Abstract
In this paper, I look at two different sets of practices that are part of the risk management apparatus in place in Québec & Canada to apprehend and control risks associated with food consumption. More specifically, I contrast diabetes and chemical contaminants risk management testing practices, so as to compare how both frame and approach risks, in a context where recent research in social sciences, epigenetics and environmental sciences increasingly points to “environmental” pathways of disease causation while many chronic conditions remain highly individualized in public and health discourses. The analysis pays close attention to the different temporalities discursively created, considered, and neglected in these practices in order to understand how risk is approached and worked on. This highlights the power relations that inform how we care (or not) for (certain) bodies, inflecting in particular ways their—uneven—becomings. I argue that the Canadian biotechnological apparatus of testing practices meant to apprehend and control diet-related risks contributes to foreclosing the temporalities of health and illness considered and acted upon. As such, the apparatus contributes to (re)producing inequalities, here mostly health related ones, as well as creating differentiated biological materialities.
As health authorities in Canada and beyond urge governments to take action to address the “obesity crisis” and the associated “diabetes epidemic” (Ellison, McPhail and Mitchinson 2018; McCullough and Hardin 2013; McNaughton 2013b), the health risks associated with these chronic—“non-communicable”—conditions are approached as manageable through the individual's diet, and framed as the result of “bad” lifestyle choices and food consumption behaviors (Bennett 2019; Frazer and Walker 2021; Gálvez, Carney and Yates-Doerr 2020; Overend et al. 2020; Durocher 2023). Yet, studies in social sciences (Gálvez, Carney and Yates-Doerr 2020; Guthman et al. 2014; Guthman and Mansfield 2013; Hatch, Sternlieb and Gordon 2019; Landecker 2011; Lock and Nguyen 2018; Moran-Thomas 2019; Shantz and Elliott 2021) and in epigenetics and environmental sciences increasingly point to “environmental” causes of disease in the development of such conditions (Chevalier and Fénichel 2016; Evangelou et al. 2016; Gupta et al. 2020; Heindel et al. 2017; Neel and Sargis 2011; O’Donovan and Cadena-Gaitán 2018; Velmurugan et al. 2017). Observing a discrepancy between this body of research and how chronic conditions, including diabetes, are framed within public discourses and public and clinical health contexts prompted the question: how do health authorities approach risk in the context of increasing occurrence of diet-related chronic conditions, and fears associated with food contaminants? Does the apparatus set into place to control and manage health risks associated with food consumption reflect or integrate research insights pointing to environmental pathways of disease causation, especially in a context where many chronic conditions are highly individualized? This paper comes out of a broader research project where I investigate these questions.
Here I look at two different sets of practices that are part of the food ingestion risk management apparatus in Québec and Canada, 1 to apprehend and control risks associated with food consumption. More specifically, I contrast diabetes and chemical contaminants risk management testing practices, so as to compare how both frame and approach risks in the context of food consumption. I am interested in what these practices do (what leads to their use; what comes out of the interpretation of their results; whether they lead to subsequent risk management practices), and how they foreclose how risk is framed, which is to say, how risk is worked upon. Comparing and contrasting these practices approach and work on risk allows me to question the power relations and ideologies that inform the development of these practices—and, more broadly, the development of the apparatus meant to control diet-associated health risks—as well as how they inflect (unevenly) the becoming of bodies and health.
STS scholars have demonstrated that bodies are cared for using biomedical tools, methods and technologies in a manner that is not purely “objective,” separated from ideologies that permeate our societies: Biomedical technologies bring about transformations, resulting in newly discovered knowledge about the material world that, in turn, influences subsequent interventions… Health-related matters are routinely “objectified” as technical problems, to be solved through the application of technology and the conduct of science, and are, by definition…decontextualized in practice. Objectification tends to make opaque moral assumptions embedded in the actual application of any given technology… (Lock and Nguyen 2018, 16; see also Martin, Myers and Viseu 2015; Puig de la Bellacasa 2017)
This perspective and my interest in questioning how the testing apparatus inflects bodies and health is informed by this (largely feminist) STS scholarship, and also by the works of “new” and feminist materialists who have increasingly paid attention to matters and their constant, contingent processes of transforming and becoming (Alaimo and Hekman 2008; Coole and Frost 2010; Pitts-Taylor 2016; Roy and Subramaniam 2016). These scholars have called for looking at biology and its ever-transforming processes (notably through biotechnological and scientific developments), so as to reveal the power relations that inform the becoming of materialities, bodies, and, of particular interest to me here, health. Like many of these scholars who use the term “materiality” to mark the shift from an earlier “linguistic turn” in feminist thought (Barad 2003; Coole and Frost 2010) that frames matters as inert and passive, I examine how testing practices work with organic materialities, thus inflecting their active becoming. Sociologists, anthropologists, feminist STS scholars, and environmental justice researchers and activists have demonstrated that risk is unevenly distributed and addressed (Gravlee 2009; Harthorn and Oaks 2003; Murphy 2006, 2017; Nishime and Hester Williams 2018). Neoliberal practices of governance as well as techno-scientific and biomedical developments and practices can foster and/or reinforce social, economic and environmental inequities while differentially “allocating risks for disease and opportunities for health and well-being along existing and new lines of privilege and oppression” (Polzer and Power 2016, 11; see also Ehlers and Krupar 2019). The discrepancy between research stressing the environmental pathways of disease causation, and the focus on individual responsibility for managing conditions such as obesity and chronic diseases led me to investigate whether the biotechnological apparatus developed and used in Canada to control risks associated with food ingestion contributes to reinforcing bodily difference and health inequalities.
