Abstract
A spongy change in the spinal cord white matter was observed in four-week-old rats treated with aniline. Although this change was found to be a result of the myelin sheath splitting at the ultrastructural level, the mechanism is unknown. This study was conducted to identify the mechanism of the spongy change in aniline-treated rats. The spongy change in the spinal cord white matter was first detected on day 5 in the histopathologic examination. The incidence and severity of the lesions, especially in the lateral and ventral funiculi of the thoracic spinal cord white matter, increased prominently from day 8 to day 10. In all rats, immunohistochemical staining by anti-2′, 3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) occurred along the cytoplasmic boundaries of the normal oligodendroglia. However, mild to moderate anti-CNPase staining extended to the swollen cytoplasm of the oligodendroglia in the aniline-treated rats from day 2 to day 4. In the electron microscopic examination, free ribosomes and rough endoplasmic reticula in the cytoplasm of the oligodendroglia increased on days 3 and 4. These changes were considered to be related to CNPase expression. However, CNPase expression decreased, whereas the spongy changes were detected from day 5. The reduction in CNPase expression may contribute to the changes in the myelin morphology observed in aniline intoxication.
Introduction
Aniline is a widely used industrial chemical, primarily as an intermediate in the synthesis of organic compounds such as rubber-processing chemicals, antioxidants, dyestuff, photographic chemicals, pharmaceuticals, and agricultural chemicals (International Agency for Research on Cancer [IARC] 1982; Kouris et al. 1963; Okazaki et al. 2001a,b). Four-week-old rats given a single oral dose of aniline developed spongy changes in the spinal cord white matter (Okazaki et al. 2001a,b). Aniline is suggested to exert its effect during myelinogenesis, as indicated by the absence of spongy changes in the spinal cord of aniline-treated seven-week-old and older rats (Okazaki et al. 2001a). In the electron microscopic examination, this spongy change corresponded to the splitting of the intraperiod line of the myelin sheath (Okazaki et al. 2001a,b). Membranous aggregates were detected in the cytoplasm of oligodendroglia in the spinal cord white matter on day 4 prior to the appearance of the spongy change in this area (Okazaki et al. 2001b). However, the pathological characteristics before day 4 and the pathogenesis of these electron microscopic lesions are unknown.
Several compounds, such as hexachlorophene, triethyl-tin, isonicotinic acid hydrazide, and cuprizone, have been reported to induce spongy changes in white matter (Blackmore et al. 1972; Jacobs, Cremer, and Cavanagh 1977; Lampert, O'Brien, and Garrett 1973; Love 1988; Purves, Garrod, and Dayan 1991; Towfighi 1980; Watanabe 1980). Although the spongy changes induced by these compounds are also caused by splitting of the intraperiod line of the myelin sheath at the ultrastructural level (Blackmore, Palmer, and Noel 1972; Jacobs, Cremer, and Cavanagh 1977; Lampert, O'Brien, and Garrett 1973; Love 1988; Towfighi 1980; Watanabe 1980), the mechanism is unknown. However, it has been suggested that some of these compounds induce the spongy change by damaging the oligodendroglia, which are responsible for myelin formation (Jacobs, Cremer, and Cavanagh 1977; Love 1988).
Myelin basic protein (MBP) (Berry and Butt 1997; Campagnoni 1988; Kanfer et al. 1989; Kursula 2001; Trapp et al. 1987), myelin proteolipid protein (PLP) (Berry and Butt 1997; Campagnoni 1988; Kanfer et al. 1989; Trapp et al. 1987), galactocerebroside (Gal) (Campagnoni 1988; Kanfer et al. 1989), and CNPase (Berry and Butt 1997; Campagnoni 1988; Kanfer et al. 1989; Kursula 2001; Trapp et al. 1988) are the specific markers expressed in either oligodendroglia or myelin. In particular, CNPase expression increases from postnatal day 15 to postnatal day 30 (Kanfer et al. 1989). Therefore, it is considered that CNPase plays an important role in myelinogenesis (Kanfer et al. 1989). In addition, CNPase is distributed to the periaxonal regions of the myelin sheath and the cell membrane regions of the oligodendroglia, thus it is also considered that CNPase is one of the markers of oligodendroglia differentiation (Trapp et al. 1988). However, CNPase function in vivo is still unknown.
