Abstract
Subcutaneous injection of pharmaceutical compounds into the dorsal skin of rats is common in preclinical and nonclinical studies. However, no detailed histologic description of this anatomic location has been published to date. Following the observation of vascular lesions in the dorsum of rats in a thirteen-week toxicity study, a complementary study was performed on untreated Sprague-Dawley rats to evaluate the normal histology of the skin and subcutis, the potential effect of chronic subcutaneous injection on the morphology of the skin and its vasculature, and the spontaneous vascular pathology in the areas used as injection sites in the principal study. This study showed that saline injection did not fundamentally alter the morphology of the injection sites used for the principal study. Skin thickness was greater in males than in females. Although acellular intimal thickening occurred spontaneously in the dorsal skin of untreated males and females, only males had a spontaneous incidence of intimal hyperplasia. No site predilection for intimal lesions was apparent for either sex. Saline injection, or the physical trauma of injection, may induce intimal hyperplasia; males appear more likely to develop the lesion than do females. It is possible that acellular intimal thickening can progress to intimal hyperplasia under appropriate conditions.
Introduction
The accurate interpretation of skin lesions in toxicity studies conducted by subcutaneous injection depends on a thorough understanding of the normal histology of the site(s) injected. Such studies frequently involve the injection of multiple sites on the dorsum of the species tested. Although the histology of rat skin has been generally described, to our knowledge there is no written report of how skin histology varies with location on the dorsum. Morphometric studies have shown that the thickness of the skin at injection sites in adult and aging rats from six-month toxicity studies and carcinogenicity studies, respectively, varies with cranial-caudal distribution (Wells et al. 2007, 2008). Not surprisingly, the dermis and the hypodermis (subcutis) play the largest role in determining skin thickness.
Vascular lesions have been reported at the injection sites of subcutaneous toxicity studies (Wells et al. 2007, 2008, 2009). Scientific literature does not provide detailed information on the morphology of the vasculature of normal rat skin. An exhaustive literature search yielded only information regarding the microanatomy (angiosomes) (Taylor and Minabe 1992) and the ultrastructure (Imayama 1981) of cutaneous vasculature in rat skin.
This article describes the histology of injection sites from four levels of the rat dorsum and illustrates that, similar to skin thickness, the morphology of injection sites varies from cranial to caudal. It compares the histology of normal skin of the dorsum with that of skin subjected to repeated subcutaneous injection with saline. Finally, it describes spontaneous vascular lesions in normal (untreated) rat skin and discusses their possible relationship to aging.
Materials and Methods
In the principal study, the test compound, placebo, or sterile isotonic saline solution was administered by subcutaneous injection to 100 Sprague-Dawley Crl: CD (SD) rats (Charles River, Saint Germain sur l’Arbresle, France) over a thirteen-week period. Of the one hundred rats used for the study, five male and five female rats were administered isotonic saline and served as absolute controls. For the complementary evaluation of untreated control injection sites discussed herein, five male and five female Sprague-Dawley Crl;CD (SD) rats (Charles River, Saint Germain sur l’Arbresle, France) were housed under similar husbandry conditions at the same laboratory that performed the principal study. All procedures used for the principal study were employed for these rats, with the exception that they were not injected with any substance.
