Abstract
ST-20 (sodium 2,2-dimethylbutyrate) is a potential therapeutic agent for treatment of β-thalassemia and sickle cell disease. A subchronic oral toxicity study was conducted in Sprague-Dawley rats (10/sex/dose) at gavage dosages of 0 (vehicle control), 200, 600, or 1,000 mg/kg, once daily for up to 15 days followed by a 14-day recovery. Ataxia (females), rough coat/thin appearance (males), and decreased body weights were observed at 1,000 mg/kg. Functional observational battery (FOB) deficits were observed more frequently in females and included decreased body tone, rectal temperature, emotional reactivity, neuromotor-neuromuscular activity (as exhibited by a deficit in visual/tactile placing accuracy, ataxia, hind limb dragging, and decreased grip strength), and rearing. ST-20 caused a decrease in WBC/RBC counts and RBC parameters; increase in reticulocytes and red cell inclusion bodies; decrease in total protein, globulin, and glucose; and increase in AG ratio. Micronucleated polychromatic erythrocytes of the bone marrow increased significantly in males at 1,000 mg/kg. Mean liver and kidney weights increased, and hepatocellular hypertrophy was observed in males at 1,000 mg/kg. Toxicologic findings were fully recovered during the 14-day recovery period. In conclusion, the no-observed adverse effect level for FOB and general toxicity was 200 mg/kg following gavage administration of ST-20 for up to 15 consecutive days.
Introduction
Beta thalassemia is a potentially life-threatening disorder that is characterized by anemia resulting from deficiency of the beta-globin chains of adult hemoglobin (α2β2) due to various mutations (Steinberg and Rodgers 2001; Cunningham et al. 2004; Wonke 2001; Rund and Rachmilewitz 2005; Perrine 2005). The anemia is caused by excess, unbalanced alpha globin chains that are toxic to the red blood cell, leading to apoptosis of developing erythroblasts at the polychromatophilic normoblast stage with intramedullary hemolysis (Rund and Rachmilewitz 2005; Perrine 2005; Schrier 1997; Koury, Sawyer, and Brandt 2002; Centis et al. 2000; Testa 2004; Mathias et al. 2000; Yuan et al. 1993; Pootrakul et al. 2000; Silva et al. 1996). The disease is characterized into two major subtypes based upon severity: beta-thalassemia major and beta-thalassemia intermedia. Patients with beta-thalassemia major have severe anemia with hemoglobin (HGB) less than 6 g/dl that requires regular blood transfusions to sustain life, whereas patients with thalassemia intermedia have milder anemia with a total HGB between 6 to 10 g/dl that may occasionally require blood transfusion (Aessopos, Kati, and Meletis 2007; Cunningham et al. 2004; Wonke 2001; Rund and Rachmilewitz 2005; Perrine 2005). Although thalassemia intermedia is less severe, it is still associated with early mortality and all the complications of thalassemia major, such as iron overload, endocrinopathies, splenomegaly, bone deformities, hypercoagulability, cardiac failure, pulmonary hypertension, and increased risk of infections, including hepatitis C and its complications (Aessopos, Kati, and Meletis 2007; Cunningham et al. 2004; Wonke 2001; Rund and Rachmilewitz 2005). Therapeutic efforts are focused on correcting the underlying globin chain imbalance to reduce the anemia of beta thalassemia by inducing or reactivating fetal globin expression.
To stimulate fetal globin expression, short chain fatty acids (SCFADs) have been used in many experimental therapeutics such as reporter gene assays, transgenic murine, and nonhuman primate models; and in clinical trials (Pace et al. 2002; Steinberg and Rodgers 2001; Perrine 2005; Perrine et al. 1993; Collins et al. 1995; Ikuta et al. 1998; Dover, Brusilow, and Charache 1994; Boosalis et al. 2001; Vadolas et al. 2004; Atweh, Sutton, and Nassif 1999). Sodium phenylbutyrate and arginine butyrate increase total hemoglobin in beta thalassemia patients (Perrine et al. 1993; Collins et al. 1995; Ikuta et al. 1998; Atweh, Sutton, and Nassif 1999; Perrine et al. 2005). However, the use of these agents has been limited by rapid metabolism necessitating intravenous administration of excessively large doses, and by antiproliferative effects on erythroid cell growth, requiring intermittent use (Atweh, Sutton, and Nassif 1999; Perrine et al. 2005). In an effort to identify an orally active SCFAD with more favorable pharmacokinetic (PK) features, ST-20 was discovered after extensive screening of a panel of SCFADs and a chemical library of 13,000 compounds using computational modeling.
ST-20 stimulates in vitro and in vivo fetal globin expression and also induces erythroid cell progenitor survival and proliferation in vitro. ST-20 increases erythroid cell survival through Bcl-family antiapoptotic protein expression (Castaneda et al. 2005). These dual beneficial effects of ST-20, enhanced erythroid survival and fetal globin gene induction, are likely to be of potential benefit in the treatment of beta thalassemia. In addition, ST-20 has also shown a favorable PK profile after oral administration to baboons (Pace et al. 2002) and was chosen for further preclinical development. A toxicity study was conducted for safety evaluation of ST-20 in rat following once-daily gavage administration for up to 15 days.
Materials and Methods
Test Article and Formulation Preparation
The test article, ST-20 (NSC-741804, purity 99.2%), is chemically described as sodium 2,2-dimethylbutyrate. It belongs to a class of SCFADs and derivatives. An appropriate amount of ST-20 (manufactured by Frontage Laboratories, Malvern, PA) was dissolved in 80% of the final volume with vehicle sterile water by stirring and/or sonicating until a solution was formed. The solution was then adjusted to a pH of 7.0 ± 0.5 using HCl and/or NaOH and then diluted with sterile water to final volume. Aliquots of each target concentration were analyzed before using the formulations for dosing the animals via oral gavage. All dose formulations were within ±10% of the target concentrations.