To clarify the early spongy change in aniline-treated rats, we examined the spongy change chronologically by ultrastructural examination. In addition, to verify our hypothesis that the spongy change is attributable to activity of oligodendroglia forming the myelin sheath, we focused on the enzyme (CNPase), proteins (MBP, PLP), and lipid (Gal) that were expressed in the oligodendroglia and compose the myelin sheath. We report on the interesting findings regarding the behavior of CNPase expression.
Materials and Methods
Chemical
Aniline (C6H5NH2, 99.5% pure, Lot No. 05624BH, CAS No. 62-53-3) was obtained from Sigma-Aldrich Japan Co., Ltd. (Tokyo, Japan). Olive oil was purchased from Oriental Pharmaceutical Co., Ltd. (Yamagata, Japan) for use as a vehicle.
Animals
Three-week-old male Crl:CD (SD) IGS rats were obtained from Charles River Laboratories Japan, Inc. (Atsugi, Japan). They were given commercial diet (MF, Oriental Yeast Co., Ltd., Japan) and tap water ad libitum. Healthy rats were used for the study at four weeks of age after an acclimation period of five days. The animals were housed in polycarbonate cages. The animal room was maintained at a temperature of 20°C to 25°C, with a relative humidity of 55% ± 15% with a twelve-hr light/dark cycle.
The animals were cared for according to the principles outlined in the Guide for the Care and Use of Laboratory Animals prepared by the Japanese Association for Laboratory Animal Science and our institution.
Treatment
Twenty-one rats were stratified by body weight and randomly assigned to seven experimental groups (three rats per group) to give similar initial mean body weights. The animals were dosed once with either 1000 mg/kg aniline or olive oil by gavage. The dose level was determined based on the results of previous reports (Okazaki et al. 2001a, b). The experimental design is shown in Table 1 .
Experimental design.
Pathological Examination
Groups of aniline-treated (n = 14) and control (n = 7) rats were chronologically (days 2, 3, 4, 5, 8, 10, 12) euthanized by exsanguination from the right atrium under anesthesia with intraperitoneal injection of pentobarbital sodium (Nembutal injection, Dainippon Sumitomo Pharma Co., Ltd.) and perfused with 4% paraformaldehyde via the left ventricle. Cross-sections of the following tissues were collected: brain, spinal cord, trigeminal nerve, sciatic nerve. The samples were processed for paraffin embedding and stained with hematoxylin and eosin (HE).
Sections from the thoracic spinal cord were also subjected to an immunohistochemical staining using a labeled streptavidin-biotin system (DAKO LSAB2 System-HRP, DAKO Japan Ltd., Kyoto, Japan). Antibodies against human CNPase (1:400 in dilution, Chemicon International, Inc., Temecula, CA, USA), human MBP (1:25 in dilution, Serotec Ltd., Oxford, England), PLP (1:200 in dilution, Chemicon International, Inc., Temecula, CA, USA), and bovine Gal (1:200 in dilution, Chemicon International Inc., Temecula, CA, USA) were used as the primary antibodies. Regarding the antigen retrieval, the slides were transferred to 0.5% Immunosaver (Nisshin EM Corp., Tokyo, Japan), and heated for 25 minutes at 95°C using a 400W-MW processor (MI-77, Azumaya Corp., Tokyo, Japan) with the microwave treatment. The sources and working dilutions of the antibodies are shown in Table 2 .
Antibodies employed in immunohistochemical staining (sources and dilution).

a Double immunofluorescence staining.
