Abstract
A seven-year-old female cynomolgus monkey had a mass in the left ovary with metastasis to the lung and the right ovary. The mass of these organs showed three different characteristics, and its immunohistochemical profiles were consistent with embryonal carcinoma (EC), choriocarcinoma (CC), and epithelioid trophoblastic tumor (ETT). The EC was characterized with sheets and glandlike structures with large pleomorphic, single-nucleated epithelial cells that were immunohistochemically positive for α-fetoprotein, octamer-4, and CD30, and with multinucleated giant cells resembling syncytiotrophoblasts. The CC also represented biphasic proliferation of the cytotrophoblast positive for cytokeratin 7 (CK7), which showed negative immunoreactivity for all three of the above antibodies, and it was syncytiotrophoblast positive for human chorionic gonadotropin. The ETT showed numerous floating cells in an abundant eosinophilic extracellular matrix with vacuolated or eosinophilic cytoplasm and was immunohistochemically positive for CK7, p63, and α-inhibin, which features nodule or cordlike structures. Collectively, this neoplasm was identified as a mixed germ cell tumor with EC, CC, and ETT. To our knowledge, this is the first report of EC in nonhuman primates as a component of mixed germ cell tumor.
Keywords
Introduction
To date, several ovarian germ cell tumors have been reported in nonhuman primates. There is, however, little information regarding germ cell tumors consisting of several different elements. The term mixed germ cell tumor (MGCT) is commonly used in human pathology and is defined as germ cell tumors that include at least two different germ cell elements. On the other hand, in laboratory or domestic animal pathology, the term is not commonly used because the number of reported germ cell tumors including several elements is low and the definition of MGCT has not been clarified (Tsubota et al. 2004). In animal pathology, embryonal carcinoma (EC) is described as a tumor composed of embryonic multipotential cells that are capable of further differentiation. There are no reports of identified EC in nonhuman primates.
Here, we encountered an ovarian tumor that corresponded to an MGCT in a cynomolgus monkey. This tumor was also identified as having EC as a component, and it showed metastasis in multiple organs.
Case Report
All procedures were performed under the Rules for Animal Experiments approved by the institutional Committee of Animal Experiments. A seven-year-old female cynomolgus monkey (Macaca fascicularis) from China, acquired from HAMRI CO., LTD (Tsukuba, Japan), was brought into our laboratory for drug efficacy and/or toxicity study. Clinically, the animal developed a cough, which lasted for three months. As the cough became severe with reduction of body weight, the animal was euthanized by exsanguination under ketamine anesthesia and then necropsied. The monkey had never been pregnant.
Organs with macroscopic findings (the ovaries and the lung) were collected. They were trimmed and fixed in 10% neutral buffered formalin. Sections obtained from paraffin-embedded tissue were stained with hematoxylin and eosin (HE). Additionally, representative sections of the left ovary and the lung were stained with periodic acid–Schiff (PAS), Massons' Trichrome (MT), Alcian Blue (AB), and immunohistochemistry (IHC). Immunohistochemistry was performed using a two-step peroxidase 3,3′-diaminobenzidine staining technique with a DAKO Envision+ Kit (DAKO Japan, Tokyo, Japan) according to the manufacturer’s instructions. Staining was performed for anti-human α-fetoprotein (AFP), anti-human cytokeratin 7 (CK7), anti-human octamer 4 (OCT4), anti-human CD30, anti-human human chorionic gonadotropin (hCG), anti-human human placental lactogen (hPL), anti-human p63, and anti-human α-inhibin (α-inh). Other information including dilutions is privided in Table 1 . Except for the sections for p63, the sections were counterstained with hematoxylin to visualize nuclear stainability of neoplastic cells.
Antibodies used for immunohistochemistry.

