Abstract
This article explores the daily practices in a Danish cancer laboratory, where researchers use “personal organoids” as new translational models in the development of personalized medicine. Grown from metastatic cancer tissue of life-threateningly ill patients, personal organoids enable patient-specific drug screenings that may directly shape clinical decision-making and individual patient lives. Hereby, laboratory researchers are confronted with the patient as a person urgently dependent on their research results. We follow how the development of these potentially lifesaving personal models are enabled by “avoidance practices,” through which researchers actively sever models’ connections to the patients as biographical persons. Yet, this separation does not completely disentangle the organoids from the patients. To highlight how alterity and distance can be constitutive of valued social relations, we bring feminist science and technology studies critiques of care into conversation with anthropological accounts of kinship and personhood in South Pacific societies. Our analysis builds on these literatures, unfolding opportunities for laboratory researchers to practice patient personhood through disconnection or an “ethic of avoidance.” Our researcher interlocutors care about cancer patients by caring for organoids and thus exceed the dichotomy between person and thing and enact the patients as biological–biographical persons.
Keywords
Introduction
“I’ve had six biopsies taken. All of them from the liver, so I know exactly what it entails.” Jens, a computer programmer in his sixties, makes a painful grimace before he continues. “But, that’s what has made it possible to grow my cells [in the laboratory] and [for the oncologists] to say to me, ‘Well, we’ve tried to stuff these pills down [the petri dish] to your cells, and it looked really exciting. Let’s see if we can make a clinical treatment out of it…we can’t cure you, but we can try to keep you alive as long as possible.’”
While genetic standardization and large numbers are usually crucial to the value of preclinical models (Harrington 2013), the specificity and intimate bodily connection to one individual living patient are key to the direct clinical value of tumor organoid research (Xu et al. 2018). Personal organoids enable real-time drug screenings that may directly shape clinical decision-making and individual patient lives. When the organoids reach a sufficient size, they are treated with a variety of potentially effective drugs. If a drug inhibits organoid growth, it may also shrink the tumors in the patient. In this article, we investigate the daily work practices in which laboratory researchers grow and experiment with personal organoids and how researchers thereby become directly involved in the urgent search for personalized treatments for life-threateningly ill cancer patients. Specifically, we ask: How is the cancer patient as an embodied human person enacted and cared about in the exchanges of personal organoids between the cancer research laboratory and the experimental cancer clinic? Asking this question, we take analytical inspiration from anthropological studies that have illuminated the crucial role of everyday care practices in constituting embodied human persons (Kaufman and Morgan 2005; Taylor 2010) and generating new forms of morality (Mol 2008).
Empirically, our analysis is informed by an ethnographic study of a Preclinical Program for Cancer Precision Medicine, the PreCan program, in which scientists in a research laboratory at the University of Copenhagen collaborate closely with oncologists in a phase 1 clinic at the nearby Copenhagen University Hospital. By nurturing each patient’s genetically unique type of cancer in organoid structures, the interdisciplinary PreCan team hope to translate experimental laboratory results into effective personalization of cancer care. Organoid-based translation, however, comes with new responsibilities and ethical challenges (Boers et al. 2019; Green, Dam, and Svendsen 2022). Most research that identifies as translational is oriented toward future medical breakthroughs that may benefit an anonymous group of future patients. In contrast, the ambition of direct translation from bench to bedside implies that laboratory researchers must navigate “an actionable regime” constrained by the needs and temporality of the clinic (Nelson, Keating, and Alberto 2013). In this context, laboratory research in personalized medicine directly shapes the pathways of cancer patients who require immediate medical attention (Kerr et al. 2021; Bourret and Cambrosio 2019).
The PreCan researchers’ awareness of the clinical urgency of laboratory results motivated long working hours of tirelessly caring for the organoids. Growing cancer organoids and interpreting drug screenings are highly challenging experimental tasks, and unforeseen obstacles often led to frustrations and doubts. But the connection to individual patients and the possibility of making a difference in patient lives imbued the daily struggles of the laboratory team with a deep sense of meaningfulness. Therefore, we were surprised to learn that the laboratory researchers try to avoid direct interaction with these patients and, in some situations, also avoid patient-specific information. Where earlier research has demonstrated how disentangling human tissues from the human person works to depersonalize the tissue (Hoeyer 2013; Waldby and Mitchell 2006), our ethnographic material suggests that acts of avoidance confirm the connection between the organoids and the patients as embodied persons.
