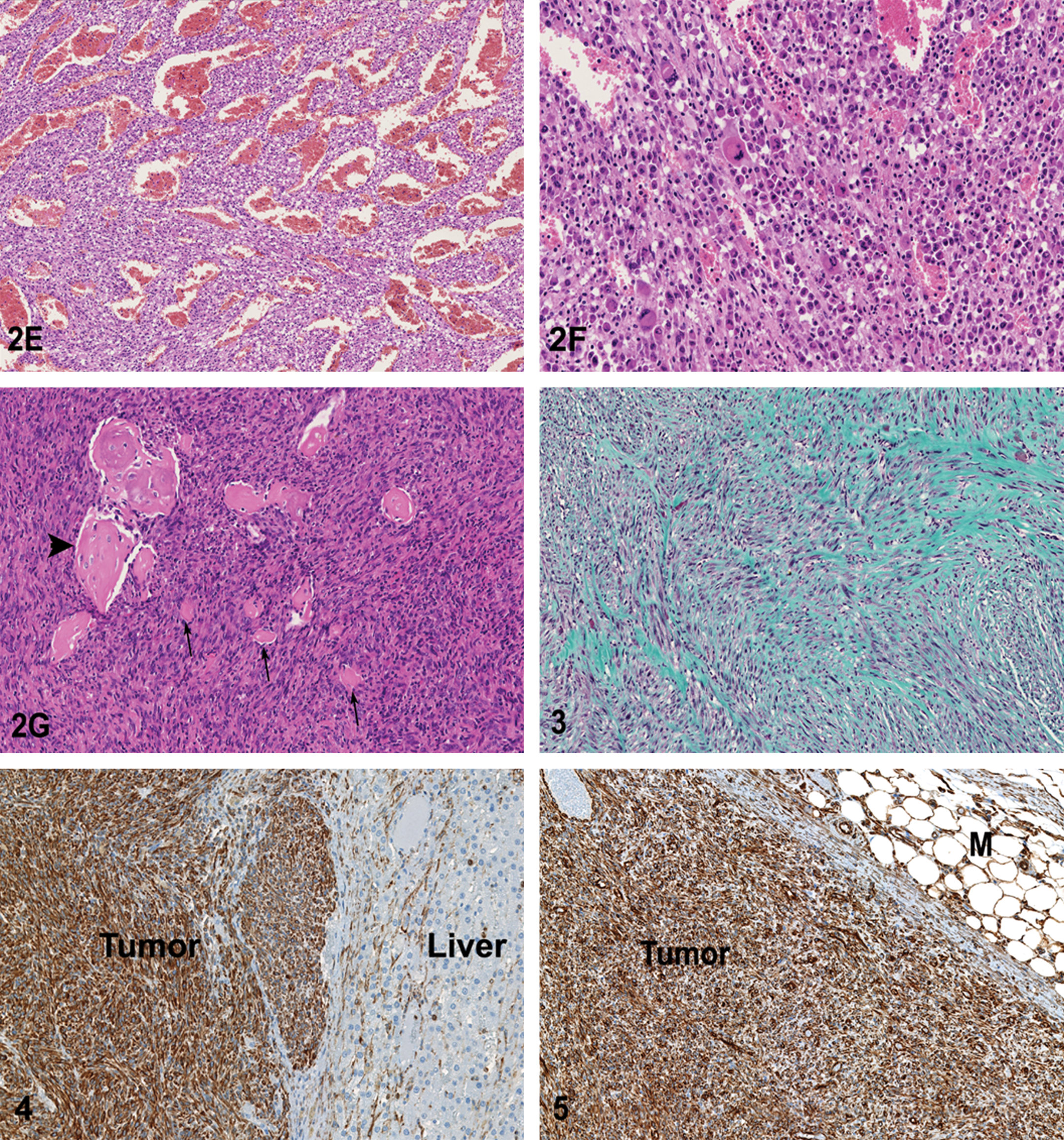
Despite these variations in morphology, 50% of telemetry-associated sarcomas were noted to make a collagenous stroma when stained with Masson’s Trichrome (Figure 3 ), consistent with a fibroblastic cellular origin. None of the tumors showed evidence of striated muscle origin on PTAH stain. Immunohistochemical examination of the tumors to elucidate histogenesis demonstrated strong cytoplasmic immunoreactivity to vimentin (Figure 4 ), confirming their mesenchymal origin. The neoplastic cells from three rats showed mild to marked positive cytoplasmic staining to SMA, consistent with smooth muscle origin, and they therefore were reclassified as leiomyosarcomas (Figure 5 ). Conversely, no tumors showed immunoreactivity for pancytokeratin (for epithelial origin), S100 (for neural crest, histiocytic, and other subtypes of mesenchymal cells), or CD31 (for leukocytic or endothelial cell origin). For all antibodies, significant immunoreactivity was present in the positive control sections but was not detected in the negative controls. Thus immunohistochemical examination confirmed the diagnosis of peritoneal sarcomatosis, with a further subclassification of fibrosarcoma in 50%, leiomyosarcoma in 37.5%, and osteosarcoma in 12.5% of tumors (Table 1 ). In summary, of a total number of fifty-five rats surgically implanted with radiotelemetry devices, 14.5% (8/55) died or were euthanized as a result of peritoneal sarcomatosis. However, as thirty-five asymptomatic animals in the original cohort were discarded without necropsy examination, the percentage that developed peritoneal sarcomatosis may be higher.
