Abstract
B6C3F1 mice chronically exposed to 3,3′,4,4′-tetrachloroazobenzene (TCAB), a contaminant of dichloroaniline-derived herbicides, developed a number of neoplastic and nonneoplastic lesions, including carcinoma of the urinary tract. Groups of fifty male and fifty female B6C3F1 mice were exposed by gavage to TCAB at dose levels of 0, 3, 10, and 30 mg/kg five days a week for two years. Control animals received corn oil:acetone (99:1) vehicle. Decreased survival of male mice in the mid-dose group and of male and female mice in the high-dose groups was related mainly to the occurrence of urethral transitional cell (urothelial) carcinoma and resulting urinary obstruction. Increased urethral transitional cell carcinomas were seen in all treated male groups in a dose-related manner as well as in the females treated with 30 mg/kg TCAB. Administration of TCAB was also associated with increased transitional cell hyperplasia of the urethra. Most nonneoplastic lesions of the urogenital tract were considered secondary to local invasion and urinary obstruction by the urethral transitional cell carcinomas. The mechanism of tumor induction is uncertain, but the high frequency of tumors in the proximal urethra of male mice suggests that the neoplasms result from the exposure of a susceptible population of urothelial cells to a carcinogenic metabolite of TCAB.
Introduction
3,3′,4,4′-Tetrachloroazobenzene (TCAB) is formed as a contaminant during the manufacture of 3,4-dichloroaniline and its herbicidal derivatives Propanil™, Linuron™, Diuron™, and Neburon™ (Bunce et al. 1979; Hill et al. 1981; Poland et al. 1976; Sundström, Jansson, and Renberg 1978). 3,3′,4,4′-Tetrachloroazobenzene is also formed by the photolysis and biolysis of 3,4,-dichloroaniline (Miller, Zisook, and Zepp 1980). In addition, environmental contamination by TCAB occurs from the degradation of chloroanilide herbicides (acylanilides, phenylcarbamates, and phenylureas) in soil by peroxide-producing microorganisms (Bartha, Linke, and Pramer 1968; Bartha and Pramer 1969; Lay and Ilnicki 1974). Since there are no commercial uses for TCAB, accurate figures concerning its production are limited. Volume estimates of TCAB contamination are based on production volumes of dichloroaniline-derived herbicides. Production of approximately ten million pounds of Propanil per year is estimated to result in production of 26,400 pounds of TCAB per year, and production of 100,000 to 1,000,000 pounds of 3,4-dichloroaniline per year is estimated to result in 8580 pounds of TCAB produced per year (U. S. EPA 1985).
Humans are exposed occupationally to TCAB during the manufacture of 3,4-dichloroaniline and its herbicidal derivatives, the application of the herbicides, and the use of 3,4-dichloroaniline in additional products, including dyes (Taylor et al. 1977). Nonoccupational human exposure may result from the consumption of dietary residues in food and water and environmental residues in soil. In 2006, the estimate for total domestic use of Propanil was approximately seven million pounds of active ingredient; 50% to 70% of the United States rice crop is treated with Propanil (U. S. EPA 2006). Analyses of soil samples from a rice field plot treated with 6.7 kg Propanil/hectare indicated a TCAB concentration of 0.09 ppm (Kearney et al. 1970). 3,3′,4,4′-Tetrachloroazobenzene was detected in the roots and shoots of soybean plants grown in soil treated with 25 ppm TCAB (Worobey 1984).
Most of the biological disposition information on TCAB has been reported in rats. 3,3′,4,4′-Tetrachloroazobenzene is absorbed systemically when exposure is via the oral, dermal, or inhalation route. It is excreted primarily in the urine and feces, with the majority excreted in the feces. Identification of urinary metabolites indicates that extensive azo reduction occurs, followed by sulfate conjugation and N-acetylation to produce conjugates of mono- and dichloroaniline derivatives. No parent compound was detected in the urine after gavage administration of TCAB. 3,3′,4,4′-Tetrachloroazobenzene may be subject to enterohepatic cycling (Pillai et al. 1996). Recent disposition studies conducted by the National Toxicology Program (NTP) in male and female Sprague-Dawley rats demonstrated a dose-dependent increase in TCAB concentrations, with the highest levels observed in fat tissue and lower levels seen in the liver and lung (NTP 2009).
There is a concern about potential adverse human health effects of TCAB based on its widespread exposure and its structural and biological similarity to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). 3,3′,4,4′-Tetrachloroazobenzene binds and activates the aryl hydrocarbon receptor, resulting in dioxin-like effects in rodents and in humans. There is no information in the scientific literature concerning the carcinogenic potential of TCAB exposure in humans and animals. This study was part of a series of two-year bioassays conducted by the NTP to evaluate the chronic pathology and potential carcinogenicity induced by dioxin, structurally related polychlorinated dibenzofurans, polychlorinated biphenyls, and mixtures of these compounds. The present study examined the carcinogenicity and chronic toxicity of TCAB in B6C3F1 mice. Previously, the NTP performed three-month studies of TCAB in male and female rats (F344/N and Harlan Sprague Dawley) and B6C3F1 mice to aid in the design and dose selection for these two-year carcinogenicity studies (NTP 1998; Van Birgelen et al. 1999). The three-month studies demonstrated that administration of TCAB to mice caused decreased thymus weights and increased liver and spleen weights. Treatment-related effects included centrilobular hypertrophy in the liver, hematopoietic cell proliferation in the spleen, and decreased sperm production in males, and squamous hyperplasia in the forestomach of males and females (NTP 1998). 3,3′,4,4′-Tetrachloroazobenzene was weakly mutagenic in Salmonella typhimurium strain TA97, but only in the presence of 30% induced rat liver S9 activation enzymes (NTP 1998). In the present publication, we report that TCAB administration to B6C3F1 mice resulted in a dose-related increase in the incidence of a unique neoplasm, urethral transitional cell (urothelial) carcinoma. The other significant neoplastic and nonneoplastic effects of TCAB are described in an NTP technical report (NTP 2009).