The severity of the spongy change and the degree of anti-CNPase immunostaining were classified into four grades, and chronologically evaluated. The spongy change and anti-CNPase immunostaining were graded as follows. For the spongy change, score 0 indicates no particular change (-); score 1, mild spongy change (+); score 2, moderate spongy change (++); and score 3, severe spongy change (+++). For anti-CNPase immunostaining, score 1 indicates slightly positive reaction is observed along the cytoplasmic boundaries of the small oligodendroglia (+); score 2, slightly to moderately positive reaction is observed throughout the slightly swollen cytoplasm of the oligodendroglia (++); score 3, moderately positive reaction is observed throughout the slightly swollen cytoplasm of the oligodendroglia (+++); and score 4, strongly positive reaction is observed throughout the swollen cytoplasm of the oligodendroglia (++++).
Double immunofluorescence labeling was performed using anti-cow glial fibrillary acidic protein (GFAP, 1:400 in dilution, DAKO Japan Ltd., Tokyo, Japan) and anti-human CNPase (1:100 in dilution, Chemicon International, Inc., Temecula, CA, USA) antibodies. Immunoreactions were visualized by rhodamine for anti-GFAP antibody and by Alexa Fluor 488 for anti-CNPase antibody on the same sections. The sections were examined with a fluorescence microscope (BX-51, Olympus Ltd., Tokyo, Japan).
For electron microscopy, tissue fragments of the thoracic spinal cord were fixed in 4% paraformaldehyde for approximately twenty-four hours and postfixed in 1% osmium tetroxide for two hours. Sections of both gray and white matter were embedded in epoxy resin. Semithin sections were stained with toluidine blue. Ultrathin sections stained with uranyl acetate and lead citrate were examined with a transmission electron microscope (Hitachi H-7600, Hitachi High-Technologies Corp., Tokyo, Japan).
Results
Histopathological Findings
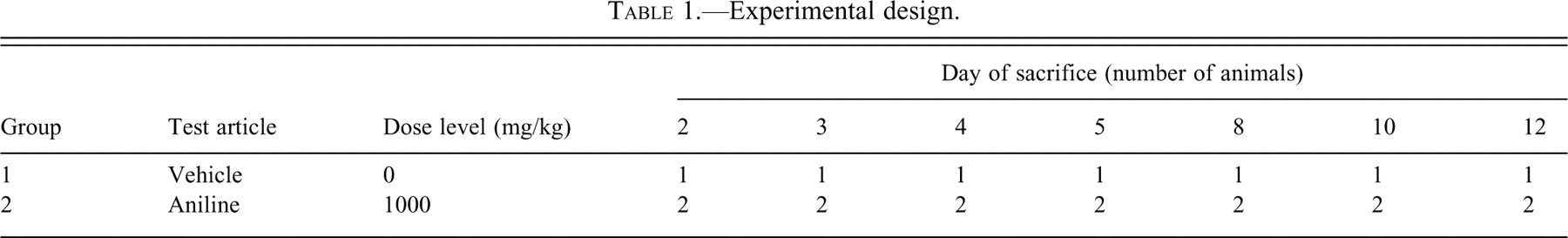
Spongy change was first detected on day 5 and thereafter. The lesions in the spinal cord were most severe, especially in the lateral and ventral funiculi in the thoracic spinal cord white matter (Figure 1 ).
Transverse section of the thoracic level in the spinal cord from rats treated once with 1000 mg/kg of aniline. Severe spongy changes were observed in the white matter after the aniline treatment on day 10. The box indicates in area of inset. Inset, high magnification of the severe spongy changes. HE staining.
Immunohistochemical Findings
Chronological changes were identified by CNPase immunostaining, whereas no changes were observed when the sections were immunostained for MBP, PLP, or Gal.
