Abstract
Itai-itai disease (IID) of humans is one of the most severe forms of chronic cadmium (Cd) intoxication. Itai-itai disease occurs mainly in post-menopausal women and is characterized by osteoporosis with osteomalacia, renal tubular disorder, and renal anemia. Some researchers insist the major cause of IID is not Cd, but rather malnutrition, especially hypovitaminosis D. We administrated a low concentration of Cd chloride intravenously to ovariectomized female rats that were fed a vitamin D–deficient diet or a normal diet for fifty weeks. The vitamin D–deficient diet decreased serum concentration of vitamin D, but it did not affect the metabolism of the kidney or bone. Cadmium treatment alone induced a decrease in serum concentration of vitamin D, as well as renal dysfunction, renal anemia, and abnormal bone metabolism. Osteoporosis with osteomalacia, tubular nephropathy, fibrous osteodystrophy, and bone marrow hyperplasia occurred following Cd treatment. In rats treated with Cd and administered a vitamin D–deficient diet, the toxic effects of Cd on kidney, bone, and hematopoiesis were enhanced in comparison to rats treated with Cd and a normal diet. The present experiment demonstrated that hypovitaminosis D did not evoke morphologic features of IID in humans but did enhance Cd-induced toxicity in the rat model of this disease.
Introduction
Cadmium (Cd) is one of the most toxic heavy metals, and it coexists ubiquitously with zinc, copper, and lead in the environment (WHO/IPCS 1992). Environmental and occupational exposure to this metal causes renal and bone toxicity, as well as respiratory difficulty and some types of cancer (Järup, Berglund et al. 1998). For many years, Cd has been thought to affect the body at high-level exposure only in Cd-polluted areas (Kawamura et al. 1978; Kjellström, 1992). However, recent surveillance has shown that low to moderate exposure to Cd can cause renal damage or bone disease in polluted areas of many countries, including Japan, Sweden, Belgium, Poland, and China (Alfvén et al. 2000; Berglund et al. 2000; Buchet et al. 1990; Cai et al. 1998; Hu 2000; Ikeda et al. 2000; Järup, Alfvén, et al. 1998; Järup, Berglund, et al. 1998; Nordberg et al. 1997).
Itai-itai disease (IID, or “Ouch-ouch disease” in English) of humans is one of the most severe forms of chronic Cd intoxication. The disease occurs mainly in post-menopausal women and is characterized by osteoporosis with osteomalacia, renal tubular disorder, and renal anemia (Blainey et al. 1980; Kazantzis 2004; Nogawa et al. 1987). Some researchers insist that the major cause of IID is not Cd, but rather malnutrition, especially hypovitaminosis D (Takeuchi 1973). Many researchers have tried to reproduce histopathological features characteristic of IID in various animals by chronic Cd exposure (Furuta 1978; Hamada et al. 1991; Itokawa et al. 1974; Kajikawa et al. 1981; Kawashima et al. 1988; Ogoshi et al. 1989), but few of them were successful in reproducing osteoporosis with osteomalacia characteristic to IID. In our previous report (Hiratsuka et al. 1996), osteoporosis with osteomalacia, tubular nephropathy, and renal anemia were induced in ovariectomized (OX) female rats by i.v. injections of Cd for fifty weeks without the involvement of malnutrition. In the present study, the effects of hypovitaminosis D on the rat model was examined to determine whether hypovitaminosis D could be the major cause of IID in humans or one of the enhancing factors of Cd toxicity.
Materials and Methods
Experimental Animals and Treatment
In total, eighty female Sprague Dawley rats (Crj: CD (SD) IGS, Charles River Japan, Kanagawa, Japan) were obtained at six weeks of age, and the ovaries were completely excised bilaterally under anesthesia with sodium pentobarbital (50 mg/kg i.p.). At ovariectomy, the body weights ranged from 164.9 to 199.1 g. Complete disappearance of the estrous cycle was confirmed by vaginal smear testing in all animals. Three weeks later, the rats were randomly assigned to four groups, and the administration began at ten weeks of age. Group composition was as follows: (1) saline injection and normal diet (Saline + Vit. D); (2) saline injection and vitamin D–deficient diet (Saline - Vit. D); (3) CdCl2 injection and normal diet (Cd + Vit. D); and (4) CdCl2 injection and vitamin D–deficient diet (Cd - Vit. D). The saline and CdCl2 injection group consisted of eighteen and twenty rats, respectively. The + Vit. D groups were given the diet (Oriental Yeast Co., Tokyo, Japan) containing 3 IU/g vitamin D (cholecalciferol), and - Vit. D groups were given the diet (Oriental Yeast Co.) containing < 0.3 IU/g vitamin D. All the animals were allowed to drink well water admixed with NaClO (about 2 ppm) from automatic dispensers ad libitum. The body weights at the initiation of administration ranged from 297.0 to 360.2 g. The animals were individually housed in stainless steel wire cages (W × H × D: 260 × 200 × 380 mm) in a barrier system area. The animal rooms were thermostatically controlled at 21°–27°C and maintained at a relative humidity of 35–75%, with a twelve-hour light/dark cycle (lighting from 7:00 AM to 7:00 PM). The rooms were ventilated thirteen to fifteen times/hour. This study was conducted in accordance with all relevant guidelines concerning experimental animals and animal welfare set by Japanese government and the testing facility.
Experimental Design
The rats were injected into the tail vein with saline (1.0 ml/kg) or CdCl2 dissolved in saline (0.5 mg/kg) five days a week for up to fifty weeks. The dose of Cd (as CdCl2) was decided by referring to the results of previous thirteen- and fifty-week administration experiments (Hiratsuka et al. 1996; Katsuta et al. 1994). Individual dose level was adjusted weekly based on the most recently recorded individual body weights. Prior to the necropsy at twenty-four and fifty weeks, all surviving rats (three to six rats/group) were deeply anesthetized with sodium pentobarbital and exsanguinated from the posterior vena cava.
