Abstract
Interaction of more than two chemicals from foods is a very important factor for carcinogenic risk assessment and management. 2-Amino-3,8-dimethylimidazo[4,5-f]quinoxaline (MeIQx), one of the most abundant carcinogenic heterocyclic amines in cooked foods, is speculated to be a human liver carcinogen. MeIQx is metabolically activated by CYP1A2 and then N-acetyltransferase (NAT), findings that suggest that its carcinogenic potential might be enhanced by simultaneous exposure to chemical(s) inducing CYP1A2. Therefore, we here investigated the effects of alpha- and beta-naphthoflavone as CYP1A2 inducers on MeIQx-induced rat hepatocarcinogenesis in a medium-term rat liver bioassay. Unexpectedly, no modifying influence of naphthoflavones on MeIQx-induced hepatocarcinogenesis was demonstrated with reference to glutathione S-transferase placental form (GST-P) positive foci in the liver, although up-regulation of CYP1A2 was detected on Western blot analysis. Activity of NAT was not affected. In MeIQx-treated rats, CYP1A expression was mainly detected in zone 3 of the liver where GST-P positive foci were preferentially located, while naphthoflavones alone or combinations of naphthoflavones and MeIQx induced CYP1A expression in zone 1. This difference in intralobular distribution of CYP1A might be related to the fact that MeIQx hepatocarcinogenesis was not modified by the two CYP1A inducers.
Introduction
Chemical interactions are important for environmental carcinogenesis with multiple chemical exposure. Most carcinogenic substances are enzymatically activated to electrophilic metabolites that react with macromolecules such as DNA, RNA, or protein. Increase of carcinogenic risk through up-regulation of activation system(s) is therefore a great concern. For example, two chemoprevention trials on lung cancer carried out in Finland (1985-1994) and the United States (1983-1996), the ATBC study and CARET, both revealed increases in lung cancer development among smokers given beta-carotene (Albanes et al. 1996; Omenn et al. 1996). Later studies performed to clarify mechanisms underlying the unexpected results revealed that the antioxidant administered as a chemopreventive agent activated cigarette-associated carcinogenic nitrosamines (Patterson et al. 1997; Al-Wadei, Takahashi, and Schuller 2006). Paradoxical effects on carcinogenesis have also been detected with phenobarbital (PB) treatment combined with other chemicals. PB was reported as a promoter on rodent hepatocarcinogenesis by Peraino, Fry, and Staffeldt (1971) but was also found capable of becoming an inhibitor of mouse hepatocarcinogenesis by Diwan et al. (1984). These phenomena were also detected in rat (Barbason et al. 1986; Natria, Watanabe, and Kitagawa 1980). Discrepancies are reported to be associated with strain differences, period of treatment, and/or carcinogenic pathways in several studies (Lee 2000; Lee, Ooasa, and Osanai 1998).
MeIQx, one of the most abundant carcinogenic heterocyclic amines in cooked foods (Wakabayashi et al. 1993), is speculated to be a human liver carcinogen (Lynch et al. 1992; Stillwell et al. 1997). Many studies have demonstrated that it is metabolically activated to genotoxic intermediates by CYP1A2-mediated N-hydroxylation in the liver followed by N-acetyltransferase (NAT)–mediated O-esterification (Kato and Yamazoe 1987; Yanagawa et al. 1994). Therefore, it is possible that the carcinogenic action of MeIQx, an environmental carcinogen, would be facilitated by induction of P450s by other chemicals concomitantly present in the diet. We therefore, investigated possible interactions of MeIQx and CYP1A2 inducers in foods such as methyl-5-(phenylthio)-2-benzimidazole carbamate (fenbendazole) and caffeine in previous studies (Kuribayashi et al. 2006; Suzuki et al. 2002). Despite induction of CYP1A2, carcinogenic effects of MeIQx were not modified.
In the present study, we used alpha- and beta-naphthoflavone (ANF and BNF, respectively) as CYP1A inducers in rat liver (Shoda et al. 2000) to obtain further data on interactions between chemicals in the environment. Using a medium-term rat liver bioassay system (Ito, Tamano, and Shirai 2003; Shirai et al. 1999) that has proven useful for investigation of hepatocarcinogenic effects caused by multiple chemicals (Shirai, Hirose, and Ito 2006), we again did not find appreciable effects on MeIQx-induced hepatocarcinogenesis with either ANF or BNF. However, we also found intralobular distributional differences in CYP1A expression in the liver between MeIQx-treated and ANF- or BNF-treated rats, which could have mechanistic implications.