Figure 3. Abdominal mass, Sprague Dawley CD rat. Masson’s Trichrome stain highlights the collagen, indicate fibroblastic origin for the neoplastic cells in most animals. Masson’s Trichrome stain; 10×.
Figure 4. Abdominal mass, liver, Sprague Dawley CD rat. Immunohistochemistry for vimentin. Most neoplastic cells and a few nonepithelial (interstitial) cell hepatic stroma showed strong immunoreactivity in the cytoplasm. Avidin-biotin immunoperoxidase method and 3,3′-diaminobenzidine as chromogen. Hematoxylin counterstain; 20×.
Figure 5. Abdominal mass-mesentery, Sprague Dawley CD rat. Immunohistochemistry for smooth muscle actin. Most neoplastic cells, and prexisting smooth muscle cells of small blood vessels showed strong immunoreactivity in the cytoplasm. Avidin-biotin immunoperoxidase method and 3,3′-diaminobenzidine as chromogen. Hematoxylin counterstain; 20×.
Results of immunohistochemical stainsa of peritoneal sarcomatosis.
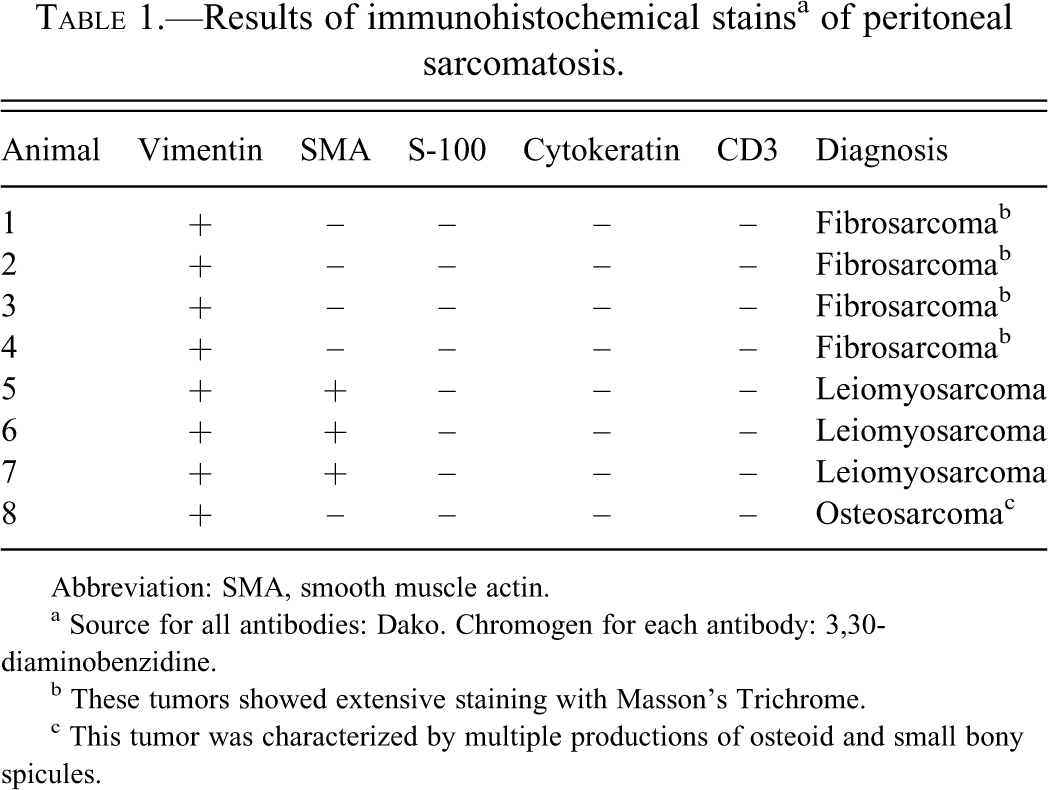
Abbreviation: SMA, smooth muscle actin.
a Source for all antibodies: Dako. Chromogen for each antibody: 3,30-diaminobenzidine.
b These tumors showed extensive staining with Masson’s Trichrome.
c This tumor was characterized by multiple productions of osteoid and small bony spicules.
Discussion
Peritoneal sarcomatosis from sarcomas arising adjacent to intraperitoneal telemetry implants is reported to be a relatively significant problem in SD CD rats in the current study, and it accounted for significant animal attrition with this model in a previous study in our facility (unpublished data). The authors of this report present the first reported cases (eight rats) of sarcoma one year after abdominal implantation