To clarify this connection, we draw on a key distinction by feminist science and technology studies (STS) scholars between “caring for” and “caring about” (Friese 2019; Puig de la Bellacasa 2017; Schrader 2015). Bringing feminist STS literature on the complexities of care in technoscience into conversation with anthropological theories of kinship and personhood, which position alterity and distance as constitutive features of valued social relations and personhood, we argue that the intimate practical labor of caring for organoids as biological extensions of patients is a way to care about cancer patients as biographical persons with little time left. Building on these joint insights, we conceptualize an “ethic of avoidance” by which our study unfolds a possibility for practicing patient personhood through disconnection. Avoidance practices help laboratory staff to overcome the distinction between person and thing, thus enabling them to care about patients as biological–biographical persons.
Care and Avoidance in Translational Research
In conversations and interviews, both oncologists and researchers in PreCan referred to the cancer organoids as a kind of avatar of the patient. These descriptions point to the inherent potential of personal organoids to blur the boundary between the model in the laboratory and the patient in the clinic. Similarly, STS scholars have previously shown that in everyday laboratory practices, research organisms designed to bridge laboratory research and clinical treatment hold the potential to vex distinctions between person and thing (Franklin 2005; Hoeyer 2013; Morgan 2009). Drawing upon the substantial feminist STS scholarship on care in science and clinical practice (Haraway 2008; Mol 2008; Puig de la Bellacasa 2017), social scientists focusing on animal modeling have documented how highly contingent care practices allow laboratory animals to act not only as biogenetic proxies for humans but also to be recognized as fellow sentient beings (Davies 2012; Friese and Latimer 2019; Svendsen and Koch 2013), with potential to step into the identities of the patients they model (Dam, Sangild, and Svendsen 2018; Svendsen 2021). Care in laboratory science, such scholars have shown, thus generates new forms of subjectivities that invigorate moral responses (Sharp 2019).
Much like animal researchers care for animal models in translational research, the PreCan researchers attended closely to the growth and “well-being” of the organoids. But although the organoids were treated as living entities of high scientific and clinical value, the PreCan researchers never allowed the personal organoids to acquire the identities of the patients with whom they shared cancer genetics. Rather, the researchers’ close daily involvement and care for the organoids seemed to be enabled by avoidance practices, through which researchers continuously worked to sever the connections between the organoids as biological representations of patient tumors and the biographical patient as a person grounded in time, place, and kinship (Svendsen 2015). The coexistence of involvement and avoidance, which constitutes the fulcrum of our further analysis, has previously been described by feminist STS scholars of care in technoscience. Foregrounding the “darker side” of care, they show that “care is a selective mode of attention” (Martin, Myers, and Viseu 2015, 3) that can result in neglecting of certain ways of being (Giraud 2019), but also that nonresponse and distance are sometimes acts of care (Martin, Myers, and Viseu 2015; Puig de la Bellacasa 2017).
Drawing on these insights, we suggest that in the context of personal cancer modeling, laboratory researchers’ way of avoiding interaction with patients is not a matter of neglect. Rather, it is a mutually constitutive mode of attention that confines life-threateningly ill patients to the clinical space, thus confirming their moral status as embodied human persons entitled to clinical care. This observation resonates with anthropological studies that have foregrounded boundary making as constitutive of intimate relations and the social formation of personhood (Candea 2010; Stasch 2009, 2011; Strathern 1988, 1996). For example, Stasch (2009) contends that intimate kinship relations among the Korowai people in Indonesia rely heavily on otherness and distance. Similarly, we argue that the PreCan researchers combine intimacy and distance in their struggle for a kind of “intimate alterity” (Stasch 2009, 117). In this context of personalized cancer medicine, they enact patient personhood by distancing the intimate laboratory practices of caring for organoids from the biographical human persons treated and cared for in the clinic.
Personalized Cancer Medicine in Denmark
In December 2016, the Danish Government launched the National Health Strategy entitled “Personalized Medicine for the Benefit of Patients” (Ministry of Health and Danish Regions 2016). In line with health policies in other countries, the Danish strategy stresses coordination and collaboration across health care and research as key to the vision of personalized medicine: to bring emerging knowledge of individual genetic variation into clinical application (Dickenson, van Beers, and Sterckx 2018). Oncology is a field where the clinical use of genomic tumor profiling is rapidly progressing (Schwartzberg et al. 2017; Kerr et al. 2021), and the Danish strategy for personalized medicine thus portrays oncology as a particularly promising field. Currently, however, most personalized cancer treatments in Denmark take place in one of the six experimental clinics in the country with the phase 1 clinic at Copenhagen University Hospital in the forefront.
