Abstract
To better understand the role of neuroendocrinological regulation in adolescent mental health, stress reactivity needs to be analyzed through both the autonomic nervous system (ANS) and the hypothalamic–pituitary–adrenal (HPA) axis. Accordingly, this study examined how adolescents’ internalizing and externalizing mental health symptoms are associated with their salivary alpha-amylase and cortisol levels, responses, and coordination (symmetry versus asymmetry). We utilized a developmentally salient stress task of mother–adolescent conflict discussion. Eighty 18–20-year-old late adolescents (55% girls) participated in a home laboratory assessment involving a 10-min conflict discussion with their mothers. Five adolescent saliva samples were collected to measure alpha-amylase and cortisol levels before, immediately after, and in 10-min intervals following the conflict discussion, to indicate stress reactivity. Adolescents had reported their internalizing (depression, anxiety, somatization) and externalizing (inattention, hyperactivity, anger control problems) symptoms 1 year earlier as part of a prospective family study. Internalizing symptoms were associated with adolescents’ high baseline cortisol levels, but not with cortisol responses or alpha-amylase levels or responses. In contrast, externalizing symptoms were associated with blunted alpha-amylase responses. Neither internalizing nor externalizing symptoms were associated with asymmetry between alpha-amylase and cortisol reactivity. The mother–adolescent conflict discussion was relevant as a stress stimulus to induce neuroendocrinological stress responses in adolescents. The nature of mental health problems was important for stress reactivity, yet, we found no evidence about mental health problems being related to endocrinological asymmetry in adolescents.
Introduction
Adolescence is an important transitional period, characterized by intensive social, emotional, and hormonal changes (Crone & Dahl, 2012). Optimal development involves fluent coordination between these domains, aiding adolescent adjustment to increasing social demands and stressors. The autonomic nervous system (ANS) consisting of the sympathetic (SNS) and parasympathetic (PNS) nervous systems and the hypothalamic–pituitary–adrenal (HPA) axis form the core bodily stress response system that establishes adaptive functioning and underlies mental health (Del Giudice et al., 2011).
Production of both SNS-regulated alpha-amylase and HPA-regulated cortisol undergo considerable changes in adolescence (Susman et al., 2010). As this period is also characterized by heightened risk for mental health problems (Hazen et al., 2010), it is vital to understand the associations between mental health and alpha-amylase and cortisol. While the basal levels and responses to stress are agreed to be important for neuroendocrinological functioning, less research has focused on the role of endocrinological symmetry between alpha-amylase and cortisol (Jones et al., 2020). This study analyzes how internalizing and externalizing symptoms in late adolescence are associated with dynamics between alpha-amylase and cortisol stress reactivity in a normative adolescent sample. Finally, previous research has mainly focused on either performance-related or social evaluative stress (Figueiredo et al., 2020; Wadsworth et al., 2019), whereas we focus on the more developmentally salient stress evoked by adolescent–mother conflict discussion.
Dual Stress Response System
As the functioning of ANS and HPA are closely integrated and cross-regulated, they can be considered dual components of the human stress response system (Ellis et al., 2006). The Adaptive Calibration Model (ACM) by Del Giudice and colleagues (2011) informs about the integrated functioning of alpha-amylase and cortisol responses to stress and threat. Based on the evolutionary developmental framework (Boyce & Ellis, 2005), the ACM postulates that multiple components of the stress response system, differing in timing and sensitivity to the type of stressor, work together to serve human growth, maturation, mental health, and social bonding in diversely demanding environments (Del Giudice et al., 2011).
In line with that, the SNS activates an acute, “fight or flight” response by increasing heart rate and respiration and by releasing the catecholamines epinephrine and norepinephrine (Nater & Rohleder, 2009). Salivary alpha-amylase serves as a useful biomarker of the rapid SNS reactivity (Granger et al., 2007). It concentrates immediately, peaks at approximately 5–10 min and is attuned by 15 min after the stress trigger (Nater et al., 2013). The HPA axis, in turn, represents a delayed, long-term response to stressors that cannot be attuned by the ANS alone. Cortisol elevation starts at around 5 min, peaks between 10 and 30 min and returns to baseline usually within 1 hr after the stress trigger (Allwood et al., 2011; Del Giudice et al., 2011).
Considering the nature of stress, alpha-amylase has been suggested to be especially reactive to novelties, opportunities, and challenge (Schumacher et al., 2013; Van Stegeren et al., 2008), whereas unpredictable and uncontrollable events and threats to interpersonal security are known to trigger cortisol reactivity (Strahler et al., 2017). The potential stress-specific activation of alpha-amylase and cortisol may serve adaption by allowing a better goodness-of-fit in responding to demands that involve stressors of different nature (Van Stegeren et al., 2008).
Mental Health Problems and Stress Response System
Dysfunctional ways of regulating stress typically involve either neuroendocrinological hyper- or hypoactivation, evident in excessive or insufficient basal levels and increased or decreased reactivity to stress (Del Giudice et al., 2011; Guerry & Hastings, 2011). There is some evidence that these follow the well-established symptom dimensions (Kotov et al., 2017), in that hyperactivation associates with internalizing and hypoactivation with externalizing symptoms (Segal, 2016). It is noteworthy that there is some evidence also for the narrow symptom-specific dysregulation patterns, particularly regarding depression and anxiety (Fiksdal et al., 2019; Steudte-Schmiedgen et al., 2017; Yoon & Joormann, 2012). Yet, systematic reviews have also pointed out inconsistencies in the findings and criticized small samples and effect sizes (Figueiredo et al., 2020; Jones et al., 2020). Considering the high comorbidity between the narrow symptom categories, we focus on the broader dimensions of internalizing and externalizing.
Concerning internalizing symptoms (e.g., depression and anxiety), systematic reviews generally suggest an association with increased cortisol levels and responses (Hartman et al., 2013; Wadsworth et al., 2019). The meta-analysis by Lopez-Duran and colleagues (2009) evidenced both higher cortisol basal levels and stronger responses to stress in depressed as compared to non-depressed adolescents. Similarly, adolescents with generalized or social anxiety showed increased cortisol levels and responses (Funke et al., 2017; Segal, 2016). There is, however, also some evidence of depressive adolescents and adults showing low cortisol levels and responses (Burke et al., 2005; Harkness et al., 2011), particularly in chronic conditions (Booij et al., 2013).
Internalizing problems have also been found to associate with dysregulated SNS (Segal, 2016), which is likely to represent the intensive distress and vigilance to threat inherent in the symptoms. Along these lines, studies confirm that severe internalizing symptoms (Wadsworth et al., 2019) and anxiety disorders (Schumacher et al., 2013) in children and adolescents are associated with high basal level and increased responses of alpha-amylase.