The subcutaneous route was chosen for administration in the principal study because this is the route of administration of the test compound in humans. Rats were maintained and handled according to international standards of animal health and welfare in an AAALAC-accredited facility. Seven injection sites, identified by tattoo prior to study initiation, were used in rotation for compound administration, so that each site was injected once every seven days. Animals were nineteen to twenty weeks of age at the end of the study. All animals from both studies were euthanized by carbon dioxide inhalation and exsanguination, and necropsied immediately after death; animals from the principal study were euthanized in equalized group order. Samples were routinely collected and processed for histology. Three sections of each injection site (middle, lateral, and medial) from all animals were evaluated. Sections were cut at three to five microns and stained with hematoxylin and eosin. The location of the injection sites is illustrated in Figure 1 .
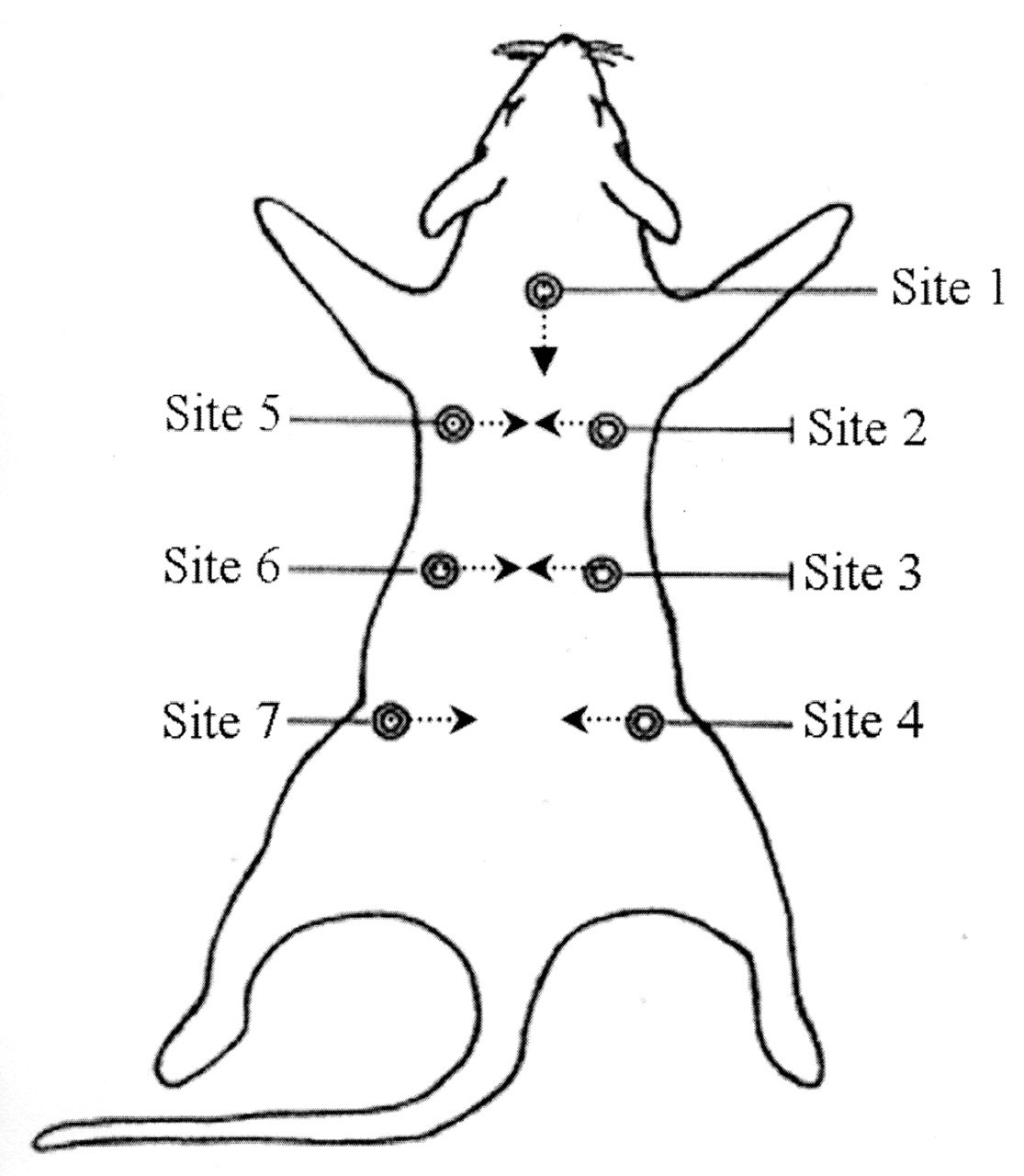
Location of injection sites 1–7.
Statistical analysis of the incidence of intimal hyperplasia was performed using the Fisher exact test (Conover 1980a). Statistical analysis of severity grades for intimal hyperplasia at individual injection sites was performed using the Kruskal-Wallis Test (Conover 1980b). The software package used to perform these tests was Roelee, version 2.051 (P. N. Lee Statistics and Computing, Surrey, UK; release date: December 2005).
Though no injections were administered to the rats used for the complementary study, the tissues evaluated from these animals are referred to as injection sites in this paper for the sake of comparison to the results of the principal study.
Results
Histology of Untreated Skin at Injection Sites
The histology of rat skin has already been described (Ellwell, Stedham, and Kovatch 1990; English and Munger 1994). However, to our knowledge, no description of the differences that may be observed from one region of the dorsum to another has been published to date. We present our observations using the terminology from the English and Munger description, which divides the skin and subcutis into epidermis, dermis, stratum adiposum, cutaneous muscle of the trunk (referred to as the panniculus herein), and stratum fibrosum.
Epidermis
This uppermost part of the skin contained up to six cell layers. The stratum basale was prominent, whereas the stratum spinosum was often inapparent. The stratum granulosum was generally easily identifiable; keratohyalin granules were not always apparent, but up to three or four layers of granules were evident in many cases. Nuclei of cells in the stratum granulosum were often difficult to identify. The thickness of the stratum corneum was variable. Occasional touch domes (English and Munger 1994) or
Dermis
This consisted of densely packed, variably oriented, thick bundles of collagen. Clusters of hair follicles (vellus and guard hairs) and associated sebaceous glands were evident, and hairs in all phases of development were observed. The depth of the hair bulbs varied from mid- to upper dermis for telogen hairs to resting within the stratum adiposum for anagen hairs. Arrector pili muscles were easily identifiable. At low magnification, vessels and nerves in the central part of the dermis were not as easily identifiable as they were in the subcutis. However, vessels and nerves at the base of the dermis (between zones of adipose tissue from the stratum adiposum) were more readily observed.
Stratum Adiposum
This part of the skin was the most variable in appearance. It ranged from being a continuous band of relatively uniform width between the dermis and the panniculus or stratum fibrosum to a jagged, irregular layer of adipose tissue with triangular, “sawtooth” (Figure 2 ), or crescent-shaped foci of fat interrupted by varying lengths of dermal collagen. It was more prominent when anagen follicles were present in the section. This layer was generally separated from the panniculus muscle by a connective tissue band that varied in thickness but was often quite thin. Vasculature and nerves in this region were very easily seen.
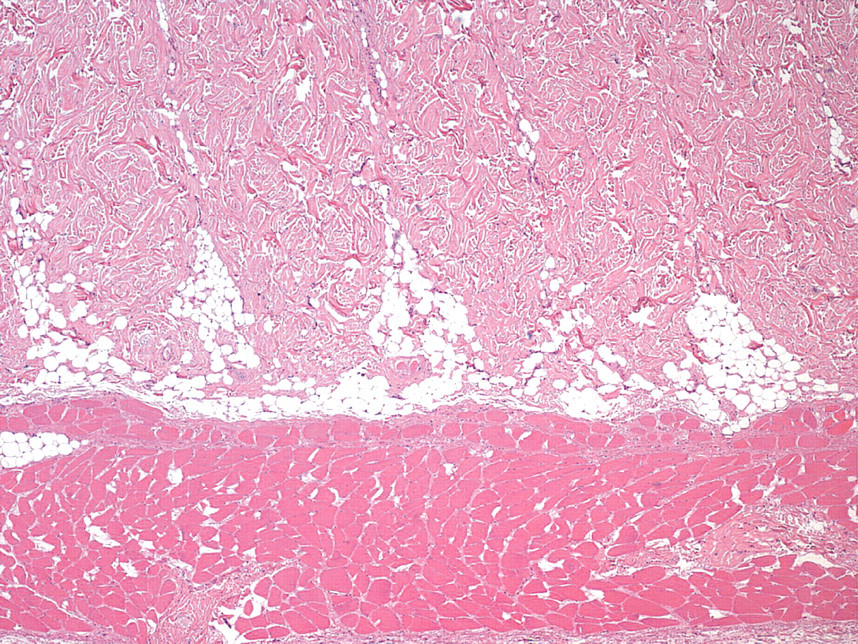
Stratum adiposum–“sawtooth” appearance (original magnification: 4×). H&E stain.