This experimental clinic is organized as a professional phase 1 unit, which means that the clinic collaborates with pharmaceutical companies in early drug development. Motivated by the poor outcome of traditional phase 1 studies, the publicly financed “Genomic Project” was launched in 2013 as a personalized genomic screening program. The program investigates the clinical utility of molecular tumor profiling. All patients eligible for phase 1 trials are offered enrollment in the project, yet for about 80 percent of these patients, their genomic profile does not lead to participation in targeted trials (Tuxen et al. 2019). To increase the percentage of phase 1 patients offered targeted treatment, the genomic project has since 2017 been expanded to include a Preclinical Program for Cancer Precision Medicine (the PreCan program) funded by the state-financed Innovation Fund Denmark. Motivated by a common interest in helping cancer patients via genetic research, PreCan was designed as a collaboration between the head of the phase 1 unit and a leading professor of a research laboratory at the University of Copenhagen. The PreCan program requires two patient biopsies. One is used for an extra molecular analysis to explore potential for treatment matches between genetic mutations in the patient’s tumor and possible targeted treatments. Another biopsy is used for in vitro cultivation of tumor organoids. If the cells form into organoids, they can serve as the basis for patient-specific drug screenings, sparing vulnerable patients from receiving ineffective drugs (see Figure 1).
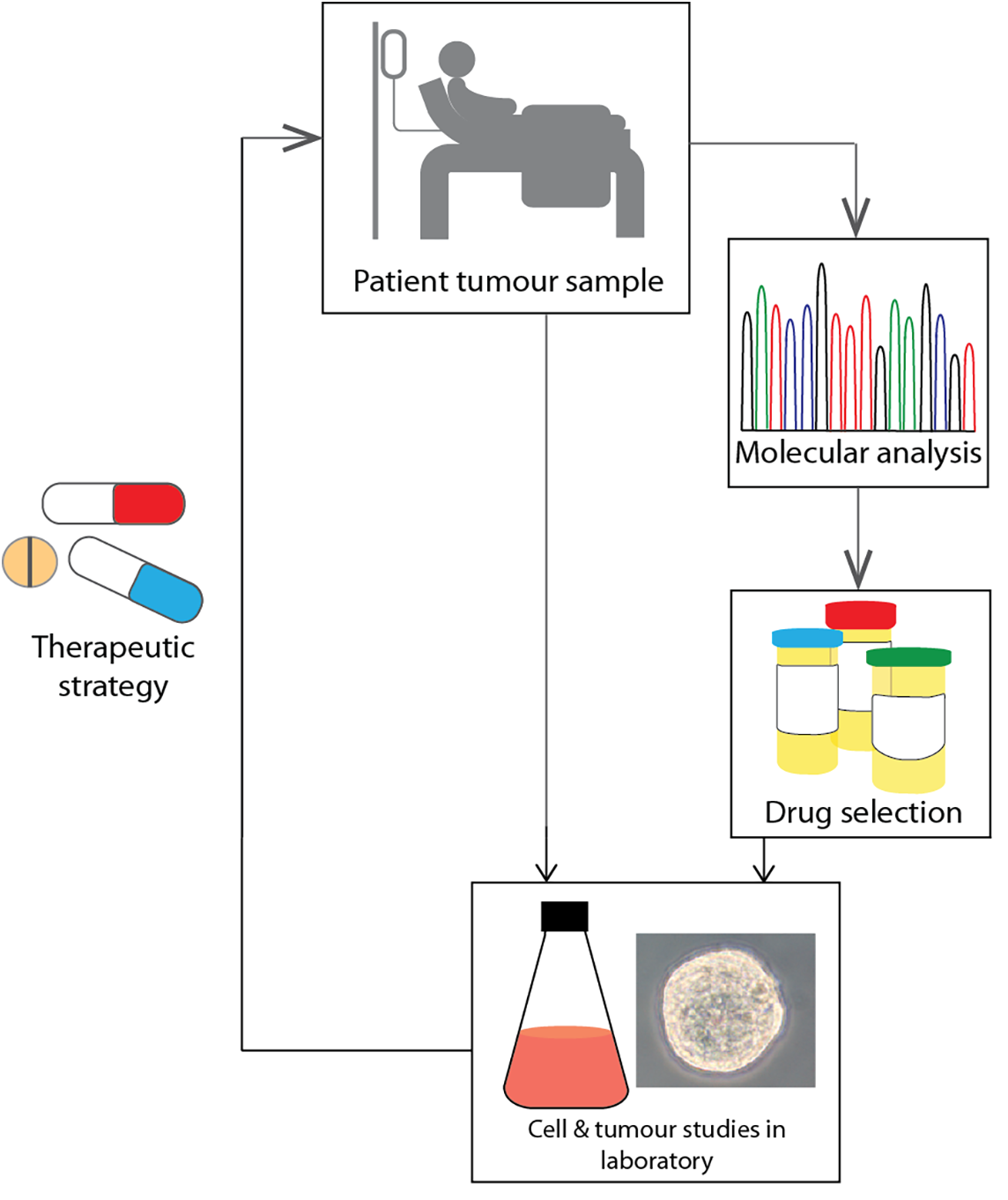
The ideal translational path from patient tumor samples to therapeutic strategy. Source: Image reprinted with permission from the PreCan team.