Concerning externalizing symptoms (e.g., aggression and conduct disorders), systematic reviews suggest a general association with low cortisol basal levels and decreased responses to stress (Figueiredo et al., 2020; Guerry & Hastings, 2011). For instance, adolescents with externalizing (Hartman et al., 2013) and attention deficit hyperactivity disorder (ADHD) symptoms (Van West et al., 2009) showed lower cortisol levels and responses to stress than controls. Other studies found that aggression and conduct disorders were associated with adolescents’ lower cortisol levels, but not with altered responses (Alink et al., 2008).
Research further illustrates that externalizing symptoms are associated with hypoactivation of both HPA and SNS. For example, adolescents with externalizing problems showed decreased cortisol responses and low basal alpha-amylase levels (Bae et al., 2015), and those with severe aggression showed both decreased cortisol and alpha-amylase reactivity, indicated by levels and responses (Gordis et al., 2006). Wadsworth and colleagues (2019) further elaborated that externalizing symptoms were particularly evident among adolescents with both low cortisol and low alpha-amylase levels during the recovery from stress. However, there are also studies showing similar dysregulated stress reactivity, regardless of the nature (internalizing vs externalizing) of mental health problems (Lorber, 2004; Vigil et al., 2010).
Cortisol and Alpha-Amylase Asymmetry
The high interdependence between ANS and HPA makes it reasonable to anticipate some symmetry between the systems, meaning that marked activation in one is expected to be accompanied by activity in the other. Instead, a failure in coordination, that is, asymmetry between these systems, can indicate dysregulation relevant for mental health problems (Bauer et al., 2002; Schumacher et al., 2013). Such asymmetry is indicated by a disproportionate ratio between alpha-amylase and cortisol levels and/or responses. Asymmetry is thus best apparent in interactive effects, for example, by symptoms associating with heightened cortisol reactivity only when alpha-amylase reactivity is blunted, or vice versa (Allwood et al., 2011; Bauer et al., 2002).
Reviews provide some evidence that internalizing (but not externalizing) symptoms are associated with asymmetry between cortisol and alpha-amylase levels and responses (Jones et al., 2020; Wadsworth et al., 2019). As examples of asymmetric co-activation, clinically significant internalizing problems have been found to associate with the combination of heightened alpha-amylase and blunted cortisol reactivity in children (Bae et al., 2015). However, other studies have reported internalizing symptoms to be associated with the opposite asymmetric pattern, that is, blunted alpha-amylase and heightened cortisol reactivity (Ali & Pruessner, 2012; Allwood et al., 2011). Further research is thus needed to clarify the role of asymmetry between cortisol and alpha-amylase reactivity in adolescent mental health.
Interpersonal Stress
While adolescents strive toward autonomy, their relationship with parents, especially the mother, remains an important socioemotional context for their development and wellbeing (Rosenthal & Kobak, 2010). Hence, it is not surprising that the quality of the parent–adolescent relationship is important for adolescent mental health (Zhang et al., 2021). Researchers agree that the most stressful stimuli in childhood reflect threat to the primary source of attachment security, and in adolescence conflicts with parents can signify fear and threat of losing support and acceptance of the attachment figure (Gunnar et al., 2009).
However, only a few studies regarding adolescent mental health and neuroendocrinological reactivity have used adolescent–parent conflict discussion as a stress stimulus. Both internalizing and externalizing symptoms (Klimes-Dougan et al., 2001) as well as social withdrawal and social anxiety (Granger et al., 1994) were found to be associated with adolescent heightened cortisol responses to conflict discussion. Despite the importance of interpersonal conflict with parents as a cause of adolescent stress, previous studies have predominantly measured stress reactivity related to performance and social evaluative stress (such as public speaking in Trier’s Social Stress Task; Gunnar et al., 2009) and peer exclusion (Stroud et al., 2009). This study is one of the first to examine adolescent neuroendocrinological stress reactivity including both alpha-amylase and cortisol in the context of adolescent–mother conflict discussion.
Aims of the Study
This study aims to describe associations between adolescent mental health problems and the levels, responses, and coordination between salivary alpha-amylase and cortisol in relation to the interpersonal stress of an adolescent–mother conflict discussion. First, we separately examine the associations between internalizing (depression, anxiety, somatization) and externalizing (inattention, hyperactivity, anger control problems) symptoms and the basal levels and responses of alpha-amylase and cortisol. Based on previous research (Figueiredo et al., 2020; Hartman et al., 2013), we hypothesize that adolescent internalizing symptoms are associated with heightened and externalizing symptoms with blunted levels and responses of alpha-amylase and cortisol across the stress task. Second, we examine how adolescent internalizing and externalizing symptoms are associated with the mutual coordination, that is, symmetry versus asymmetry, between alpha-amylase and cortisol reactivity. Based on previous research (Jones et al., 2020; Wadsworth et al., 2019), we assume internalizing symptoms to be associated with asymmetric responses, indicated either by blunted alpha-amylase and heightened cortisol, or by heightened alpha-amylase and blunted cortisol.
Methods
Participants
A subsample of 80 adolescents at the age of 18–20 years (M = 19.27, SD = 0.48) were recruited during late 2018 to early 2020 from a prospective Finnish family study (Miracles of Development) to take part in a home laboratory assessment. The original sample, recruited in pregnancy, consisted of couples with infertility history and successful assisted reproductive treatment (ART; n = 484) and naturally conceiving couples (NC; n = 469). They further participated in questionnaire study waves during the child’s infancy, middle childhood, and late adolescence. The late adolescence questionnaire study involved 449 adolescents, 553 mothers, and 340 fathers, and took place about a year before the home laboratory assessment (late 2017 to early 2019), when the adolescents were 17−19 years old (M = 18.23, SD = 0.34). A more detailed description of the original sample is presented elsewhere (Tammilehto et al., 2021).
A disproportionate stratified sampling procedure was used with the aim to collect a subsample of 100 late adolescents for the analysis of neuroendocrinological stress reactivity. However, due to the COVID-19 pandemic, sampling was interrupted in March 2020, and only 80 adolescents were examined. The sampling started in November 2018. Stratification was based on a risk index, focusing on multiple family risks during the pregnancy and infancy. The risk index was a sum of 21 variables that cover mothers’ and fathers’ mental health, interparental relationship, and parenting stress. Stratification was balanced using child sex and ART status. Thus, the subsample was representative of adolescents with high variability in the early life family environment (i.e., ranging from very low to very high risk). For details about the procedure, see the study by Ilomäki and colleagues (2022).
The project complies with the Code of Ethics of the World Medical Association (WMA) Declaration of Helsinki 1964–2014. The Ethical Board of Helsinki University Central Hospital approved the methods and data collection of the questionnaire study (HUS/1566/2017) and the home laboratory study (HUS/1477/2018). During both study phases, participants gave their written informed consent. In case the family indicated a need for psychological help, a psychotherapist in the research group was available for consultation. Furthermore, adolescents exhibiting clinically significant non-normality in neuroendocrinological stress levels or responses were contacted and referred to further examination.
