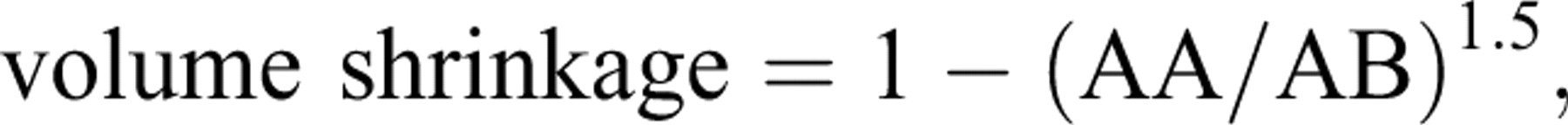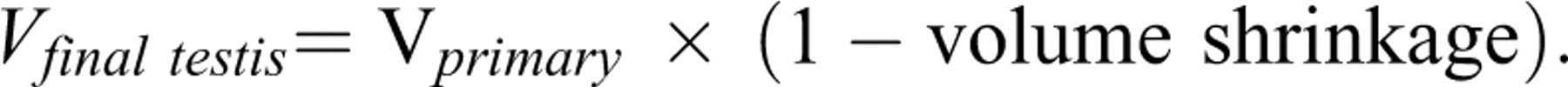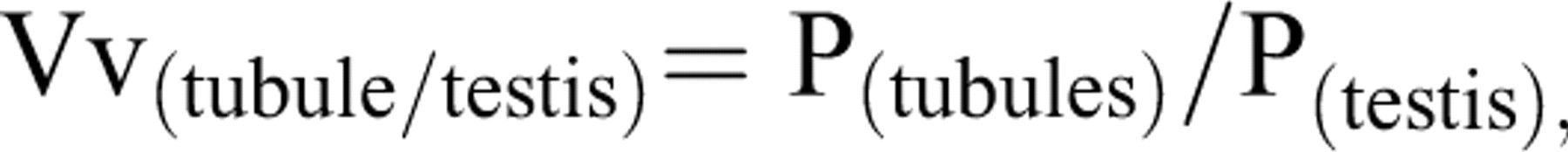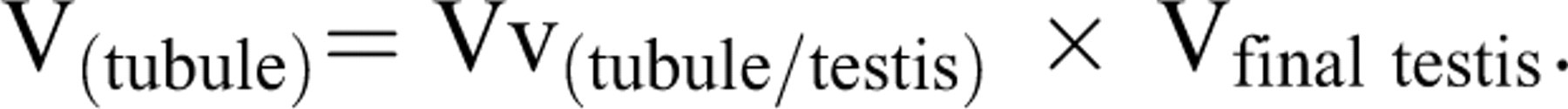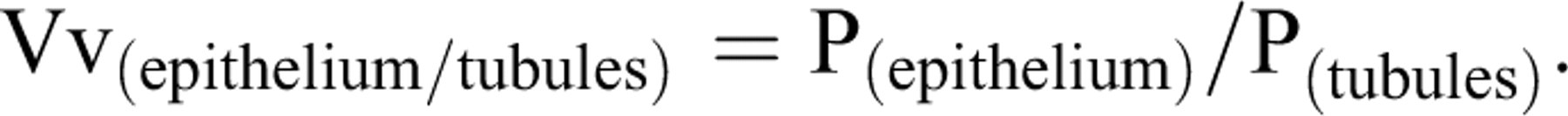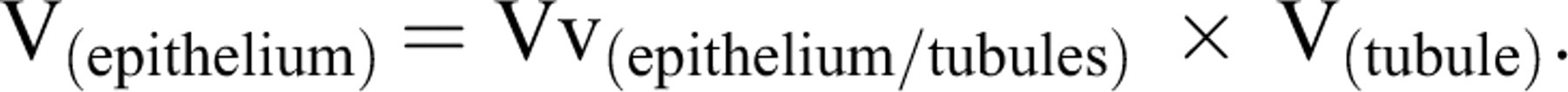Abstract
Metronidazole (MTZ) has negative effects on sperm analysis and testis structure. Curcumin is the principal curcuminoid found in turmeric and exhibits antitumor, anti-inflammatory, and anti-infectious activities with low toxicity. To evaluate stereological changes of seminiferous germinal epithelium by MTZ and ameliorative effects of curcumin, Balb-c mice were divided into 6 groups. The control, second, third, fourth, fifth, and sixth groups were administrated distilled water, high doses of MTZ (500 mg/kg/day), MTZ (500 mg/kg/day) + 100 mg/kg/d curcumin, therapeutic doses of MTZ (165 mg/kg/day), MTZ (165 mg/kg/day) + 100 mg/kg/day curcumin, and 100 mg/kg/day curcumin, respectively. Testis weight, testis volume, total epithelial volume, spermatocytes, and spermatid number showed a significant reduction in MTZ-treated (high or therapeutic doses) animals in comparison with the controls. Curcumin can protect spermatocytes after high or therapeutic doses of MTZ treatment. But curcumin is able to protect the other parameters only in the mice receiving therapeutic doses of MTZ. The total number of sertoli cells and spermatogonia did not show any significant difference in the mice that received MTZ. MTZ can reduce germinal epithelium volume and the number of spermatocytes and spermatids. Ameliorative effects of curcumin can mainly be observed in the mice receiving therapeutic doses of MTZ.