As part of the 2017 expansion, the developed organoids will eventually serve as the basis for a biobank for future drug testing in collaboration with medical companies. Yet at the time we followed the PreCan program, this had not been established; we thus focus on the role of organoids guiding personalized treatment selection.
The Ethnographic Study
This article draws on ethnographic fieldwork primarily conducted from December 2017 to June 2019. In the phase 1 clinic, first author, Mie S. Dam, observed initial consultations, where the aims of the Genomic Project and the PreCan project were explained to cancer patients by an oncologist. When a patient consented to participate, Mie went with the patient to have biopsies performed at the Hospital’s ultrasound department. As part of the fieldwork in the nearby cancer laboratory, Mie followed laboratory researchers as they collected biopsies. Mie spent full days observing how the laboratory team dissected the needle biopsies into tiny pieces, dissociated cancer cells from the tissue, and cultivated the cells in a 3D cell culture. Mie followed how the researchers nurtured and nourished the cells to grow into organoids, and how in vitro drug screenings were conducted to identify a personalized treatment strategy for a specific patient.
Together, Mie and second author, Sara Green, went to biweekly tumor board meetings, observing how an interdisciplinary team of oncologists, molecular biologists, pathologists, and a clinical geneticist deliberated to match patients to targeted treatments based on their genomic profiles. Furthermore, Mie attended monthly PreCan team meetings hosted by the phase 1 clinic. Here, the laboratory team informed the oncologists about the status of organoid drug screenings and discussed practical challenges and adjustments in laboratory procedures. The entire team then discussed potential new treatments for individual patients. In addition to participant observation, semi-structured interviews were conducted with clinical (five) and scientific collaborators (three) in PreCan and with patients (two) treated in the program during the period of fieldwork. Danish research ethics regulations do not require approval for qualitative interview and observation studies, but according to Danish law, our study has been registered by the Data Protection Authorities (Datatilsynet) and all informants have been anonymized.
In the following, we draw primarily on fieldwork from the cancer laboratory and situations where daily laboratory life included interactions and intersections with the phase 1 clinic. First, we investigate the way personal organoids as biological extensions of patient tumours motivated intimate care practices in the laboratory and facilitated a unique collaboration across the cancer laboratory and the experimental cancer clinic. Second, we focus on the ways collaborative work revolving around specific patients coexisted with avoidance practices that served to prevent direct interactions between the laboratory researchers and the patients as biographical persons.
Caring for Personal Organoids
One Wednesday morning in May 2019, Mie entered the cancer laboratory to observe postdoctoral researcher Helena who is the laboratory manager of the PreCan Program. Like many other mornings, Helena began her day at the bench preparing for the patient biopsies scheduled at the hospital later that morning. Mie did her best to follow Helena in the laboratory, where she alternated between adjusting laboratory apparatus, taking bottles of reagents from the fridge, and frequently checking her mobile phone. During this time, Helena quickly turned her head around explaining to Mie that she never knew exactly when they would call her from the hospital, but when they did, she had to leave immediately to collect the biopsies, and she wanted everything to be ready for the planned laboratory procedures. While checking her phone again, Helena added that she was always worried that the hospital would forget to call her in time to collect the freshly biopsied material. “If they don’t call me, then I am not the one to blame, if the organoids won’t grow,” she stated in a frustrated tone of voice, just before the phone rang. Helena and Mie dropped their lab coats and hurried to the hospital about 500 meters down the street.
For Helena and her three laboratory colleagues, moving quickly from the laboratory bench to the hospital ward and back again to speed up translation of personalized treatment strategies is far from just a political trope. Rather, it is very much a practical and material necessity imposed on them by the biological degeneration process that begins as soon as the needle biopsies have been performed and metastatic cancer tissue has left the patient’s body. In interviews and informal conversations, the laboratory team distinguished their work from standard laboratory work in being “so close to the patient in what you do” and they emphasized that being “helpful to someone” is “highly motivating” and “the driving force” to them. Yet, Helena’s busy mornings in the laboratory and her words about who to “blame if the organoids won’t grow” demonstrated that the embodied connection to individual patients also brought about new responsibilities, anxieties, and dilemmas. These were intensified by a combination of practical challenges, uncertainties about the value of the organoids as model systems, and the precarious situation of life-threateningly ill cancer patients. Comparing a biopsy to a “gunshot,” the laboratory researchers expressed that they were highly aware of the pain and risks this method involves for the patients and that this knowledge made them feel morally obliged to make the most of the limited tumor tissue they had at their disposal.