Panniculus
This muscle layer varied in thickness according to the location of the injection site. In females, it sometimes appeared as a split layer of tissue in the area where mammary tissue was abundant. Muscle was sometimes interrupted by foci of adipose tissue containing connective tissue bundles or sheaths that in turn contained vessels and nerves.
Stratum Fibrosum
Representing the lower limit of the subcutis, this layer consisted of fine-caliber collagen bundles with a variable amount of myxomatous material observed between the fibers. Vessels (mostly very small caliber) and nerves were easily identifiable in this layer. The collagen bundles could be densely packed in a single band or separated by zones of adipose tissue in which foci of connective tissue contained vessels and nerves (Figure 3 ). Connective tissue bundles containing vessels and nerves could also be viewed independent of foci of adipose tissue.
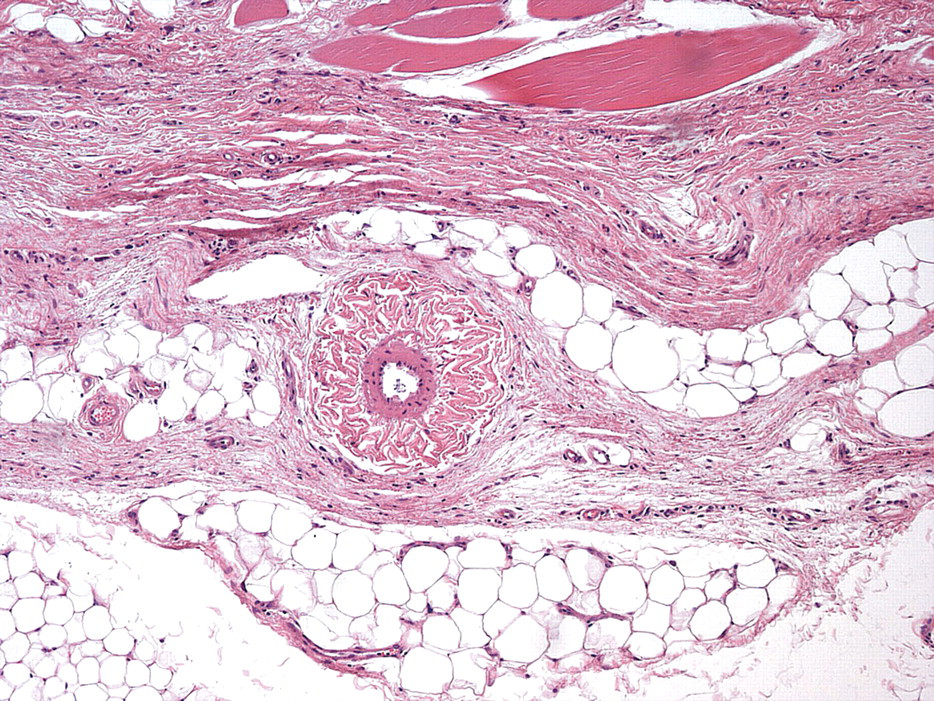
Stratum fibrosum (original magnification: 10×). H&E stain.
Sex-Related Differences in Histology of Normal Dorsal Skin
The only consistent sex-related difference observed between the skin and subcutis of the injection sites evaluated was skin thickness. For any given site, thickness was greater in males compared to females (Figures 4A and 4B ). Males also had more variability in epidermal thickness and panniculus:dermis thickness ratio than did females. If stratum spinosum was observed in the epidermis, it was more likely to be identifiable in female rats. See Table 1 for a list of the histological features of normal dorsal skin by location.
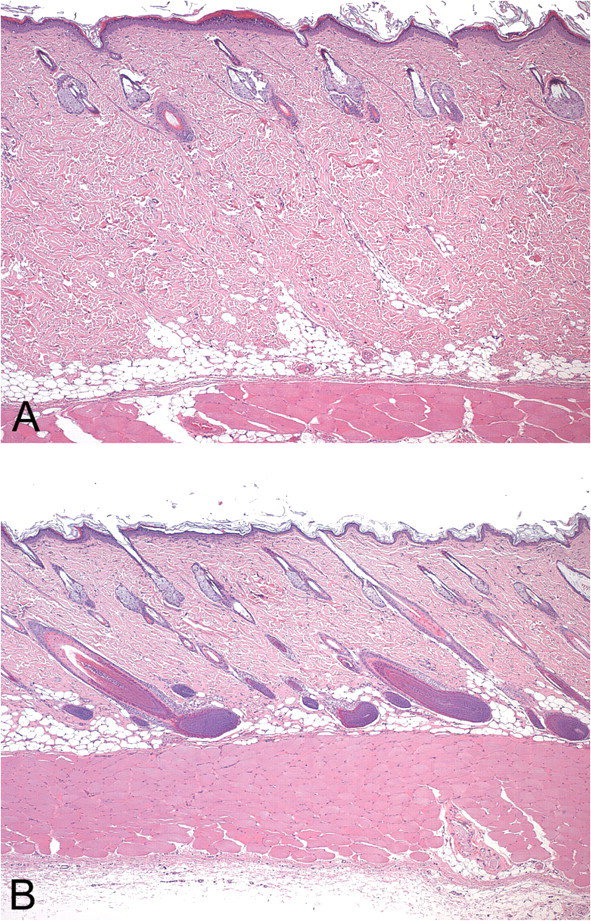
Comparison of male (4A) and female (4B) injection sites (Injection Site 2; original magnification: 4×). H&E stain.