Materials and Methods
Chemical and Dose Formulations
3,3′,4,4′-Tetrachloroazobenzene (CAS No. 14047-09-7) was obtained from AccuStandard, Inc. (New Haven, CT, USA) in one lot. 3,3′,4,4′-Tetrachloroazobenzene was identified by infrared and proton nuclear magnetic resonance spectroscopy, by gas chromatography (GC) coupled with mass spectrometry, and by melting point analysis. The purity of TCAB was determined by Karl Fischer titration to determine moisture content; elemental analysis for carbon, hydrogen, nitrogen, and chlorine; and GC with flame ionization detection, which indicated a purity of 99.8% or greater. The dose formulations were prepared by mixing TCAB with corn oil containing 1% USP-grade acetone to give the required concentrations; they were prepared fresh approximately once a month and were shown to be stable during that period. Periodic analyses of the dose formulations of TCAB were conducted using GC with electron capture detection. All analyzed dose formulations were within 10% of the target concentrations.
Animals and Animal Maintenance
The studies were conducted in the AAALAC accredited facility of Battelle Columbus Laboratories (Columbus, OH, USA). Male and female B6C3F1 mice were obtained from Taconic Farms, Inc. (Germantown, NY, USA) and quarantined for eleven or twelve days before dosing; mice were approximately five to six weeks old at the beginning of the studies. Animals were distributed randomly into groups of approximately equal initial mean body weights and identified by tail tattoo. Male mice were housed individually, and female mice were housed five per cage in polycarbonate cages containing irradiated Sani-Chips hardwood chip bedding (P. J. Murphy Forest Products Corp., Montville, NJ, USA). Tap water and feed (irradiated NTP-2000 pelleted diet, Zeigler Brothers, Inc., Gardners, PA, USA) were available ad libitum. The room temperature was 72°F ± 3°F, and the humidity was 50% ± 15% throughout the study. The fluorescent light cycle was twelve hours per day, and there were at least ten room air changes per hour. Animals were killed by carbon dioxide asphyxiation.
Animal use was in accordance with the United States Public Health Service policy on humane care and use of laboratory animals and the Guide for the Care and Use of Laboratory Animals. Animal handling and husbandry met all NIH guidelines (Grossblatt 1996). These studies were conducted in compliance with the Food and Drug Administration Good Laboratory Practice Regulations (U. S. FDA 1987).
Experimental Design
Groups of fifty male and fifty female mice were administered 0, 3, 10, or 30 mg TCAB/kg in corn oil:acetone (99:1) by gavage five days per week for 105 weeks. The dosing volume was 10 mL/kg body weight. The dose levels were selected based on the results from NTP three-month toxicity studies (NTP 1998; Van Birgelen 1999). Animals were observed twice a day for clinical signs of toxicity. Individual animal weights were recorded on day 1, weekly for thirteen weeks, monthly thereafter, and at the end of the studies.
Pathology
Complete necropsy and microscopic examination were performed on all animals that died early or were killed at termination. At necropsy, all organs and tissues were examined for grossly visible lesions. Tissues were fixed and preserved in 10% neutral buffered formalin according to NTP protocol (NTP 2009), trimmed and processed, embedded in paraffin, sectioned to a thickness of 5–6 µm, stained with hematoxylin and eosin (H&E), and examined microscopically. The proximal urethra was examined microscopically in all male and female mice, and the ureter was examined microscopically in all male mice. Additional details regarding the pathology data generation and pathology peer review are available elsewhere (Boorman et al. 1985; NTP 2009).
Based on the findings of urethral transitional cell carcinoma in the current two-year study, a retrospective study was performed to microscopically examine the urinary bladder and urethra from the three-month mouse study (NTP 1998).