I thus question how testing practices dedicated to preventing and controlling health risks associated with food consumption frame and approach risks and disease causation pathways so as to expose the power relations that permeate these practices and the apparatus of diet-associated risk management they constitute. To do this critical work, I pay close attention to the different temporalities discursively created, considered, and acted upon in the context of these practices.
Why Temporalities?
Risk exists only in a temporal sense due to “its relation to an unknown future;” it “expresses not something that has happened or is happening, but something that might happen” (Reith 2004, 386). Western biomedicalized societies (Clarke et al. 2010; Crawford 1980) are characterized by the apprehension and anticipation of health risks through regimes of anticipation that permeate how risks and the future are apprehended, worked on, and how bodies are cared for (Adams, Murphy and Clarke 2009). Anticipation has become “a form of care and risk management” (Molas and Whittaker 2023; see also Harthorn and Oaks 2003). Anticipation is a broad, overarching temporal regime (and concept). In this article I delve into how it orients risk prevention and control in specific ways, in the context of diet-related testing practices, drawing on Sharma's (2013, 2014) work on temporality. Sharma (2014) adopts a critical approach to time to question the power relations negotiated through the creation of multiple, differentiated temporalities that produce social difference, as people inhabit and experience time differently. Inspired notably by Massey's work on time and space, Sharma (2013, 312) understands time to be “multiple, relational, and deeply uneven.” While she uses this cultural and critical approach to time to question how people experience and inhabit time, I draw on her method to question the different temporalities at play within diet-related risk management practices. By saying “at play,” I mean that I draw on STS to investigate how bio-technological practices contribute to creating and framing temporalities to be considered and/or neglected, as well as temporalities to be acted upon—and how this transpires.
Time materializes in bodies and, as many socio-cultural anthropologists, feminist, and feminist STS scholars have argued, body materialities and health are inflected by different environmental and socio-cultural processes operating along the lines of race, gender, and class (Ehlers and Krupar 2019; Fausto-Sterling 2005; Gravlee 2009; Lock and Nguyen 2018; Murphy 2006, 2012; Pitts-Taylor 2018; Smith and Vasudevan 2017). These processes follow different temporalities, meaning different timeframes, timing, duration, evocations/constructions of the past, present and future. This has been investigated, for instance, by social scientists analyzing how inequalities and adversity become embodied, often integrating developments from epigenetics into their analysis (Landecker 2011; Lappé and Hein 2021, 2023; Mansfield 2017; Poleykett and Jent 2023). Health and medical sociologists and socio-cultural anthropologists have also investigated the different temporalities at play in the development of chronic diseases or chronic disease epidemics. Other scholars have paid attention to how temporalities are produced, conveyed or experienced in care practices (for example, Sager and Zuiderent-Jerak 2021) and how temporalities are conveyed and mobilized to create a sense of urgency contributing to stigmatizing certain bodies and limiting the types of bodies and health deemed “acceptable” (Felt et al. 2014; Tidgwell et al. 2018). I mobilize these different works on temporalities throughout my analysis of the multiple temporalities at play in the biotechnological practices constitutive of Canada's diet-related risk management apparatus. I argue that the existing risk management apparatus contributes to (re)producing inequalities— especially health-related ones, and to creating differentiated biological materialities as it forecloses the temporalities of health and illness considered and acted upon.
Here again, I draw on Sharma, who explains that temporalities, as power relations, must be “understood in terms of [their] discursive power” (Sharma 2017, 195). The practices analyzed in this paper are all about managing diet-associated risks in the present, so that future health issues can be prevented. But health and illness—and the materialization of inequalities in differentiated health and organic materialities—are inflected by different temporalities. Paying closer attention to the different temporalities discursively created, conveyed, and foreclosed in these practices helps in understanding how risk is approached and worked on (or not), and as such allows for highlighting the power relations that inform how we care (or not) for (certain) bodies, hence inflecting in particular ways their—uneven—becomings.
In the upcoming sections, I first provide more information on the practices in focus. I then analyze different “phases” of the risk management practices and discuss the different temporalities taking shape throughout. I conclude with a discussion contrasting the different temporalities permeating the two sets of practices and what it means for how risk is approached and worked on. I also offer a reflection on what a greater collectivization of health risks might look like, specifically for testing practices.
The Practices in Focus—a few Words About Methods
To gather the materials analyzed in this paper, I conducted 10 semi-structured interviews with Public Health researchers and program managers at both federal (Canada) and provincial (Québec) levels, as well as with clinical practitioners (clinical physician, dietician). 2 These interviews helped me understand and trace back the array of testing practices used to monitor and control health risks currently associated with the diet. The two sets of practices analyzed in this paper are part of a longer list of practices I identified that either directly test food and body materialities; are informed by such testing (like the nutrient file database); or are part of the broader food and bodies health risk governance apparatus (Canadian Community Health Survey, for example). I focus on these two sets of practices for two reasons: (1) they involve organic materialities testing, thereby informing how risks will be acted upon; (2) analyzing both practices together affords insight into the different ways in which they frame and act on risk (hence questioning the power relations underlying their use and development), but also to get directly at what inspired my questioning. In a context where social science, epigenetics, and environmental science research increasingly points to “environmental” pathways of disease causation, many chronic conditions are still highly individualized in public and health discourses: how are risks associated with food ingestion actually approached and worked upon in the apparatus of practices set into place in Canada to manage and control them?