of telemetry devices in rats. These tumors were intimately associated with the implants and in some cases infiltrated into and partially replaced adjacent organs including the mesentery, liver, spleen, pancreas, diaphragm, and skeletal muscles of the abdominal wall. The natural incidence of spontaneous sarcomas of mesenchymal origin (soft tissue sarcomas) and gastrointestinal stromal tumors are rare in rats (Boorman and Everitt 2005; Greaves and Barsoum 1990); however, the incidence of the peritoneal sarcomas in this study is almost one in seven.
Masson’s Trichrome staining showed evidence of collagen production by 50% of tumors, and immunohistochemistry supported a sarcomatous origin in all eight cases. Based on histopathology, histochemistry, and immunohistochemistry techniques, the histiogenesis of the tumors was consistent with fibroblastic fibrosarcoma (four of eight cases), leiomyosarcoma (three of eight cases), and osteosarcoma (one of eight cases). The transducer (the part in the abdomen) of both types of the telemetry devices used in this study is coated with silicone elastomer, which suggests that there was no link between the type of implant and the types of tumors reported here.
Although the mechanism of carcinogenesis could not be definitively determined, the fact that the tumors in our cases were grossly and histologically intimately associated with the implants, and that spontaneous peritoneal sarcomatosis in rats is rare, suggests a causal relationship to the telemetry devices. The putative mechanism of carcinogenicity is therefore considered to be foreign body–induced tumorigenesis resulting from mild, continuous, chronic irritation.