Study Procedure
Late adolescence questionnaire data were used in this study for the part of adolescent self-reported mental and somatic health and demographic factors as well as mother-reported socio-economic factors. For the home laboratory assessment, a researcher (who was also a psychologist) met the adolescent–mother dyads either at their home or at a family clinic, according to their preferences. All visits took place in the late afternoon during the same hours and lasted for about 2 hr.
Before the visit, the researcher sent instructions regarding the saliva sampling to the participating adolescents and mothers by e-mail or SMS. Adolescents were asked not to eat or drink anything, nor smoke for an hour before the session, and to refrain from alcohol use and heavy exercise from the previous night onwards. These requirements were aimed to prevent acute substance effects on HPA axis and ANS functioning (Strahler et al., 2017).
Figure 1 presents the phases and time points of cortisol and alpha-amylase assessments. To sum it up, after arrival, the researcher introduced the tasks at hand and asked participants to fill in questionnaires about factors that may affect cortisol and alpha-amylase functioning. The participants were then asked to rinse their mouths (10–15 min before the first saliva sampling). The actual procedure involved: (a) a relaxation period when participants were asked to sit or lie down comfortably and listen to a 10-min relaxation tape that consisted of a progressive muscle relaxation and guided imagery; (b) the first (pre-conflict task) saliva sample collection; (c) defining the conflict discussion topic, that is, the adolescent recorded her or his topic together with the researcher; (d) the adolescent and the mother listened to the recording without the researcher’s presence and then discussed about the conflict for 10 min, they were instructed to use the whole time and the discussion was video recorded; (e) the second (post-conflict task) saliva sample collection immediately after the conflict discussion; (f) evaluation of subjective stress during and satisfaction with the conflict discussion; (g) the third (early recovery phase) saliva sample collection at 10 min after the end of conflict discussion; (h) the fourth (mid-recovery phase) saliva sample collection after a free resting period and exactly 20 min after the end of conflict discussion; and (i) the fifth and final (late recovery phase) saliva sample was collected at 30 min after the end of conflict discussion. Importantly, from early recovery to late recovery phases, the participants were instructed to engage in calm activity, reading, or listening to music.
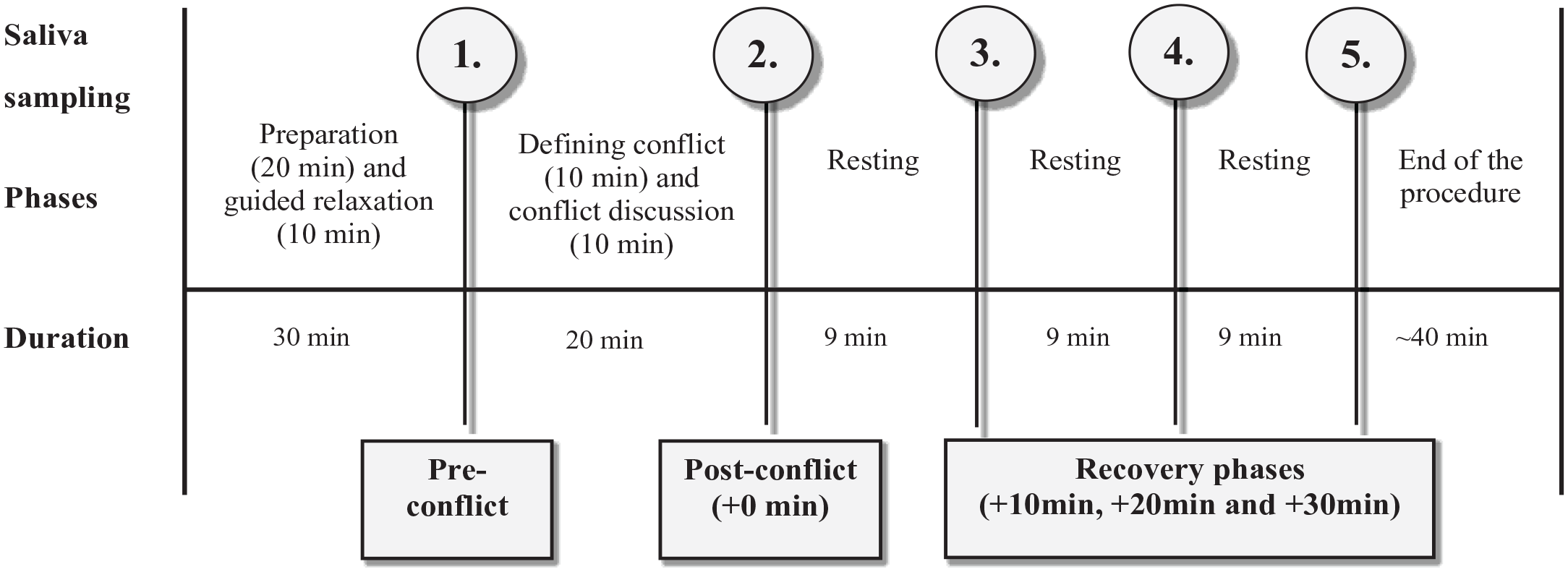
Phases and Time Points of Saliva Assessments.
Measures
Mental Health Problems
Adolescent mental health problems were measured using the Self-Report of Personality─Adolescent (SRP─A) scales of the Behavior Assessment System for Children, Third Edition (BASC-3; Reynolds & Kamphaus, 2015). Six symptom scales were utilized: depression (12 items; e.g., “I just don’t care anymore”), anxiety (13 items; e.g., “I worry but I don’t know why”), somatization (7 items; e.g., “I have trouble breathing”), attention problems (8 items; e.g., “I am easily distracted”), hyperactivity (8 items; e.g., “I have trouble sitting still”), and anger control problems (10 items; e.g., “When I get angry, I want to hurt someone”). The adolescents used dichotomous (1 = True or 0 = False) and 4-point Likert-type scale (from 0 = Never to 3 = Almost always) to answer whether or how often they had had the problem during the last month. Sum scores were constructed for internalizing (depression, anxiety, and somatization) and externalizing (attention problems, hyperactivity, and anger control problems) symptoms, with higher scores indicating more severe symptoms. The reliabilities were α = .83 for internalizing and .71 for externalizing symptoms.
Demographic and Health Information
Adolescents’ sex was determined from the original data collected right after birth. Adolescents self-reported on their age, education, and living arrangements. They further estimated their general somatic health, chronic illnesses, and psychiatric/neuropsychiatric disorders. Mothers reported about parental divorce and family’s level of monthly income. Right after the conflict discussion, adolescents self-evaluated the amount of subjective stress during the discussion (from 1 = not at all to 5 = extremely stressed).