When Mie first entered the cancer laboratory, she had spent months of fieldwork in the phase 1 clinic following oncologists and patients in their persistent, but often unsuccessful, attempts to stop the uncontrolled growth of cancer. In the clinic, cancer cells were enacted as aggressive and proliferating intruders in patients’ bodies and lives. In contrast, the laboratory researchers enacted the cancer cells as fragile seeds, sown and nurtured to grow into (potentially lifesaving) preclinical models of specific patients. To increase the chance of these cancer cells growing into personal organoids, the researchers need as many viable cancer cells as possible. For this purpose, a quick and careful transfer from the human body to laboratory well plates is the critical first step. The cell cultures are then stored in an oxygenated refrigerator-like incubator with a temperature of precisely 37 degrees.
When Mie visited the laboratory for the very first time, in spring 2018, a postdoctoral researcher in the group named Kaya referred to the incubator as “almost like an entire hospital.” Kaya opened it to show Mie the rows of black well plates and added, “There is a patient in each plate.” Although the organoids look no different from ordinary cell cultures, this choice of terminology clearly articulates the direct connection to individual patients. As Helena later explained in an interview: It is there [on my mind] all the time, that there’s a patient at the other end, right? We can’t just throw the cells out if it doesn’t work, you know, you do everything to save whatever you can save.
Spending full days in the laboratory, Mie observed how the researchers experimented with new types of growth factors in the organoid culture. They split big organoids into new wells to optimize their growth conditions, tried out different kinds of antibiotics in the organoid media, and followed ever-stricter hygiene requirements to avoid contamination. Filling up stacks of notebooks, Helena tirelessly and meticulously noted down every change in procedures, describing in detail how she interpreted the reactions of organoids in every single plate. In this way, she strove to find her way in a trackless territory. Helena let the organoids guide her research through the way they appeared in the microscope or reacted to drug screenings and thereby developed her “capacity to read the other” (Clark 2007, 62). These skills were crucial to enable the fragile cancer cells to survive in the laboratory. As living biological extensions of specific patients, the organoids stand in for individual patients in drug screenings.
The researchers’ intimate care for the organoids was thus bound to the patients enrolled in PreCan as very real elements in the experimental system (see also Friese 2013). Kaya’s words about each plate containing a patient illustrate that in these practices, each plate of organoids appeared as “a microcosm of relations” and as “part of” the biographical person (Strathern 1988, 131) they had been derived from. Yet, surprisingly, the researchers explained that, at the bench, the organoids are “just the biological material” they are working with, adding that they do not think of a plate of organoids as “Mr. Jensen,” or even as belonging to Mr. Jensen. In this context, the organoids never take on the identity of the individual person they model. How can we make sense of how this contrasts with the researchers’ words that the “patient at the other end” is always on their minds?
Earlier STS scholars of biomedical research have demonstrated that human bodily material must become a disentangled resource before it enters experimental endeavors (Hoeyer 2013). This is perhaps most clearly seen in how making human embryos ethically available for biomedical exchange requires separation of stem cells from the human donor (Mitchell and Waldby 2006; Svendsen and Koch 2008). While the PreCan researchers’ referring to the personal organoids as “just biological material” similarly disentangles the human cancer tissue from the individual patient and turns it into an impersonal resource, this interpretation cannot fully account for the organoids’ continued connection to individual persons. As embodied human persons, the patients imbue the laboratory work with purpose and urgency. Their health status and treatment plans structure the priorities in the laboratory and the close collaborative relationship to the phase 1 oncologists. In this way, the PreCan project constitutes a bio-clinical borderland, in which laboratory research and clinical care are mutually constitutive; clinicians and researchers literally work with (different versions of) the same patient.