The two sets of practices I focus on here test organic materialities to assess, prevent and control risks associated with food consumption on a longer-term basis: (1) blood testing to assess the risk of developing metabolic/chronic conditions such as diabetes, and (2) biomonitoring practices dedicated to controlling the levels of chemical contaminants in food and bodies. For food, I analyze the Canadian Total Diet Study (TDS), which annually monitors the fluctuation over time in levels of chemical contaminants in foods “that are typically consumed by Canadians” (Health Canada 2003). For bodies, I pay closer attention to various human body materials’ monitoring practices and programs operated mostly at the federal level (e.g., by the Bureau of Chemical Safety of Health Canada) and also at the provincial level (e.g., by the Institut national de santé publique du Québec, INSPQ, the Québec National Public Health Institute). These biomonitoring practices seek to identify levels of contaminants present in the bodies of the Canadian population.
I draw on Sharma's approach to temporalities as well as Foucaldian discourse analysis methods (Foucault 1971; 1975; 1980; Khan and MacEachen 2021) to understand what informs these practices and the temporalities they contribute to creating, considering and neglecting. This paper features excerpts from the interviews as well as elements from the corpus of materials gathered thanks to the information provided by people I interviewed. The corpus comprises a wide range of heterogenous elements such as clinical protocols, guidelines, governmental screening programs, screening reports, etc. It is important to note that, in line with my analytical framework, these excerpts are mobilized to help the reader “see” what constitutes these practices and what informs them, including the power relations that I discuss throughout the analysis mobilizing the relevant, critical literature.
In the two following sections, I analyze different “phases” of risk management and discuss the different temporalities taking shape throughout to illustrate how risk is approached and worked on. Ultimately, this allows me to pay closer attention to the power relations at play, notably which pathways and temporalities of disease are identified and/or neglected, thus contributing to the (re)production of (health, social, environmental) inequalities.
Diabetes Risk Management Testing Practices
The different “phases” of diabetes risk management analyzed in this section unfold in this manner: I first look at the screening for the condition (the very first step initiated by practitioners in Canada) which brings me to discuss the temporalities at play in epidemiology and in the creation of risk factors; I then turn my attention to diagnosing, which is the moment where blood test results are interpreted and the future is apprehended; I complete this section with the analysis of what follows a diagnostic, which leads to assessing and acting on identified causes of the disease.
Screening and Testing
When it comes to preventing and controlling the onset of diabetes, clinicians in Canada undertake the screening of a patient. This happens on many different occasions: in the context of a routine health checkup, “systematically when a patient comes to the emergency room” (Interview with family doctor), or when a patient requires a consultation with a doctor to investigate given symptoms (that may or may not be linked to diabetes). Screening includes invasive approaches (e.g., blood testing) and non-invasive ones (e.g., a risk calculator questionnaire) that can eventually lead to blood testing if the risk of developing the condition is considered high. Non-invasive screening is typically done using a risk calculator such as FINDRISC (Finnish Diabetes Risk Score) or CANRISK (Canadian Diabetes Risk Questionnaire) and includes screening of “risk factors” such as age, weight (obesity in particular), lifestyle habits, and the identification of an “at-risk” population: [FINDRISC reveals] if we need to do the blood test every year, either by capillary blood glucose or by blood testing, which is the measure of glycated hemoglobin.…Someone who doesn’t have risk factors, who is thin, who runs, who eats healthy, who doesn’t eat processed food or anything like that, or who doesn’t have precedents, that person would not score for[/with] a FINDRISC. (Interview with family doctor)
Individuals who “rank” as “High risk” or “Very high risk” (see Figure 1) of developing the condition are more likely to be tested or to be tested on a regular basis so the condition can be diagnosed and treated early on. This risk calculator's “scale” is based on the statistical prevalence determined by epidemiological studies. There is something quite circular at play here: epidemiology determines what is considered a risk factor, which determines that certain bodies more than others are to be tested; the results of testing practices subsequently inform epidemiological studies. 3 Epidemiology is constituted by statistics that draw from the aggregation of past incidence or prevalence, which is used to extrapolate and predict the future. While the materiality analyzed is that of an individual body, it is interpreted in light of populational/statistical studies, which means that future health, or health issues, is/are predicted based on this statistical data—it is not an actual calculation of that specific individual body's “chances” of developing a particular condition.
