Abstract
In the field of developmental neuropsychology, interesting patterns of association between functional brain development at birth and neurodevelopmental outcomes during early childhood have been recently highlighted. In this work, we investigated a population of preterm neonates (n = 32) and (1) derived perinatal functional connectivity (p-FC) markers of prematurity, through direct comparison with a group of at-term borns and (2) tested the role of these markers as predictors of cognitive (COG), language (LANG), and socioemotional (SE) outcomes measured at 6, 12, 24, and 36 months. We did this by means of regression models in which functional markers were tested as predictors, after accounting for variability captured by metrics derived from current clinical practice and which were previously reported to be associated with a higher risk for adverse neurodevelopmental outcomes (i.e., pregnancy, perinatal weight and physiological conditions, and structural brain development variability). Our results revealed that (1) p-FC between the bilateral cerebellar hemispheres predicted SE outcomes at 6 months; (2) p-FC between the bilateral visual cortices predicted COG outcomes at 12 months; (3) p-FC between cortices related to language and emotional control predicted SE outcomes at 12 months; (4 and 5) p-FC between somatosensory motor and higher-order control cortices predicted LANG outcomes at 24 months and COG outcomes at 36 months; and (6 and 7) p-FC between language-related cortices predicted SE and COG outcomes at 36 months. Linear mixed model analyses with pattern mixture modeling, performed to comprehensively explore the longitudinal impact of significant perinatal functional predictors, demonstrated the importance of specific perinatal functional predictors of global neurodevelopment over time. We discuss our results by suggesting that perinatal functional imaging of the brain at rest could represent a very valuable and unique addition to current preterm clinical care routines, to improve pediatric clinical practices of the near future.
Introduction
Theoretical Background
Over the last four decades, a clear association between preterm birth and long-term cognitive and behavioral sequelae has been established (Bhutta et al., 2002; Brydges et al., 2018).
Throughout years, clinical factors heightening the risk for the occurrence of pre- and perinatal brain injury leading to adverse neurodevelopmental outcomes (Volpe, 2009a) have been identified, including conditions of the mother (Kundu et al., 2021) and the fetus during pregnancy (Murray et al., 2015), as well as weight (De Rose et al., 2021), vital systems functionality (Baron et al., 2011), and associated procedures for their treatment (Vinall et al., 2014) at birth. Furthermore, the introduction of structural magnetic resonance imaging (MRI) into pediatric clinical practice has prompted major advances in the detection and characterization of disruptive neurodevelopmental events and, consequentially, for the investigation of their association with neurodevelopmental outcomes (Counsell et al., 2003).
However, at present, our understanding of and ability to predict neurodevelopmental outcomes following the occurrence of different forms of brain injury suffers from a substantial inhomogeneity. Namely, on one hand, neurodevelopmental sequelae following severe brain injury (e.g., cystic white matter lesions following hypoxic-ischemic events or high-grade germinal matrix-intraventricular hemorrhage) have been extensively investigated, and their occurrence is highly predictable (Hielkema & Hadders-Algra, 2016). On the other hand, cognitive and behavioral outcomes following milder (e.g., punctate white matter lesions) or subtle (e.g., gray and white matter dysmaturation) disruptions are less understood, and their occurrence and specific manifestations are far less predictable. This difference pivots on at least two major factors. First, the occurrence of major insults is clearly detectable through perinatal structural MRI, and associated neurocognitive sequelae start manifesting in the weeks following delivery and in terms of major alterations of neurocognitive development (e.g., cerebral palsy and sensorimotor impairments). Conversely, minor insults are more difficult—or not possible—to detect at imaging examinations and show no visible signs of cognitive or behavioral impairments throughout the perinatal period (Inder et al., 2023); rather, manifest behavioral pictures may occur at later developmental stages in childhood, in the form of subtler impairments of all major cognitive functions (e.g., attention, memory, language, executive control, and self-regulation) (Bhutta et al., 2002; Chan et al., 2016). Second, it has to be taken into consideration that cognition and behavior (as well as their development) are more closely captured through the study of brain function, rather than structure (Park & Friston, 2013). To this end, structural investigations may limit our ability—and precision—to detect salient perinatal neurodevelopmental variability disclosing further cognitive and behavioral outcomes.
Functional imaging of the neonatal brain could help to overcome these limitations. First, considering that functional brain development occurs on the neurobiological substrate laid through structural brain development, functional investigations could represent a means to capture the occurrence of developmental disruptions (even the subtler), through the detection of their reflections on functional networks maturation (Hillary & Grafman, 2017). Second, capturing variability in functional brain organization could allow for the detection of perinatal neurodevelopmental variability more directly associated with cognitive and behavioral development, and could thus provide a contribution in increasing the specificity of predicted outcomes (Menon, 2011).
Evidence so far appears to support this view. Modulation of functional connectivity strength of all major resting-state networks (i.e., sensorimotor, language, executive control, and default mode) has been reported in premature newborns (PTBs) with no detectable structural alterations (Gozdas et al., 2018; Smyser et al., 2016), and estimates of subcortical-cortical perinatal functional connectivity have been shown to allow for a reliable classification between PTBs and at-term born (ATBs) peers (i.e., 80.2% mean accuracy) (Ball et al., 2016).
Moreover, associations between functional connectivity metrics at birth and neurodevelopmental outcomes in infancy and early childhood have been reported, showing a considerable degree of consistency between metrics source areas and networks and the specific associated outcomes. For instance, in our previous studies, we demonstrated that activity in the temporal cortices (Baldoli et al., 2015), as well as connectivity strength between the medial prefrontal cortex and the executive control network (Della Rosa et al., 2021), was associated with cognitive outcomes at 6 months. Toulmin and colleagues (2021) revealed functional connectivity between the thalamus and both premotor association cortices and primary sensorimotor cortices to be correlated to motor and cognitive outcomes at 20 months, respectively. Finally, functional connectivity of the ventral attention and default mode networks has been shown to be associated with development of behavioral inhibition at 24 months (Sylvester et al., 2018), while connectivity between the amygdala and both insular and cingulate cortices to predict internalizing symptoms at the same timepoint (Rogers et al., 2017).
We thus believe that perinatal rs-fMRI of the preterm brain holds the potential to become a pivotal addition to pediatric clinical practices of the near future. However, toward this aim, the predictive power of rs-fMRI metrics needs to be established first; particularly, the specific contribution of metrics derived from rs-fMRI needs to be assessed while taking into account neurodevelopmental outcomes variability which can be captured through metrics derived from information gathered as part of current pediatric clinical practice, and which may potentially impact brain functioning not only at birth but also mirroring in neurodevelopmental projections after birth.
Aims
To this end, the objectives of this study were twofold: (1) to identify perinatal functional markers of prematurity, between a group of PTBs with no major alterations of the brain parenchyma and for whom rs-fMRI data were collected in a short perinatal window right after birth (i.e., contextually to structural MRI scanning performed as part of preterm clinical routine practices), and a group of ATBs (i.e., Experimental Aim 1); (2) to test the identified functional markers of prematurity as predictors of cognitive (COG), language (LANG), and socioemotional (SE) developmental outcomes at 6, 12, 24, and 36 months (as assessed through Bayley-III scales), while accounting for outcomes variability captured by neurodevelopmental risk factors related to pregnancy (maternal and fetal conditions), weight and physiological conditions (i.e., vital systems functionality) between birth and discharge, and structural brain development (i.e., Experimental Aim 2).
Materials and Methods
Participants
Preterm Neonates
PTB neonates were selected from a larger sample of n = 51 (mean GW at birth 31.2 ± 2.1; range, 27.1–35.9) subjects who underwent a research protocol entailing the acquisition of rs-fMRI data at birth and follow-up neuropsychological examinations performed using the Bayley-III test battery (Bayley, 2006) (see Supplementary Materials, Section 1.1 for details on participants’ selection and inclusion criteria). As a result of the selection procedure, 19 subjects were excluded from the study, leading to a final sample of n = 32 PTB subjects. The mean GW at birth was 30.7 ± 1.9 (range, 27.1–34.7) (see Table 1 for descriptive statistics of PTBs and ATBs birth parameters). The study protocol was approved by the San Raffaele hospital ethical committee (date of approval, 10 April 2014). Written and verbal informed consent was provided by the PTB newborns’ parents.
Sample Descriptive Statistics.
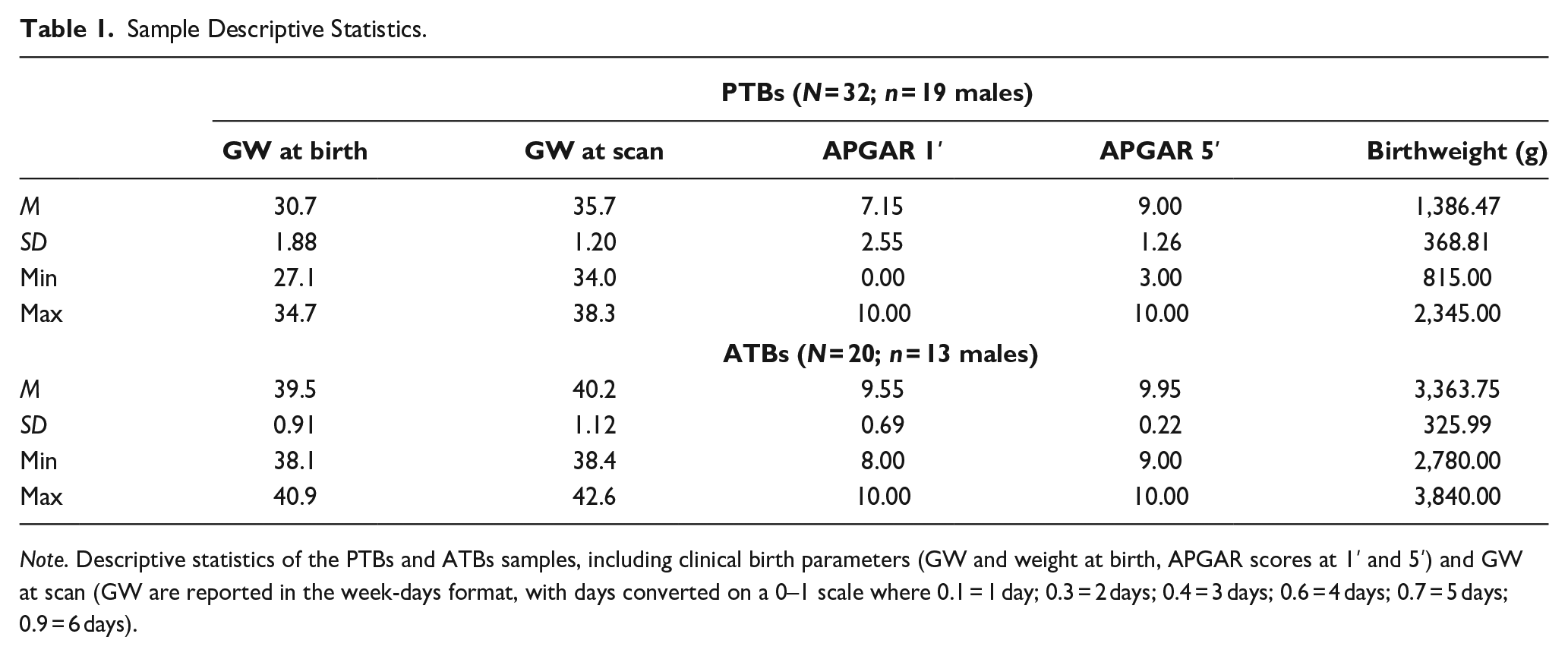
Note. Descriptive statistics of the PTBs and ATBs samples, including clinical birth parameters (GW and weight at birth, APGAR scores at 1′ and 5′) and GW at scan (GW are reported in the week-days format, with days converted on a 0–1 scale where 0.1 = 1 day; 0.3 = 2 days; 0.4 = 3 days; 0.6 = 4 days; 0.7 = 5 days; 0.9 = 6 days).
