Abstract
Biomass burning is a major source of indoor air pollution in rural India. The authors investigated in this study whether cumulative exposures to biomass smoke cause activation of the serine/threonine kinase Akt in airway cells and peripheral blood lymphocytes (PBL). For this, the authors enrolled 87 premenopausal (median age 34 years), nonsmoking women who used to cook with biomass (wood, dung, crop wastes) and 85 age-matched control women who cooked with cleaner fuel liquefied petroleum gas. Immunocytochemical and immunoblotting assays revealed significantly higher levels of phosphorylated forms of Akt protein (p-Aktser473 and p-Aktthr308) in PBL, airway epithelial cells, alveolar macrophages, and neutrophils in sputum of biomass-using women than control. Akt activation in biomass users was associated with marked rise in generation of reactive oxygen species and concomitant depletion of superoxide dismutase. Measurement of particulate matter having a diameter of less than 10 and 2.5 µm in indoor air by real-time aerosol monitor showed 2 to 4 times more particulate pollution in biomass-using households, and Akt activation was positively associated with particulate pollution after controlling potential confounders. The findings suggest that chronic exposure to biomass smoke activates Akt, possibly via generation of oxidative stress.
Introduction
Akt or protein kinase B (PKB) is a serine/threonine kinase. It controls key cellular processes like glucose metabolism, cell cycle progression, apoptosis, and cell survival (Monick, Carter, et al. 2001; Brunet et al. 1999; Burow et al. 2000; Eves et al. 1998; Kulik and Weber 1998; Madrid et al. 2000; Monick, Mallampalli, et al. 2001). Activation of Akt requires its translocation to the plasma membrane and binding with phosphatidylinositol triphosphate (PIP3) via pleckstrin homology (PH) domain. Phosphorylation of Akt at Thr308 by phosphoinositide-dependent kinase 1 (PDK1) partially activates Akt (Alessi et al. 1996), and its full activation is achieved by additional phosphorylation at Ser473 by phosphoinositide-dependent kinase 2 (PDK2) (Toker and Newton 2000). Following activation, Akt detaches itself from the cell membrane, migrates to the cytoplasm or the nucleus, and starts mediating pro-survival and anti-apoptotic effects in part via phosphorylation and inhibition of the Bcl-2 homolog BAD, phosphorylation and inactivation of FoxO subfamily of forkhead transcription factors, and transcriptional activation of Yes-associated protein (Datta et al. 1997; Brunet et al. 1999). In addition, Akt mediates cell proliferation and migration via the phosphorylation and activation of the endothelial nitric oxide synthase and glycogen synthase kinase-3β, which is functional during cell cycle progression, and inactivation of p21CIP1 and p27KIP1, which are inhibitors of cyclin-dependent kinases (Fosbrink et al. 2006; Diehl et al. 1998). Akt activation facilitates cell survival even after DNA adducts formation (West et al. 2003). In conformity with this, excessive activation of Akt has been implicated in the development of a wide range of human cancers (Scheid and Woodgett 2001). Akt mediates its tumor-promoting activity by a variety of mechanisms that include down-regulation of its inhibitor phosphatase and tension homologue deleted on chromosome 10 (PTEN), activation of the proto-oncogene Ras, and up-regulation of growth factor receptors (Luo, Manning, and Cantley 2003).
Change in Akt expression can be clinically relevant. Up-regulation of phosphorylated Akt (p-Akt) is considered an early event in the pathway of bronchial (Tsao et al. 2003; Chun et al. 2003; Balsara et al. 2004; Al-Saad et al. 2009), oral (Watanabe et al. 2009), and mammary (Al-Bazz et al. 2009) carcinogenesis. Besides, Akt activation may cause relaxation of vascular smooth muscle cells via increased nitric oxide production (Carrillo-Sepúlveda et al. 2010), suppression of gastric acid secretion (Rotte et al. 2010), airway inflammation via activation of pro-inflammatory mediators (Beaver et al. 2009; Lee et al. 2010), and perpetuation of inflammation by delaying neutrophil apoptosis (Salamone et al. 2010).
A large majority of rural women of the developing world including India are chronically exposed to high level of indoor air pollution (IAP) for daily household cooking with traditional biomass fuel (BMF) such as dried cow dung cake, firewood, and agricultural wastes. Smoke emitted from burning biomass contains a wide spectrum of pollutants that include carbon monoxide; coarse, fine, and ultrafine particulate matters; oxides of nitrogen and sulphur; formaldehyde; transitional metals; volatile organic compounds including benzene; and polycyclic aromatic hydrocarbons such as benzo(a)pyrene (Zhang and Smith 1996). Some of these pollutants like benzo(a)pyrene and benzene are established human carcinogens (Smith 2000). Women who used to cook with BMF in poorly ventilated kitchen for 2 to 6 hours a day are believed to be inhaling carcinogens equivalent to smoking two packs of cigarettes per day (Pandey et al. 1989). It is reasonable to assume therefore that chronic exposures to biomass smoke may impair cellular functions, especially in those cells that are at the direct route of exposure. Given the importance of Akt in regulating cell survival, growth, and apoptosis, it thus seems important to study its activity in relation to biomass smoke exposures. To our knowledge, no such study has been carried out so far. Against this background, we examined in this study the impact of IAP from BMF use on Akt activation in exfoliated AEC and inflammatory cells present in expectorated sputum as well as in PBL in a group of never-smoking, premenopausal women from eastern India who used to cook exclusively with BMF for the past 5 years or more. We have compared the findings with an age-matched group of control women from the same neighborhood who cooked with cleaner fuel liquefied petroleum gas (LPG).
Materials and Methods
Subjects
A total of 172 premenopausal women aged between 28 and 42 years from rural areas of West Bengal, a state in eastern India, were enrolled in this study after obtaining written informed consent. They attended health checkup camps organized in different villages with the active cooperation of the local administrative bodies and nongovernmental organizations. Among the participants, 87 women (aged 28–42 yr, median 34 yr) were cooking daily for 3–6 hours exclusively with wood; cow dung; and agricultural refuse, such as bamboo, jute stick, paddy husk, hay, and dried leaves for the past 5 years or more. Accordingly they were grouped as biomass-users. The remaining 85 women, aged 27–42 yr, median age 33 yr, were from the same villages, but they used to cook with cleaner fuel LPG and were considered as control. Some households, especially those using biomass as cooking fuel, lacked a separate kitchen. Women of these households used to cook in a space adjacent to the living room.
Inclusion and Exclusion Criteria
The inclusion criteria were apparently healthy, premenopausal married women actively engaged in household cooking for the past 5 years or more who were nonsmokers and nonchewers of tobacco and had a body mass index > 15 and < 30 kg/m2. Women using oral contraceptive, had a history of malignancy, or were currently under medication were excluded.
During personal interview with female members of the research team, each participant was requested to furnish information about age, education, family size and income, habit, cooking time per day, years of cooking, fuel and oven type, ventilation and location of kitchen, and general health problems in past 3 months and past 1 year. As most of the participants were poorly educated, the researchers recorded their responses in structured questionnaire forms on their behalf. The Ethics Committee of Chittaranjan National Cancer Institute approved the study protocol. The research was conducted according to the principles of the Helsinki Declaration.
Collection of Blood and Expectorated Sputum
Venous blood (5 ml) was collected in vacutainer tubes (Becton Dickinson [BD], San Jose, CA, USA) containing K2EDTA as anticoagulant. Blood was collected at a fixed time of the day (9.30–10.30 hours) to minimize diurnal variation. Early morning expectorated sputa were collected in sterile plastic cups for 3 consecutive days to harvest airway cells following the procedure of Erkilic et al. (2003). Three smears were prepared from the nontransparent highly viscous part of each freshly collected sputum sample on clean glass slides from each day’s sample—one for Papanicolaou (Pap) staining and two for immunocytochemistry (ICC), one for p-Aktser473 and another for p-Aktthr308. After air drying, one slide marked for Pap staining was fixed with 95% ethanol and the other two marked for ICC were fixed in ice-cold methanol for 20 min at the site of collection. The remaining part of the expectorated sputum was collected in sterile plastic screw-cap tubes containing 20 ml of phosphate-buffered saline (PBS) with 0.1% dithiotheritol.
PAP Staining for Cytopathology of Airway Cells
Ethanol-fixed slides were brought to the laboratory and were stained with Papanicolaou (Pap) staining method following the procedure of Hughes and Dodds (1968). The slides were coded and examined under light microscope (Leitz, Germany) at 400x and 1,000x magnification. At least 10 high power fields (hpf; 40x objective and 10x eyepiece) per slide were examined. Sputum cytology and differential distribution of non-squamous AEC, AM, and inflammatory cells were performed following the established criteria (Grubb 1988). The slides were coded and cytological examinations were carried out blindly. The 3-day average value of each parameter was taken as the representative data of each participant.
