Abstract
Background: Mitochondrial disease (MD) is a group of disorders caused by dysfunctional mitochondria, the organelles that generate energy for the cell. Malnutrition in patients with MD may lead to increased mitochondrial dysfunction, which may enhance already existing symptoms. The aim of this study was to investigate whether patients with MD have an insufficient or unbalanced food intake and to establish which nutrients and product groups are particularly compromised in this patient group. Methods: In this observational, cross-sectional, retrospective study, sixty 3-day nutrition diaries of adult patients with MD were analyzed and compared with the Dutch recommended daily allowance and the Dutch National Food Consumption Survey (DNFCS). Results: The intake of all macronutrients and micronutrients of patients with MD was significantly different from Dutch recommended daily allowance values with the exception of fat and iron. In particular, protein and calcium intake in patients with MD was significantly lower when compared with the DNFCS. Interindividual differences were high. Also, intake of fiber, sugars, saturated fat, and vitamin D differed from recommendations for the overall population. In comparison with DNFCS, the intake of dairy products and drinks was significant lower in patients. Conclusions: Our study demonstrates that many patients with MD have an inadequate diet. Specifically, intake of protein, calcium, dairy products, and fluids were low. Overall, eating a healthy diet seems as difficult for patients with MD as for the general population. Since interindividual differences are high, individual diet counseling is recommended for all adult patients with MD.
Clinical Relevancy Statement
For patients with mitochondrial disease (MD), a healthy and adequate diet is of utmost importance because of the increased risk for malnutrition, gastrointestinal and metabolic problems, muscle weakness, and fatigue. We found that patients with MD have an inadequate diet, rendering them at risk for malnutrition. These findings are clinically relevant for clinicians and dieticians because a cure for MDs is lacking and treatment remains symptomatic. Since malnutrition can be treated, with the promise to improve quality of life, offering individual diet counseling seems a promising treatment option for adult patients with MD.
Introduction
Mitochondrial disease (MD) is a group of genetic disorders caused by dysfunctional mitochondria: the organelles that generate energy for the cells. Mitochondria are present in every cell of the human body—except red blood cells—and convert the energy of nutrients into ATP, which powers most cell functions. Tissues that require major amounts of energy, such as the brain, skeletal muscle, and heart, are most commonly and severely affected.
MD is a heterogeneous, multisystemic, and progressive disease. The heterogeneity of MD is caused by dualistic genome control by mitochondrial DNA and nuclear DNA. The m3243A>G mutation is the most frequently reported genotype. MD may present from infancy to adulthood. Patients can be strongly affected and may die at childhood or present with a milder disease phenotype. Maternally inherited diabetes and deafness, mitochondrial myopathy, and chronic progressive external ophthalmoplegia are the most frequently reported phenotypes in adult patients. 1
Patients with MD frequently suffer from conditions that may affect nutritional intake and metabolism, such as gastrointestinal problems, dysphagia, fatigue, or diabetes. Accordingly, in a previous study in patients with MD caused by the m3243A>G mutation, we showed that 89% of patients suffer from chronic gastrointestinal manifestations, mainly constipation, bloating, and flatulence. These patients have a higher risk of malnutrition, and the severity of the gastrointestinal problems, as well as that of the overall disease, correlated with a lower body mass index (BMI). 2
Vice versa, it is known from other diseases, such as anorexia 3 or cancer cachexia,4,5 that malnutrition per se causes secondary mitochondrial dysfunction. From data of children with mitochondrial disorders, it was suggested that by improving the nutrition status in patients with MD, ATP production may be improved. 6 MD cannot be cured, and treatment remains symptomatic with the aim to improve quality of life. However, malnutrition can be treated, with the promise to improve quality of life, as has been shown in other diseases, such as head and neck cancer 7 and chronic obstructive pulmonary disease. 8
Vitamins B and D, iron, and calcium are relevant micronutrients in mitochondrial disorders because deficiencies in these micronutrients may induce frequently observed complaints in these patients, such as fatigue and muscle pains. Also, these micronutrients play an important role in mitochondrial function.9,10 Although micronutrient supplementation has been often discussed as a possible treatment strategy in patients with MD, the effects are as yet not proven.9,11 In addition, data on intake in patients with MD of these micronutrients are lacking.