Macroscopically, there was a grayish-white, 3 × 4 × 2 cm mass in the left ovary (Figure 1A ). The section of the left ovary was grayish- or pinkish-white with scattered dark brown foci (Figure 1B). On the other hand, wine-colored hematoid substance was observed in the section of the right ovary, and an obvious mass had not formed. The lung exhibited multiple masses in every lobule, and the surface of the masses showed many scattered grayish-white nodules.
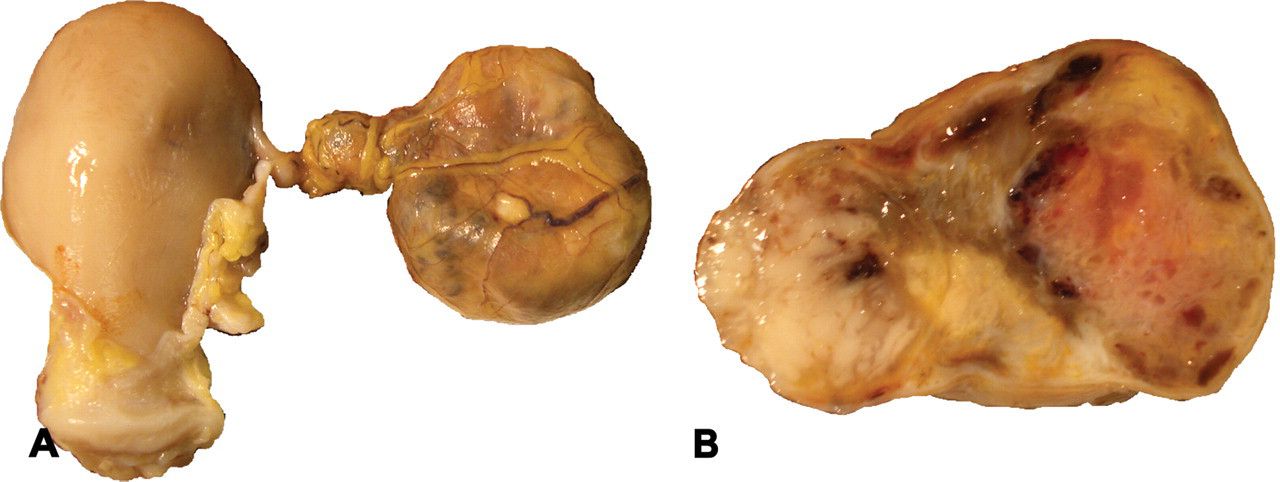
Macroscopic appearance of the tumor. The left ovary neighboring to the uterus (A). Section of the left ovary (B).
Microscopically, neoplastic lesions replaced almost all of the left ovary and the lung. The central portion of each mass indicated extensive necrosis or hemorrhage, whereas the lesion was localized as a focus in the right ovary. The stroma around neoplastic cells displayed marked infiltration of lymphocytes, plasmacytes, macrophages, and neutrophils. The left ovary exhibited some massive fibrotic areas with calcification or hemosiderin deposition. Vascular invasion was obeserved in every section.
The neoplastic lesion was composed of three different elements with transitional areas. About three-quarters of the lesion displayed a biphasic proliferation pattern by mononucleated cells (MNC) and multinucleated giant cells (MGC). Half of this area showed sheetlike structures with atypical glands composed of solid aggregates of large, epithelial-like polygonal cells. Mononucleated cells and MGC made a mosaic-like pattern by the shading of cytoplasmic stainability (Figure 2A ). The MNC showed bright, rich, often vacuolar, cytoplasm and vesicular atypical nuclei. In contrast, the MGC were characterized by scant basophilic cytoplasm, including multiple nuclei. Some of them contained cytoplasmic vacuoles of various sizes and were occasionally dilated like cysts filled with cell debris or erythrocytes. Mononucleated cells and MGC in this area were complexly intermixed, but each cell border was clearly recognized (Figure 2B).
