Abstract
Engineered living materials (ELMs) are composite technologies that respond to environmental cues, and are able to remodel, self-organise and self-heal. Proposed as a fusion of synthetic biology and classical materials science, ELMs are seen as having the potential to transform domains such as healthcare or transport infrastructure, and to underpin future circular bioeconomies. In this paper, drawing on work that explores the pragmatic dimensions of developing new biomaterials in collaboration with microorganisms themselves, we focus on how our interdisciplinary research team responded to the surprising behavior deployed by encapsulated bacteria in the biofactory system we designed. We suggest that—and describe how—this surprise prompted us to think like bacteria—to become bacteriocentric—and how this enables the deployment of this mode of practice in the building of ELMs. We propose that this alternation between surprise and control is essential to the technological development of ELMs, and to our interdisciplinary collaboration. We suggest that bacteriocentricity could be a powerful way to problematize and explore the role of bacteria in the making of sociotechnical worlds.
Engineered living materials (ELMs), defined as any “composite material that has a biologically derived component and a synthetic component” (Nguyen et al. 2018), are technologies that respond to environmental cues, being able to remodel, self-organize and self-heal. The Royal Society (2021), using the alternative definition of “animate materials,” notes that the defining characteristics of this type of materials are that they are active, adaptive and autonomous. Emergent ELM applications include design of macroscopic functional materials deploying fungi, mammalian cells, or consortia of unicellular organisms.
Engineered living materials can be understood as the fusion of synthetic biology and classical materials science, and are seen to have the potential to transform domains such as healthcare or transport infrastructure. More specifically, ELMs have been proposed as underpinning a shift toward an economy where value is produced by recycling, re-use and repurpose (Ellen MacArthur Foundation 2019 ; European Commission 2023). Bringing these economic scenarios to bear requires, according to strategic funders such as the European Innovation Council, developing “engineered living materials with multiple predictable dynamic functionalities, shapes and scales” with environmental sustainability (EIC 2021, 3; also Srubar and Will 2021).
Due to such policy investments, biotechnologies such as ELMs have been linked with bieconomy's reliance on what Felt and colleagues (2007) have labeled the regime of techno-economic promises, and particularly with the functional conjunction of the bioeconomy with biological potentiality and commodification of hope (Roy 2018), often underpinned by the extraction of future revenues from life's processes as assets (Birch 2017; Muniesa 2014). Understanding how biotechnological projects articulate a political economy of hope relies on careful empirical understanding of how scientific and engineering practices interact with innovation processes and actors, and often with mapping the diversity and dynamic tensions within the sociotechnical logics that make up new technologies.
We take our interdisciplinary research project—a collaboration between biophysicists and a social scientist focused on an ELM prototype—a membrane-encapsulated microbial cell factory—as a case study to explore the dynamic interchange between an approach aiming to “rationally design” biomaterials, strongly aligned with the linear model of innovation, and a style that takes ecology and complexity as the model of biotechnological development (Dunham 2007, 1741; Rabinow and Bennett 2012, 157; Szymanski and Henriksen 2022). 1 Kearnes, Kuch, and Johnston (2018) identified similar modes of ordering biotechnological materials in their research on synthetic biology and nanotechnology: one strongly underpinned by an engineering approach and another, which, drawing on Bensaude-Vincent (2013, 117), they define as the chemical approach, “allow[ing] for surprise, hazards and opportunities to occur.” They argue that, while the former is usually presented as overriding biotechnology's R&D practices, the two styles of practice are equally important for experimental development of new technologies, and are entwined with differing social and political stakes and responsibility distributions across innovation networks (see also Frow and Calvert 2013).
What is unique about our project is the privileged window we had to understand the value of this chemical style of working with ELMs, and in particular how it is underpinned by fostering interspecies relationships. In the paper, we describe how the surprising and intriguing behavior of encapsulated bacteria in our experimental system motivated the interdisciplinary team to think like bacteria. This ability to become temporarily “bacteriocentric” enabled an openness to surprise, which, according to the multispecies theorist Despret (2015; 2020; 2021), is at the core of a research practice where non-humans are enrolled as partners in inquiry. Coming to inhabit what we, following Latour (2014), label as the “metamorphic zone” prompted a series of questions about bacterial sensing and behavior that have become central to the unfolding of our project and the nature of our collaboration, which we describe in the paper. In so doing, the paper brings together two strands of STS that, as Szymanski, Smith and Calvert (2021) argue, have much to gain from mutual dialogue: the social study of innovation and multispecies studies.
Drawing often on the work Haraway (2008, 2016) on the centrality of withness for processes of relational material, interspecies becoming, various scholars have traced and detailed forms of pragmatic engagement with the microbial world that do not align with the dominant anti-microbial approach or with the politics of anthropocentrism (e.g., Hinchliffe 2022). Indeed, one of the key contributions of the social studies of microbes is the conceptual recognition and empirical attention to practices in which humans work with rather than on microbes (e.g., Kilpisjärvi Collective cited in Brives, Rest and Sariola 2021). This is evident, for example, in current practices of fermentation around the world where practitioners' attunement to sensorial marks of microbial metabolism is key to the co-creation of foodstuffs (Hey 2021; Nading 2016). Our paper engages specifically with the social studies of microbes, to make sense of the agential role of bacteria in opening up the direction of scientific investigation into the capacities and possible functions of our ELM prototype.