Radiotelemetry has numerous benefits for animal welfare and validity of scientific data. Additionally, it is possible to maintain instrumented animals for repeated investigations over the long term in drug discovery programs, creating cost efficiency and reduction of the total numbers of animal used (Deveney et al. 1998; Kramer et al. 2001). However, clinical and scientific evidence supports the concept that the development of malignancy in relation to the use of implanted biomaterials, although a rare occurrence, is a potential complication of their use (Keel et al. 2001; Kirkpatrick et al. 2000; Memoli et al. 1986; Scheon 2004; Visuri et al 2006).
Several animal models in rats that have been implanted with orthopaedic hardware and prosthetic joints, and various biomaterials, have shown that up to 67% of implant sites developed either a sarcoma or a tissue reaction considered “proliferative, probably preneoplastic” (Keel et al. 2001; Kirkpatrick et al. 2000; Memoli et al. 1986; Scheon 2004). A high proportion of rats with subcutaneously implanted biomaterials (polymers, metals, and ceramics) as an experimental model developed sarcomas around the implantation site (Kirkpatrick et al. 2000). The fact that malignancies have arisen in close proximity to metallic implants in these studies is strong evidence that there may be a causal relationship (Scheon 2004; Sinibaldi et al. 1976).
Vaccinations and injections have been found to lead to sarcomas in cats (Kass et al. 2003), dogs (Vascellari et al. 2003), and ferrets (Munday et al. 2003). In humans, there are a number of reports of pacemaker implantation site malignancy that have involved both sarcomas and carcinomas (Bhandarkar et al. 1993; Biran et al. 1979; Carpentier et al. 2000; Fraedrich et al. 1984; Okamoto et al. 2010), and similarly, one of 104 dogs with pacemaker implantation is reported to have developed fibrosarcoma at the pacemaker generator site, thirty months after implantation (Johnson et al. 2007).
Several studies and some case reports that indicate that microchip–transponder implants have been associated with neoplasia in different animals including laboratory mice and rats, and pet dogs (Ball et al. 1991; Blanchard et al. 1999; Elcock et al. 2001; Jansen et al. 1999; Lambooij 1995; Mader et al. 2002; Pessier et al. 1999; Siegal-Willott et al. 2007; Sura et al. 2011; Tillmann et al. 1997; Troyk 1999; Vascellari et al. 2004; Vascellari et al. 2006). The majority of transponder-associated tumors have been mesenchymal in origin, and the mechanism of carcinogenicity is thought to be foreign body–induced tumorigenesis (Brand et al. 1976; Elcock et al. 2001).
One of the major factors thought to influence foreign body–induced tumor formation is inflammation. This effect is usually transient and is known to occur at the implantation sites of microchips (Lambooij 1995; Mader et al. 2002). Acute tissue injury and tissue inflammation have a role in the development of cancer, as they are accompanied by the expansion of stem cell pools and by the transient activation of the Hh and Wnt signaling pathways (Balkwill and Mantovani 2001; Beachy et al. 2004; Cousins and Werb 2002; Tillmann et al. 1997). Stem cells have an increased likelihood of accumulating multiple additional mutations that may be required to increase the rate of cell proliferation and produce clinically significant cancers (Lessard and Sauvageau 2003). Furthermore, inflammation, specifically the chronic type, is associated with cancer, and the chronic inflammatory process itself provides the prerequisite environment for the development of malignancy (O’Byrne and Dalgleish 2001).
Chronic inflammation involves monocytes, macrophages, lymphocytes, plasma cells, and fibroblasts and is also mediated by a range of substances including the complement system, cytokines, growth factors, and reactive oxygen species. The end point is tissue destruction and fibrosis, and such chronic inflammatory conditions may become persistent. Indeed, over recent years, the association with malignancy has grown stronger and a range of tumors has now become linked with chronic inflammation and chronic tissue injury in humans. Examples of chronic tissue damage with this association to neoplasia include exposure to toxins such as organic chemicals (Blair and Kazerouni 1997; Ekbom et al. 1990; Lieber et al. 1979; Wu et al. 2001), chronic bacterial and parasitic infections including Helicobacter pylori, Spirocerca lupi, and other pathogens (Uemura et al. 2001), and chronic inflammatory conditions, such as sclerosing cholangitis and inflammatory bowel disease (Burak et al. 2004; Ekbom et al. 1990). Under conditions of chronic injury, continued signaling pathway activation and presumed expansion of stem cell pools would persist as long as repeated injury prevents full regeneration. This state of continuous stimulation of Hh and Wnt signalling pathways and progenitor cell activation resembles the continuous pathway activity and cell division seen in cancer (Lessard and Sauvageau 2003; Watkins et al. 2003).