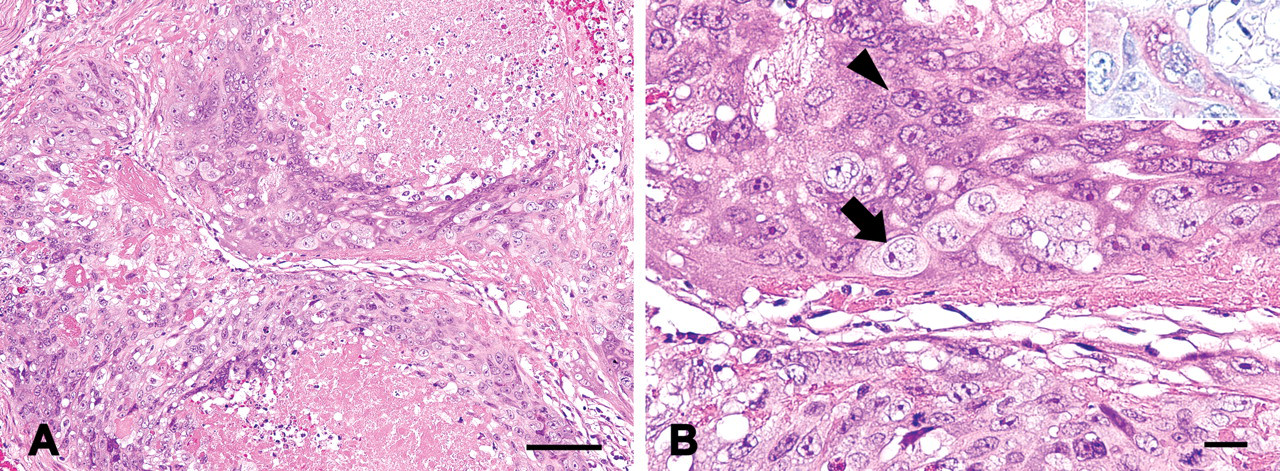
Histological appearance of the tumor: embryonal carcinoma. Shading between the cytoplasm of mononucleated cells (MNC) and multinucleated giant cells (MGC) made this area mosaic (A). Biphasic proliferation consisting of MNC (arrow) and MGC (arrowhead) were seen; hematoxylin and eosin staining. Inset: Most MGC were positive for periodic acid–Schiff stain, whereas MNC were not; periodic acid–Schiff (B). Bars: (A) 100 µm, (B) 20 µm.
The remaining half of the biphasic proliferation showed almost the same structure as the previous one. However, the stainabilities of MNC and MGC were relatively uniform (Figure 3A ). Mononucleated cells in this area were distinct from those in the previous areas, which had amphophilic or eosinophilic cytoplasm, a chromatin-scattered nucleus, and a small cell body. The cell body and nucleus of MGC were slightly smaller than MGC in the previous area, whereas the cytoplasmic and nuclear stainability was analogous (Figure 3B). In these biphasic areas, mitotic figures were easily found.