Introduction
Metronidazole (MTZ) is active against both anaerobic bacteria and protozoa (Freeman, Klutman, and Lamp 1997). It has been reported that six-week treatment with metronidazole decreases testicular and epididymal weights and testicular and epididymal spermatid counts and causes abnormal sperm morphology with degeneration of seminiferous tubules (Grover et al. 2001). It has been shown that after treating rats with MTZ (400 mg/kg per day), degeneration of the seminiferous epithelium in the testes of rats was observed; the tubules were generally devoid of primary or secondary spermatocytes and spermatids (McClain, Downing, and Edgcomb 1989). Qualitative aspects of histological alteration of seminiferous epithelium have been studied, but quantitative and stereological study of the MTZ on the epithelium has received less attention. In the present study, testis volume, total germinal epithelium volume, number of sertoli cells, spermatogonia (type A and B), spermatocytes, and long and round spermatid were evaluated using stereological methods.
The effects of curcumin on the sperm parameters are researched in this study. Curcumin is the main coloring principle of turmeric (Curcuma longa; Sharma, Gescher, and Steward 2005). Turmeric reveals antitumor, anti-inflammatory, anti-infectious (Duvoix et al. 2005) and antioxidant (Cohly et al. 1998) activities with low toxicity. Protective effects of curcumin on L-thyroxine–induced testicular oxidative stress (Sahoo, Roy, and Chainy 2008) or aflatoxin-induced toxicity in mice have been shown (Mathuria and Verma 2008). Thus, on the basis of these findings, the present study investigated the potential ameliorative effects of curcumin against high or therapeutic doses of MTZ-induced toxicity on the testis volume, total germinal epithelium volume, number of sertoli cells, spermatogonia (type A and B), spermatocytes, and long and round spermatids.
Materials and Methods
Ethics
All animal experiments were approved by the Animal Ethics Committee of Shiraz University of Medical Sciences.
Animals
To evaluate histological changes by MTZ and ameliorative effects of curcumin, 48 Balb-c mice weighing between 35 and 40 g were selected from the laboratory animal center of Shiraz University of Medical Sciences. The animals were housed in cages where humidity and temperature were kept constant. They had free access to food and water. The mice were acclimatized under laboratory conditions for two weeks prior to the experiment and were divided into six groups, each including eight mice. The control group received distilled water. The second group was administrated high doses of MTZ (500 mg/kg/day; Freeman, Klutman, and Lamp 1997), the third group high doses of MTZ (500 mg/kg/day) and 100 mg/kg/day curcumin (Shu et al. 2009), and the fourth group therapeutic doses of MTZ (165 mg/kg/day; Freeman, Klutman, and Lamp 1997). The fifth group received therapeutic doses of MTZ (165 mg/kg/day) and 100 mg/kg/day curcumin, and the sixth group received 100 mg/kg/day curcumin. All of the administrations were done by oral gavages for thirty days.