At-Term Neonates
ATB neonates were pooled from a larger cohort of mothers following a normal pregnancy and delivery (n = 31 subjects, mean GW at birth 39.2 ± 1; range, 37–40.9), who voluntarily adhered to the San Raffaele Hospital research protocol on premature birth, and served as a control group for Experimental Aim 1 (see later).
As a result of the selection procedure, 11 ATB subjects were excluded, leading to a sample of n = 20 subjects (see Supplementary Materials, Section 1.1 for details on participants’ selection and inclusion criteria). The mean GW at birth was 39.5 ± 0.9 (range, 38.1–40.9). The study protocol was approved by the San Raffaele hospital ethical committee (date of approval, 4 May 2018). Written and verbal informed consent was provided by the ATBs newborns’ parents.
Data Acquisition
For Experimental Aim 1, perinatal rs-fMRI data were collected for both the PTBs and ATBs samples. For Experimental Aim 2, clinical data and Bayley-III follow-up outcome scores were collected for the PTBs group only. Detail on rs-fMRI data and clinical data acquisition is provided in the Supplementary Materials, Sections 2.1 and 2.2.
Data Processing
Pre and Perinatal Conventional Clinical Neurodevelopmental Risk Scores
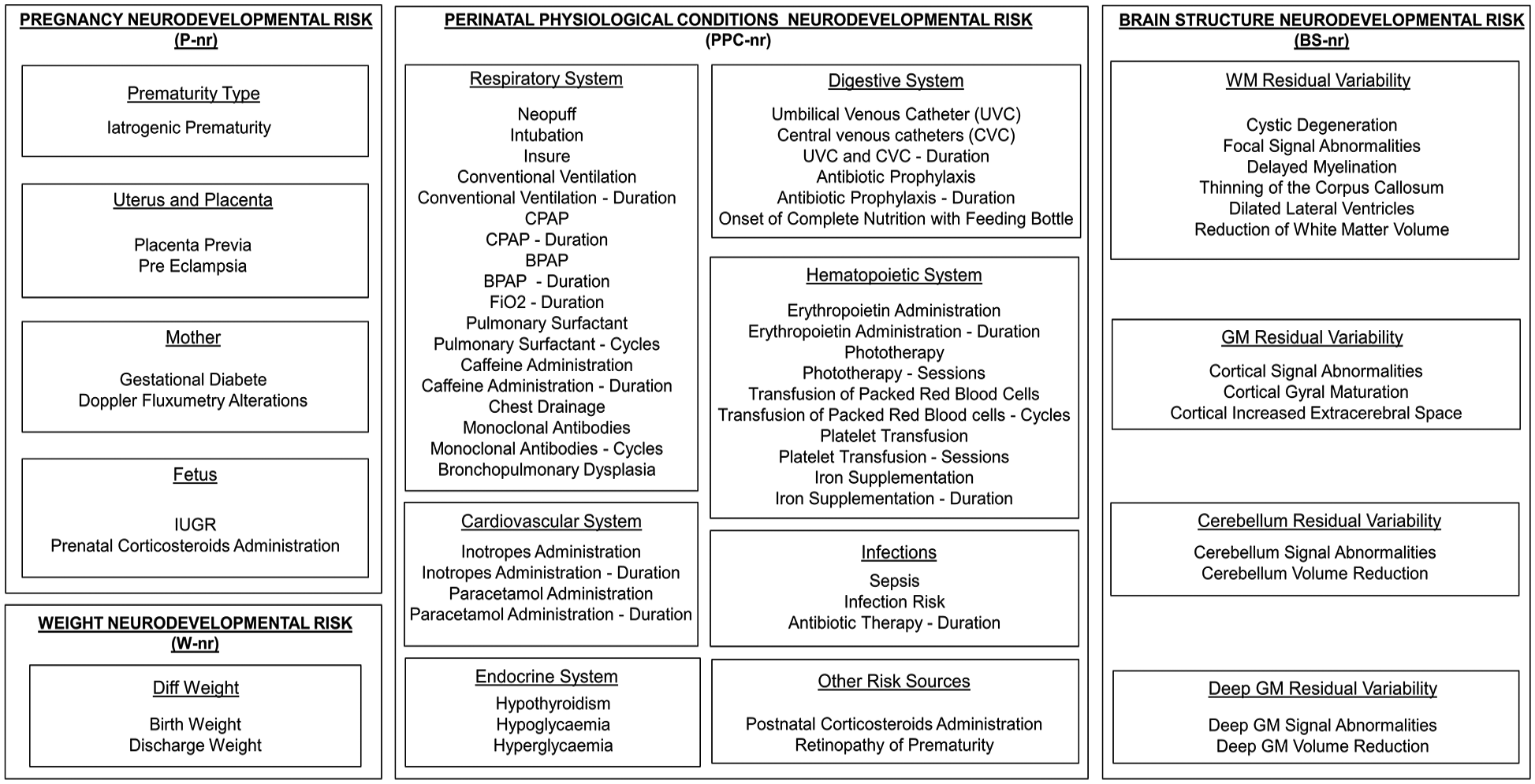
A summary “neurodevelopmental risk” (nr) score was estimated for each identified conventional source of clinical variability. The summary nr scores included the following:
A “pregnancy” neurodevelopmental risk (P-nr) score, capturing variability related to prematurity type, clinical conditions of the mother, of the uterus and placenta and of the fetus.
A “perinatal weight” neurodevelopmental risk (W-nr) score, capturing variability related to ponderal growth of the neonate between birth and discharge.
A “perinatal physiological conditions” neurodevelopmental risk (PPC-nr) score, capturing variability related to the wealth of all major vital systems-that is, respiratory, cardiovascular, hematopoietic, digestive, endocrine-, infections, and other relevant sources of physiological variability between birth and discharge.
A “perinatal brain structure” neurodevelopmental risk (BS-nr) score, calculated using the scoring system provided by George and colleagues (2017) and capturing residual structural variability related to white matter, gray matter, deep gray matter, and cerebellum development.
Details on nr scores composition are provided in Table 2. Details on nr scores calculation and composition (i.e., specific variables included and aggregation steps toward summary neurodevelopmental risk scores) are provided in Supplementary Materials, Section 3.1, along with details on variables distributions.
Neurodevelopmental Risk Scores Composition.
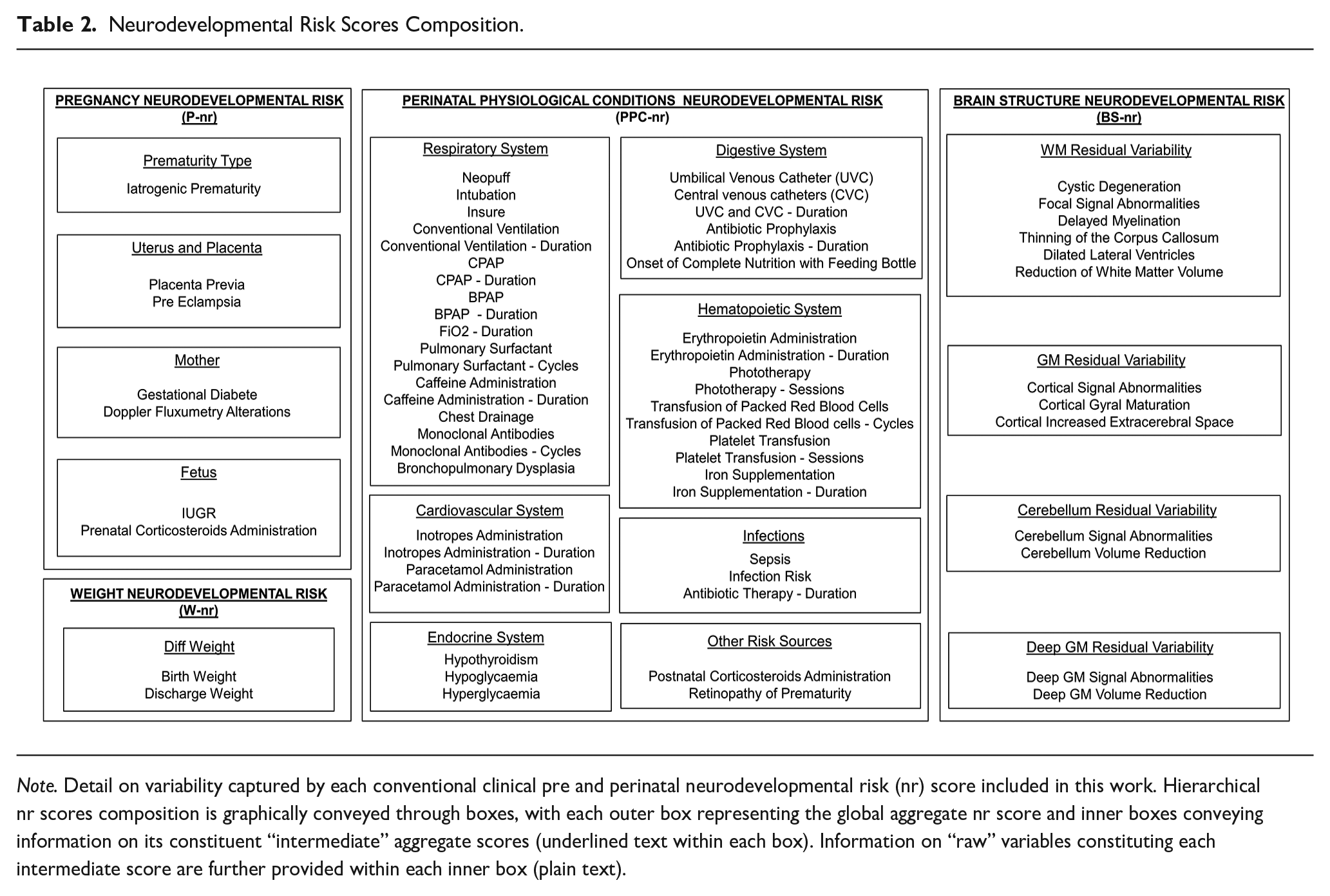
Note. Detail on variability captured by each conventional clinical pre and perinatal neurodevelopmental risk (nr) score included in this work. Hierarchical nr scores composition is graphically conveyed through boxes, with each outer box representing the global aggregate nr score and inner boxes conveying information on its constituent “intermediate” aggregate scores (underlined text within each box). Information on “raw” variables constituting each intermediate score are further provided within each inner box (plain text).