Assays for Serum Vitamin D
At necropsy, a blood sample was obtained from the posterior vena cava for hematology, blood chemistry, and assays for serum vitamin D and/or bone metabolic markers. At week 50, serum concentrations of 1,25-dihydoroxycholecalciferol (1,25[OH]2D3) and 25-hydoroxycholecalciferol (25[OH]D) were determined using commercially available enzyme-linked immunosorbent assay (ELISA) kits (Immundiagnostik AG, Germany) and a microplate reader (SpectraFluor; Tecan, Salzburg, Austria).
Urinalysis
At sixteen, twenty-four, and fifty weeks, four to six rats in each group were housed in individual metabolism cages for about sixteen hours and urine was collected. Total urine volume was recorded and aliquots of urine were frozen for determination of calcium (Ca; methylxylenol blue method, Wako Pure Chemical Industries, Ltd., Osaka, Japan), inorganic phosphorus (IP; purine-nucleoside phosphorylasexanthine oxidase method, Wako), N-acetyl-β-D-glucosaminidase (NAG; enzyme method, Kyowa Medics Co., Ltd., Tokyo, Japan), β2-microglobulin (ELISA method, Panapharm Laboratories Co., Ltd., Japan), pyridinoline (Pyd; Sumitomo Seiyaku Biomedical Co., Ltd., Japan), and deoxypyridinoline (Dpd; Sumitomo Seiyaku Biomedical Co., Ltd.). β2-microglobulin, Pyd and Dpd were assayed using a SpectraFluor microplate reader. Ca, IP, and NAG were assayed using an auto analyzer (7170; Hitachi Ltd., Japan).
Hematology and Blood Chemistry
The hematology parameters evaluated were erythrocyte count (RBC; laser optical method), hematocrit (Ht; [RBC × MCV]/103), hemoglobin concentration (HB conc.; cyanic methemoglobin method), mean corpuscular volume (MCV; laser optical method), mean corpuscular hemoglobin (MCH; calculated from HB conc. and RBC), mean corpuscular hemoglobin concentration (MCHC; calculated from HB conc., RBC and MCV), and reticulocyte count (RNA staining laser optical method) using a hematology system (ADVIA 120; Bayer Medical Ltd., Germany). The blood chemistry parameters evaluated were asparate aminotransferase (AST; UV-kinetic method), alanine aminotransferase (ALT; UV-kinetic method), gamma glutamyltranspeptidase (γ-GT; L-γ-glutamyl-3-hydroxymethyl-4-nitroanilide reaction), alkaline phosphatase (ALP; p-nitrophenylphosphate reaction), blood urea nitrogen (BUN; urease-GlDH method), creatinine (Jaffé method), IP (PNPXOD method), Ca (MXB method), iron (Fe; bathophenanthroline direct method), unsaturated iron-binding capacity (UIBC; bathophenanthroline direct method), and total iron-binding capacity (TIBC; calculated from Fe and UIBC) using an auto analyzer (7170; Hitachi Ltd.).
Assays for Bone Metabolic Markers
Serum concentrations of parathyroid hormone (PTH), calcitonin, and osteocalcin (bone glaprotein, BGP) were determined using commercially available kits (Immutopics, Inc., San Clemente, CA, USA), the immunoradiometric (IRMA) assay method, and a Gamma counter (COBRA Quantum 5005; PerkinElmer, Inc., USA).
Histopathology
The liver, kidneys, and parathyroid glands were collected at necropsy from all surviving animals. These organs and tissues were fixed in 10% neutral-buffered formalin, embedded in paraffin wax, sectioned, and stained with hematoxylin and eosin (HE). In addition, the right femurs and thoracic vertebrae of the animals were fixed for two days in 0.5% cyanuric chloride in methanol containing 1% (0.1 M) N-methylmorpholine (Yoshiki 1973), decalcified in 10% formic acid formalin, and were processed routinely for HE staining.
Bone Mineral Density
Bone mineral density (BMD) of the left femur (proximal, middle, and distal portions) was measured by dual energy x-ray absorptiometry (DCS-600; ALOKA CO., Ltd., Tokyo, Japan).
Statistical Analysis
Values obtained from rats in the Saline - Vit. D group and Cd treatment groups were compared with those from the Saline + Vit. D group. Bartlett’s test was used to compare the variances among the groups. If the groups were accepted to be homogeneous, Dunnett’s multiple comparison test was used for comparison of the groups. If the data of each group were heterogeneous in Bartlett’s test, Steel’s multiple comparison test was used. The Wilcoxon test was used for the comparison of semiquantitated histopathological findings.
Results
Body Weight
Body weight gain consistently decreased in the Cd-treated groups from five to fifty weeks, and the body weight of the Cd-treated rats was significantly low from thirteen weeks (Figure 1). There were no clear effects of vitamin D deficiency on the body weight change throughout the administration period.