Materials and Methods
Chemicals
MeIQx was synthesized in the NARD Institute (Osaka, Japan), with a purity of >99.9%. Diethylnitrosamine (DEN) was purchased from Tokyo Kasei Kogyo Co. Ltd. (Tokyo, Japan). ANF and BNF were purchased from Sigma-Aldrich Inc. (St. Louis, MO, USA).
Animals
Animal experiments were performed under protocols approved by the Institutional Animal Care and Use Committee of Nagoya City University School of Medical Sciences. Five-week-old male F344 rats were obtained from Charles River Japan Inc. (Atsugi, Japan). They were housed in plastic cages with hardwood chip bedding in an air-conditioned room at 23 ± 2°C and 55 ± 5% humidity with a 12-hr light/dark cycle and maintained on a basal diet (Oriental MF, Oriental Yeast Co., Tokyo, Japan) and tap water ad libitum.
Experimental Procedure
Animals were randomly divided into six groups of fifteen rats each. All groups received an intraperitoneal injection of DEN at a dose of 200 mg/kg b.w. as an initiation procedure. Starting 2 weeks later, they were administered basal diet containing 100 ppm MeIQx alone or in combination with 200 ppm ANF or BNF for 6 weeks. Other groups were given 200 ppm ANF, BNF, or basal diet alone. All rats were subjected to two-thirds partial hepatectomy at the end of week 3. Body weights and food consumption were recorded every week, and all surviving animals were killed under ether anesthesia at week 8. The livers were immediately excised, weighed, and cut into 2- to 3-mm-thick slices, one from the caudate lobe and two from the right lateral lobe. The slices were fixed in formalin for immunohistochemical examination. The remaining livers were immediately frozen in liquid nitrogen and stored at –80°C until processed.
Immunohistochemistry
Liver sections were treated with rabbit anti-rat glutathione S-transferase placental form (GST-P) antibody (MBL, Nagoya, Japan) or goat anti-rat CYP1A antibody (Daiichi Kagaku Yakuhin Co. Ltd., Tokyo, Japan) and then sequentially with secondary antibody and avidin-biotin reagents (Vectastain ABC Elite Kit, Vector Laboratories Inc., CA, USA). Anti-rat CYP1A antibody detected both CYP1A1 and 1A2 but not other CYP isotypes. The sites of peroxidase binding were demonstrated with diaminobenzidine as the substrate. Sections were then counterstained with hematoxylin for microscopic examination. Carcinogenic and promotional effects were quantitatively assessed with reference to the numbers and sizes of GST-P positive foci (Ito, Tamano, and Shirai 2003; Shirai et al. 1999). Areas of GST-P positive foci larger than 0.2 mm in diameter in the liver were determined with the aid of a video image processor (VIP-21C, Olympus Co., Tokyo, Japan). Staining intensity for CYP1A was quantitatively assessed with an Image Processor for Analytical Pathology (IPAP-WIN, Sumika Technos Co., Osaka, Japan) to give optical densities.
Definition of Zones in the Liver
For determination of intralobular differences of CYP1A expression in the liver, we defined the liver cells adjacent to Glisson’s capsule as in zone 1, adjacent to central vein as zone 3, and not adjacent to both of them as zone 2. We also defined GST-P positive foci adjacent to Glisson’s capsule as in zone 1 (Figure 1A), adjacent to central vein as zone 3 (Figure 1C), and adjacent to both Glisson’s capsule and central vein or not adjacent to either of them as zone 2 (Figure 1B).