Stereological Study
On the last day of the experiment, the animals were weighed and then anesthetized. Animal fixation was by cardiac perfusion with formaldehyde buffer. The testis was weighed, and the primary volume (Vprimary) was measured using the immersion method (Scherle 1970). Briefly, a container with distilled water was placed on the scale and weighed, and then the testis suspended by a thin thread was immersed in the container so that it was fully covered by water and did not touch the bottom of the container. The new weight in grams, minus the weight of the container and water, divided by the specific gravity of distilled water (~1.0) was the volume of the testis in cubic centimeters. The final volume of the testis should be estimated in a stereological study to prevent reference trap (Braendgaard and Gundersen 1986). Different methods, such as Cavalieri methods, can be used for estimating the testis volume, but this technique needs consecutive sections, and it is time-consuming. In this study, the final testis volume was estimated after tissue processing and staining without the need for consecutive sections, after shrinkage estimation. Estimation of some of the stereological parameters such as shrinkage requires isotropic uniform random sections (i.e., the testis has a random position and orientation during sectioning and all its parts have an equal chance to be sectioned; Mühlfeld, Nyengaard, and Mayhew 2009). These sections were obtained by the orientator method (Figure 1). Briefly, the testis was placed on a circle divided into 10 equal distances; a random number between 0 and 10 was selected, and the testis was sectioned into two halves, with a blade at that direction. The cut surface of each half of the lobe was then placed on the 0-0 direction of the second circle with ten unequal cosine-weighted divisions, and the second cut was done. Each half was sectioned into slabs with a blade, placed in the direction of the second cut. Then, the eight to ten slabs were collected. A circle was punched from a testis slab by a trocar (Figure 2). The diameters of the circular piece of the testis were measured by a micrometer and the area of the circle estimated using a usual formula for calculating the area of a circle. The cut surfaces of the slabs and circular piece were embedded in a paraffin block, and only 2 sections with 5- and 15-μm thicknesses were obtained. The sections were stained with Heidenhain’s azan. After staining, the area of the circular piece was measured again, and the volume shrinkage (Mühlfeld, Nyengaard and Mayhew 2009) was calculated as
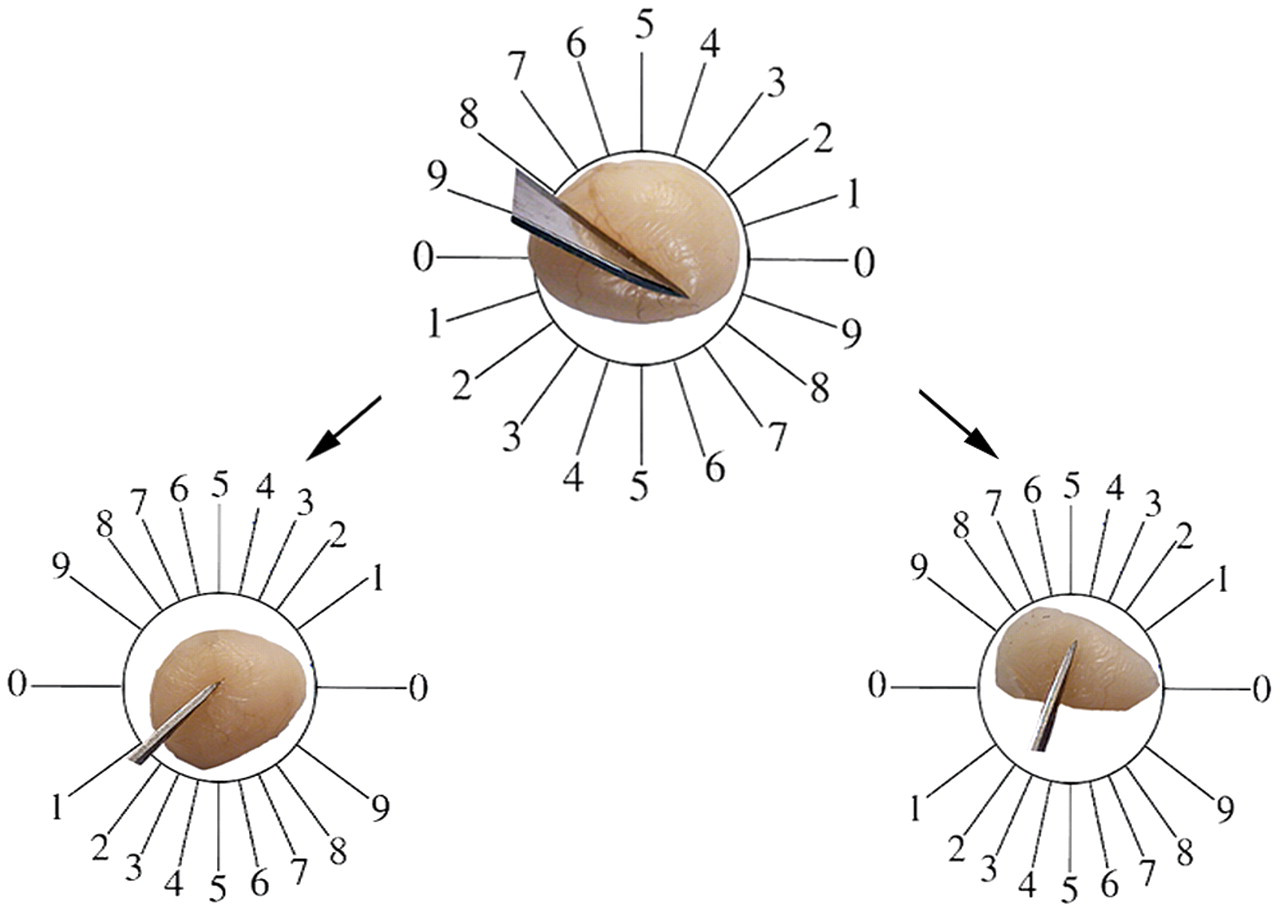
Orientator method, to achieve the isotropic uniform random sections. (Top) The whole testis is placed on a circle divided into 10 equal distances. A random number between 0 and 10 is selected (here 8), and the testis is sectioned into two halves, with a blade at that direction. (Bottom) The cut surface of each half of the testis is then placed on the 0-0 direction of the second circle with ten unequal cosine-weighted divisions and the second cuts done (here 1 and 4).