Animals
Sprague-Dawley rats (male 200–260 g and female 160–220 g) from Charles River Laboratories Portage, MI, were individually housed in polycarbonate cages during the quarantine and study periods. The rats were provided with Harlan Teklad Global Diet (Harlan 2018C) daily. Water from the Columbus, Ohio municipal system was used without further treatment and supplied, ad libitum, via an automatic watering system. Animal care, housing, and environmental conditions were according to AAALAC recommendations, requirements stated in the Guide for Care and Use of Laboratory Animals and stated by the U.S. Department of Agriculture through the Animal Welfare Act requirements.
Experimental Design
Forty male and 40 female rats (10/sex/group) were assigned to three dose groups and a vehicle control group (sterile water) as shown in Table 1 . The study was a stagger start with the males dosed 4 days before the females at constant volume (5 ml/kg). The doses were selected to include and exceed the proposed therapeutic dose range in human (< 25 mg/kg). The genotoxicity group (5 rats/sex/group) received 14 days of treatment and was necropsied on day 15 for micronuclei sample collection, clinical pathology, and histopathology. The neurologic pharmacology group (5 rats/sex/group) received an additional dose on day 15 in order to obtain FOB measurement approximately 2 hr post dose and was sacrificed on day 29. The neurologic pharmacology group was used for FOBs and clinical pathology on day 15 and also served as the recovery group with a 14-day recovery period (day 29 necropsy).
Study design.

a Dose volume was 5 mL/kg.
b Neurologic Pharmacology Group was dosed daily for 15 days and used for FOBs/clinical pathology on day 15 and served as a recovery group for clinical pathology/necropsy/histopathology data on day 29.
c Genotoxicity Group was dosed daily for 14 days and used for micronuclei sample collection/clinical pathology/necropsy/histopathology on day 15.
Parameters Evaluated
Clinical Observations
Observations for morbidity and mortality were made twice daily throughout the study. Pretest clinical observations were recorded once on day –3. Starting on day 1, clinical observations were conducted at least twice on each day of dosing (predose and postdose) and at least once daily thereafter.
Body Weights
Body weights were recorded on day –3 for each respective stagger start for the purpose of randomization. Body weights were recorded for all animals on days 1, 4, 8, 11, 15, 18, 22, 25, and 29. All body weights collected on dosing days were collected prior to dosing and used for dose volume determinations.
Functional Observational Battery (FOB)
Functional observational battery data were collected approximately 2 hours (approximate Cmax) after the 15th dose using a series of 40 tests. FOB tests included home cage observations (posture, tremor activity, convulsive activity, lethargy/arousal, eyelid closure), removal (ease of removing, ease of handling, hand to hand transfer, general condition/appearance, urine stain, fecal stain, salivation, piloerection, fur appearance, vocalization, lacrimation, palpebral reflex responsivity, papillary diameter test, pupil response), open field observations (lines crossed, rearings, urine pools, fecal boluses, posture, tremor activity, convulsive activity, gait, gait score, stereotype, bizarre behavior, spontaneous vocalization), and reflexes (approach response, touch response, startle response, tail pinch response, placing of paws on grid, grip strength, righting reflex air, righting reflex surface and body temperature).
Each animal was examined by two trained technicians (blinded and nonblinded to the treatment) in the following order: the technician nonblinded to treatment dosed the animals, observed the animal in its home cage, and observed the animal during removal and handling. The technician blinded to treatment observed the animal in an open field box, tested the animal’s reflexes, and took the animal’s body temperature.
Clinical Pathology
Clinical pathology samples were collected and analyzed by a board-certified pathologist (M.J.R.) at the following time points: on day 15 from the genotoxicity group animals (5/sex/group) and on days 15 (after the FOB) and 29 from the neurologic pharmacology group animals (5/sex/group). Hematology analysis included white blood cell (WBC)/red blood cell (RBC) counts, hemoglobin (HGB), hematocrit (HCT), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), platelets (PLT), differential leukocyte counts (neutrophils, monocytes, eosinophils, basophils, lymphocytes), reticulocytes (RET), and nucleated RBCs (nRBC). Clinical chemistry analysis included alkaline phosphatase (ALP), aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyl transferase (GGT), total protein (TP), albumin (ALB), glucose (GLU), blood urea nitrogen (BUN), creatine (CREA), phosphates (PO4), creatine kinase (CK), lactose dehydrogenase (LDH), sodium (NA), potassium (K), chloride (CL), globulin (GLOB), albumin/globulin ratio (AGR), total bilirubin (TBILI), direct bilirubin (DBILI), and indirect bilirubin (IBILI). Coagulation parameters included prothrombin time (PT), activated partial thromboplastin time (APTT), and fibrinogen (FIB).
Micronucleus (MN) Analysis
The micronucleus analysis was performed by Integrated Laboratory Systems, Inc. (Durham, NC), to evaluate the genotoxic potential of ST-20. On day 15, bone marrow samples were collected from the femur of the genotoxicity group animals (5 rats/sex/group) between 18 and 24 hours post–day 14 dosing. The slides were prepared, fixed, and shipped by overnight mail to ILS, Inc. The slides were stained with acridine orange and then rinsed with sodium phosphate buffer, air-dried, and scored by two scorers blinded to the identity of the slides. Acridine orange is a fluorochrome that differentially stains DNA/RNA-containing micronuclei from other basophilic granules. The frequency of micronucleated polychromatic erythrocytes (PCEs) was evaluated in 2,000 PCEs per animal. The number of PCEs per 1,000 bone marrow erythrocytes per animal was also determined to evaluate effects of ST-20 on bone marrow erythropoiesis.