Statistical Analysis
Dose-related effects on survival were tested using Cox’s method (1972) to identify pairwise differences from the control group and Tarone’s life table test (1975) to identify trends. Incidences of neoplasms are calculated as the number of animals bearing the lesion at a specific site divided by the number of animals examined microscopically at that site. Because some animals did not survive the entire two years, the Poly-3 test (Bailer and Portier 1988; Piegorsch and Bailer 1997; Portier and Bailer 1989) was used to compare survival-adjusted incidences. The Poly-3 test is a modified Cochran-Armitage linear trend test that adjusts the denominator of the incidence to more closely approximate the number of animal years at risk. Animals surviving until terminal sacrifice and animals dying early with the lesion of interest are assigned a risk weight of one. Animals dying early without the lesion of interest are assigned a risk weight equal to the proportion of the study that they survived raised to the third power. The value of 3 is based on the shape parameter of a Weibull hazard function that has been shown to reasonably fit the cumulative lesion incidence of a variety of neoplasms in NTP’s two-year mouse and rat studies (Bailer and Portier 1988; Portier, Hedges, and Hoel 1986). The number of animals at risk in a specific dose group is the sum of risk weights of all animals in that dose group and constitutes the denominator of the survival-adjusted incidence. The Poly-3 test statistic is corrected for continuity and its variance is adjusted to reflect the fact that the risk weights are random variables (Bieler and Williams 1993). Tests of significance on the survival-adjusted incidences included tests for trends over dose and pairwise comparisons of each dose group with the controls.
Results
Survival and Body Weight
The survival in 10 and 30 mg/kg males and 30 mg/kg females was markedly reduced (11%, 0%, and 39% respectively) and significantly lower than that of the vehicle controls (Table 1 ). All 30 mg/kg males died before the end of the study. Most of the deaths in the 10 and 30 mg/kg males were considered related to transitional cell (urothelial) carcinomas of the urethra and resulting urinary obstruction. Urethral transitional cell carcinomas, malignant schwannomas, and fibrosarcomas of skin contributed to decreased survival in 30 mg/kg females.
Survival of B6C3F1 mice exposed to 3,3′,4,4′-tetrachloroazobenzene.
a Kaplan-Meier estimates.
b Mean of all deaths (uncensored, censored, and terminal sacrifice).
* Significantly different (p ≤ .01) from the vehicle control group by the life table pairwise comparisons (Cox, 1972).
Mean body weights of treated males and females were similar to or greater than those of the vehicle controls throughout most of the study. Mean body weights of 10 and 30 mg/kg males were 10% and 8% less than those of the vehicle controls at the last weighing at weeks 101 and 73, respectively.
Pathology
Urethra
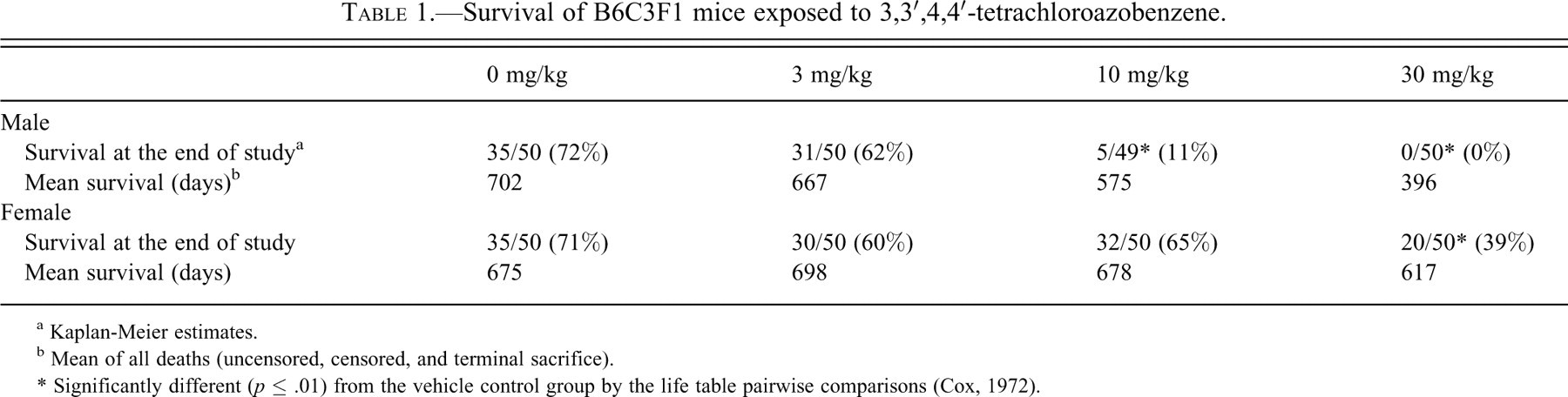
There was a dose-related increase in transitional cell (urothelial) carcinoma of the urethra (survival-adjusted rates of 0%, 71%, 100%, and 100% in the vehicle control, 3, 10, and 30 mg/kg groups, respectively) (Table 2 ) of TCAB-treated males, but not in the females, in which only two incidences of urethral carcinoma occurred in the high-dose group (Table 2). Carcinomas were characterized by a poorly demarcated, hypercellular proliferation of transitional epithelium with a variably solid and sometimes glandular appearance often circumferentially involving the urethra. The lesions showed prominent cellular atypia and frequent mitotic figures. In some cases, it was apparent that the neoplastic process was confined to the submucosal gland area, whereas in more invasive lesions, both the urethral lining and the submucosal compartment were involved (Figure 1A–C). The urethral carcinomas usually invaded adjacent tissues and/or had spread to the prostate gland, seminal vesicle, coagulating gland, ductus deferens, kidney, ureter, urinary bladder, skeletal muscle, and lung. Local invasion occurred in the majority of male and female mice (up to 100%) bearing urethral transitional cell carcinomas. Distant metastasis was rare and was found in the lungs of only one male mouse. In male mice, carcinomas and hyperplastic lesions appeared to arise from the surface or glandular epithelium of the proximal urethra (membranous urethra) near the excretory ducts of the accessory sex glands (Figure 2A–C). Neoplastic cells eventually invaded and replaced adjacent ducts and/or acini of the urethral glands and in advanced cases effaced the submucosa and outer muscular wall (Figure 1A). The invasive malignant tumors were primarily a solid transitional type with occasional regions forming acinar/glandular structures and/or squamous differentiation (Figure 1B, 1C).