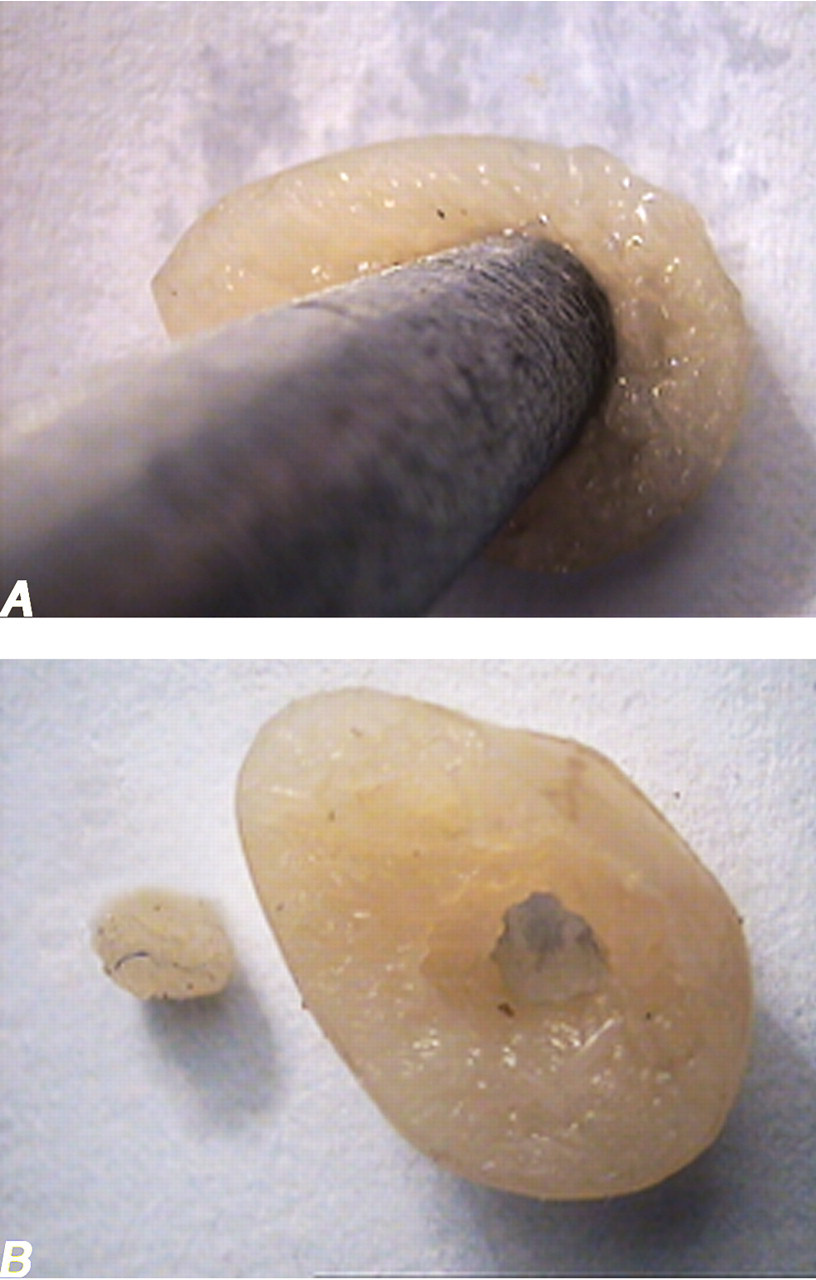
(A) A circle is punched from a testis slab by a trocar. (B) The diameter of the circular piece and the area of the circle are estimated.
where AA and AB are, respectively, the area of the circular piece after and before processing, sectioning, and staining.
After estimating the shrinkage, the final volume of the testis (the reference space) was estimated using
Each sampled section was analyzed using a video-microscopy system made up of a microscope (E-200, Nikon, Japan) linked to a video camera (Sony, Japan, SSC Dc 18P), a computer, and a flat monitor. On average, for estimating each parameter, ten to fourteen microscopic fields were examined in each testis for any estimation. In selection of microscopic fields, a systematic random sampling procedure was applied (i.e., the position of the first field was selected randomly outside the sections) on one corner of the microscopic slide. Then, the slide was moved at equal intervals along the x- and y-axis using a stage micrometer. This procedure was continued until all of the sections had been studied. Test probe or stereology grids were laid on the microscopic images by means of the stereology software designed at our laboratory (Morphometry & Stereology Research Laboratory, Shiraz University of Medical Sciences, Shiraz, Iran).