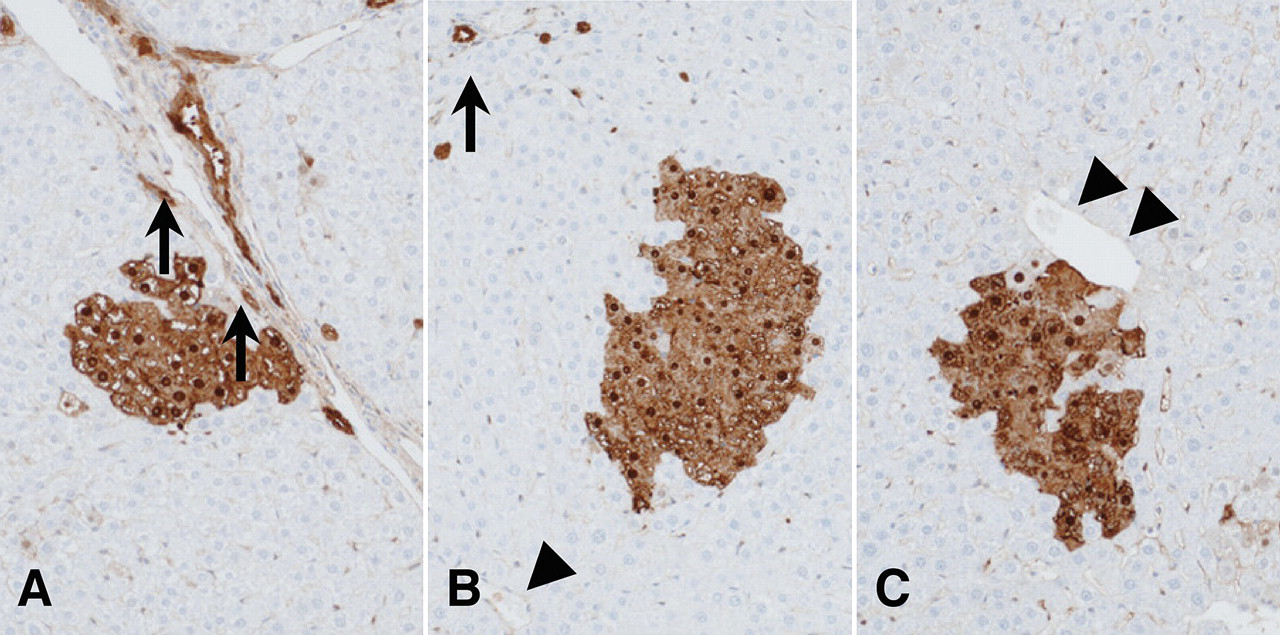
Localization of GST-P positive foci in rat livers. GST-P positive focus adjacent to a Glisson’s capsule (zone 1; A), a central vein (zone 3; C), and neither (zone 2; B). Arrows indicate a Glisson’s capsule; arrowheads indicate a central vein.
Western Blot Analysis
Microsomal fractions of livers prepared from the animals in each group were subjected to 10% SDS-polyacrylamide gel electrophoresis and transferred to nitrocellulose membranes (Bio-Rad Laboratories, Hercules, CA, USA). After blocking, the membranes were incubated with goat anti-rat CYP1A antibody or anti-beta-actin (Sigma-Aldrich Inc.) for 1 hr at room temperature and then secondary antibody for 1 hr. After soaking in ECL detection reagents (GE Healthcare, Buckinghamshire, UK), the membranes were autoradiographed (Hyperfilm-ECL, GE Healthcare).
Quantitative Reverse Transcription (RT)–PCR
Total RNAs were isolated using ISOGEN (Wako, Tokyo, Japan), and cDNA was synthesized with Superscript II (Life Technologies Japan Ltd., Tokyo, Japan) primed with oligo(dT) primers from 1 μg of total RNA as the template. For quantification, aliquots of 2 μl of cDNA samples were subjected to quantitative PCR in 20 μl reactions using FastStart DNA master SYBR Green I and a Light Cycler apparatus (Roche Diagnostics, Mannheim, Germany). The primers used were as follows: for NAT2, 5′-CCTGCCATCTTTCGTTTGAC-3′ and 5′-GAGCCAACCAAACAGTGAAC-3′; for NAT1, 5′-GGACCAAATCAGAAGAGAGC-3′ and 5′-CCAAAGAAATGCCAAATGTG-3′; for GAPDH, 5′-GCATCCTGCACCACCAACTG-3′ and 5′-GCCTGCTTCACCAC CTTCTT-3′. Initial denaturation at 95°C for 10 min was followed by 40 cycles of denaturation at 95°C for 15 sec, annealing at 60°C for 5 sec, and elongation at 72°C for 30 sec, with fluorescence detection performed at 87°C after each cycle. GAPDH mRNA levels were used to normalize for sample cDNA content.
Statistical Analyses
Differences between control or MeIQx-treated or ANF/BNF-exposed groups were assessed by ANOVA, which when significant (p < .05) was followed by Dunnett’s test (GraphPad Prism 5, GraphPad Software, Inc., La Jolla, CA, USA).
Results
Body and Liver Weights and Food Consumption
During the experimental period, minimal differences in body weight gain were observed among the groups, and final body weights did not significantly vary (Table 1 ). Absolute and relative liver weights of rats treated with MeIQx and/or ANF or BNF were significantly higher than those in the control group (Table 1, data for relative liver weights not shown). There were no significant differences in food consumption among the groups (data not shown).
Effects of treatment with MeIQx and/or naphthoflavone on body and liver weights, GST-P positive foci.