Necropsy and Microscopic Pathology
The genotoxicity group was euthanized on day 15, and the neurologic pharmacology group was euthanized on day 29. The rats were fasted overnight prior to necropsy. The following tissues were collected for histopathology examination: adrenal glands, aorta, bone, femur, bone marrow (sternum, costochondral junction), brain, cecum, colon, duodenum, epididymides, esophagus, eyes, heart, ileum, jejunum, kidneys, liver, lungs, lymph nodes (mandibular, and mesenteric), mammary gland, ovaries, pancreas, parathyroid gland, pituitary gland, prostate gland, salivary gland (mandibular), sciatic nerve, seminal vesicle, skeletal muscle (thigh), skin (ventral abdomen), spinal cord (thoracolumbar), spleen, stomach (forestomach, glandular), testes, thymus, thyroid glands, trachea, urinary bladder, and uterus.
The tissues were examined, sampled, and fixed in 10% neutral-buffered formalin, processed to approximately 5–micron sections for hematoxylin and eosin staining, and examined microscopically by a board-certified pathologist (D.Y.V.). All tissues were examined from all rats necropsied on day 15. Potential targets (liver and spleen) based on histopathological examination of the tissues obtained from day 15 necropsy were examined from rats necropsied on day 29 (recovery animals). Microscopic findings were graded based on severity from minimal (grade 1) to marked (grade 4).
Organ Weights
The following organs were collected for weight measurements: adrenal glands, brain, epididymides, heart, kidneys, liver, lungs, ovaries, spleen, testes, thymus, thyroid, and uterus.
Data Analysis
FOB Assessment/Statistical Analysis
FOB data summarization and statistical analyses were conducted independently for each parameter and for males and females. The following statistical analysis approaches were used in this study to analyze FOB data, with the specific method being dependent on the type of variable.
Categorical Variables (Score, Yes/No)
For categorical variables, a mean response model was used to predict the likelihood of observing a particular pattern of responses among the possible response categories, as a function of dose group. This model was fitted to categorical data using the CATMOD procedure in the SAS system (SAS Institute, 2004). A chi-square test was used to test the significance of the dose group effect. When this effect was significant at a .05 level, additional chi-square tests were performed within the model fit to compare each of the three test article dose groups to vehicle. Each comparison to vehicle was done at a .05/3 = .0167 significance level, to control the overall error rate across all three comparisons to no more than .05.
The frequencies of responses within each category and each dose group were tabulated. The p-values were reported from the chi-square tests for dose group effects and comparisons to vehicle.
Continuous Variables
For continuous variables, a one-way analysis of variance (ANOVA) model was fitted to the observed responses using the MIXED procedure in the SAS system. The model included dose group as a fixed effect and assumed the residual error (i.e., difference in observed and model-predicted values) followed a normal distribution with zero mean. An F-test was used to test the significance of the dose group effect. When this effect was significant at a .05 level, additional t-tests were performed within the model fit to compare each of the three test article dose groups to vehicle. Each comparison to vehicle was done at a .05/3 = .0167 significance level, to control the overall error rate across all three comparisons to no more than .05.
Results
Clinical Observations and Body Weights
ST-20 treatment did not cause mortality in any dose group. Animals dosed at 200 or 600 mg/kg exhibited no adverse clinical signs that could be considered test-article-related. Females dosed at 1,000 mg/kg exhibited ataxia (incoordination) in 4 of 10 females (since all animals were still on study) on days 6 through 8 and, for males, rough coat and thin appearance in 2 of 5 males (since 5 animals were euthanized on day 15) on days 16 or 18. The animals dosed at 1,000 mg/kg had significantly decreased mean body weights on day 15 (10% in males and 8% in females as compared to vehicle control animals). There were no drug-related clinical signs or no significant differences in mean body weight observed during the 14-day recovery period.
FOB Data
Of the 40 tests, parameters that showed statistical differences are shown in Table 2 . Both male and female rats treated with the 600 or 1,000 mg/kg dose of ST-20 exhibited diminished body tone. Both male (1,000 mg/kg dose group) and female (600 and 1,000 mg/kg dose groups) rats exhibited decreased rectal temperatures.
Significantly affected FOB parameters.
