Abstract
GJA1 gene (Connexin43, also known as Cx43) is the most abundant gap junction protein isoform in animal cells and is associated with bone development in embryos. The objective of the present work was to evaluate in vivo osteal development in GJA1-deficient fetal mice through determination of the histological and molecular alterations induced by partial or total deletion of the GJA1 gene. Heterozygous C57BL/6 mice (HT) harboring a null mutation of the GJA1 gene were mated, and pregnant females were submitted to euthanasia and Caesarean section from 12.5 to 19.5 days post coitum (dpc). HT (GJA1+/–) and homozygous (GJA1–/– ) knockout (KO) mutants and wild-type (WT) fetuses were identified by polymerase chain reaction (PCR), and development curves were constructed on the basis of fetus weight and crown-rump length. Histopathological, histochemical, and real-time PCR analyses were performed in order to assess the expression of markers associated with bone development, namely, osteocalcin, osteopontin, alkaline phosphatase, RUNX2, GJA1, GJC1 (Cx45), and GJA3 (Cx46). HT and KO fetuses exhibited delays in the differentiation of osteoblasts and, consequently, in bone development in comparison with the WT group. Additionally, less deposition of mineralized and osteoid matrix was observed in GJA1-deficient fetuses. Bone development in KO fetuses was delayed through the moment of birth, but in HT animals the delay only extended until 17.5 dpc, following which development was normalized. The expression of genes coding for osteocalcin, osteopontin, alkaline phosphatise, and RUNX2 were also delayed in GJA1-deficient fetuses. Animals that exhibited a lower expression of GJA1 presented delayed expression of the GJC1 and GJA3 genes and their corresponding protein products in the bone tissue. The results of the present study contribute to our understanding of the function of GJA1 during bone development and suggest that GJC1 could play a role in restoring intercellular communication in GJA1-deficient mice.
Introduction
Osteogenesis is a complex process involving synchronized cell differentiation coordinated by a genetic program that controls the mechanisms of cell differentiation and function and also their interaction with components of the extracellular matrix (Erlebacher et al. 1995; Karsenty et al. 1999). New bone material is formed according to two main processes, namely, (a) endochondral ossification, in which a cartilage template is vascularized, occupied by osteocytes, and then mineralized, and (b) intramembranous ossification, in which the osteoblasts assemble and mineralize the tissue without the presence of a cartilage template (Stains and Civitelli 2005).
The complex control of cell differentiation and synchronization that occurs during bone development is mediated by the intercellular diffusion of signaling molecules through gap junctions. This exchange process, termed gap- junctional intercellular communication (GJIC) (Loewenstein and Kanno 1966; Civitelli 2008), occurs through channels comprising connexin proteins (Cx) (Willecke et al. 2002) that span the plasma membranes of adjacent cells and facilitate the selective passage of water-soluble molecules (< 1.2 kDa), such as ions, nucleotides, cellular messengers (cAMP and inositol 1,4,5-triphosphate), and secondary metabolites (Civitelli 2008).
Connexin43 (Cx43; gene is known as GJA1) is the most abundant member of the gap junctional intercellular communication family present during embryo development and has been detected in mouse embryos since first cell divisions (Yancey, Biswal, and Revel 1992). The importance of the GJA1 gene has been demonstrated in knockout (KO) mice models in which total deletion of the GJA1 gene (GJA1–/– mutants) caused cardiac malformations and the consequent death of animals soon after birth (Reaume et al. 1995; Ya et al. 1998).
In bone tissue, gap junctional intercellular communication proteins are directly associated with the coordinated development and remodeling of the mineralized matrix components (Lecanda et al. 2000; Stains and Civitelli 2005). More specifically, GJA1 can be found in several types of bone cells, including chondrocytes, osteoclasts, and osteocytes, while osteoblasts express the GJC1 and GJA3 isoforms together with GJA1.
It has recently been shown that mutations in GJA1 are correlated with the rare human congenital disorder oculodentodigital dysplasia (ODDD), the manifestations of which include malformations of the skull, phalanges, and long bones (Richardson et al. 2004). Although individuals suffering from ODDD may exhibit various patterns of mutations, all involve alterations of amino acids that compromise the function of GJICs (Paznekas et al. 2003).
In vitro studies have shown that Cx43 protein (protein from GJA1 gene) not only plays a role in the differentiation of osteoblasts and the functionality of osteocytes, but is also closely related with the expression of GJC1 (Lecanda et al. 1998). Moreover, in comparison with normal osteoblasts, GJA1–/– mutant cells cultivated in vitro exhibit reduced expression of the genes coding for the bone-related proteins osteocalcin and osteopontin, decreased secretion of type I collagen, and diminished mineral deposition (Chung et al. 2006). Additionally, it is reported that GJA1–/– osteoblasts over express GJC1 protein in order to compensate for gap junctional intercellular communication deficiency (Stains and Civitelli 2005). In contrast, no differences in the abundance or distribution of GJA3, a monomeric protein restricted to the trans Golgi apparatus, were observed in GJA1–/– cells, suggesting that this isoform does not play a compensatory role (Lecanda et al. 2000).
Using the GJA1-deficient mouse model, we determined whether genetic deficiency of Cx43 affects skeletal development in vivo. Intramembranous ossification of the cranial vault were delayed in the mutant embryos, and cranial bones originating from migratory neural crest cells were also hypoplastic, leaving an open foramen at birth (Lecanda et al. 2000).
Although numerous studies have focused on GJA1-mediated intercellular communication between bone cells, the function of the GJA1 gene in vivo remains unclear. The objective of the present work was to evaluate osteal development in GJA1-deficient fetal mice by determination of the histological and molecular alterations induced by partial or total deletion of the GJA1 gene.