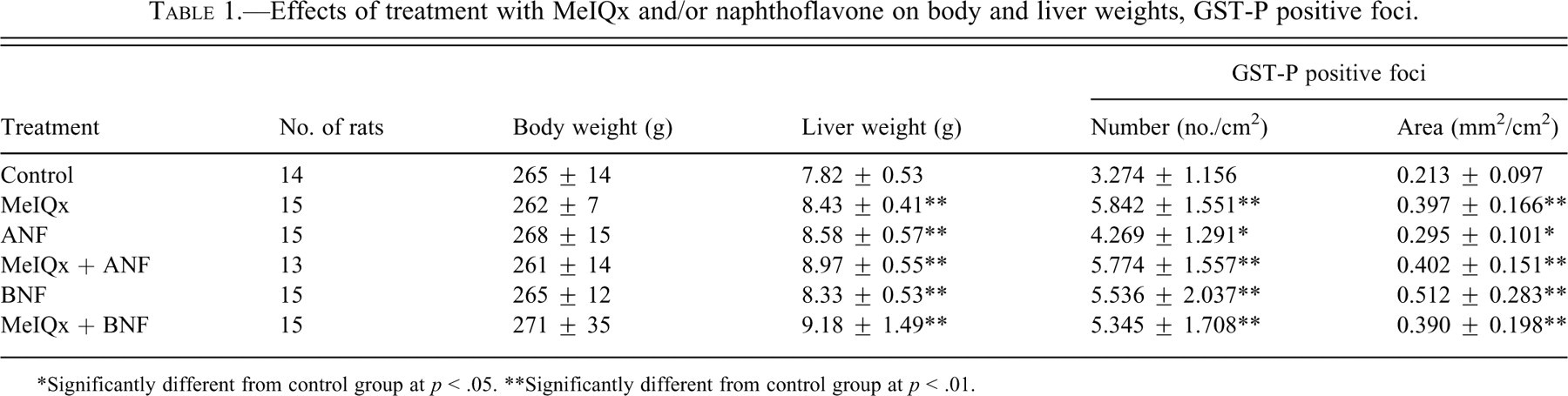
*Significantly different from control group at p < .05.
**Significantly different from control group at p < .01.
GST-P Positive Foci in the Liver
The numbers and areas of GST-P positive liver cell foci were clearly increased by treatment with MeIQx (Table 1). Additionally, the numbers and areas of GST-P positive foci were also increased by treatment with ANF and BNF alone slightly (Table 1). However, combination treatments of MeIQx and naphthoflavones (ANF or BNF) did not cause increase as compared to treatment with MeIQx alone regarding the numbers and areas of GST-P positive foci (Table 1).
Localization of GST-P Positive Foci in the Liver
For determination of MeIQx-induced carcinogenic effect on the liver, localization of GST-P positive foci was investigated in all groups. The percentages of GST-P positive foci in each zone of the liver for all groups are presented in Table 2 . The percentage of GST-P positive foci in Zone 3 was significantly greater in the livers of rats treated with MeIQx (18.7%) compared to the controls (10.2%), ANF (9.4%), and BNF (9.2%). Coadministration of MeIQx and BNF (8.7%) reduced the percentage of GST-P positive foci in zone 3 significantly compared to MeIQx-treated rats (18.7%). However, coadministration of MeIQx and ANF was without additional influence (18.7%).
Zone differences in the liver of each treatment.