Incidences of neoplasms and nonneoplastic lesions of the urethra in B6C3F1 mice exposed to 3,3′,4,4′-tetrachloroazobenzene for two years.
a Number of animals with lesion/number of animals necropsied.
b Poly-3 estimated neoplasm incidence after adjustment for intercurrent mortality.
* Significantly different (p ≤ .01) from the vehicle control group by the Poly-3 test.
Carcinoma in the urethra of a male B6C3F1 mouse administered 10 mg/kg TCAB by gavage for two years. Note the poorly demarcated cellular proliferation of neoplastic epithelium with a glandular appearance (arrows). The tumor is occluding the lumen of the urethra and shows invasion into the submucosal compartment. Hematoxylin and eosin, 25×. (B). Higher magnification of Figure 1A. Note the cellular proliferation of neoplastic epithelium invading into the submucosal compartment and showing a glandular appearance (arrows). Hematoxylin and eosin, 250×. (C) Higher magnification of another area of Figure 1A. Note the solid and less pleomorphic invasive tumor with transitional cell differentiation (arrows). Hematoxylin and eosin, 250×.
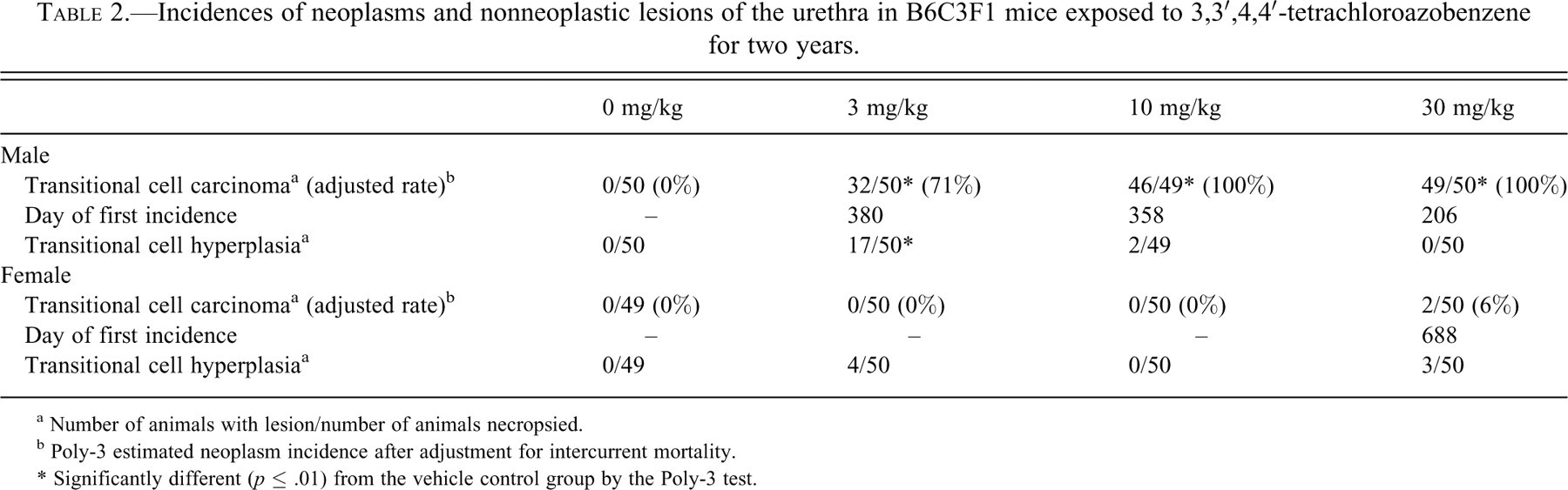
Transitional cell hyperplasia of the urethra occurred in 3 and 10 mg/kg males and in 3 and 30 mg/kg females (Table 2) and was characterized by a localized and well-demarcated increase in the number of luminal or submucosal urothelial cells (Figures 2 and 3).
Marked hyperplasia in the urethra of a male B6C3F1 mouse administered 3 mg/kg TCAB by gavage for two years. Note the hyperplasia arising from the proximal urethra (membranous urethra) near the excretory ducts (large arrow) of the accessory sex glands (small arrow). A coagulum (copulation plug) is present in the urethral lumen (asterisk). Hematoxylin and eosin. (B) Higher magnification of normal part of urethra of Figure 2A. Note the presence of periurethral glands (urethral glands of Littré; arrows), which are located in the lamina propria of the membranous urethra. Hematoxylin and eosin. (C) Higher magnification of a hyperplastic area (arrow) seen in Figure 2A. The hyperplasia involves the urethral glands and surface lining urothelium near the excretory ducts (asterisk).