Yet over the course of fieldwork, we learnt that it was often of paramount importance to separate the needs of the patients “at the other end” and the “needs” of the organoids, as they do not always coincide. A key laboratory task was to do the “nitty gritty” work of optimizing organoid cultivation to identify the conditions that would allow organoids to grow into sufficiently large testing panels and—in principle—to live perpetually in the laboratory. But most patients are “close to the end of their days” and do not have time to wait. Therefore, the researchers must balance experimental requirements for sufficient time to deliver robust and reproducible data and clinical demands for fast test results. Ideally, growing more organoid material for each patient would allow the researchers to conduct multiple drug screenings to validate results through comparison of outcomes. However, the available time window to expand the organoid material is highly constrained by the risk that the patient will become too ill to receive any experimental treatment at all (see also Green, Dam, and Svendsen 2021, 2022). In a team meeting, Helena presented some issues with the interpretation of some drug screening results. She explained: I really care about the patients. It’s so sad when I learn that someone has died. Then I think that maybe I was one week too late with a drug screening, even if I know that one week is not what makes the difference when the patients are that sick, it still makes me think about it. And that’s also why I get so frustrated when the organoids won’t grow, or the drug screenings don’t work…. Because of the patients, I want to be as sure as I can possibly be [before I recommend a drug].
Feminist STS scholars have distinguished between the practices of “caring for” and “caring about” (Friese 2019; Puig de la Bellacasa 2017; Schrader 2015). While caring for entails the goal-oriented labor of caring for somebody or something (Mol 2008), caring about refers to an affective relation which does not have a specific subject of care (Schrader 2015). Schrader (2015, 668) elaborates that in “institutionalized modes of caring, caring for somebody in need is surely possible without caring about somebody” and that “[c]aring about someone does not have to…translate into a specific action.” As we shall see in the next part of the analysis, the PreCan researchers managed the uncertainty regarding the outcome of their careful and meticulous work in the laboratory by insisting that they do not want to care for patients. Rather, they care about patients as embodied biographical persons by intimately caring for organoids as “just biological” extensions of patients.
Caring about Patients through Avoidance Practices
When following Helena collecting biopsies, Mie noticed that the researchers ripped off the label taped to the lid of the Styrofoam box before leaving the ultrasound department to walk back to the laboratory. The label stated the name and ten-digit personal identification number (CPR number [Det Centrale Personregister]) containing information on the patient’s date of birth and gender. Similarly, back at the university, the researchers always headed directly to the bench to exchange the CPR number on the test tubes in the box with prepared labels stating a consecutive PreCan number. At first, Mie assumed that the practice of separating cancer tissue from personal identification number was a matter of careful administration, treating the biopsies with ethical caution, because they are genetically similar to the patients. Helena confirmed that she is always very careful not to keep such personal information together with patient tissue. Yet while Helena was a very conscious data manager, the separation between tissue and patient identity also helped her in other ways. Helena paused before adding: Actually, I feel better if I don’t look too much at the patient’s name and CPR number, when I collect the biopsies. I hate it when it’s a young woman my age or even younger. Then I just really hope that she doesn’t have any kids.
For instance, Helena recounted that even though the biopsies must be as fresh as possible, she does not want to arrive too early at the ultrasound department and risk encountering the patient in the ward. Helena explained that it would be “really awkward” for her to stand there next to a patient lying in a hospital bed; she “wouldn’t know what to say at all.” Helena also recounted that a nurse had once approached her because one of the patients had questions about “all the biopsies and everything.” “I instantly refused,” she recalled. Explaining her immediate reaction, she emphasized that it is not her responsibility to talk to the patients. At yet another occasion, Helena told Mie that one of the oncologists had warned her about the father of a young woman with aggressive and treatment-resistant cancer. In desperation, the father had contacted the phase 1 clinic several times a week ever since his daughter had been enrolled in the PreCan program to ask if they had found a treatment for her. The oncologist had told Helena that the father would probably “track down” Helena too and contact her. Helena ended her story by emphatically uttering: But I am not going to answer him. They [the oncologists] will have to take care of him. It is simply not possible for me to take care of everything.
Feminist STS critiques of care help illuminate what is at stake in these boundary making practices. In their introduction to a special issue on care in technoscience, Martin, Myers, and Viseu (2015) argue that the potentiality of care is grounded in a feminist ethic of “response-ability” (Haraway 2008; Schrader 2010), defined as “the practice of making oneself available to respond without knowing which phenomena will call upon one’s attention or what form the response should take” (Martin et al. 2015, 11). From this perspective, at first glance, it may seem paradoxical that the PreCan researchers, who respond attentively to the unforeseeable cancer organoids because they care profoundly about their corresponding patients, avoid responding when the patients call upon their attention as embodied human persons. Yet “[c]rucially, non-response is also a legitimate response…at times not reacting, not intervening, and not ‘casting one’s lot’ may be the most responsible action” (Martin, Myers, and Viseu 2015, 11). Accordingly, when asked to elaborate on her relationship to the patients, Helena explained that as she is not trained in communicating with life-threateningly ill patients in experimental treatment, she feared that her answers to patients inquiring about organoid-based test results would devastate already vulnerable patients. To her, nonresponse was thus also a matter of care and respect.