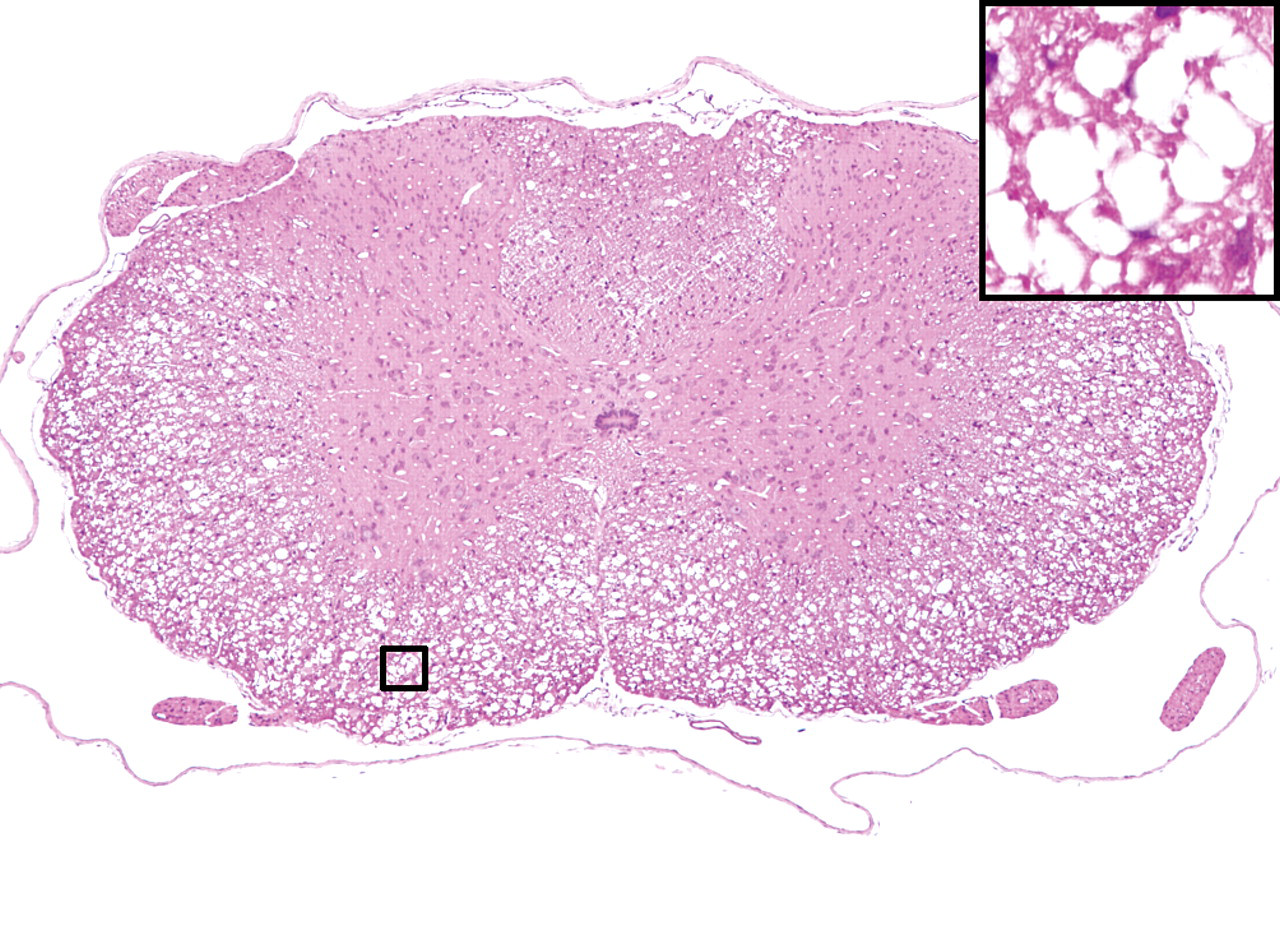
In aniline-treated rats, slight to moderate immunostaining of CNPase extended to the swollen cytoplasm of the oligodendroglia on day 2. The swelling of the oligodendroglia was obvious, and strong immunostaining of CNPase extended to the whole cytoplasm of the oligodendroglia from day 3 to day 4 (Figure 2B and 2C). Compared to the controls, there was no difference in the size or intensity of the positive reaction of the oligodendroglia on day 12. In the controls, the CNPase positive reaction was observed along the cell membrane of the normal oligodendroglia from day 2 to day 12 (Figure 2A).