Cytopathological Diagnosis and Specimen Adequacy Criteria
The sputa were cytopathologically diagnosed into six categories following the criteria of Neumann et al. (2009): category I. nondiagnostic, insufficient pulmonary material; category II. nondiagnostic, distorted, poorly preserved or stained cells; category III. benign, normal cells are present; category IV. atypical cells are present, probably benign; category V. atypical cells are present, suspicion of malignancy; and category VI. malignant cells present. When a sputum slide contained at least 50 alveolar macrophages and was grouped in category III or above, it was considered an adequate specimen of the lower airways (Neumann et al. 2009). Squamous metaplasia and dysplasia were diagnosed on the basis of cytological criteria (Grubb 1988; Naib 1996). Metaplastic cells appear as a single or loose cluster of bright orange or dark blue-green-stained cells with “glassy” appearance. The cell shape is often irregular, size varies between 10–25µm and the chromatin is dense, greater than normal, and nucleoli are seldom seen. The characteristic features of dysplasia are polymorphism in size and shape of the cell and the nucleus, markedly increased nuclear-cytoplasmic ratio, abnormality in chromatin distribution, nuclear membrane indentations, and intensely acidophilic cytoplasm.
Lymphocyte Separation from Peripheral Blood
PBL were separated from EDTA-anticoagulated whole blood by centrifugation in density gradient (Histopaque®-1077, Sigma Chem, St. Louis, MO, USA) for 10 min at 400 x g at 4°C. The lymphocytes were collected in microfuge tubes, resuspended in 500 µl of ice-cold phosphate buffered saline (PBS). Smears of lymphocytes were prepared on clean glass slides for immunocytochemistry (ICC) for localization of phosphorylated Akt (p-Akt) protein.
Localization of p-Akt by ICC
Immunocytochemistry of PBL and sputum slides were done following a standard procedure (Mondal et al. 2010). In brief, slides containing PBL and sputum cells were air dried and fixed in ice-cold methanol for 30 min, air dried, washed in PBS thrice, and blocked in 3% BSA (Sigma–Aldrich Chemicals, Saint Louis, MO, USA) for 1 hr at room temperature. The slides were overnight incubated in darkness with rabbit polyclonal p-Aktser473 (Santa Cruz, CA, USA) and p-Aktthr308 (Abcam, Tokyo, Japan) primary antibodies (diluted 1:50 and 1:200 in 1% BSA, respectively) in a humid box at 4°C. After washing with PBS, slides were incubated with anti-rabbit IgG, F(ab')2-HRP (Santacruz, USA) secondary antibody diluted 1:500 in 1% BSA for 90 min. The slides were developed by incubating with substrate for HRP for 45 min in darkness followed by washing with distilled water and counterstaining with hematoxylin, dehydration in graded ethanol, and mounted in distrene plasticizer xylene (DPX). The slides were coded and examined blindly. For each participant, one sample of PBL and three samples of sputum obtained from 3 consecutive days were examined. The 3-day average values of p-Aktser473 and p-Aktthr308 expressions in sputum cells were taken as the representative data of each participant.
Cell Lysate Preparation for Immunoblot and SOD Assay
Whole sputum collected in PBS with 0.1% dithiotheritol was centrifuged for 10 min at 400 x g at 40C. The supernatant was discarded, and the cells were suspended in fresh PBS and centrifuged again and the cell pellet was collected. Whole-cell protein from sputum and PBL were obtained by lysing the cells on ice with 500 µl of lysis buffer (0.05 M Tris [pH 7.4], 0.15 M NaCl, 1% Nonidet P-40, with added protease and phosphatase inhibitors: 1 protease minitab [Roche Biochemicals, Indianapolis, IN, USA]/10 ml and 1x phosphatase inhibitor mixture [no. 524625; Calbiochem, Darmstadt, Germany]) followed by four 20 s pulses of sonication. Samples were kept in ice in between the sonication pulses. Lysates were centrifuged at 15,000 x g for 10 min at 40C in an ependorf microcentrifuge. The supernatant was collected and used for immunoblotting (sputum and PBL) and SOD (sputum) assay.
Immunoblot Analysis of p-Akt
Cleared lysates were subjected to SDS–PAGE (10% matrix) according to the method of Laemmli (1970). Protein concentrations in the lysates were measured using the Bradford assay. Proteins (60 µg) separated by SDS-PAGE were electrophoretically transferred to nitrocellulose membrane (ECL; Amersham Biosciences, Arlington Heights, IL, USA) using a mini transblotter (Bio-Rad, Hercules, CA, USA). Membranes were blocked, then incubated consecutively with rabbit polyclonal primary antibodies (1:1,000 dilution) raised against human p-Aktser473, p-Aktthr308, and total Akt (Cell Signaling Technology, Beverly, MA, USA) and HRP-conjugated anti-rabbit IgG secondary antibody (1:2,000 dilution). Immunoreactive proteins were visualized with enhanced Chemiluminescence method using luminol reagent kit (Santa Cruz Biotechnology, Santa Cruz, CA, USA). Protein levels in each band were quantitated using a Fluor-S scanner and Quantity One software for analysis (Bio-Rad). The data were analyzed using GraphPad Prism software (San Diego, CA, USA).
Flow Cytometric Measurement of ROS Generation
Generation of reactive oxygen species (ROS) was measured in leukocytes (granulocytes, monocytes, and lymphocytes) present in anticoagulated venous blood and sputum cells by flow cytometry using DCF-DA following the procedure of Rothe and Valet (1990). For this, 10,000 events were acquired in a flow cytometer (FACS Calibur with sorter, Becton Dickinson [BD], San Jose, CA, USA) using Cell Quest software (BD). Respiratory burst and generation of ROS by cells were associated with emission of green fluorescence that was recorded in fluorescence channel-1 and was expressed as mean fluorescence intensity (MFI) in arbitrary unit. In case of blood, granulocytes, monocytes, and lymphocytes were gated on the basis of their characteristic forward and side scatters on dot plot and MFI of each population was recorded, while MFI of the total cells present in sputum was recorded.
Spectrophotometric Measurement of SOD
The activity of the antioxidant enzyme superoxide dismutase (SOD) was assayed in blood erythrocytes and sputum cell lysate spectrophotometrically following the procedure of Paoletti et al. (1986). EDTA anticoagulated blood was centrifuged at 200 x g at 40C for 5 min, the RBC pellet was collected and washed in 0.9% saline and then lysed with cold distilled water (1: 9, v/v). Following centrifugation at 500 x g for 10 min, the supernatant was collected. In spectrophotometric cuvette, 800µl of TEA-DEA buffer containing 100 mM each of triethanolamine (TEA) and diethanolamine (DEA) (Qualigens, Mumbai, India), 40µl of 7.5 mM nicotinamide adenine dinucleotide reduced disodium salt (NADH, SRL, Mumbai, India), pH 7.4, 25µl of a mixture (1:1, v/v) of 0.2 M ethylene diaminetetraacetic acid disodium salt (Sigma-Aldrich Chem, St. Louis, MO, USA) and 0.1 M manganous chloride (SD-Fine Chem, Mumbai, India), and 100µl of sample (RBC or sputum cell lysate) were added and mixed well. The absorbance (OD) was measured at 340 nm in a spectrophotometer (Shimadzu, Kyoto, Japan) immediately (0 min) and 1, 2, 3, 4, and 5 min after addition of mercaptoethanol. SOD activity was calculated from the standard curve and was expressed as U/ml.
Measurement of PM 10 and PM 2.5 in Indoor Air
Particulate matter with aerodynamic diameter less than 10µm (PM10) and 2.5µm (PM2.5) were measured in the cooking areas with real-time laser photometer (DustTrak™ Aerosol Monitor, model 8520, TSI Inc., Shoreview MN, USA) that contained 10-mm nylon Dor-Oliver cyclone and operated at a flow rate of 1.7 L/min, measuring particle load in the concentration range of 1µg to 100 mg/m3. The monitor was calibrated to the standard ISO 12103-1 A1 test dust. We used two monitors for simultaneous measurement of PM10 and PM2.5. Air sampling was done in each household for three consecutive days, 8 hours/day (7.00–15.00 hrs) covering both cooking and noncooking time. The mean of 3 days was used as the indoor air quality of a single household. Biomass-using women cook in a sitting position 2–3 ft away from the open chullah (oven). Accordingly, the monitor was placed in the breathing zone of the cook 2.5 ft above the floor level on a wooden stool 3 ft away from the chullah. LPG users, on the other hand, cook in a standing position, and the monitor was therefore placed at a height of 4.5 ft. Since laser photometers make an overestimation of PM levels by 2–3 fold (Lehocky and Williams 1996), the raw data require reduction using correction factors. We reduced the 8-hr raw data by dividing with a correction factor of 2.5 for PM10 (Siddiqui et al. 2009) and 2.77 for PM2.5 (Lehocky and Williams 1996; Chung et al. 2001).
Statistical Analysis
Results are presented as mean ± standard deviation (SD) or median with range. Analysis between groups was performed using Student’s t-test, chi-square test, or Mann-Whitney U test, as applicable. The possibility of an association between measured parameters with age, BMI, family income, education, cooking hours per day, lifetime duration of cooking (cooking-years), kitchen location, family size, number of smokers in family, use of mosquito repellant, and PM10 and PM2.5 levels in cooking areas was tested using univariate regression analysis. Variables that showed significant association were later included in a backward stepwise multiple regression model to adjust for their effects. Statistical analyses were performed using EPI info 6 and SPSS statistical software (Statistical Package for Social Sciences for Windows, release 10.0, SPSS Inc., Chicago, USA) and p < .05 was considered significant.