Materials and Methods
Details of the study were submitted to and approved by the Committee of Ethics in Research of the University of São Paulo (protocol no. 1305/2008).
Origin of Mouse Fetuses
Heterozygous C57BL/6 mice harboring a null mutation of the GJA1 gene (Reaume et al. 1995) were used in the experiments. Pairs were housed together overnight and copulation was confirmed by observation of a vaginal plug the next morning, thus establishing the date at 0.5 days post coitum (dpc). Pregnant females were submitted to euthanasia and subsequent Caesarean section in the morning of a day within the period 12.5 to 19.5 dpc. Fetuses (n = 40) were weighed and the crown
Histopathological Analysis
Fetuses obtained between 12.5 and 16.5 dpc were fixed in 10% formalin for two days while those acquired between 17.5 and 19.5 dpc were similarly fixed for four days. Larger fetuses (i.e., 18.5 and 19.5 dpc) required incisions in the surface to facilitate the infiltration of the fixative agent. Fixed fetuses were embedded in paraffin, cut into 5 µm sections, and stained with hematoxylin eosin stain for histopathological analysis. The analysis of bone development was performed following methodology described previously (Komori et al. 1997; Bialek et al. 2004), and the results obtained for WT fetuses (controls) were compared with those for KO and HT fetuses from the same litter. Six individuals of each genotype from three different litters were used in the experiments.
Immunohistochemical Analysis
Evaluation of the deposition of mineralized matrix in the developing fetuses was performed in two stages. First, the total mineralization of the fetal skeleton was analyzed according to the methodology of McLeod (1980), which involved staining with a solution containing 0.1% alizarin red and 0.3% alcian blue followed by diaphanization with 1% potassium hydroxide in 20% glycerol. In the second stage, specific areas of mineralized matrix deposition were detected in fetal sections following treatment with 10% silver nitrate for 60 min under intense light and subsequent staining with 1% alcian blue and eosin according to the Von Kossa method as modified by Lillie (1965).
Evaluation of the deposition of osteoid matrix in the developing fetuses was performed using an immunofluorescence technique for the detection of type I collagen. Histological sections were submitted to antigen unmasking by treatment with target retrieval solution (pH 6.0; Dako, Carpinteria, CA, USA) for 40 min in a water bath at 95oC followed by digestion with 0.1% pepsin in hydrochloric acid for 20 min at 37 oC. Treated sections were incubated overnight at 4oC with anti-collagen type I primary antibody (1:25; Rockland Immunochemicals, Gilbertsville, PA, USA) and subsequently with anti-rabbit IgG-FITC secondary antibody (1:100; Dako) for 2 hr at room temperature. Negative controls consisted of fetal sections that had not been treated with primary antibody as well as sections of central nervous tissue present in the fetus.
The serial sections were photographed, and morphometric analyses of the areas of mineralized or osteoid matrices present were performed with the aid of Image ProPlus 4.5 (Media Cybernetics, Silver Spring, MD, USA) software. The areas of mineralized or osteoid matrices were expressed as percentages of the total area of the rib cage. Experiments were carried out in duplicate, and the rib cages of all animals (n = 6) were examined.
RT-PCR Analysis
Quantitative real-time (RT) PCR was used to analyze the marker genes associated with bone development. Following fixation of fetuses (n = 6) with cold ethanol for 1 hr, the bones were isolated and any attached skin, muscle, and tendons removed under the stereomicroscope (SZX10 model, Olympus, Center Valley, PA, USA). mRNA was extracted from bone material with TRIzol® reagent (Invitrogen, Carlsbad, CA, USA), and the corresponding cDNA synthesized using Superscript II Reverse Transcriptase (Invitrogen) and oligo(dT) primers. RT-PCR was performed with the aid of Applied Biosystems (Foster City, CA, USA) TaqMan Gene Expression Assay kits used according to the manufacturer’s instructions. The primers and probe sets employed were specific for osteocalcin (Applied Biosystem cat. no. Mm03413826_mH), osteopontin (cat. no. Mm00436767_m1), alkaline phosphatase (cat. no. Mm00475834_m1), RUNX2 (cat. no. Mm00501580_m1), type I collagen (cat. no. Mm01165187_m1), GJA1 (cat. no. Mm00439105_m1), GJC1 (cat. no. Mm00433624_m1), GJA3 (cat. no. Mm00444263_s1), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH; cat. no. Mm99999915_g1). The analysis of relative gene expression data was performed according to the method of Livak and Schmittgen (2001) using the GAPDH gene as internal control and WT fetuses as controls for normalization.
Immunofluorescence Analysis
Connexins Cx45 and Cx46 (GJC1 and GJA3, respectively) were evaluated using an immunofluorescence technique. Histological sections (n = 6) were submitted to antigen unmasking by treatment with Tris-EDTA buffer (pH 9.0) for 20 min in a water bath at 95oC. Sections were subsequently incubated overnight at 4oC with anti-Cx45 (Invitrogen) and anti-Cx46 (kindly supplied by Dr. Michael Koval, Institute for Environmental Medicine, University of Pennsylvania School of Medicine, Philadelphia, PA, USA) monoclonal antibodies (Sanches et al. 2009) at dilutions of 1:3000 in TNB buffer (0.1 M Tris-HCl, 0.15 M NaCl, 0.5% blocking reagent, pH 7.5). Immunolabeling was performed by treating sections with biotinylated anti
Statistical Analysis
Data were checked for normality of distribution and homogeneity of variance and subsequently analyzed by ANOVA. Tukey tests, with the significance level set at 5%, were applied in order to assess the significance of differences between treatment means.