Based on the findings of urethral transitional cell carcinoma in the current two-year study, a retrospective study was performed to microscopically examine the urinary bladder and urethra from the three-month mouse study. Ten animals per dose group were treated with 0, 0.1, 1, 3, 10, or 30 mg/kg TCAB (NTP 1998). Only one 30 mg/kg male and one 0.1 mg/kg male were found to have mild or moderate transitional cell hyperplasia of the urethra.
Ureter
One 10 mg/kg male and one 30 mg/kg female had transitional cell carcinoma, and one 30 mg/kg female had transitional cell hyperplasia of the ureter (Table 3). The hyperplasia and carcinomas had the same morphological characteristics as those seen in the urethra (Figure 3C–D). Chronic, active inflammation and dilatation in the ureter seen in the treated male mice were interpreted as secondary to the neoplastic lesions in the urethra (Table 3).
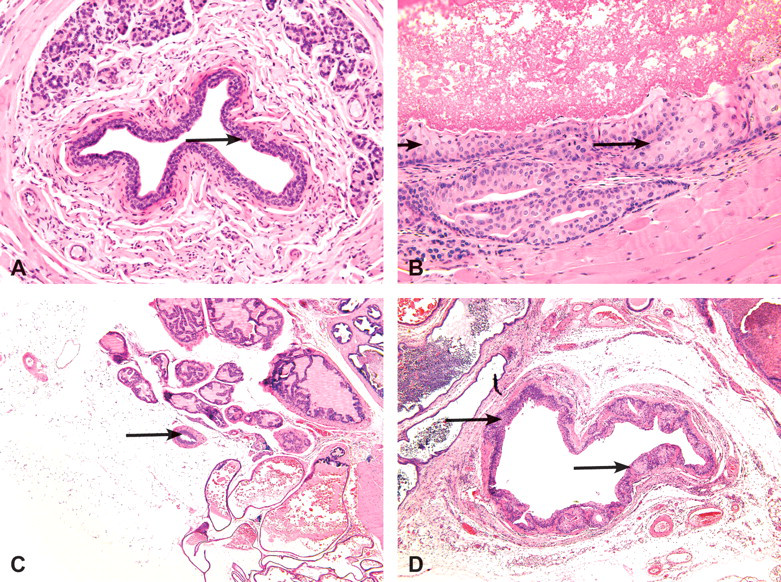
Normal epithelium (arrow) lining the urethra in a male B6C3F1 vehicle control mouse at two years in the gavage study of TCAB. Hematoxylin and eosin, 250×. (B) Transitional cell hyperplasia in the urethra of a male B6C3F1 mouse administered 10 mg/kg TCAB by gavage for two years. Note the localized and well-demarcated increase in the number of transitional cells (arrows). Hematoxylin and eosin, 250×. (C) Normal transitional epithelium (arrow) lining the ureter in a female B6C3F1 vehicle control mouse at two years in the gavage study of TCAB. Hematoxylin and eosin, 50×. (D) Transitional cell hyperplasia in the ureter of a female B6C3F1 mouse administered 30 mg/kg TCAB by gavage for two years. Note irregular hyperplasia of the urethral lining cells (arrows). The urethral lumen is markedly dilated and interpreted as secondary to urethral tumor. Hematoxylin and eosin, 50×.
Incidences of neoplasms and nonneoplastic lesions of the ureter in mice exposed to 3,3′,4,4′-tetrachloroazobenzene for two years.
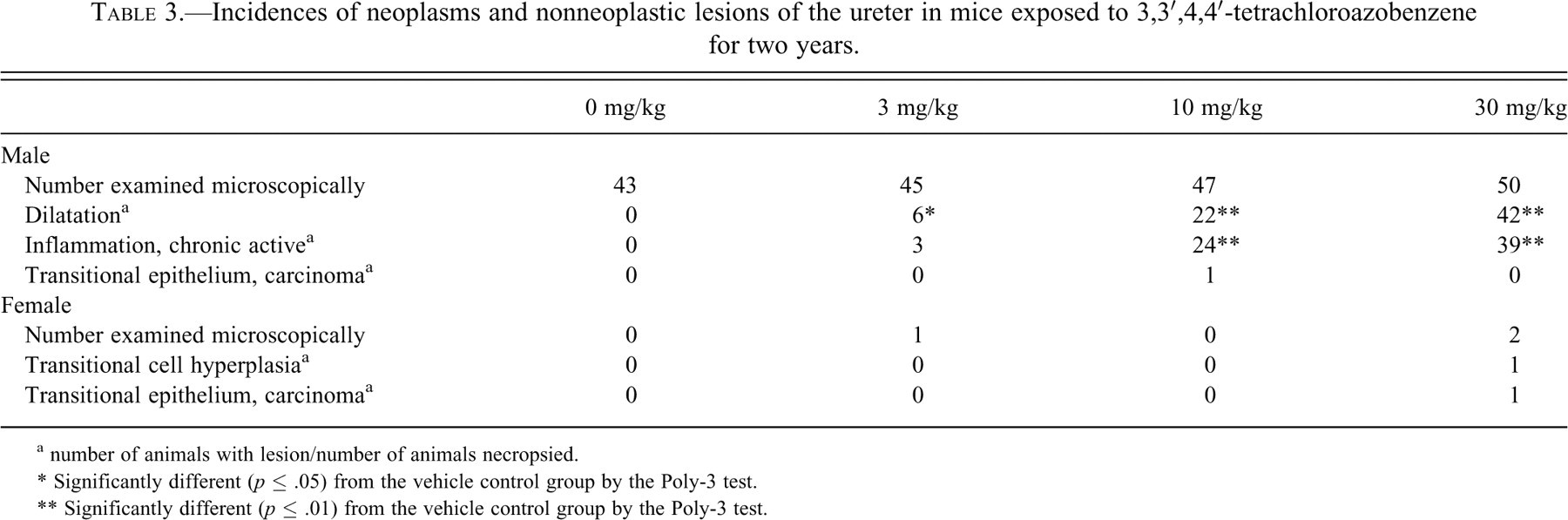
a number of animals with lesion/number of animals necropsied.