Factors Potentially Affecting Saliva Analyses
Factors that might affect saliva analyses were measured by 14 items, developed particularly for this study and based on literature (Strahler et al., 2017). They covered chronic and transient illnesses, dentistry, medications, contraceptives, nicotine, alcohol, and caffeine use, heavy exercise, sleep quality, and major losses and stress during the past 6 months, as well as stressful events during the day of the saliva collection. Adolescent medication use was asked about generally, and also specifically concerning thyroxin and corticosteroids.
Saliva Samples and Adolescent−Mother Conflict Discussion
ANS functioning was measured by alpha-amylase level in the first four collected saliva samples (pre-conflict task, post-conflict task, and recovery phases at 10 and 20 min), indicating rapid neuroendocrinological responses to the acute stressor. HPA axis functioning was measured by cortisol level in the five saliva samples (pre-conflict task, post-conflict task, and recovery phases at 10, 20, and 30 min), indicating slower neuroendocrinological responses.
Salivary samples were stored at −5°C in the laboratory of the Finnish Institute of Occupational Health. Alpha-amylase and cortisol were analyzed using a commercial expanded range high sensitivity Enzyme Immunoassays, EIA kit (Salimetrics; State College, PA). Inter- and intra-assay critical values were less than 5% and 10%, respectively.
Statistical Analyses
To answer the first research question about the associations between mental health symptoms and alpha-amylase and cortisol levels and responses, we used longitudinal linear mixed modeling (LMM) with restricted maximum likelihood estimation. In these models with alpha-amylase and cortisol values at each time point as outcomes, we included (a) the main effects of internalizing and externalizing symptoms on basal levels, (b) the linear and quadratic effects of time elapsed (in minutes since the pre-conflict task assessment), and (c) the interaction effects between internalizing and externalizing symptoms and linear and quadratic time. Random intercepts and random slopes for the effect of time on alpha-amylase and cortisol levels were included when they significantly improved model fit according to the log-likelihood test. Autoregressive covariance structures were included if models converged and they improved fit according to the log-likelihood test. Otherwise, variance component structures were used.
Our second research question was about mental health problems associating with asymmetry between alpha-amylase and cortisol. Similar to the study by Martinez-Torteya et al. (2017), we used areas under the curve (AUC) to analyze alpha-amylase and cortisol responses. However, in contrast to the study by Martinez-Torteya et al. (2017), who were interested in total response, we examined the change (increase or decrease) from pre-conflict task into the post-conflict task and recovery phases for both alpha-amylase and cortisol by using the AUC with respect to increase (AUCI; Pruessner et al., 2003). We calculated the AUCI using the summation of trapezoids method presented by Pruessner et al. (2003). Importantly, we tested the effects of asymmetry utilizing an interaction term of alpha-amylase AUCI and cortisol AUCI, computed using mean centered values. These interaction terms (as well as the corresponding main effect terms) were included in regression analysis as independent variables, and adolescent’s internalizing and externalizing symptoms as dependent variables. We used Spearman correlation analyses for examining associations between study variables, and analysis of variance (ANOVA) for associations between categorical demographics, factors potentially affecting the saliva analyses, and alpha-amylase and cortisol reactivity.
Data were missing for some mental health variables for 9 out of 80 participants and for the included confounding factors for 5 out of 80 participants. Data were excluded in a pairwise manner. Due to missing data, 67 participants contributed data to longitudinal LMM and regression models. Those excluded did not differ from those included in the LMM in terms of sex distribution, nor in basal levels or AUCI of cortisol and alpha-amylase. We carried out analyses of the two research questions by using R 4.0.5 (R Core Team, 2021) and the nlme 3.1-152 R package (Pinheiro et al., 2021) for LMM.
Covariates and Outliers
Adolescent sex was included as a covariate in all analyses as the literature suggests neuroendocrinological sex differences (Kudielka & Kirschbaum, 2005). Furthermore, contraception was used as a covariate in all analyses, as contraceptive medication containing estrogen is known to affect stress hormone levels and responses (Gervasio et al., 2022). In LMM, the effects of the covariates were estimated for both the basal level and the linear and quadratic shapes of the responses.
The data were screened for physiologically impossible or totally implausible values of alpha-amylase or cortisol, but none were identified. All cases with very high values (> 3 SD above mean) in (a) baseline values of alpha-amylase or cortisol, (b) alpha-amylase or cortisol responses (AUCI), or (c) the interaction of the AUCI of alpha-amylase and cortisol (a total of 6 out of 80 cases) were individually examined. One participant reported using glucocorticoid medication. As a sensitivity analysis, we ran all our statistical analyses excluding this participant, but this did not affect the results. We report the results with this participant included.
To counter effects of extreme outliers, we winsorized alpha-amylase and cortisol values that were more than three standard deviations above the mean. Five values of four participants for alpha-amylase and 12 values of four participants for cortisol were winsorized due to high deviation from the mean. To account for non-normality, we further square-root transformed alpha-amylase values and log-transformed cortisol values, in line with conventional practices in the field (Martinez-Torteya et al., 2017).
Results
Descriptive Statistics
Table A1 (in Supplementary Appendix) presents demographic characteristics of the subsample. Approximately half (55%) of the adolescents were girls, and almost a half (49%) were conceived with ART. Three quarters (76%) lived with the nuclear family. About 17% had divorced parents. A majority (81%) studied in high school and about a tenth in vocational school. Almost half of the families had monthly income of more than 7,500 euros, close to average for Finnish families (Official Statistics of Finland, 2022). A majority (82%) of adolescents estimated their health as good or pretty good, and about a tenth reported a diagnosed psychiatric or neuropsychiatric disorder (e.g., depression, eating disorder, or ADHD).
Table A2 (in Supplementary Appendix) summarizes the presence of factors potentially affecting the saliva analyses. About a third of the adolescents had had some transient illness in the past 6 months and 17% had chronic illness. More than a half had used painkillers during the two previous weeks and 18% were on regular medication. Two participants used an asthma spray and one used thyroxine medication, but none used cortisone cream or pills. Nearly half of the girls indicated use of hormonal contraception. Regular smoking was rare (5%), but caffeine intake (80%) was common. Two-thirds (67%) reported moderate (i.e., one or two portions per week) alcohol use. About 13% of the adolescents reported major losses and stress during the past 6 months, and 13% reported stressful events during the saliva assessment day. The mean level of subjective stress during the conflict discussion was 1.97 (SD = 1.00), and did not differ significantly between boys (M = 1.82) and girls (M = 2.10), t(71.62) = 1.27, p = .208.