Results
Fetal Growth Was Not Affected in GJA1-Deficient Mice
The developmental parameters of wild-type fetuses and those of heterozygous GJA1+/– and homozygous GJA1–/– knockout mutant fetuses were monitored from 12.5 days post coitum onward. Fetal development curves, constructed on the basis of weight and crown
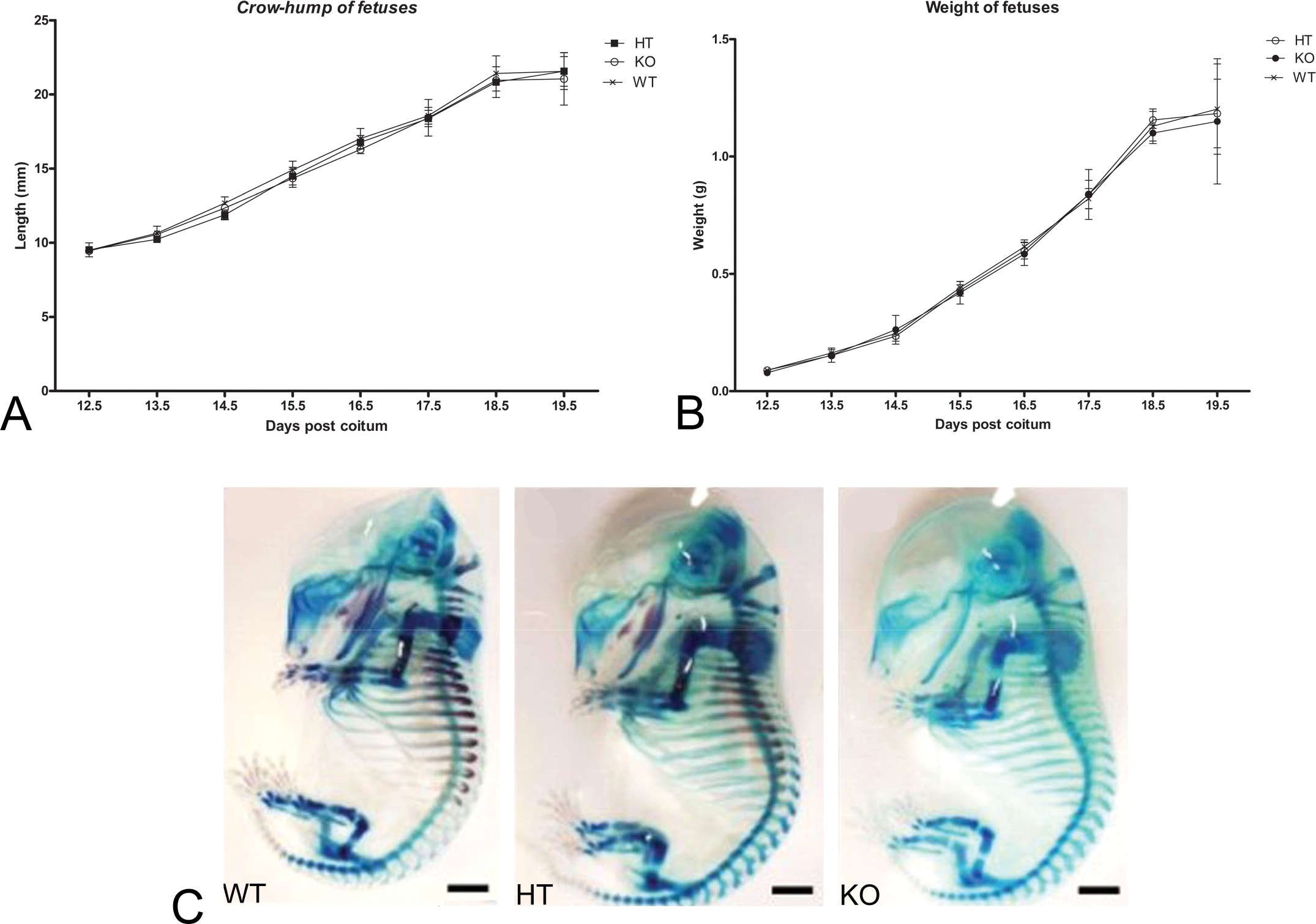
Growth curves of mouse fetuses at different gestational times (dpc = days post coitum). (A) Variation in the crown-rump length of fetuses from wild type (WT), heterozygous C57BL/6 Cx43+/– mutants (HT) and Cx43–/– knockout mutants (KO). (B) Variation in the weight of fetuses from WT, HT, and KO and genotypes. There were no significant differences between genotypes with respect to crown-rump length and weight of fetuses according to ANOVA (p < .05).
Bone Cell Differentiation Was Delayed in GJA1-Deficient Mice
Morphological evaluation of HT and KO fetuses revealed that mineralization was delayed in all bones in comparison with the WT group. Thus, WT fetuses clearly exhibited extensive deposition of mineralized matrix at 15.5 dpc while KO fetuses showed no signs of this process during the same period (Fig. 1C). HT fetuses, on the other hand, exhibited an intermediary state of osteal mineralization at 15.5 dpc.
Histological analysis of ribs showed that partial or total absence of GJA1 induced major alterations in cell differentiation during the development of bone tissue. At the beginning of the experimental period (13.5 dpc), chondrocytes in WT fetuses were in an advanced stage of differentiation and the cartilage template that would give rise to the rib cage had already formed. However, the rib regions of HT and KO fetuses were still colonized by undifferentiated cells at this stage, thus demonstrating a delayed cellular differentiation (Fig. 2 , panels A-C).