Histological features of dorsal rat skin by anatomic location.

Abbreviations: F, female; M, male.
a Thickest site in males and females
The cranial-most injection site sampled in this study was located in the center of the neck region. In male rats, the epidermis was two to four layers thick compared to one to three layers in females. In males, the stratum adiposum often had a “sawtooth” appearance, whereas it was less pronounced to almost inapparent in females. In both sexes, the panniculus muscle was often not present in a continuous band, but rather was located at one end of the section only. It was roughly 25–33% of the thickness of the dermis in males and 33–100% of the thickness of the dermis in females. In the area where panniculus was not present, the stratum adiposum rested directly on the stratum fibrosum. When abundant subcutaneous tissue was present in the tissue sample, separation of the stratum fibrosum into bands by adipose tissue and/or by connective tissue sheaths that contained vessels and nerves was observed.
For paired injection sites, the histology was sufficiently similar to allow them to be described together.
Injection Sites 2 and 5 (near the scapulae) were the thickest sites in both males and females. The epidermis was two to six layers thick in males compared to one to three layers thick in females. The stratum adiposum was more often contiguous in females than in males, with little to no “sawtooth” appearance in females compared to males. It was separated from the panniculus muscle by a thin band of connective tissue, the thickness of which varied more in males than in females. The panniculus muscle was roughly 50–125% of the thickness of the dermis in males and 67–100% of the thickness of the dermis in females. Occasionally, bundles of panniculus muscle were interrupted by the presence of one or more foci of densely packed collagen containing vessels and nerves, with or without associated adipose tissue. The stratum fibrosum was separated into bands by adipose tissue and/or by connective tissue sheaths that contained vessels and nerves (similar to those viewed in panniculus muscle). In females, mammary tissue was found within and/or beneath this layer (Figure 5 ).
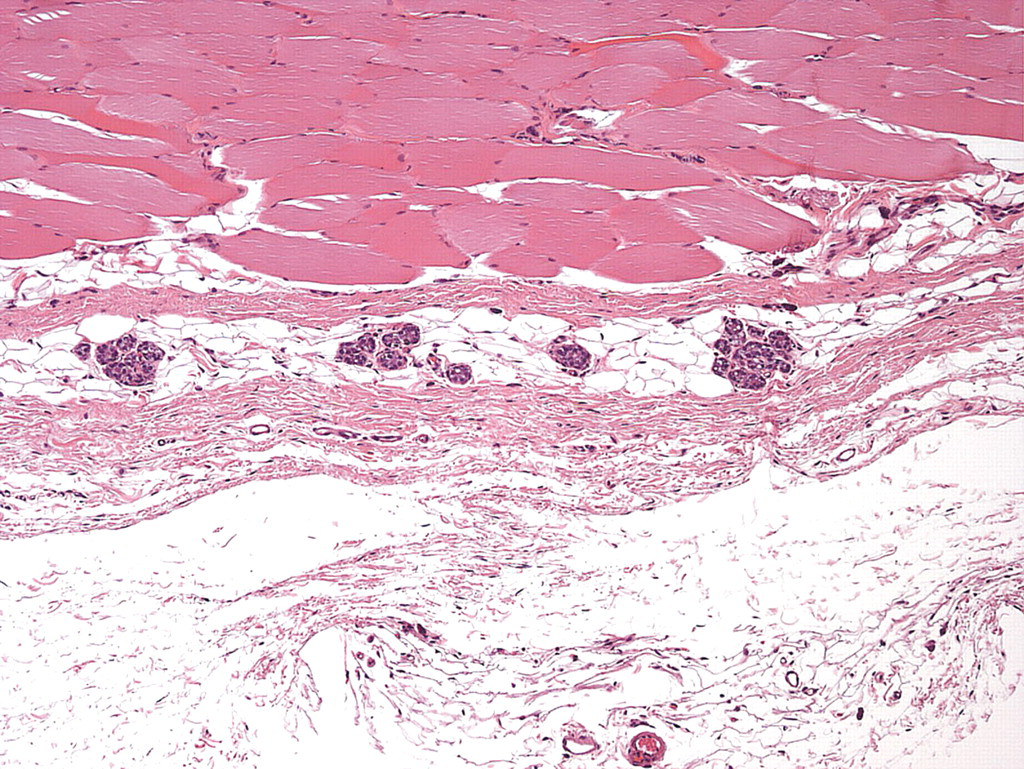
Stratum fibrosum with mammary tissue (original magnification: 10×). H&E stain.
For injection sites 3 and 6 (middle back), the epidermis was two to five layers thick in males and one to three layers thick in females. The stratum adiposum was partially to completely contiguous in both sexes, with a prominent “sawtooth” appearance in males and a more uniform appearance in females. The connective tissue band that separated the stratum adiposum from the underlying panniculus muscle was sometimes discontiguous. The panniculus varied from 33–50% of the thickness of the dermis in males to 33–100% of the thickness of the dermis in females. As seen at injection sites 2 and 5, the stratum fibrosum was sometimes separated into bands by adipose tissue and/or by connective tissue sheaths that contained vessels and nerves. In females, it was rare to see mammary tissue at these sites.
For injection sites 4 and 7 (near the pelvis), epidermal thickness was roughly the same in males and females (one to three or four cell layers thick). Dermal thickness was most comparable between males and females at these sites. The stratum adiposum was discontinuous, and males tended to have much less fat than did females at this level of the dorsum. The “sawtooth” appearance was present in males and females. The panniculus was roughly 25–50% of the thickness of the dermis in males and females. The stratum fibrosum was similar in appearance to that found in other injection sites, with mammary tissue rarely seen in females.
Spontaneous Vascular Pathology
Spontaneous vascular lesions consisted of intimal thickening and intimal hyperplasia (Figures 6 and 7 ).