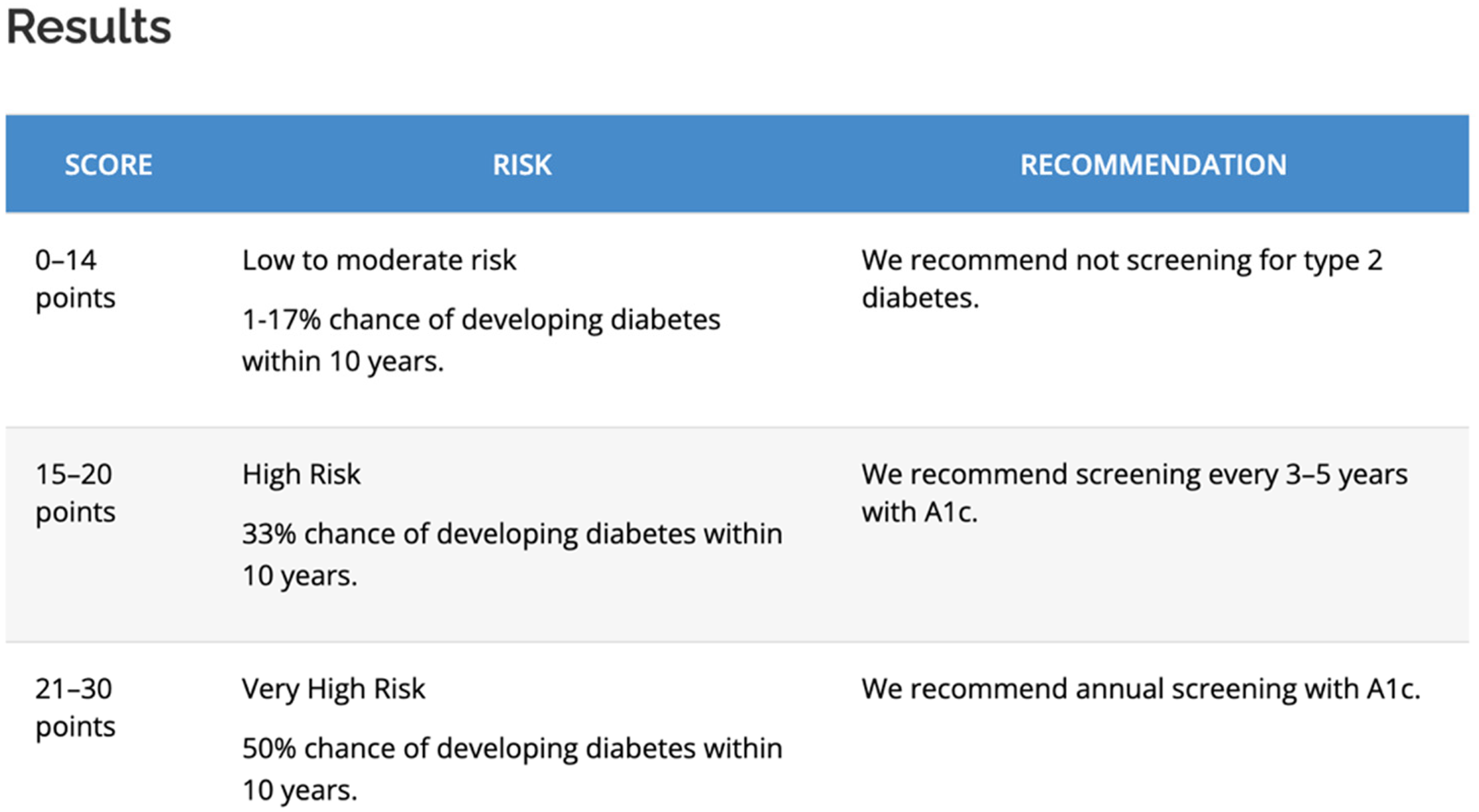
Screenshot of the FINDRISC score analysis grid used by clinicians to evaluate the need for more invasive screening practices including blood testing. https://canadiantaskforce.ca/tools-resources/type-2-diabetes-2/type-2-diabetes-clinician-findrisc/ Accessed 8 August 2024.
These statistically identified relations are used to infer causality, even when the latter cannot be directly demonstrated. As Giroux (2015) describes while discussing the origins of the risk factor approach in epidemiology in the context of cohort studies on cardiovascular disease (CVD), there is often conflation between the predictive value of the risk factor and the etiology of a condition. She provides the example of weight which, while “an important etiological factor in CVD” (Giroux 2015, 183), is almost useless as a predictor of the condition (see also Fletcher 2022). Through the projection of a future to come and its materialization in the present via the calculation of risk, the relation between illness and its causal pathways and incidence is “black boxed” (Berlivet 2005; Fletcher 2022; Giroux 2015; Rothstein 2003; Shim 2002). This tends to naturalize and render invisible the created nature of risk factors (Shim 2002), as well as the identified links of causality. With the risk factors approach, the (created, collective) past (in the form of statistical prevalence) is projected into the (individualized) future, determining body and health classification in the present. While the predicted future is drawn from the analysis of a multiplicity of bodies, the futures predicted and acted upon are very much individual(ized).
This contributes to individualizing the risk, framing it as belonging to the individual's body, according to “independent,” biological characteristics and/or behaviors such as eating habits (Fletcher 2022; Gravlee 2009; Hatch 2016; Lock and Nguyen 2018; Montoya 2011; Ng 2023; Rock 2003; Shim 2002). This has big implications for how disease causation pathways are investigated, as well as for the meanings attributed to bodily difference. The risk factor approach which informs the screening practices described above contributes to further increasing surveillance of certain bodies, including racialized (Hatch 2016; Keval 2016; Montoya 2011; Ng 2023; Rock 2003; Shim 2002) and fat ones (McCullough and Hardin 2013; McNaughton and Smith 2018), increasing their likelihood of becoming the object of the biomedical gaze (Giroux 2015; Shim 2002).
Diagnosing
When screening leads to blood testing or encompasses blood testing (e.g., invasive approaches), the interpretation of the blood results may lead to a diagnosis (see Figure 2). A diagnosis brings the patient under biomedical control and contributes to the re-classification of the individual from “normal”/“healthy” to “abnormal”/“unhealthy.”

Screenshot of an excerpt from the Diabetes Canada clinical guidelines featuring the diagnosis chart, used with permission. https://guidelines.diabetes.ca/cpgrefguide Accessed 2024-08-08.