Resting-State fMRI Data Preprocessing
A dedicated, semi-automated pipeline, running on SPM12 (v7771—https://www.fil.ion.ucl.ac.uk/spm/software/spm12/), was created for preprocessing and spatial normalization of the rs-fMRI time-series to the Kuklisova-Murgasova et al. (2011) template anatomy (see Supplementary Materials, Section 3.2 for preprocessing details).
Bayley-III PTB Follow-Up Sample Characterization and Data Scoring
Among the total sample of PTBs (n = 32; mean GW at birth 30.7 ± 1.9) included in the study, 31 attended the 6-months evaluation timepoint (mean GW at birth 30.7 ± 1.9), 29 attended the 12-months evaluation timepoint (mean GW at birth 30.8 ± 1.8), 30 attended the 24-months evaluation timepoint (mean GW at birth 30.7 ± 1.8), and 22 attended the 36-months evaluation timepoint (mean GW at birth 30.8 ± 1.7). The mean GW at birth of each timepoint subsample fell within the lower and upper limits of the 95% confidence interval around the mean GW at birth of total sample (i.e., n = 32; 95% CI = [27, 34.4] GW). In addition, follow-up confidence interval evaluation indicated that, with 95% certainty, no significant “follow-up variation” emerged from one timepoint to the next and between all pairs of consecutive timepoints for all Bayley-III scales (see Table 3 and Supplementary Materials, Section 3.3 for complete details).
Bayley-III Follow-Up Variation.
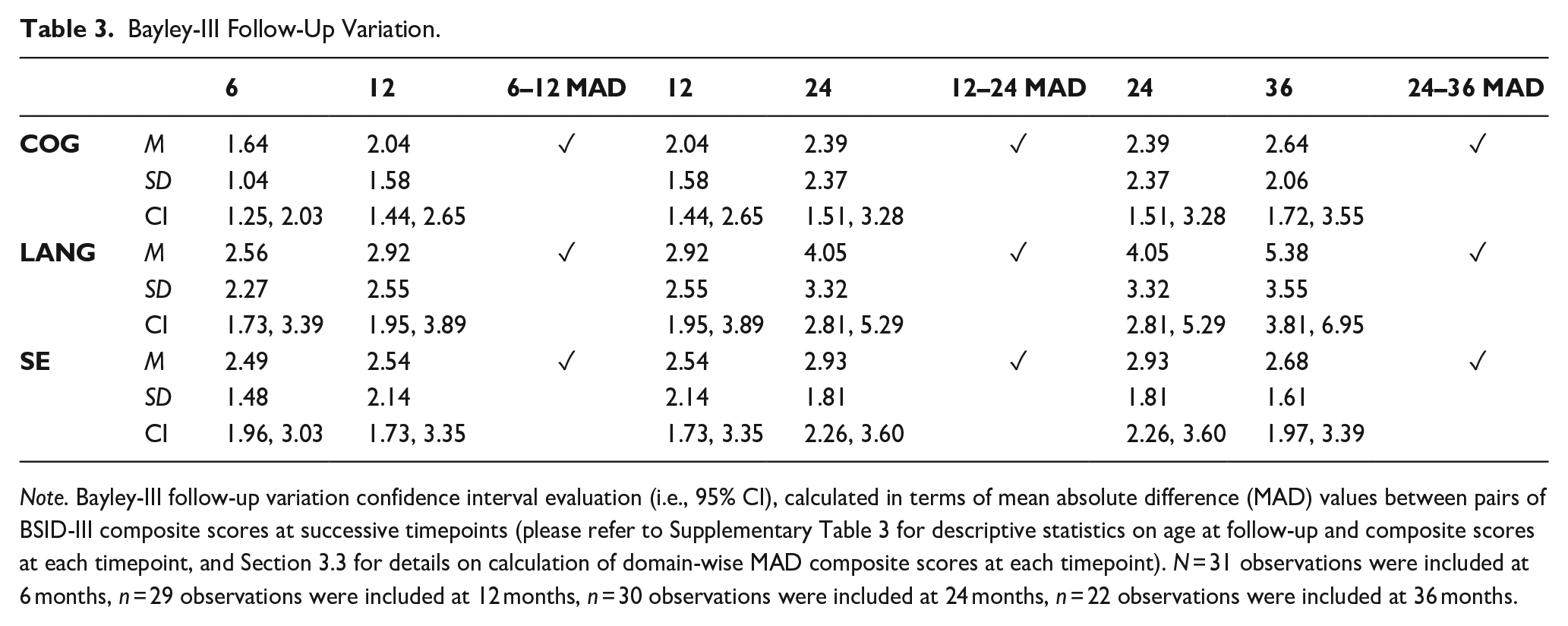
Note. Bayley-III follow-up variation confidence interval evaluation (i.e., 95% CI), calculated in terms of mean absolute difference (MAD) values between pairs of BSID-III composite scores at successive timepoints (please refer to Supplementary Table 3 for descriptive statistics on age at follow-up and composite scores at each timepoint, and Section 3.3 for details on calculation of domain-wise MAD composite scores at each timepoint). N = 31 observations were included at 6 months, n = 29 observations were included at 12 months, n = 30 observations were included at 24 months, n = 22 observations were included at 36 months.
Subsamples of participants included at each timepoint were quantitatively deemed (a) representative of the total sample of included PTBs (n = 32), in terms of mean GW at birth; and (b) representative of the subsample (n = 31) of PTBs included at the first timepoint (i.e., 6 months) across all consecutive paired follow-up timepoints for each scale, in terms of Bayley-III follow-up.
COG, LANG, and SE Bayley-III composite scores were finally computed for each subject at each timepoint (Bayley, 2006) (please see Supplementary Materials, Section 3.3 for details on composite scores distributions at each timepoint).
Data Analysis
Identification of Perinatal rs-fMRI Markers
Regions of Interest
Regions of interest (ROIs) included subcortical nuclei and primary and associative cortices belonging to functional systems underlying all major aspects of neurocognitive functioning, and previously reported to characterize—to varying degrees—neonatal brain functional development (see Zhang et al., 2019, for a review). A total of 51 ROIs was defined. The complete list of included ROIs, along with their localization, can be appreciated through Figure 1 (Results section). Details on ROIs selection and creation procedures are provided in Supplementary Materials, Section 4.1.
Experimental Aim 1
ROI-to-ROI Analysis—Identification of Perinatal Functional Markers of Prematurity
The effects of prematurity on perinatal functional connectivity were investigated using a ROI-to-ROI analysis using the CONN toolbox v21a (Whitfield-Gabrieli & Nieto-Castanon, 2012), including the 51 ROIs. A two-tailed t-contrast testing for differences between PTBs (1) and ATBs (−1)—while controlling for the effects of acquisition parameters and sex—was specified to test for the effects of prematurity.
First, the effects of prematurity on the brain’s functional architecture were explored, in terms of spontaneous ROIs grouping into ensembles of functionally coherent activity (functional system—FS). To this end, results were explored using the agglomerative hierarchical clustering (complete-linkage) algorithm, setting the criterion to .05 to favor functional similarity over spatial proximity clustering (Whitfield-Gabrieli & Nieto-Castanon, 2012). The multivariate pattern analysis (MVPA omnibus) inferential approach (Nieto-Castanon, 2022) was employed for FSs identification, using a p < .05 threshold corrected for false discovery rate (FDR). Second, perinatal functional connectivity markers of prematurity were identified as clusters of within- or between-FSs connection pairs marked by significantly different (i.e., higher or lower) connectivity strength for the PTBs compared to the ATBs group (i.e., p < .05 FDR-corrected, MVPA omnibus), using a p < .01 uncorrected threshold at the connection level (please see the ‘Step 1’ panel in Figure 2). A contrast coding for the main effect of each group was also specified, to explore the functional architecture features of the PTBs and ATBs brains independently and support interpretation of results of the main contrast. Results from this contrast are provided for consultation in Supplementary Materials, Section 4.2.
Experimental Aim 2
Perinatal Functional Markers of Prematurity—Metrics Creation and Bootstrapped Correlation Analyses With Bayley-III Outcomes
A summary metric score was then calculated for each identified functional connectivity marker of prematurity, for further modeling of perinatal neurodevelopmental variability captured by rs-fMRI in correlation and regression analyses. First, for each PTB subject, R-to-Z correlation values were extracted from each significant connection pair (i.e., ROI-to-ROI correlation) and corrected for the GW at scan, to control for the effects of different brain maturation rates at the time of rs-fMRI data acquisition. Second, marker-specific corrected R-to-Z connectivity values were aggregated into a “Summary Z Connectivity Metric” (S-ZCM) score (Della Rosa et al., 2021) (please see the ‘Step 2’ panel in Figure 2).