Patients with MD often report that they lack energy to prepare and consume healthy meals. This may lead to an imbalanced diet with high intake of snacks and sugar. This is a concern because snacks and sugar have low nutritional value, which means that patients with a normal or high BMI may be malnourished because of inadequate protein and/or micronutrient intake.
Before designing a proper dietary intervention for patients with MD, the malnutrition problem should be analyzed in detail. Disease-related malnutrition, according to Stratton, 12 involves a shortage and/or disbalance of nutrient intake, leading to negative changes in body composition, functioning, and clinical outcome. Malnutrition is a complex problem, and according to the definition of Stratton, undernutrition as well as obesity could be a status of present malnutrition, and both are known to occur in patients with MD.
In the present study, we focused on the definition of malnutrition 12 (ie, nutrient intake). We investigated whether patients with MD have an insufficient and/or unbalanced food intake. Furthermore, we analyzed which food groups and nutrients are particularly compromised in this patient group. The intake of the patients with MD was compared not only with the recommended daily allowances but also with the Dutch National Food Consumption Survey (DNFCS). 13
Materials and Methods
In this observational, cross-sectional, retrospective study, nutrition diaries of 80 adult patients (>18 years of age) with MD were collected between the start of 2013 and the end of 2016. All patients were treated at the national expertise center for mitochondrial disorders: Radboud Center for Mitochondrial Medicine (www.RCMM.info). Nutrition diaries were included from patients who gave written informed consent. Patients were excluded when the MD was not genetically proven and when food diaries were not completed for a minimum of 3 days. The study was approved by the medical Ethics Committee of the Arnhem and Nijmegen region.
Patients completed the nutrition diaries after reading written instructions from a preprinted form. They were instructed to register, very accurately, all foods and drinks that were consumed during 3 days—preferably, 2 weekdays and 1 weekend day. Patients were asked for clarification of their nutritional intake in case the diary was not sufficiently clear. In unresolved cases, portion sizes were estimated with the Dutch food measurements codebook. 14 Calculation procedures, including the estimations, were documented in a log for consistency.
Anthropometrics were measured at the outpatient clinic. Height was measured in a standing position with a calibrated ruler in centimeters (Seca, Hamburg, Germany). Weight was measured in kilograms on a calibrated electronic scale (Seca). Information about genotype, phenotype, and presence of diabetics was collected from the electronic patient files.
The nutrition diaries were analyzed according to a standardized procedure. Nutritional intake was calculated twice by 2 independent research dieticians using Foodfigures software based on the Dutch food file 2013 database (NEVO, Bilthoven, The Netherlands). No differences were observed between analyses.
Foodfigures analyses were made separately on a nutrient level and on a food product group level. Total daily protein, fat, saturated fat, and carbohydrate were calculated (g/d). Relevant micronutrients 9 were calculated only if they could be reliably obtained and calculated through a nutrition diary. This was the case for vitamin D, calcium, vitamin B12, and iron. The calculations were solely based on the nutrition diaries, including enteral nutrition but without the micronutrient supplements. The micronutrient supplements were being checked and reported separately.
To determine if the food intake of patients was sufficient, it was compared with various references. The energy intake was compared with the patient’s individual estimated requirement. The energy requirement was calculated with the Harris and Benedict equation 15 with a standardized low activity factor of 40%, based on previous actometer measurements in this patient population. 16 Protein intake was compared with the standard recommendation for healthy adults of 0.8 g/kg. For protein, we calculated the requirements with body mass, and a correction for weight was applied in patients with a BMI outside the healthy range. Weight was corrected to a BMI of 20 kg/m2 for patients with a BMI <20 kg/m2. For a BMI >30 kg/m2, weight was corrected to a BMI of 27.5 kg/m2. 17 The fiber intake was compared with the Dutch adult fiber guidelines of 3.4 g/MJ. The intake of carbohydrate and fat was compared with standards of the Health Council of The Netherlands (carbohydrate energy, 40%; fat energy, 20%–40%). For the micronutrients and product groups, the Dutch recommended daily allowance (DRDA) was used as reference. Finally, the intake of nutrients and product groups was compared with the DNFCS. 13 Because references differed with age and sex, absolute numbers were converted into percentages of the reference values, in which 100% equals the recommendation. For fat, the upper limit of the healthy range (energy, 40%) was set equal to 100%.