Yet, Rock (2005, 479–480) argues that “diabetes is a disease not because of elevated blood glucose per se but because of the health risks this causes.” Sweeter blood does not mean “unhealthiness” in itself (or in the present) but it is approached as such because it leads to increased chances of developing microvascular (kidney damage, vision loss) and macrovascular complications in the future. Hence, while diabetes could be approached as a “risk factor” for the possible development of other health issues in the future, it became—like other risk factors, such as obesity to diabetes—a “disease in [its] own right” (Fletcher 2022, 62), acted upon in the present to prevent the development of possible health issues in the future. Epidemiology—including the statistical and biomedical technologies dedicated to measuring the sweetness of the blood—underpins not only the screening of the patient, but also the very “definition and classification of diabetes as a disease” (Rock 2003, 154).
The apprehension of future complications is what justifies the focus on screening and diagnosing the condition: 4 since it is assumed that there is a possibility of reverting possible future complications through early diagnosis and intervention in the present, screening for the condition becomes “ethically imperative” (Rock 2003, 140). As Adams, Murphy and Clarke put it, “Anticipation is not just betting on the future; it is a moral economy in which the future sets the conditions of possibility for action in the present, in which the future is inhabited in the present” (Adams, Murphy and Clarke 2009, 249). The blood test provides a sort of snapshot of the body's composition at that moment, which leads to assessing health status based on the apprehended future, and to further actions based on this interpretation. The diagnosis thus contributes to bringing the future in the present along with a certain linearity in the chain of “events” to be expected—the anticipated future sets the conditions of possibility for action in the present, hence shaping that possible future.
The focus on diagnosing with the hope of curtailing future health issues thus contributes to disregarding the “complex and interconnected ‘web’ of both causal and protective factors that together determine an individual's health status” (Shim 2002, 132) and biological state (e.g., sugar levels in the blood). In other words, the focus on extrapolating the future so as to act on the present through testing and diagnosis overlooks “the question of incidence” (Rock 2005, 479). This shifts the gaze away from the many factors that can contribute to elevated blood glucose, which may ironically also contribute to higher risk of health conditions (including some currently associated with elevated blood glucose), such as macrovascular ones, “even if blood glucose levels do not (yet) meet the diagnostic criteria for diabetes” (Rock 2005, 479–480). Ironically, the analysis of the bodily materials and the interpretation of its results might not lead to identifying the multiplicity of causes that might have led to elevated blood sugar, or that might NOT lead to elevated blood sugar but could still lead to higher risk of CVD.
Cause Assessment and Treatment
The results of blood testing practices are interpreted by clinicians in such a way that current health status is assessed, future health is predicted, and past behaviors that could have led to these results are investigated and targeted for change in the present. For instance, the most recent protocol for type 2 diabetes published by the Canadian Task Force on Preventive Health Care stresses that change of lifestyle behaviors is key to prevention and control in the context of pre-diabetes, and to treatment in the context of diagnosed diabetes: Individuals with a prediabetes diagnosis are strongly encouraged to undertake lifestyle modifications that include increasing physical activity, altering one's diet and weight loss.…The goals of treatment for type 2 diabetes are similar to prediabetes treatment, in that lifestyle modification is a cornerstone of management. (Sherifali 2011, 9–10)
From 2005 onwards, non-modifiable factors or causes of diabetes such as age, genetics, and family history have begun to slip into the background (and down the list), while those factors seen as modifiable lifestyle factors—notably weight, diet, and exercise—are given greater prominence. In short, there is a discernible shift in emphasis away from the non-modifiable to what is deemed open to change and intervention.
In that context, high blood sugar becomes loaded with moral judgments as it is framed, like obesity, as self-inflicted—the result of wholly changeable and risky lifestyle behaviours, factors, and choices” (McNaughton and Smith 2018, 123; see also Bennett 2019; Bombak et al. 2020; McNaughton 2013a, 2013b). Even though “[t]he exact etiology for diabetes remains undetermined” (Bombak et al. 2020) and there is currently no cure that exists for it, the measurement of the condition and of the “outcomes” of practices adopted to “fix it” (e.g., better glucose readings interpreted as the result of weight loss or change in lifestyle habits) make the condition appear not only manageable, but also preventable.
Emphasizing individual behavioral change as both a treatment for the condition (where the future is inflected by actions in the present) and its cause (a faulty past that must be “corrected”) puts the focus on individual responsibility. At the same time, it contributes to restraining the emergence and trajectory of the condition to the timeframe of the individual, which contributes to erasing the “profound social and economic inequalities underlying incidence of the disease” (Rock 2005, 467; see also Gálvez, Carney and Yates-Doerr 2020), as well as the possible environmental factors of disease causation. I am referring here to (sometimes intergenerational) trauma (e.g., Mendenhall 2020; Mulligan 2016; Lee, Tsenkova and Carr 2014); environmental racism, sugar ecologies and racial capitalism (Doucet-Battle 2021; García et al. 2023; Hatch, Sternlieb and Gordon 2019; Kaufman and Hajat 2021; Moran-Thomas 2019; Pulido 2000; Saxton 2021); past generations’ famine and malnutrition (Landecker 2011; Painter et al. 2008); stress and anxiety linked to systemic, chronic inequalities and systemic discriminations (Gálvez, Carney and Yates-Doerr 2020; Geronimus 2001; Gravlee 2009; Manderson and Warren 2016), to name a few examples mentioned in recent literature. These studies reveal how socio-environmental “events” materialize in the body through various biological and biochemical processes (i.e., epigenetics) following many different temporalities—timeframes, duration, timing, etc. that extend beyond the individual's will, power, and key to my analysis, lifetime. Diabetes prevention and control testing practices and the way their results are interpreted to assess cause and orient treatment do not acknowledge these processes.