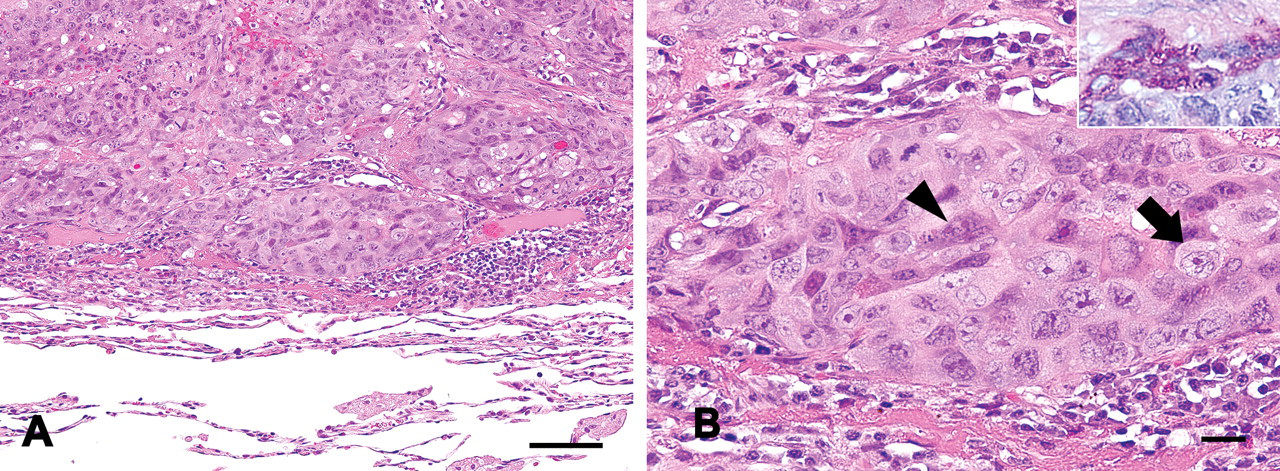
Histological appearance of the tumor; choriocarcinoma. In contrast to embryonal carcinoma, uniform dense stainability was shown (A). Biphasic proliferation consisting of mononucleated cells (arrow) and multinucleated giant cells (arrowhead) were seen; hematoxylin and eosin staining. Inset: Most multinucleated cells were positive for periodic acid–Schiff stain, whereas mononucleated cells were not; periodic acid–Schiff (B). Bars: (A) 100 µm, (B) 20 µm.
In the residual quarter lesion, neoplastic cells formed nests and cords surrounded by an extensive eosinophilic hyaline-like matrix (Figure 4A ). Most neoplastic cells had vesicular nuclei in bright, rich cytoplasm, whereas some cells showed basophilic cytoplasm and nuclei. They were mononucleated, with round to oval nuclei containing prominent nucleoli (Figure 4B). Additionally, oval or column-shaped cells showed palisading proliferation on surrounding fibrotic parenchyma or blood vessels. Multinucleated giant cells were not observed in these hyaline-like matrix areas.
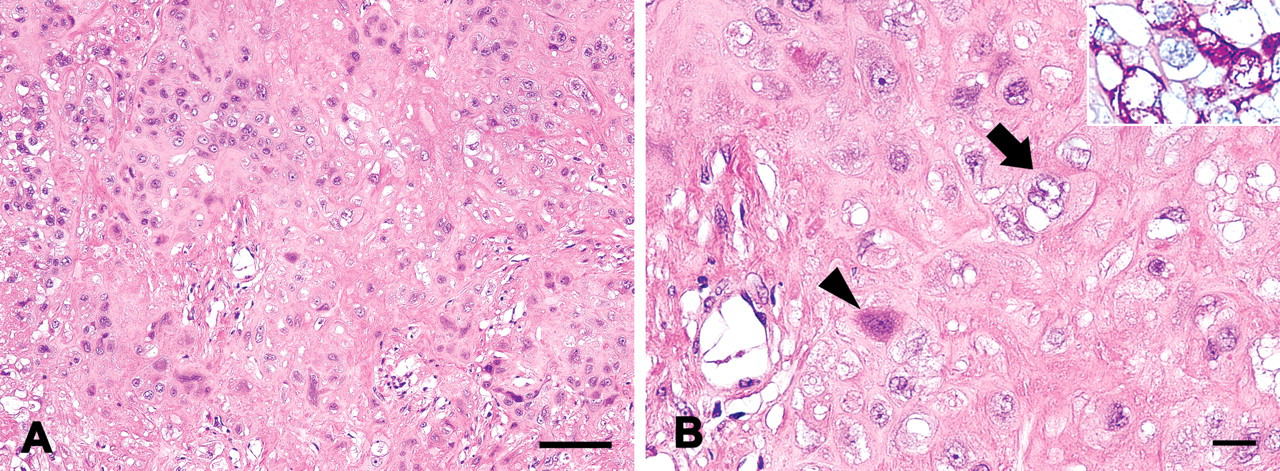
Histological appearance of the tumor; epithelioid trophoblastic tumor. Many floating cells in the eosinophilic hyaline-like matrix were seen (A). Most neoplastic cells had vesicular nuclei in bright, rich cytoplasm (arrow), whereas some cells showed basophilic cytoplasm and nuclei (arrowhead); hematoxylin and eosin staining. Inset: Periodic acid–Schiff positive granules were seen in most neoplastic cells; periodic acid–Schiff (B). Bars: (A) 100 µm, (B) 20 µm.