Immunohistochemical staining of CNPase in the thoracic spinal cords. Positive reaction was observed along the cell membrane of the normal oligodendroglia in the controls (A, arrows). In aniline-treated rats, moderate to severe positive reaction was also observed in the swollen cytoplasm of the oligodendroglia (arrows) on day 3 (B) and day 4 (C). Inset, high magnification.
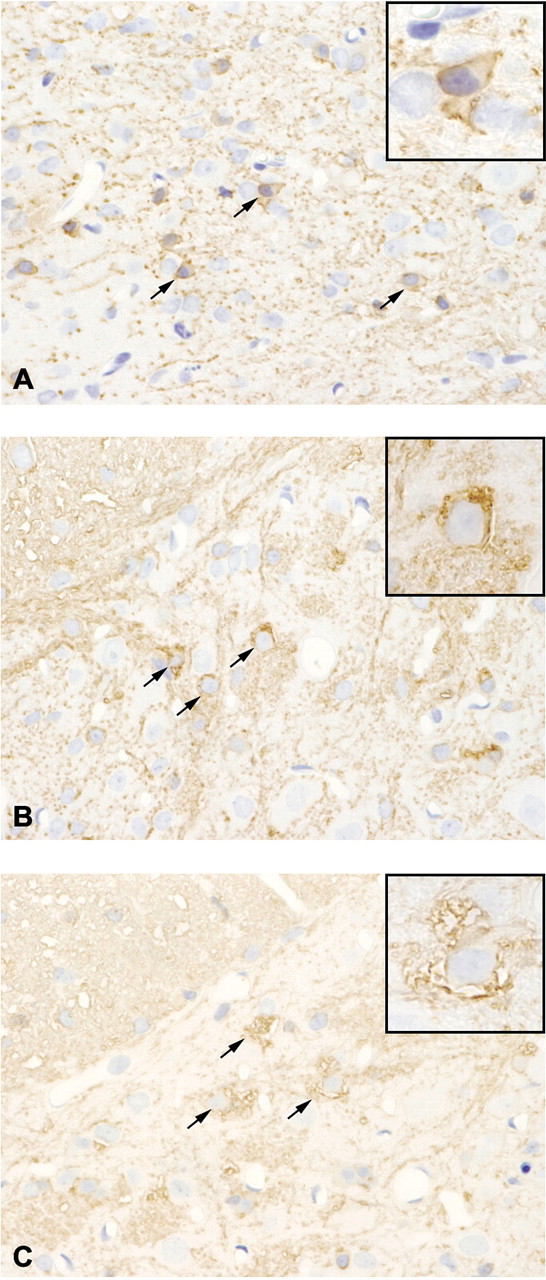
In double immunofluorescence labeling, the CNPase-labeled oligodendroglia indicated by green fluorescence (Figure 3 ) were distinguishably swollen in comparison to the GFAP-positive astrocytes, indicated by red fluorescence (Figure 3). No regions contained both green and red fluorescence (Figure 3).
Double immunofluorescence labeling for CNPase and GFAP of the thoracic level in the spinal cords. The CNPase-positive oligodendroglia visualized by Alexa Fluor 488 (green) were distinguishably swollen in comparison to the GFAP-positive astrocytes visualized by rhodamine (red). None of the cells attracted both immunolabels.
Electron Microscopic Findings
Splitting of the myelin sheath was found on days 2 and 3 (Figure 4A and 4B). The cytoplasm of the oligodendroglia invaded the myelin sheath. In addition, a small amount of membranous aggregates (MA) was seen in the cytoplasm of the oligodendroglia (Figure 4C and 4D). The MA included flocculent contents or myelin structures. The incidence of myelin sheath splitting increased on day 4. The free ribosomes and rough endoplasmic reticula in the cytoplasm of the oligodendroglia increased on days 3 and 4 (Figure 5B), which coincided with the immunohistochemical findings, suggesting enhanced protein synthesis. Mitochondrial swelling was also observed. The incidence and severity of the myelin sheath splitting increased prominently from day 8 to day 12. In addition, MA also increased intensively corresponding to the myelin sheath splitting.