Bootstrapped Correlations, Regression and Linear Mixed Model Analyses
Bootstrapped Correlation Analyses
The association between each identified S-ZCM score and Bayley-III COG, LANG, and SE outcome scores at 6, 12, 24, and 36 months was assessed with correlation tests, using SPSS, v21. Statistical significance was assessed using a p < .05 threshold, along with bootstrapping of confidence intervals (n = 1,000 iterations). Only significant correlations between S-ZCM scores and Bayley outcomes were carried on to regression analyses (please see the ‘Step 3’ panel in Figure 2).
Regression Analyses
Associations between S-ZCM and Bayley-III outcomes surviving bootstrapped correlations analysis were tested for prediction. Specifically, to assess whether perinatal rs-fMRI could represent a valuable addition to information already gathered as part of current preterm care clinical practices (i.e., Experimental Aim 2), a hierarchical multiple linear regression model was specified for each significant S-ZCM score—Bayley-III outcome correlation, in which the Bayley-III outcome was included as the dependent variable and the S-ZCM score as candidate predictor, after accounting for variance captured by clinical neurodevelopmental risk scores. Namely, in each model neurodevelopmental risk scores related to (1) pregnancy (P-nr), (2) weight (W-nr), (3) physiological conditions (PPC-nr) and (4) residual brain structure variability (BS-nr) were added at blocks 1 to 4, respectively, followed by the (5) S-ZCM score at block 5. Multicollinearity between all variables was assessed before entering regression analyses, using a R > .7 threshold (Alin, 2010). Functional markers S-ZCM scores providing a significant contribution in outcomes variance explanation were deemed as functional predictors (please see the ‘Step 4’ panel in Figure 2).
Linear Mixed Model (LMM) Analyses
LMM analyses were conducted to explore the longitudinal impact of the identified perinatal functional predictors of time-point scale-specific BSID-III outcomes (i.e., S-ZCM scores yielding significant R2 changes in hierarchical regression models) (see Table 6) on the global change in predicted COG, LANG, and SE outcomes over time (i.e., the follow-up interval range of time-point scale-specific BSID-III outcomes for each significant perinatal functional predictor) for the entire cohort of PTBs (n = 32 subjects).
Pattern mixture modeling, which codes different patterns of missing data (i.e., no missing, sporadic missing, dropouts), was first used to evaluate whether changes in the developmental outcome scores over time were related to missing data patterns. Dummy variables were created for each missing data pattern and LMMs were conducted to evaluate if each identified missing data pattern predicted the global developmental outcome variable or interacted with time to predict changes in neurodevelopmental scores over time.
Second, a final linear mixed model was specified to investigate the ability of the significant perinatal functional predictors to predict the global developmental outcome variable (Global NeuroDevelopment) across BSID-III COG, LANG, and SE scales (Domain) and different time points (TimePoint) (i.e., 6, 12, 24, and 36 months) (please see the bottom panel in Figure 2). The fixed effects included the intercept, TimePoint, Domain, and the functional predictors. Interaction terms between TimePoint and each predictor, Domain and each predictor, and TimePoint and Domain were also included. A random intercept for each subject was included to account for the within-subject correlation over time and a restricted maximum likelihood (REML) estimation method was used. For the scope of this analysis, only significant main effects of perinatal functional predictors and significant interactions between TimePoint and predictors are reported.
Results
ROI-to-ROI Analysis
The effects of prematurity on perinatal brain’s functional architecture revealed the emergence of n = 8 FSs, which were labeled according to their association with major brain functional systems. Namely:
A “Cerebellum” (CRBM) FS, including the bilateral cerebellum;
A “Thalamo-Striatal” (TS) FS, including the bilateral thalamus, lentiform and caudate nuclei;
A “Somatosensory Motor” (SSM) FS, including the bilateral primary somatosensory and motor cortices;
A “Visual” (VIS) FS, including the bilateral occipital cortices;
A “Memory System” (MEM) FS, including the brainstem and bilateral hippocampus, parahippocampal cortex, and amygdala;
A “Auditory and Language System” (AL) FS, including the bilateral primary auditory cortices and Wernicke area and the right middle temporal gyrus and temporal pole;
A “Rostral Emotional Control System” (REC) FS, including the bilateral subthalamic nucleus, anterior insula, rostral ACC, and medial prefrontal cortex;
A “Higher-Order Cognition System” (HOC) FS, including the bilateral posterior cingulate/retrosplenial cortex, posterior parietal cortex, inferior parietal cortex, intraparietal sulcus, the left middle temporal gyrus and temporal pole and the bilateral frontal eye fields, dorsolateral prefrontal cortex, and inferior frontal gyrus. A graphical visualization of the identified FSs is provided in Figure 1.
Functional connectivity markers of prematurity were identified in terms of clusters of ROI-to-ROI connections both within and between the identified FSs, and were labeled accordingly. Results are presented in Table 4, in which complete detail on the identified markers, their composition (i.e., specific connections), and statistical significance are provided. Connections and markers functional connectivity strength values are provided in a complementary table mirroring (Table 4), along with reference literature highlighting the putative relevance of each FS for early cognitive and behavioral development (please see Supplementary Materials, Section 4.3).
Perinatal Functional Connectivity Markers of Prematurity.
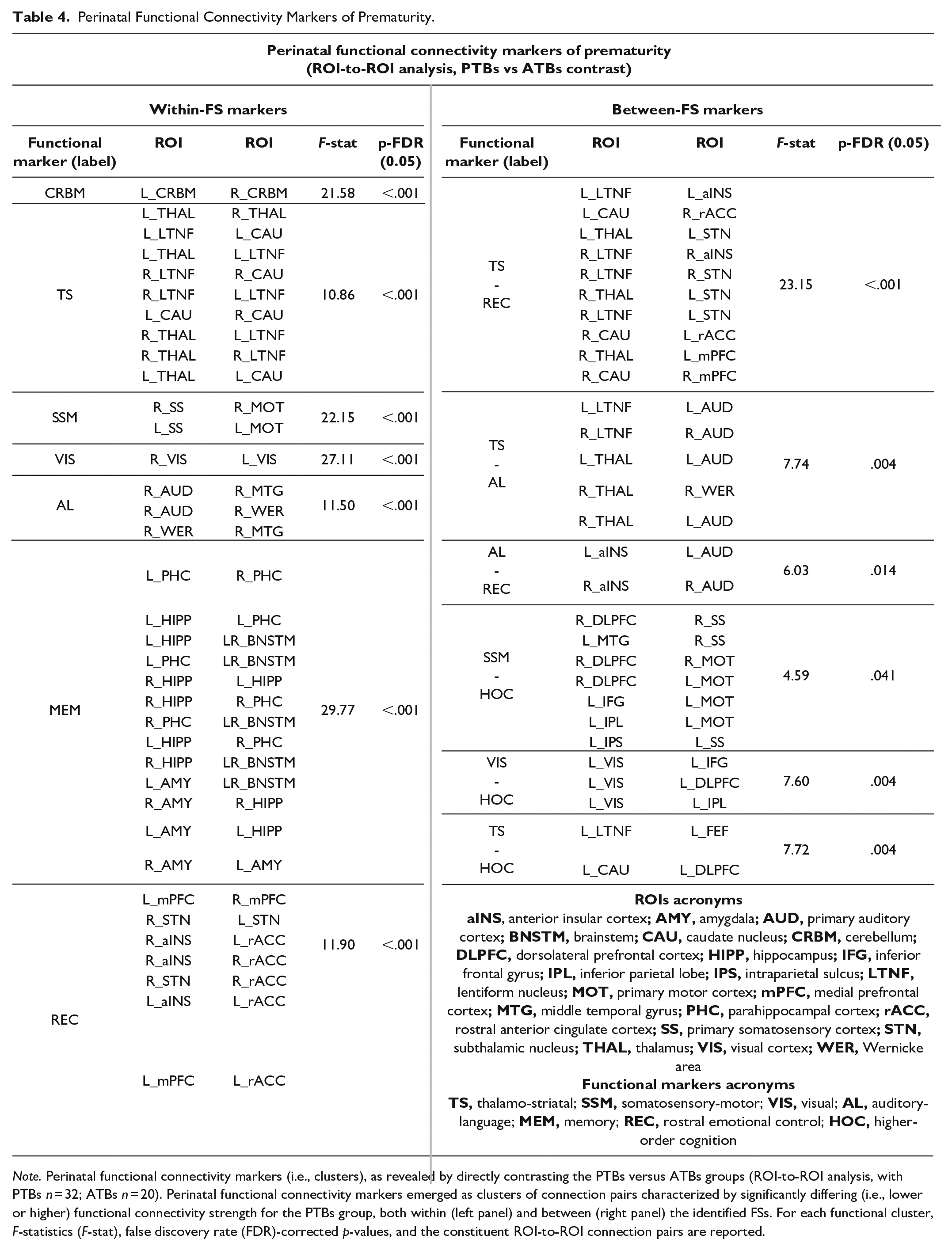
Note. Perinatal functional connectivity markers (i.e., clusters), as revealed by directly contrasting the PTBs versus ATBs groups (ROI-to-ROI analysis, with PTBs n = 32; ATBs n = 20). Perinatal functional connectivity markers emerged as clusters of connection pairs characterized by significantly differing (i.e., lower or higher) functional connectivity strength for the PTBs group, both within (left panel) and between (right panel) the identified FSs. For each functional cluster, F-statistics (F-stat), false discovery rate (FDR)-corrected p-values, and the constituent ROI-to-ROI connection pairs are reported.
