Abstract
Alzheimer’s disease (AD) in humans is a progressive neurogenic disease that can be linked with such characteristic pathological findings in the cerebrum as senile plaques (SPs), neurofibrillary tangles (NFTs), cerebral amyloid angiopathy (CAA), and neuronal loss. In the present study, the authors investigated the age-related morphological changes in 12 middle-aged and 12 young cynomolgus monkeys. Low numbers of neurons and astrocytes in the hippocampal region in cynomolgus monkeys accompanied ageing, and there was a high number of microglial cells; however, no clearly neurotoxic abnormalities due to β-amyloid were noted before the age of 20 years. The onset of SPs and CAA in the cerebrum in cynomolgus monkeys can occur before the age of 20 years. SPs were almost all categorized as diffuse plaques (DPs); they did not have amyloid cores and were unaccompanied by neuritic degeneration. In cynomolgus monkeys, SPs (DPs) occur before the appearance of CAA. From the above, it was concluded that cynomolgus monkeys showed pathological changes due to ageing similar to those related to Alzheimer’s disease in humans, even before they were 20 years old.
Introduction
In the increasingly ageing society of today, the problem of the neurodegenerative condition Alzheimer’s disease (AD) has become global in scope. Excluding the case of familial AD (FAD) (Tanzi and Bertram 2001), the great majority of AD cases occur as sporadic AD (SAD) in elderly people, although the pathogenetic mechanism has long been unclear.
In humans, AD is a progressive neurogenic disease regarded as a cardinal sign of dementia, which can be linked with such characteristic pathological findings in the cerebrum as senile plaques (SPs), neurofibrillary tangles (NFTs), cerebral amyloid angiopathy (CAA), and neuronal loss (Selkoe 2001). SPs are small deposits of β-amyloid protein (Aβ). There are three types of SPs: primitive plaques (PPs), which are swellings of portions of neuritic degeneration; classical plaques (CPs), which have an amyloid core in the central part; and diffuse plaques (DPs), which do not represent neuritic degeneration (Nakamura et al. 1995). β-amyloid protein deposits in the cerebral parenchyma and small blood vessels of the meninges and capillaries represent CAA. β-amyloid protein is generated by abnormal cleavage of the amyloid precursor protein (APP), an intrinsic membrane protein of unknown function; and when flocculation occurs, it shows strong toxicity to the cerebral cortex and hippocampus neurons. As a result, fibrillary tangle structures, NFTs, are formed in the cytoplasm of neurons. NFTs are produced by phosphorylated tau proteins tightly bound to cellular microtubules and considerably reduce neuron performance. When the accumulation of NFTs becomes advanced, neurons undergo necrosis and, as a result, the patient exhibits severe dementia. This is called the amyloid cascade hypothesis of AD onset (Hardy and Selkoe 2002).
In recent years, the monkey has been widely used in safety studies because it is taxonomically close to humans and has similar pharmacological and toxicological reactions and drug absorption, distribution, metabolism, and excretion. Amongst monkey species, the cynomolgus monkey is widely used in safety studies for reasons such as ease of breeding. However, up until now there have been very few reports on spontaneous occurrence of pathological changes with ageing in monkey species; in particular, reports on age-related changes in the brain are limited, and reports on the occurrence of SPs are very few. The cynomolgus monkey has come to be the focus of attention for establishing AD models because SPs, the main pathological change associated with AD, spontaneously occur in this species after reaching a certain age. In the present study, we investigated the presence and distribution of SPs, NFTs, and CAA and neuronal loss in both middle-aged and young cynomolgus monkeys, using various special staining and immunohistochemical methods. This is among the first studies to report on the numbers of neurons, astrocytes, and microglial cells in the hippocampus in the cynomolgus monkey. Furthermore, cerebrums with age-related lesions were compared with those of young monkeys.
Materials and Methods
Animals
Twenty-four female cynomolgus monkeys (Macaca fascicularis) were used in this study: 12 young monkeys (aged 3 to 5 years) and 12 middle-aged monkeys (aged 18 or 19 years).
All procedures involving animals were approved by the Institutional Animal Care and Use Committee of Shin Nippon Biomedical Laboratories Ltd. and performed in accordance with standards published by the National Research Council (Guide for the Care and Use of Laboratory Animals) as the U.S. National Institutes of Health policy on human care and use of laboratory animals. In accordance with these standards, every effort was made to ensure that the animals were free of pain and discomfort. The animal room was maintained at a temperature within the range of 23–29°C, and humidity within the range of 35–75%, with 15 air changes/hour, and artificial illumination for 12 hours/day (06:00 to 18:00). The animals were housed individually in stainless steel cages (Taiyo Stainless Co., Ltd., Kagoshima, Japan). Approximately 108 g of solid food (Teklad Global Certified 25% Protein Primate Diet, Harlan Sprague Dawley Inc., Madison, WI, USA) was provided to each animal daily at approximately 15:00. Water, certified to meet the water quality standards required by the Japanese Waterworks Law, was available ad libitum from an automatic supply system (Edstrom Industries, Inc., Waterford, WI, USA).