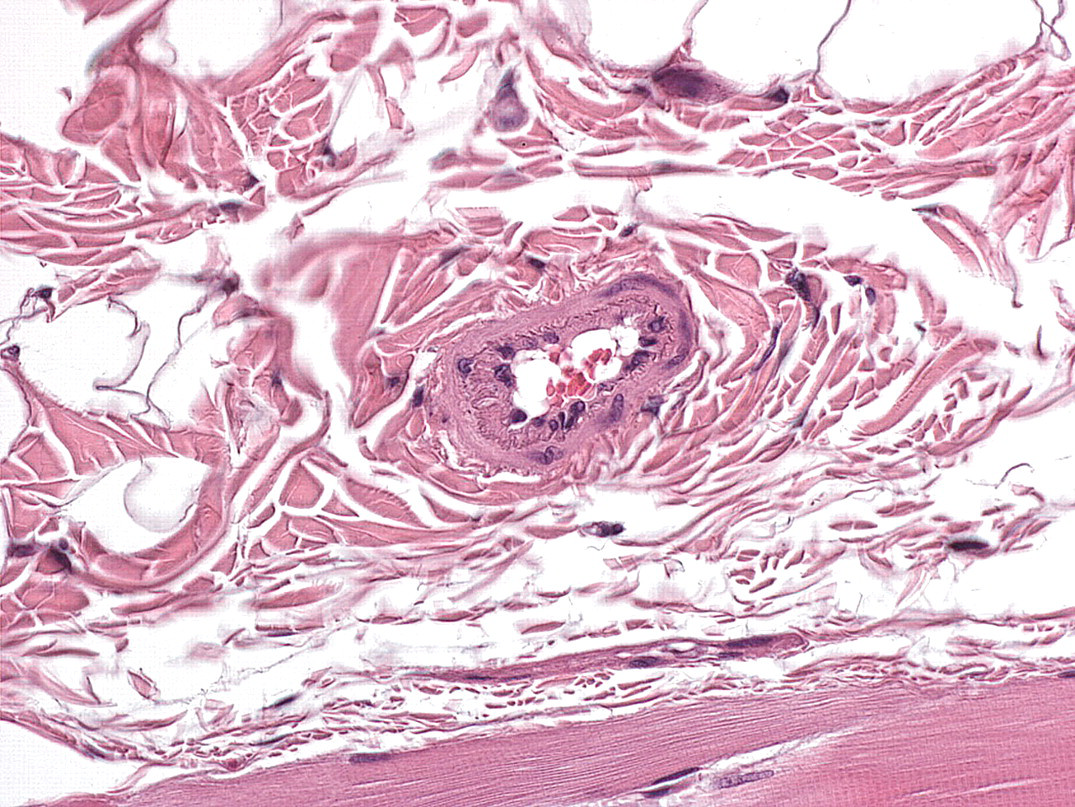
Intimal thickening, dermis: characterized by what is likely to be an increase in extracellular matrix, without an increase in cell numbers, between the endothelium and the internal elastic lamina of small vessels in the dermis and subcutis (original magnification: 40×). Severity grade: mild. H&E stain.
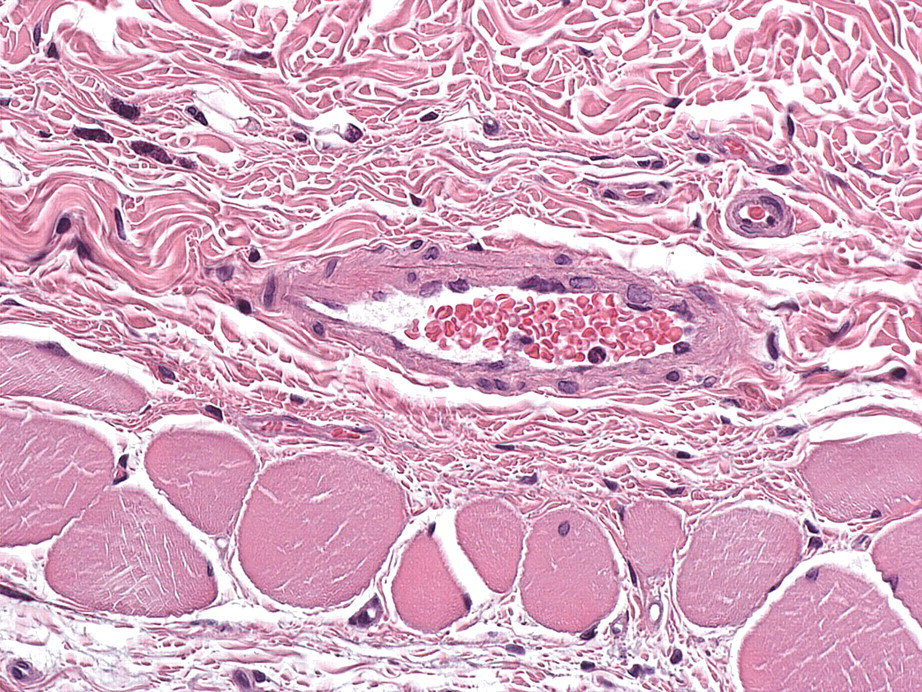
Intimal hyperplasia, dermis: characterized by proliferation of cells (thought to be smooth muscle cells) between the endothelium and the internal elastic lamina (original magnification: 40×). Severity grade: minimal. H&E stain.
Intimal thickening and intimal hyperplasia caused increased thickness of the vascular wall and decreased luminal patency. Severity of these lesions was based on the degree of compromise of the vascular lumen, regardless of vessel size and number. Hyperplasia affected either part of or the entire circumference of the intima, whereas intimal thickening was generally circumferential.
Comparison of Results with the Principal Study
Normal Histology
The histology of the injection sites from the saline controls of the principal study was comparable to that observed in untreated controls from the present study. Saline injection did not fundamentally alter the morphology of the skin at any injection site.