Descriptive statistics were used to present the nutrient intake. Data were checked for normality, and means were presented with their standard deviations or medians and ranges. In case of normal distribution, 1-sample t test was used to calculate the significance of the difference between the mean intake of the patients with MD and the reference data. Correction for multiple testing was performed with the Bonferroni method, and only results with a P < .0025 were considered significant. Pearson correlation analysis was performed between food groups and relevant nutrients that significantly differed from the DNFCS.
Results
Research Population
Eighty nutrition diaries were collected from the population of Dutch adult patients with MD from the Radboud Center for Mitochondrial Medicine. Twenty reports were excluded (see Figure 1). The study population available for data analysis consisted of 60 nutrition diaries from 60 patients with MD.
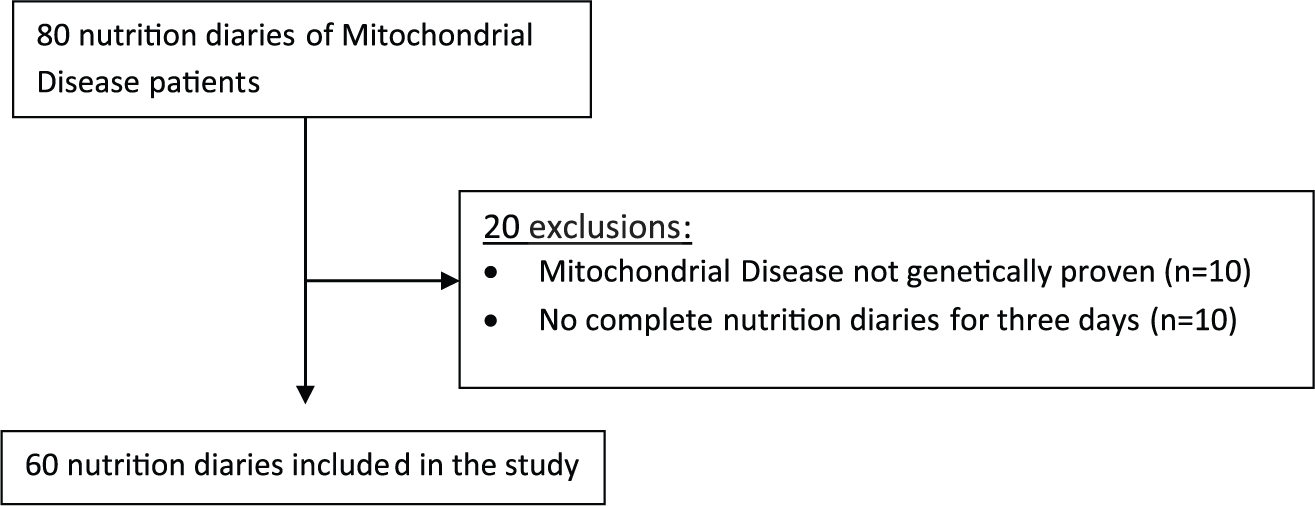
Patient report exclusion schedule.
Patients Characteristics
Table 1 shows the characteristics of the MD study population. Nutrition diaries from 19 men (32%) and 41 women (68%), with a mean age of 49.2 ± 13.1 years and a mean BMI of 24.3 ± 4.6 kg/m2, were included in the study. The most frequent MD phenotypes were mitochondrial myopathy (35%), maternally inherited diabetes and deafness (30%), and chronic progressive external ophthalmoplegia (30%). Thirty-three percent of patients had diabetes, of which 80% were insulin dependent.
Characteristics of Patients With Mitochondrial Disease.