All animals were anesthetized by an intravenous injection of sodium pentobarbital (Tokyo Chemical Industry Co., Ltd.) solution (64.8 mg/mL, 0.4 mL/kg) into the cephalic vein of the forearm after being weighed and were then euthanized by exsanguination. The brains from all animals were weighed and fixed in 10% neutral buffered formalin.
Necropsy and Organ Weight
The brains were examined grossly and weighed using an electronic balance (HF-3000, A&D Co., Ltd., Tokyo, Japan). The relative brain weights were calculated from body weight on the day of necropsy.
Histopathology
The brains were trimmed, and the parietal lobes, temporal lobes, and diencephalons were embedded in paraffin, sectioned, and stained with Hematoxylin-Eosin (HE), Periodic Acid-Methenamine-silver (PAM), Per’ls Prussian Blue, and Thioflavine S (Thio-S).
Immunohistochemistry
For immunohistochemical investigations, a mouse monoclonal antibody against Aβ (4G8; Signet, Dedham, MA, USA), a mouse monoclonal antibody against neuron anti-neuronal nuclei (NeuN; Chemicon, Temecula, CA, USA), a rabbit polyclonal antibody against microglial cells (Iba1; Wako, Osaka, Japan), a mouse monoclonal antibody against astrocytes (S100 antibody, Abcam, Cambridge, UK), and a rabbit polyclonal antibody against tau protein (Rabbit polyclonal to Tau [phosphor S422], Abcam) were used for detecting total Aβ, neuron, microglial cells, astrocytes, and phosphorylated tau protein, respectively.
Sections were de-paraffinized, treated with 70% periodic acid, and then left at room temperature for 30 minutes, followed by dropwise addition of Protein Block Serum-Free (DAKO, X0909, approximately 100 μL) for 30 minutes. Primary antibody dilutions were prepared with 4G8 diluted 500-fold with PBS solution (TaKaRa, Lot No.: 2001) or with Rabbit polyclonal to Tau (phosphor S422) diluted 100-fold with antibody diluent with background reducing components (DAKO, Lot No.: S3022). Sections were incubated free-floating in primary antibody solution overnight at 4°C, and then washed briefly with buffer, followed by dropwise addition of secondary antibody (Envision+ Mouse/HRP and Envision+ Rabbit/HRP: DAKO, K4006 and K4002, respectively). Immunoreactive elements were visualized by treating the sections with DAB+ Liquid (DAKO, K3467).
Morphometry
The brain was placed with the base upside and cut vertically at the optic chiasma. This single cut section was prepared routinely and stained immunohistochemically using NeuN, S100, and Iba1. Only the hippocampal area was used for morphometric analysis. The numerical cell density of NeuN-positive neurons, S100-positive astrocytes, and Iba1-positive microglial cells was evaluated quantitatively, using computerized image analysis and morphometric techniques (Soft Imaging System, GmbH). Briefly, the total area of hippocampus was estimated; NeuN-positive, S100-positive, and Iba1-positive cells in the hippocampus were counted automatically; and the number of each kind of cell per unit hippocampal area was calculated and expressed as number per mm2 of hippocampal area.
Statistic Analysis
Brain weights (absolute and relative), neuron counts, astrocyte counts, and microglial cell counts were compared between middle-aged and young monkeys. The data were analyzed for homogeneity of variance by the F-test. All variances were homogeneous; Student’s t-test was performed. These statistical analyses were conducted using MUSCOT statistical analysis software (Yukms Co., Ltd., Tokyo, Japan) at significance levels of 5% and 1%, and indices are shown at both p < .05 and p < .01.
Results
Gross Examination
No abnormalities were observed in the brain of any middle-aged or young cynomolgus monkey.
Brain Weights (Absolute and Relative to Body Weight)
The range, mean, and standard deviation of absolute and relative organ weights for each group are shown in Table 1 . Relative brain weights in the middle-aged monkey group were statistically significantly lower than in the young monkey group (p < .05), but no significant differences were noted in absolute brain weight.
Body weight, absolute and relative brain weights.
* Indicates significant differences from the young monkey group (p < .05).
Histopathological Findings
Histopathological findings in the brain are summarized in Table 2 .
Individual findings in middle-aged cynomolgus monkeys.

A dash (—) indicates none.