Bootstrapped Correlations, Regression and Linear Mixed Model Analyses
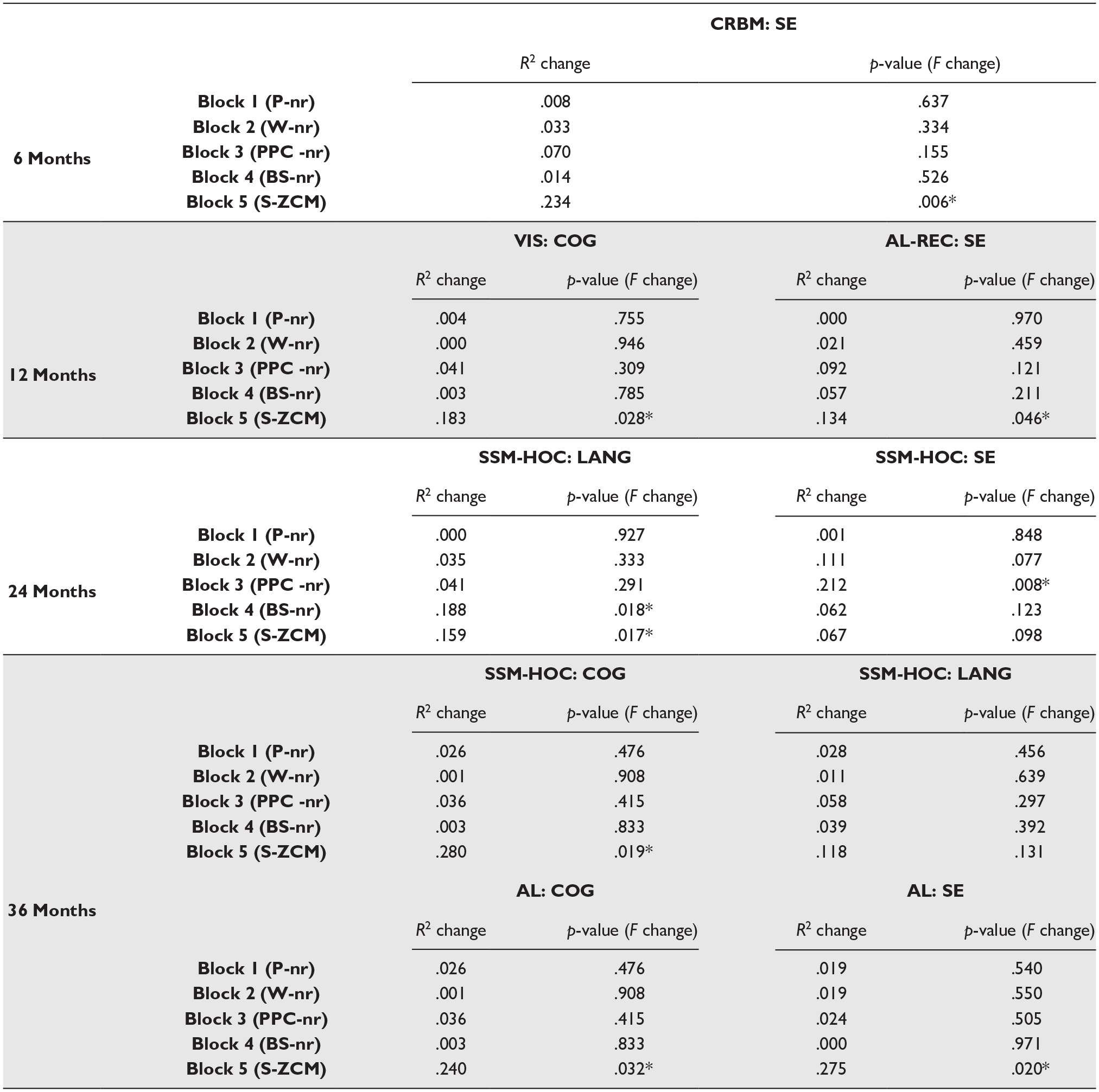
Among the significant functional connectivity markers of prematurity identified through the ROI-to-ROI analyses (n = 13), nine survived bootstrapped correlation testing for association with Bayley outcomes and seven emerged as significant predictors of neurodevelopmental outcomes in hierarchical multiple regressions models accounting for pre- and perinatal variability.
Correlation coefficients and p-value significance for association of functional markers with Bayley outcomes following bootstrapped correlation, as well as beta coefficients, t-test values, and p-value significance following regression analyses for functional predictors are provided in Table 5. In addition, R2 % change and F change significance values at each regression block for each model are provided in Table 6.
Perinatal Functional Predictors Identification.
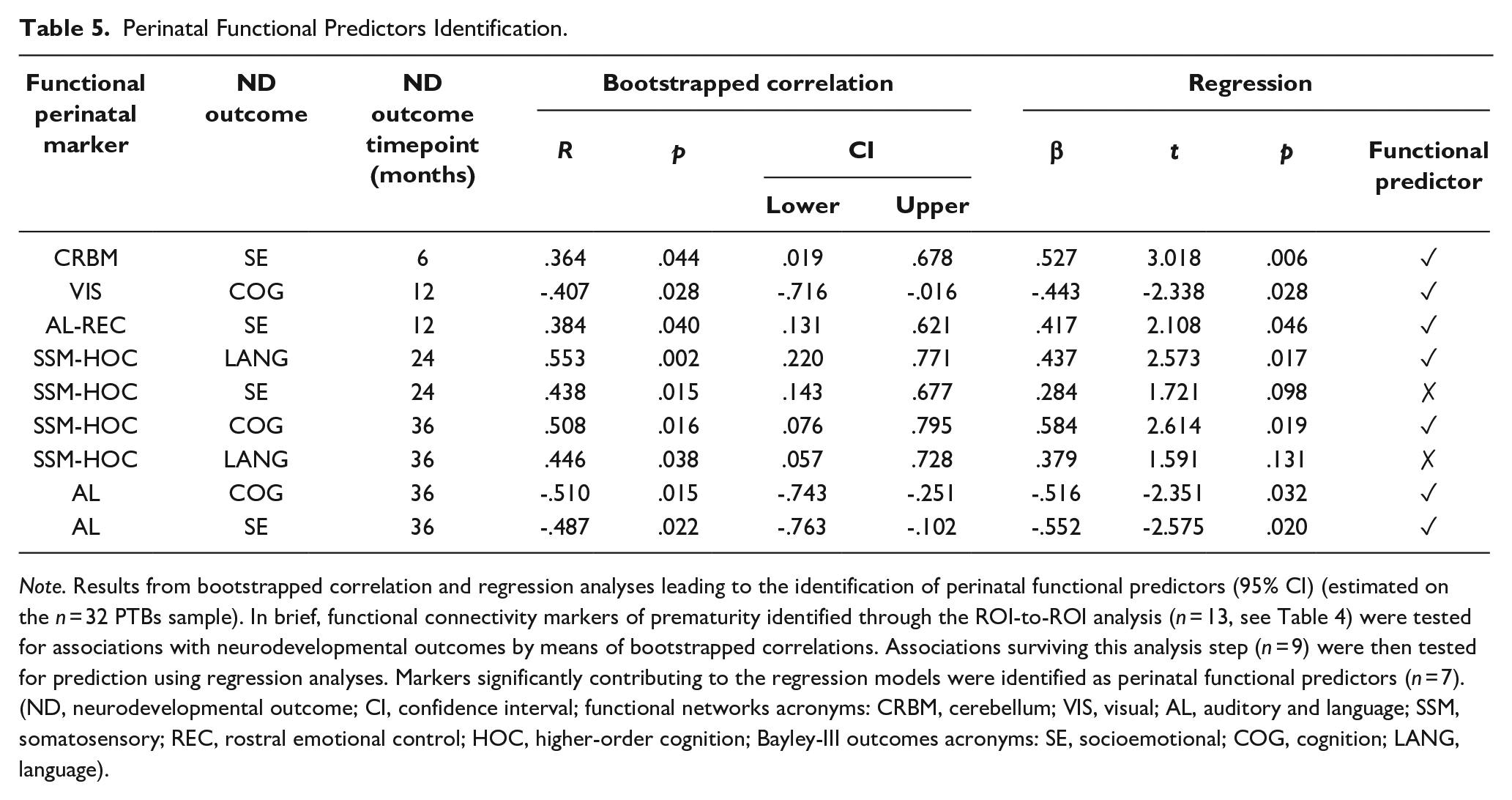
Note. Results from bootstrapped correlation and regression analyses leading to the identification of perinatal functional predictors (95% CI) (estimated on the n = 32 PTBs sample). In brief, functional connectivity markers of prematurity identified through the ROI-to-ROI analysis (n = 13, see Table 4) were tested for associations with neurodevelopmental outcomes by means of bootstrapped correlations. Associations surviving this analysis step (n = 9) were then tested for prediction using regression analyses. Markers significantly contributing to the regression models were identified as perinatal functional predictors (n = 7). (ND, neurodevelopmental outcome; CI, confidence interval; functional networks acronyms: CRBM, cerebellum; VIS, visual; AL, auditory and language; SSM, somatosensory; REC, rostral emotional control; HOC, higher-order cognition; Bayley-III outcomes acronyms: SE, socioemotional; COG, cognition; LANG, language).
Hierarchical Multiple Regression Analyses Results.