Results
Demography
Demographic and socioeconomic characteristics of study population are summarized in Table 1 . The LPG and BMF users were comparable with respect to age, body mass index, cooking years, hours of cooking per day, environmental tobacco smoke for presence of a smoker in the family, food habit, and use of mosquito repellant. However, BMF users were less educated (p < .05 in Mann-Whitney U test), and their family income was significantly lower than that of their neighbors who used to cook with LPG (p < .05 in Student’s t-test). Moreover, 42.5% of BMF-using households lacked a separate kitchen against 17.7% of LPG-using households, and the difference between these two groups in this regard was significant (p < .05) in chi-square test.
Demographic and socioeconomic characteristics of biomass fuel and LPG-using women.
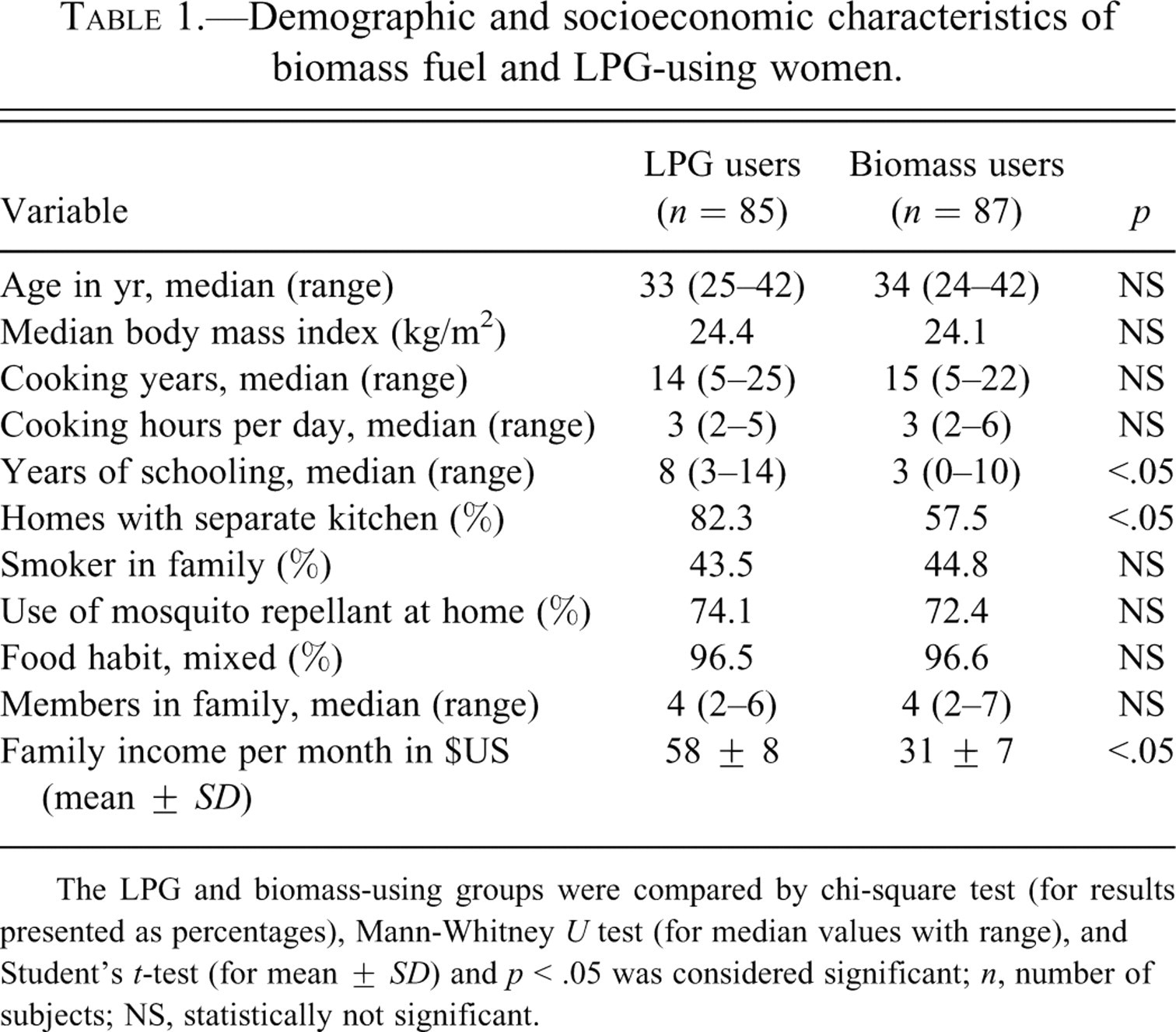
The LPG and biomass-using groups were compared by chi-square test (for results presented as percentages), Mann-Whitney U test (for median values with range), and Student’s t-test (for mean ± SD) and p < .05 was considered significant; n, number of subjects; NS, statistically not significant.
Particulate Pollutants in Indoor Air
The levels of particulate air pollutants in indoor air in cooking areas were significantly higher (p < .001) in BMF-using households than that of LPG-using households during cooking as well as noncooking time (p < .001) (Table 2 ).
Comparison of particulate pollution in indoor air of cooking areas between LPG- and BMF-using households.
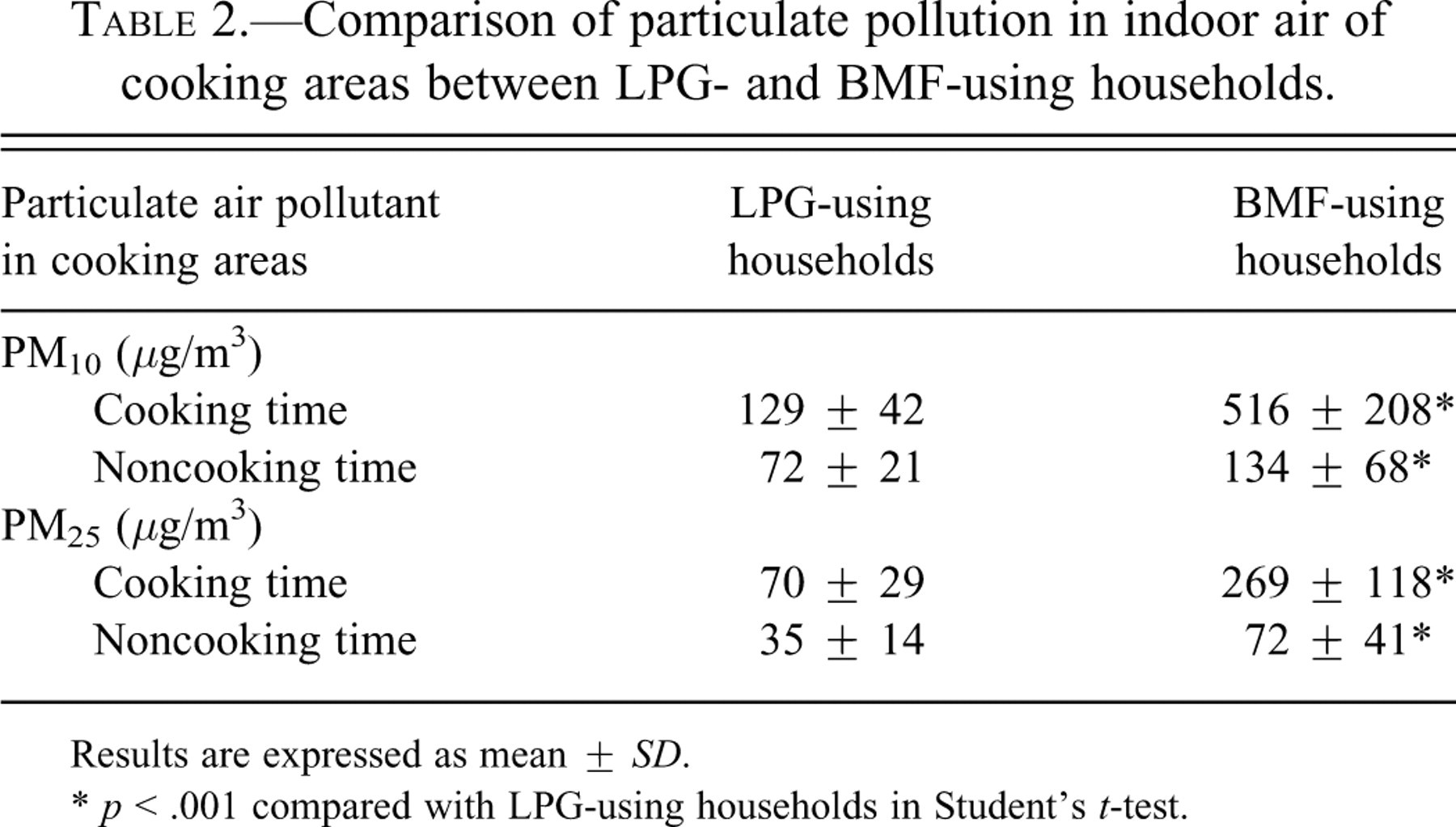
Results are expressed as mean ± SD.