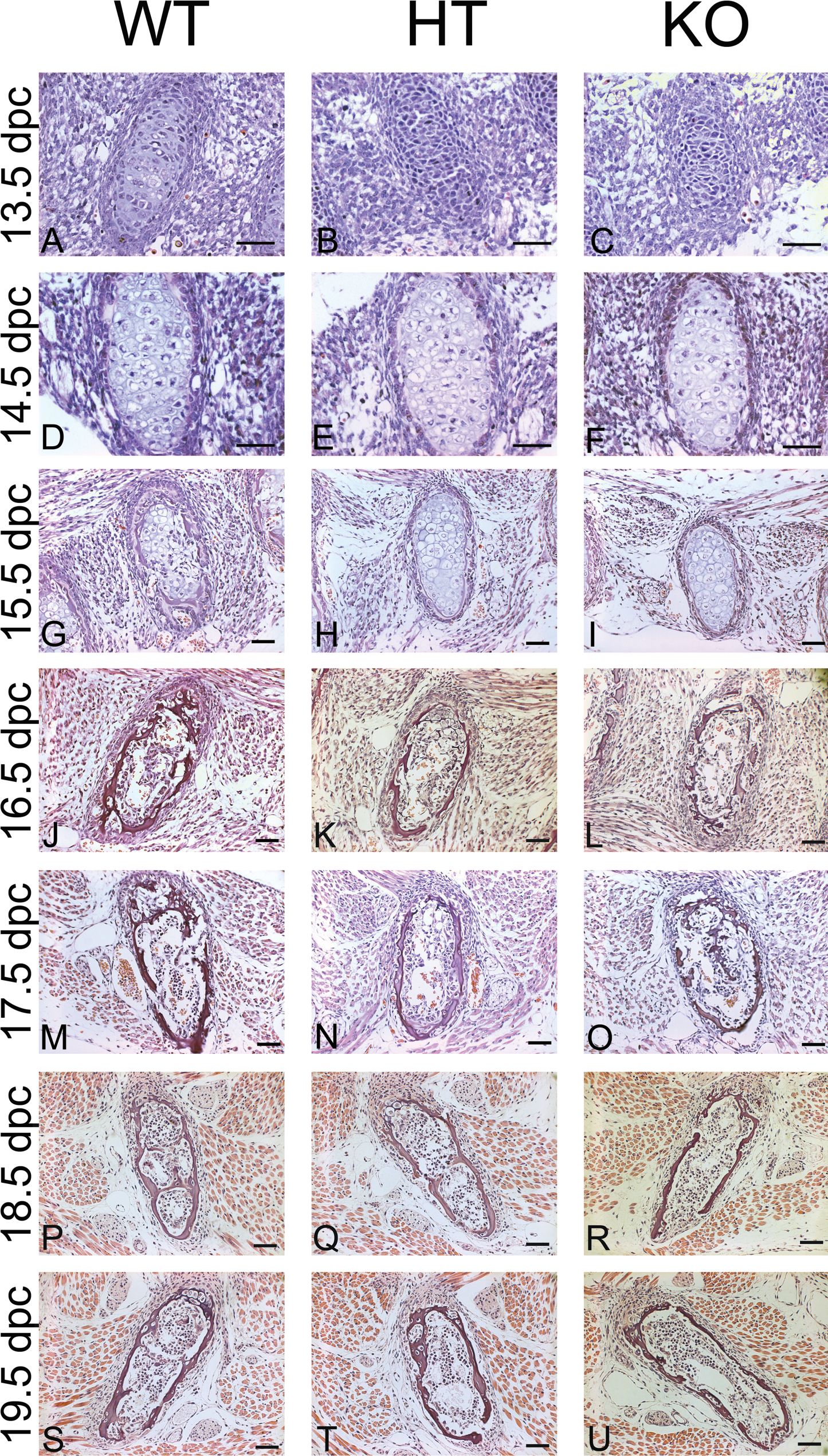
Histological characteristics of fetal rib cages at different gestational times (dpc = days post coitum). Staining with hematoxylin eosin from 12.5 to 19.5 dpc: KO mutant fetuses showed delayed cell differentiation and bone development during the whole experimental period in comparison with the WT group; HT fetuses exhibited similar characteristics to their KO counterparts at 13.5 dpc, but a more mature stage of development similar to that of WT mice from 17.5 dpc onwards. Bar = 50 µm.
At 15.5 dpc, WT fetuses showed intense differentiation of osteoblasts into osteocytes, with migration of the latter to the center of the cartilage structure and deposition of osteoid matrix. In contrast, chondrocytes in the HT and KO fetuses had not been replaced by more mature bone cells and the cartilage templates were populated by hypertrophic chondrocytes (Fig. 2, panels G-I). Indeed, the replacement of chondrocytes by osteocytes in GJA1-deficient fetuses occurred only after 16.5 dpc, while at this time, WT fetuses already showed intense deposition of mineralized matrix in the rib cage with the presence of strong trabeculae throughout the entire structure (Fig. 2, panels J-L).
At 18.5 dpc, no histological differences could be detected between WT and HT fetuses
The delay in bone development in HT and KO fetuses was confirmed by morphometric analysis in which the areas of mineralized and osteoid matrices were compared with the total area of the rib cage. As shown in Fig. 3A, the deposition of osteoid matrix within HT and KO mice was less than that observed in the WT group between 13.5 and 15.5 dpc. Delayed bone development was even more apparent from the rate of deposition of mineralized matrix (Fig. 3B). Thus, the mineralized areas in KO fetuses were smaller than in their WT counterparts from the start of osteal mineralization (15.5 dpc) right through until the moment of birth. HT fetuses also exhibited reduced deposition of mineralized matrix in relation to the WT group up to 16.5 dpc, although from 17.5 dpc onward such differences were not statistically significant.