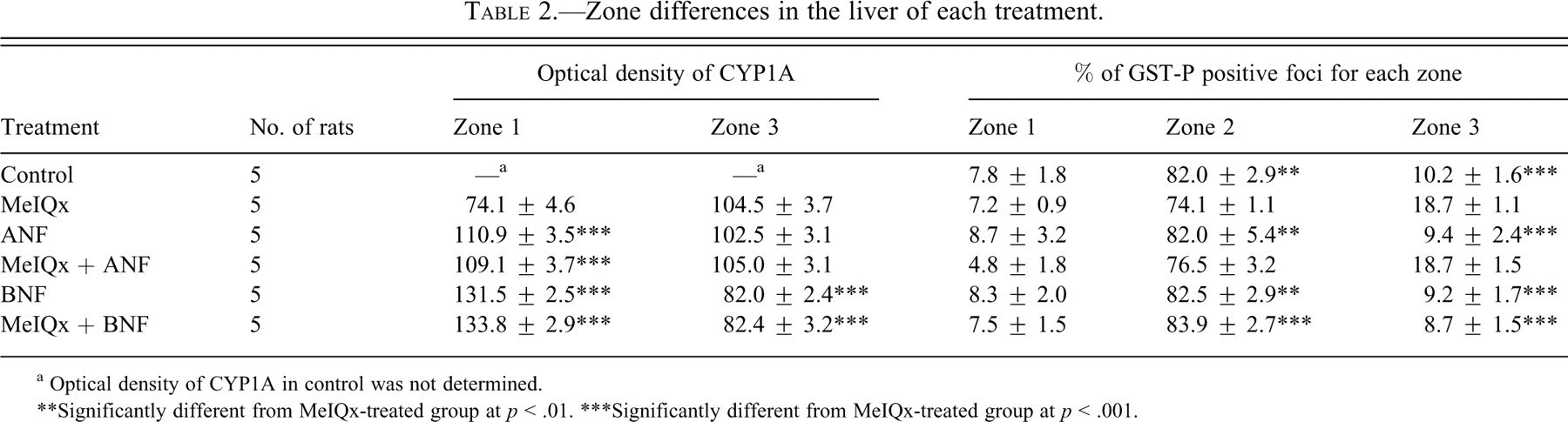
a Optical density of CYP1A in control was not determined.
** Significantly different from MeIQx-treated group at p < .01.
*** Significantly different from MeIQx-treated group at p < .001.
Expression of CYP1A and NAT in the Whole Liver
Data for CYP1A protein expression from Western blot analysis are presented in Figure 2A. CYP1A2 protein expression in livers of rats treated with MeIQx was higher than in controls. Further significant increment was noted with napthoflavones (ANF and BNF). Remarkable induction of CYP1A1 protein expression was also observed in livers of rats treated with napthoflavones (ANF and BNF), alone or with MeIQx.
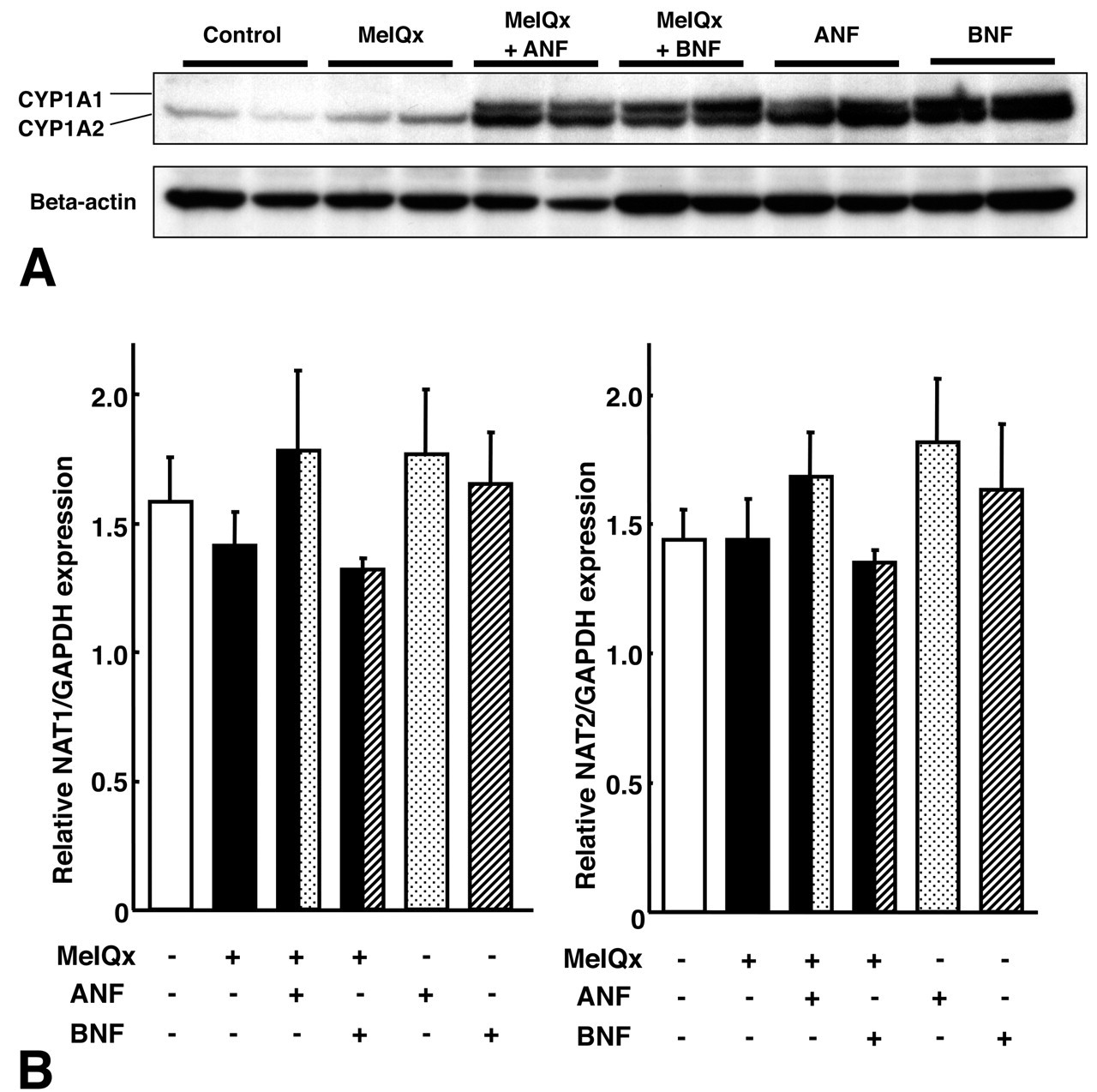
Expression of CYP1A and NAT in rat livers. A: Expression of CYP1A1, 1A2, and actin as shown by Western blotting. B: Expression of NAT1 and NAT2 in a quantitative RT-PCR.
For metabolic activation of MeIQx after the CYP1A2-induced metabolism, expression of NATs is important factor for MeIQx-induced hepatocarcinogenesis. On quantitative RT-PCR analysis, there were no significant differences in expression of NAT1 or NAT2 among the groups (Figure 2B).
Expression and Localization of CYP1A in the Liver