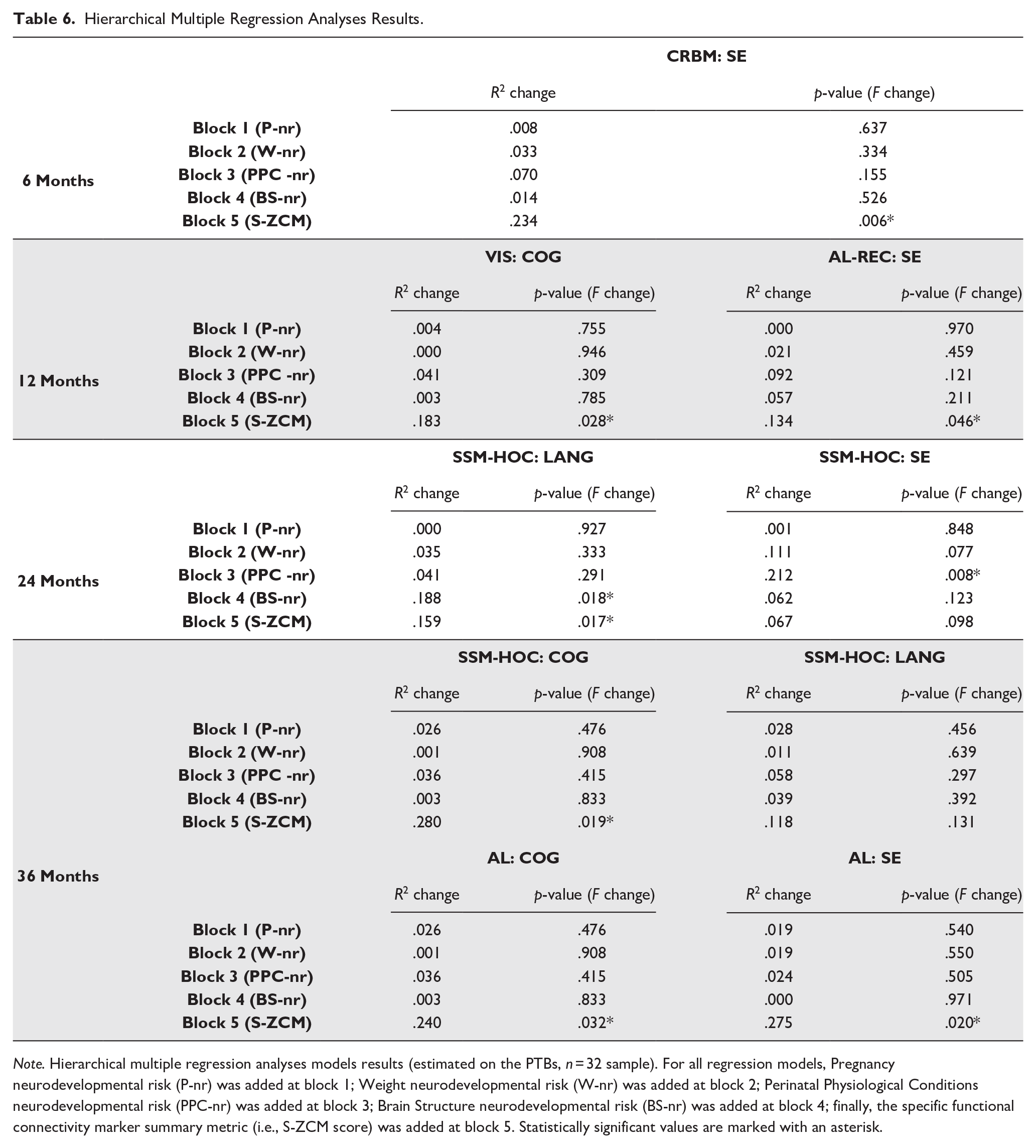
Note. Hierarchical multiple regression analyses models results (estimated on the PTBs, n = 32 sample). For all regression models, Pregnancy neurodevelopmental risk (P-nr) was added at block 1; Weight neurodevelopmental risk (W-nr) was added at block 2; Perinatal Physiological Conditions neurodevelopmental risk (PPC-nr) was added at block 3; Brain Structure neurodevelopmental risk (BS-nr) was added at block 4; finally, the specific functional connectivity marker summary metric (i.e., S-ZCM score) was added at block 5. Statistically significant values are marked with an asterisk.
Pattern mixture modeling found no significant differences in the change in Global NeuroDevelopment outcome between participants with missing data and those without missing data over time. There was a significant main effect of the CRBM perinatal functional predictor on Global NeuroDevelopment (β = 31.98, SE = 16.05, t(129.04) = 1.99, p = .048), indicating that higher CRBM S-ZCM scores at birth were associated with higher Global NeuroDevelopment scores over time. A significant main effect was also found for the AL-REC perinatal functional predictor (β = 26.10, SE = 11.96, t(128.67) = 2.18, p = .031). Higher AL-REC S-ZCM scores at birth were predictive of higher Global NeuroDevelopment scores over time.
There was a significant interaction between TimePoint and the CRBM perinatal functional predictor, particularly at 24 months (β = 41.06, SE = 17.14, t(117.30) = 2.40, p = .018), suggesting that the positive effect of the CRBM perinatal functional predictor on Global NeuroDevelopment scores over time was stronger at 24 months compared to other time points. A significant interaction was also found between TimePoint and the AL perinatal functional predictor at 24 months (β = 24.98, SE = 12.17, t(112.07) = 2.05, p = .042), indicating that the positive effect of the AL perinatal functional predictor on Global NeuroDevelopment scores over time was also more pronounced at 24 months. In addition, a significant negative interaction between TimePoint and the SSM-HOC perinatal functional predictor at 12 months (β = −13.80, SE = 4.80, t(119.43) =−2.88, p = .005) was evident. This negative interaction suggests that higher SSM-HOC S-ZCM scores at birth are associated with lower Global NeuroDevelopment scores at 12 months as compared to other timepoints and with respect to the reference timepoint (i.e., 36 Months).
Discussion
Perinatal Functional Connectivity Markers of Prematurity
Following birth, neurocognitive development undergoes an incredibly rapid maturation trajectory, with the infant shifting from a predominantly reflexive, exogenously driven behavioral repertoire to increasingly controlled and goal-directed interactions with the surrounding physical and social environment paralleled by the emergence of increasingly complex cognitive skills (D. H. Feldman, 2004; Langer, 1990, 2000; Piaget, 1935; Walle & Campos, 2014). Subcortical-cortical and cortical-cortical functional systems supporting this steep neurodevelopmental trajectory are ontogenetically scheduled to be trained prenatally, with a spurt in maturation of cortical-cortical circuitry particularly occurring during the third trimester of gestation (Canini et al., 2020; Jakab, 2019). Premature birth opens a window of vulnerability on the physiological development of brain functional systems upholding neurodevelopment, observed so far in terms of altered resting state networks emergence and configuration characterizing the preterm population scanned both prior to and at term-equivalent age (Bouyssi-Kobar et al., 2019; Cao et al., 2017; Doria et al., 2010; Fransson et al., 2007, 2009, 2011; Gao et al., 2014; He & Parikh, 2016; Hu et al., 2022; Smyser et al., 2010, 2016; Van Den Heuvel et al., 2015; Wang et al., 2021). In line with this evidence, our ROI-to-ROI analysis revealed the existence of perinatal functional connectivity markers of prematurity, as compared to at-term birth, in terms of a modulation of connectivity (1) within the cerebellar, thalamo-striatal, and primary cortical components of the sensorimotor systems (CRBM, TS, SSM, and VIS markers); (2) between the thalamo-striatal sensorimotor system component and rostral emotional control cortices (TS-REC marker); (3) between the thalamo-striatal and primary cortical sensorimotor system components and lateral parietal and prefrontal cortices related to higher-order executive control (i.e., TS-HOC, SSM-HOC, VIS-HOC markers); (4) within the auditory/language system (AL marker); (5) between the auditory/language system and both thalamo-striatal motor nuclei (TS-AL marker) and rostral emotional control areas (AL-REC marker); (6) within the rostral emotional control system (REC marker); and (7) within the medial temporal lobe memory system (MEM marker). In other words, the effects of prematurity were observed in terms of a capillary modulation of functional connectivity patterns occurring within and between all major functional systems of the developing brain, underlying crucial aspects of early neurocognitive development (see Table 4 and Supplementary Table 5 for details). Provided the experimental aims of this work, we will further discuss the role of perinatal functional markers, emerging as neurodevelopmental outcomes predictors and the alignment observed between specific functional predictor/s, neurodevelopmental outcome/s, and the dominant neurocognitive milestones related to the predicted outcome emerging at specific developmental timepoints.
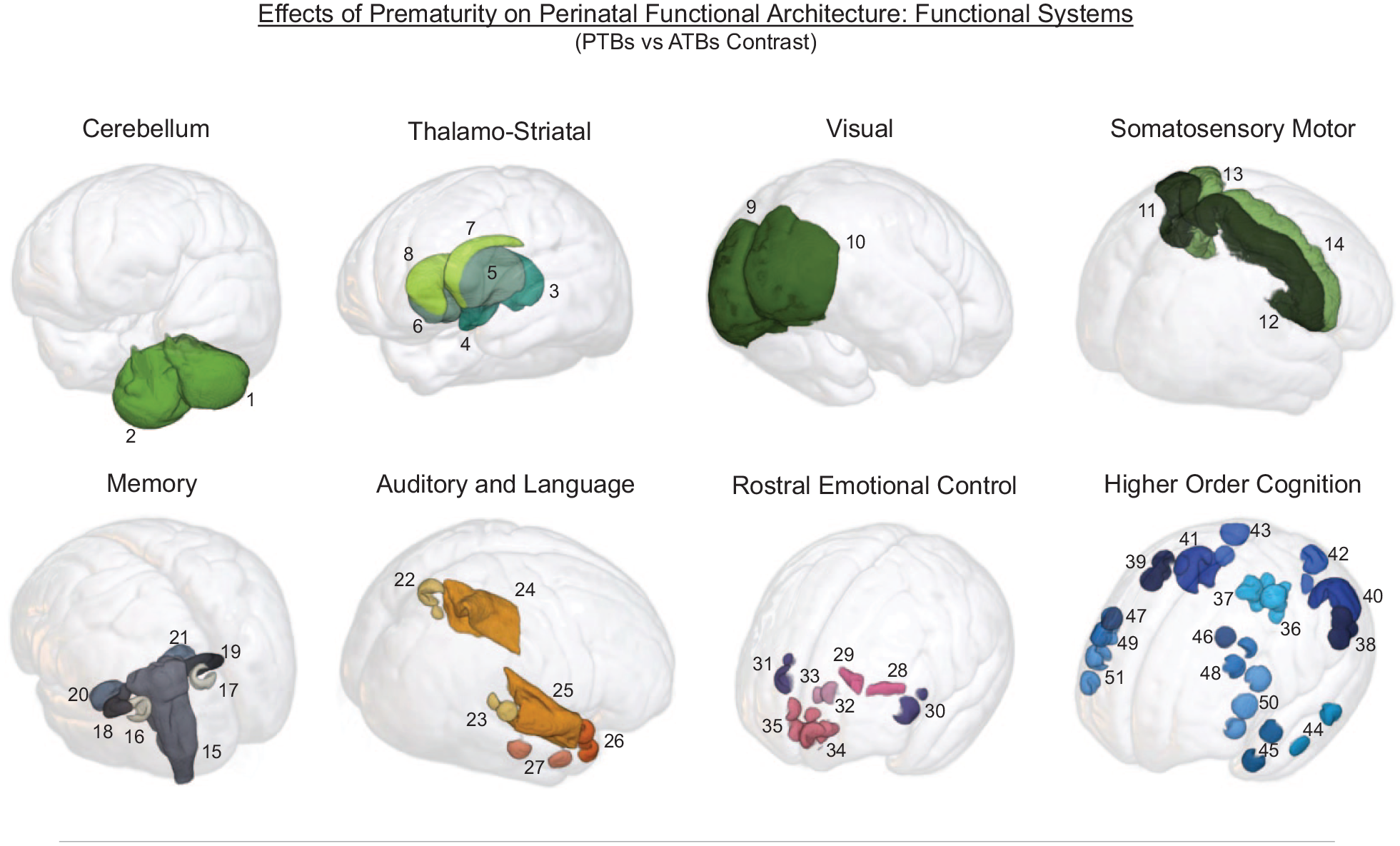
Effects of Prematurity on the Perinatal Functional Architecture. The effects of prematurity on perinatal functional organization, as revealed through the investigation of spontaneous ROIs grouping into functional systems (ROI-to-ROI analysis, PTBs vs ATBs contrast; with PTBs n = 32, and ATBs n = 20). An overlay of ROIs used for signal extraction on the Kuklisova-Murgasova template anatomy is provided for each identified functional system.