The head of the PreCan laboratory team, Professor Walters, provided further insights into the researcher’s practices of nonresponse. At a PreCan team meeting taking place in the phase 1 clinic, Mie shared her somewhat paradoxical observations with the entire team and asked what “keeping a distance from the patients” meant to the researchers. Professor Walters answered that “staying behind the curtains” is necessary for her dedicated team to “sleep at night” and “concentrate on their part of the job”, which is to carry on in the laboratory despite constant challenges and failures. In the following discussion, she reminded the team about one single situation, where postdoctoral researcher, Kaya, ended up next to a patient while the biopsies were performed. “And that was just so hard on her,” Professor Walters recounted. In an interview, Kaya likewise told Mie that she would not be able to cope with the challenges of her working life if she had to meet the patients every time. She could not forget about that single patient during her work in the laboratory, as “it just makes a huge difference when there is a face.” These reflections from Helena, Professor Walters, and Kaya illuminate that “staying behind the curtains” is not only a question of laboratory researchers avoiding their own discomfort. Rather, they spell out what we conceptualize as an “ethic of avoidance,” by which patients—and researchers—are mutually constituted by drawing boundaries of moral relations and responsibilities. Through their avoidance practices, the laboratory researchers are concomitantly protecting the patients from potentially harmful communication and preserving themselves to provide the best research to inform clinical patient care.
In this way, our analysis confirms that care is a mutually constitutive practice that also involves exclusions (Giraud 2019; Puig de la Bellacasa 2017). Yet while Giraud’s “ethics of exclusions” reminds us to pay attention to entities that are foreclosed when the focus is narrowly on relationality and entanglement, our empirical material flesh out the constitutive moral value of avoidance and disconnection. In PreCan, exclusion of the biographical patient from laboratory care practices does not result in neglect. Rather, it exemplifies that “[c]are is not about fusion: it can be about the right distance” (Puig de la Bellacasa 2017, 5). In anthropology, distance and avoidance has also been theorized as productive in the social formation of persons and valued social relations (Candea 2010; Stasch 2009, 2011; Strathern 1988, 1996). For instance, Stasch’s (2009, 2011) ethnography about kinship relations among the Korowai people in Indonesia vividly unfolds the moral and relational value of avoidance practices. He observes that “often a relation is centrally defined by a mismatched joining of intimacy in one medium and otherness in a different medium” (Stasch 2009, 17). This can help us conceptualize the apparently contrasting ways laboratory researchers engage intimately with organoids in the laboratory, while practicing their engagement with embodied patients in the clinic through avoidance and exclusion.
Among the Korowai, mothers-in-law and sons-in-law, who often visit each other’s houses, must stay out of each other’s sight, and avoid bodily contact, just as they are not allowed to address each other directly. Such avoidance is a practice of discretion and carefulness toward a social situation that would otherwise be uncomfortable. Thus, upholding the avoidance imperative, mother-in-law and son-in-law become “a pair” that in performing this “reciprocal engagement” expresses the closeness and moral value of their relational bond (Stasch 2009, 87). Just as Korowai in-laws often visit each other’s houses and are thus not completely separated in space, the laboratory researchers regularly visit the hospital, which we may think of as the “house” of the patients. This means that keeping the patients at a distance is not clear-cut, as it may be for researchers working with residual human tissues for the purpose of future medical progress. In contrast, refraining from face-to-face encounters and direct verbal or written communication requires continuous attention. Under such circumstances, “[a]voidance indexes attentive restraint toward another, and thus creates intensified relatedness through that restraint” (Stasch 2011, 102).
We interpret the researchers’ continuous attention to upholding avoidance—like in Korowai kinship relations—as a practice that confirms the moral value of the patients as biographical persons as well as their continued connection to the organoids. A common Korowai explanation for such avoidance practices is that mothers-in-law and sons-in-law are “scared, panicked, uncomfortable” (Stasch 2011, 87) in each other’s presence. Likewise, we may interpret the researchers’ practices of “staying behind the curtains” as a way to protect themselves from being “scared, panicked, uncomfortable” at the personal tragedy behind each PreCan number, which they are not trained to cope with. If the researchers met with the patients each time, we may contemplate that the patients’ faces would occupy the researchers’ minds and extend the uncanny feelings of “intellectual uncertainty…something one does not know one’s way about” (Freud 1919, quoted in Friese 2013, 134) into the laboratory space and make them unable to “concentrate on their part of the job.”