* p < .001 compared with LPG-using households in Student’s t-test.
Sputum Cytology
Biomass users had elevated number of total cells in sputum (89.6 ± 24.5 vs. 64.1 ± 11.2 cells/hpf, p < .01) than controls. Sputa of BMF users contained more AEC (11.9 ± 3.3 vs. 6.3 ± 2.21 cells/hpf, p < .01), neutrophils (57.9 ± 18.6 vs. 46.6 ± 11.8 cells/hpf, p < .01), eosinophils (1.6 ± 0.8 vs. 0.4 ± 0.2, cells/hpf p < .01), lymphocytes (4.5 ± 1.3 vs. 3.3 ± 1.1, cells/hpf p < .05), and AM (12.9 ± 6.9 vs. 6.7 ± 3.1 cells/hpf, p < .01) than that of control. Moreover, AEC metaplasia was recorded in 20.7% of BMF-using women in contrast to 8.2% of control, and mild to moderate dysplasia was present in sputa of 4.5% BMF and 1.2% LPG users (p < .01 in chi-square test).
Expression of p-Aktser473 and p-Aktthr308
Phosphorylated forms of Akt, p-Aktser473, and p-Aktthr308 were localized by ICC and immunoblotting. Expression of p-Aktser473 was mostly nuclear, while p-Aktthr308 was expressed chiefly in the cytoplasm (Figures 1–4). Compared with control, a statistically significant increase in the percentages of p-Aktser473- and p-Aktthr308–expressing epithelial cells, neutrophils, and AM in sputum and lymphocytes in peripheral blood were recorded among BMF users (Table 3 ). BMF users had 3 times more p-Aktser473-expressing AEC and PBL than controls. Similarly, they had 4 times more airway neutrophils and AMs that expressed p-Aktser473 when compared with controls. On the other hand, BMF users showed 5-fold increase over control in the percentage of basal cells of the airways that expressed p-Aktthr308 in their cytoplasm. The percentage of p-Aktthr308-expressing cells was 2- to 2.7-fold higher for the remaining cell types among BMF users (Table 3).
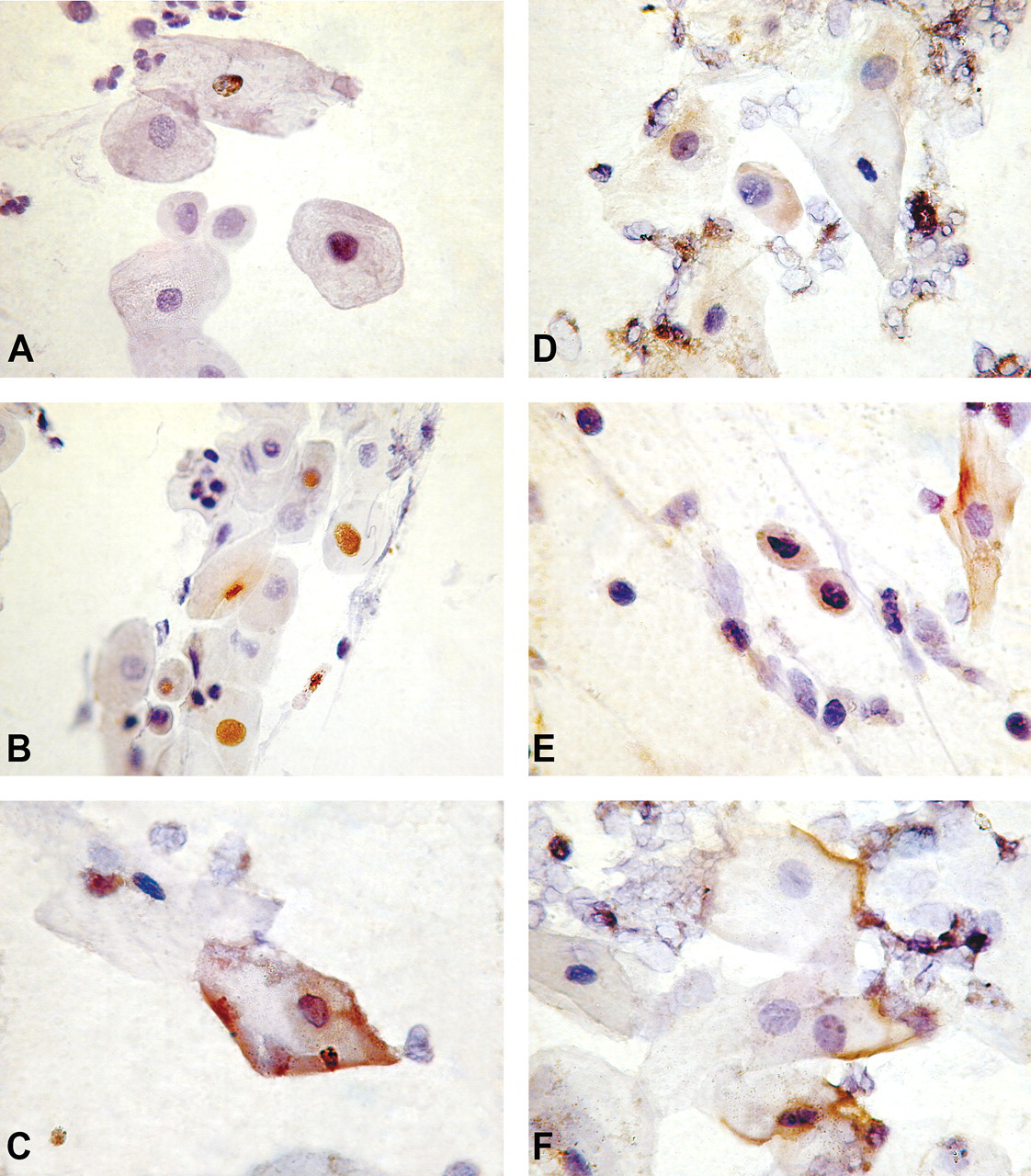
Immunocytochemical localization of phosphorylated forms of Akt protein in AEC present in sputum of biomass- and LPG-using control women. The cell nuclei were counterstained with hematoxylin. Compared with relatively weak nuclear expression of p-Aktser473 in AEC of control women (A), strong p-Aktser473 expression was observed in nuclei (B) as well as in cell membrane (C) of AEC of biomass users. Similarly, strong p-Aktthr308 expression was detected in nuclei and cell membrane of AEC of biomass-using women (E,F), while weak cytoplasmic expression was observed in AEC of control women (D). Magnification x 1,000.
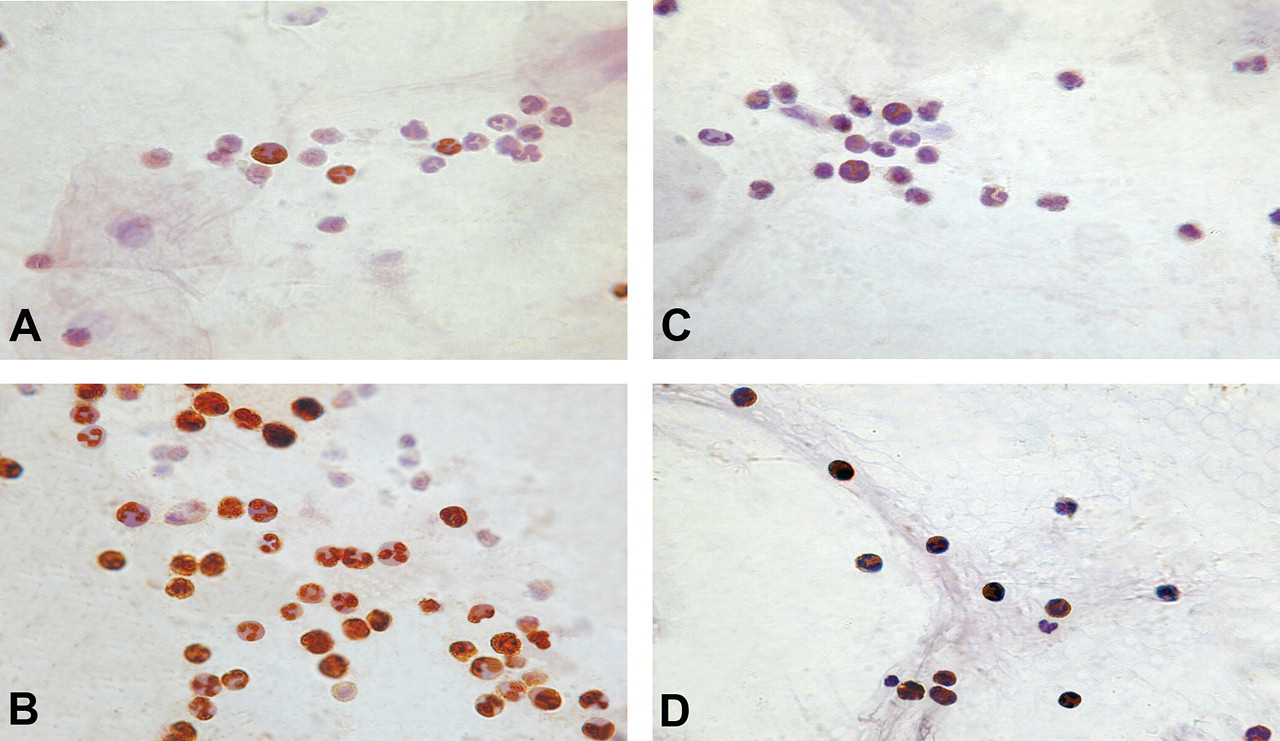
Immunocytochemical localization of phosphorylated forms of Akt in sputum neutrophils of biomass- and LPG-using control women. The cell nuclei were counterstained with hematoxylin. A greater proportion of sputum neutrophils of biomass-using women expressed p-Aktser473 in nuclei (B) than that of control (A). p-Aktthr308 expression in general was weak and chiefly cytoplasmic and biomass-users illustrated greater frequency of positive cells (D) than the control (C). Magnification x 1,000.