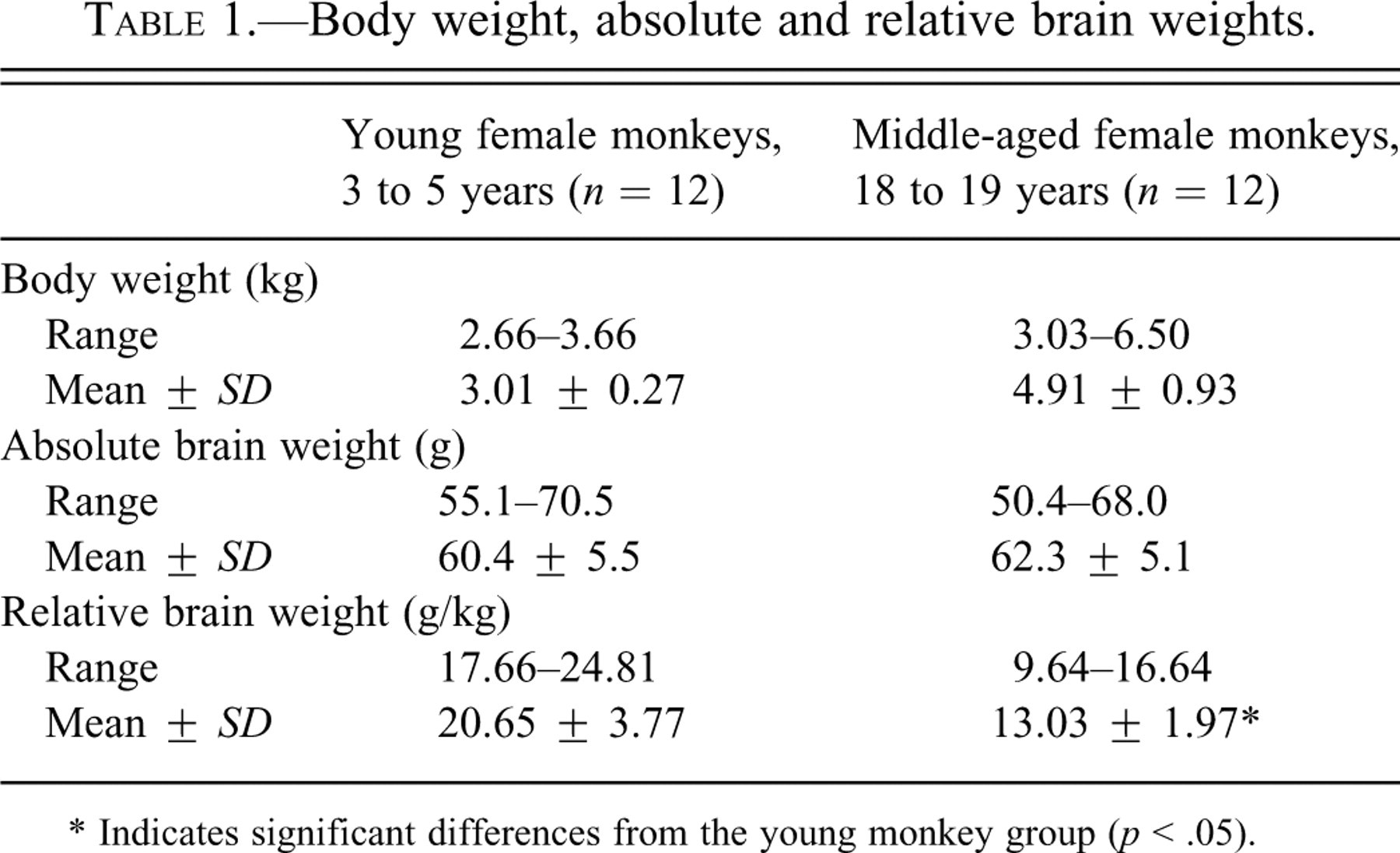
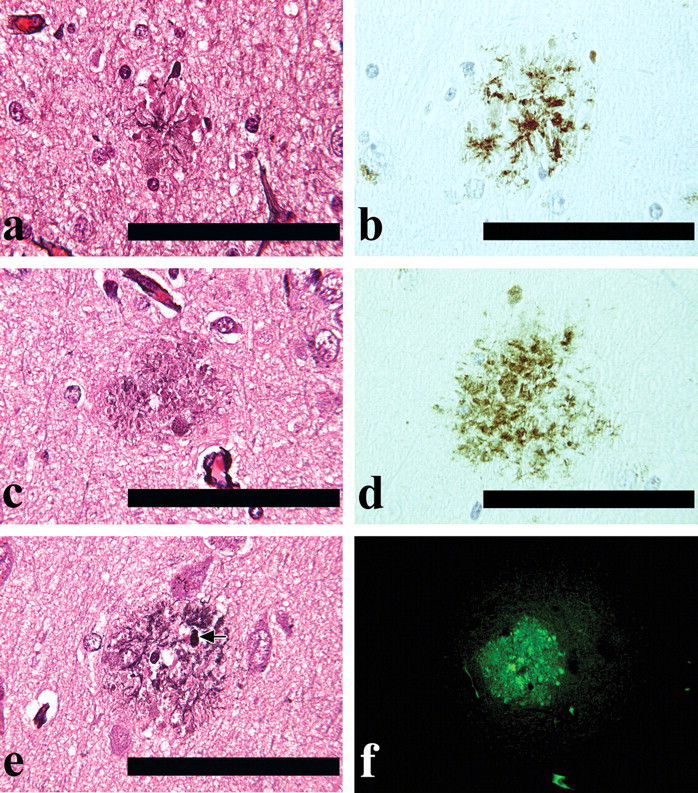
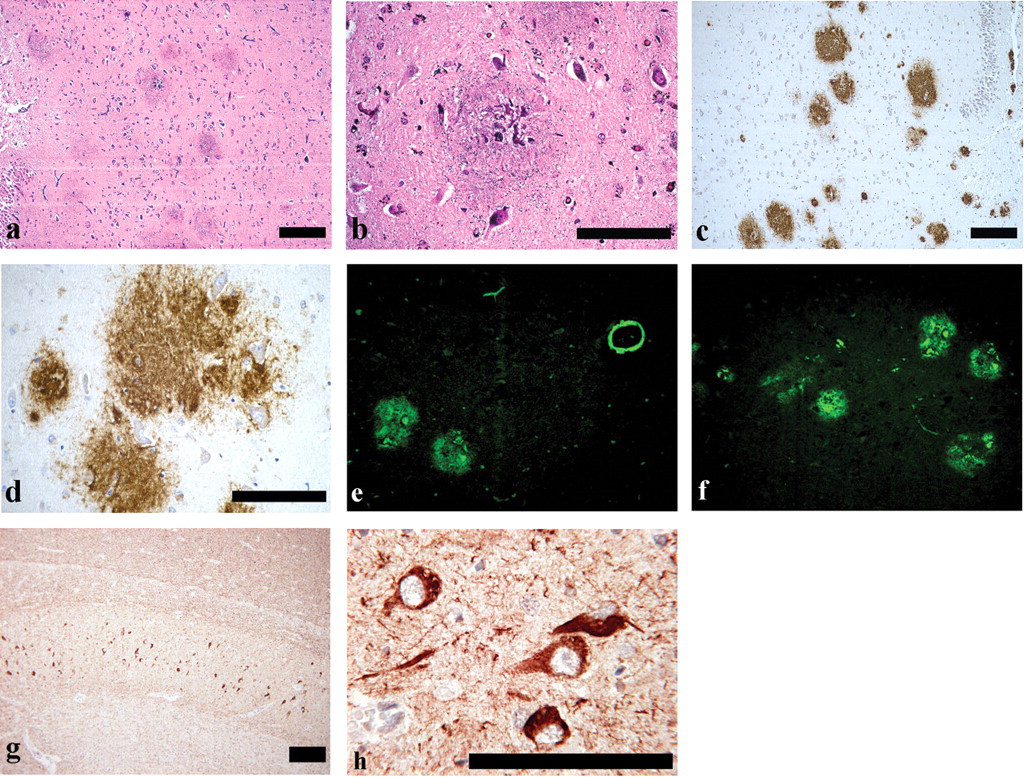
SPs were observed throughout the cerebrum for all middle-aged monkeys, but not for young monkeys (PAM and Thio-S staining, and 4G8 immunohistochemical staining, Figures 1a-f). The distribution of the SPs in the cerebrum is shown in Figure 2. The SPs were spherical to oval structures approximately 100 μm in diameter and were disseminated throughout the cortex and hippocampus and, to a slight extent, in the basal ganglia (all SPs were found in the gray matter of the brain). Almost all SPs were classified as DPs formed as aggregated Aβ; they did not have an amyloid core in the central part and did not represent neuritic degeneration. Accumulations of reactive astrocytes and microglial cells were not observed around the DPs. CPs were observed in the cortex and hippocampus in the cerebrum from one middle-aged monkey (a 19-year-old), but not in those from young monkeys. Furthermore, slight accumulations of reactive astrocytes and microglial cells were observed around the CPs.