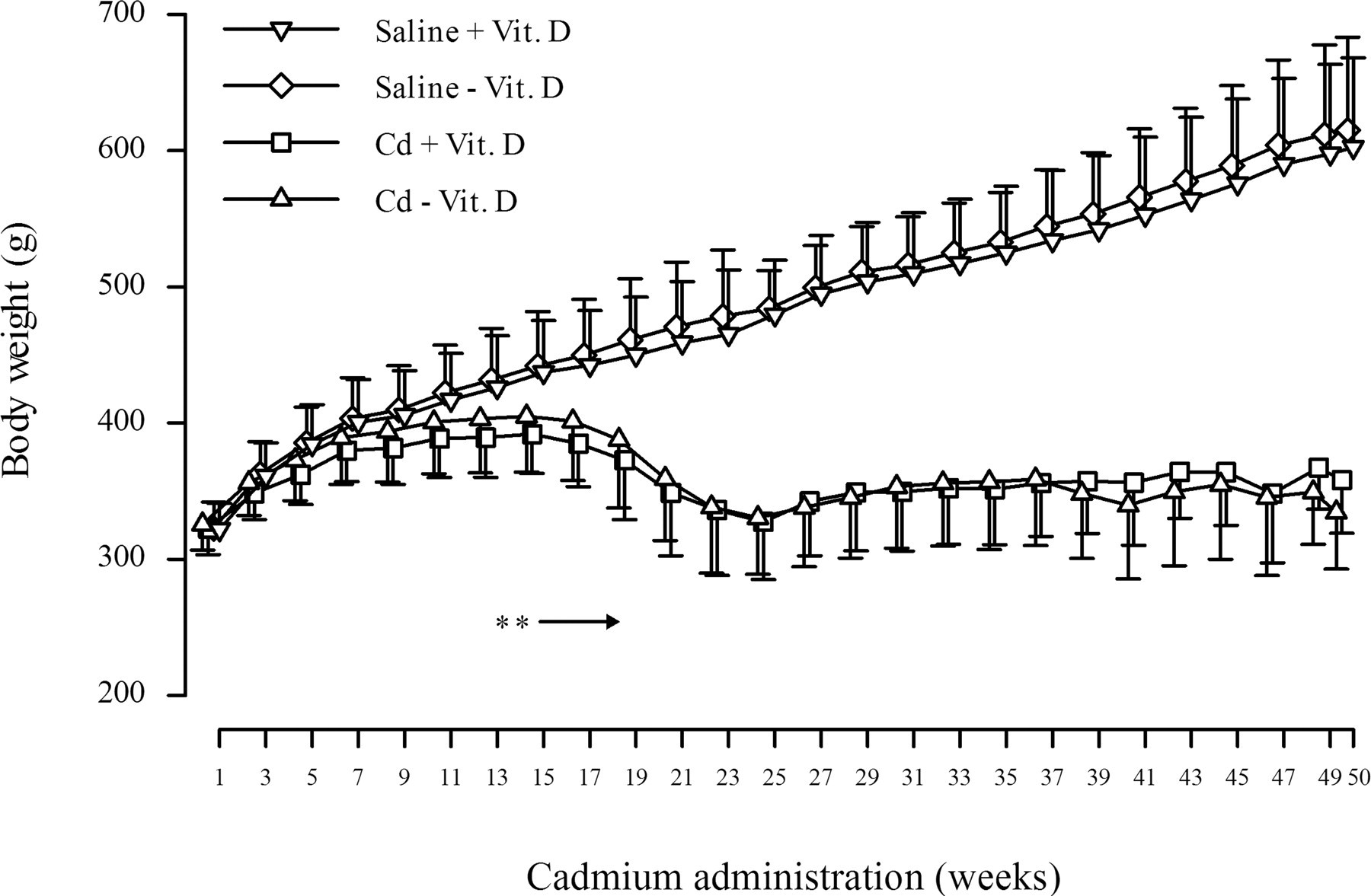
Body weight changes of the rats treated with cadmium and/or vitamin D deficiency. Each point represents the mean ± SD (n = 12 at weeks 1 to 23, n = 3, 5, or 6 at weeks 25 to 50). Asterisk (*) indicates significant difference from the saline + Vit. D group (p < .01).
Concentration of Serum Vitamin D
In the Saline - Vit. D group, serum levels of 1,25(OH)2D3 and 25(OH)D were significantly decreased, and a decrease of 25(OH)D was more remarkable (Table 1). In contrast, Cd treatment alone induced decreases of 1,25(OH)2D3 to the same extent. The decrease of vitamin D in the Cd - Vit. D group was the sum of the effects of Cd treatment and vitamin D deficiency, and the serum concentration of 1,25(OH)2D3 was higher than that of 25(OH)D.
Serum vitamin D concentrations of the rats treated with cadmium and/or vitamin D deficiency for fifty weeks.
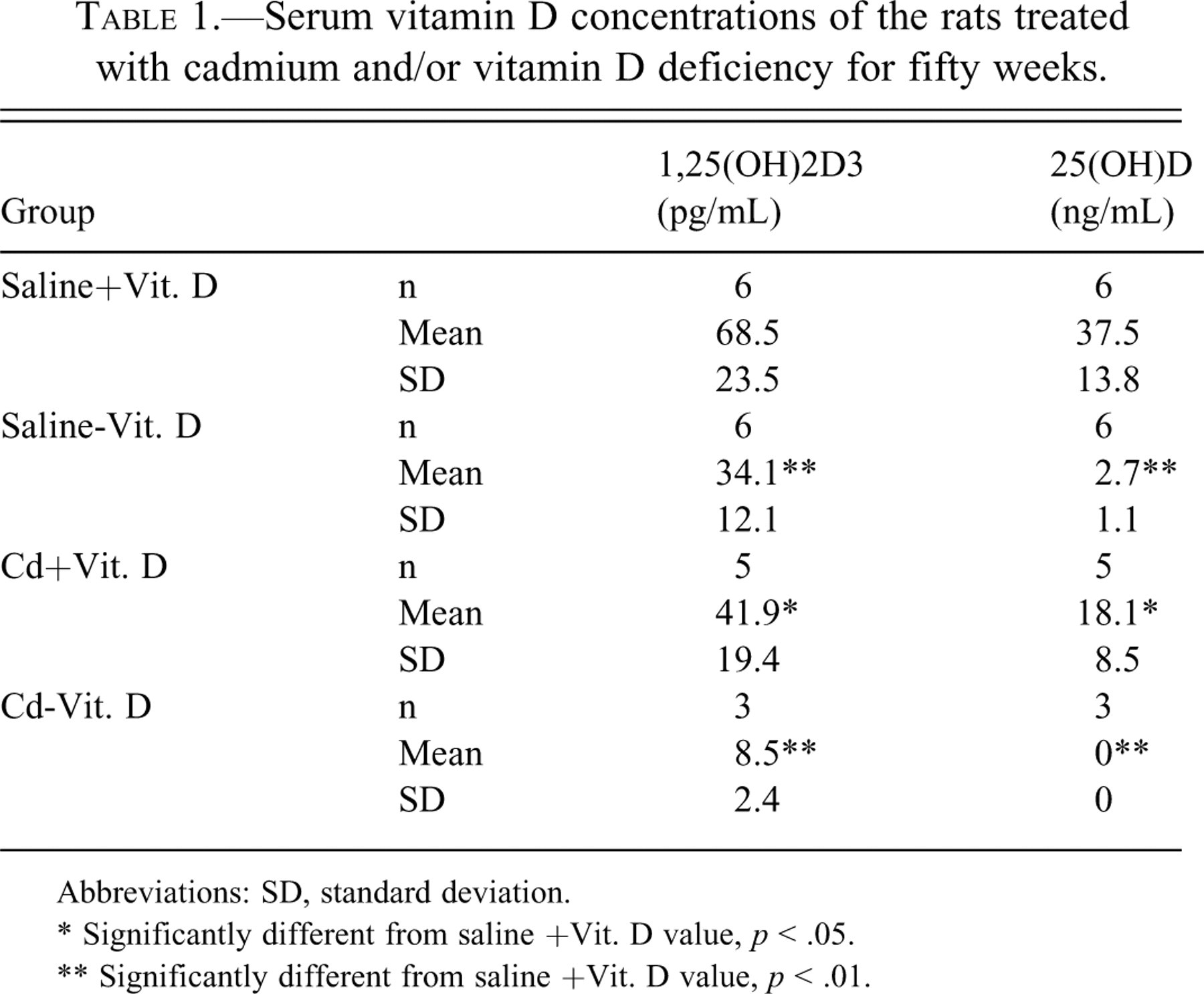
Abbreviations: SD, standard deviation.