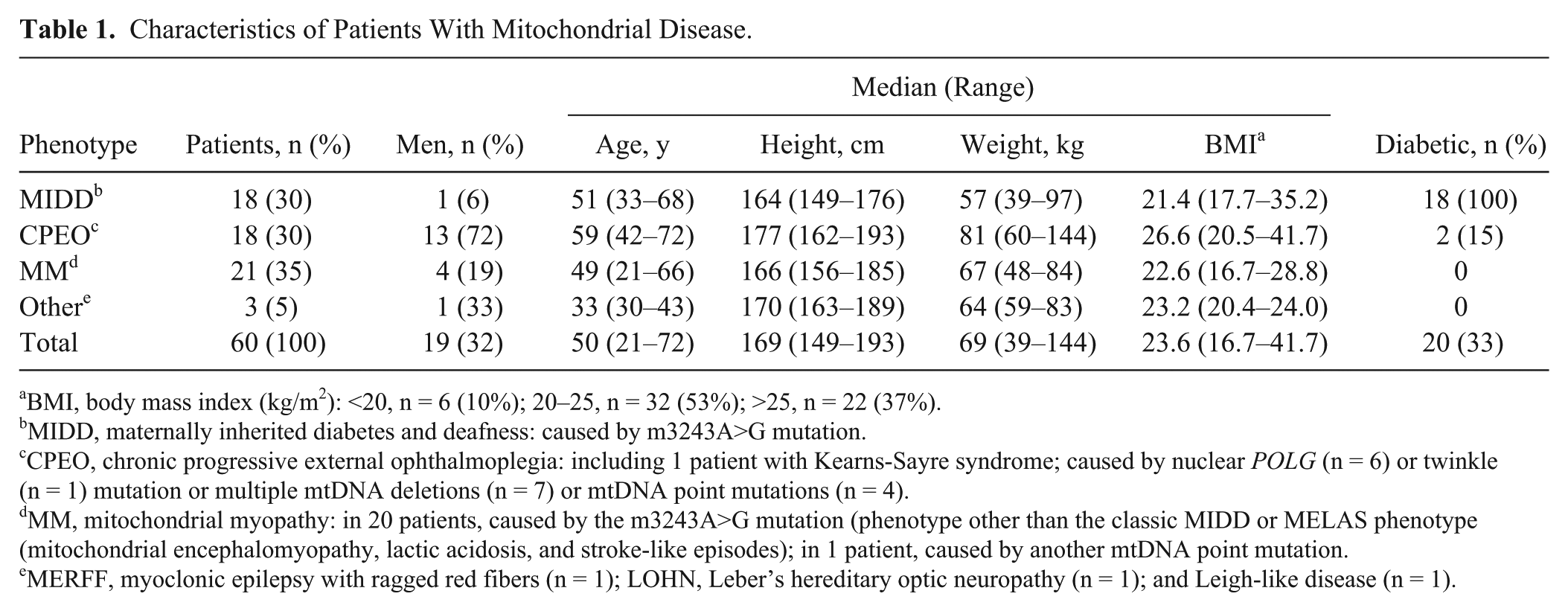
BMI, body mass index (kg/m2): <20, n = 6 (10%); 20–25, n = 32 (53%); >25, n = 22 (37%).
MIDD, maternally inherited diabetes and deafness: caused by m3243A>G mutation.
CPEO, chronic progressive external ophthalmoplegia: including 1 patient with Kearns-Sayre syndrome; caused by nuclear POLG (n = 6) or twinkle (n = 1) mutation or multiple mtDNA deletions (n = 7) or mtDNA point mutations (n = 4).
MM, mitochondrial myopathy: in 20 patients, caused by the m3243A>G mutation (phenotype other than the classic MIDD or MELAS phenotype (mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes); in 1 patient, caused by another mtDNA point mutation.
MERFF, myoclonic epilepsy with ragged red fibers (n = 1); LOHN, Leber’s hereditary optic neuropathy (n = 1); and Leigh-like disease (n = 1).
Macronutrients and Micronutrients
Figure 2 shows the mean intake of micronutrients and macronutrients in relation to reference values with corresponding standard deviations, as compared with the average intake of the DNFCS. The mean macronutrient and micronutrient intake of the patients with MD was different from reference values (P < .0025) with the exception of fat and iron. While the intakes of energy, fiber, vitamin D, and calcium were lower than the reference data, the intakes of carbohydrate, protein, saturated fat, and vitamin B12 were higher than recommendations. Variation in nutritional intake was high, especially in vitamin B12.