Our analysis suggests that the experimental ELM prototype was generated through a dynamic and continuous fluctuation between a situation where multispecies entanglement dominated, and a second step where “beings [were] redistributed into different regimes of action” (Despret 2020, 186) to arrive at materials with novel properties. We argue that bringing bacterial agency to bear on technoscientific innovation requires careful, pragmatic wavering between control and surprise. As an interdisciplinary team we are interested not only in evidencing this argument but also in engendering new practices, that is to say, in intervening (Zuiderent-Jerak 2015) by considering how our analysis could move us toward “new paradigms shared with the microorganisms themselves” (Szymanski and Calvert 2018, 5).
The next section focuses on providing a thick and contextualized description of the surprising behavior deployed by encapsulated bacteria in our experiments, the consequences of this for our project's aims and focus, and for the scientific and technological understanding of cell membrane interfaces. We draw on this description to conceptualize the teams' engagement with this unexpected behavior as an active problematization of a bacterial being otherwise. We discuss the possible implications of this conceptualization in the last section of the paper and suggest that our dynamic model is relevant to explore future uses of bacteria–membrane interfaces and the ELM technology.
Bacterial Surprise
I met Margarita in her office in the morning to discuss the [then] upcoming program and activities for our interdisciplinary workshop/symposium on new “smart” living materials and responsible innovation. I had brought a draft outline of the presentation and activities schedule and was ready to present the main idea behind it. I sat at the small roundtable where we normally assembled as a team and opened my laptop intently, but noticed Margarita was still sitting at her desk by the ample window that lights her southwesterly facing office.
“Do you want to see something really cool?” Margarita probed teasingly as she moved the chair toward the table.
“What?” I asked, rejigging to an interested bodily stance.
As an answer, Margarita pushed her swiveling chair back to her desk across the room and started looking for a file in her computer folders. She clicked noticeably on a video file and announced:
“Lucas sent it to me yesterday. Look.”
On the computer screen, against a light grey background, there was a scattering of still, small dark dots and larger circles. But then there was some movement: a darker circle was—seemingly—being dragged by a moving line of dots across the screen. I approached the computer, and Margarita played the video file again. My blank expression may have prompted an explanation.
“The bacteria are pulling the vesicle.”
“Wow,” I said, but still not grasping the logic and significance of the image.
Margarita ran the short microscopy video again a few times. “So cool.” After hesitating for a moment, I gave in and asked the “stupid question:” “Why is it so cool?”
We moved back to the roundtable, Margarita still amused by my question.
“It's not supposed to happen. It's like if you wanted to make a boat move from inside,” Margarita replied, physically mimicking a person sitting in the boat while jolting their body forward, “the boat wouldn't move.”
With this analogy, I suddenly realized why the video was fascinating and mirrored Margarita's smile. “Any movement forward will be reverted … So why is this happening?” she marveled.
In this field note, Tiago tried to convey the exhilaration brought by the video, as well as his slow reckoning of its significance, of why it was so cool. The contrast between our perspectives is useful because it highlights the multiple layers of instrumental, conceptual, technical and visual elements that equipped Margarita and her biophysicist colleagues to become so excited about a 10-s video showing, to Tiago's untrained eye, dots moving along what he was tempted to describe, at the time, as a “black blob.” What for Tiago was a somewhat patterned arrangement of motions on a screen, for Margarita it was “bacteria pulling the vesicle.” However, even this explanatory description of the movements was not enough to fully elucidate the special quality of the actions displayed on the screen. The video was also remarkable because it was evidence of something that “is not supposed to happen,” that it was an apparent breach of “the laws of physics.” Trying not to embarrass Tiago for not remembering Newton's third law of action and reaction, Margarita used an embodied example: in order to propel forward, a boat in water needs to couple with the outside media, for example by oars or sails. However, in the experimental system the team had created, the bacteria were encased inside a vesicle—a droplet covered by a lipid membrane—similar to how the interior of biological cells is encased by a lipid membrane. Thus, the bacteria are isolated/uncoupled from the exterior by the vesicle membrane, and the vesicles were expected to remain stationary. However, they did not.
Afterwards, Margarita continued to respond to Tiago's “stupid question” by recalling that she and the lab team had very quickly come to the agreement that the video was evidence of an unexpected and interesting finding. First, it was surprising that such a behavior would manifest at such an early stage of the research, at a time when Lucas, the doctoral researcher working on the microbial cell factory Margarita and Wilson Poon had originally envisaged, was still “characterizing” and “developing” the experimental system. This involved checking how “basic” parameters such as temperature, viscosity and composition of the medium and lipid vesicles, might affect the motility of the specific type of bacteria they were working with: a modified, “smooth swimming” strain of E. coli (AD83). From this perspective, the team first thought the phenomenon was an artifact, probably due to the experimenters' lack of familiarity with the newly developed system. However, the “mistake” continued to happen, if only rarely.
The realization that the dragging effect was not an artifact marks the beginning of the transformation from a description of the actions observed on the screen, to an understanding of what is doing those actions that Margarita had proposed (see field note above). This transformation was enabled—among other things—by the apparent breach of the laws of motion, which made the phenomenon especially interesting for a team of biophysicists: how do encapsulated bacteria generate and transmit forces across the membrane that allow them to deform the vesicle and drag it? What could explain the deformation—the “extreme physical distortion” (Dillon 2012, 193)—of the lipid vesicle?
A few weeks later, Tiago arranged to visit Lucas in the lab. Lucas had kindly scheduled to “encapsulate” some bacteria that morning to show the procedure, having had to coordinate with the lab manager the day before to obtain “model” bacterial colonies on agar plates so that, on the day, at room temperature, they were exhibiting the desired smooth motility. After picking Tiago up at reception, they collected the bacteria from the incubator together and walked to the main lab. At Lucas's own bench, he demonstrated how to set up the experimental system the team had devised. This involved creating an emulsion by agitating oil, medium, and the model bacteria for a specific amount of time in a Vortex mixer, and then stabilizing the resulting emulsion to confine the bacteria in the interior of the resulting vesicles. Tiago was impressed by the cookery-type simplicity of the procedure and more amazed by the results: observed under the microscope, the preparation visibly showed the formation of dozens of vesicles, each containing a few moving and living bacteria (Figure 1).