On immunohistochemistry of CYP1A in rat livers, intralobular distributional differences in staining were observed. Data for optical density of CYP1A in each zone for the liver for the different groups are presented in Table 2. The zones in the liver were defined the same as the definition of localization of GST-P positive foci. Immunostaining in the livers of MeIQx-treated rats was mainly in zone 3 (Figure 3A) as in control rats (data not shown). In contrast, high optical density of immunostaining was apparent in zone 1 of the liver in rats treated with ANF (110.9 [O.D.]; Figure 3B) and BNF (131.5 [O.D.]; Figure 3D) compared to with MeIQx only (74.1 [O.D.]; Figure 3A). Only low optical density of immunostaining in zone 3 of the liver was detected in rats treated with BNF (82.0 [O.D.]; Figure 3D) compared to MeIQx (104.5 [O.D.]; Figure 3A) or ANF (102.5 [O.D.]; Figure 3B). Findings with combinations of MeIQx and napthoflavones (ANF or BNF; Figures 3C and Figures 3E, respectively) were the same as with napthoflavones alone.
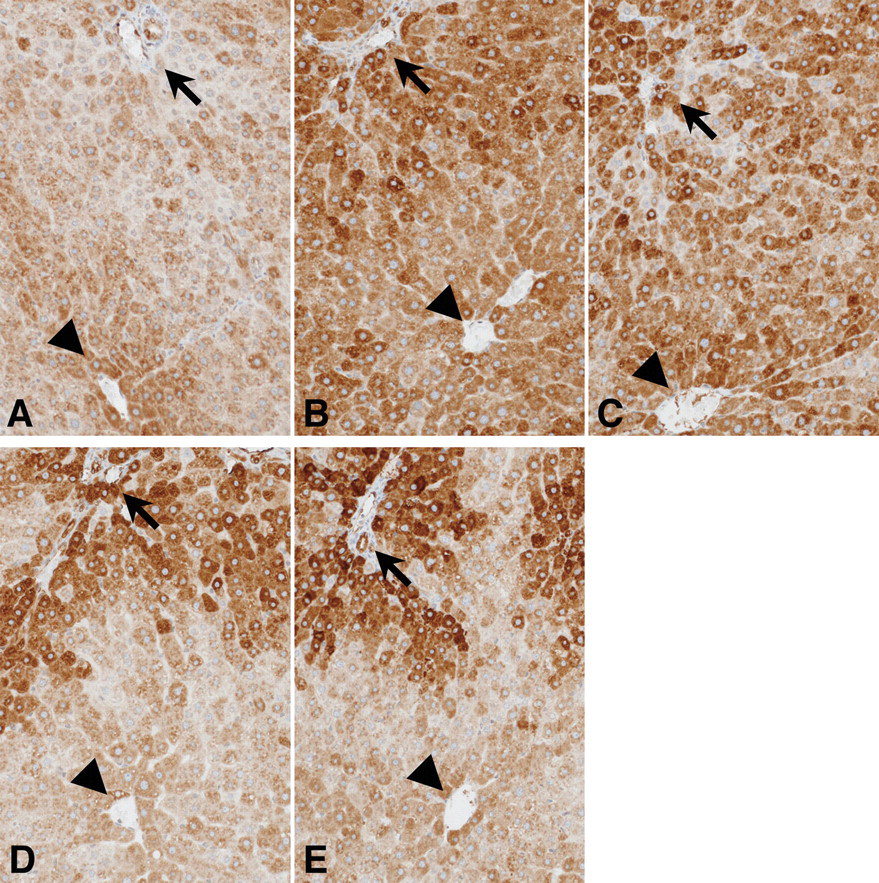
Immunohistochemistry of CYP1A in rat livers. A: MeIQx; B: ANF; C: MeIQx + ANF; D: BNF; E: MeIQx + BNF. Arrows indicate a Glisson’s capsule; arrowheads indicate a central vein.