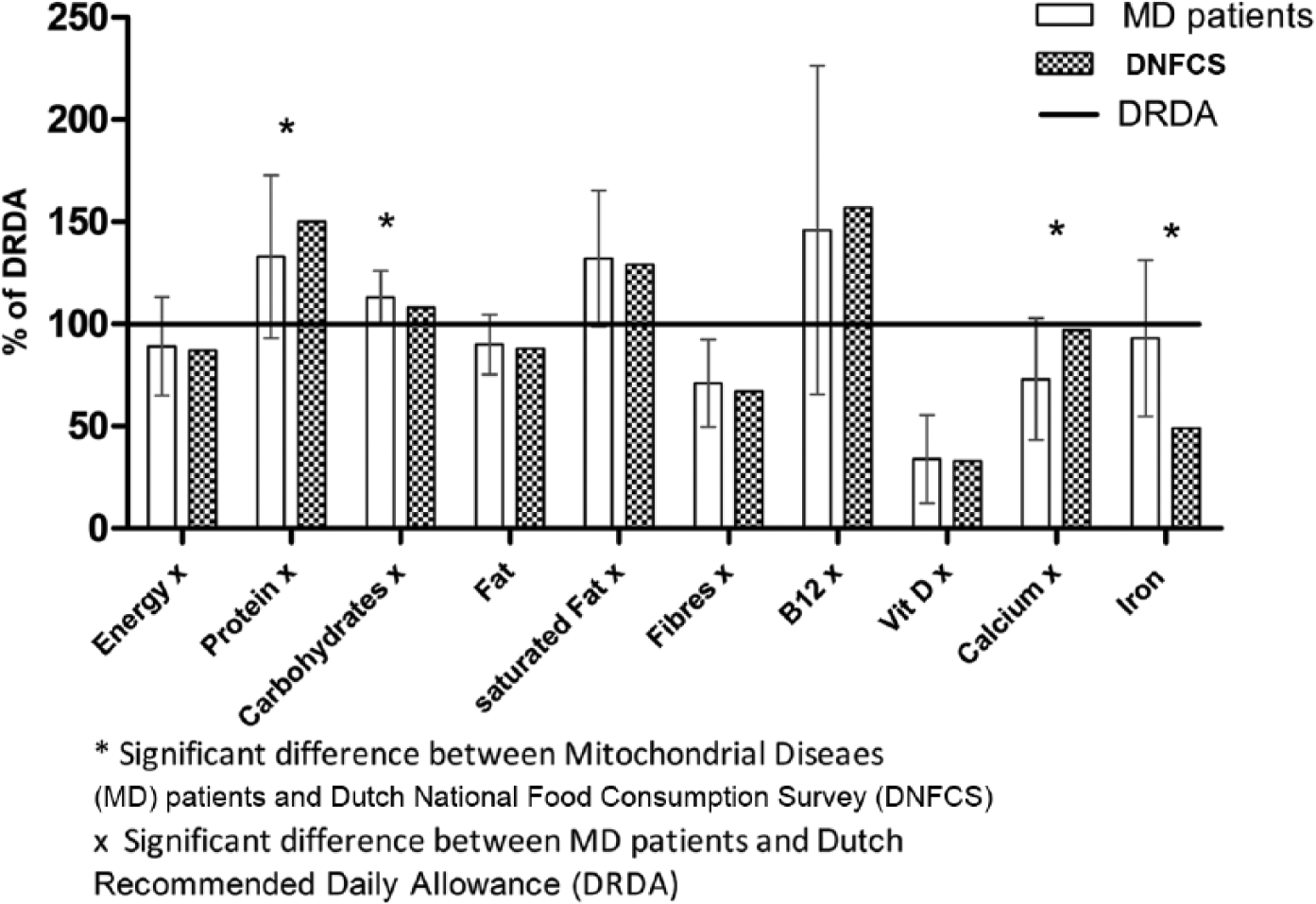
Intake of macronutrients and micronutrients in patients with MD and DNFCS, expressed as percentage from DRDA. Values are presented as mean ± SD.
Protein intake was higher than the DRDA of 0.8 g/kg. The mean protein intake in the patients with MD was 1.1 ± 0.34 g/kg. As compared with the DNFCS, the intake of protein and calcium was significantly lower among patients with MD. In contrast, the intake of carbohydrates and iron was significantly higher in this group.
Product Groups
Figure 3 demonstrates the product group intake relative to the reference values and compared with the average intake of the DNFCS.