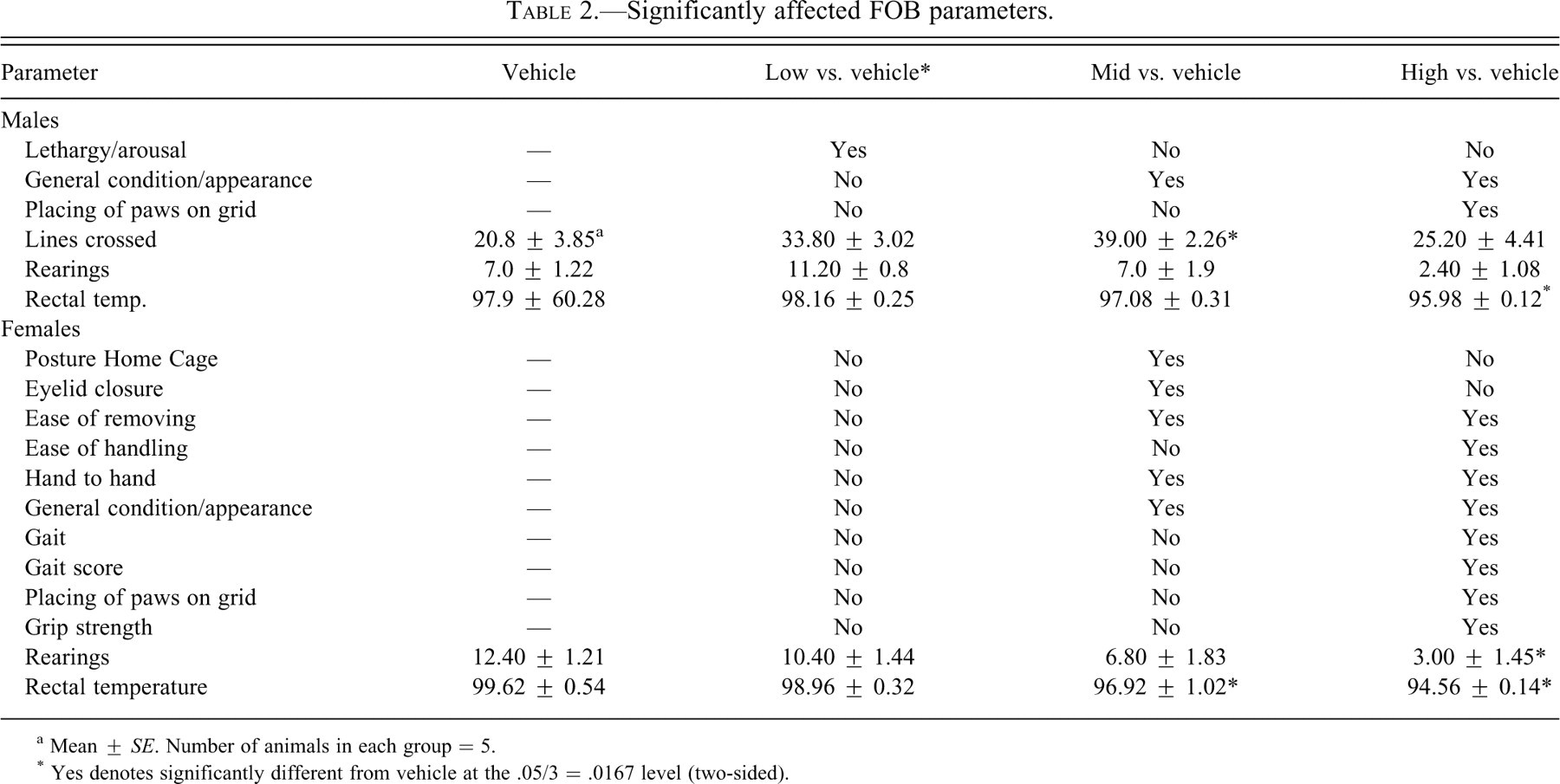
a Mean ± SE. Number of animals in each group = 5.
* Yes denotes significantly different from vehicle at the .05/3 = .0167 level (two-sided).
Female rats treated with the 600 or 1,000 mg/kg dose of ST-20 exhibited reductions in emotional reactivity, as indicated by three FOB handling tests (ease of removing test, ease of handling test, and hand-to-hand transfer test).
There was evidence of neuromotor/neuromuscular system dysfunction in both male and female rats, although the effects were more severe in females. Male rats in the 1,000 mg/kg dose group exhibited a deficit in visual/tactile placing accuracy. Two of the 5 male rats in the 1,000 mg/kg dose group exhibited either slight ataxia or dragging of the hind limbs in the open field. Female rats exposed to the 1,000 mg/kg dose exhibited significant decrease in grip strength.
Males treated with the 200 mg/kg dose of ST-20 exhibited a statistically significant increase in arousal in the home cage, while males treated with the 600 mg/kg dose demonstrated significantly increased locomotor activity (lines crossed) in the open field compared to the vehicle group. These effects were not dose-dependent; however, a dose-dependent decrease in number of rearings was observed in both sexes.
Clinical Pathology
Coagulation
None of the coagulation results indicated any effects due to ST-20 administration.
Hematology
On day 15, decreased RBC, HGB, HCT, MCH, and MCHC were observed in the 1,000 mg/kg dose group (male) or in the 600 and 1,000 mg/kg dose groups (female) given ST-20 (Table 3 ). The differences were statistically significant for most of these parameters in the 1,000 mg/kg dose group rats of both sexes and the 600 mg/kg dose group females.
Summary of significant changes in hematology and clinical chemistry values, day 15.