Estimation of Seminiferous Tubule and Germinal Epithelium Volume
To estimate the total volume of the seminiferous tubules and germinal epithelium, 5-μm sections were used. A multicascade design was applied to estimate the stereological parameters. A grid of points was overlaid on the monitor image of the testis (Figure 3 ). At the first level of multicascade estimation, the volume density of the tubules (Vv(tubule/testis)) or the ratio of the testis volume occupied by tubules was obtained using point counting at final magnification of 19× and the following formula:
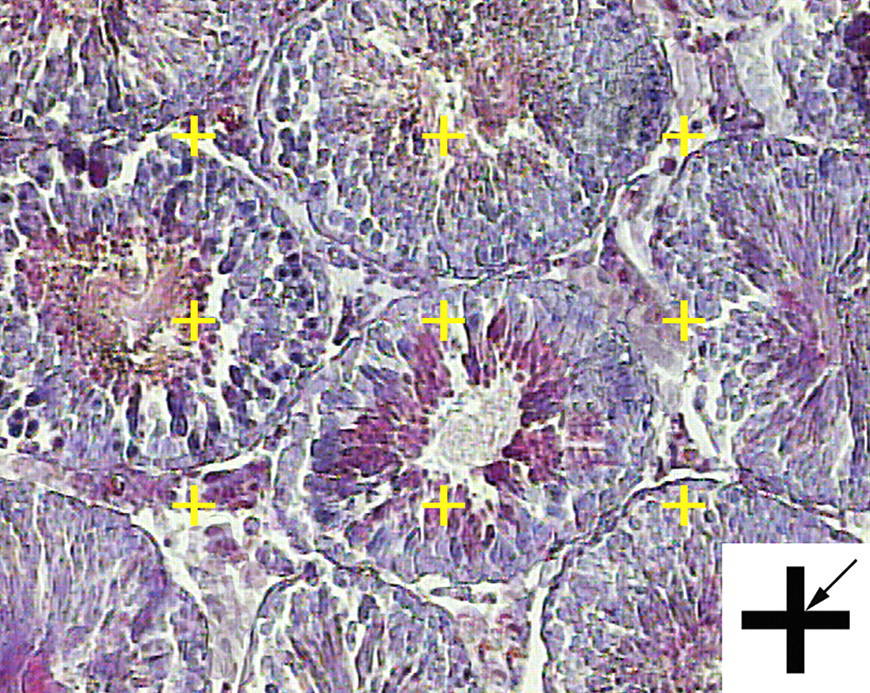
Volume density estimation using point counting. The total number of points hitting the interstitial tissue, lumen, or epithelium is counted and divided by the total number of points hitting the reference space (here testis). At the corner of this figure, a point is presented. The right upper corner of the cross is considered as the point (arrow), and it is counted only if the right upper corner hits the tissue.
where the P(tubules) and P(testis) were the number of points hitting the profiles of the tubules and testis, respectively. It is only in the knowledge of the size (volume) of the reference space that the character of any variation (or lack of variation) can be fully understood. A wrong conclusion has been made by ignoring the reference volume and comparison of densities only. This phenomenon has been termed the reference trap (Braendgaard and Gundersen 1986). To prevent the reference trap, the total (absolute) volume was obtained by multiplying the density by the final testis volume, that is,
At the second level of the multicascade design, a grid of points (Figure 3) was overlaid on the image of tubules at the magnification of 180× to estimate the volume density of the germinal epithelium Vv(epithelium/tubule), using the following formula:
The total (absolute) volume of the epithelium V(epithelium) was obtained using the following formula:
Estimation of the Total Number of Sertoli Cells, Spermatogonia, and Spermatids
To estimate the total number of sertoli cells, spermatogonia (type A and B), spermatocytes, and spermatids (round and long), the 15-μm-thickness sections were used. By means of the stereology software, an unbiased counting frame was superimposed on the images of the testis sections viewed on the monitor. The position of the microscopic field was selected by systematic random sampling. A high numerical aperture 100× magnification (NA = 1.4) oil immersion lens was used. The number of cells was estimated using the disector method at the final magnification of 4700×. Disector is a method in which the cells were selected with uniform random probability free from assumption of size and shape. By optical disector (Figure 4), the cells are sampled in thick microscopic sections observed with a microscope (Mühlfeld, Nyengaard, and Mayhew 2009). An unbiased counting frame with inclusion (right and upper) and exclusion (left and lower) borders was superimposed on the images (Figure 4). This frame avoids the edge effect and biased counting of the particles, and all of the nuclei profiles regardless of their shape are counted by the frame and have the same probability of being sampled. The optical section was moved downward on the z-axis. Using a microcator (Heidenhain MT-12, Germany) that measures the z-axis (depth), any cell whose nucleus was in focus at the first 5-μm plane was ignored. Any nucleus that came into maximal focus within the next traveling 5-μm optical plane (height of disector) was selected if it laid in the counting frame or touched the inclusion borders and did not touch the exclusion borders of the frame (Figure 4). The numerical density (Nv) or number of the cells in the unit volume of the germinal epithelium was estimated using Nv = ∑Q/∑A × h, where ∑Q was the number of nuclei coming into focus, ∑A was the total area of the unbiased counting frame in all fields (area of each frame was 168 μm2), and h was the height of the disector (5 μm here; Mühlfeld, Nyengaard, and Mayhew 2009). On average, 100 to 120 cells were counted in each animal. The total number of the nuclei was estimated by multiplying the numerical density (Nv) by the total volume of the epithelium (V (epithelium) ).