In a context of increasing occurrence of diet-related chronic conditions such as diabetes, and of emerging studies pointing to the environmental pathways of disease causation, I turn my attention to the second set of practices used to control and prevent health risks associated with food consumption: those dedicated to controlling the risks associated with the ingestion of pesticides and chemical contaminants. How do the latter approach and work on risks, given the well-established permeability of bodies to their environments?
Pesticides/Chemical Contaminants Risk Management Practices
The following sub-sections are dedicated to analyzing different “phases” of pesticide and chemical contaminant risk management practices. Two main groups of practices are of concern here: (1) those that test levels of chemicals in foods in circulation in Canada; and (2) those that focus more on levels of chemicals contained in Canadian human bodies. After discussing the temporalities at play in testing practices of food and bodies, I pay closer attention to the temporalities at stake in the context of risk assessment, which leads me to pay attention to the evaluation of risk, exposure, and hazard, as well as the development of threshold limits. I analyze how risk is approached and worked on for this other type of testing, and how it may (re)produce health, social, environmental inequalities.
Food and Body Testing
Food Testing
Governmental authorities in Canada use various practices to monitor the presence and varying levels of contaminants in foods in circulation. The Bureau of Chemical Safety (BCS) is the entity within Health Canada responsible for setting threshold limits of chemical residue found in food, environments and bodies in the country. Once the BCS has established a threshold for a given contaminant, the Canadian Food Inspection Agency (CFIA) and the provincial body (in the case of Québec, the Québec Ministry of Agriculture, Fisheries and Food, MAPAQ) carry out regular monitoring activities to ensure compliance and watch trends and anomalies through food sample analyses. While these ongoing monitoring activities are conducted with the aim of ensuring compliance, the TDS conducted by the BCS, is meant to analyze long-term trends, identify possible new threats, and inform analyses of exposure to given contaminants through food consumption. The yearly TDS “provides estimate levels of exposure to chemicals that Canadians in different age-sex groups accumulate through the food supply” (Health Canada 2003) by testing a pre-determined selection of foods. BCS relies on surveys conducted with or by Statistics Canada (e.g., Canadian Community Health Survey) to select which foods to test, part of a “typical” Canadian diet. 5
The TDS is done to observe long-term trends (are chemicals found in food changing? Are the quantities detected varying?) and providing information for long-term overall exposure analyses. The TDS results are subsequently used in the evaluation of exposure and risk analysis as they provide information on what Canadians are exposed to through their diet. Timing matters here: this type of testing is not done ahead of consumption with the objective of informing eaters about what their food contains. The concern is with variations in contaminant amounts in respect to established thresholds, and the accumulation within the diet identified through populational surveys deemed “safe.” It thus follows a different timeframe than actual consumption: it is done so that authorities can better manage the average amount of contaminants consumed over the long term, not so that prevention occurs BEFORE a specific individual ingests at a given time. This somewhat “decontextualized” approached to food testing also contributes to putting the focus uniquely on a statistical average of food matters and what has permeated it, thereby neglecting the consideration for the actual situated body that ingests.
Moreover, there is “no systematic, comprehensive national surveillance of environmental exposures, food contaminants and risks” (Health Canada 2005; see complete list of limitations identified in food and surveillance activities in Canada), which means that not all food produced, put into circulation, and consumed in Canada is actually tested to evaluate the amount and type of chemical residue it may contain.
Importantly, only a pre-determined set of chemicals including “trace elements, pesticides, radionuclides, and a variety of other industrial chemicals” (Health Canada 2003) are monitored, which means that attention is directed at certain specific types and sources of exposure, while others may be neglected. What is investigated is limited to what is “known,” what is currently envisioned as within the realm of possibility, making it hard to act on forms of harm that are still invisible to our current methods and technologies (Murphy 2006; Saxton 2021; Vogel 2013) but might still materialize and accumulate in bodies following heterogenous timeframes and durations.
Human Biomonitoring
Various types of human biomonitoring activities are done both at the federal (BCS; Health Canada) and the provincial level (INSPQ), with the most comprehensive work being undertaken at the federal level. Large-scale biomonitoring surveys, such as the Canadian Health Measures Survey (CHMS), collect information on the general health and lifestyle habits of a sample of Canadians, with each testing cycle sampling “approximately 5,000 people over a two-year period” (Health Canada 2012) through interviews, physical measurements and biomonitoring. Samples of blood and urine are collected “to provide information on chronic and infectious diseases, nutrition and environmental exposures” (Health Canada 2012). Health Canada defines the human biomonitoring activities they undertake as follows: the measurement, in people, of a chemical or the products it makes when it breaks down. This measurement (called the level or concentration) is usually taken in blood and urine and sometimes in other tissues and fluids such as hair, nails and breast milk. The measurement indicates how much of a chemical is present in a person.…Large national or regional-scale biomonitoring surveys help measure our exposure to chemicals, and help us determine if the exposure is changing over time. (Health Canada 2010)
In the context of human biomonitoring activities in Canada, risk is not assessed with relation to an individual body that ingests or is about to ingest: it is assessed at a populational scale. In that respect, risk assessment is in some ways “disembodied,” in that risk is not evaluated for an actual body ingesting, an actual body whose cumulative exposure and actual level of contamination could be assessed, traced back and acted upon—an individual body situated and analyzed in context. Risk to hazards is, as Gabrielson (2016, 183) puts it, “abstract[ed] …through a universal language based upon a fictional body (the average body)” in that it is evaluated for no one's actual body, but rather for a fictional one created out of statistical averages used in risk assessment analysis. The potential health risks foreseen and acted upon in terms of levels of contaminants deemed “safe” to consume are those calculated for a statistical abstraction (the fictional body), not an actual body situated in time and space, with layers of accumulation intermingling with what is being consumed. Yet biomonitoring activities focused on observing variations in decontextualized “units” do not pay close attention to how new inputs intermingle in bodies that cumulate “toxic layers” and “chronic harms” through their lifetime and those of previous generations (Saxton 2021; Moran-Thomas 2019), further inflecting metabolisms and biological processes, and contributing to “recursive cascades” of chronicities in the making (Manderson and Warren 2016).