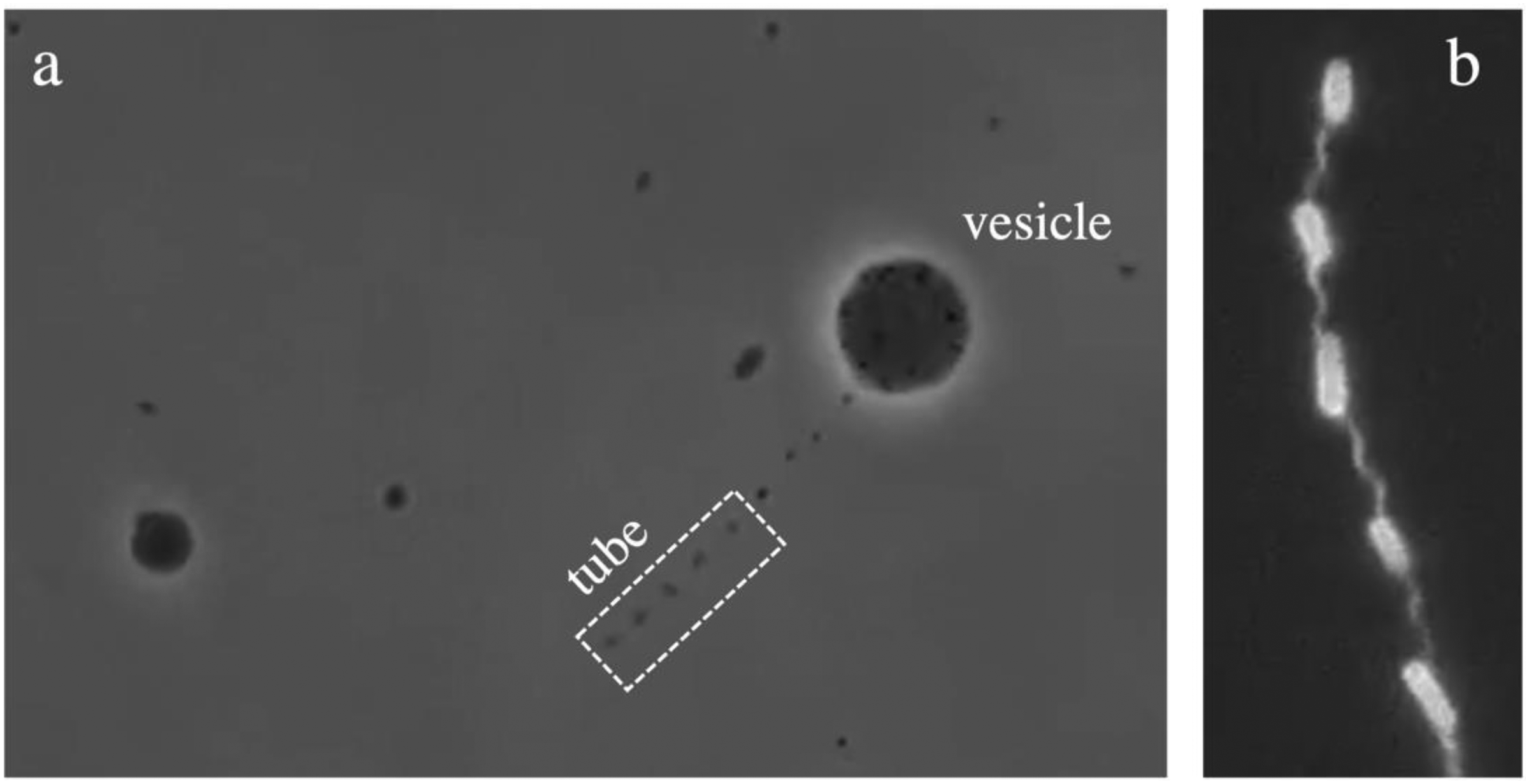
Bacteria encapsulated in lipid vesicles show emergent “cooperative” motility behavior. (a) A vesicle containing many bacteria (small dark spots), which can extract a tube. (b) A higher magnification image of the tube shows the bacterial bodies enclosed in the tube and the helical shape of the tube produced by the bacterial tails. (Image credit: Lucas Le Nagard).
As Lucas explained while they were looking and assessing the quality of the resulting vesicles, now the “real work” could start. With this expression, Lucas was referring to the painstaking toil of preparing, observing and filming the bacteria-filled vesicles in the outer solution, and waiting for something to happen. Of course, there was much more to this waiting than standing around. Indeed, waiting involved looking patiently, shifting magnification, changing light settings, observing different sections of the slide, finding something potentially interesting, recording it, annotating the observation, looking at the footage, comparing it with other recordings, and looking at the slide again, and again. On the day, it also involved speaking those actions out loud, for Tiago's benefit, while ironically lamenting the fact that nothing was really “happening.” This lack of action was in many ways adding significantly to Lucas's understanding of the system, and to identifying the conditions leading to the vesicle's deformation and the dragging effect: the absence of this outcome, while demonstrating that they still could not produce it at will, added further layers in the understanding of the phenomenon.