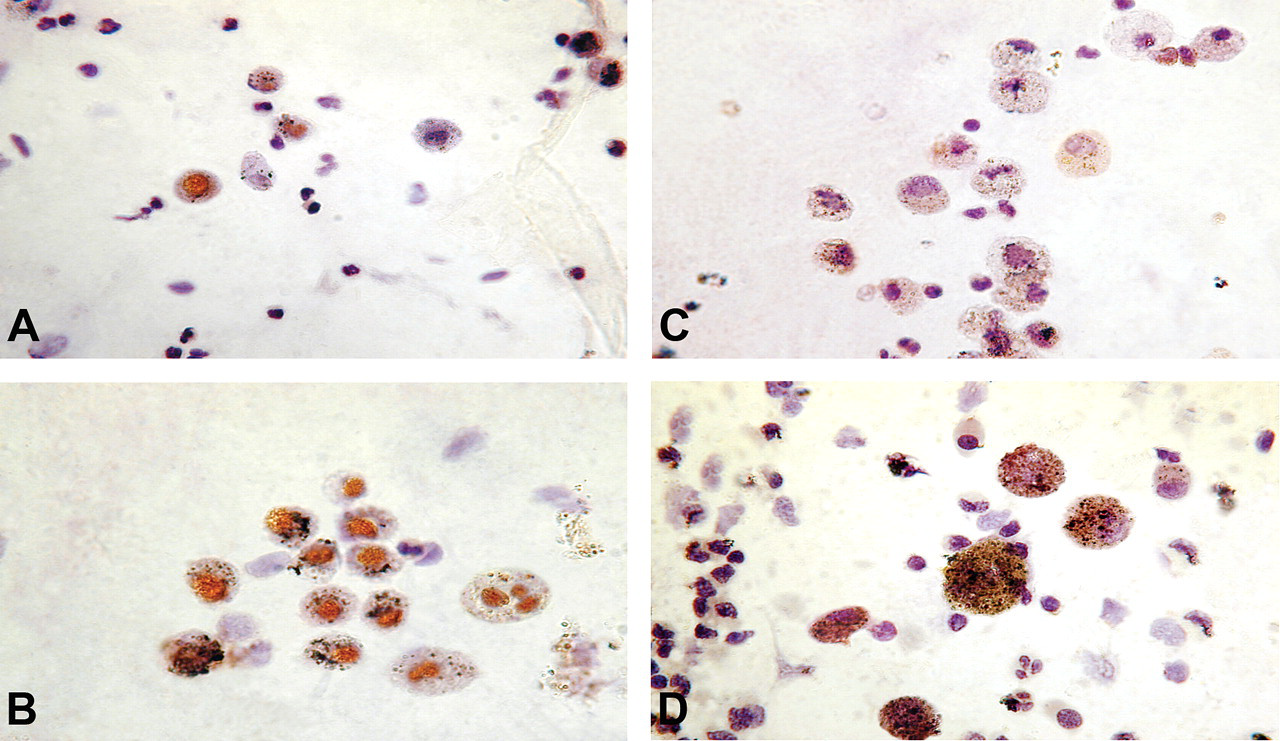
Immunocytochemical localization of phosphorylated forms of Akt in AM present in expectorated sputum of biomass- and LPG-using control women. The cell nuclei were counterstained with hematoxylin. Although the staining intensity of p-Aktser473 in AM from control (A) and biomass-using women (B) is similar, the frequency of positive cells is greater in B than in A. Expression of p-Aktthr308, on the other hand, was weak and chiefly cytoplasmic, and biomass users had greater frequency of p-Aktthr308 positive AM (D) than that of control (C). Magnification x 1,000.
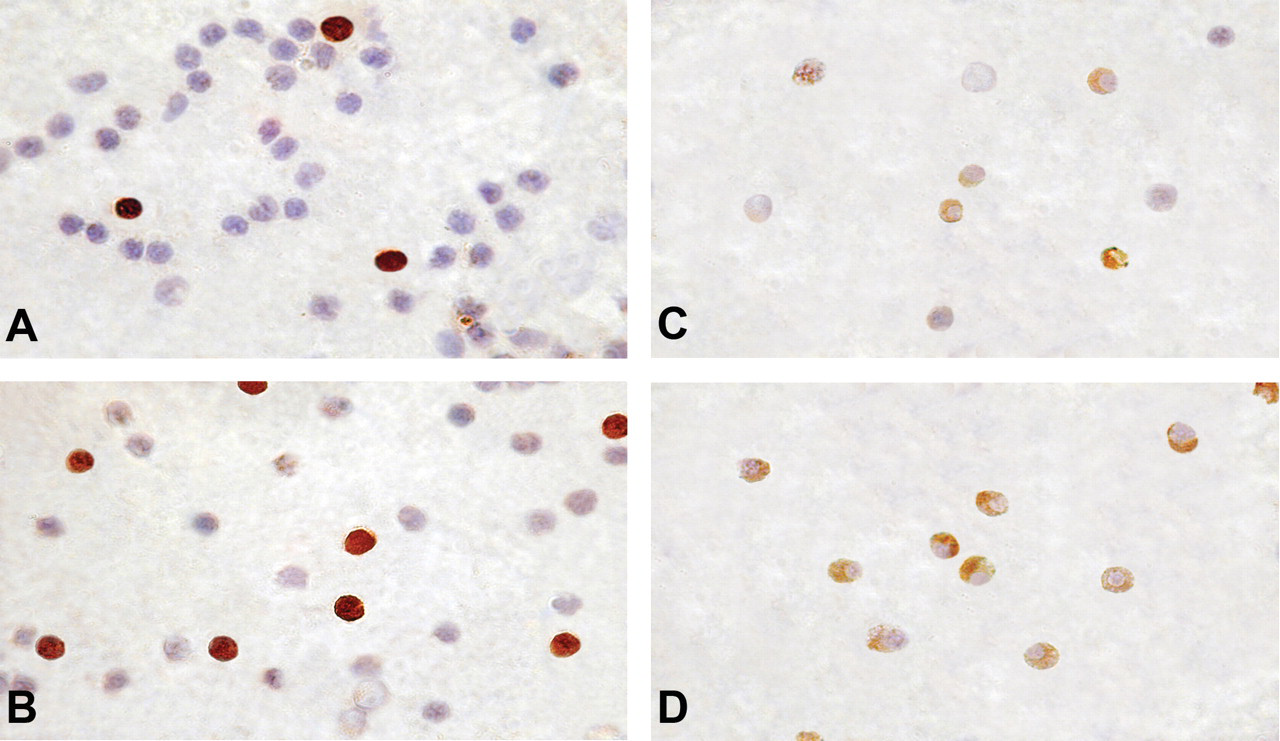
Immunocytochemical localization of phosphorylated forms of Akt in PBL of biomass- and LPG-using control women. The cell nuclei were counterstained with hematoxylin. Expression of p-AktSer473 was strong and both nuclear and cytoplasmic. Biomass-using women had higher frequency of p-AktSer473–expressing PBL (B) than control (A). Expression of p-AktThr308 in PBL of control women was negligible (C). In contrast, strong, cytoplasmic p-AktThr308 expression was found in a large numbers of PBL of biomass users (D). Magnification x 1,000.
Changes in nuclear and cytoplasmic expression of two forms of phosphorylated Akt in LPG- and BMF-using women.