Risk Assessment—Exposure and the Evaluation of Risk/Hazard
As described above, the TDS and human biomonitoring activities are oriented toward observing varying amounts of chemicals in foods and body materialities, so as to make sure threshold limits are respected in the long run to curtail health risks associated with the ingestion of chemical contaminants. The establishment of maximum residue authorized in food is done with the evaluation of potential hazard (evaluation of toxicity and characterization of the dose–response of a chemical) along with the evaluation of exposure. Exposure calculations are done by combining data on presence and concentration of chemicals with data about what Canadians eat, or with data collected through human biomonitoring activities. For the evaluation of hazard, BCS relies on toxicological data in “existent literature and international review” as well as its own “in-house experiments” (Interview with BCS). Toxicological data is often produced following a model where chemicals are singled out and analyzed “out of context,” for instance in a laboratory (Liboiron 2021; Roberts 2021a; Saxton 2021). In other words, the possible harm caused by a chemical agent (or a few) on an organism is evaluated within a framework that is very linear in its approach to time, and very unidimensional and unitemporal in its approach to space. This framework falls short of taking into account the particularities and specificities of the organism in situated context and exposing the complexity of the effects of multiple chemicals intermingling in a situated body. When hazard is evaluated on the basis of epidemiological observations, links of causality need to be established via statistical calculations for risk managers to be able to act: only one study isn’t sufficient to influence decisions such as public policies with regard to contaminants. It requires more than one study, led in different contexts, and if we see that, finally, each time, it's the same link we observe, we end up…. It's a balance of probabilities. It's a kind of scientific consensus.…There are nine causality criteria that are recognized: Hill's criteria we call them in epidemiology. So, temporality: does the effect arrive after the exposure, that's obvious. Is it repeated? …Do we observe a dose–response relation? Do we see that the effect is stronger when there are more substantial exposures? Are there mechanisms of action that can explain the relationship we observe? …So when there are many elements together that converge, it's not only a correlation, it's a causal relation.…At a certain point, as an institution, we don’t wait to have all the answers to our questions to [act:] at a certain point, we have enough evidence to say that there is a reasonable presumption that there is a link between this exposure and this risk. (Interview with INSPQ researcher) The complex temporality of epigenetic processes also highlights the inadequacies of contemporaneous spatial measures of exposure).…Research has also revealed some epigenetic processes can be heritable, leading to intergenerational effects….Studies of epigenetic processes invite a relational perspective where the dynamics of time are privileged, and more so, epigenetic studies necessitate a lifecourse approach that pays attention to timing as well as social and historical context. (Prior, Manley and Sabel 2019, 537–538)
Health Canada acknowledges the limits of its biomonitoring activities when it comes to risk control and prevention, as the excerpt from the Health Canada website shows (see Figure 3). It acknowledges that it is often hard to know what are the sources or pathways of exposure because human biomonitoring analyzes bodies “after the fact,” once chemicals have already permeated the human body:
Likewise, the researchers I met fully acknowledge the limits of their practice and of the current state of knowledge that circumscribes what protective and legislative measures can be developed to manage and control risks associated with the ingestion of chemical contaminants. The acknowledgment of these limitations does not mean that the threshold approach to risk control is shattered; it only means that “precautionary measures” are used to “compensate” for uncertainty (e.g., lowering authorized thresholds). The use of precautionary measures allows for the apparatus to persist and continue to follow an approach to risk management that assumes that disease/harm follows linear, unitemporal causal pathways, and thus that it can be prevented or at least limited if acted upon with the appropriate set of measures (e.g., developing and enforcing threshold limits).

Screenshot from Health Canada's website, exposing some of the limitations of biomonitoring activities and the data it generates (Health Canada 2010).
Thinking Along the Different Temporalities of Risk Management in Food and Bodies Materiality Testing
I have described the different temporalities at play in diet-related risk management practices, to discuss how risk is approached and worked on. I have argued that temporalities in diabetes risk management practices are centered on the individual, insofar as risk is framed and analyzed within the timeframe of the individual—tied to their essentialized biological features or linked to individual behaviors or lifestyle “choices.” I have argued that the interpretation of blood test results evacuates the body's situated position and experience; what matters is to establish a diagnosis based on the analysis of the organic materiality tested and act on future health risks. The results of these testing practices eventually move from the clinical context to a larger, populational one (e.g., the Canadian Community Health Survey), feeding into further decontextualized epidemiological data, contributing to the invisibilization of the multiple, situated, socio-historical and environmental elements of context that shape bodily materialities and health (Roberts 2021b; Yates-Doerr 2020). How these testing practices are used and interpreted excludes consideration for processes that extend beyond an individual's lifetime, that can materialize in differentiated health. This contributes to furthering the imaginary that bodily processes are controlled by the individuals themselves, hence excluding disruptions caused by the broader environment that extends beyond the skin and willpower of the individual, and those that follow diverse temporalities.