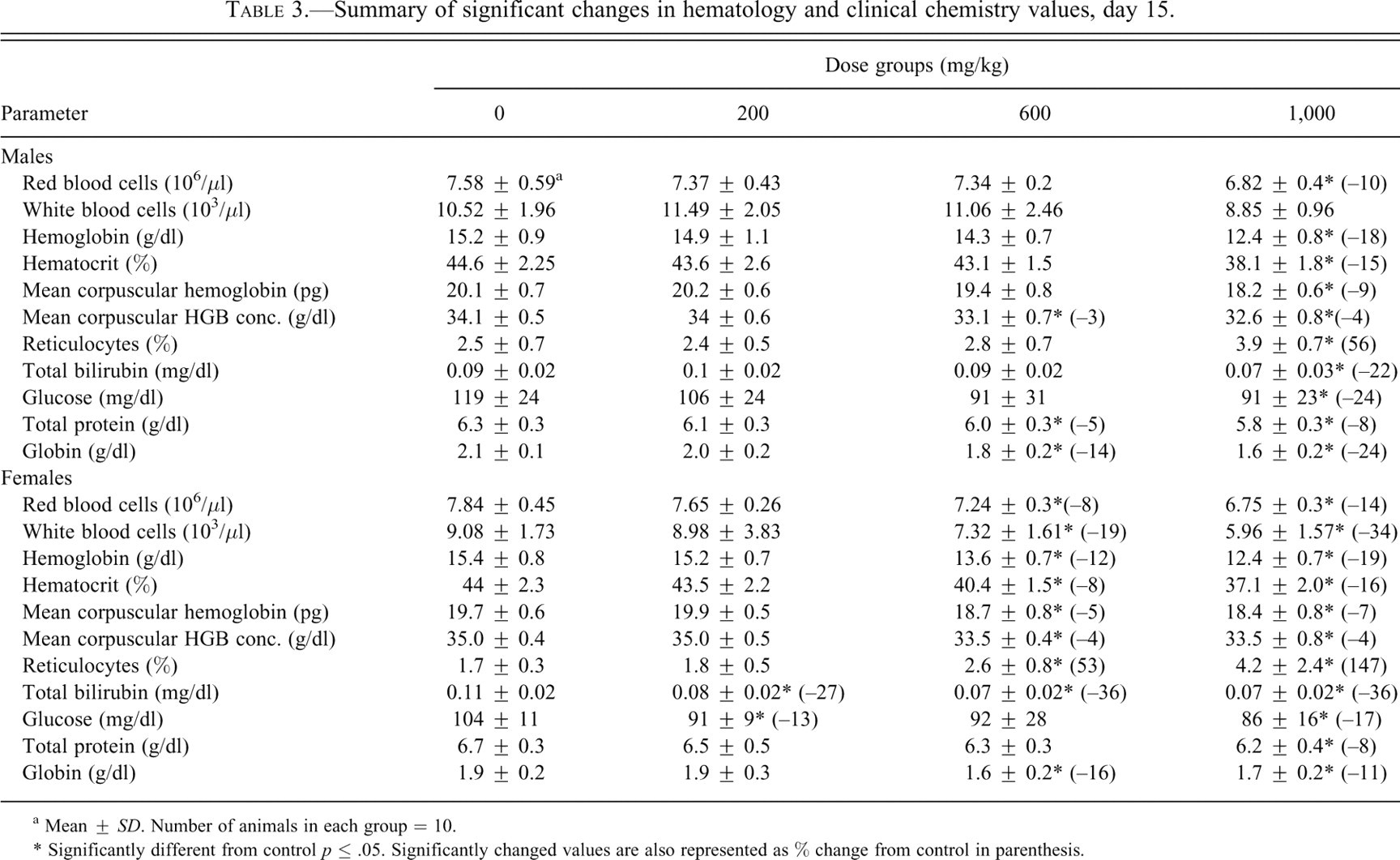
a Mean ± SD. Number of animals in each group = 10.
* Significantly different from control p ≤ .05. Significantly changed values are also represented as % change from control in parenthesis.
The increased RET counts observed in the 1,000 mg/kg dose group rats of both sexes and the 600 mg/kg dose group females were statistically significant (Table 3).
Also on day 15, a morphologic change in RBCs (inclusion bodies that are interpreted to be Pappenheimer Bodies) was noted microscopically in 6 of 10 males and in 10 of 10 females from the 600 mg/kg dose group, and in 10 of 10 males and females at 1,000 mg/kg (Figure 1 ). The severity observed of intracytoplasmic body involvement increased with dose and was more severe in females.
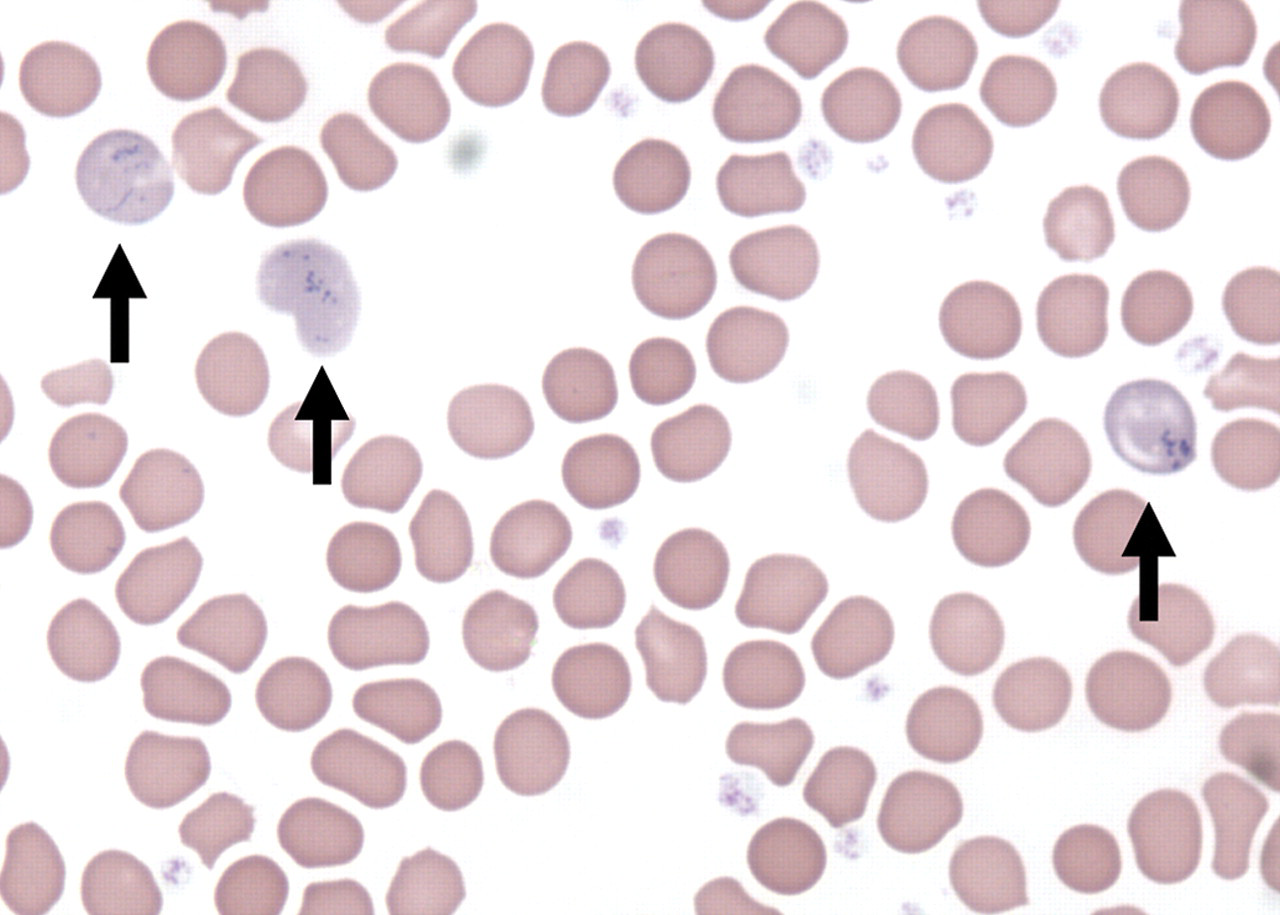
Light micrograph of blood smear obtained on day 15 from a female rat treated with 1,000 mg/kg ST-20 for 14 days. Arrows indicate red cell inclusion bodies interpreted to be Pappenheimer bodies. Giemsa stain, 1,000X magnification.