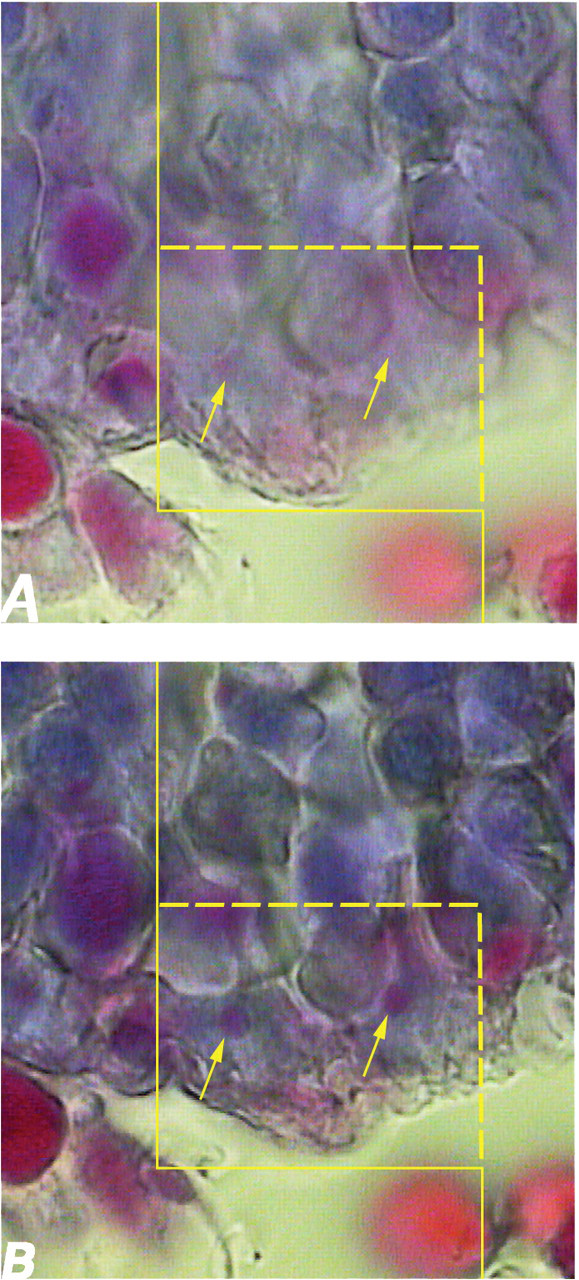
(A) To estimate the total number of the cells (here sertoli cells) using the optical disector method, an unbiased counting frame is superimposed on the images. (B) Any nuclei of sertoli cells that come into maximal focus within the traveling optical section (height of disector) are counted (here two; arrows).
Statistical Analysis
The data are reported as means and standard deviations. Statistical comparisons between group means were done by Mann-Whitney U test and one-way analysis of variance. P values less than 0.05 were considered significant.
Results
All data, P values, and differences are presented in Tables 1 and 2, and here the main differences are reported.
Mean ± Standard Deviation of the Testis Weight (mg), Volume (mm3) of the Testis, and Total Germinal Epithelium in the Control and the Mice Treated with High or Therapeutic Doses (500 or 165 mg/kg/day, Respectively) of Metronidazole (MTZ) with or without Curcumin (CUR)
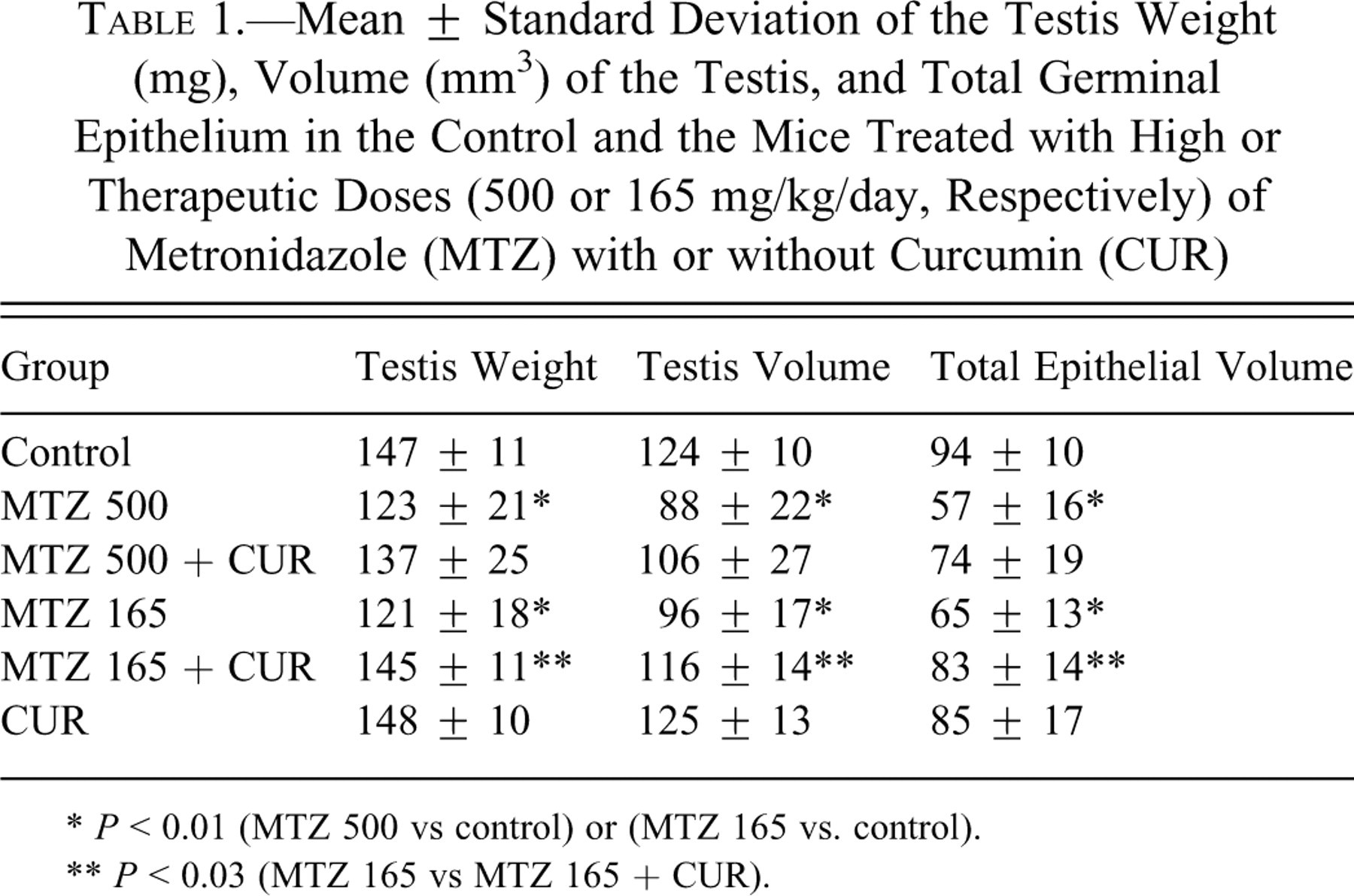
* P < 0.01 (MTZ 500 vs control) or (MTZ 165 vs. control).