* Significantly different (p ≤ .05) from the vehicle control group by the Poly-3 test.
** Significantly different (p ≤ .01) from the vehicle control group by the Poly-3 test.
Urinary Bladder
Transitional cell hyperplasia was observed in the urinary bladder in 8% (four of fifty) of the males and females treated with 30 mg/kg TCAB (p < .01) and 2% (one of fifty) of the males treated with 3 mg/kg (Table 4 ). Microscopically, transitional cell hyperplasia was characterized by a localized and well-demarcated increase in the number of transitional epithelial cells.
Incidence of selected nonneoplastic lesions in kidney and urinary bladder in B6C3F1 mice exposed to 3,3′,4,4′-tetrachloroazobenzene.
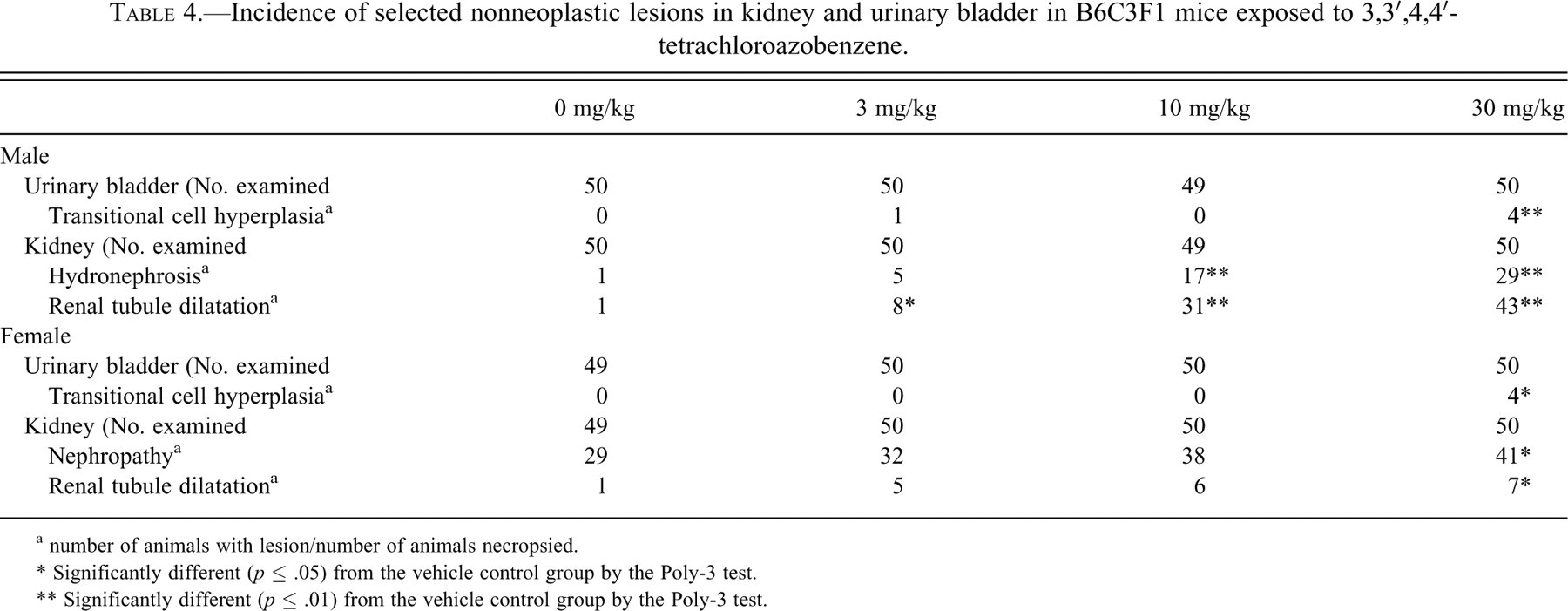
a number of animals with lesion/number of animals necropsied.
* Significantly different (p ≤ .05) from the vehicle control group by the Poly-3 test.