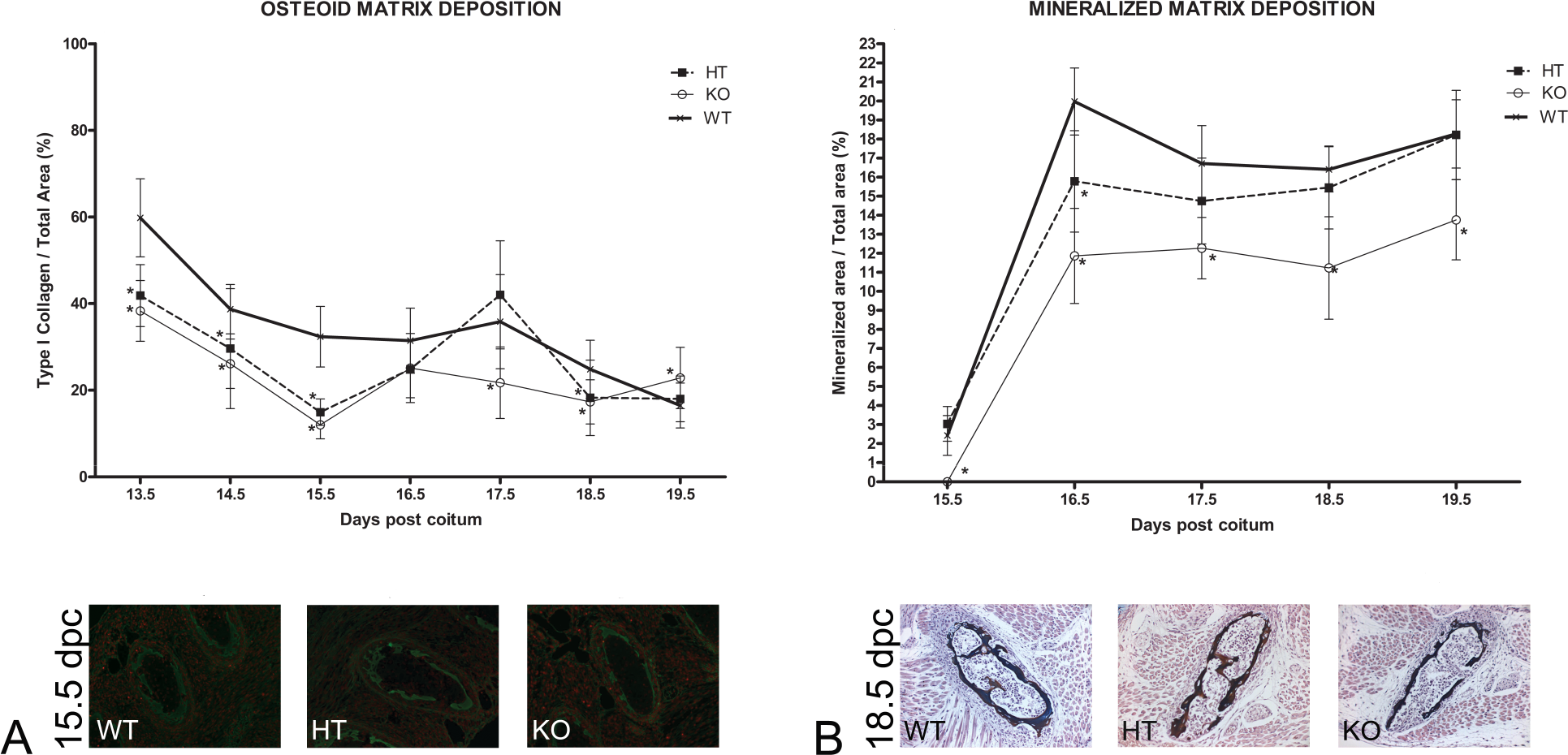
Morphometric characteristics of fetal rib cages at different gestational times (dpc = days post coitum). (A) Deposition of type I collagen (osteoid matrix) in fetal rib cages from 13.5 to 19.5 dpc: immunohistochemical localization of type I collagen revealed that knockout (KO) and heterozygous (HT) fetuses presented smaller depositions of osteoid matrix between 13.5 and 15.5 dpc in comparison with the wild-type (WT) group. (B) Deposition of mineralized matrix in fetal rib cages from 15.5 to 19.5 dpc: immunohistochemical localization of type I collagen demonstrated that in comparison with the WT group, deposition of mineralized matrix in KO fetuses was lower during the entire experimental period, whereas in HT fetuses such deposition was reduced until 17.5 dpc but was comparable with the WT group thereafter. Comparisons involved littermate fetuses (n = 6) from different genotypes. Statistical differences, determined according to ANOVA (p < .05), are indicated by asterisks.
Expression of Bone Development Marker Genes Was Altered in GJA1-Deficient Mice
The molecular mechanisms associated with delayed bone development were investigated by analyzing the expression of appropriate marker genes, the products of which, namely, osteocalcin, osteopontin, alkaline phosphatase, runt-related transcription factor 2 (RUNX2), GJC1, and GJA3, either function as transcription factors or participate directly in the formation of the mineralized structure (Stein et al. 2004; Chung et al. 2006). There were no significant differences in the levels of osteocalcin mRNA between WT, HT, and KO fetuses until 17.5 dpc, but in comparison with the WT group, levels were reduced from 18.5 dpc onward in the KO group and at 19.5 dpc in the HT group (Fig. 4A). Compared with the WT group, levels of osteopontin mRNA were lower in HT and KO fetuses at 16.5 dpc, although the profile changed after 17.5 dpc with the HT and KO groups exhibiting peaks of expression (at 18.5 and 19.5 dpc, respectively) to compensate for the delay in bone development (Fig. 4B). The pattern of expression of the alkaline phosphatase gene was essentially similar to that of osteopontin after 18.5 dpc (Fig. 4C). Significant alterations in the expression of the RUNX2 transcription factor gene were also detected such that in contrast to WT fetuses, RUNX2 mRNA levels showed distinct peaks at 17.5 dpc within the HT group and at 19.5 dpc within the KO group (Fig. 4D).