Perinatal Functional Connectivity Markers and Outcomes Prediction
“Cerebellum” Perinatal Functional Predictor: SE 6 Months
Perinatal functional connectivity strength between the left and right cerebellum predicted socioemotional development at 6 months, in line with the central role of the cerebellum in procedural skills learning (Ito, 2000; Paulin, 1993), and with early neurocognitive development features characterized by a progressive emancipation from reflexive sensorimotor repertoires and by the emergence of controlled perceptual and motor interactions (e.g., voluntary tracking of objects and reach-to-grasp behaviors) (Limperopoulos et al., 2007; Shultz et al., 2018) occurring before the establishment and consolidation of cortical executive control circuits (Koziol et al., 2012). The cerebellum undergoes a dramatic growth between 24 and 40 weeks (i.e., a 5-fold volume increase) playing a central role in early cortical development (Volpe, 2009b). Accordingly, cerebellar perinatal damage—often associated with prematurity—and developmental malformations typically result into disruptions of foundational aspects of neurocognitive development, including delayed acquisition of motor milestones, impaired gross and fine motor skills, attention-deficit hyperactivity disorder (ADHD), developmental dyslexia and affective disorders (Brossard-Racine et al., 2015; Stoodley & Limperopoulos, 2016; Volpe, 2009b), and—most notably—autism spectrum disorders (ASD) (D’Mello & Stoodley, 2015; Limperopoulos et al., 2007).
“Visual” Perinatal Functional Predictor: COG 12 Months
Cognitive development at 12 months was predicted by perinatal functional connectivity strength of the visual system. After birth, development of the visual system is fast and follows a trajectory of functional integration of primary and extra-striate visual cortices with temporal, parietal, and prefrontal cortices, culminating in the maturation of the ventral and dorsal visual pathways occurring between 9 and 12 months (Mercuri et al., 2007). Anchored in connectivity with the temporal lobe, the ventral (“vision for perception”) pathway holds a semantic functionality, underlying perceptual encoding and recognition of objects, faces, and scenes allocentric properties and spatial relationships. Anchored in connectivity with parietal cortices, the dorsal (“vision for action”) pathway underlies visual control of actions, including real-time mapping of the objects’ position in space as well as encoding, selection, execution, and monitoring of action opportunities endowed by the object’s properties (Koziol et al., 2012; Milner & Goodale, 2008). In turn, the maturation of these pathways (and of underlying functional circuitry) grounds the emergence of visual modules for the control of gaze (saccades, pursuit, and head orientation), reaching and grasping behaviors and crawling (for a review, see Braddick & Atkinson, 2011), underlying the marked increase in the infant’s control on sensorimotor repertoires observed by the end of the first year (R. Feldman, 2004; Langer, 1990; Shultz et al., 2018). As put forward by Posner and Rothbart (Posner & Rothbart, 2000; Rothbart et al., 2011), orienting could underlie the first instantiations of self-regulatory control, followed by the emergence of central executive control networks (Rothbart et al., 2011—and see the next paragraph). Visual deficits are common among preterm borns and particularly affect processing of motion and visual sensorimotor control abilities (i.e., dorsal pathway development) further resulting into motor, visuo-spatial memory and attention/executive control deficits (Atkinson & Braddick, 2007; Leung et al., 2018).
“Auditory and Language AL—Rostral Emotional Control” Perinatal Functional Predictor: SE 12 Months
Modulation of functional connectivity strength between the primary auditory and the anterior insular cortex did also emerge as an outcome predictor at 12 months, and particularly of socioemotional development. Integration with cortical executive control systems throughout the first year does not only map onto development of visuo-motor control systems but also marks the emergence of self-regulatory dynamics, through which the infant gains increasing control capability over visceral and emotional distress (Ruff & Rothbart, 1996; Voigt et al., 2013). Attentional redirection from internal discomfort feelings to external perceptual soothing cues operated by the caregivers is critical to this end (Als et al., 2004; Calkins & Hill, 2007; Posner & Rothbart, 2000; Shultz et al., 2018). The auditory/vocal communication channel (along with tactile stimulation and attentional visual reorientation) plays a particularly prominent role in the establishment and consolidation of early infant-caregivers’ interactions (Matsuda et al., 2014; Shultz et al., 2018), allowing the newborn to encode physiological and emotional states into behavioral expressions directed to the caregivers and, complementarily, to decode feedback provided by them (Oller et al., 2019; Shehata-Dieler & Wermke, 2018). It is through these socioemotional interactions with the caregivers that the infant progressively comes to learn the (relational) meaning of its behavioral repertoires, and that the groundings of semantic knowledge are thus established (R. Feldman, 2007; Topping et al., 2013). Functional integration between the auditory system and the insula plays a key role in coding of emotional speech prosody (Filippa et al., 2022) and direction of attentional resources toward salient sounds, tuning prototypical cognitive mechanisms for language acquisition (Uddin et al., 2017). This is in line with the known role of the anterior insular cortex as a pivot of the salience network as well as—as more recently highlighted—as a “gatekeeper of executive control” (Molnar-Szakacs & Uddin, 2022). Accordingly, poorer regulation of emotional reactivity during the first months of postnatal life is associated with poorer domain-general executive control ability measured during childhood (Posner & Rothbart, 2000; Ursache et al., 2013), an evidence which is well in line with the emergence of executive function deficits (Taylor & Clark, 2016) and emotionally dysregulated profiles (see below) observed in the preterm population.
“Somatosensory Motor—Higher Order Cognition” Perinatal Functional Predictor: LANG 24 Months and COG 36 Months
Building on previously acquired perceptual, motor, communicative, and emotional control repertoires, upright locomotion emerges toward the end of the first year, marking a bloom of infants’ interactions with the environment and—in parallel—of cognitive development throughout the second and third year (Walle & Campos, 2014). Accordingly, and interestingly, our results further revealed that strengthening of perinatal functional connectivity between the primary sensorimotor cortices (Bulea et al., 2015; Siegel et al., 2015) and regions related to (1) domain-general (IPS, DLPFC) (Bressler & Menon, 2010; Duncan, 2010) and semantic (MTG, IFG) (Whitney et al., 2011) executive control, as well as to (2) automatic processing of semantic information (i.e., IPL) (Noonan et al., 2013) predicted language and cognitive development at 24 and 36 months. Functional integration (and balance) between sensorimotor and cortical control systems underlies flexible selection and retrieval of relevant semantic information based on ongoing internal and external task demands, monitoring, and real-time adjustment of behaviors based on interaction outcomes. To this end, functional integration is not only crucial for further refinement of sensorimotor repertoires, but represents the very groundings for the emergence of higher-order cognitive abilities (Mesulam, 1998). Accordingly, disruption of functional integration between sensorimotor and higher-order systems is associated with a plethora of neurodevelopmental disorders and psychiatric conditions (Banaschewski et al., 2001; Mosconi & Sweeney, 2015). Furthermore, as shown by Cao and colleagues (2017) (further expanding findings from Fransson et al., 2011), functional development of sensorimotor cortices during the preterm period plays a central role in the emergence of the brain’s global functional architecture. Accordingly, functional connectivity strength between subcortical and cortical components of the sensorimotor system and association cortices at birth has been shown to be associated with cognitive/behavioral outcomes in childhood in preterm born newborns (Peyton et al., 2020; Toulmin et al., 2021).