* Significantly different from saline +Vit. D value, p < .05.
** Significantly different from saline +Vit. D value, p < .01.
Urinalysis
β2-microglobulin significantly increased in the Cd-treated groups from sixteen to fifty weeks. Although an elevation of NAG was observed from sixteen weeks in the groups, the change disappeared at week 50. Urinary excretion of Ca in Cd-treated rats tended to decrease or significantly decreased at weeks 16 and 24, respectively, and then changed to an increase at week 50. Excretions of the bone metabolic markers, such as Pyd and Dpd, slightly increased at week 16 in the Cd-treated groups, and the excretion of Pyd continuously increased at weeks 24 and 50. The Saline - Vit. D group showed no urinary changes throughout the administration period.
Hematology
The Cd-treated groups revealed significant decreases in HB conc., Ht, MCV, MCH, and MCHC, and an increase in reticulocyte count (Table 2 ). At week 50, these changes became more prominent, except for MCV, MCH, and MCHC. The degree of anemia at week 50 was more severe in the Cd - Vit. D group than in the Cd + Vit. D group. None of these changes appeared in the Saline - Vit. D group during the administration period.
Hematology data of the rats treated with cadmium and/or vitamin D deficiency for twenty-four and fifty weeks.
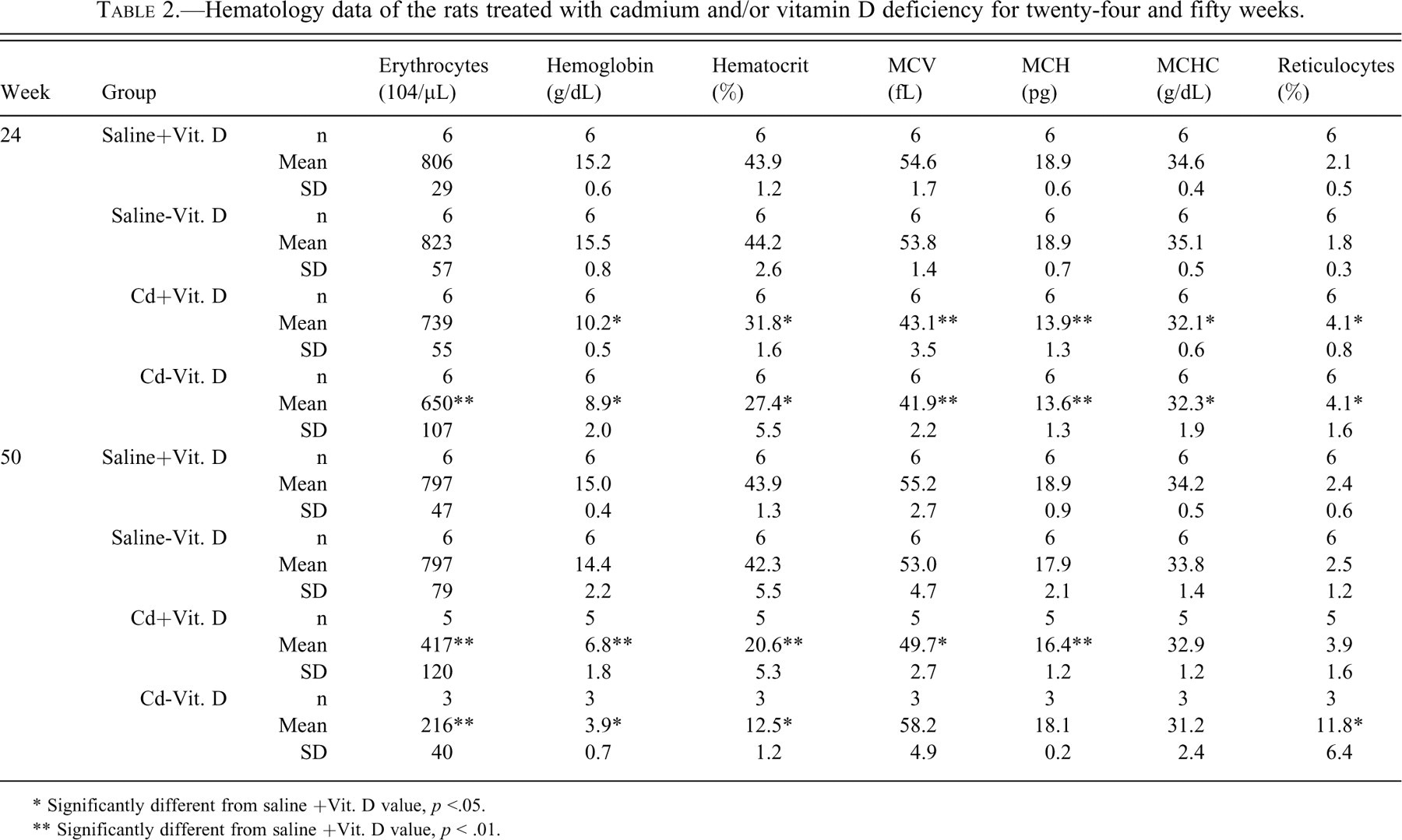
* Significantly different from saline +Vit. D value, p <.05.