** Significantly different (p ≤ .01) from the vehicle control group by the Poly-3 test.
Kidney
In males, there were significant increases (p < .01) in the incidences (16–86% in dosed groups compared to 2% in the vehicle controls) of renal tubule dilatation (all dosed groups) and hydronephrosis (34% and 58% for 10 and 30 mg/kg groups, respectively, compared to 2% in the vehicle controls) (Table 4). In the 30 mg/kg females, there were significant increases (p < .05) in the incidences of nephropathy and renal tubule dilatation (82% compared to 58% in the vehicle controls and 14% compared to 2% in the vehicle controls, respectively; Table 3). Dilatation affected cortical and medullary tubules, whereas hydronephrosis was evident from the dilatation of the renal pelvis. The dilatation of the tubules and hydronephrosis were likely owing to increased pressure from tumor-related obstruction of urine outflow at the urethra. Microscopically, nephropathy consisted of focal to multifocal regenerative renal tubules surrounded by a thickened basement membrane, glomerular thickening, tubular protein casts, and interstitial chronic inflammatory infiltrates with fibrosis.
Increased incidences of other nonneoplastic lesions (data not shown) in the kidneys were interpreted as either nonspecific or secondary to the urethral tumors, including chronic active inflammation, thrombosis, glomerular hyalinization (presumptive amyloidosis), mineralization, and renal papillary necrosis.
Discussion
3,3′,4,4′-Tetrachloroazobenzene was nominated by the United States Environmental Protection Agency for toxicity and carcinogenicity testing based on its structural and biological similarity to TCDD and potential for human exposure from the consumption of crops contaminated with 3,4-dichloroaniline-derived herbicides including Propanil, Linuron, and Diuron. In this two-year carcinogenicity bioassay, an uncommon neoplasm, urethral urothelial (transitional cell) carcinoma, developed in male and female mice administered TCAB by gavage for two years.
Chronic exposure of TCAB in the male mice was associated with urethral transitional cell carcinoma, with 100% incidence in the two highest dose groups (10 and 30 mg/kg), resulting in compromised survival of male mice. Two similar neoplasms were observed in the high-dose females. Low incidences of treatment-related urethral transitional cell hyperplasia were observed in the three-month mouse study, indicating that these lesions could be induced following subchronic exposure. In the two-year study, the neoplasms were often invasive into the surrounding tissues and spread to other organs (prostate gland, seminal vesicle, coagulating gland, ductus deferens, kidney, ureter, urinary bladder, skeletal muscle, and lung). 3,3′,4,4′-Tetrachloroazobenzene administration was also associated with increased incidences of nonneoplastic lesions in the urinary tract and genital system of male mice (NTP, 2009). Most of the lesions in the urinary tract were considered to be secondary to obstruction and inflammation caused by urethral carcinomas and hyperplasia.
The urothelium covers the luminal surface of almost the entire urinary tract, extending from the renal pelvis, through the ureter and bladder, to the proximal urethra (Gaillard 1999). The urethra of the male mouse is divided into the membranous (proximal) urethra and the penile (distal) urethra (Gaillard 1999). The membranous urethra, urethral diverticulum, and penile urethra are lined by transitional epithelium. In mice, the membranous urethra can be distinguished from the penile urethra by the presence of numerous glands called the urethral or periurethral glands (also called the urethral glands of Littré), which are located in the lamina propria of the membranous urethra. The urethral glands are lined by cuboidal epithelium and empty into the lumen of the membranous urethra. Studies in the literature suggest that the urethral glands in male mice are testosterone dependent and may play a role in localized secretory immunity (Parr et al. 1992, 1994). In male rats and mice, the glands contribute to the formation of semen and the copulation plug.
The urethra of female rodents is relatively shorter than that in males. As in males, the lumen is lined by transitional epithelium and urethral glands are located in the lamina propria. In the female rat, occasional clusters of urethral glands are observed with ducts that empty into the lumen. The urethral glands are most developed at about 30 days of age and then undergo degeneration (Jokinen 1990). These differences may partly explain the presence of fewer urethral tumors in female mice.
Most of the tumors in this study were noted to be derived primarily from the transitional epithelium of the urethra in TCAB-exposed male mice and primarily from the proximal (membranous) urethra. Since the distal urethra was absent in most of the urethral sections, we could not perform microscopic evaluation of the distal urethra. It is not clear why transitional epithelium of the urethra was selectively affected. Some urethral carcinomas appeared to have arisen from the urethral glands. Other neoplasms probably arose from the surface urothelium and invaded down into urethral gland ducts and acini. Most of the neoplasms had typical transitional cell (urothelial) differentiation, with variable abundance of glandlike structures and/or squamous differentiation. It is common to see glandular or squamous differentiation in transitional cell carcinomas in animals (Meuten 2002). The glandular pattern often makes the distinction of prostatic and urethral carcinoma difficult (Meuten 2002). The pattern of involvement of the submucosa and glands of the membranous urethra suggests possible origin from the glands; however, development and outward invasion from the urethral surface epithelium cannot be excluded. More studies are needed to elucidate the mechanisms underlying this regional-specific response.