(a) Classical plaque (PAM stain). Scale bar: 100 μm. (b) Classical plaque (4G8 immunohistochemical stain). Scale bar: 100 μm. (c) Diffuse plaque (PAM stain). Scale bar: 100 μm. (d) Diffuse plaque (4G8 immunohistochemical stain). Scale bar: 100 μm. (e) Classical plaque (PAM stain), (arrow) enlarged nuritic degeneration. Scale bar: 100 μm. (f) Classical plaque (PAM stain), (arrow) enlarged nuritic degeneration. Scale bar: 100 μm.
Distribution of SPs and axonal spheroids in the cerebrum in middle-aged female cynomolgus monkeys.
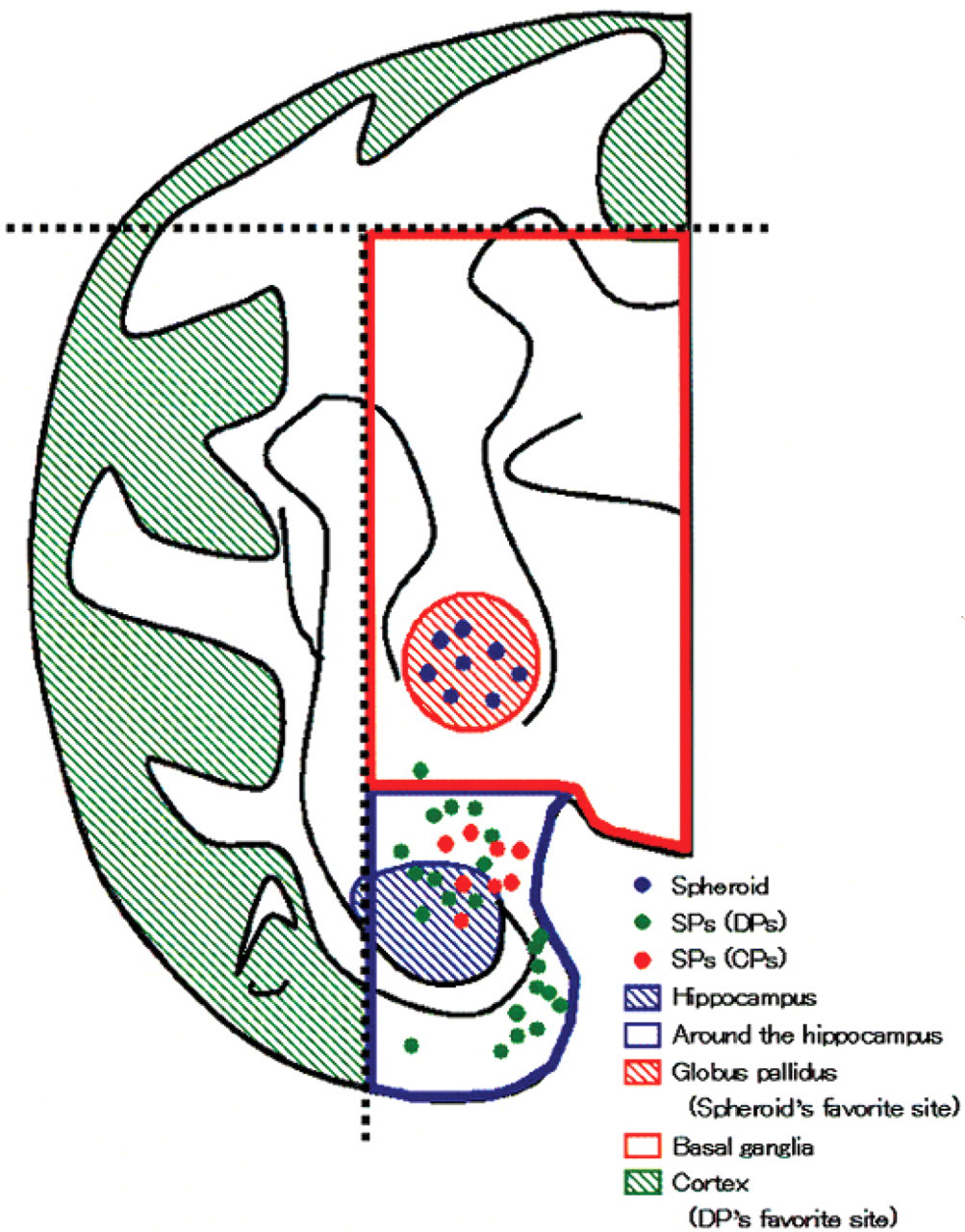
CAA was observed in the cortex and hippocampus in the cerebrum from one middle-aged monkey (a 19-year-old; the animal in which CPs were observed), but not in those from young monkeys (Thio-S staining and 4G8 immunohistochemical staining, Figures 3a and d). All of the CAA observed in the cortex or hippocampus was located near the CPs (Thio-S staining and 4G8 immunohistochemical staining, Figures 3a and d). All CAA revealed that Aβ was present in the walls of cerebral blood vessels. There were no morphological changes in the thickness of the walls of cerebral blood vessels.