** Significantly different from saline +Vit. D value, p < .01.
Blood Chemistry
Serum concentrations of BUN, creatinine, and IP were significantly higher in the Cd-treated groups at weeks 24 and 50. There were significant increases or a tendency to increase in serum AST, γ-GT, and ALP activities in the Cd-treated groups at weeks 24 and 50. In addition, Cd treatment resulted in significant increases of UIBC and TIBC concentrations, which were accompanied by a decrease in Fe concentration at weeks 24 and 50. There were no clear influences of vitamin D deficiency in these parameters.
Bone Metabolic Markers
No treatment-related changes were observed in the Saline - Vit. D group. As for the Cd-treated groups, serum levels of PTH and BGP were significantly elevated.
Gross Findings
A total of nine and eleven out of twenty rats died in the Cd + Vit. D and Cd - Vit. D groups, respectively, between week 40 and week 50. The livers and kidneys of the deceased animals were slightly atrophic and indented on their surfaces. There was no clear effect of vitamin D deficiency on the liver or kidney. The animals that survived Cd treatment also exhibited hepatic and renal changes similar to the rats that died of Cd treatment at week 24, but to a milder degree, and these changes became more severe at week 50.
Histopathology
The hepatocytes of Cd-treated rats showed an increase in individual cell necrosis and/or focal necrosis, enlargement of the cytoplasm, and nuclei with prominent nucleoi, irrespective of dietary vitamin D concentrations (Table 3). Mitosis and binucleated hepatocytes were also occasionally seen in the livers of Cd-treated rats at week 50. Cd-treated rats showed tubular nephropathy with interstitial fibrosis at weeks 24 and 50 (Figures 2a and 2b). The renal change was more severe at week 50 than at week 24 (Table 3).
Histopathologic findings of the rats treated with cadmium and/or vitamin D deficiency for twenty-four and fifty weeks.
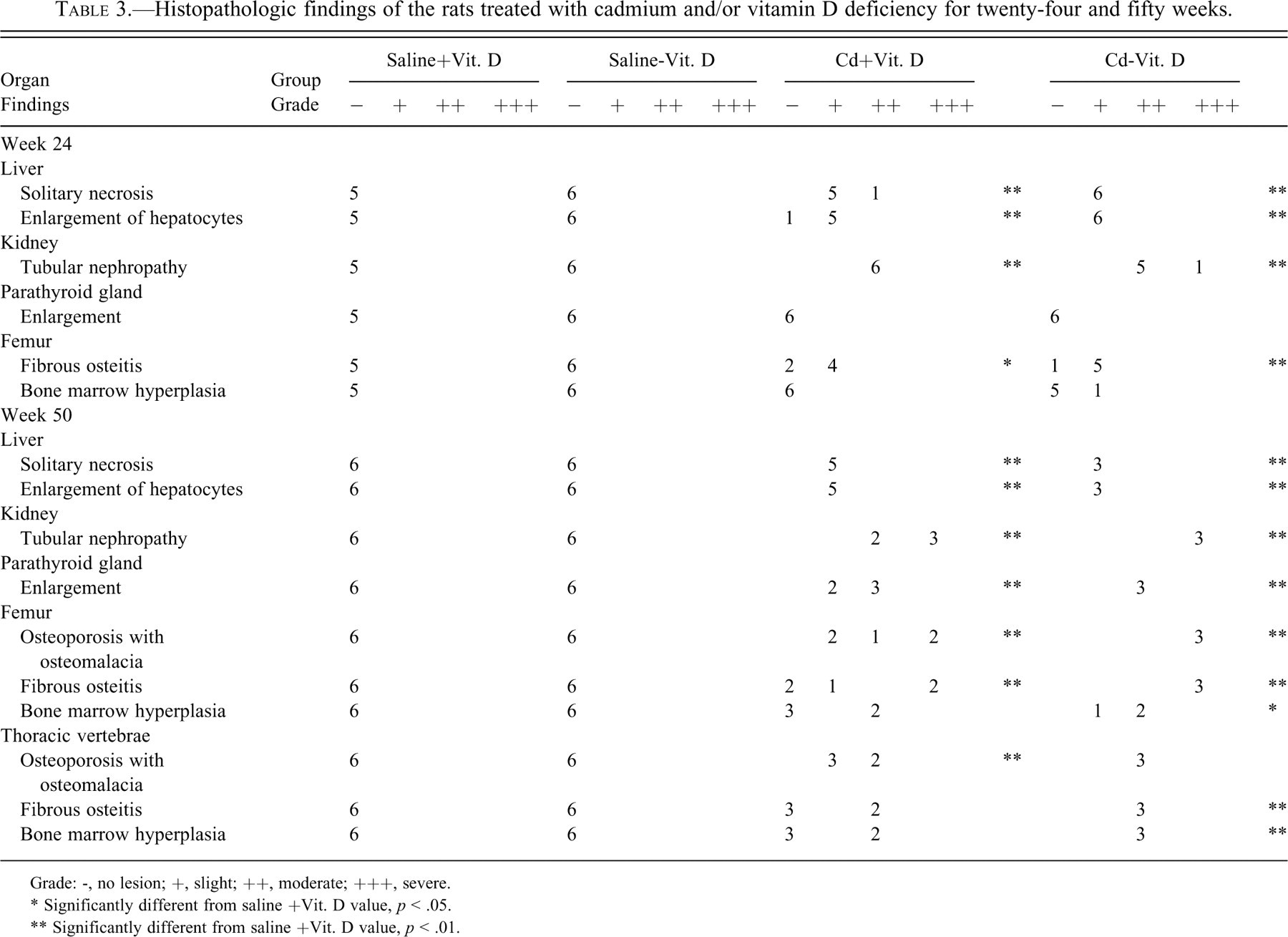
Grade: -, no lesion; +, slight; ++, moderate; +++, severe.