** P < 0.03 (MTZ 165 vs MTZ 165 + CUR).
Mean ± Standard Deviation of the Number (×106) of the Different Cells of Germinal Epithelium in the Control Group and in the Mice Treated with High or Therapeutic Doses (500 or 165 mg/kg/day, Respectively) of Metronidazole (MTZ) with or without Curcumin (CUR)
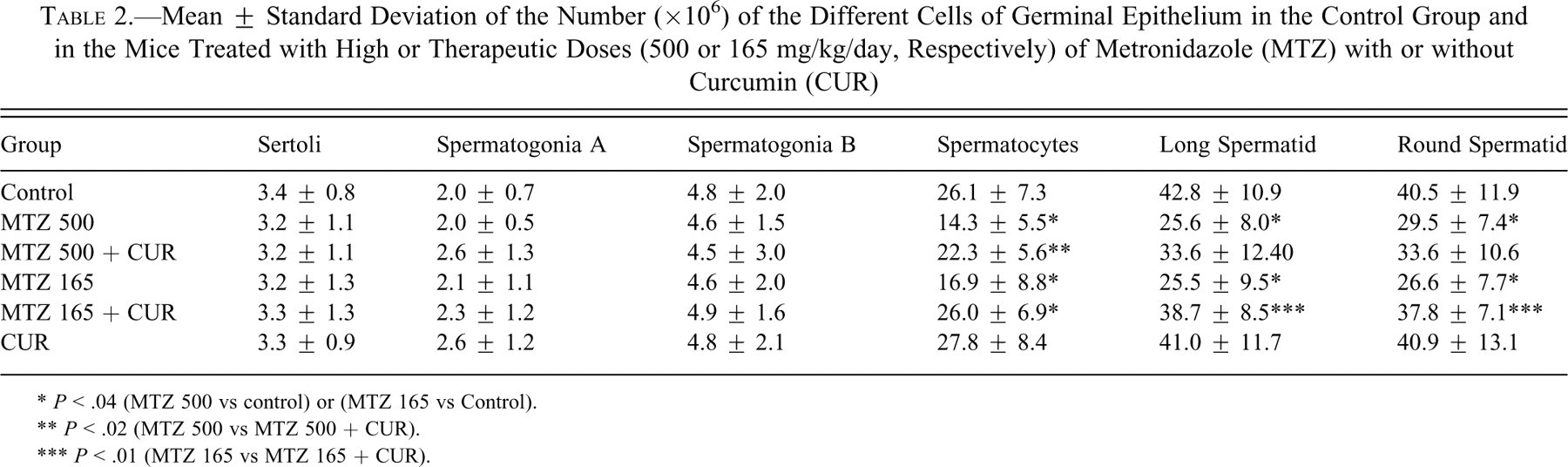
* P < .04 (MTZ 500 vs control) or (MTZ 165 vs. Control).
** P < .02 (MTZ 500 vs MTZ 500 + CUR).
*** P < .01 (MTZ 165 vs MTZ 165 + CUR).
Testis Weight and Volume
The testis weight was reduced ~17% in the high or therapeutic dose of MTZ-treated animals in comparison with the controls (P < 0.01). The testis volume was reduced ~29% and ~23% in high or therapeutic doses of MTZ-treated animals, respectively, in comparison with the controls (P < 0.01). Combined treatment with high doses of MTZ and curcumin did not ameliorate the reduction, but testis weight and volume increased after treatment of mice with therapeutic doses of MTZ and curcumin, in comparison with the related MTZ treatment only (P < 0.03; Table 1).