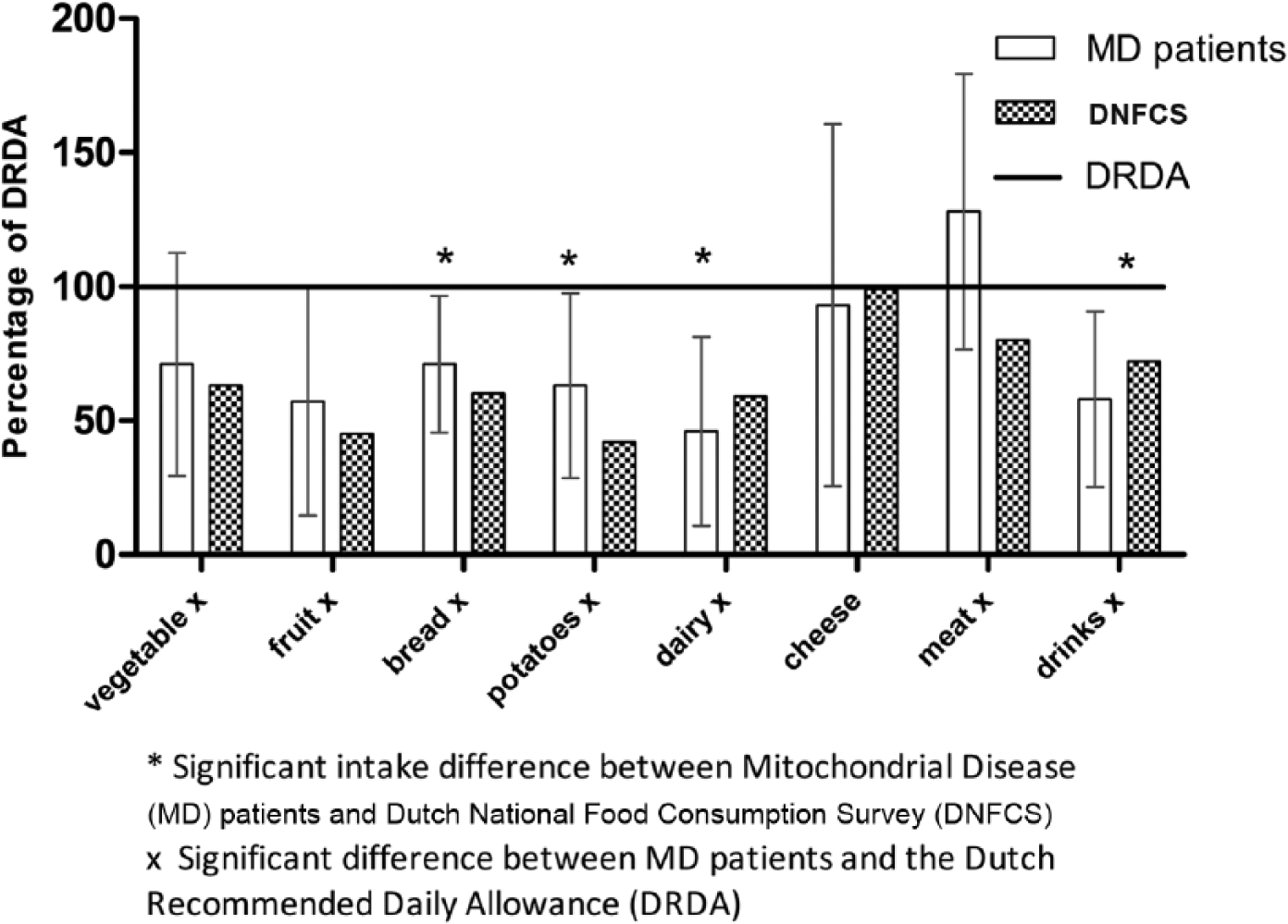
Product group intake in patients with MD and DNFCS, expressed as percentage from DRDA. Values are presented as mean ± SD.
The intake of food per product groups of the patients with MD was significantly lower than the reference values (P < .0025), with the exception of “cheese.” The intake of food for each product groups was below the DRDA, while the intake of meat was higher than the DRDA. The intake of “dairy products” and “drinks” was significant lower for the patients with MD versus the DNFCS. Furthermore, although below DRDA reference, the intake of food from the “bread and potatoes” group was significantly higher among patients with MD as compared with the DNFCS.
Correlation of Dairy Products With Protein and Calcium
A correlation of 0.74 (P < .001) was observed between the consumption of calcium and dairy products. Furthermore, a correlation of 0.4 (P = .002) was observed between the consumption of protein and dairy products.