* Significantly different from saline +Vit. D value, p < .05.
** Significantly different from saline +Vit. D value, p < .01.
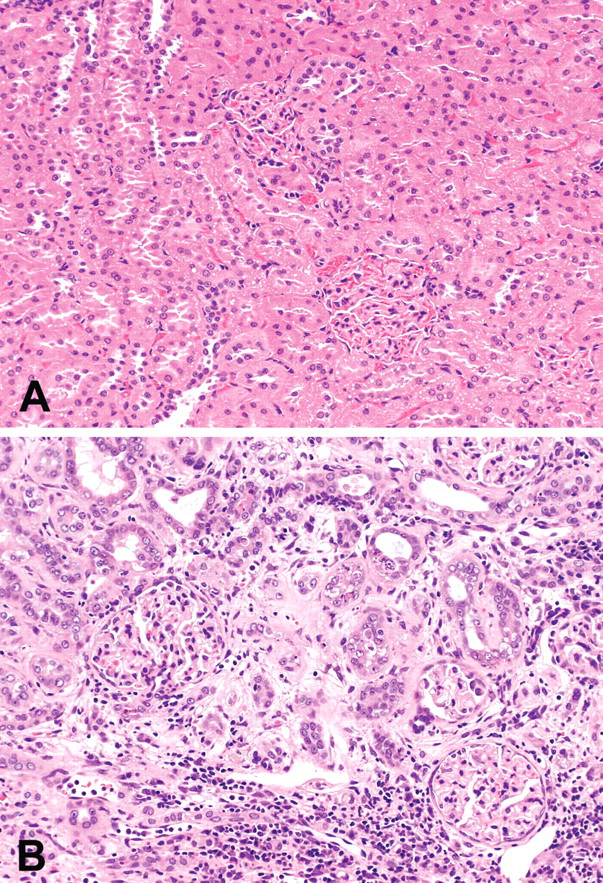
The kidney of the rats in Saline - Vit. D group (A) and Cd - Vit. D group (B) at fifty weeks of treatment. Interstitial fibrous tissues accompanying inflammatory cell infiltration fill the gaps among small-diameter tubules and the epithelial cells of the tubules are regenerative (B). Hematoxylin and eosin stain; ×200 (A, B).
At week 24, nine of twelve femurs in Cd-treated groups showed mild fibrous osteodystrophy. Bone marrow hyperplasia was observed in the femur of one rat from the Cd - Vit. D group. At week 50, the femurs and thoracic vertebrae in Cd-treated groups showed prominent fibrous osteodystrophy, osteoporosis with osteomalacia, and bone marrow hyperplasia (Figures 3a and 3b). The extent of the former two osseous changes was more severe in the Cd - Vit. D group than in the Cd + Vit. D group, and more prominent in the femur than in the vertebrae (Table 3). Cortical and cancellous bones were remarkably porotic or atrophic, and there was formation of osteoid seams at the inner surface of Haversian canals and endosteum. Fibrotic tissues proliferated within the pores, at the inner surface of the endosteum (Figure 4), and around the bone spicules of the trabecular bone at metaphysis. Microfractures of cancellous bone accompanying collagenous callus were sometimes observed at epiphysis of the femur in the Cd - Vit. D group. Bone marrow hyperplasia was prominent in all three rats of the Cd - Vit. D group and in two of the five rats in the Cd + Vit. D group. The femurs and thoracic vertebrae of non-Cd-treated groups were unremarkable at weeks 24 and 50.
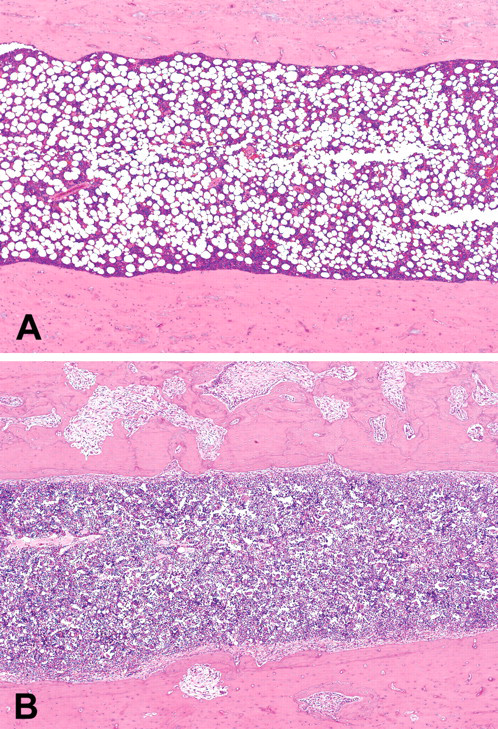
Diaphysis of the femurs of the rats in the Saline - Vit. D group (A) and Cd - Vit. D group (B) at fifty weeks of treatment. Bone marrow hyperplasia, osteoporosis, and fibrous osteodystrophy (B). Hematoxylin and eosin; ×40 (A, B).