Additional factors may be involved in implant-related tumor induction. Implanted biomaterials may release trace amounts of residual compounds such as monomers, catalysts, plasticizers, and antioxidants that were used during the implant manufacturing process. It is possible that such contaminants could be associated with sarcoma development (Scheon 2004). Research also has shown that, through a phenomenon known as the Oppenheimer effect, or Oppenheimer phenomenon (Brand 1994; Oppenheimer et al. 1952), implants of solid materials with a large surface area in soft tissue have induced sarcomas in rodent animal models, even though the material has no inherent toxic or tumorigenic properties (Kirkpatrick et al. 2000; Scheon 2004). There was no reported leakage or an obvious outer damage of the telemetry devices used in this study, and they were functioning properly until the end of the study.
Another factor that could not be totally ruled out and that could have played a role in the development of sarcoma in the cases reported here is the localized cumulative low-electromagnetic exposure related to the presence of the transponder. However, available studies on the role of electromagnetic fields in the development of glioma and acoustic neuroma among users of wireless telephones are of insufficient quality, consistency, and statistical power to permit a conclusion regarding the presence or absence of a causal association between exposure and cancer (Lehrer et al. 2011).
In this study, the incidence of telemetry-associated peritoneal sarcomatosis (< 15% of implanted SD rats) is considered to constitute a significant issue for both animal welfare and data validity in affected animals. However, the risk of this complication needs to be weighed against the benefits to animal welfare, ease of data collection, and improved data quality associated with the use of telemetry devices. In order to maximize animal welfare, we recommend not keeping telemetered animals for more than twelve months post-implantation, as the incidence of clinical sarcomatosis was noted to rise significantly with post-surgical intervals longer than twelve months in our facility (data not shown). Additionally, vigilant and thorough clinical examination and body weight monitoring are recommended in animals that have had surgical implants for longer than eight months, in order to facilitate early detection of peritoneal sarcomatosis and minimize the impact on animal welfare from the condition. However, more research into the oncogenic potential of implanted abdominal telemetry is needed, and veterinarians and researchers using telemetered animals for long time periods need to be vigilant for this complication.
Conclusion
Radiotelemetry presents numerous benefits to the quality of animal welfare, validity of scientific data, and the reduction and refinement of laboratory animal use, as it facilitates the maintenance of instrumented animals for repeated investigations over the long term in drug discovery programs. However, as the incidence of telemetry device–associated peritoneal sarcomatosis may approach 15% in SD rats, it presents a significant complication for data integrity and animal welfare. Proactive clinical monitoring and limiting post-surgical implantation survival times to twelve months is recommended to maximize both animal welfare and scientific integrity in this valuable animal model.
Footnotes
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article. The authors received no financial support for the research and/or authorship of this article.
Acknowledgments
We offer special thanks to Benjamin Stone and Rosie Ochs of Pfizer Ltd., Sandwich, Kent, UK, for preparation of slides; and Dr. Marcus Andrews for performing immunohistochemistry. We thank all animal research scientists of Pfizer Ltd., Sandwich, Kent, UK, involved in this study for their collaboration. In addition, we thank Drs. Thomas Brodie and Nabeel Mehdi, veterinary pathologists, for their help editing the manuscript and for their constructive guidance.