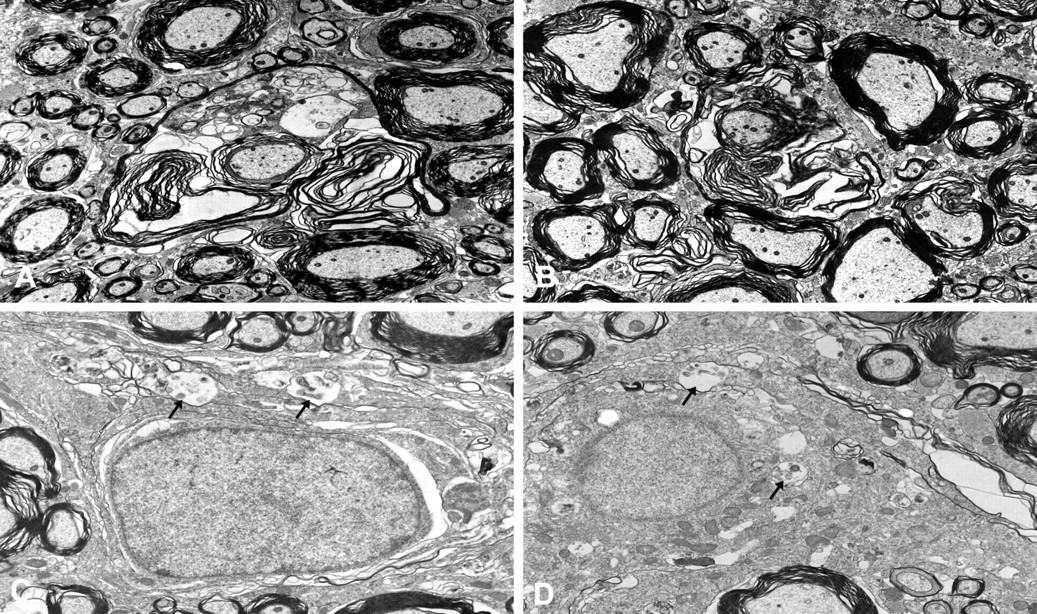
Electron photomicrographs of the white matter of the thoracic level in the spinal cords. Severe splitting of the myelin sheath was observed after the aniline treatment on day 2 (A) and day 3 (B). Small amounts of membranous aggregates (arrows) were also observed in the cytoplasm of the oligodendroglia on day 2 (C) and day 3 (D).
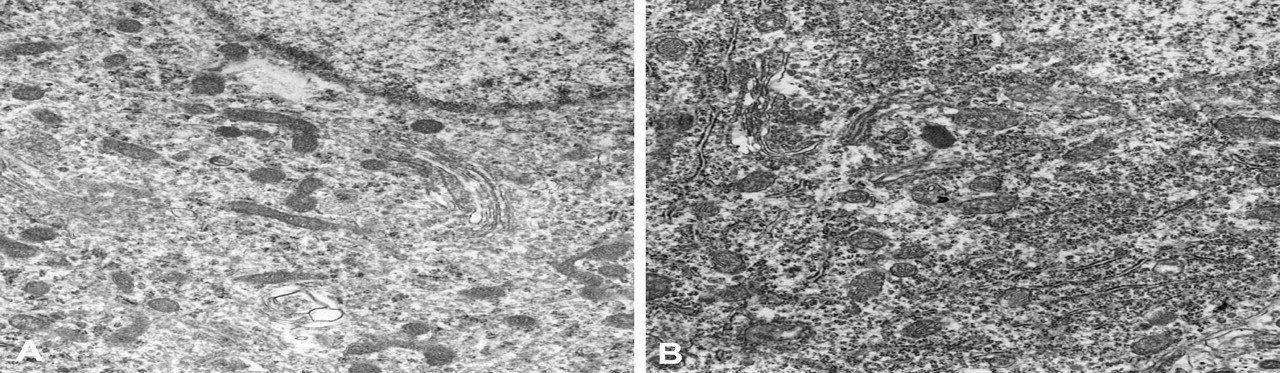
Electron photomicrographs of the white matter of the thoracic level in the spinal cord. Free ribosomes and rough endoplasmic reticula in the cytoplasm of the oligodendroglia were increased on day 4 (B) following the aniline treatment compared to the control (A).