To consider the need to include covariates in the main analyses in addition to sex and hormonal contraception, we examined whether demographics and factors potentially affecting the saliva analyses would be associated with the main study variables (i.e., mental health, alpha-amylase, and cortisol). ANOVAs showed that smoking was significantly associated with heightened mental health problems, for both internalizing, F(1,70) = 8.09, p = .006, η2 = .11, and externalizing symptoms, F(1,70) = 14.82, p = .0001, η2 = .18. As only a few (n = 5; 6%) adolescents smoked regularly and smoking was associated only with mental health (not with alpha-amylase or cortisol), we decided not to include it as an additional covariate.
Table A3 (in Supplementary Appendix) shows the mean levels of alpha-amylase and cortisol at different assessment time points, separately for boys and girls. These mean levels are also visualized in Figures A1 and A2 (in Supplementary Appendix).
Table 1 shows the correlations between the study variables, including maximum elevation (i.e., peak minus the pre-conflict task) values of alpha-amylase and cortisol. Female sex correlated with lower levels of maximum cortisol elevation (r = –.39) and lower AUCI of cortisol (r = –.38), and hormonal contraception correlated with lower maximum cortisol elevation (r = –.23). Externalizing symptoms (r = –.26) correlated with lower levels of maximum alpha-amylase elevation.
Correlations between Levels of Alpha-Amylase and Cortisol, Mental Health Variables, and Demographic Variables among Adolescents During the Family Conflict Discussion.
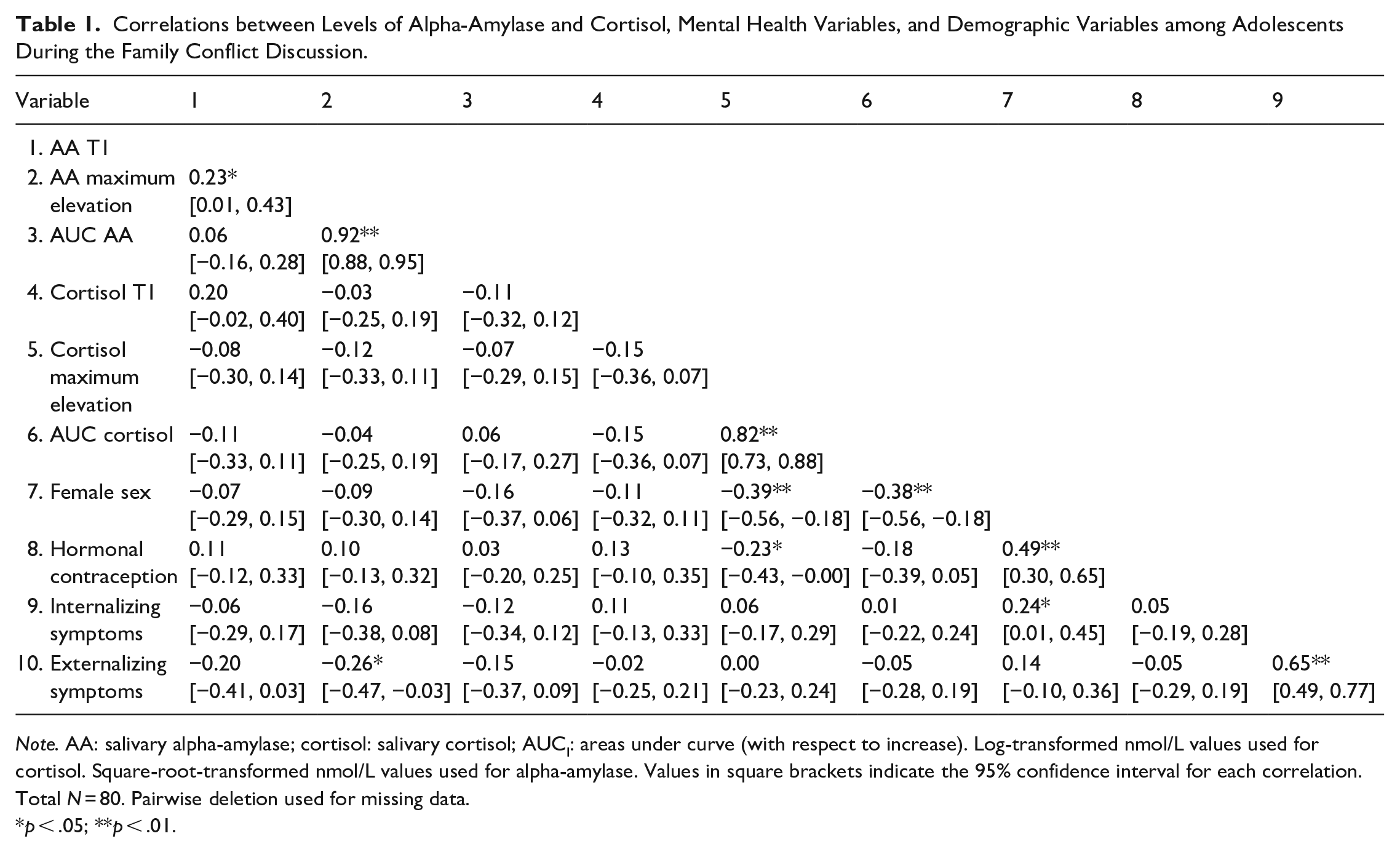
Note. AA: salivary alpha-amylase; cortisol: salivary cortisol; AUCI: areas under curve (with respect to increase). Log-transformed nmol/L values used for cortisol. Square-root-transformed nmol/L values used for alpha-amylase. Values in square brackets indicate the 95% confidence interval for each correlation. Total N = 80. Pairwise deletion used for missing data.
p < .05; **p < .01.
Mental Health and Alpha-Amylase Levels and Responses
Table 2 presents results on adolescents’ mental health problems associating with basal alpha-amylase levels (main effect) and responses to the conflict discussion (Time1 for linear and Time2 for quadratic shapes). Allowing for random intercepts and slopes improved model fit. Autoregressive covariance structure could not be tested due to model non-convergence. As hypothesized, externalizing symptoms were associated with blunted alpha-amylase responses, as indicated by significant interaction terms (i.e., Time1 × Externalizing for linear and Time2 × Internalizing for quadratic effects). Against our hypothesis, however, externalizing symptoms did not significantly associate with low basal levels of alpha-amylase (non-significant externalizing main effect), and internalizing symptoms did not associate with either high basal levels or heightened responses of alpha-amylase. Concerning covariates, girls showed more blunted alpha-amylase responses than boys (significant Time1 × Sex and Time2 × Sex effects).
Fixed Effects of Internalizing and Externalizing Mental Health Symptom on Levels of Salivary Alpha-Amylase at Different Time Points for Adolescents Experiencing Family Conflict Discussion.