As the day progressed and they prepared two more batches of emulsion, Lucas's activity centered on trying to understand how the ratio of nutrients between the medium and the vesicle impacted on the occurrence of membrane distortion. Was a higher concentration of nutrients outside the membrane making the bacteria wanting to cross the boundary and leading to the extrusion of membrane tubes (see Figure 1) and the “dragging” phenomenon? That did not appear to be the case, and subsequent work focused more intensely on the mechanical properties of the membrane. Was the composition of the outer solution having an effect on vesicle adhesion to the plate glass? This reasoning was aided by the publication of two other studies on similar systems (Takatori and Sahu 2020; Vutukuri et al. 2020) and by established biophysics knowledge that vesicle deformation and tube formation can be aided by making the vesicles less tense by applying osmotic disbalance. However, to understand “how bacteria do what they do and how they do it,” as Lucas put it, he could not simply apply existing knowledge to the new situation. He had to understand, through embodied operations and instrumented observations, how swimming bacteria respond to the confined environment of the vesicle, and how the lipid membrane responds to produce membrane tubes and induce vesicle motion. Although Lucas came to understand how to experimentally make bacteria extract membrane tubes from the vesicles, the mechanics of vesicle dragging remained a mystery.
It took several attempts developing different modes of visualization and skillful ordering and re-ordering of the experimental system to solve the riddle, embodied by Margarita through the model of boat driving. It is well known that bacteria propel in aqueous media by rotating long helical tails (Wadhwa and Berg 2022), but how this motility mechanism was being deployed in pulling the vesicle remained unclear. Over time, the team consistently observed that as swimming bacteria inside the vesicle encounter its wall, they push on it and extrude thin membrane tubules, provided the vesicles are sufficiently soft. Since bacteria are tightly wrapped in the tubes, their rotating helical tails deform the lipid tubes in a similar helical fashion (Figure 1). This observation revealed the mechanism for vesicle propulsion: the tubes, shaped by the bacteria, were acting as giant pulling tails—as oars—for the vesicles (Le Nagard et al. 2022).
The discovery of such intimate and surprising physical coupling between motile cells and lipid membranes not only prompted scientific questions on the mechanical properties of the lipid membrane but also had consequences for the further framing of our project and what it meant for our collaboration.
The project had been originally conceived as an interdisciplinary collaboration in which we aspired to create an experimental living material, to be developed through a set of participatory activities engaging stakeholders in its design and application. The original project proposal stated our aim to use giant lipid vesicles (a “membrane bag”) to encapsulate and organize living bacteria in a bio-factory that is capable of growth, replication, and specific activity, e.g., motion or response to external stimuli. We believe that such an open-ended material innovation project will open new possibilities for collective experimentation between scientists and other members of society.
2
In this, we were drawing on the idea of biofactory, an established approach of using cells as producers/metabolizers of recombinant proteins and natural products, or as catalyzers of biological transformations of industrial interest. However, our focus was not on the production of high-value-added substances such as biopolymers as most of the research in this field does, but on understanding the physical and chemical interactions between the bacteria and the capsule underpinning such processes. We wanted to develop strategic knowledge about bacteria–membrane interactions in biofactories. Specifically, we wanted to manipulate bacteria's own lipid secretion to grow and multiply the vesicle container, so that we could create a living, multi-compartment, functional structure.
We also sought to involve stakeholders at an early stage in the development of the system. Our idea was to harness the open-ended technological character of our biofactory, and the fuzzy boundaries of expertise linked to the inherent uncertainty of research-in-action, to allow carefully selected stakeholders to participate in designing possible applications for the system. We planned to deploy participatory design methodologies “to generate ideas for the future applications of the bacterial factory” (Staykova, Moreira & Poon 2018, 3).
The unexpected behavior of the encapsulated bacteria challenged both of these aims and our correlated roles within the team. On the one hand, it questioned our immediate focus on creating a multifunctional bacterial factory, shifting the project in a more exploratory direction, concerned with understanding, at a fundamental level, the interactions between artificial membranes and living bacteria. This new emphasis widened the uncertainty about what such biohybrid systems could do, which fundamentally undermined the application of standard procedures of stakeholder engagement (Blok 2019). These, we came to realize, require a degree of closure on the technology's key properties and the delineation of possible, even if fuzzy, applications, from which “values” and “concerned groups” or stakeholders can be broadly derived. As the project shifted focus toward basic research, and the technological horizon was becoming more blurred, unexpectedly, our interdisciplinary collaboration was also affected.
In the first version of the project, our roles were, to a certain degree, well defined. Lucas, Margarita and Wilson were responsible for the experimental work of data collection and analysis. Initially, Tiago's role was a well-recognized function that STS scholars play in interdisciplinary projects, as the “representative of the public” (Balmer et al. 2015, 9, 16) with an ambition to perhaps become more of a “co-producer of knowledge.” The thrill sparked by the bacteria's unexpected behavior, and its opening of fundamental questions about bacteria–vesicle interactions, challenged this arrangement. As a result, it became temporarily unclear what Tiago's role was in the project, what social science expertise could be relevant to a “basic science” project, which meant that Tiago often fell back on the “classic” STS ethnographer-observer distanced role, providing thick analytical descriptions of scientists working in the lab (see above).