Other hematologic changes found on day 15 were noted in PLT and WBC counts. Changes in PLT counts were small in magnitude with no morphological changes. WBC counts decreased in the 600 mg/kg dose group females and 1,000 mg/kg dose group rats of both sexes; the decreases in 600 and 1,000 mg/kg dose group females were statistically significant. The total WBC changes consisted of changes in differential counts: decreased neutrophils, total lymphocytes, monocytes, and basophils.
At the end of the 14-day recovery period (day 29), no hematologic changes were present in treated rats indicating reversible changes.
Clinical Chemistry
On day 15, decreased TP and GLOB were noted in the 600 and 1,000 mg/kg dose group rats of both sexes (Table 3). The TP decreases were statistically significant in the 600 mg/kg dose group males and the 1,000 mg/kg dose group males and females. The GLOB decreases were statistically significant in both sexes at both dosages. These decreases in the 600 and 1,000 mg/kg dose group rats of both sexes were accompanied by increased albumin/globulin ratios (AGR), because albumin remained nearly constant in these groups as globulin decreased.
Decreased serum GLU was noted in the treated groups; the decreases were statistically significant in females from 200 mg/kg dose group and 1,000 mg/kg dose group rats of both sexes (Table 3). These decreases in GLU levels were small in magnitude and low in dose dependency.
At the end of thr 14-day recovery period (day 29), there were no significant changes observed in the clinical chemistry parameters indicating reversible changes.
Micronucleus Analysis
ST-20 treatment did not result in higher frequency of micronucleated PCEs at any dose level in females. However, in males, a significant increase in the frequency of micronucleated PCEs was observed at the 1,000 mg/kg dose (mean: 0.33 ± 0.16 MN PCEs/100PCEs) compared with the vehicles (mean: 0.14 ± 0.13 MN PCEs/100PCEs) based on a one-tailed Fisher’s exact test. A linear regression test indicated a dose-response trend only in the males.
ST-20 did not significantly change the percent PCEs, indicating it did not alter bone marrow erythropoiesis at the doses tested.
Organ Weights
At the day 15 sacrifice, there were dose-dependent increases in kidney and liver weights in both sexes. Significant increases in absolute (16%) and relative (26%) liver weights were seen in 1,000 mg/kg dose group females. In males, only relative (22%) liver weight was significantly increased at 1,000 mg/kg dose. Significant increase in absolute (12%) kidney weight was seen in 1,000 mg/kg group males, whereas relative kidney weight was increased in both males (24%) and females (18%). Relative organ weights were affected more in magnitude, mainly because of decreases in body weights. Organ weights at 200 and 600 mg/kg dose levels remained unaffected.
At the end of the 14-day recovery period (day 29), there were no statistically significant differences noticed in organ weights, indicating reversible changes.
Histopathology
Microscopic changes of minimal severity were observed in the liver and spleen. Centrilobular hepatocellular hypertrophy was observed in 3 of 5 males from the 1,000 mg/kg dose group at day 15 (Figure 2 ). Splenic hematopoietic cell proliferation was present in females that received 600 mg/kg and 1,000 mg/kg doses at greater incidence than in control females (Figure 3 ).