Untreated controls (complementary study) and saline controls (principal study): Incidence (average severity) of intimal hyperplasia at injection sites (
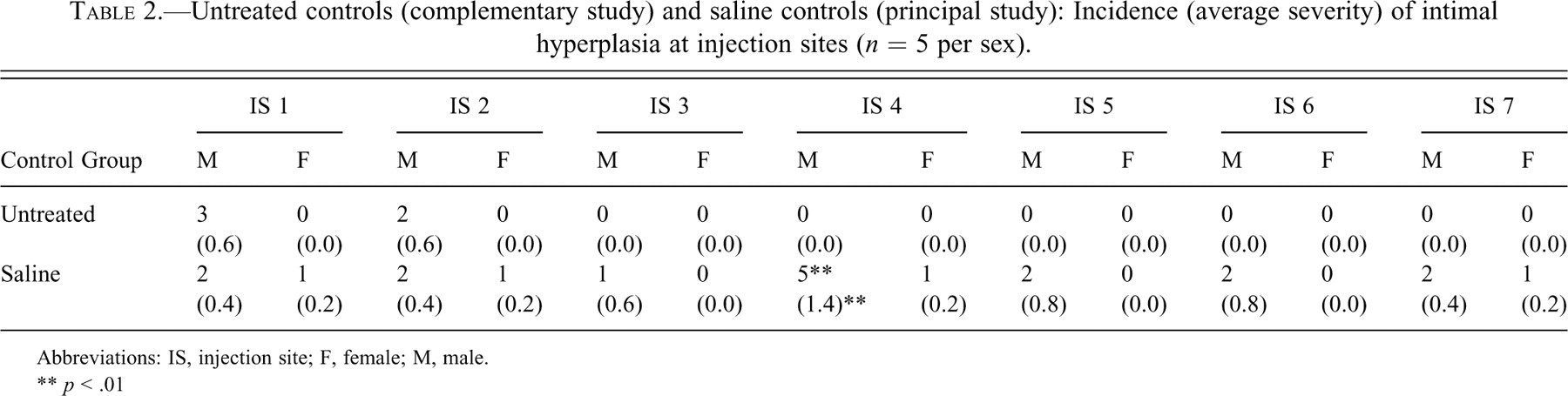
Abbreviations: IS, injection site; F, female; M, male.
**
All saline control males (five of five) from the principal study and all untreated control males (five of five) from the present study had at least one injection site at which intimal hyperplasia was observed. A comparable incidence of intimal hyperplasia was found between these groups at injection sites 1 and 2, whereas saline controls had a higher incidence and severity of this change at injection site 4 (
Comparison of Intimal Thickening and Intimal Hyperplasia
Because we considered the possibility of a common pathogenesis between intimal thickening and intimal hyperplasia, we compared the incidence and average severity of intimal thickening in untreated controls and intimal hyperplasia in saline controls. Data are presented in Table 3 .
Untreated controls (complementary study) and saline controls (principal study): Incidence (average severity) of intimal lesions at injection sites (
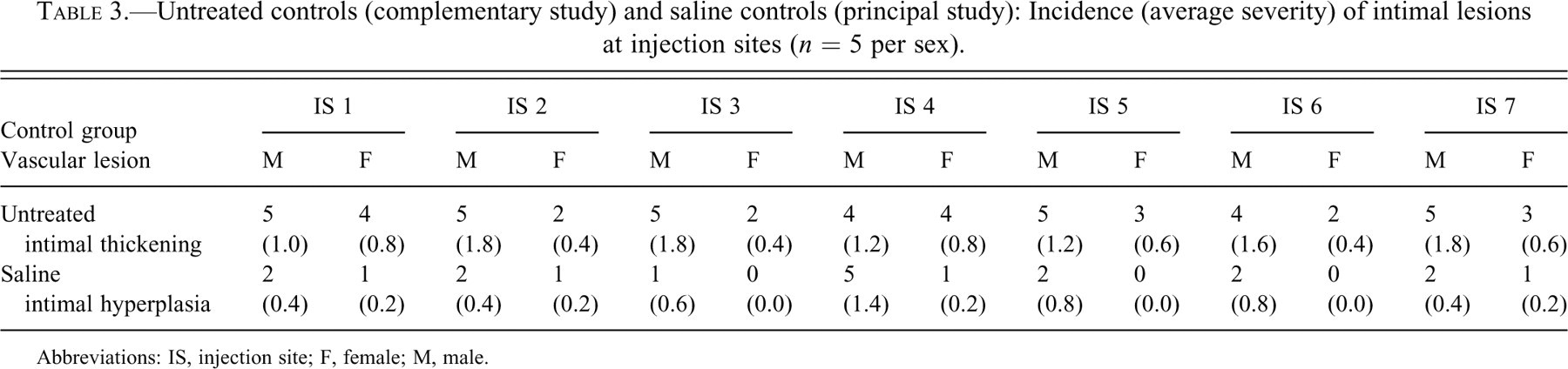
Abbreviations: IS, injection site; F, female; M, male.
In untreated and saline-control males, intimal thickening and intimal hyperplasia, respectively, were observed at all injection sites. The incidence of intimal hyperplasia was comparable to that of intimal thickening at all sites except Injection Site 3 in males. Both lesions were observed to occur with minimal to moderate (grades 1–3) severity. When individual injection sites were evaluated for severity, average severity scores were notably higher for intimal thickening than for intimal hyperplasia at all sites except for injection site 4 in males. No consistent correlation could be found between injection site location and incidence or severity of thickening and hyperplasia.
Intimal thickening was observed at all sites in untreated female controls, but intimal hyperplasia was observed in only four of seven sites in saline-treated female controls. Though no consistent correlation could be found between injection site location and incidence of intimal lesions, all injection sites for female untreated and saline controls had lesions with the same degree of severity (minimal).