Crucially, the new project framing foregrounded the risks of relying solely on the regime of technoeconomic promise for generating new engineered living materials, particularly their foreshadowing of the circular economy, as discussed in the Introduction. This model restricted the material imagination the project required, preventing us from exploring the full potential of the ELM field within the politics of environmental care. In this, a major shift in the project came from Margarita's increasing interest in multispecies studies, facilitated by informal engagement with academicians working on multispecies ethnography, including those linking it to work in synthetic biology, 3 and a formal collaboration with design scholar Laura Forlano (2017) within the exploratory phase of our project. 4 We wondered whether we could draw on multispecies literature to understand the technological and epistemic dynamics in the lab and, further, to use those insights to reformulate our approaches to experimental system design and to the ELM field in general. In this process, a more collaborative, reflexive form of working emerged, with both of us invested in exploring the possibility of intervening in the ELM field, and in the design of experimental materials by adopting a bacteriocentric point of view. We explore the conceptual underpinning of this possible intervention in the next section.
Metamorphic Encounters
In the previous section, we described how the surprising behavior of bacteria encapsulated in lipid vesicles provoked a series of transformations in the focus, experimental approach, roles and relationships in our ELM research project. One of the key changes was related to the shift from seeing the dragging behavior as an artifact to grasping it as a phenomenon in its own right. In this seemingly simple conversion lies the pragmatic distinction that underpins the establishment of a fact; an alteration that was borne out of multiple trials, which showed, in the recordings and notes produced in the lab, that the dragging effect was something that happened under “some conditions,” unknown at the time Margarita shared the video.
Drawing on Garfinkel, Lynch and Livingston's (1981) in-depth analysis of the actual moment of discovery of the optical pulsar, Latour (2014) suggested that this reconfiguration can be interpreted, using semiotic terms, as a shift from performance to competence, that is to say, from a description of actions to a description of actors doing such actions. There is a similar dynamics in the field note provided above: on the one hand, a reading of the video as a recording of dots and blobs artificially moving on the screen, and on the other, a description of it as footage of bacteria physically pulling a vesicle in a medium, in seeming defiance of the laws of physics. The consequences of this translation—i.e., the conversion of the dragging performance into a competence—emanated to the whole project, shifting its aims and focus, including the very collaborative arrangement we had put together, which is described at the end of the previous section. This was crucial because in our lab experiment, this competence could not be explained by the properties of the individual components but only from the emerging, mediated interplay of bacterial activity and the artificial scaffolds.
This speaks directly to an STS view of experiments as events capable of generating unanticipated outcomes, a capacity that Rheinberger (1997, 32-33) considers unique to the experimental sciences, and which is key to the shaping of future biological and social relationships that arises from those events (Latour 2021). How to think about such novel experimental findings, however, is a recurring problem, because there is a constant temptation to take a shifting relational enactment for a primary source of agency: that is to say, to assume an original foundation in the causal chain rather than taking it as the outcome of an unfolding configuration of interactions and attachments (concepts, standardized organisms, instruments, graphs, etc.), particularly if taking the point of view of the experimenter.
To address this issue, in her critical analysis of practices of animal research, Despret (2015) argues that novel epistemic claims underpinned by model organisms such as our E. coli batches rely on a forked but interchanging pragmatic engagement with them, as both standardized instruments and co-producers of knowledge. In this, Despret proposes that researchers' capacity to think like their organism of choice is fundamental to their ability to design the experimental setting, re-formulate the question, and devise the tests and tools—to set up the “right conditions,” referring back to our case—that allow them to build epistemic claims on general principles of life or biophysical systems. For this, they have to inescapably rely on an embodied and situated understanding of the organism they are working with. We offered a detailed description of these engagements in the field note above, starting with Margarita's spontaneous impersonation of someone attempting and failing to move a boat while sitting inside it by jolting their body forward.
In doing these maneuvers, Despret (2015) would argue, researchers draw on a way of thinking about organisms that is similar to that proposed by Uexküll (2010). For Uexküll, organisms cannot be conceived as mere machine-like repositories of genetic programs responding to the environment and a general set of stimuli, but should instead be understood as actively transforming the very environment they inhabit. This activity is encapsuled in the concept of Umwelt, through which organisms establish and deploy their own world as a species-specific functional circle of perception and action. This complex circular activity may be defined as organism-as-environment. In this respect, thinking like an organism entails a situated, mediated corporeal imagining of this complex, species-specific unfolding circular activity with the environment.
In our project, making an embodied sense of bacteria's surprising action involved drawing on what Dexter Dyer (2003) once described as bacteriocentricity. Although formulated to guide the work of her fellow field microbiologists, we suggest that the concept of bacteriocentricity also underpins much experimental work in biophysics and our own ELM design. Being bacteriocentric is to put [one]self in the place of bacteria, to try to experience the world as they experience it. [It] is a useful point of view to cultivate [when] you are trying to observe and understand the bacterial world … through the mental exercise of “being a bacterium” [or] “thinking like a bacterium”—trying to guess what parts of the microenvironment are essential, what aspects might be combined to create the right conditions. (Dexter Dyer 2003, 7; our emphasis)
5
This shifting of perspective is essential, Dexter Dyer argues, for the work of any microbiologist, cheese maker—or biophysicist, we would argue—culturing and/or experimenting with bacteria. Dexter Dyer's emphasis on the importance of scientists experiencing the world in the way bacteria do is redolent of Uexküll's proposal for a biosemiotics, as evidenced in the above reference to the microenvironment. Crucially, it overlaps with Despret's conceptualization of the importance of thinking like model organisms for experimental research. 6 Bacteriocentricity, we suggest, has been a resource for physicists trying to explain the microbial world mechanistically.