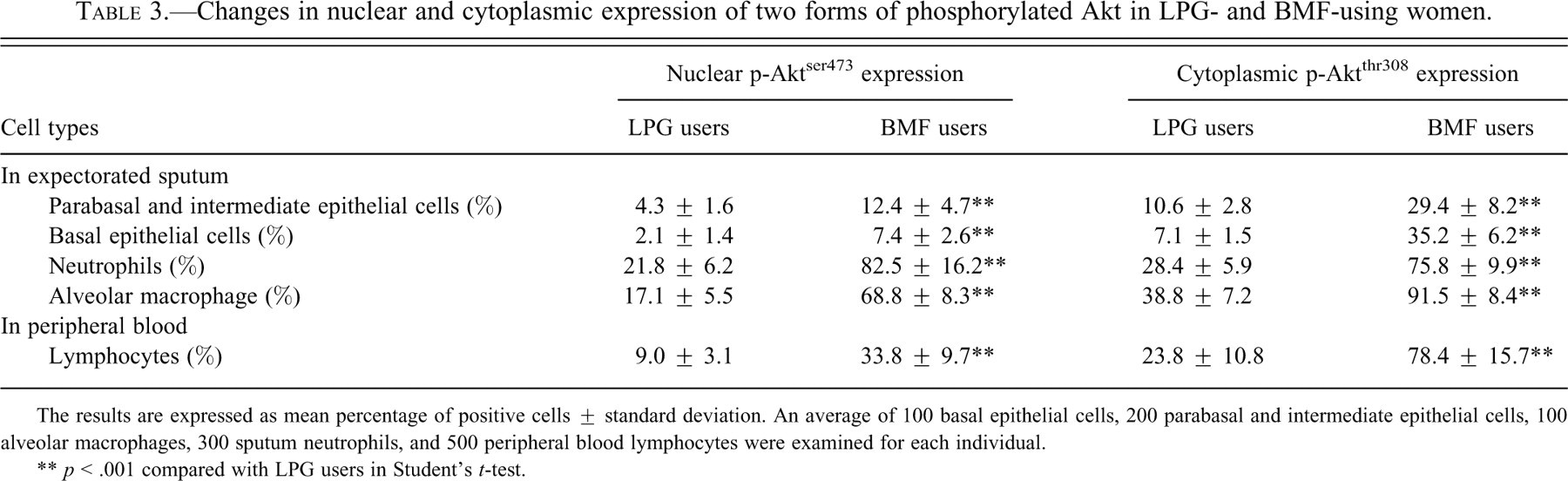
The results are expressed as mean percentage of positive cells ± standard deviation. An average of 100 basal epithelial cells, 200 parabasal and intermediate epithelial cells, 100 alveolar macrophages, 300 sputum neutrophils, and 500 peripheral blood lymphocytes were examined for each individual.
** p < .001 compared with LPG users in Student’s t-test.
Compared with cytologically normal AEC, the percentage of p-Akt–expressing cells was remarkably higher (p < .001) among metaplastic and dysplastic cells both among LPG and BMF users. Expression of p-Aktser473 was found in 56% and 62% of metaplastic cells and 82% and 86% of dysplastic cells in LPG and BMF users, respectively. Similarly, p-Aktser473 expression was recorded in 81% and 83% of metaplastic cells and 92% and 91% of dysplastic cells in sputa of LPG and BMF users, respectively.
Controlling age, family income, and kitchen location as possible confounders, multivariate logistic regression analysis showed a strong positive association between percentage of p-Aktser473-expressing AEC with PM10 (odds ratio [OR] = 1.33, 95% confidence interval [95% CI] 1.09–1.62) and PM2.5 (OR = 1.45, 95% CI 1.12–1.74) level in indoor air.
Immunoblotting revealed greater presence of p-Akt in PBL and sputum cells of women who used to cook with BMF (Figure 5 ). Akt phosphorylation positively correlated with years of cooking with biomass without altering the total Akt protein level (Figure 5).
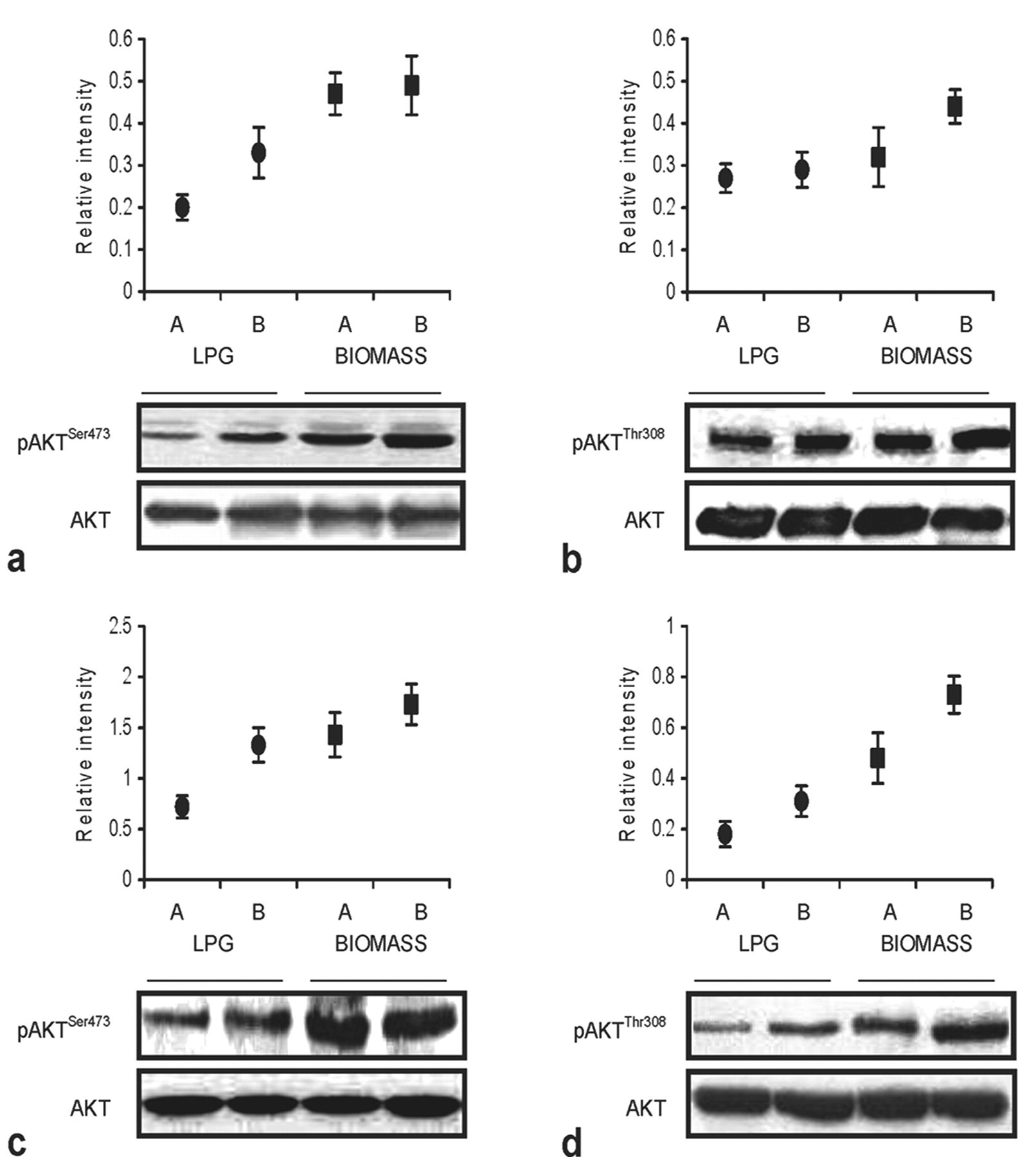
Western blot analysis of p-AktSer473 (a,c) and p-AktThr308 expression (b,d) in PBL (a,b) and airway cells (c,d) of premenopausal women who were cooking with LPG or biomass fuel for the past 10–19 (A) and 20–29 (B) years. Whole cell lysates (60μg/lane) were used for immunoblotting. The degree of Akt phosphorylation were quantified by densitometry and normalized with total Akt. The blots and relative intensity of p-Aktser473 and p-Aktthe308 proteins shown above them illustrate higher levels of phosphorylated Akt among biomass users. All results are representative of one of four independent experiments with comparable results.
ROS and SOD Levels
Flow cytometric analysis showed a significant rise in MFI DCF-DA among BMF users, suggesting increase in ROS generation in peripheral blood leukocytes (granulocytes, monocytes, and lymphocytes) and un-fractionated sputum cells that contained alveolar macrophages, epithelial cells, airway neutrophils, eosinophils, and lymphocytes (Figure 6A). The MFI of DCF-DA was 36% higher in peripheral blood granulocytes (572.5 ± 136.5 vs. 420.6 ± 143.3 in control, p < .0001), 25% higher in monocytes (306.4 ± 124.8 vs. 245.6 ± 102.5, p = .0006), and 14% higher in lymphocytes (179.4 ± 91.1 vs. 157.5 ± 72.7, p = .0836) of biomass users, relative to controls. ROS generation was doubled in sputum cells of biomass users when compared with that of control (MFI 708.8 ± 274.9 vs. 346.4 ± 188.1, p < .0001). On the other hand, biomass-using women had 39% and 28% lower levels of SOD in erythrocytes (156.8 ± 65.2 vs. 255.1 ± 59.3 U/ml in control, p < .0001) and sputum cells (565.3 ± 115.2 vs. 785.4 ± 183.8 U/ml, p < .0001), respectively, in comparison with control (Figure 6B).