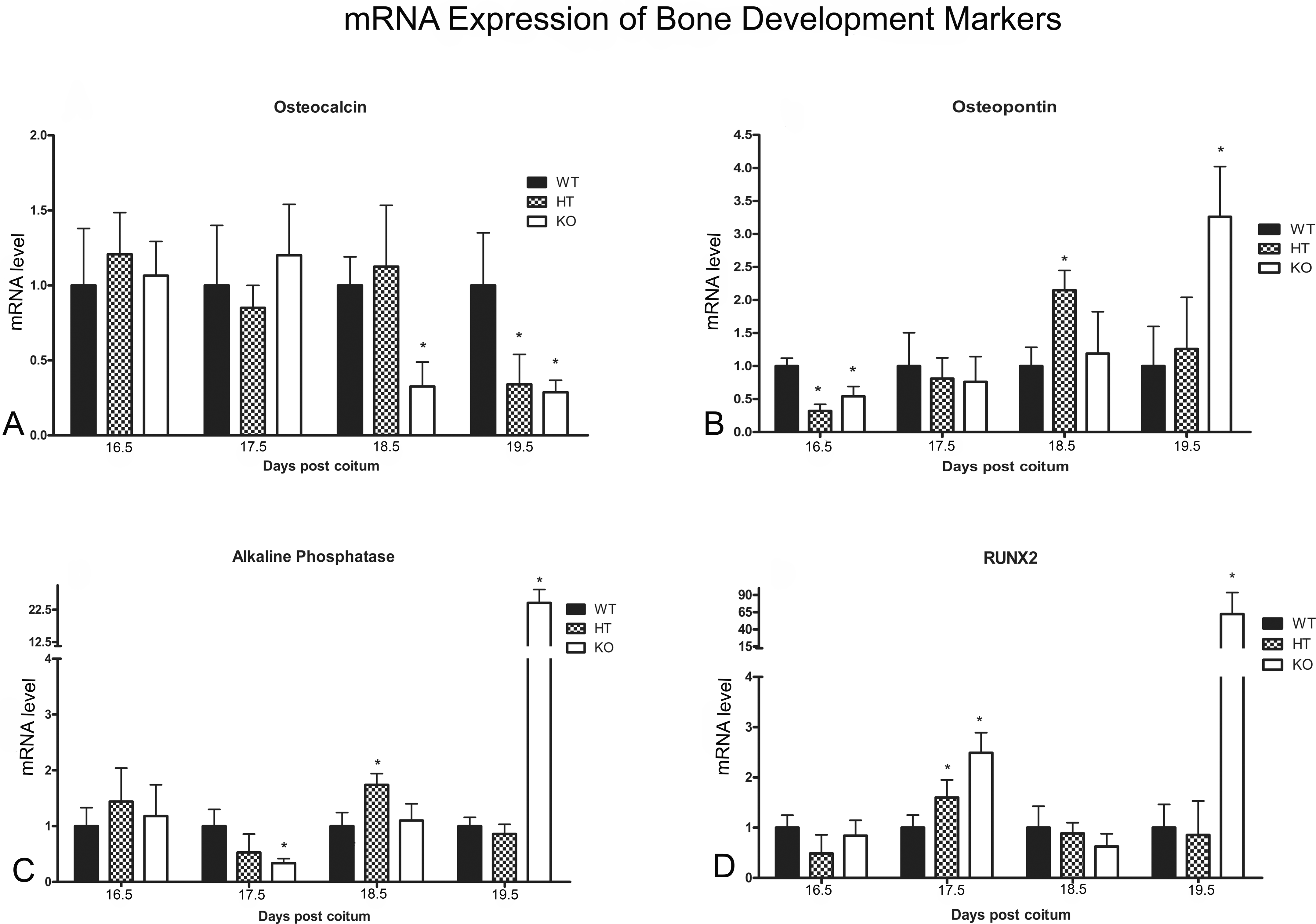
mRNA levels for the marker genes associated with bone development in wild-type (WT) fetuses and heterozygous (HT) and knockout (KO) mutants at different gestational times (dpc = days post coitum) as determined by quantitative real-time polymerase chain reaction (RT-PCR). (A) Alterations in osteocalcin mRNA levels can be observed at 18.5 dpc in HT fetuses and at 18.5 and 19.5 dpc in the KO group. (B) Alterations in osteopontin mRNA levels can be observed at 16.5 and 18.5 dpc in HT fetuses and at 16.5 and 19.5 dpc in the KO group. (C) Alterations in alkaline phosphatase mRNA levels can be observed at 18.5 dpc in HT fetuses and at 17.5 and 19.5 dpc in the KO group. (D) Alterations in the expression of RUNX2 transcription factor gene can be observed at 17.5 dpc in HT fetuses and at 17.5 and 19.5 dpc in the KO group. Comparisons involved littermate fetuses (n = 6) from different genotypes. Statistical differences, determined according to ANOVA (p < .05), are indicated by asterisks.
GJC1 Expression Was Delayed in Cx43-Deficient Mice
Similar to other types of tissue, bone cells contain more than one connexin isoform in their GJICs (Stains and Civitelli 2005), and hence the gene expression and protein profiles of Cx45 (GJC1 gene) and Cx46 (GJA3 gene) were evaluated in GJA1-deficient mice. WT fetuses exhibited a progressive increase in the intensity of immunofluorescence corresponding to Cx45 from 15.5 dpc onward, with a maximum value determined at 17.5 dpc, while GJC1-associated fluorescence in KO fetuses could be with maximum intensity attained at 19.5 dpc (Fig. 5A). The levels of GJC1 mRNA within HT and KO fetuses showed statistically significant increases only at 18.5 and 19.5 dpc, respectively, whereas no significant changes in GJC1 expression were observed within the WT group during the experimental period (Fig. 5B).