To fully understand the implications of this suggestion we have to start by exploring what imagining a physical world completely different from that of humans and animals might entail. In the early studies of bacterial motility—of how and why bacteria move in aqueous media—adopting a bacteriocentric approach meant trying to appreciate, and even embody, the physical and chemical properties of the environment from the standpoint of bacteria, so as to design the best instruments and experiments to trace such phenomena. In the microbial world, motion is greatly resisted by viscous forces, in conditions equivalent to “a man swimming as his own sperm cells,” in a “swimming pool in molasses [where] we can only move [our hands as slowly as] the hands of a clock” (Purcell 1977, 6). Thus, Purcell (1997)—better known for his work on nuclear magnetic resonance, drawing on Taylor (1952)—proposed that in order to propel forward in such conditions, microorganisms can only use non-reversible motions, such as waving flexible oars (as in the case of sperm cells) or rotating the long thin helical tail at the end of their bodies (as in the case of bacteria). This was elaborated by Purcell's colleague Berg (2003), who used this insight to build an experimental system where E. coli bacteria are glued on a rigid glass and their helical motion recorded.
Despret proposes that model organism researchers, such as Purcell and Berg, are able to produce mechanistic, causal explanations from within complexity because they usually rely on a two-fold epistemic dynamic: a first step, where multispecies entanglement dominates, and a second step, where separation of categorical and ontological domains is the central concern (Despret 2020). Haraway (2016; 2008) urges us to think about the first movement as a process of more-than-human becoming, a situation where human and non-human actors exchange qualities, learn from each other, form alliances and care relationships. From this perspective, being bacteriocentric means coming to inhabit a particular world or bacterial Umwelt, an operation that is, despite Dexter Dyers's own words, more than a “mental exercise:” it requires an incarnate, instrumented imagining of a different perceptual world-in-action, a grasping, even if intuitive and not fully verbalized, of a being otherwise.
This operation takes place in what Latour labeled the metamorphic zone (Latour 2014, 94) and Haraway (2008, 219) designated as a contact zone. Despite its spatial implication, the notion explicitly avoids drawing on regional, ontologically rigid models to understand the relationship between humans and non-humans (Latour 2013, xxvi). In this, the idea of metamorphic zone aims to describe a particular quality of relationships between species (metamorphosis) where entities shift and transform, a phase that occurs only within a set of specific conditions (the zone), and could be seen as conceptually more akin to a geological stratum (Latour 2014). Imagining this “being otherwise” in the metamorphic zone is well exemplified in the specificity of Purcell's example of bacteriocentricity above—the pool of molasses—or in the operations performed by Lucas in the laboratory to capture the “right conditions” in which the tubes form. To understand this configurational layer, it is necessary to grasp the proliferation and multiplication of possible ways of being: imagining a radically different form of motility or proposing a distinctively bacterial “sense of aesthetics” (Berg 2003, 15). In other words, to imagine this zone, rather than start with known regions/ontological domains, it is necessary to formulate problematizing, searching, tentative descriptions of the dynamic and open set of entities generated within empirical ontologies.
Despret's concern, and Latour's, as we intimated in the beginning of this section, is that post hoc, polished scientific descriptions of what happens in the metamorphic zone downplay its reliance on the type of movements, relationships and mediated exchanges, which make possible something like bacteriocentricity. Once knowledge is established, through the complex chains of references that STS researchers have detailed for many years, the rat or the bacteria are discarded (often physically), and are no longer part of the story. Thus, taking the rodent merely as a laboratory model, researchers can avoid a “questioning on the subject of the rat” (Despret 2015, 15), despite their own experience of handling, following, observing and caring for them as organisms-as-environment (Moreira 2022). Similarly, framing E. coli solely as a model organism for behavioral or metabolic genetics (Zimmer 2012) devalues the wealth of knowledge researchers built—and methodologically rely upon to build experiments—about the bacterial Umwelt. How are we to avoid this reduction? What do we gain by allowing for more-than-human transformations and exchanges in the making of new ELMs and living technologies? We address these questions in the following section.
Expanding Bacteriocentricity as Framework for ELM Research
In the previous section, we explored how projects such as ours are confronted with a tension between following the more-than-human transformations and exchanges that make creating new knowledge possible, and having to provide a finished account of how plans of action and hypotheses yielded results. We proposed that bacteriocentricity was a key part of the originality and imagination that underpinned the lab team's discovery of the new biophysical behavior—the intimate physical coupling between motile cells and lipid membranes. Yet following Despret's analysis of model organism research, we were also concerned with how the surprising exchanges and the care enacted in the metamorphic zone may be discarded in favor of narratives where agency/power is discursively attributed to only one or a restricted type of actor in the zone (usually the “subject” or the “object”).
In our project, two easily recognizable narratives were readily available: Science as it is often portrayed in defenses of its “objectivity,” and Technology. To a certain extent, the issues arising from drawing on the first narrative to make sense of what happened in the lab and under the microscope was already critically dealt with in the previous section by detailing the more-than-human basis of experimental work. Furthermore, there are similarities in the structure between the two narratives, with technological mastery often depicted as a form of control equivalent to that shown by “great men of Science.” For example, focusing on synthetic biology, Rabinow and Bennett (2012, 16) have argued that post-genomic techniques have led to “the intensification of an engineering disposition in biology,” seeking perfect alignment between form (in the genome) and function (behavior), and a weakening of craft-based practice within technoscientific innovation. The latter enables the generative ordering of experimental practices, opening aspects and proliferating questions beyond the strict aims of the design/control process.
The narrative of technological design is also dominant in the field of ELM. With the combination of synthetic biology and materials science, the field has reinforced the engineering mode of the practice of biology by linking it with specific sociotechnical expectations of environmental, biomedical, industrial and military applications. This mode of practice is underpinned by the overarching aim to “rationally design” tools and applications in the field of ELM as a whole (Lantada, Korvink and Islam 2022; Rodrigo-Navarro et al. 2021; Srubar and Will 2021), and its reliance on the linear model of innovation, evident in the vision for ELM articulated by institutions such as the European Innovation Council cited above.