With the results of localization of GST-P positive foci, coadministration of ANF did not modify MeIQx-induced ratio of GST-P positive foci and CYP1A expression in zone 3; however, BNF reduced both of them.
Relation between GST-P Positive Foci and Expression of CYP1A
In the GST-P positive foci, expression of CYP1A was decreased compared to surrounding parenchyma of both MeIQx-treated (Figure 4C) and BNF-treated rats (Figure 4F). The same was the case for ANF treatment and combined MeIQx and napthoflavone (ANF or BNF) treated rats (data not shown).
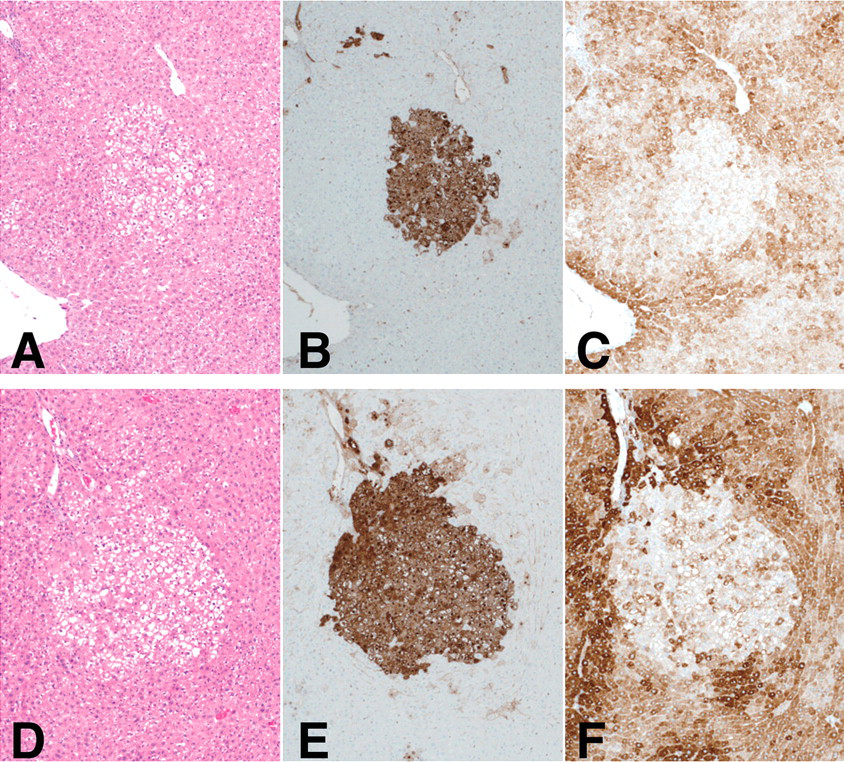
Relation between GST-P positive foci and CYP1A expression in rat livers. A, D: H & E; B, E: GST-P; and C, F: CYP1A antibody. GST-P positive foci detected in MeIQx-treated (A ~ C) and BNF-treated rats (D ~ F).
Discussion
The medium-term rat liver bioassay, which was used in the present study, is known to be a reliable test method for the detection of carcinogens as well as promoters of hepatocarcinogenesis (Ito, Tamano, and Shirai 2003; Shirai 1997; Shirai et al. 1999). The model has been applied for investigation of modifying effects of additional chemicals (Puapairoj et al. 1994; Suzuki et al. 2002) as well as combined effects of multiple carcinogens (Hasegawa et al. 1996; Ito et al. 1995, 1991; Shirai, Ogawa, and Takahashi 2006).