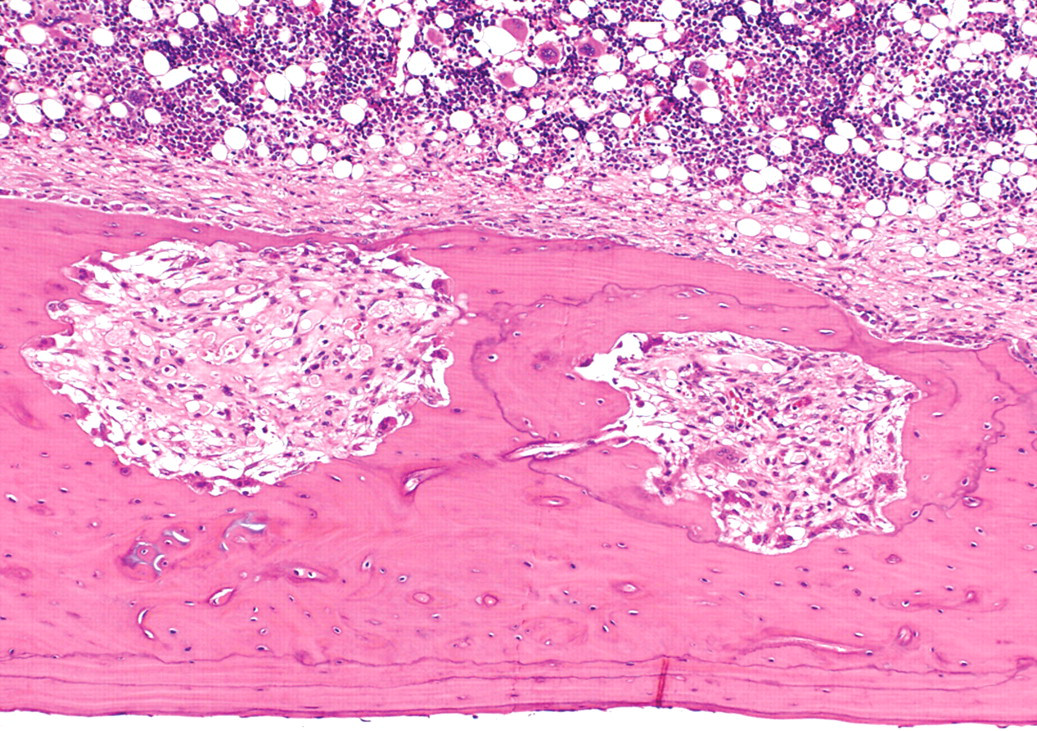
Diaphysis of the femur of a rat in the Cd - Vit. D group at fifty weeks of treatment. Fibrous osteodystrophy of cortical bone. Hematoxylin and eosin stain; ×100.
The parathyroid glands of all rats necropsied at week 24 were unremarkable. In contrast, the glands of the Cd-treated rats were slightly or moderately enlarged in comparison with those of the non-Cd–treated rats at week 50. In the enlarged glands, each lobule was enlarged owing to hypertrophy and hyperplasia of the chief cells with pale and granular cytoplasm. Mitotic figures of chief cells were occasionally observed in the enlarged parathyroid glands.
Bone Mineral Density
At week 24, the BMDs of all treatment groups were not significantly different from those of the Saline + Vit. D group. At week 50, the BMDs of the femur were significantly or insignificantly low in the Cd-treated groups (Figure 5). The osteoporosis at the middle and distal portions of the femur was slightly more severe in the Cd - Vit. D group than in the Cd + Vit. D group.
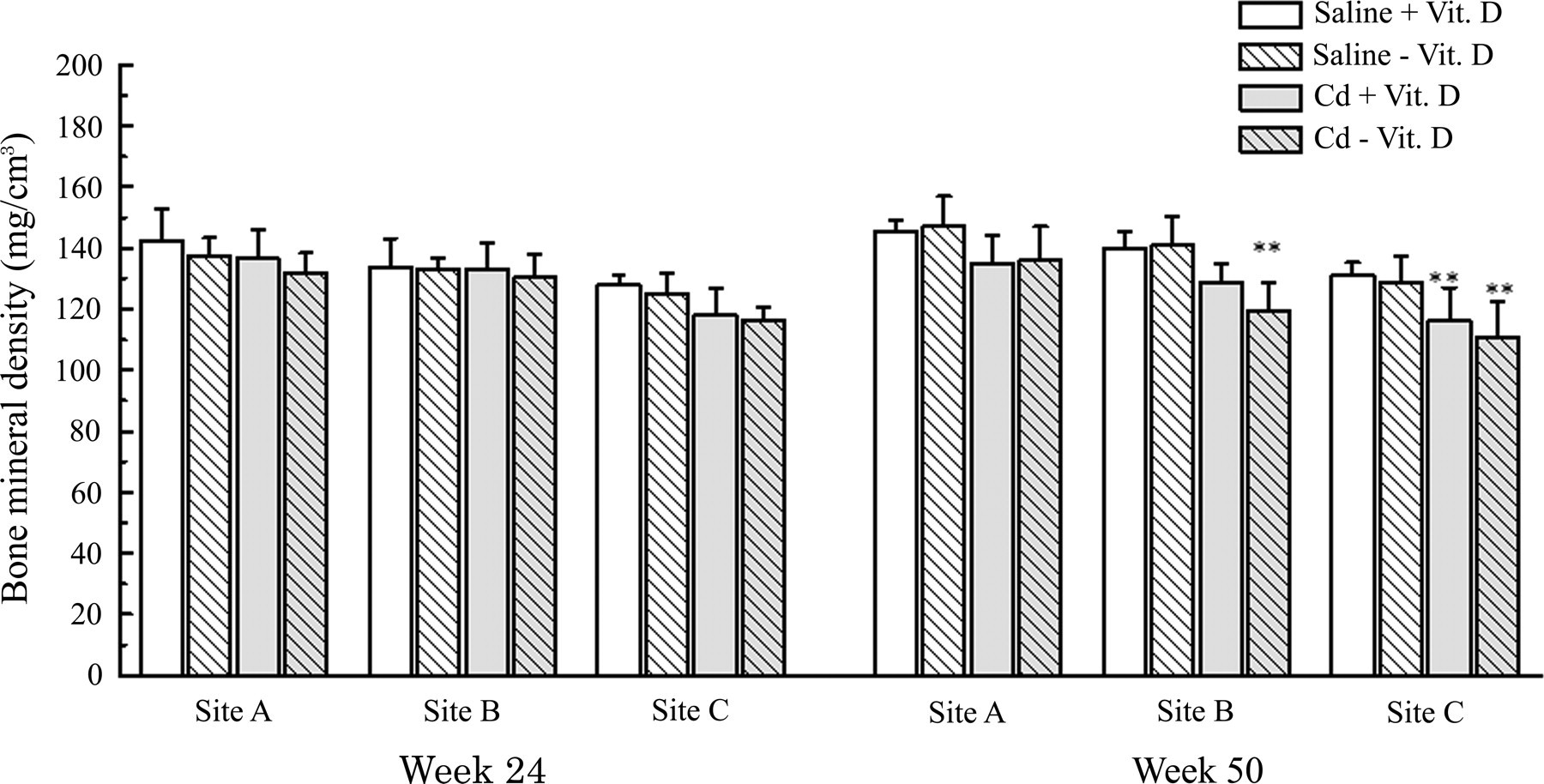
Bone mineral density of the rats treated with cadmium and/or vitamin D deficiency. Asterisk (*) indicates significant difference from the saline + Vit. D group (p < .01). Site A: 1/4 part from proximal metaphysis of the femur. Site B: Diaphysis (central part of the femur). Site C: 1/4 part from distal metaphysis of the femur.