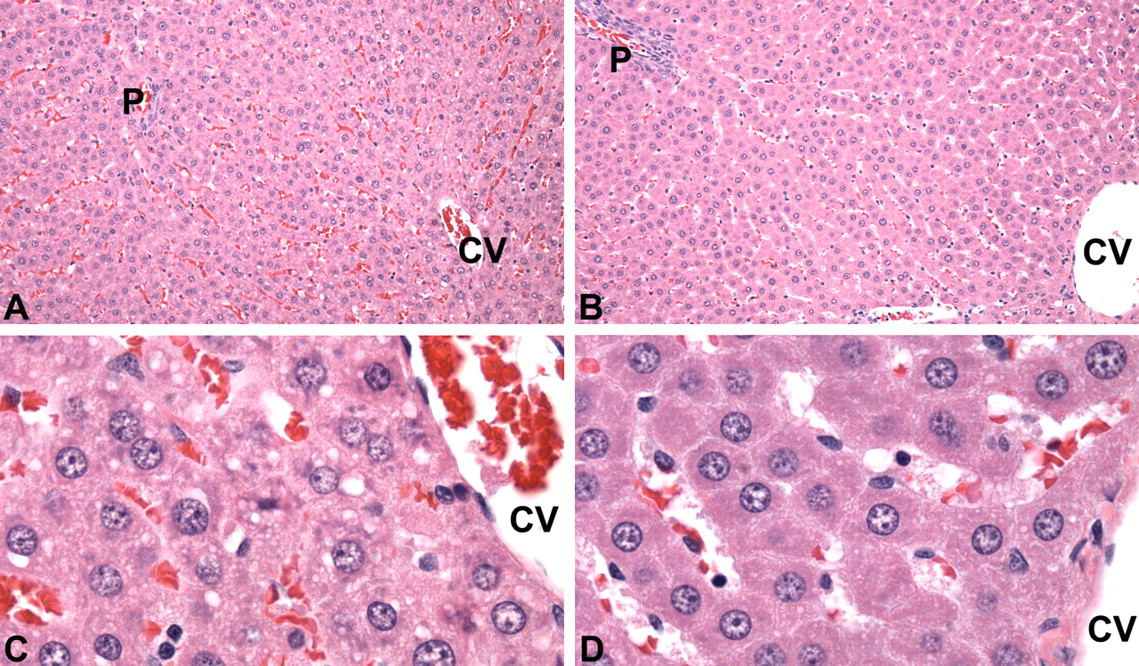
Light micrographs of liver sections obtained on day 15, H&E stain. (A) Liver of a vehicle control male rat. Central vein marked “CV,” portal triad marked “P,” 20X magnification. (B) Liver of a male rat treated with 1,000 mg/kg ST-20 for 14 days. Hepatocytes are minimally enlarged. Central vein marked “CV,” portal triad marked “P,” 20X magnification. (C) Vehicle control male rat liver illustrated in A. Central vein marked “CV,” 100X magnification. (D) Male rat liver from 1,000 mg/kg dose group illustrated in B. Central vein marked “CV.” Centrilobular hepatocytes are minimally enlarged. 100X magnification.
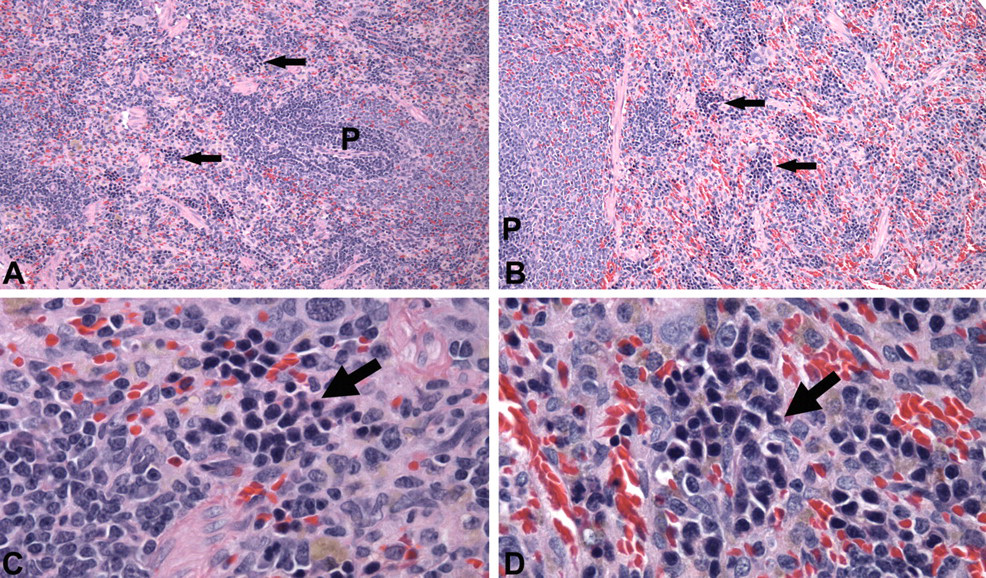
Light micrographs of spleen sections obtained on day 15, H&E stain. (A) Spleen of a vehicle control female rat with the greatest amount of extramedullary hematopoiesis within the control group. Arrows indicate islands of hematopoietic cells. “P” marks periarteriolar lymphoid sheaths. 20X magnification. (B) Spleen typical of all female rats treated with 1,000 mg/kg ST-20 for 14 days. Arrows indicate islands of hematopoietic cells that are minimally larger than in the illustrated control spleen. “P” marks periarteriolar lymphoid sheaths. 20X magnification (C) Vehicle control female rat spleen illustrated in A. Arrow indicates an island of hematopoietic cells (erythroid series). 100X magnification. (D) Female rat spleen from 1,000 mg/kg dose group illustrated in B. Arrow indicates an island of hematopoietic cells (erythroid series). 100X magnification.
Hepatic or splenic changes were not observed in any rats from recovery group (day 29).
Discussion
ST-20 (sodium 2,2 dimethylbutyrate) is a potential therapeutic agent for treatment of β–thalassemia and sickle cell disease at proposed human doses of ≤ 25 mg/kg once per day. Butyrate stimulates production of fetal hemoglobin through induction of the fetal (γ) globin gene promoter, which thereby reduces globin chain imbalance in beta thalassemia, which in turn reduces anemia, or inhibition of polymerization of sickle hemoglobin; both effects ameliorate the clinical courses of the respective disease conditions (Steinberg and Rodgers 2001; Perrine 2005). Butyric acid is reported to cause toxicity that included decreased alertness, ataxia, hypokalemia, hematologic changes (reduced reticulocytes, WBC, HCT, and PLT counts), neuropathologic changes (cystic necrosis in the dentate nuclei, demyelination in the globus pallidus), and multiorgan system failure when infused in excess of 4 g/kg/day in primates (Blau et al. 1993). In this study, we did not observe these changes except ataxia and reduced WBC/HCT; this difference may be accounted for by lower doses (≤1,000 mg/kg), formulation, oral route of administration, species, and butyric acid analog.