Kearnes, Kuch and Johnston (2018, 9) have suggested that in synthetic biology laboratory practice, top-down computational models of engineering biological materials co-exist with an alternative narrative of “design as emergent,” aligned with an openness to surprise that is inherent to life (see also Dade-Robertson and Zhang 2022). This duality, they argue, underlies the innovative capacities of synthetic biology rather than being evidence of an opposition between new and obsolete modes of practice. The same tension is also key to Bensaude-Vincent's (2013; 2011) history of synthetic biology as a discipline, and to her exploration of the establishment of materials science and nanotechnology. Notably, Latour (2013, 207-232) proposes that this heterogeneity is central to technology as a mode of existence: technological objects are constituted by this wavering, or “zigzags” between improvised and planned action (see also Suchman 2007), and between engineering control and creative surprise.
This work on the generative tensions that are inherent to technoscientific innovation, taken together, offers a way to reopen the dynamics that Despret (2020, 187) viewed as preventing us from being attentive to the “surprising kinship of ways of affecting, of influencing, of coming to bear on the behavior of [non-human] others.” Telling different stories about life in the metamorphic zone allows us to re-engage and remake the worlds we inhabit (see also Despret 2021). As proposed in the last section, a possible version of those different accounts is encapsulated in the idea of bacteriocentricity. We argue that to move technoscientific practices toward “new paradigms shared with the microorganisms themselves” (Szymanski and Calvert 2018, 5), we should extend the bacteriocentric point of view beyond its use as sense-making template, into a technique for scientific inquiry and invention. From this perspective, as a way of asking questions of experimental systems, bacteriocentricity is not simply a “frame of mind” or a “mental state” but, as suggested above, an active problematization, that is, a tentative way of engaging with the “art of weaving sense in situations requiring an ongoing reinvention of the way lives, human and non-human, are involved in each other” (Stengers 2021, 89). For this approach to become generative, however, we need to embed bacteriocentricity in the wavering between control and surprise in living materials design. Below, we detail how we endeavor to do so.
A significant example of bacteriocentric problematization in action in our work was the realization that the vesicle dragging effect was highly specific, which is to say that it was an “obligatory point of passage” for the lab team's understanding and experimental stabilization of the “right conditions” for its occurrence. Key in this was the realization that dragging occurs only when motile bacteria are placed in a unique artificial environment: a lipid membrane soft enough to be deformed and shaped by persistent locomotive forces, while keeping its integrity as a compartment. Relatedly, drawing on the bacteriocentric imagining of the physics of swimming in molasses, the lab team came to understand that only bacteria that have distinct tails (flagella), and likely only the type of bacterial strain (AD83) used in the experiments, in which the characteristic run-and-tumble bacterial motility has been replaced by smooth directed swimming, can shape the vesicle membrane into a propelling tube. In this, the dragging effect, caused by the harmonized tractor propeller formed by groups of bacteria swimming inside the tube, spoke directly to an engineering understanding of the system, which is currently being investigated further: it opened questions about how other bacterial swimming modalities interact with lipid membranes and how the unexpected bacteria–membrane interplay may be harnessed to engineer moving functional components and “bio-bots.” In that way, bacteriocentricity meant not only thinking through the biophysics of bacterial motility in the Umwelt we artificially imposed, but allowed us to also appreciate the biological variability and the “character” of specific microbial strains, as well as the engineering potential of such variability.
A key corollary of the encapsulated bacteria's unexpected behavior was also that it redirected our attention into thinking about the role of bacteria–membrane interactions in the origins of symbiosis between cells. Part of the sense-making involved drawing links between the phenomena under the microscope and the biophysics that underpin the symbiogenesis hypothesis of eukaryotic cells (Margulis 1991), and the dynamics of holobionts in evolution (e.g., Bordenstein and Theis 2015). The team realized that very little was known about how, in the evolution of life, some bacteria were able to physically enter the interior of other cells and, once inside, how they modified and transformed the host membranes into their own protective niches, to create the higher forms of life seen today. Similar interactions between bacteria and membranes underlie the mechanisms of bacterial infection. While in mainstream biology these processes are usually explained in terms of evolution and natural selection acting on unsuspecting microorganisms (Becking's famous tenet: “everything is everywhere but the environment selects”), our laboratory work brought to bear the central role of bacterial agency in these processes: their capacity to actively sense and modify their environment in ingenious and surprising ways is essential to both pathogenesis and evolution.
Linking our experimental findings to symbiogenesis also enabled us to think very differently about engineered living materials. On one hand, it brought into focus the active role of bacteria and their environment in the design process. The question was now a different one: if symbiosis between cells, which is fundamental to all aspects of life on earth today, is the result of bacteria–environment enactments, can we use the active and innovative capacity of microbes as a fundamental engineering stipulation to be explored in further system design? The experimental prototype thus opened the possibility of drawing on the coupling of living cells with membrane scaffolds to arrive at new biohybrid technologies. Can ELMs be designed as dynamic synthetic ecologies that grow out of the symbiotic interaction between microbial species, their artificial scaffolds, users' needs and environmental cues? Such bacteriocentric problematization requires a more open-ended style that is amenable to surprise, allowing new design opportunities to occur. It could perhaps rely more systematically on the listen-parse-response (LPR) cycle proposed by Szymanski and Henriksen (2022). In other words, instead of “forcing” bacteria through genetic engineering to produce or functionalize materials for human benefit (often inefficiently), we could rely on their sensorial, agential capacities to guide them to enter into mutualistic relationships with their artificial material scaffolds. For example, we are currently experimentally working on biohybrid capsules, which can direct themselves, drawing on the ability of bacteria to move along chemical gradients (chemotaxis) to locations inside where they can grow and replenish themselves, and further modify the environment.