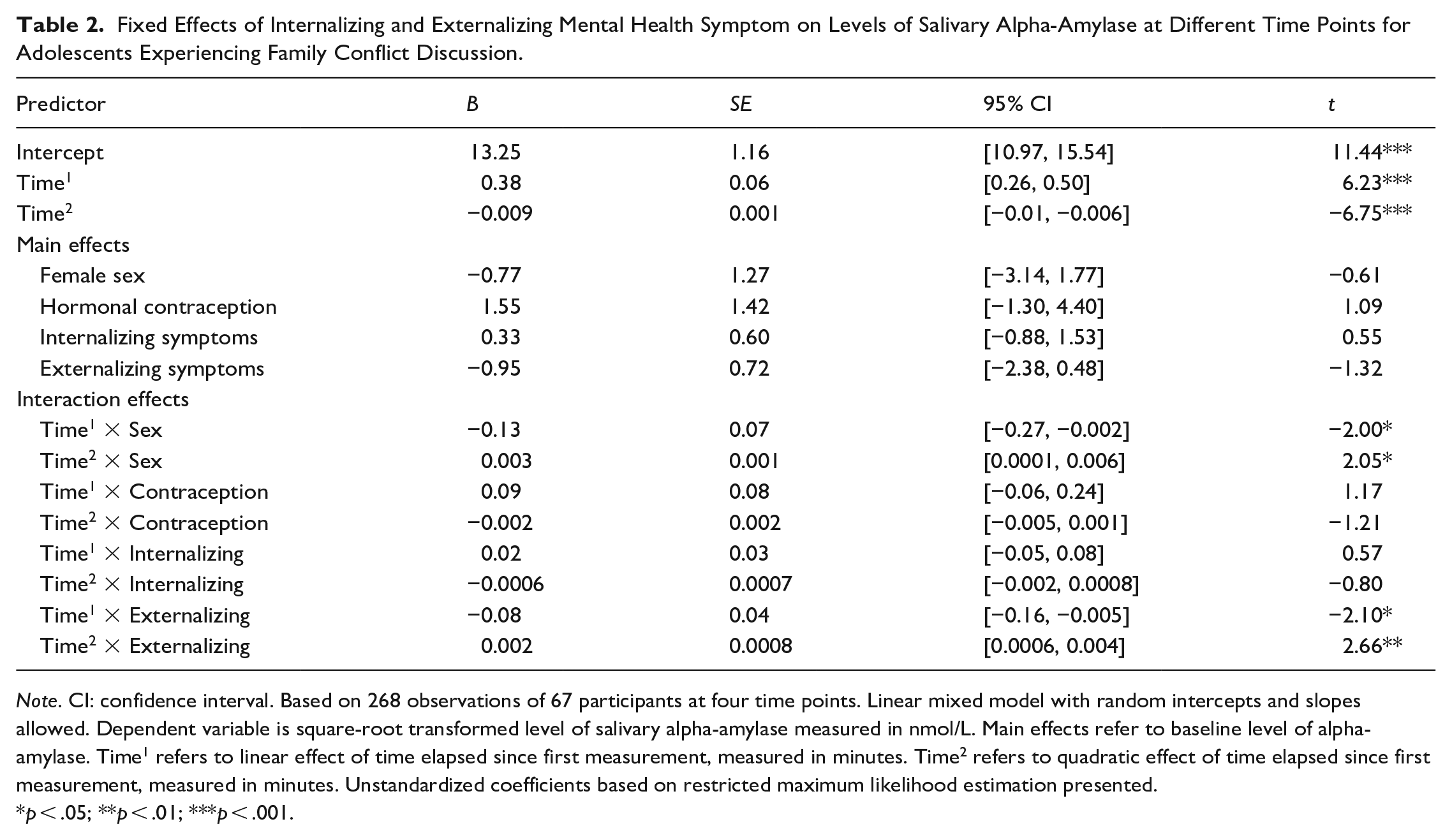
Note. CI: confidence interval. Based on 268 observations of 67 participants at four time points. Linear mixed model with random intercepts and slopes allowed. Dependent variable is square-root transformed level of salivary alpha-amylase measured in nmol/L. Main effects refer to baseline level of alpha-amylase. Time1 refers to linear effect of time elapsed since first measurement, measured in minutes. Time2 refers to quadratic effect of time elapsed since first measurement, measured in minutes. Unstandardized coefficients based on restricted maximum likelihood estimation presented.
p < .05; **p < .01; ***p < .001.
Mental Health and Cortisol Levels and Responses
Table 3 presents results on adolescents’ mental health problems associating with basal cortisol levels (main effects) and responses to the conflict discussion (Time1 for linear and Time2 for quadratic shapes). Allowing for random intercepts and slopes improved model fit. Autoregressive covariance structure did not improve model fit, and was not included. As hypothesized, internalizing symptoms were associated with high basal cortisol levels, indicated by significant main effect of these symptoms. Yet, against our hypothesis, internalizing symptoms were not associated with heightened cortisol responses to the conflict discussion, as indicated by non-significant interaction effects with time (i.e., Time1 × Internalizing for linear and Time2 × Internalizing for quadratic effects). Furthermore, against our hypothesis, externalizing symptoms did not significantly associate with either lower basal levels or blunted responses of cortisol. Concerning covariates, results show that sex predicted basal levels (main effect) and cortisol responses in terms of both the linear (Time1 × Sex) and quadratic (Time2 × Sex) effect of time. Controlling for other covariates, girls showed lower basal levels of cortisol and more blunted cortisol responses than boys.
Fixed Effects of Internalizing and Externalizing Mental Health Symptoms Predicting Levels of Cortisol at Different Time Points for Adolescents Experiencing a Family Conflict Discussion.