In the present study, two naphthoflavones, ANF and BNF, applied as alternative CYP1A2 inducers did not demonstrate enhancement of MeIQx carcinogenicity in the medium-term rat liver bioassay model, contrary to our expectation. Concerning carcinogenicity of naphthoflavones, both ANF and BNF did not exert carcinogenicity in 2-year animal carcinogenicity studies (http://ntp.niehs.nih.gov/ntp/htdocs/Chem_Background/ExSumPdf/Benzoflavone.pdf), but recently BNF was reported to have tumor-promoting effects on rodent liver (Shoda et al. 2000), which is supported by the result of the present study. Effects of ANF against CYP1A2 were contradictive; inhibiting CYP1A2 function in vitro (Cho et al., 2001, 2003) and inducing CYP1A2 mRNA expression in human hepatocellular carcinomas (Ishida et al. 2002). The reason for such contrasting action of ANF on CYP1A2 is uncertain. On the other hand, effects of BNF have been reported to induce the CYP1A2 expression (Ishida et al. 2002; Shoda et al. 2000). As demonstrated in the present study, both ANF and BNF induced CYP1A2 expression in the liver at a detectable level by Western blotting (Figure 2A). Despite this fact, neither chemical altered MeIQx hepatocarcinogenicity. The findings suggested that induction of CYP1A2 by the naphthoflavones did not facilitate MeIQx metabolic activation as expected. The reasons for the unexpected results are unclear, but our investigation of the intralobular distribution of CYP1A2 might provide a clue. Several studies previously showed that chemical induction of P450s occurs with intralobular differences among chemicals (Buhler et al. 1992; Oinonen et al. 1996; Tritscher et al. 1992; Tsutsumi et al. 1989). In the present case, the immunohistochemical distribution of CYP1A varied among the three chemicals: predominant expression was noted in zone 3 in the MeIQx and control cases; in zone 1 with BNF more broad and diffuse in the case of ANF. Coadministration of MeIQx and ANF resulted in broadly diffuse expression, exactly the same as with ANF alone, indicating the activity in zone 1 to be enhanced. The MeIQx and BNF pattern was very similar to that of BNF alone, indicating that the activity in zone 1 was increased but that in zone 3 was decreased (Table 2). These distributional shifts of CYP1A expression probably resulted from stronger CYP1A inducible ability of ANF and BNF compared with that of MeIQx.
Concerning the zonal distribution of GST-P positive foci, MeIQx shifted the ratio toward zone 3 as compared to the control, and the same was the case for MeIQx- and ANF-treated rats. However, with MeIQx and BNF or BNF alone, the pattern of GST-P positive foci returned to that of the controls (Table 2). Thus, it could be that influence of hepatocarcinogenesis of MeIQx was exerted on activation of CYP1A in zone 3. However, in this study, CYP1A2 was not separated from CYP1A1 and CYP1A2 in the results of lobular localization of CYP1A expression detected by immunohistochemistry. However, protein expression of CYP1A1 and CYP1A2 separately was only detected in the whole liver tissues by Western blotting. More detailed studies were needed for determination of the relation between MeIQx carcinogenesis and CYP1A2 expression in zone 3. Expressions of NATs in the liver did not differ among the groups. Therefore, there might be no different modification of the second step of MeIQx metabolism for carcinogenesis.
Expression of CYP1A in the GST-P positive foci, regardless of the groups, was reduced (Figures 4C and 4F, data not shown), in line with our earlier findings (Suzuki et al. 2004). GST-P positive foci are known to be preneoplastic lesions in rat liver (Ogiso et al. 1985; Sato et al. 1984), highly expressing glutathione transferases that are very active in antioxidant pathways (Hayes, Flanagan, and Jowsey 2005; Ketterer 1988). These data suggest that MeIQx may be detoxified rather than genotoxically activated in GST-P positive foci.
In summary, ANF and BNF (especially BNF) induce CYP1A mainly in zone 1. Therefore, ANF and BNF did not affect the hepatocarcinogenicity of MeIQx, which induces GST-P positive foci mainly in zone 3. The intralobular distribution of metabolic enzyme expression in the liver may be an important factor for hepatocarcinogenesis in vivo.
Footnotes
Acknowledgment
The authors would like to thank Dr. Malcolm Moore for helpful review of this article.
This work was supported in part by a Grant-in-Aid for cancer research from the Ministry of Health, Labour and Welfare of Japan and a grant from the Society for Promotion of Pathology in Nagoya, Japan.