When in June 2019, master student Louisa showed Mie how to change the organoid-media in all well plates, she explained that it is important for her “not to have a face on some of the organoids in order to hope equally for each organoid” and thus stay equally careful and concentrated during the demanding and sometimes monotonous laboratory work. Louisa’s interpretation of the productive aspects of distance resonates with Candea’s (2010) ethnography of meerkat–researcher relationships in the Kalahari. Here, abstaining from direct interaction with individual meerkats was a hard-earned form of engagement that enabled the researchers to form a caring and impartial relationship to the entire meerkat population. In combining intimacy and distance, the PreCan researchers likewise struggled for a kind of “intimate alterity” (Stasch 2009, 117) in which intimately caring for all organoids as biological extensions of individual patients is a way to care about all cancer patients, while keeping the biographical human person contained and cared for in the clinic, by oncologists trained in caring for patients in crisis. When the researchers depersonalize organoids by avoiding connections to patient information and interactions, we should not interpret their actions as a reduction of organoids to a thing or as a devaluation of the patient as person. Rather, the PreCan researchers’ avoidance practices make patient and organoid become “a pair” (Stasch 2009, 87) that exceeds the distinction between person and thing and confirms the moral status of the relation between organoid and biological–biographical person.
Conclusion
In personalized oncology, laboratory researchers and clinical oncologists have developed patient-specific tumor organoids as new translational models to bridge the gap between standardized cell and animal models in the laboratory and the variation of individual patients in the clinic. Such personal models rest on complex laboratory techniques and on closer alignment of the daily work practices in the laboratory and the clinic. In this paper, we have followed the Danish preclinical research program PreCan and explored how personal organoids unify laboratory researchers and clinical oncologists in caring about the same life-threateningly ill cancer patients. As a kind of patient avatars, personal organoids hold an intimate bodily connection to specific patients and introduce new hopes and treatment possibilities, not only for an anonymous group of future patients but also for individual living cancer patients. Yet our ethnographic study has demonstrated that these new translational models also come with new epistemic uncertainties and moral responsibilities for laboratory researchers, who become directly involved in the urgent surge for personalized cancer treatments.
We have shown that the possibility for real-time translation across laboratory and clinic situates organoid researchers in a bio-clinical borderland in which they must simultaneously attend to the “needs” of the organoids and to the clinical situation of the patients. In this balancing, the researchers acted in seemingly contrasting ways that shed new light on the complexity of practicing care and personhood in personalized cancer research. While the researchers cared intimately and tirelessly for the organoids in the laboratory because they perceived them as biological extensions of living patients in urgent need for treatments, they never allowed the organoids to step into the identities of the patients they modelled. On the contrary, the researchers refrained from personal interactions with these patients, so as not to imagine them as biographical persons grounded in time, space, and kinship.
Bringing feminist STS distinctions between caring for and caring about into conversation with anthropological theories of kinship, which position alterity and distance as constitutive of valued social relations and personhood, we have conceptualized an “ethic of avoidance.” With this concept, avoidance practices are brought into relief as an important aspect of the laboratory team’s care about patient personhood. By upholding distance from the patients, as embodied human persons with little time left, the researchers could better manage epistemic uncertainties and unforeseen obstacles, and care equally and persistently for the organoids in the laboratory. Yet, awareness of the individual patients “at the other end” was ever-present. In order to deliver reliable test results to guide clinical decision-making, the researchers continuously had to plan and align experimental work in the laboratory to the clinical situation of the patients, just as they regularly visited the hospital to collect biopsies or attend meetings with clinical collaborators. In real-time translational research, the severance of connections between laboratory organisms as biological models and the biographical human patients thus requires continuous attention. Therefore, in this context, avoidance practices are not a matter of neglect but a form of caring engagement that constitutes patients as integrated biological–biographical persons. Rather than opting for a dichotomous division between person (the patient) and thing (the organoid), the researchers mobilized an intimate alterity that enabled them to care about patients by caring for organoids. Hereby our ethnography of personal cancer models illuminates the ethical importance and the constitutive value of contesting certain relations in care and in the formation of patient personhood.
Footnotes
Acknowledgements
We wish to thank the staff in the cancer laboratory and in the experimental cancer clinic who kindly let us into their daily work and took time to discuss their experiences of translational cancer research with us. Our sincere thank-you also goes to the patients who allowed us to follow them into the intimate space of experimental cancer treatment. We thank Tyler Zoanni and Mette N. Svendsen for insightful comments and guidance. We also want to thank Laura E. Navne, Iben M. Gjødsbøl, Clémence Pinel, Anja B. Jensen, Olivia Spalletta, and the entire MeInWe team for their comments on earlier versions of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research project “Personalized Medicine in the Welfare State” (MeInWe) is supported by The Carlsberg Foundation (Semper Ardens grant CF17-0016). Green has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 101006012.