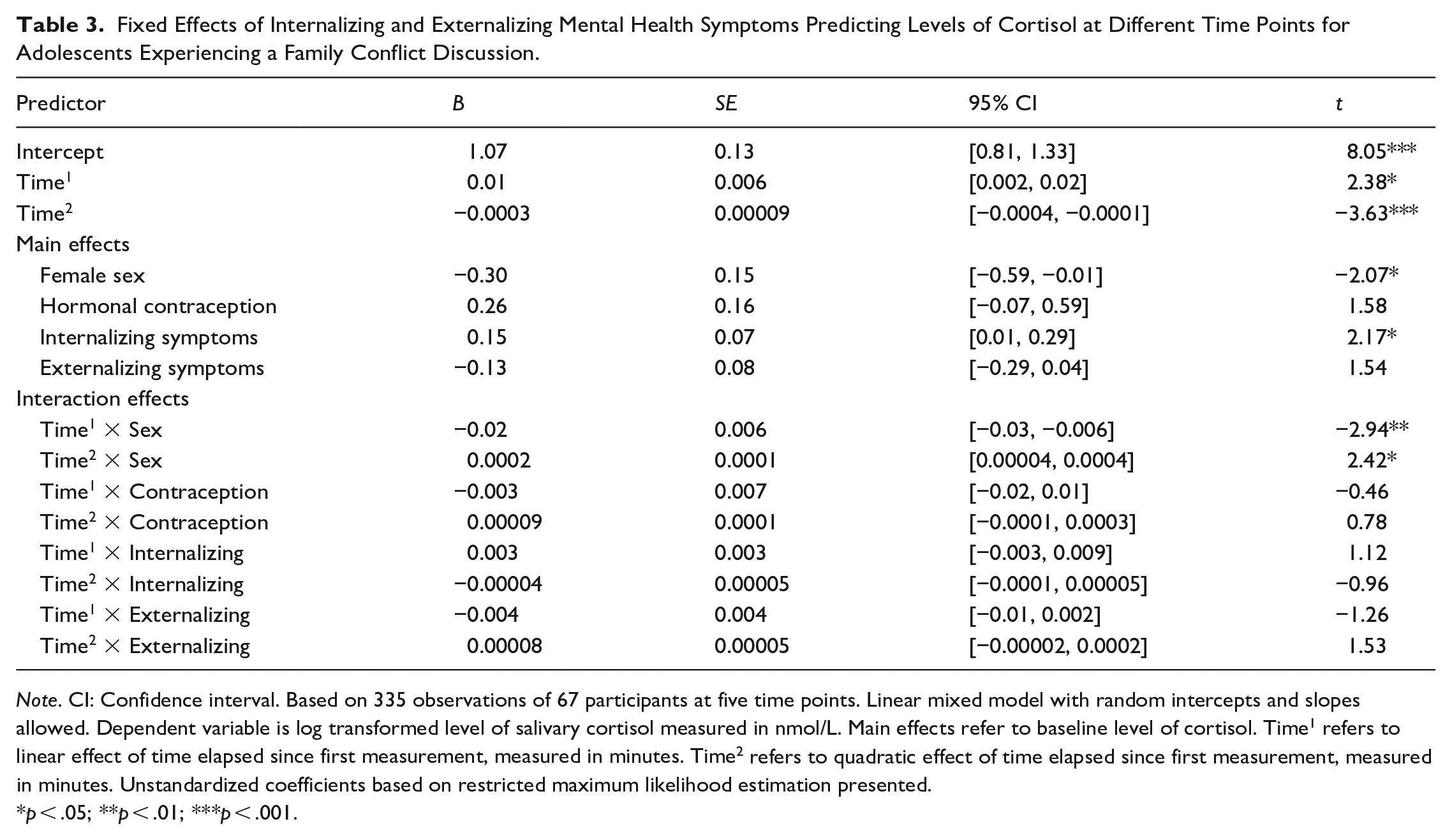
Note. CI: Confidence interval. Based on 335 observations of 67 participants at five time points. Linear mixed model with random intercepts and slopes allowed. Dependent variable is log transformed level of salivary cortisol measured in nmol/L. Main effects refer to baseline level of cortisol. Time1 refers to linear effect of time elapsed since first measurement, measured in minutes. Time2 refers to quadratic effect of time elapsed since first measurement, measured in minutes. Unstandardized coefficients based on restricted maximum likelihood estimation presented.
p < .05; **p < .01; ***p < .001.
Asymmetry between Alpha-Amylase and Cortisol Reactivity
Against our hypotheses, the results showed no associations between adolescent mental health and the asymmetry of alpha-amylase and cortisol reactivity. More precisely, in regression analyses, neither internalizing nor externalizing symptoms were associated with the total amount of alpha-amylase and cortisol responses (i.e., AUCI) nor their interaction (i.e., AUCI cortisol × AUCI alpha-amylase), all p > .05. Thus, the results did not provide support our hypothesis that high levels of internalizing symptoms would be associated with alpha-amylase−cortisol asymmetry. See Table A4 (in Supplementary Appendix) for details.
Discussion
Interpersonal conflicts are stressful in general and also for adolescents who seek autonomy from their parents and build novel bonds outside the home. We analyzed how mental health problems in late adolescence were associated with the levels, responses, and coordination (symmetry vs asymmetry) between ANS-regulated alpha-amylase and HPA-regulated cortisol in a conflict discussion with the mother. Based on previous research, we hypothesized internalizing symptoms to associate with neuroendocrinological hyperactivation and externalizing symptoms with hypoactivation (Figueiredo et al., 2020; Hartman et al., 2013). In line with our hypotheses, the nature of symptoms turned out to be decisive: internalizing symptoms associated with heightened baseline cortisol levels, whereas externalizing symptoms associated with blunted alpha-amylase responses to the adolescent–mother conflict discussion. Against our hypothesis, however, adolescent internalizing symptoms did not to associate with the asymmetry between alpha-amylase and cortisol reactivity.
The main function of alpha-amylase and cortisol production is stress adaptation and homeostatic maintenance of optimal neuroendocrinological activity (Del Giudice et al., 2011). People suffering from mental health problems typically show either excessive or insufficient basal levels and altered responses to stress, reflecting regulatory dysfunction (Guerry & Hastings, 2011; Jones et al., 2020). Our findings concur with studies of depressive or anxious adolescents showing neuroendocrine hyperactivity, especially heightened cortisol levels (Hartman et al., 2013; Lopez-Duran et al., 2009), and aggressive or antisocial adolescents showing hypoactivity, indicated by blunted reactivity (Figueiredo et al., 2020). However, the associations between mental health and alpha-amylase and cortisol in our study were not as comprehensive as expected. Internalizing symptoms were associated only with high basal cortisol levels, but not with cortisol responses or alpha-amylase levels or responses during the conflict discussion. Externalizing symptoms in turn were associated only with blunted alpha-amylase responses, but neither with low basal alpha-amylase levels nor with cortisol levels or responses.
Dynamic biological, psychological, and behavioral processes may explain our findings about the specific associations between internalizing and externalizing symptoms and neuroendocrinological levels and responses to interpersonal stress. Internalizing symptoms are characterized by maladaptive regulation of negative emotions, which can be exemplified in rumination (Olatunji et al., 2013). Although the focusing on one’s negative emotions and thoughts reflects a mental attempt to solve problems, the judgmental and repetitive nature of rumination tends to maintain dysphoric mood and distress (Michl et al., 2013). High cortisol basal levels and increased responses to stress reflect dysfunctional maintenance of physiological homeostasis (Allwood et al., 2011), which was found to be associated with rumination (Zoccola & Dickerson, 2012). Other emotion regulation and personality factors may similarly play important roles in explaining the association. For example, neuroticism and introversion as personality traits have been found to be linked with less-than-optimal cortisol regulation patterns (Hauner et al., 2008).