(a) Classical plaque and amyloid angiopathy located near the classical plaque (4G8 immunohistochemical stain). Scale bar: 100 μm. (b) Axonal spheroid (HE stain). Scale bar: 100 μm. (c) Lipofuscin deposition (HE stain). Scale bar: 100 μm. (d) Classical plaque and amyloid angiopathy located near the classical plaque (Thio-S stain). Original magnification ×40.
NFTs and micro-hemorrhage could not be confirmed in the cerebrum of any monkey, which were stained immunohistochemically with anti-phosphorylated tau protein and Beriln blue, respectively.
Axonal spheroids were observed in the globus pallidus of the basal ganglia in all middle-aged monkeys (HE staining, Figure 3b), but in only one young monkey. Axonal spheroids appeared as roughly spherical bodies 30-50 μm in diameter.
Lipofuscin deposition was observed in the cortex, basal ganglia, and hippocampus (whole area of brain) in all middle-aged monkeys (HE staining, Figure 3c). Lipofuscin was significantly increased in middle-aged monkeys in comparison with young monkeys, and the granules were larger, more numerous, and in some cases aggregated. The majority of the lipofuscin granules were found in neurons, but the amount of pigmentation was also increased in glial cells and neutrophils.
Morphometry in the Hippocampus
The numbers of the NeuN-positive neurons, S100-positive astrocytes, and Iba1-positive microglial cells in the hippocampus were shown in Figure 4 .
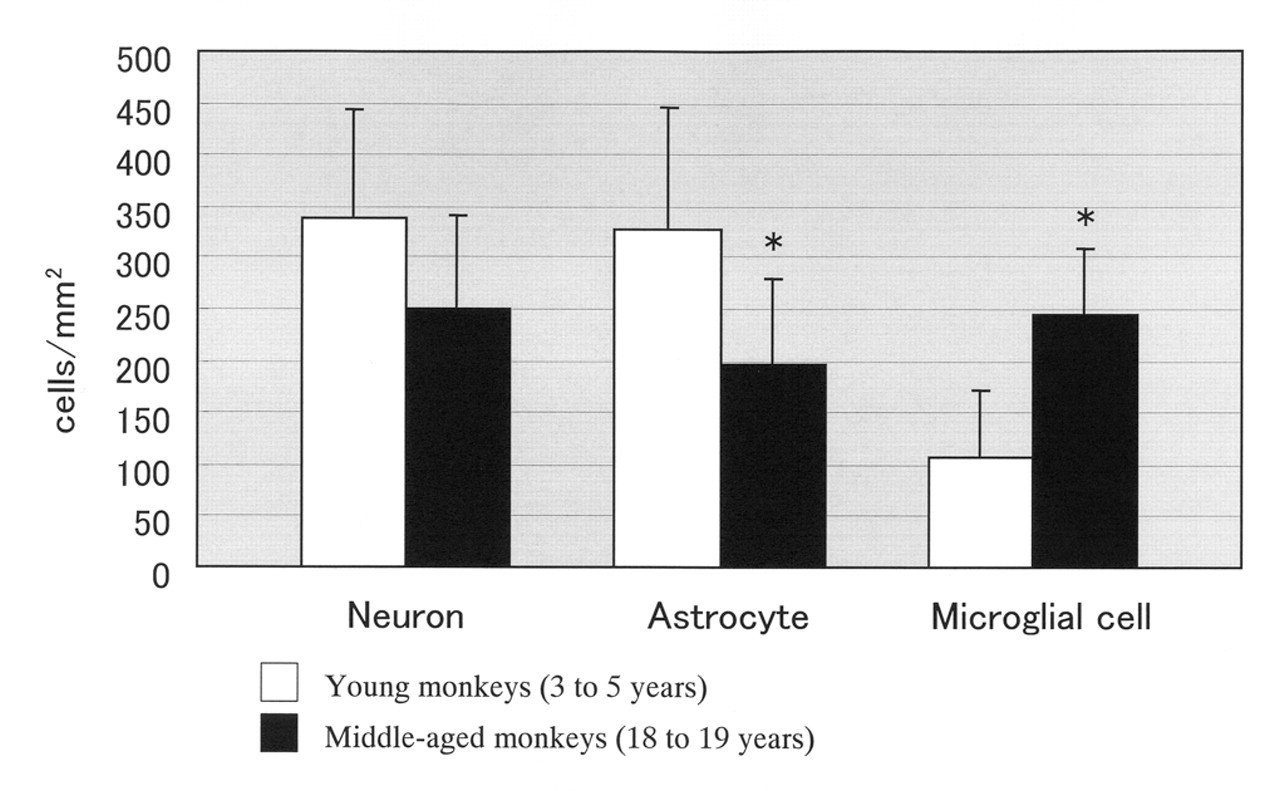
Comparison of cell counts for neurons, astrocytes, and microglial cells in the hippocampal region. *Indicates significant differences between age groups (p < .05).