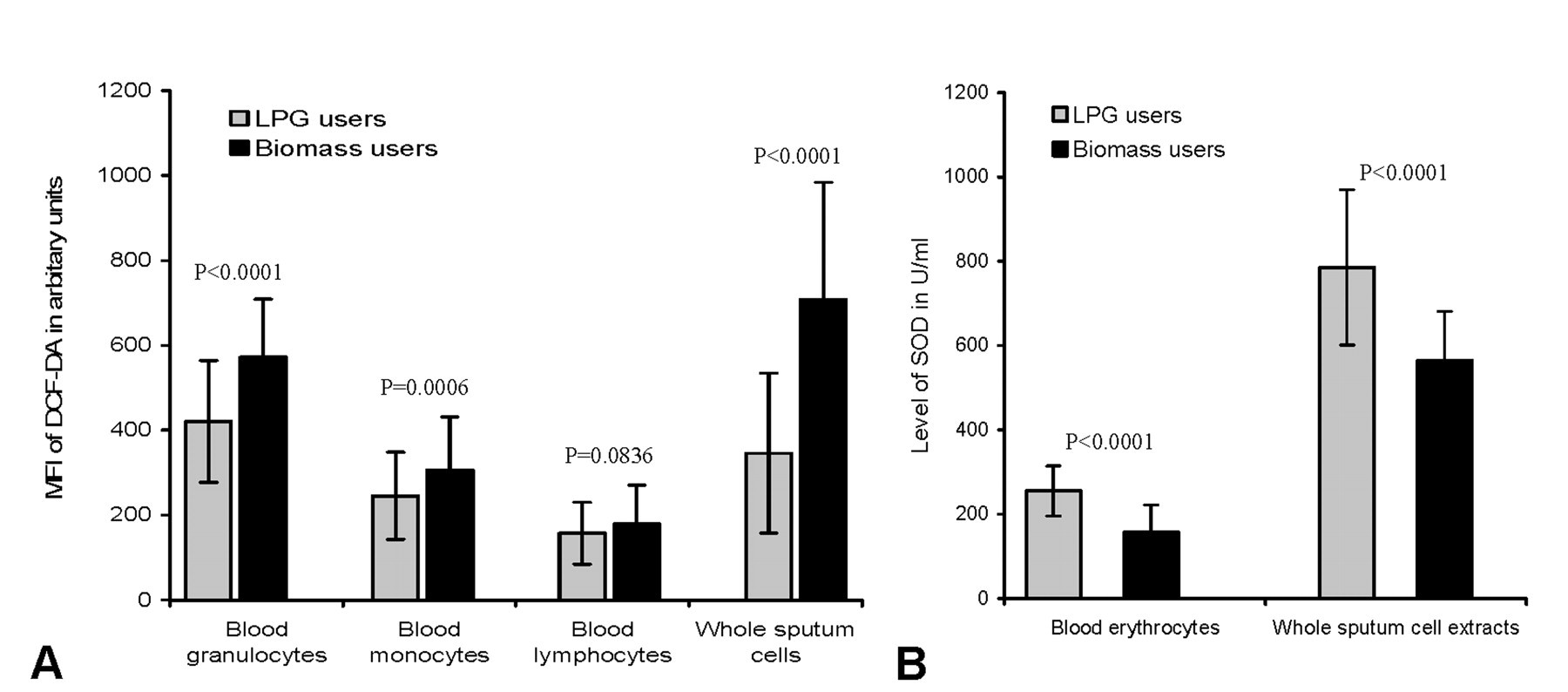
Histograms showing higher levels of reactive oxygen species (ROS) generation in peripheral blood leukocytes and sputum cells (A) and lower levels of superoxide dismutase (SOD) enzyme in erythrocytes and sputum cells (B) of biomass-using women in comparison with LPG-using control. Bars represent standard deviation of mean.
Controlling age, family income, and position of the kitchen as possible confounders, multivariate logistic regression analysis indicated a strong positive association between MFI of DCF-DA and level of PM10 (OR = 1.25, 95% CI 1.07–1.45) and PM2.5 (OR = 1.51, 95% CI 1.20–1.93).
ROS, SOD, and Akt Activation
Figure 7 demonstrates progressive rise in the percentages of p-Aktser473-positive AEC and PBL in relation to years of cooking with BMF. Cooking years correlated positively with generation of ROS in AEC, in terms of MFI of DCF-DA (Figure 7a), and negatively with concentration of SOD in erythrocytes (Figure 7b). Highest percentages of p-Aktser473-positive AEC (22.7%) and PBL (40.1%) were recorded in women having longest duration of cooking with biomass (20–24 yrs). Women in this exposure category showed greatest elevation in ROS (DCF-DA MFI 743.50) and maximum depletion of erythrocyte SOD (135 U/ml).
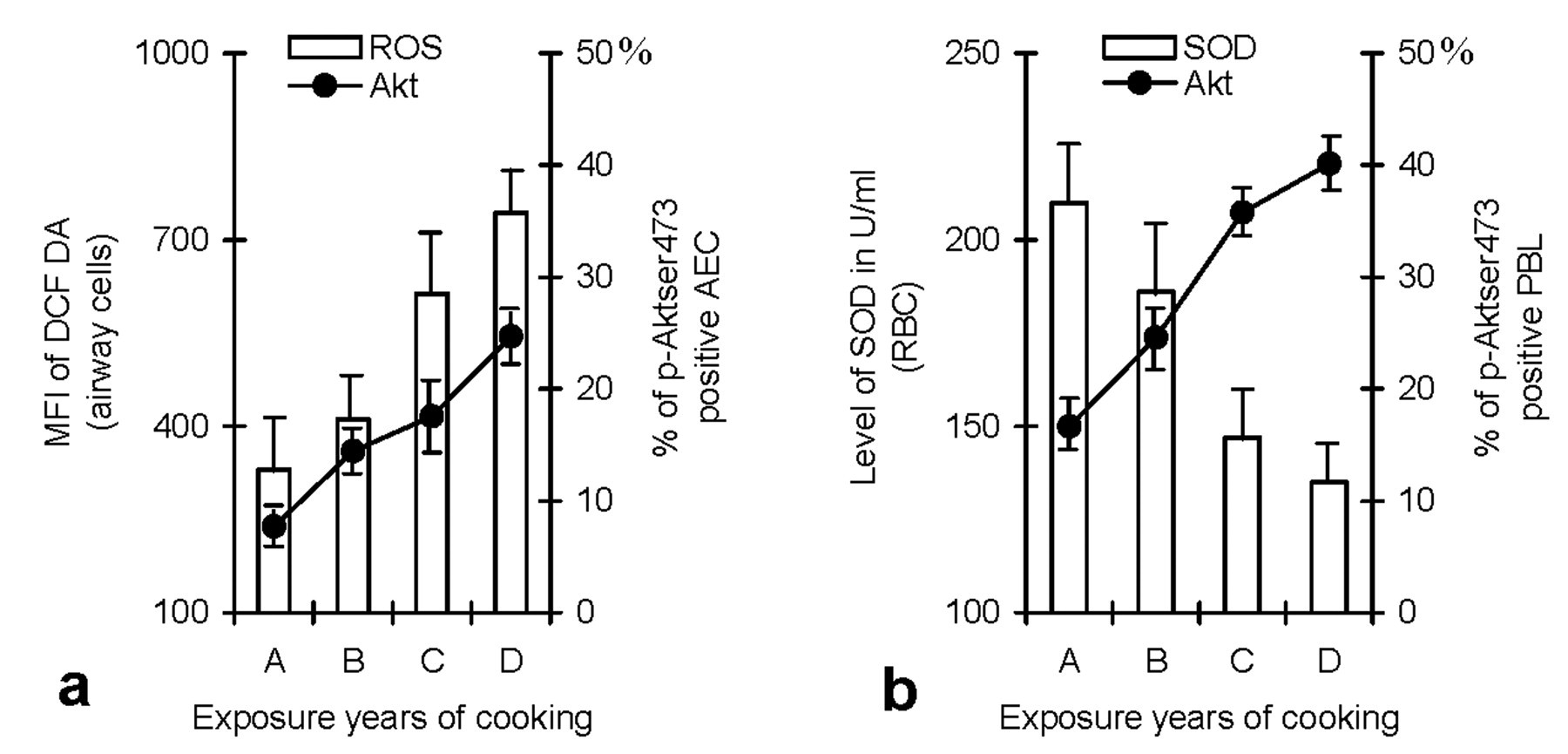
Histograms showing close relationship between years of cooking with BMF, ROS, and SOD levels, and activation (phosphorylation) of Akt. Increasing cooking years were associated with progressive rise in the percentage of p-Aktser473-expressing AEC (a) and circulating lymphocytes (b). The changes were paralleled by increase in ROS generation in airway cells (a) and depletion of SOD in erythrocytes (b). A, 5–9; B, 10–14; C, 15–19; and D, 20–24 years of cooking experience. Bars represent SD.