Considering links between behavior and biological processes can help explain why adolescents’ externalizing symptoms were associated with blunted alpha-amylase responses. According to the sensation seeking model (Zuckerman et al., 1980), low neuroendocrinological activity creates an unpleasant physiological state that evokes a need for stronger stimulation, which in turn is characteristic to externalizing behaviors, such as conduct disorders and aggression. This “unresponsivity” characteristic of externalizing problems also concur with findings that low neuroendocrinological activity is associated with fearlessness in the face of danger and insensitivity to other people’s emotions (Blair, 2001). This may, in turn, hinder anticipation and learning of consequences that typically inhibit aggression and deceit in interpersonal relations (Figueiredo et al., 2020). Yet, previous research has observed these processes in relation to more extreme forms of antisociality (Wright et al., 2019). Our findings are important as they reveal a similar link between externalizing symptoms and blunted alpha-amylase responses in a normative late adolescent group.
Coordination between alpha-amylase and cortisol reactivity has been suggested as important for mental health, with asymmetry indicating dysregulation and thus mental health problems (Bauer et al., 2002; Wadsworth et al., 2019). Surprisingly, however, our results provided no indication about the role of asymmetry in adolescents’ mental health. Hence, we found no support for the hypothesis that internalizing symptoms would associate with the pattern of low alpha-amylase and high cortisol levels and responses or vice versa. Reviews emphasize that results concerning coordination between alpha-amylase and cortisol are still mixed and based on small samples or specific groups, such as male participants only (Jones et al., 2020). It is possible that the links between mental health and asymmetry are more complex than currently understood.
It is noteworthy that in modeling asymmetry, we relied on a method introduced by Martinez-Torteya et al. (2017), where coordination is reflected as the interaction of alpha-amylase and cortisol AUC. This method emphasizes asymmetry in the overall neuroendocrinological responses. Alternatively, for example, the interaction could have been analyzed in separate sequences or focusing on individual points of measurement. Yet, as the rates of reaction, peak, and recovery differ both between alpha-amylase and cortisol and between individuals, such approaches would not have been straightforward to accomplish. However, caution is needed before making definitive conclusions about the role of asymmetry between ANS and HPA. For example, it is possible that asymmetry between ANS and HPA may not always indicate regulation problems but can rather reflect a sequential nature of the stress response system: a stressful situation may not trigger a cortisol response if the stress can be efficiently managed by the faster-responding mechanisms of the ANS (Del Giudice et al., 2011). Instead, HPA may only be engaged to challenges that require substantial, longer-lasting mobilization of resources. More research with varying approaches to the asymmetry and interactions are needed.
Adolescent sex was important in endocrinological reactivity, as girls, compared to boys, showed lower basal levels and cortisol and alpha-amylase responses in the conflict discussion with mother. The result concurs with research reporting more blunted cortisol and alpha-amylase reactivity to psychological stressors in women compared to men, who typically show increased reactivity (Kudielka & Kirschbaum, 2005). Different from earlier performance-related stress tasks, our task involved interpersonal stress with the mother, which may represent gender-specific stressor to adolescents. However, boys and girls did not differ in their subjective evaluation of the stress. Our result is an important contribution to the literature, as findings on sex differences in adolescence are inconsistent and research on alpha-amylase is scarce (Panagiotakopoulos & Neigh, 2014; Strahler et al., 2017). It is noteworthy that besides adolescent sex, other demographic factors were not significant to the cortisol and alpha-amylase levels or responses in our study.
Finally, our sample represented a unique reproductive group, as half of the participating adolescents were conceived with ART. Some research shows higher medical risks in offspring after ART (Bergh & Wennerholm, 2020). These risks can suggest vulnerability to neuroendocrinological dysfunction in ART children and adolescents, but there are still very few studies (Belva et al., 2013). Our previous research, based on the same prospective study, found only minor differences in the daily cortisol levels between ART and NC children in middle childhood (Vänskä et al., 2016), and the current findings did not show any differences in cortisol or alpha-amylase reactivity between ART and NC adolescents. Therefore, our results can provide some reassurance that infertility and ART do not place children’s neuroendocrinological development at a long-term risk.
Strengths and Limits of the Study
Our results confirmed some general observations regarding the biobehavioral associations between internalizing and externalizing problems and alpha-amylase and cortisol. The salivary products of the ANS and HPA showed distinct response profiles for alpha-amylase as a rapid mobilizing response to stress and for cortisol as a later response important for recovery (Jones et al., 2020; Nater & Rohleder, 2009). The alpha-amylase response reached a peak and recovered more quickly than the cortisol response. Furthermore, sex differences in girls’ blunted and boys’ heightened cortisol and alpha-amylase reactivity support earlier findings (Kudielka & Kirschbaum, 2005; Panagiotakopoulos & Neigh, 2014).
Yet, our study also has limitations regarding the research setting and sample size. Instead of a task-performance or social-evaluative situation, the adolescent stress responsivity in our study was analyzed in the context of an adolescent–mother conflict discussion. Although our research setting can be considered as relatively ecologically valid, it can as well be criticized for not including fathers. Furthermore, our results are based on five time points for cortisol and four for alpha-amylase assessments. In an ideal setting, there would have been two baseline assessments, instead of our one, before the stress task to account for anticipatory stress (Gordis et al., 2006; Gunnar et al., 2009). Although the data collection was carried out mainly in the adolescents’ homes, the novel situation and the psychologist conducting the assessment may have initiated endocrinological responses. We did, however, include a relaxation task at the beginning of the experiment to reduce anticipatory stress.
Finally, our analyses were based on one-time measurements of mental health and neuroendocrinological stress reactivity. As such, our results are correlational in nature and do not allow for conclusions about causal influences (Hamaker, 2023). Future studies could analyze directional effects using repeated measures panel designs. Furthermore, our sample size was relatively small (80 adolescents in total), which led to only moderate statistical power that may have prevented some effects from reaching statistical significance in our sample. Due to the small sample, we could only study the associations between adolescent mental health problems and neuroendocrinological reactivity within the whole sample. Yet, the associations may have differed between girls and boys, and thus using sex as a moderator rather than covariate might have been informative. A larger sample would also have allowed investigating the role of salient emotional, cognitive, and social processes in mediating and moderating the mental health and neuroendocrinological associations, such as the emotional quality of the adolescent–mother relationship.
Supplemental Material
sj-docx-1-jbd-10.1177_01650254231208965 – Supplemental material for How is mental health associated with adolescent alpha-amylase and cortisol reactivity and coordination?
Supplemental material, sj-docx-1-jbd-10.1177_01650254231208965 for How is mental health associated with adolescent alpha-amylase and cortisol reactivity and coordination? by Mervi Vänskä, Samuli Kangaslampi, Jallu Lindblom, Raija-Leena Punamäki, Mirva Heikkilä, Lotta Heikkilä, Aila Tiitinen and Marjo Flykt in International Journal of Behavioral Development
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was a part of the Miracles of Development research project supported by the Academy of Finland (#3266413) and Juho Vainio Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