I have highlighted how Canada/Québec's pesticides/chemical contaminants risk management apparatus is not aimed at preventing the penetration of contaminants in bodies, nor at providing insights into how new chemical contaminants intermingle with what has already permeated bodies and inflected their metabolism and bodily processes. Using previous STS work attending to the limits and issues of the threshold paradigm in environmental risk management, I discuss how setting threshold limits as the measure for safety is based on abstract calculations, not on the actual analysis of a situated body's risks, which tends “to…uproot the body from the social and geographical landscapes of exposure” (Murphy 2006 in Gabrielson 2016, 183). Discussing the temporalities in chemical contaminant risk management testing practices, I established that current methods and technologies cannot investigate or trace back the accumulation of a multiplicity and heterogeneity of exposures over time, for which moments of effectivity might also differ. Current risk assessment practices rely on the identification and assessment of specific, usually linear spatio-temporal “events” (e.g., an exposure to a specific contaminant that could result in a given health issue). My analysis reveals that the existing apparatus does not account for “the infrastructure of chemical relations that surround and make us [which] largely resides in the realm of the imperceptible” (Murphy 2017, 495–496) and for how the social and the biological constantly intermingle in continual flows of “accumulating exposure” (Prior, Manley and Sabel 2019, 531–532).
The apparatus of testing practices analyzed here, while informed by regimes of anticipation, forecloses the different temporalities considered and hence acted upon in the development of ill-health. As argued throughout the paper using works by medical anthropologists, STS scholars and environmental feminists among others, the temporalities at stake in the development of health issues are not restricted to what the individual alone can act on, notwithstanding the contemporary tendency to individualize risks in clinical settings (Borovoy and Hine 2008).
Because of its limits, which are intimately linked to the shortcomings of current scientific knowledge and available technologies, but also because of its very existence, the apparatus set into place to manage and control risks associated with food ingestion fails to address the many social, structural, and environmental markers of inequality that materialize in uneven health, following different timeframes, durations, and taking effect or affecting at different moments and according to different timings. In other words, the biotechnological apparatus meant to control risks associated with food ingestion currently fails to grasp and act on processes that unfold following many different temporalities, and sometimes through cascades of inter-actions (Green and Lynch 2022; Manderson and Jewett 2023; Manderson and Warren 2016). When not addressed, these can further increase the chance of developing ill health, uneven bodies becoming, following existing lines of oppression and marginalization, while many conditions are essentialized, individualized and stigmatized in current biomedicalized settings and culture (Gravlee 2009; Lock and Nguyen 2018).
The apprehension of the future through the practices analyzed in this article leaves little room to consider other temporalities than those framed within these practices, as well as little room to consider broader contexts. Yet the last decades of research in epigenetics, on the microbiome, and in endocrinology reveal how the body cannot and should not “be treated as separate[d] from environmental flows, a point that has been made by critics and scholars of biomedical paradigms” (Guthman and Mansfield 2015, 558).
Approaching health and illness as informed by collective processes, systems and structures (which is different again from identifying “social determinants of health,” see Yates-Doerr (2020)) could mean investigating collective pathways of disease causation, which would also mean developing new tools and methods of inquiry to better integrate consideration for contexts. 6 Such modes of knowledge production and mobilization could open the door for future avenues of inquiry, where chronic conditions could be looked at with a broader, more environmental, social and systemic lens, so that bodies would be re-situated within contexts that necessarily inform their materiality and health status. 7 Such work might lead to the realization that higher rates of chronic diseases among marginalized, racialized groups in Canada and in North America more generally (Bennett 2019; Czyzewski 2011; Dennis and Robin 2020; Doucet-Battle 2021; Hatch 2016; Ng 2023) have less to do with purported individual biological features, behaviors and foodscapes, and much more to do with environmental racism (e.g., Waldron 2018; Murphy 2017) and ongoing histories of discrimination and oppression, including colonialism (e.g., Dennis and Robin 2020). In other words, the accumulation of exposure to social and environmental inequities. This could also eventually lead to better interventions in clinical settings, where data generated via materiality testing could be approached as having a (hi)story, a situated context of emergence (which would also help with getting rid of the stigma surrounding diagnosis). Most importantly, this would also mean rerouting the amount of energy and resources put into investigating individual responsibility with regard to health and risk prevention, and investing instead in addressing environmental, collective pathways of disease causation, to tackle the social and environmental inequities that materialize in differentiated bodies and health.
Footnotes
Acknowledgements
I would like to thank Branwyn Poleykett, Emily Yates-Doerr and the colleagues from the Origin Stories of Harm workshop (held in Amsterdam in November 2022) for their careful engagement with previous versions of this paper, leading to a stronger, clearer and more significant argument. I would also like to thank Samuel Thulin for his thorough revision and commenting on the different versions of this piece, as well as the Science, Technology, & Human Values editorial team and the anonymous reviewers whose comments allowed me to improve this paper with each round of revisions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Social Sciences and Humanities Research Council of Canada.