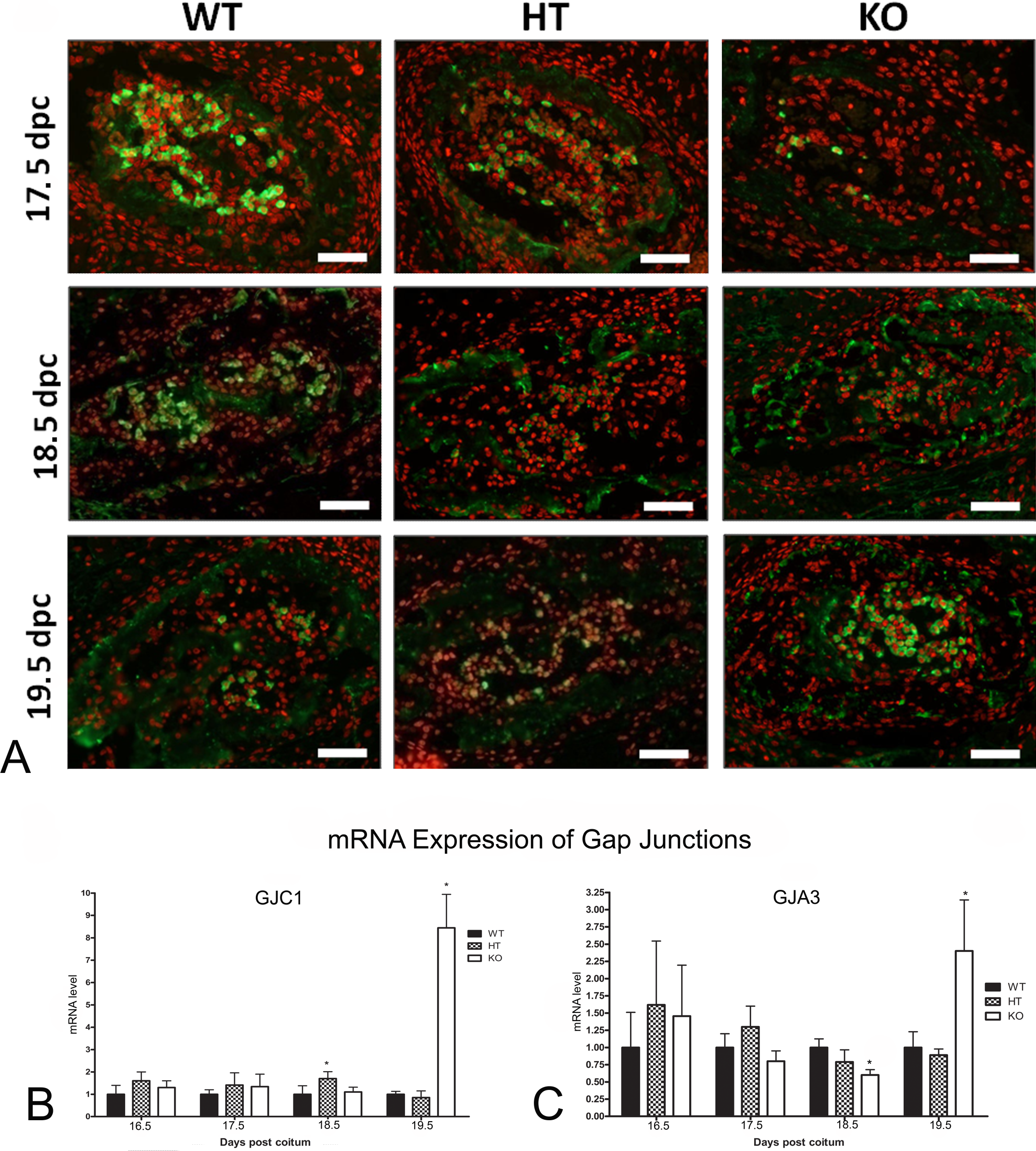
mRNA levels and cytoplasm fluorescence, as determined by real-time polymerase chain reaction (RT-PCR)and immunoassay, respectively, of the connexin isoforms GJC1 and GJA3 in the fetal bone tissue of wild-type (WT) fetuses and heterozygous (HT) and knockout (KO) mutants at different gestational times (dpc = days post coitum). (A) Immunodetection of Cx45 in the ribs of 15.5 to 19.5 dpc fetuses: The absence of GJA1 delayed the transcription of GJC1. (B) The relative levels of GJC1 mRNA were comparable with the results obtained by immunofluorescent analysis of the protein product. (C) The relative levels of Cx46 mRNA demonstrated a delayed expression in KO mice. Bar = 50 μm. Comparisons involved littermate fetuses (n = 6) from different genotypes. Statistical differences, determined according to ANOVA (ρ <.05), are indicated by asterisks.
Cx46 could be immunodetected only in aberrant cell cytoplasm and no significant differences could be established between the three genotypes during the experimental period (data not shown). The level of GJA3 mRNA within KO fetuses was lower than that of the WT group, although in the KO group GJA3 expression peaked at 19.5 dpc (Fig. 5C).