The number of NeuN-positive neurons in the hippocampus was lower in the middle-aged monkeys when compared with that in the young monkeys; however, the numerical differences were not statistically significant.
The number of S100-positive astrocytes in the hippocampus was statistically significantly lower in the middle-aged monkeys when compared with that in the young monkeys (p < .05).
The number of Iba1-positive microglial cells in the hippocampus was statistically significantly higher in the middle-aged monkeys when compared with that in the young monkeys (p < .05).
Discussion
Comparison of Neuron, Astrocyte, and Microglial Cell Counts in the Hippocampal Region
In the hippocampal region, the numbers of neurons and astrocytes per unit area were slightly lower in the middle-aged than in the young cynomolgus monkeys. However, neuronal and astroglial loss were not recognized in HE-stained specimens. This can be expected to occur to a degree as a result of degeneration/necrosis/loss of the neurons that progressed with ageing. Although SPs and CAA were observed in our monkey case, NFTs was not observed, even though there was accumulation and agglutination of β-amyloid. It was considered that the minimal quantities of β-amyloid observed in our cynomolgus monkeys were associated with neuronal loss—suggested only by decreased numbers of neurons per area of hippocampus—but did not result in large-scale neuronal necrosis. On the contrary, the number of microglial cells was significantly higher in the middle-aged monkeys, reflecting an upgraded function for removing the deposited amyloid and/or degenerated necrotic neurons and matrix.
Senile Plaques
Among the histopathological findings in the region of the cerebrum for middle-aged cynomolgus monkeys, the most distinct was SPs at an incidence of 100% (DPs: 100%, CPs: 8.3%, PPs: 0%). As shown above, almost all SPs were classified as DPs, which are unaccompanied by neuritic degeneration, and they were diffusely distributed in the cerebral cortex in and around the hippocampus of the gray matter region. DPs were mostly observed in the cortices of the temporal and parietal lobes and were found in the basal ganglia in only one case (a 19-year-old). In this animal, CPs were also observed in the cerebral cortex and the hippocampus, with slight surrounding accumulations of reactive astrocytes and microglial cells. It has been reported that SPs change from diffuse to mature plaques with age (Tomlinson, Blessed, and Roth 1968, 1970); the formation of SPs in the present study was consistent with this hypothesis. SPs are sometimes seen in rhesus monkeys in the late teens, and almost always at 25 years old and above, and it has been reported that the occurrence of SPs in squirrel monkeys is also possible at 10 years and above and widespread once they pass 20 years old (Cork and Walker 1988). The age of onset of SPs in cynomolgus monkeys is thought to resemble those in rhesus and squirrel monkeys.
Cerebral Amyloid Angiopathy
CAA was observed in only one case (a 19-year-old) in the cerebral cortex and the hippocampus. It has been reported that SP formation occurs before vascular amyloid deposition in rhesus monkeys and after vascular amyloid deposition in squirrel monkeys (Cork and Walker 1988). It was considered, therefore, that the chronological sequence of SP formation and vascular amyloid deposition in cynomolgus monkeys more resembles that in rhesus monkeys than that in squirrel monkeys. CAA was observed in the capillaries and parenchymal arterioles in the cortex and was often located near mature plaques (CPs). These findings are consistent with the speculation of Nakamura et al. (1995a, 1995b, 1996, 1998). There have been reports that the walls of blood vessels in which amyloid is deposited weaken and sometimes rupture, leading to hemorrhage (Anderton 1997); however, hemorrhage was not noted in this study.
In humans and in monkey species, amyloid deposits in the SPs and CAA result from autoagglutination of amyloid protein becoming entirely detached due to an abnormal APP degradation process (Kimura et al. 2001, 2003, 2004, 2005). Also, it is thought that there is a relationship between the number of blood vessels with amyloid and the number of mature SPs (CPs and PPs). SPs have for a long time been assumed to originate from a vascular abnormality; however, it was thought that in monkeys, vascular amyloid and brain parenchyma amyloid were related to a slight degree. These findings are also consistent with the speculation of Nakamura et al. (1995a, 1995b, 1996, 1998).