ST-20-related clinical observations were limited to animals dosed at 1,000 mg/kg, which exhibited ataxia (females), rough coat and thin appearance (males), and decreased body weights. ST-20 induced a larger number of FOB deficits in females including decreased body tone, rectal temperature, emotional reactivity, neuromotor-neuromuscular activity, and rearing. The observed deficits could be the result of secondary effect (anemia) caused by the test article and/or due to a direct effect of the test article on the nervous system. There were no correlative neuropathological findings observed in any dose group in both males and females.
SCFA derivatives have been shown to increase HGB, HCT, and RBCs in nonhuman primates at doses up to 500 mg/kg (Pace et al. 2002). However, in this study, ST-20 at higher doses decreased RBC, HGB, HCT, MCV, MCH, and MCHC with concomitant increases in reticulocytes and red cell inclusion bodies. The increased reticulocyte counts were interpreted to be a physiologic compensatory response to the reduction in RBCs. Dose-dependent increases in the red cell inclusion bodies that are interpreted to be Pappenheimer Bodies are indicative of abnormal red cell production, which may have decreased the lifespan of erythrocytes to cause anemia. Histopathologic examination of the bone marrow indicated no remarkable changes in bone marrow cellularity and/or myeloid-to-erythroid ratios that could help explain the observed changes in the hematologic parameters. Thus, the underlying mechanism for observed hematologic effects at the doses tested is not completely understood.
Fifteen days of daily ST-20 treatment did not alter biomarkers of liver injury (ALT, AST, ALP, GGT), indicating the test article does not induce liver damage at the doses and the duration of treatment tested. However, ST-20 decreased TP and GLOB with increased AGR in the 600 and 1,000 mg/kg rats of both sexes. SCFAs have been shown to cause potassium and protein loss and inhibit endogenous respiration in fungal cell cultures, which is thought to be due to perturbation of cell membrane selective permeability (Teh 1974). We did not see hypokalemia, an SCFA class effect, in rats, which may be accounted for by differences in dose, route, or species. A small non-dose-dependent decrease in GLU of unknown mechanism was noted in all dose groups in both sexes. Lack of GLU dose response can be explained by a compensatory mechanism of the liver. The changes in GLU, TP, GLOB, and AGR may simply be due to reduced food consumption (food consumption was not recorded in this study) and/or changes in cell membrane selective permeability as indicated by in vitro study (Teh 1974).
Bone marrow micronucleus analysis was conducted to fulfill regulatory requirements for genetic toxicity testing (International Conference on Harmonization 1997). Micronuclei are nuclear leftovers of a chromosome fragment and/or a whole chromosome formed during mitosis that does not pass on to daughter nuclei. ST-20 significantly induced micronuclei in male rats gavaged with a daily dose of 1,000 mg/kg for 14 days, but for females, no increase in the frequency of micronucleated PCEs was observed at any dose level. Although the mean increase (mean: 3.3 ± 1.6 MN PCEs/1,000PCEs) was statistically significant, it is not considered biologically relevant, since the increase was observed only in the high-dose males and is within the range of the laboratory’s historical negative control values (range: 0 to 3.5 MN PCEs/1,000PCEs); and the mean increase is similar to the female vehicle value (mean: 3.0 ± 1.2 MN PCEs/1,000PCEs) and is far below the range of laboratory’s historical positive controls (range: 9 to 27 MN PCEs/1,000PCEs, mean ± SD: 19.2 ± 5.5 MN PCEs/1,000PCEs). In addition, females appeared to be more severely affected by the test article and yet showed no effect on micronucleated PCEs.
Centrilobular hepatocellular hypertrophy of minimal severity was observed in 1,000 mg/kg dose group males, and liver weights were also significantly increased in the 1,000 mg/kg groups (both sexes). In females, splenic hematopoietic cell proliferation was noted in the 600 mg/kg and 1,000 mg/kg dose groups. Splenic changes were considered to be secondary to the test-article-induced anemia. Microscopically, the bone marrow appeared normal; in addition, ST-20 did not alter bone marrow erythropoiesis based on the percent PCEs in the bone marrow. It is possible that increases in peripheral blood reticulocytes could be due to extramedullary (such as splenic) hematopoiesis in response to demand for increased erythrocytes observed with ST-20 toxicity. Similar findings of increased peripheral blood reticulocytes associated with splenic hematopoietic cell proliferation and no changes in bone marrow have also been reported with a few other chemicals, for example, gallic acid, trinitrotoluene, and aniline (Niho et al. 2001; Levine et al. 1984; Pauluhn 2004). In rat, aniline-induced extramedullary hematopoiesis in the spleen at lower doses than the doses required to induce bone marrow hematopoiesis indicating spleen erythropoiesis could be more responsive than the bone marrow (Pauluhn 2004). In addition, it has been shown in rats that hypoxia is associated with extramedullary hematopoiesis in spleen (Stutte et al. 1986). Moreover, in rat, erythropoietic stimuli can induce 5 to 6 additional cell divisions in spleen but only 2 to 3 additional divisions in the bone marrow, resulting in significant increase in the spleen’s contribution to erythropoiesis (Pantel et al. 1990).
In conclusion, the study indicates that the hematologic system is the most sensitive target organ system for ST-20 toxicity. Toxicity produced by 15-day oral administration of ST-20 is reversible upon a 14-day recovery period. The no-observed adverse effect level (NOAEL) for FOB and general toxicity was 200 mg/kg (a human equivalent dose of approximately 32 mg/kg) following gavage administration of ST-20 for up to 15 consecutive days.
Footnotes
Acknowledgments
The authors acknowledge ILS, Inc., Durham, North Carolina, for conducting the micronucleus analysis.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article. The authors declared the following source of funding: Supported by NCI contract N01-CM-42200; NIH grants R01 DK-52962, R42 HL-62715, and NHLBI; and NIDDK under NIH-RAID Pilot Program.