Snacks, Soft Drinks, and Monosaccharides and Disaccharides
Patients consumed 72 ± 56.5 g of snacks per day, which is not significantly different from the average Dutch intake. The mean intake of soft drinks was higher among the patients, while the intake of sugars was significantly lower than the average Dutch intake but still above recommendations according to DRDA. When data were categorized by diabetes status among patients with MD, this lower intake of monosaccharides and disaccharides was found in only the diabetic group (n = 20; Table 2).
Mean Intake of Snacks, Soft Drinks, and Monosaccharides and Disaccharides in Patients With MD.
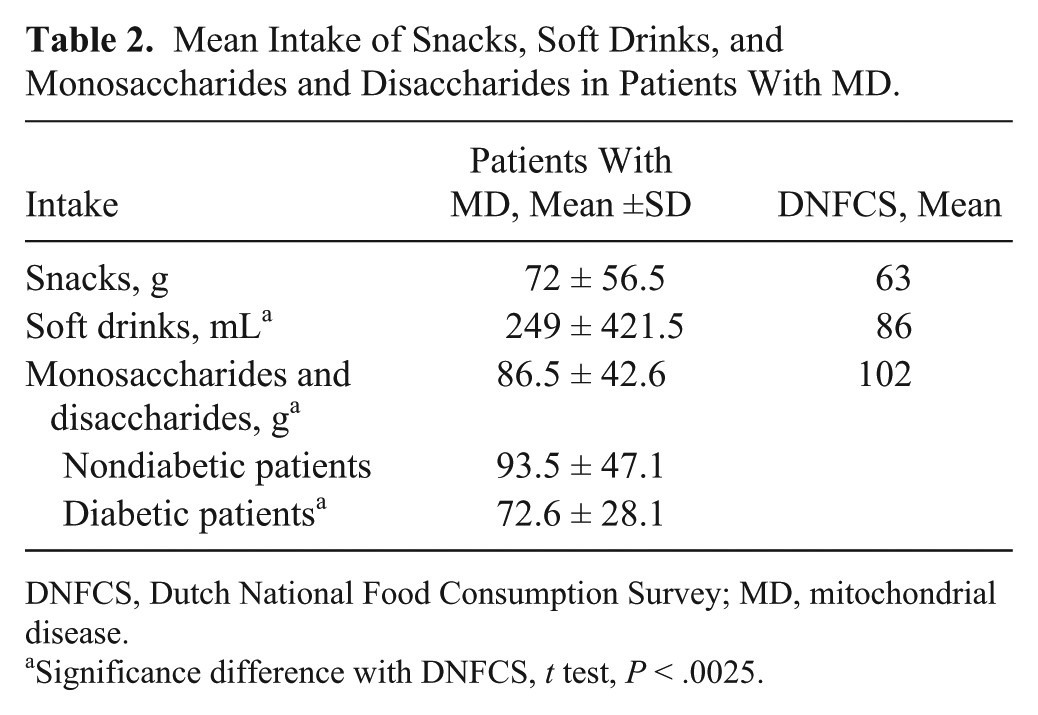
DNFCS, Dutch National Food Consumption Survey; MD, mitochondrial disease.
Significance difference with DNFCS, t test, P < .0025.
Supplement Use
Information was obtained on the nutritional supplement use of 44 patients. Of these, 68% consumed ≥1 nutritional supplements. Coenzyme Q10 was mostly used (44%), followed by multivitamin supplements (32%). One patient used enteral nutrition.