Chronological Spongy Change and CNPase Immunoreaction
In the graph representing the severity of the spongy change and the degree of the CNPase-positive reaction, CNPase expression peaked on days 3 and 4 and then decreased immediately thereafter (Figure 6 ). Expression of CNPase decreased, whereas spongy changes were detected from day 5 and peaked from day 8 to day 10.
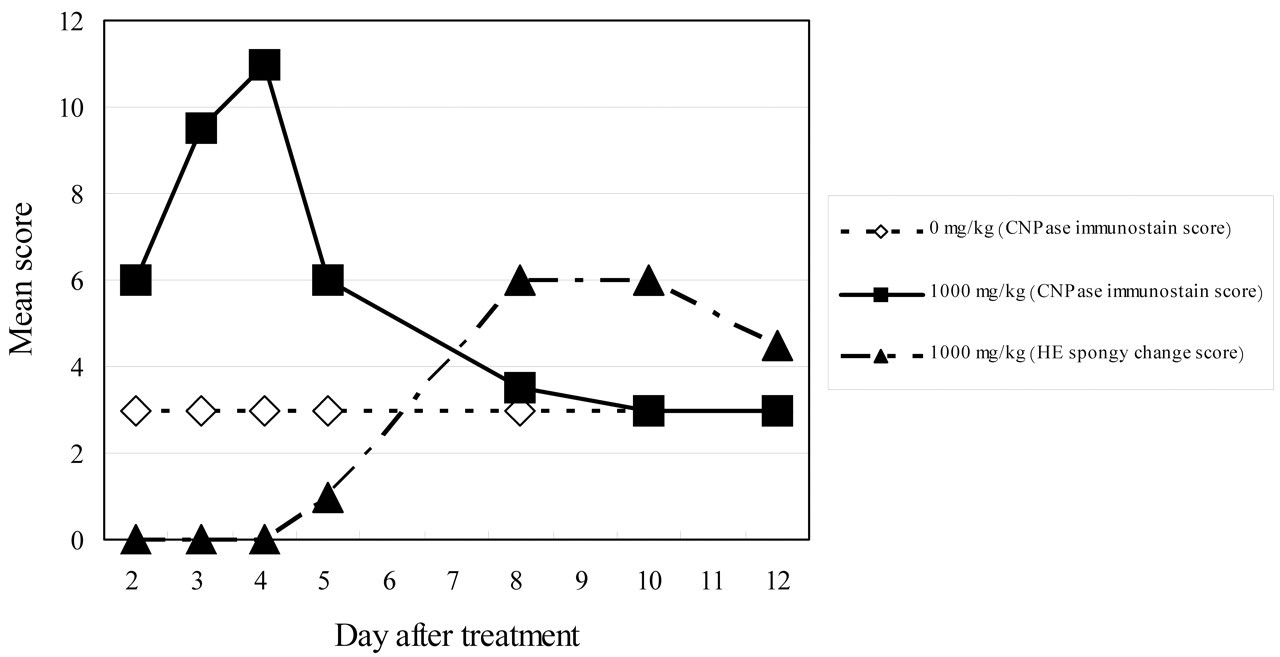
Mean score of the severity of the spongy change and the degree of the CNPase immunostaining in the spinal cords. Expression of CNPase peaked on days 3 and 4 and then decreased from day 5 to day 12, whereas the spongy changes were detected from day 5 and peaked from day 8 to day 10.
Discussion
Although the spongy change induced by the aniline treatment was detected from day 5 in the histopathological examination, the splitting of the myelin sheath was observed on days 2 and 3 at the ultrastructural level. Okazaki et al. reported that the splitting of the myelin sheath was detected on day 8 (examination point: days 4, 8, 12, 15, 22, 30, and 60) (Okazaki et al. 2001b). As a result of the early stage examination following the aniline treatment, this change was first observed on days 2 and 3 ultrastructurally. In addition, MA in the oligodendroglia was observed on day 2 and thereafter. The incidence and severity of MA increased prominently and the MA occupied most part in cytoplasm of individual oligodendroglia by day 8. These changes were considered to be the early stage of the spongy change induced by aniline.