Another consequence of adopting the bacteriocentric approach to technology development is that we lost sight of a clear user domain and sociotechnical horizon to which the system spoke. While drawing on the bacterial factory model that emplaced the project in the world of biomedicine or food production where those approaches are most commonly used, the new system does not—yet—have a possible application. As we argued above, this meant that standard stakeholder mapping tools were difficult to implement. Adapting bacteriocentricity to ELMs as problematization meant drawing on interdisciplinary work that shapes and traces the relationship between microbes and humans in terms of assemblages, problematizing their shifting relationship in specific settings (e.g., Brives, Rest and Sariola 2021). We came to see humans both as skillful users and as a living component of ELMs, whose needs evolve and adapt in concert with the other living and non-living components of the material. This means that we needed methods that can tap into existing knowledge of living with living materials to define, anticipate and explore future uses of the technology (e.g., Tomico, Wakkary and Andersen 2023).
In collaboration with design scholar Justin Marshall, we have developed and conducted a series of speculative design workshops with members of the public to explore such possible uses. Using narrative and visual prompts, we sought to provide the means by which imaginaries could be generated and made manifest through a talk-in-interaction. The prompts were intended to shift the focus from a functional, human-centered approach to one that emphasized the bacteriocentric point of view and interspecies relationships, in order to recast the problem of how to “live with” other living things. These workshops were successful in generating a variety of rich collective insights about the work of maintenance and care of living, which are unpredictable things that we report elsewhere (Moreira, Marshall and Staykova 2023). How to reverse-translate and act on these forms of engagement with materials into the design of ELM as an intervention remains our shared aim in the collaboration.
Conclusion
Our point of departure for this paper was current proposals to engineer new materials and systems that can support circular bioeconomies. Our paper argues that rather than mere technical solutions to the environmental problems created by industrial modes of production, ELMs might play an important role in problematizing and experimenting with the relationship between biological and non-biological, and more-than-human materials in technoscientific innovation. Drawing on work that explores the pragmatic dimensions of developing new biomaterials where the agency and participation of microorganisms themselves are acknowledged, accounted for and harnessed, we focused on how our interdisciplinary research team responded to the surprising behavior deployed by encapsulated bacteria.
We analyzed how the unexpected deforming and dragging behaviors prompted the lab team to engage in what we, after Dexter Dyer (2003), called bacteriocentricity, which we conceptualized as an active problematization of affordances and implications of the bacterial Umwelt. Drawing on our and colleagues' lab work as the main source of data, we suggested that this bacteriocentricity was responsible for driving sense-making and experimental tinkering in a way that made visible and brought to bear the relationship and exchanges characteristic of the “contact” or “metamorphic” zone. Wanting to avoid the habitual way in which these surprising exchanges are discarded in favor of technical mastery and control narratives, we proposed that emphasizing—extending—bacteriocentricity is essential to alternating between surprise and planned design in technological development in ELMs, and have detailed how we are continuing to do so in our collaboration.
As noted in the Introduction, our work aligns with current work in multispecies studies in detailing, this time in a lab, how humans become attuned—through a series of mediators—to the microbes with which they work. This ability to think like bacteria is essential, we argued, to be able to design, conduct and analyze microbiology or biophysics experiments with bacteria. While this embodied and highly equipped knowledge is normally relegated to the “experimental techniques” section or delegated to technicians, we have demonstrated that this embodied imagining and openness to surprise makes those very experiments possible. We argued that it is important to not dispose, in our story making, of the shared withness so that they come to bear on the making of new ELMs. In so doing, however, there is a need to directly shape future experiments and technological development. A key contribution of our paper is to propose a synthetic ecology approach in ELMs, which fruitfully navigates the two-fold dynamic alternation between mediated withness and situations of engineered, planned redistribution of ontological domains (control). As we create synthetic material ecosystems and get acquainted with their emergent behaviors, it is possible that bacteriocentricity may have to be extended to account for further hybrid identities and symbiosis within ELMs.
Our project was a fruitful, if convoluted, collaboration between biophysics and STS, particularly on how STS ways of reasoning pragmatically supported extending and giving space to surprise within technoscientific practice. However, it also made it clear that STS needs to develop a more detailed empirical and conceptual understanding of the structure of exchanges in the “metamorphic zone” beyond Latour's (2013) own brief description, which is explored above. While we agree with Latour and Stengers that to make the exchanges and care between humans and non-humans matter means learning and re-learning, within specific ontologies, what transformation is required of us in that encounter, there might be specific modes of practice that are intrinsic to the metamorphic zone, which we need to identify, map and interrelate. We suggest that bacteriocentricity as active problematization of the bacterial Umwelt might be a fundamental pragmatic disposition in the various mundane scientific and technological interactions we have with bacteria (Moreira 2023). As we argued, bacteriocentricity should not be seen as a template of technological solution but as a powerful way to problematize and explore the role of bacteria in the making of sociotechnical worlds. If, as we suggested, thinking like bacteria figured centrally in making E. coli the “workhorse of biomolecular biology and biotechnology,” there are many bacteriocentric STS stories to tell to fundamentally understand and reshape the dynamics of contemporary science and technology. We hope to have inspired others to continue bringing the agential role of bacteria to bear on those dynamics.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Royal Society (grant number APX/R1/191020).