Discussion
Histology of Normal Skin of the Rat Dorsum
The division of skin into epidermis, dermis, and subcutis is widely accepted. However, where the dermis ends and the subcutis begins is a subject for debate. English and Munger (1994) define the rat subcutis as containing the stratum adiposum, the layer of adipose tissue found above (external to) the panniculus muscle, the panniculus muscle (found on the trunk only), and the stratum fibrosum. This definition implies that the dermis contains no adipose tissue. In contrast, Elwell, Stedham, and Kovatch (1990) indicate that “the demarcation between the dermis and the subcutis is not always distinct because fat cells are prominent in the deep dermis as well as in the subcutis.” Although we used the English and Munger schema to describe the histology of the injection sites for this study, we considered the stratum adiposum to be part of the dermis and the upper limit of the panniculus muscle to be the boundary between the dermis and subcutis. This definition is consistent with the clinical definition of the subcutis for subcutaneous injection in regulatory toxicity studies, in which the compound to be injected is targeted to the area beneath the panniculus muscle.
For any given injection site, male skin was thicker than female skin. Though we did not undertake morphometric evaluation of the skin samples in this study, previous reports support this observation. In control Sprague-Dawley rats from a two-year subcutaneous carcinogenicity bioassay, differences in the thickness of the epidermis, dermis, and hypodermis (subcutis) were observed craniocaudally in rats of both sexes. These differences were always greater in males than in females (Wells et al. 2007). Similarly, in control Sprague-Dawley rats from a six-month subcutaneous toxicity study, significant differences in skin thickness were observed cranially to caudally in males and females (Wells et al. 2008). In both of these studies, as in the present study, skin was found to be thickest in the area of the scapulae.
The ratio between the thickness of the dermis and panniculus was frequently larger and more widely varied in females than in males, because the dermis of female rats was consistently thinner than that of males, whereas the thickness of the panniculus muscle was more comparable between the sexes. This qualitative observation is corroborated by the results of the morphometric studies mentioned above, in which female rats from a carcinogenicity study and a six-month toxicity study had a thinner dermis at all injection sites compared to their male counterparts (Wells et al. 2007, 2008).
Spontaneous Vascular Pathology in the Skin of the Rat Dorsum
Intimal hyperplasia was observed in control rats in the principal study; this diagnosis triggered the literature search and the detailed review of the skin of age-matched, untreated rats in the complementary study. Intimal hyperplasia is commonly seen following vascular injury. The classical pathogenesis of the lesion is as follows: endothelial cell denudation and thrombus formation > accumulation of mitogens at site of injury > activation of smooth muscle cells (SMC), which become proliferative and secretory > migration of SMC through breaks in the internal elastic lamina to the intima > SMC proliferation and synthesis of extracellular matrix (Schiller et al. 2002). This situation results in decreased lumen diameter and decreased vessel patency.
In addition to the above mentioned pathogenesis, it has been shown that intimal hyperplasia can occur following mechanical trauma of the perivascular tissue without physical denudation or trauma to the endothelium. Booth et al. (1989) induced this lesion beneath intact endothelium by placing a hollow silicone collar around the carotid artery in rabbits. Hyperplasia was verified by the investigators using light microscopy, scanning and transmission electron microscopy.
In the current study, intimal hyperplasia was found only in the skin of untreated control male rats and at only two injection sites. In contrast, it was found at all injection sites of all saline control males and at five of seven injection sites in three of five saline control female rats in the principal study. Therefore, we hypothesize that the injection of saline, or the trauma induced by injection, may have augmented the incidence of intimal hyperplasia in the principal study. No consistent correlation was found between injection site location and incidence or severity of this lesion.
In addition to intimal hyperplasia, we observed intimal thickening in the present study, which was characterized by an increase in extracellular matrix, without an increase in cell numbers, between the endothelium and the internal elastic lamina. This terminology was chosen to distinguish the lesion from intimal hyperplasia, in which increased numbers of cells were observed between the endothelium and the internal elastic lamina. In the human literature, the term “intimal thickening” has been used as a synonym of intimal hyperplasia for many years (Schoen and Cotran 1999), particularly in research papers that discuss catheter-induced vascular injury in rats (Bochaton-Piallat et al. 1995; George et al. 2004; Verheyen et al. 1988). This literature includes papers that present research performed with a somatostatin analog (Ulus et al. 1998; von Essen et al. 1997; Yumi et al. 1997). “Intimal thickening” has also been used as a synonym for intimal hyperplasia in regulatory toxicity studies (personal observation, 2008). Consequently, we suggest that “acellular intimal thickening” is a better term to denote increased matrix without increased cell numbers because it eliminates any possible confusion about the morphology of the vessels concerned.
The pathogenesis of acellular intimal thickening, as described in this paper, is not certain. What the matrix beneath the endothelium represents in the present study is not known. In cases in which thickening is slight, endothelial cell hypertrophy may be the cause. Thickening (from one to two orders of magnitude) of endothelial cell cytoplasm of capillaries and venules resulting from increased numbers of endoplasmic reticulum cisternae, enlarged Golgi apparatus, numerous free ribosomes and other organelles has been observed in the parotid glands of rats injected with isoproterenol (Hand and Ho 1985). Decreased numbers of pinocytic vesicles and loss of fenestrae were also observed in affected vessels. A twofold increase in endothelial cell volume and an accompanying two- to threefold decrease in luminal volume have been reported in the retinal capillaries of cynomolgus monkeys after intravitreal injection of vascular endothelial growth factor (Hofman et al. 2001).
In humans, endothelial cell hypertrophy has been reported in patients with urticarial dermatitis (Jones and Eady 1984) and hypertrophic cardiomyopathy with dilated cardiomyopathy-like features (Takemura, Takatsu, and Fujiwara 1998). We were unable to find reports of endothelial cell hypertrophy in untreated or nondiseased animals or humans.