Other Histopathological Findings
Large quantities of lipofuscin deposits in all animals and large granules across an extensive area were observed. Axonal spheroids that were identified as “Pallido-nigral axonal spheroids” (Fukuda et al. 2005) were observed in the globus pallidus of the basal ganglia in all animals. These findings were significantly increased in middle-aged monkeys when compared with young monkeys. It is thought that lipofuscin deposition and axonal spheroids occur as functions of ageing, in a similar manner to that in humans and in other monkey species (Cork and Walker 1988). Degeneration of the neurons in the hippocampal region was observed to a slight degree in all middle-aged animals; however, no great differences were noted when compared with young monkeys. NFTs and neuronal loss and hippocampus and cerebral cortex atrophy were not observed. Some cases had developed as far as β-amyloid deposition, but toxicity to neurons was not noted to any degree, and it was thought that these cases had not developed as far as NFTs.
Brain Weight
A statistically significantly low relative brain weight was noted in middle-aged cynomolgus monkeys when compared with young cynomolgus monkeys. However, atrophy was not observed in the brain itself in gross examination. Furthermore, female cynomolgus monkeys become obese at the age of 5 to 7 years as their body weight increases (Tanaka et al. 1995). From these reports, the change was considered to be a result of increased body weight and not an age-related brain abnormality.
In the present study on 18- to 19-year-old cynomolgus monkeys (the equivalent of 45 to 60 years old in humans), as histopathological findings in the cerebrum that indicate AD in humans (shown in reference figures: Figure 5), SPs and CAA were observed, but NFTs were not observed. The numbers of neurons and astrocytes were decreased, and the number of microglial cells increased, with age in the hippocampal region. It is reported that SPs, as abnormal processing of APP, appear before NFTs in the pathological formation process of AD (Hardy et al. 1998). Among the 18- to 19-year-old monkeys, individuals showed relatively large amounts of β-amyloid deposition and SPs and CAA, and it was thought that a pre-Alzheimer’s condition had been reached.
(a) Senile plaques in human hippocampus with Alzheimer’s disease (PAM stain). Scale bar: 200 μm. (b) Classical plaque in human hippocampus with Alzheimer’s disease (PAM stain). Scale bar: 100 μm. (c) Senile plaques in human hippocampus with Alzheimer’s disease (4G8 immunohistochemical stain). Scale bar: 200 μm. (d) Senile plaques in human hippocampus with Alzheimer’s disease (4G8 immunohistochemical stain). Scale bar: 100 μm. (e) Senile plaques and amyloid angiopathy in human hippocampus with Alzheimer’s disease (Thio-S stain). Original magnification ×40. (f) Senile plaques in human hippocampus with Alzheimer’s disease (Thio-S stain). Original magnification ×40. (g) Neurofibrillary tangles in human hippocampus with Alzheimer’s disease (anti-phosphorylated tau protein immunohistochemical stain). Scale bar: 200 μm. (h) Neurofibrillary tangles in human hippocampus with Alzheimer’s disease (anti-phosphorylated tau protein immunohistochemical stain). (Note that the tangle fills the cell body as well as extending into the apical dendrite and the basal dendrites.) Scale bar: 100 μm.
β-amyloid produces SPs and CAA resulting from its agglutination and possesses toxicity to neurons, producing NFTs and inducing apoptosis (Hardy and Selkoe 2002; Kimberly et al. 2003). It is thought that this process takes a long time, several years or decades. This is why the onset of AD is mainly in old age, although it can be at 30 to 40 years in cases of FAD.
Amyloid production and deposition, particularly the deposition of SPs and CAA, both occur in animals in old age (Cork and Walker 1988; Nakamura et al. 1995a, 1995b, 1996, 1998; Kimura et al. 2001, 2003, 2004, 2005). In humans, there is further progression to reach a stage of NFT production. However, cynomolgus monkeys reach the stage of amyloid deposition, although other illnesses occur before NFTs can be produced, cutting their life span. The rate of ageing is much slower for the brain than other organs, and as a result, humans live until NFT production and neuron loss can occur, while laboratory animals rarely remain alive until that stage. In other words, the fact that the brain ages proportionally less progressively than other organs presents a challenge in providing ideal laboratory animal models of AD.
Taken together, the results of the present study showed the following: Age-associated hippocampal changes included decreased numbers of neurons and astrocytes accompanied by increased numbers of microglial cells. The onset of SPs and CAA in the cerebrum in cynomolgus monkeys can occur before the age of 20 years (18 to 19 years). SPs were almost all categorized as diffuse plaques (DPs); they did not have amyloid cores and were unaccompanied by neuronal degeneration. In cynomolgus monkeys, SPs (DPs) occur before the appearance of CAA.
From the above, it was concluded that cynomolgus monkeys showed pathological changes due to ageing similar to those related to AD in humans, even before they were 20 years old. It was suggested that if the lifespan of cynomolgus monkeys were prolonged or their β-amyloid or tau protein production were accelerated, they would have the potential of being an appropriate AD model.