Although the nature of MA in the cytoplasm of the oligodendroglia is unknown, MA is observed in the cases of abnormal utilization of lipid components (Ghadially 1988; Okazaki et al. 2001b). It is also observed in the cytoplasm of the oligodendroglia in black tremor hamsters (Kim et al. 2003) and quaking mice (Watanabe and Bingle 1972) with impaired myelinogenesis in the central nervous system. Accordingly, it is suggested that MA in the cytoplasm of the oligodendroglia is attributed to abnormal myelinogenesis.
The expression of immunostained CNPase increased from day 2 and peaked on days 3 and 4. In the electron microscopic examination, free ribosomes and rough endoplasmic reticula in the cytoplasm of the oligodendroglia increased on days 3 and 4, which indicated enhanced protein synthesis (Ghadially 1988). On the other hand, the spongy change in the spinal cord was detected from day 5, after which CNPase expression decreased remarkably. As for the organophosphorous compounds that induce degenerative change in the central nervous system of hens, it has been reported that expression of CNPase was decreased by tri-o-cresyl phosphate, di-isopropyl fluorophosphates, or leptophos (Olajos 1987), and increased by S, S, S-tri-n-butyl phosphorotrithioate (Abou-Donia et al. 1986; Olajos 1987).
It has also been reported that CNPase mRNA and protein increased rapidly and peaked by 30 minutes after surgery in the rat model of total section of the spinal cord, and gradually decreased to the control level by 1 week (Morin-Rechaud, Feldblum, and Privat 1998). Accordingly, acceleration of CNPase synthesis is suggested in the oligodendroglia from immediately after the aniline treatment to the onset of the spongy change. From the CNPase changes observed in the rat model or by treatment of compounds inducing neurodegenerative lesions other than aniline, it was suggested that the cellular activity of oligodendroglia was accelerated by the toxicity of aniline, resulting in changes in CNPase distribution over time. On the other hand, no changes were observed in MBP, PLP, or Gal in the present study. CNPase is distributed mainly between the myelin sheath and the axon, whereas MBP and PLP are in the myelin sheath (Brunner et al. 1989; Ferreira et al. 2007). The difference in the distribution pattern may be attributed to differences in enzyme, protein, or lipid expression after the aniline treatment. Consequently, it has been suggested that aniline specifically affected the region containing CNPase, eventually increasing CNPase production. Furthermore, Gal is reported to rapidly increase in rats between postnatal days 10 and 17 (Bichenkov and Ellingson 1999). As Gal was hardly stained even in the normal spinal cord, Gal may rarely occur in four- or five-week-old rats. Our study also suggested a close relation between CNPase and the spongy change in the spinal cord, as a number of changes were detected in CNPase, despite the absence of any changes in MBP, PLP, or Gal.
In summary, CNPase expression, myelin sheath splitting, and membranous aggregartes were found from day 2, the earliest time point evaluated, after treatment with a single dose of aniline at 1000 mg/kg. CNPase activity peaked on day 4, however, it decreased remarkably from day 5, which corresponded with the onset of spongy change detectable by light microscopy. It was concluded that although it was not apparent whether the CNPase expression was attributable to the direct or indirect action of aniline on the oligodendroglia, aniline was considered to specifically affect the region containing CNPase. Therefore, a close relation between CNPase and spongy change in the spinal cord was suggested. Further investigations are required to clarify the still unknown role of CNPase in the impaired central and peripheral nervous systems.
Footnotes
Acknowledgments
The authors thank Mr. Hiroyuki Ishii and Ms. Atsuko Ohnobu for assistance in the investigation, and Mr. Takayoshi Ito and Ms. Chie Shiroumaru for their excellent technical assistance. The authors also thank Mr. Steve Yamakami, Ms. Kanae Tamatsukuri, Ms. Ai Ishi, and Ms. Azumi Takahashi for language editing.