Discussion
The results presented herein demonstrate that fetuses with diminished GJA1 expression exhibit delayed bone development. Although previous studies have revealed that differentiation of GJA1-deficient osteoblasts and osteocytes cultivated in vitro tended to be delayed (Lecanda et al. 1998, 2000; Stains and Civitelli 2005), this investigation provides more evidences of such a delay using histological, morphological, and molecular analyses in in vivo systems, therefore our focus was to analyze bones with cartilaginous template, endochondral ossification. GJA1 is the most abundant connexin isoform in normal bone tissue and its function is to facilitate the passage of ions and secondary messengers (Niessen et al. 2000) that are essential for the transmission of information between cells and, consequently, for their coordinated differentiation (Mesnil 2004). The absence of GJA1 would give rise to a decrease in the permeability of cells that make up bone tissue, resulting, most likely, in the observed delay in cell differentiation.
The results of both histological and histochemical analysis of fetal bone tissue confirmed that animals bearing either partial (HT) or total (KO) deletion of the GJA1 gene exhibited delayed bone differentiation. This delay was perceptible since the very early stages of fetal development (13.5 dpc), during which stage the precursor cells are transformed into specialized cells of the skeleton. Throughout the entire experimental period, KO fetuses were clearly in an earlier stage of differentiation compared with WT fetuses. However, despite the delay in differentiation observed at the initial stages, HT fetuses were fully developed at 17.5 dpc and presented bone characteristics similar to those of their WT counterparts.
Chung et al. (2006) have previously reported the occurrence of osteopenia in adult mice bearing targeted deletions of the GJA1 gene in their bone cells. The present study is in accord with this finding since it demonstrates that the deposition of mineralized matrix in GJA1-deficient fetuses is reduced, and consequently, these animals present a lower bone density compared with the controls. The abnormalities found for us are correlated with the pattern of bone development gene markers. The abnormalities found in our study correlated with the pattern of bone development gene markers. For example, at 16.5 or 18.5 dpc the KO animals have a delay in bone histology and less mRNA levels and gene expression of osteopontin.
The use of bone development markers to determine alterations in growth patterns has been fully reported (Schiller et al. 2001; Stains et al. 2003; Chung et al. 2006). The monitoring regime employed in the present study was designed with the objective of identifying the crucial stages at which delayed osteogenesis could be unambiguously detected. Comparing GJA1-deficient fetuses with the wild type, it was possible to observe significant differences in the expression of the various gene markers at specific stages of fetal development. The key gene in this process is RUNX2, the expression of which not only induces the differentiation of mature osteoblasts into osteocytes (Komori et al. 1997; Stein et al. 2004), but is also responsible for the proliferation of osteoblasts (Pratap et al. 2003) and the control of expression of osteocalcin and osteopontin genes (Komori et al. 1997; Stein et al. 2004). The absence of RUNX2 results in the lack of cell differentiation and mineralization of bone tissue. Apparently, a reduction in GJA1-mediated communication causes problems in the dissemination of the signaling molecules that activate the RUNX2 gene and, consequently, delays development in KO animals.
The findings reported herein are analogous to those obtained from studies of human ODDD, the cause of which is a mutation in the GJA1 gene. Currently, twenty-eight different mutations in this gene are known to affect the phenotype (Vitiello et al. 2005) by producing abnormal channel proteins that drastically reduce the permeability of various cells including osteoblasts and osteocytes (McLachlan et al. 2005). The deficient intercellular communication and delayed bone differentiation presently reported in the KO murine model appears to be similar to that observed in human ODDD, a disease that also results from problems in the differentiation of osteoblasts at the stage of bone development (McLachlan et al. 2008).
Studies involving osteoblasts of the fetal calvaria cultivated in vitro have revealed an inverse relationship between the expression of the GJA1 and GJC1 genes, in that decreased expression of the former corresponded with increased expression of the latter (Lecanda et al. 2000). Although increased expression of GJC1 was not observed in the present in vivo study, a delay in GJC1 gene expression and protein production was detected in the bone tissue. Apparently, GJC1 is associated with the differentiation of osteoblasts and its levels are reduced in mature osteocytes.
The hypothesis that the GJC1 may partially compensate for the absence of the GJA1 gene in bone tissue (Stains and Civitelli 2005) is supported by the fact that in KO fetuses the expression peak of RUNX2 occurred at 19.5 dpc, and this coincided with an increase in GJC1 expression and in the intensity of GJC1 protein in the developing bone. It is possible that GJC1 facilitated widespread diffusion of messengers that were accumulated in some cells, thus inducing large-scale activation of RUNX2 and a consequent increase in expression of osteopontin and alkaline phosphatase genes at that time.
With regard to GJA3, although its expression also increased in KO fetuses at 19.5 dpc, immunohistochemistry revealed that the protein product was located mainly in the cytoplasm. The cytoplasmic localization of GJA3 has already been reported for osteoblasts originating from the calvaria and bone neoplasia (Koval et al. 1997; Sanches et al. 2009). Currently, the function of GJA3 is not well understood, but it may possibly be associated with the protection of cells against hypoxia (Banerjee et al. 2010).
In conclusion, the present study demonstrated that a reduction in GJA1 induces a delay in bone development in mouse fetuses as shown by real-time polymerase chain reaction of the corresponding gene markers. These in vivo results corroborate previous in vitro studies and underline the importance of GJC1 during normal bone development and in compensating for the absence of GJA1.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
Fundação de Amparo a Pesquisa do Estado de São Paulo, FAPESP, supported the first author (Chaible, LM) with a fellowship and also gave financial support to develop the study (Dagli, ML). Authors received no financial support from the pharmaceutical industry.
Acknowledgments
We wish to thank Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support (Grant no. 2007/55274-7), including an MSc scholarship awarded to one of us (LMC) during the studies at the Experimental and Comparative Pathology Program of the School of Veterinary Medicine and Animal Science of the University of São Paulo, Brazil. The sponsoring foundation played no role in the design of study; collection, analysis, and interpretation of data; writing the manuscript; and in submitting the paper for publication.