When thickening is prominent, the deposition of subendothelial matrix can contribute to the abnormal morphology of affected vessels. Several investigators have observed and characterized age-related increases in the thickness of the subendothelial space in large vessels of the thoracic and abdominal cavities of the rat. Fornieri, Quaglino, and Mori (1992) performed scanning electron microscopic analysis of the aorta in male Wistar rats at 1, 18, 60, 240, 480, and 720 days after birth. They found that although the tunica intima consisted essentially of internal elastic lamina up to sixty days of age, an increasing amount of extracellular matrix separated the endothelium from the internal elastic lamina in older rats. At twenty-four months, this matrix was sometimes “swollen” and contained abundant glycosaminoglycans. Toda (1978) performed electron microscopy on sections of abdominal and thoracic aorta from Wistar rats sacrificed at multiple time points between zero and three months and four and thirty-six months of age. In the subendothelial space, he found nothing at five days, multiple cystic structures and granular to rod-shaped elastin fibers at fifty days, the appearance of granular basement membrane at three months, and increased amounts of this material as well as cellular debris and fragments of internal elastic lamina at twelve months. Guyton, Lindsay, and Dao (1983) found basement membrane that merged with clumps and masses of granular material, microfibrils and fibrillar elastin in the subendothelium of the aortas of male Sprague-Dawley rats aged eleven to sixteen months, compared to elastin without microfibrils and basement membrane-like material only in rats aged ten weeks. These investigators also found cystic structures containing granular material and collagen fibers in the subendothelial space in older rats. They reported that the subendothelium increased fivefold in thickness in these animals compared to their ten-week-old counterparts. Bilato and Crow (1996) note that many of the structural alterations in great vessels that are attributable to aging may also affect the arteriolar bed.
Ikegami et al. (2002) induced reversible endothelial cell hypertrophy and widening of the subendothelial space in the mesenteric and pancreatic arteries by a twenty-four-hour continuous infusion of the dopamine agonist and vasodilator fenoldopam in male F344 rats. Increased rough endoplasmic reticulum and enlarged nucleoli were observed in the endothelium at one and three days postinfusion, whereas amorphous, electron-dense material was seen in the subendothelial space at three days postinfusion. The authors attributed the intracellular changes to increased protein synthesis, as marked positive reactions for von Willebrand’s factor and Factor VIII were identified immunohistochemically compared to saline-infused controls. No evidence of thrombosis was observed. The endothelium returned to normal by seven days postinfusion. The investigators reported evidence of medial hemorrhage and necrosis, which are known effects of dopaminergic agents on the vasculature (Mitsumori 1990).
In a time-course study of the aortic changes observed in the spontaneously hypertensive rat, Limas, Westrum, and Limas (1980) observed gradual thickening of the intima with ground substance, elastin, and collagen fibers for up to twenty weeks prior to the appearance of smooth muscle cells and fibroblasts in the subendothelial space that signaled the transition of the lesion to intimal hyperplasia. The ground substance in the subendothelial space was ruthenium red–positive, which the authors considered to be consistent with the presence of acid mucopolysaccharide. (Elastin microfibrils, which are also ruthenium red–positive [Fanning and Cleary 1985], may have contributed to what Limas, Westrum, and Limas observed.) Endothelial cell nuclei were described as being oriented parallel to the internal elastic lamina at five weeks, but at an angle or perpendicular to the lamina at twenty weeks (a similar orientation was observed at the light microscope in the current study). Increased endothelial cell permeability was demonstrated by the retention of injected horseradish peroxidase.
Although Limas, Westrum, and Limas (1980) acknowledge that patchy widening of the aortic subendothelial space (acellular intimal thickening) has been observed in several animal species and that this can be considered part of the normal aging process, they considered the lesions in the spontaneously hypertensive rat to be pathologic because of the magnitude and the progression of the changes observed compared to normotensive controls. They did not find evidence of endothelial erosions (fibrin, erythrocytes, or platelets in the subendothelial space) as an initiator of vascular injury, which implies that disruption of the endothelial cell layer is not required for intimal thickening (or intimal hyperplasia) to occur. There was no evidence of endothelial disruption in our principal study or in our current study, which is compatible with the presumption that this event is not required for the generation of intimal thickening or intimal hyperplasia.
Based on the information cited above, it is reasonable to infer that acellular intimal thickening can progress to intimal hyperplasia. Acellular intimal thickening was found at all injection sites in untreated male and female rats in the current study. In contrast, there was an apparent sex predilection and limited distribution of intimal hyperplasia in this study. Our animals were nineteen to twenty weeks old at study termination; it is likely that they were not old enough to have developed intimal hyperplasia, similar to the hypertensive rats that did not display evidence of subendothelial cell proliferation until after twenty weeks of age.
It is possible that the intimal thickening observed in our untreated control rats is age related and that vessels with this morphology are predisposed to develop intimal hyperplasia, similar to the way that certain areas of “adaptive intimal thickening” in humans (intimal hyperplasia as defined in this paper) are predisposed to develop atherosclerosis (Stary et al. 1992). Given that saline control animals from the principal study had intimal hyperplasia at more injection sites than did untreated controls (Table 2), saline injection, or the trauma from injection, may have initiated events leading to the increased incidence of intimal hyperplasia.
In conclusion, the histology of the dorsal skin of untreated control rats from the present study and saline control rats from the principal study was comparable. Saline injection did not fundamentally alter the morphology of the injection sites used for the principal study. Skin thickness was greater in males than in females. Although acellular intimal thickening occurred spontaneously in the dorsal skin of untreated male and female control rats, only males had a spontaneous incidence of intimal hyperplasia. No site predilection for intimal lesions was apparent for either sex. Saline injection, or the physical trauma of injection, may induce intimal hyperplasia; males appear more likely than females to develop the lesion. It is possible that acellular intimal thickening can progress to intimal hyperplasia under appropriate conditions.
Footnotes
The authors have not declared any competing interests.