Discussion
By repeated i.v. injection of Cd to OX female rats over fifty weeks, osteoporosis with osteomalacia, renal tubular nephropathy, and renal anemia, which were the main pathological changes of IID, could be reproduced in the present study. These results confirm our previous reports in which the characteristic pathologies of human IID were reproduced by Cd treatment for thirteen weeks (Katsuta et al. 1994) and fifty to seventy weeks (Hiratsuka et al. 1996) in OX rats. In the present study, fibrous oteodystrophy was also observed in the Cd-treated rats. Fibrous osteodystrophy is one of the osseous changes distinctive to hyperparathyroidism (Lotinun et al. 2006; Rude 1996) and may occur secondary to renal dysfunction (Amann et al. 1994; Jablonski et al. 1993).
The rats fed a vitamin D–deficient diet showed a significant decrease of active and inactive forms of serum vitamin D [1,25(OH)2D3 and 25(OH)D]. Vitamin D is an important hormone regulating homeostasis of Ca and P, and severe vitamin D deficiency is known to increase the risk of rickets (Hirota and Hirota 2006; Lips 2006). However, in the present study, the rats given a diet deficient in vitamin D did not show biochemical abnormality in blood or urine, suggesting metabolic disorders of the bone and kidneys. Vitamin D is produced in the skin through ultraviolet irradiation or obtained from food. The inactive form of vitamin D undergoes its first step of activation, namely 25-hydroxylation in the liver followed by 1α-hydroxylation in the kidneys, to make the biologically active compound 1,25(OH)2D3. The active metabolite has many functions related to Ca regulation and bone metabolism, including intestinal Ca and P absorption, Ca mobilization in the bone, and resorption of P and Ca in the renal tubular cells (Deluca 2004; Jones et al. 1998; Lips 2006). Although vitamin D interacts with PTH (Deluca 2004; Jones et al. 1998), hypovitaminosis D does not influence PTH under sufficient serum levels of Ca (Dabbagh et al. 1989; Kollenkirchen et al. 1991), and many researchers (Fox 1992; Lee et al. 1991; Masuyama et al. 2003) have reported a vitamin D–independent Ca homeostasis system. Therefore, hypovitaminosis D alone might not affect Ca homeostasis and bone remodeling owing to the vitamin D–independent system in the present experiment.
The mechanisms of Cd-induced osteoporosis with osteomalacia, which is the main pathological finding of IID, are not fully understood. The inactive form of vitamin D is activated at the proximal tubules of the kidneys, and dysfunction of the tubules occurs in the early phase of chronic Cd toxicosis (Kazantzis 2004; Kjellström 1992) and IID (Shigematsu, 1984; Takebayashi et al. 2000). Thus, the disorder of vitamin D hydroxylation in the kidneys is regarded as one of the important mechanisms of bone pathology in IID patients (Aoshima and Kasuya 1991; Kido 1995; Tsuritani et al. 1992). The rats treated with Cd showed a decreased serum 1,25(OH)2D3 concentration owing to renal tubular nephropathy, which was accompanied by a remarkable elevation of serum PTH; thus, the indirect action of Cd on bone via renal damage was demonstrated in the present experiment. Histological and roentgenological examinations of the bone showed that the fibrous osteodystrophy, osteoporosis with osteomalacia, and reduction of BMD detected in the femur were more severe in Cd - Vit. D rats than in Cd + Vit. D rats. These results suggested that dietary hypovitaminosis D enhanced Cd toxicity on the bone. Similar findings were reported by Kobayashi et al. (1979), in which osteomalacic changes were prominent in the rats subjected to long-term feeding of the diet containing Cd and low concentrations of Ca and vitamin D.
Severe anemia appeared in the Cd treatment groups after twenty-four and fifty weeks in the present experiment. The anemia at week 24 accompanied decreases of MCV and MCH, whereas the anemia at week 50 was normocytic and normochromic. These differences indicate the shift from iron-deficiency anemia at week 24 to renal anemia at week 50 owing to the advance of renal tubular destruction resulting in a poor erythropoietin response by Cd treatment (Hiratsuka et al. 1996). A diet deficient in protein, calcium, phosphorus, and fiber enhances Cd accumulation in the kidney in rats under Cd toxication (Muto and Omori 1977), and malnutrition plays an important role in the early development of Cd toxicosis, in particular for the development of renal damage. The present experiment demonstrated the aggravation of Cd-induced renal dysfunctions and renal anemia following feeding of a vitamin D–deficient diet. In conclusion, malnutrition (based on a vitamin D–deficient diet) alone does not evoke histopathological features of IID in the rodent model but enhances the Cd-related toxicity characterized by osteoporosis with osteomalacia, renal tubular nephropathy, and renal anemia.
Footnotes
Acknowledgments
The present study was supported in part by the Japanese Environmental Agency. The authors wish to thank Mr. Masahiro Nasu and Ms. Miwako Ishii for their excellent technical assistance.