Discussion
The objective of this study was to examine whether cumulative inhalation of biomass smoke causes activation of the proto-oncogene Akt in those cells that are at the direct route of exposure to inhaled pollutant. We studied Akt activity also in PBL, because biomass smoke contains high concentration of ultrafine particles (UFPs; particles having aerodynamic diameter of less than 0.1 µm) that can cross the alveolar-capillary barrier and then circulate throughout the body, causing systemic damage through generation of oxidative stress (Bräuner et al. 2007). We found marked increase in the percentages of p-Aktser473- and p-Aktthr308-positive airway cells and PBL among BMF users, suggesting up-regulation of Akt activity in these cells. Akt is one of the key regulators of cell proliferation and death. It prevents apoptosis, and this effect is mediated by its influence on downstream targets including forkhead proteins (Datta et al. 1997), BclXL (Jones et al. 2000), NF-κB (Madrid et al. 2000), Bad (Datta et al. 1997), caspase-8 (Jones et al. 2002), and glycogen synthase kinase (Brazil, Park, and Hemmings 2002). Transcription factor FOXO 3A exerts tumor-suppressive functions by inducing transcription of p27CIP1 (Di Cristofano et al. 2001); and p-Akt phosphorylates this transcription factor, causing its functional inactivation and export from the nucleus (Brunet et al. 1999). Apoptosis of T and B-lymphocytes were found inhibited in transgenic mice that overexpressed active Akt (Bommhardt et al. 2004). Conversely, inhibition of PI3K-Akt signaling pathway induced apoptosis in rat hepatic stellate cells, even under strong mitotic stimulation by platelet-derived growth factor (Wang et al. 2008).
The participants of this study were never-smokers and nonchewers of tobacco. Moreover, exposure to environmental tobacco smoke for the presence of smoker in the family was similar among BMF and LPG users. Therefore, greater expression of p-Akt in BMF-using women cannot be explained by tobacco use. The villages where the participants resided were far from the highways and busy road traffic. Bicycle and cycle rickshaw were the principal mode of transport, and there were no air-polluting industries within 5 km radius. Thus, ambient air pollution levels in the study areas seemed negligible. Besides, the BMF and LPG users were neighbors; hence the impact of outdoor air pollution was similar in these two groups. The major difference between these two groups was significantly higher exposure to particulate air pollution among BMF users, and this could explain to a large extent the observed increase in Akt activation. There has been progressive increase in the percentages of p-Akt-expressing cells in association with increasing cooking years. However, we found a strong correlation between age and years of cooking with biomass (r = .0094) or LPG (r = .0092). Thus, age appears to be an important contributor to higher p-Akt expression among women with longer cooking years. In conformity with this, p-Akt expression has been found significantly higher in colorectal carcinoma patients aged 60 years or more when compared with younger patients (Khor et al. 2004).
Some studies have shown Akt activation in association with environmental carcinogenesis (West et al. 2003). Since biomass smoke contains a host of mutagens and carcinogens including benzo(a)pyrene, 1,3-butadiene, and benzene (Wafula et al. 1990; Zhang and Smith 1996), it is possible that increased expression of p-Akt was mediated by these mutagens. Alternatively, biomass smoke can activate Akt via generation of oxidative stress, as ROS can activate Akt (Esposito et al. 2003). This appears to be a distinct possibility as we found rise in ROS and concomitant depletion of SOD, suggesting oxidative stress among BMF users. Like the present finding, depletion of antioxidant enzymes and enhancement of lipid peroxidation have been reported in Turkish women who cooked with BMF (Gani et al. 2000). Even short-term exposure to biomass smoke has been shown to reduce plasma antioxidant levels in laboratory animals (Sezer et al. 2006).
AMs are the first line of cellular defense against inhaled pollutants in the lung. Human AM plays a critical role in host defense and in the development of inflammation and fibrosis in the lung. Proliferation of respiratory epithelium, increase in the number of AM, emphysematous changes, and bronchoalveolar hemorrhage have been observed in rabbits exposed to dung cake smoke (Fidan et al. 2006). In line with these observations, we found increased number of AM and inflammatory cells in expectorated sputum of BMF-using women, suggesting airway inflammation. AMs have constitutive Akt activation, and the cells are deficient in PTEN (Flaherty, Monick, and Hinde 2006). This could be a molecular adaptation to survive for prolonged periods of time in vivo as the estimated turnover time of AM in humans is 81 days (Thomas et al. 1976). Akt is involved in the signal transduction pathway mediating delay of neutrophil apoptosis induced by inflammatory mediators (Rane and Klein 2009), and its inhibition was found to enhance neutrophil death. Conversely, neutrophils with increased Akt activity have a prolonged lifespan (Zhu et al. 2006). Therefore, Akt activation in AM and airway neutrophils, as observed in BMF users, may give these cells survival advantage and perpetuate inflammation. The net effect could be respiratory impairment. In agreement with this, BMF users of this study had greater prevalence of upper (sore throat, sneezing, running nose) and lower respiratory symptoms (wet and dry cough, wheeze, chest tightness); higher prevalence of chronic obstructive pulmonary disease (COPD); and significant decline in pulmonary function especially in forced vital capacity (FVC), forced expiratory volume in one second (FEV1), FEV1/FVC ratio, and mid expiratory flow rate (FEF25-75%) when compared with their neighbors who used to cook with LPG (data not shown).
The major strengths of the present study are the identified human cohorts and their focused and detailed examination of Akt activity in differentiated airway cells and PBLs. However, the study has its limitations. First, we have only partially characterized indoor air pollution by measuring PM10 and PM2.5 levels in cooking areas, leaving emission of gaseous pollutants such as oxides of nitrogen and carbon monoxide and their possible impact on Akt expression undetected. Second, we have no data on personal exposure of these pollutants. Third, the study did not attempt to identify potential toxic components responsible for these biological changes. For example, we did not carry out physicochemical analyses of PM10 and PM2.5 such as level of polycyclic aromatic hydrocarbons including benzo(a)pyrene, elemental carbon, organic carbon, particle size, and endotoxin content. Endotoxin seems important because it elicits inflammation and Akt activation, and airborne endotoxin concentrations in biomass, burning homes of Malawi and Nepal have been found significantly higher than those found in homes in developed countries (Semple et al. 2010). Fourth, we carried out indoor measurements and biological sampling for a limited number of days. Therefore, seasonal variation and climatic changes could have influenced the measured parameters. However, monitoring of PM was done in LPG- and biomass-using homes of a village simultaneously, as was sampling of sputa and blood. Thus, seasonal effect does not appear to be a significant confounder. The participants of this study who cook regularly were simultaneously exposed to cooking oil fumes and cooking fuel emissions. Therefore, up-regulation of Akt can be attributed in part to cooking-oil fume, as it is capable of inducing up-regulation of p-Akt in bronchial cells (Hung et al. 2007). However, both LPG and biomass users of this study were exclusive users of mustard oil as cooking medium, and the type of cooking and food habit was similar in these two groups. Therefore, cooking-oil fume exposure appeared to be similar in both groups, excluding this as a contributor to Akt activation among BMF users.
In essence, the study has shown up-regulation of Akt activity in women who used to cook with BMF and are therefore highly exposed to IAP. p-Akt was detectable in AEC of control women also. This is consistent with the report that showed p-Akt expression in 27.3% of normal human bronchial biopsy specimens (Tsao et al. 2003). Biomass users of this study had significant rise in the prevalence of metaplasia and dysplasia among AEC. This can be one of the reasons of high p-Akt activity in exfoliated epithelial cells in biomass users, because p-Akt expression has been found markedly increased in metaplasia and dysplasia of AEC, particularly the latter (Tsao et al. 2003). Elevated p-Akt activity has been demonstrated in metaplastic and dysplastic areas of the bronchial epithelium of patients with lung cancer, whereas normal and hyperplastic bronchial epithelia of these patients exhibited little or no activity (Balsara et al. 2004). Compared with metaplastic and dysplastic cells that were 44–88% positive for p-Akt, Tsao et al. (2003) found that only 33% of non-small cell lung cancer specimens expressed p-Akt. It was hypothesized that Akt activation is an early and frequent event in lung cancer development (Tsao et al. 2003; Balsara et al. 2004). In support of this, pharmacological inhibition of PI3K and consequent down-regulation of Akt phosphorylation has been shown to inhibit premalignant and malignant growth in human bronchial epithelium (Chun et al. 2003). In view of these reports, overexpression of phosphorylated (active) Akt in exfoliated airway cells of biomass users indicate greater risk of bronchogenic carcinoma in these women. Indeed, chronic exposures to biomass smoke have been implicated as a major risk factor for lung cancer among women in India (Smith 2000; Smith and Mehta 2003; Behera and Balamugesh 2005). Currently, lung cancer is the fifth leading site of cancer among nonsmoking women in eastern India, where this study was conducted (Nandakumar et al. 2004). Since millions of rural women of the country are exposed to IAP from biomass smoke, the present findings have public health relevance and warrant immediate measures to reduce IAP from BMF use.
Footnotes
Acknowledgments
The authors gratefully acknowledge the financial support received from Central Pollution Control Board, Government of India, Delhi, in carrying out this study.