Typical immunoreactivities for ovarian tumors identified in a literature search are shown in Table 2 (Allison et al. 2006; Chadra et al. 1989; Cheng et al. 2004; Emanuel et al. 2006; Kalhor et al. 2009; Lau and Chang 2006; Mazur et al. 2005; Suster et al. 1998; Tavassoéli et al. 2003; Ulbright. 2005). Immunohistochemistry was performed for this tumor, and the results are shown in Table 3 . First, differential identification was conducted on the biphasic proliferation area consisting of MNC and MGC. In this area, strong positive reactivity of MNC for CK7 ruled out the possibility of yolk sac tumor (YST), dysgerminoma, and granulosa cell tumor (GCT). Furthermore, epithelial-derived tumor (e.g., squamous cell carcinoma) was also dismissed, since MGC showed identical reactivity for hCG (Figure 5A ). Epithelioid trophoblastic tumor (ETT) and placental site trophoblastic tumor (PSTT) were distinguished based on histological characteristics. These results suggest that the area is composed of EC and/or choriocarcinoma (CC). To identify them, supplemental examination for an EC marker (OCT4, CD30, and AFP) was perfomed. Interestingly, the result was consistent with the histological charasteristics. The MNC in the mosaic-like pattern showed positive immunoreactivity for all EC markers, including CD30 (Figure 5B). In contrast, the MNC in the relatively uniformly stained areas exhibited no immunoreactivity for any of them (Figure 5C). In addition, specific immunohistochemical reactivities supported that the biphasic proliferationn area was composed of EC and CC (Table 3).
Typical immunohistochemical reactivity of ovarian tumors.
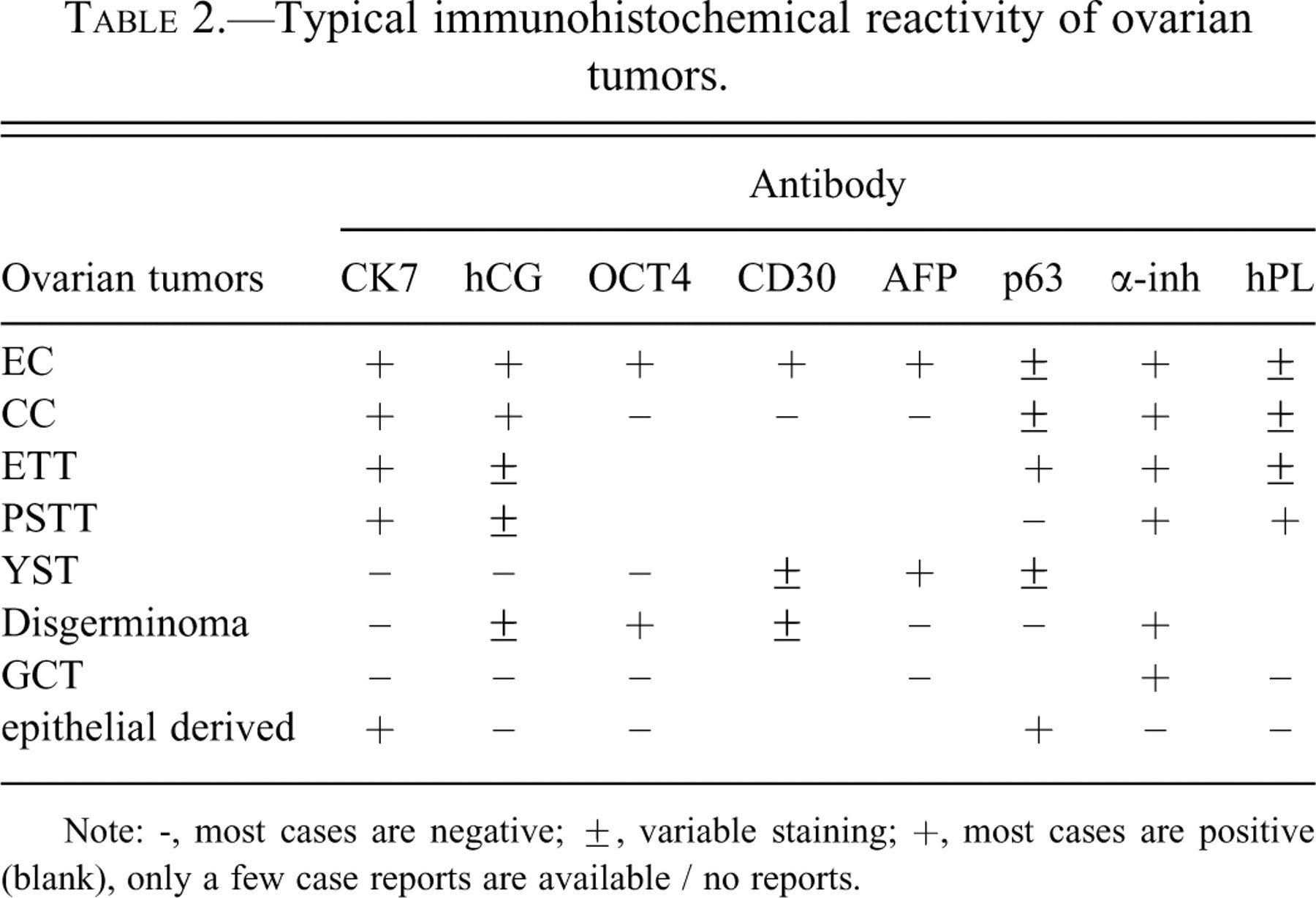
Note: -, most cases are negative; ±, variable staining; +, most cases are positive (blank), only a few case reports are available / no reports.
Summary of histochemical and immunohistochemical findings.

Note: - (no reactivity), ± (discrete reactivity), + (mild reactivity), ++ (moderate reactivity), +++ (marked reactivity) (nuc), positive for cell nucleus Cell cytoplasm is positive, if there is no description.