Total Germinal Epithelium Volume
The data show that total epithelial volume in animals treated with high or therapeutic doses of MTZ has decreased ~39% and ~31% in comparison with the controls (P < 0.01). It is obvious from Table 1 that the animals that received high doses of MTZ and curcumin did not show differences in comparison with those who received high doses of MTZ. Treatment with therapeutic doses of MTZ and curcumin ameliorate the epithelial volume changes in comparison with therapeutic doses of MTZ only (P < 0.03).
Number of Sertoli Cells and Spermatogonia A and B
The total number of sertoli cells, spermatogonia A, and spermatogonia B did not show any significant difference in the mice which received high or therapeutic doses of MTZ in comparison with the controls (Table 2).
Number of Spermatocytes
The number of spermatocytes decreased ~45% and ~35% in high or therapeutic doses in MTZ-treated animals in comparison with the controls, respectively (P < 0.04). The combined treatment of curcumin with high (P < 0.02) or therapeutic (P < 0.01) doses of MTZ ameliorated these changes, and number of cells increased significantly (Table 2).
Number of Long and Round Spermatid
The number of long spermatids decreased ~40% in mice treated with high and therapeutic doses of MTZ in comparison with the control mice (P < 0.04). The number of round spermatids also decreased ~27% and ~34% in the animals treated with high or therapeutic doses in comparison with the control group, respectively (P < 0.04). No significant recovery was seen in the number of spermatids after combined treatment of curcumin and high doses of MTZ, but ameliorative effects of curcumin on the number of these cells were seen in mice after treatment with curcumin and a therapeutic dose of MTZ (P < 0.04; Table 2).
Discussion
The present study reports the ameliorative effects of curcumin on the seminiferous epithelium in the mice treated with high and therapeutic doses of MTZ. The testis volume, total germinal epithelium volume, number of sertoli cells, spermatogonia (type A and B), spermatocytes, and long and round spermatids are the parameters studied.
The first investigated parameter was testis weight and volume. McClain, Downing, and Edgcomb (1989) and el-Nahas and el-Ashmawy (2004) showed that MTZ decreased the weight of the testis. This is in line with the present results.
Another studied parameter was total epithelial volume. In a qualitative histological study, McClain, Downing, and Edgcomb (1989) reported that severe degeneration of the seminiferous epithelium was seen after treatment with MTZ. We reported the epithelial changes as total epithelial volume, which is a comparable parameter. This quantitative parameter supports the report of McClain, Downing, and Edgcomb (1989).
The present study showed that the number of sertoli cells (nursing and supporting cell of germinal cells) and spermatogonia (the generators of other germinal cells) were not altered significantly and that the changes by MTZ were started from spermatocytes (primary and secondary) and continued in spermatids. The data presented here are in the same line with those of McClain, Downing, and Edgcomb (1989), who reported that the tubules were generally devoid of primary or secondary spermatocytes and spermatids after MTZ treatment. Some authors explain that the mean serum follicle-stimulating hormone, luteinizing hormone, and testosterone levels were also lowered in MTZ-treated animals (Grover et al. 2001; McClain, Downing, and Edgcomb 1989). The toxic effects of MTZ were probably mediated by a decrease in the circulating hormones responsible for spermatogenesis (Grover et al. 2001; McClain, Downing, and Edgcomb 1989).
Interestingly, curcumin ameliorates some of these effects. The present study showed that curcumin can protect spermatocytes after high or therapeutic doses of MTZ treatment. But curcumin is able to protect the round or long spermatid, testis weight, volume, and germinal epithelium volume only in mice receiving therapeutic doses of MTZ. Protective effects of curcumin have been reported in oxidative damage induced by cisplatin, sodium arsenite (Ilbey et al. 2009; El-Demerdash et al. 2009), and aflatoxin-induced biochemical changes and ischemia-reperfusion injury in rat testis (Mathuria and Verma 2007; Wei, Yan, and Zhou 2009). It has been reported that curcumin recovers the histological structure of the testis after di-n-butylphthalate–induced testicular damage (Farombi et al. 2007).
In conclusion, high or therapeutic doses of MTZ can decrease the testis volume, total germinal epithelium volume spermatocytes, (type A and B), and long and round spermatids in mice. The number of sertoli cells and spermatogonia was not affected by MTZ. Curcumin can protect spermatocytes after high or therapeutic doses of MTZ treatment. But curcumin is able to protect the round or long spermatid, testis weight, volume, and germinal epithelium volume only the mice receiving therapeutic doses of MTZ.
Footnotes
Acknowledgment
The work was carried out at Histomorphometry & Stereology Research Centre of Shiraz University of Medical Sciences. The work was financially supported by grant 87-4412 from Shiraz University of Medical Sciences.