Discussion
The aim of this study was to describe which nutrients and/or product groups are specifically different from reference values in adult patients with MD. The main finding was that the nutritional intake of adult patients with MD is inadequate according to the DRDA and that the individual variation of intake is high. However, this also applies to the healthy Dutch population. Eating adequately seems to be difficult for everyone. It is particularly important, however, for patients with MD since an insufficient diet may increase the risk for malnutrition, leading to secondary mitochondrial dysfunction and resulting in worsening of symptoms. 6
Importantly, the average protein intake of patients with MD was significantly lower than that of the healthy Dutch population but still well above the recommendations for healthy individuals. As patients with MD are not healthy, protein requirement may be different in these individuals. For instance, among some patients with renal involvement, the protein intake should be restricted. 18 However, malnutrition is frequently found, leading to increased protein needs. Protein recommendations in malnutrition are minimal at 1.2 g/kg, 19 which is higher than the mean intake of the patients with MD. This lower protein intake could be partly explained by the very low intake of dairy products, which is specific for the patients with MD: a moderate correlation between protein and dairy products was calculated. Avoidance of dairy products may be explained by frequent gastrointestinal problems, especially related to bloating and abdominal cramps. 2 Adequate intake of dairy products is important in patients with MD, not only because of protein, but also because of calcium requirements. Correlation between dairy products and calcium intake was strong; calcium intake was too low for patients with MD, increasing the risk for osteoporosis. 20 Patients with MD already have a higher risk for osteoporosis because of low physical activity and persistent lactic acidosis. 21 In addition, vitamin D intake was, though not different between patients with MD and the DNFCS, extremely low. An adequate vitamin D status is important in MD since, in addition to the risk of osteoporosis, vitamin D deficiency may cause muscle weakness, 22 which is a key complaint among these patients. 1 Taken together, the inadequate intake of protein and dairy products is a serious concern in the diet of patients with MD.
Other relevant findings of this study were that patients with MD tend to have lower fluid intake than the DNFCS and a lower dietary fiber intake than recommended. As fluid and fiber intake is important in preventing and treating the frequently occurring gastrointestinal distress of these patients, 2 caution should be paid to these nutrients.
In our study, the energy intake was insufficient but not different from the DNFCS. It is known that the energy demands of patients with MD are usually below recommendations for healthy individuals because of the lower physical activity. 16 Because it is assumed that their nutritional requirements are not different 9 from those of typical individuals, the nutrient density of the diet should therefore be higher than in patients with normal energy demands. However, we did not find this compensating eating pattern among the patients with MD. Data from our study did not support our hypothesis that patients with MD eat more unhealthy than the general population. Although they did consume high levels of sugar, snacks, and saturated fat, this was not different from the consumption of the general Dutch population. Nevertheless, healthy eating is particularly important for patients with MD because of the increased risk for diabetes, fatigue, and metabolic syndrome.1,23 The high saturated fat intake is relevant, as cholesterol-lowering statins are discouraged in patients with MD because of the risk of statin myopathy. 24
Previous research regarding supplement use among patients with MD in a partly overlapping cohort (n = 33) revealed that 70% of them used nutritional supplements. 25 These data are corroborated by our findings in this cohort (68%). The most commonly used products were coenzyme Q10 and multivitamin supplements. The supplement use is not in line with the identified deficiencies in the diet, because calcium, vitamin D, fiber, or protein supplements were barely used. Given the increased incidence of malnutrition in patients with MD, 2 prescribing enteral nutrition or mineral and vitamin supplements tailored according to the patient’s individual deficiencies should be considered.
A strong point of this study is the relatively large group of subjects with this rare disorder. Although the patient population was heterogeneous, this study provides unique insight in the food intake of patients with MD in general. A weakness of the study is that the use of nutrition diaries may lead to bias in the results. Interpretation errors, as well as underreporting or overreporting, are possible reasons for this. 26 However, in a retrospective study, it is not possible to change the method of data collection, and nutrition diaries provide valuable information that qualitatively seems better than the frequently used dietary history. 27 Furthermore, the data collection method used in the DNFCS is the 24-hour dietary recall of any 2 nonconsecutive days. This method is different from that used in our study. These weaknesses should be taken into account when interpreting the study results.
Overall, it may be concluded that patients with MD have an inadequate diet. Calcium, dairy products, and fluid intake are often decreased. Furthermore, intake of fiber, sugars, saturated fat, and vitamin D intake differs from general recommendations. A healthy and adequate diet is especially important in patients with MD because of the increased risk of gastrointestinal problems, diabetes, metabolic syndrome, muscle weakness, fatigue, and osteoporosis. Because individual differences are considerable, a patient-tailored approach is recommended in which all adult patients with MD are provided with individual diet counseling for obtaining a healthy and adequate diet.
Footnotes
Acknowledgements
We thank Shanna de Kleer, Cassandra Arndts, and Emily Leenders for their assistance.
Financial disclosure:
None declared.
Conflicts of interest:
None declared.
Statement of Authorship
H. Zweers, M. C. H. Janssen, and G. Wanten contributed to the conception and design of the research; all authors contributed to the acquisition, analysis, or interpretation of the data; and H. Zweers drafted the manuscript. All authors critically revised the manuscript, agree to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