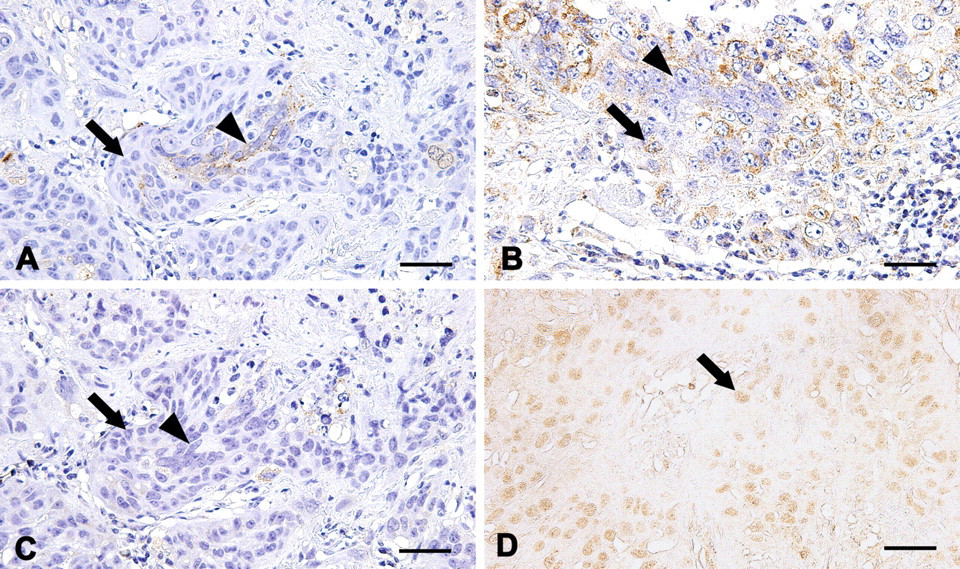
Immunostaining for hCG (A), CD30 (B and C), and P63 (D). Choriocarcinoma: hCG was specifically expressed by cytoplasm of multinucleated giant cell (arrowhead). Mononucleated cells (arrow) were negative (A). Embryonal carcinoma: CD30 was expressed by paranuclear with mononucleated cells (arrow), whereas multinucleated giant cells (arrowhead) were negative (B). Choriocarcinoma (the same area as Figure 5A): each mononucleated cell (arrow) and multinucletaed cell (arrowhead) was negative for CD30 (C). Epithelioid trophoblastic tumor: P63 was expressed by nuclei of
Second, IHC was performed on the eosinophilic matrix-rich area. Although ETT, YST, GCT, PSTT, and epithelial-derived tumors can produce hyaline-like matrix, positive immunoreactivity for CK7 eliminated YST and GCT. Placental site trophoblastic tumor and epithelial-derived tumor were excluded because of nuclear immunoreactivity for p63 (Figure 5D) and cytoplasmic immunoreactivity for α-inh, respectively. Therefore, this area was identified as ETT. Histochemically, MGC in the biphasic proliferation area and neoplastic cells in the eosinophilic hyaline-like matrix area were strongly positive for PAS (Figures 2B, 3B, and 4B). Hyaline-like matrix was focally positive for PAS, but negative for AB. Results of histochemical examination are also shown in Table 3.
Embryonal carcinoma originates from a germ cell differentiating into embryonic multipotential (Tavassoéli and Devilee 2003). It generally reveals disorganized sheets, glands, and papillary structures composed of primitive epithelial cells with pleomorphic nuclei. These cells are positive for CK7, AFP, OCT4, and CD30. In addition, EC can exhibit scattered hCG-positive syncytiotrophoblasts (Tavassoéli and Devilee 2003). In the present case, a mosaic-like biphasic proliferation area showed specific immunoreactions of MNC for CK7, AFP, OCT4, and CD30, and MGC for hCG. These results satisfied the diagnostic criterion for EC. Embryonal carcinoma is a rare germ cell tumor in rodents or domestic animals, and it has never been reported in nonhuman primates (Sawaki et al. 2000). In humans, this tumor has been occasionally reported as a component of MGCT or pure histologic type in the testis (Ulbright 2005).
Like EC, CC is a rare tumor, and only two cases have been reported in the ovaries of nonhuman primates (Farman et al. 2005; Toyosawa et al 2000). The origin of ovarian CC is recognized as a germ cell that shows differentiation to chorionic villi composed of cytotrophoblasts and syncytiotrophoblasts. A typical feature of CC is this dimorphic pattern of syncytiotrophoblast alternating with nests and sheets of cytotrophoblasts. Syncytiotrophoblasts contain multiple nuclei of various sizes. The nuclei are often pyknotic, and they can be vesicular, with prominent nucleoli. They have dense eosinophilic to amphophilic cytoplasm with small vacuoles or large lacunae that often contain erythrocytes. In contrast to syncytiotrophoblasts, cytotrophoblasts are small and uniform. They have a single nucleus with a prominent nucleolus and distinct cell borders. Immunohistochemically, syncytiotrophoblasts are positive for hCG and α-inh, and cytotrophoblasts are positive for CK7 (Mazur and Kurman 2005). These characteristics corresponded to the uniform stainability portion of the biphasic proliferation area. In humans, CC is one of the rarest germ cell tumors in the ovary and testis (Ulbright 2005).