There are different histologic subtypes of transitional cell tumors in humans and animals (Meuten et al. 2004) based on morphology. The most common variant in animals is papillary and infiltrating. Variable degrees of squamous metaplasia and glandular metaplasia may occur independently or in combination in all of the variants of TCC in animals. In humans, urothelial carcinoma is the most common tumor of the urinary bladder, accounting for approximately 90% of tumors in this anatomical location. These tumors have been classified into two major groups, infiltrating urothelial carcinoma and noninvasive urothelial carcinomas (Lopez-Beltran et al. 2004). Infiltrating urothelial carcinoma has a propensity for divergent differentiation, with the most common variant being squamous and the next being glandular (Lopez-Beltran et al. 2004).
Spontaneous and chemically induced proliferative lesions are very rare in the lower urinary tract of rodents (Wolf 2002). Searches of the published literature and the NTP historical control database failed to reveal any spontaneous or chemically induced transitional cell carcinomas in the urethra or ureter of B6C3F1 mice in two-year cancer bioassays. The exact incidences of the proliferative lesions in the ureter and urethra are not known, since these tissues are not routinely examined microscopically in NTP studies. Only one other chemical tested by the NTP, nitrilotriacetic acid trisodium monohydrate, resulted in increased incidences (33% in males and 25% in females) of ureteral transitional cell carcinoma in rats (NCI 1977). Chemically induced transitional cell tumors are rare and have been described mostly in the urinary bladder. In the urinary bladder, twenty-four chemicals induced tumors in one or more of the four sex-species experimental groups of NTP two-year rodent cancer bioassays. Only three compounds induced urinary bladder tumors in male or female mice: o-anisidine hydrochloride (NCI 1978a), p-cresidine (NCI 1979), and 1,3-dichloropropene (Telone II) (NTP 1985).
The proximal urethra is a relatively common site for transitional cell carcinoma in the dog. Bracken fern, cyclophosphamide, insecticides, benzene, nitrosamines, papilloma virus (BPV), schistosomes, and tryptophan cause transitional cell carcinoma within the urogenital tract of dogs, cattle, and/or humans (Meuten 2002). Structurally related chloroaniline compounds, such as 4-chloroaniline, are aromatic amines that were widely used in the dye, chemical, and pharmaceutical manufacturing industries. They are also persistent environmental degradation products of various herbicides. Several chloroaniline analogs were evaluated for carcinogenicity in experimental animals based on reports of high incidences of bladder cancer observed among workers in the dye manufacturing industry. However, IARC (1987) concluded that the evidence for carcinogenicity of aniline to humans was inadequate based on the conclusion that the excess of bladder cancer deaths observed in workers in the aniline dye industry was attributed to exposure to chemicals other than aniline. 4-Chloroaniline hydrochloride induced fibrosarcomas in the spleen of male rats and hemangiosarcomas in the spleen and liver of male mice (NTP 1989). Increased incidences of hepatocellular adenomas and carcinomas were also observed in male mice. Based on data in experimental animals, 4-chloroaniline was classified as possibly carcinogenic in humans (group 2B). 4-Chloro-o-phenylenediamine, another chemical containing a chloroaniline moiety, induced urinary bladder neoplasms in male and female rats (NCI 1978b). No experimental animal data are available for 3,4-dichloroaniline and a number of other chemical analogs.
The mechanism of TCAB-induced urethral transitional cell carcinoma in mice is unclear and needs further investigation. It is unclear whether the pathogenesis is mediated through the aryl hydrocarbon receptor (AhR). Tumors of the urinary tract were not observed in a previous NTP two-year gavage study of TCDD in B6C3F1 mice (NTP 1982). However, exposure to TCDD has been shown to cause proliferation of embryonic urothelium (Abbott, Birnbaum, and Pratt 1987). The epithelial cells of the ureter express AhR and epithelial growth factor receptor (EGFR) (Choi Miller, and Harper 2006). Expression of EGFR in ureter epithelium declines with age. 2,3,7,8-Tetrachlorodibenzo-p-dioxin inhibits the decrease in EGFR in ureter epithelium with subsequent proliferation of these cells (Abbott and Birnbaum 1990). Epithelial growth factor receptor has also been shown to play an important role in N-butyl-N-(4-hydroxybutyl) nitrosamine-induced bladder tumors in mice (El-Marjou et al. 2001).
This study demonstrates that administration of TCAB by gavage for two years in male B6C3F1 mice induced increased incidences of a rare neoplasm, urethral transitional cell carcinoma. This finding represents clear evidence of carcinogenic activity (according to NTP levels of evidence for carcinogenic activity) of TCAB in male B6C3F1 mice based on the dose-related increased incidence of these neoplasms. Although the incidences did not increase significantly in female mice, the presence of these rare urethral transitional cell carcinomas was considered to be related to TCAB administration. The marginally increased incidence of carcinoma of the ureter in males and females may have been related to TCAB administration. The mechanism of tumor induction is uncertain, but the high frequency of tumors in the proximal urethra of male mice suggests that the neoplasms result from the exposure of a susceptible population of urothelial cells in this region to a carcinogenic metabolite of TCAB.
Footnotes
Acknowledgments
This research was supported (in part) by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences under Research Project Number 1 Z01 ESO45004-11 BB. The authors wish to thank Drs. Flake and Masten for critical review of this manuscript.