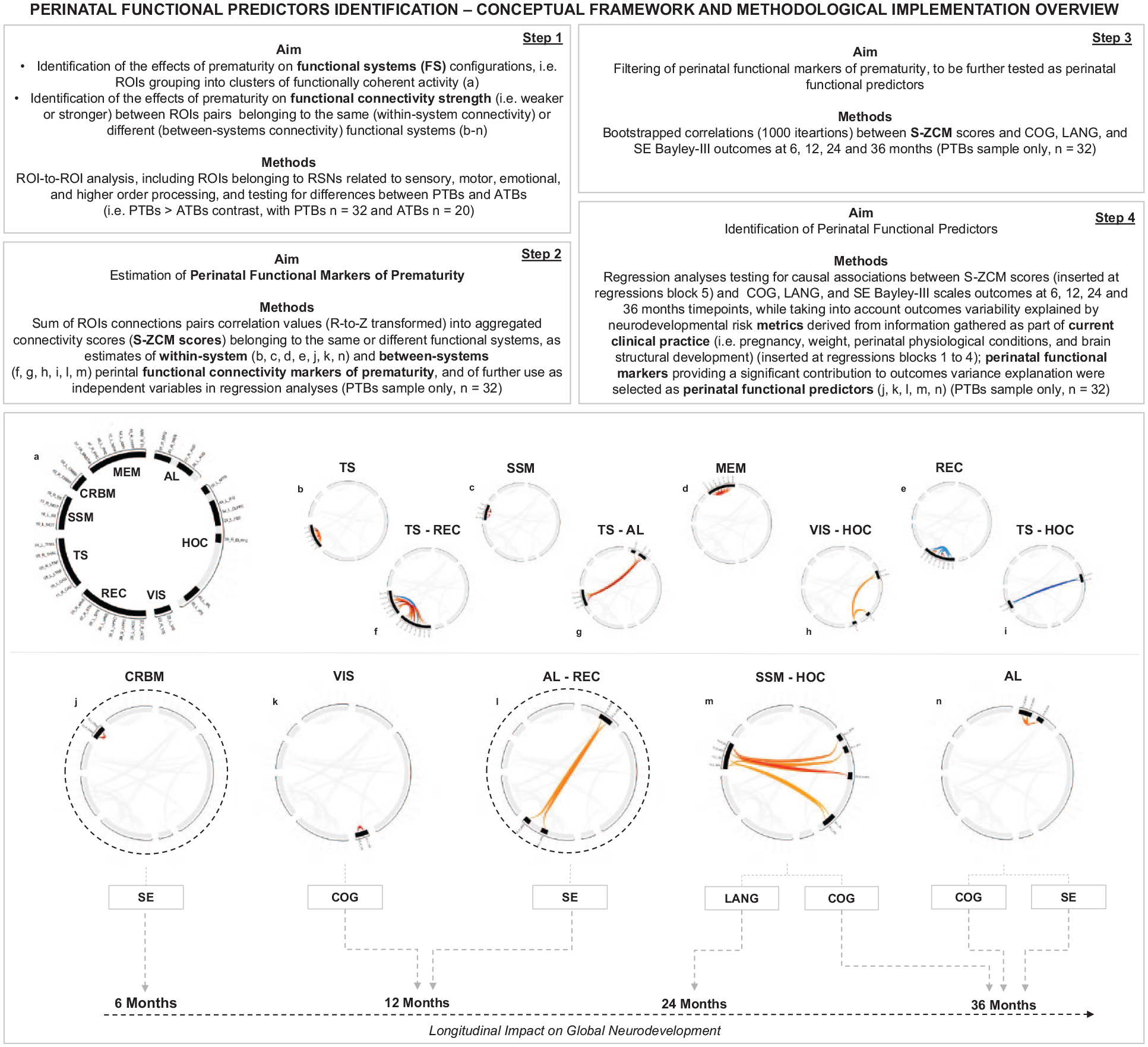
Step-wise overview of the conceptual framework employed in this study for the identification of perinatal functional predictors, along with its methodological implementation. Causal associations between perinatal functional predictors and their predicted outcomes at specific timepoints are denoted by dotted gray lines. Perinatal functional predictors further found to exert a longitudinal impact on global neurodevelopment are marked with a dotted black ring (please see results from the LMM analyses). Further details on the identified functional systems are provided in Section 2.5; further details on functional systems composition are provided in Figure 1; further details on within-system and between-systems perinatal functional markers (and/or predictors) composition (i.e., specific ROI-to-ROI connections included) are provided in Table 4 and in Supplementary Table 5. Functional systems acronyms: TS, thalamo-striatal; CRBM, cerebellum; SSM, somatosensory motor; VIS, visual; MEM, memory; AL, auditory and language; REC, rostral emotional control; HOC, higher-order cognition. Bayley-III scales acronyms: COG, cognitive; LANG, language; SE, socioemotional.
“Auditory and Language AL” Perinatal Functional Predictor: COG and SE 36 Months
Cognitive development at 36 months was also predicted by functional connectivity modulation of the auditory/language system, along with socioemotional development. Stimulation of the auditory/language system begins prenatally, through exposure of the fetus to maternal voice (Ghio et al., 2021), and immediately after birth shows a substantial maturation of functional circuitry underlying receptive speech abilities (Nallet & Gervain, 2021). Behaviorally, phonetic discrimination and prosodic information encoding abilities are already observed in the first weeks of postnatal life (Dehaene-Lambertz et al., 2006). Refining of the expressive behavioral repertoire occurring during the first years of life (Fenson et al., 1994) is paralleled by maturation of the language network and particularly characterized by functional integration between temporal and inferior frontal cortices (Skeide & Friederici, 2016). Importantly, verbal communication abilities represent a crucial channel through which parent-infant synchrony patterns are formed and consolidated early in life (R. Feldman, 2007) and these, in turn, are critical for shaping of subsequent cognitive and socioemotional developmental trajectories (R. Feldman, 2007; R. Feldman & Masalha, 2010). Accordingly, deafness (Vaccari & Marschark, 1997) and developmental language delays are associated with the emergence of poorer socioemotional development, further resulting into lower relational competence (e.g., lower prosocial peer relations, empathy, compliance, motivation mastery) and behavioral problems (e.g., internalizing/externalizing behaviors) (Henrichs et al., 2012; Thurm et al., 2018). Furthermore, language delay and associated dysfunctional socioemotional profiles do also contribute to the early onset of various psychiatric conditions, including ADHD, oppositional defiant and conduct disorder, anxiety, and depression (Im-Bolter & Cohen, 2007).
Perinatal Functional Integration With Executive-Control Cortices as a Prominent Predictive Feature
Summing up, our results suggest that the association between perinatal functional markers and their predicted neurocognitive outcomes develops along a gradient of increasing integration between sensorimotor, emotional, and language and executive function systems. To this respect, a further level of detail emerges when taking into consideration the longitudinal impact of the identified perinatal functional predictors. Segregation and integration of brain networks are two key features of brain functioning, directly grounding the emergence of task active and task passive networks interactions which are, in turn, necessary for tuning of cognitive resources and dynamic adaptation to environmental requests (Cohen & D’Esposito, 2016; Della Rosa et al., 2021; Schurz et al., 2020). Segregation is the specialization of individual networks for distinct functions, whereas integration is the interplay of multiple networks that allows for coordinated and flexible brain function. Della Rosa et al. (2021) proposed the functional homeostasis hypothesis, which states that optimal brain function is dependent on a balance of network segregation and integration. In this framework, the significant main effects of the CRBM and AL-REC perinatal functional predictors on global neurodevelopment scores over time highlight the importance of the functional balance between network specialization and flexibility (integration) in supporting cognitive, language, and socioemotional development. Significant interactions between CRBM, AL, and SSM-HOC perinatal functional predictors of neurodevelopment over time may instead reflect critical transitional periods in functional homeostasis dynamics at birth, between segregation and integration. Functional network transitioning states at birth could therefore be pivotal in shaping neurodevelopment of preterm birth infants that occur during sensitive periods of language acquisition, cognitive, and socioemotional development, demonstrating a complex interplay between more specialized and more flexible networks, and highlighting the requirement for a functional balance between these networks, already at birth, for optimal development along a gradient of increasing functional complexity and flexibility. To this regard, evidence from graph theory studies have highlighted the PTBs functional connectome development is characterized by alterations in these functional properties, with a tendency towards both higher functional networks segregation and lower integration (Bouyssi-Kobar et al., 2019; de Almeida et al., 2021; Gilchrist et al., 2022; Gozdas et al., 2018).
Conclusion
As a final consideration, we would like to highlight that the metrics derived from current clinical practice provided a limited contribution to outcomes prediction. Namely, the PPC-nr metric predicted SE outcomes at 24 months, while BS-nr predicted LANG outcomes at 24 months, with inclusion of functional markers further providing a significant increase to variability captured by the model (see regression analyses results in Table 6). Considering that subjects investigated in this study were PTBs without major pregnancy and neonatal complications and parenchymal alterations, we believe our results framework highlights how perinatal rs-fMRI allows to capture exclusive predictive neurodevelopmental variability features, especially in prematurely born newborns characterized by the absence of clear clinical indications pointing toward a higher risk for developing neurodevelopmental sequelae. This, combined with the remarkable degree of consistency between the identified functional features at birth and the predicted outcomes (as observed in previous studies), prompts us to encourage future investigations aimed at investigating perinatal rs-fMRI as a source of diagnostic and prognostic information for pediatric clinical practice of the near future.
Limitations and Future Directions
It has to be noted that, despite encouraging, our results are drawn from a limited sample of preterm newborns, hindering generalizability of our findings. In other words, they should be thus considered a starting point, rather than concluding evidence. To this end, we suggest upcoming investigations should focus on four major research focuses, to take a major step forward toward the evaluation of the clinical potential of perinatal rs-fMRI. First, longitudinal evaluations on larger cohorts will be needed in the future for further validation of these findings. Second, testing the predictive value of connectome metrics of functional integration and segregation on these cohorts will provide, in our view, further groundings for functional predictors identification and validation, as well as to increase predictions accuracy. Third, and to this same regard, functional metrics derived from EEG and fNIRS studies should also be investigated. Fourth, and crucially, investigation of postnatal variability related to parenting care, parent–infant interactions, and home environmental stimulation is a necessary step toward a full understanding of how perinatal metrics relate to neurodevelopmental outcomes measured throughout infancy.
Supplemental Material
sj-docx-1-jbd-10.1177_01650254241312136 – Supplemental material for Functional connectivity markers of prematurity at birth predict neurodevelopmental outcomes at 6, 12, 24, and 36 months
Supplemental material, sj-docx-1-jbd-10.1177_01650254241312136 for Functional connectivity markers of prematurity at birth predict neurodevelopmental outcomes at 6, 12, 24, and 36 months by Matteo Canini, Cristina Cara, Claudia Oprandi, Ana Katušić, Iris Žunić Išasegi, Antonio Messina, Alberto Andrea Zambon, Nicolo’ Pecco, Sarah Barni, Antonella Poloniato, Maria Grazia Natali Sora, Monica Falautano, Paola Scifo, Graziano Barera, Marco Tettamanti, Andrea Falini, Cristina Baldoli and Pasquale Anthony Della Rosa in International Journal of Behavioral Development
Footnotes
CRediT Authorship Contribution Statement
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by the Italian Ministry of Health’s “Ricerca Finalizzata 2016” (grant number RF-2016-02364081; Principal Investigator: Dr Pasquale Anthony Della Rosa).
Ethics Approval Statement
The study protocol was approved by the Ethics Committee of the San Raffaele Hospital (registration No. EK Nr. 2174/2016). The study was conducted according to the principles expressed in the Declaration of Helsinki.
Permission to Reproduce Material From Other Sources
This work does not contain material reproduced from other sources.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.