Epithelioid trophoblastic tumor was first reported in humans and has been recently classified (Mazur and Kurman 1994). It is known that human placenta is composed not only of cytotrophoblasts and syncytiotrophoblasts, but also of intermediate trophoplasts (IT). Intermediate trophoplasts are classified into three types; villous IT, implantation-site IT, and chorionic-type IT. The chorionic-type IT constitute the cells of the chorion laeve, where they form a cohesive layer of epithelium. Epithelioid trophoblastic tumor had been reported as a germ cell tumor exhibiting differentiation to chorionic-type IT (Mazur and Kurman 2005). In general, ETT displays a nodular growth pattern of cords and nests of monomorphic cells with eosinophilic to clear cytoplasm set in an eosinophilic, hyaline-like matrix. Well-circumscribed borders and single uniform nuclei are seen. CK7, p63, and α-inh are specific markers in IHC (Mazur and Kurman 2005). Although only one case has been reported in a cynomolgus monkey, the above criteria were fitted to this hyaline-like matrix area in the present case (Giusti et al. 2005). We considered the tumor included the area of ETT.
Taken together, this tumor was considered to be a germ cell–derived tumor with three different intermixed components. Detailed microscopic examination clearly demonstrated that three components occupied a certain number of masses, and each component seemed to contribute to form this tumor. In human pathology, ovarian tumors formed from several germ cell–derived components are treated as MGCT in the WHO tumor classification (Tavassoéli and Devilee 2003). Although the term is not totally accepted in laboratory or domestic animal pathology, three different elements were prominent in this case. Therefore, we diagnosed this case as an MGCT with EC, CC, and ETT. Obviously, this was an extremely rare case histologically, but it also might be uncommon epidermiologically. In humans, primitive germ cell tumors tend to develop in children and youth under the age of twenty-one years (Tavassoéli and Devilee 2003). Accordingly, this case was interesting in terms of developmental age, because a seven-year-old female cynomolgus monkey corresponds to a middle-aged woman. To the best of our knowledge, this is the first case of EC in nonhuman primates as a component of an MGCT. Although it is unclear whether this tumor can be induced or not, germ cell tumors (e.g., YST and terotoma) are known as inducible tumors (Ito et al. 1982). We considered this case might be spontaneous, because the animal had never been treated with any known carcinogen. The left ovary showed extensive fibrosis, whereas there was active proliferation of neoplastic cells and extended replacement of original tissue in the lung. In the right ovary, the neoplastic cells were localized as foci, and most normal tissue remained. These findings supported the possibilities that the lung and the right ovary were metastatic organs and the left ovary was primary. Hematogenous metastasis might be involved in this pathological condition, because vascular invasion was observed.
As mentioned above, CC has been reported in some nonhuman primates, though identification of this condition was made with microscopic and restrictive immunohistochemical examination such as against cytokeratin AE1/AE3 (CK) and hCG (Farman et al. 2005; Toyosawa et al. 2000). As a result of this study, EC of nonhuman primates could represent biphasic proliferation patterns of MNC and MGC. Additionally, MNC and MGC were positive for CK and hCG, respectively. These characteristics are common in CC, hence using only CK and hCG as immunohistochemical markers is insufficient for distinguishing CC from EC. In addition, employing complementary antibodies such as AFP, OCT4, and CD30 in combination with CK7 and hCG proved to be useful when identifying EC or CC.
Footnotes
Acknowledgments
We are grateful to Kimiko Hara, Fukuroi, Daiichi-Sankyo Co., Ltd., for her technical assistance in the immunohistochemistry slide preparation.
