Abstract
Subcutaneous infusion, or hypodermoclysis, is a technique whereby fluids are infused into the subcutaneous space via small-gauge needles that are typically inserted into the thighs, abdomen, back, or arms. In this review, we provide an overview of the technique, summarize findings from studies that have examined the use of subcutaneous infusion of fluids for hydration or nutrition, and describe the indications, advantages, and disadvantages of subcutaneous infusion. Taken together, the available evidence suggests that, when indicated, subcutaneous infusion can be effective for administering fluids for hydration or nutrition, with minimal complications, and has similar effectiveness and safety to the intravenous route. Of note, subcutaneous infusion offers several advantages over intravenous infusion, including ease of application, low cost, and the lack of potential serious complications, particularly infections. Subcutaneous infusion may be particularly suited for patients with mild to moderate dehydration or malnutrition when oral/enteral intake is insufficient; when placement of an intravenous catheter is not possible, tolerated, or desirable; at risk of dehydration when oral intake is not tolerated; as a bridging technique in case of difficult intravenous access or catheter-related bloodstream infection while infection control treatment is being attempted; and in multiple settings (eg, emergency department, hospital, outpatient clinic, nursing home, long-term care, hospice, and home).
Keywords
Intravenous (IV) fluids are administered in multiple clinical settings to provide hydration and/or nutrition. Notably, IV fluids are used in the treatment of dehydration, a common cause of hospitalization and death in elderly adults,1,2 particularly among those in nursing homes. 3 In the context of nutrition, IV fluids can help meet the nutrition demands of patients who require an alternative to, or supplemental, oral or enteral nutrition. Although the IV route can be effective for providing hydration or nutrition, IV access is not always possible or feasible—for example, in patients with fragile veins, those who are uncooperative, agitated, confused, or demented or in situations (eg, palliative care) where the maintenance of a venous line (either central or peripheral) may cause the patient suffering/pain. In such instances, an alternative means of fluid delivery is required. Two potential alternatives are subcutaneous infusion and intraosseous infusion. Of these, subcutaneous infusion (or hypodermoclysis) has the benefit of being the more straightforward approach.
Subcutaneous infusion, in which fluid is absorbed from the subcutaneous tissue to the circulation via the forces of diffusion and perfusion, 4 was first described in 1865 for treating dehydration in patients with cholera. 5 Since then, subcutaneous infusion has been used to provide hydration to infants, children, and adults, particularly elderly adults in whom venous access is difficult. 6 More recently, the findings from a randomized clinical trial highlighted the applicability of subcutaneous infusion for the delivery of nutrition. 7 Specifically, in a study of elderly hospitalized patients, 7 subcutaneous infusion of nutrition was noninferior to IV infusion in the occurrence of major local side effects. In addition, subcutaneous infusion was better tolerated and had similar clinical outcomes compared with IV infusion. 7
Given the findings from the aforementioned clinical trial and that subcutaneous infusion appears to be underrecognized and underused in some settings, 1 we performed a review of the literature on subcutaneous infusion of fluids for hydration or nutrition. MEDLINE, Embase, and Biosis databases were searched (inception to March 23, 2016) using the terms hypodermoclysis, clysis, and subcutaneous fluid. Articles describing subcutaneous infusion for hydration or nutrition were included, whereas those describing results from nonhuman studies or subcutaneous infusion of drugs were excluded. The aims of our review are to provide an overview of the technique and to summarize key findings from clinical studies, including those related to safety, the advantages and disadvantages of subcutaneous infusion, and its indications.
Technique
Subcutaneous infusion is a straightforward technique that can be applied with minimal training. After the skin is cleaned, a cannula (22- to 24-gauge needle) is inserted at a 45° angle into the subcutaneous space and aimed toward the head/thorax (Figure 1). Although both metal and nonmetal cannulae can be used, the duration of use may be longer with nonmetal cannulae. 8 The most common insertion sites are the lateral aspect of the abdomen, thighs (inner/outer aspect), pectoral region in men, outer surface of the forearms/upper arms (note: placement at these sites can restrict movement), or the interscapular region. Care should be taken to select sites that have adequate subcutaneous tissue (determined by pinching the skin between the fingers) and reasonable skin turgor. After insertion, the needle is covered with a bandage/transparent semipermeable dressing, as per an IV catheter, and connected to the container holding the fluid.
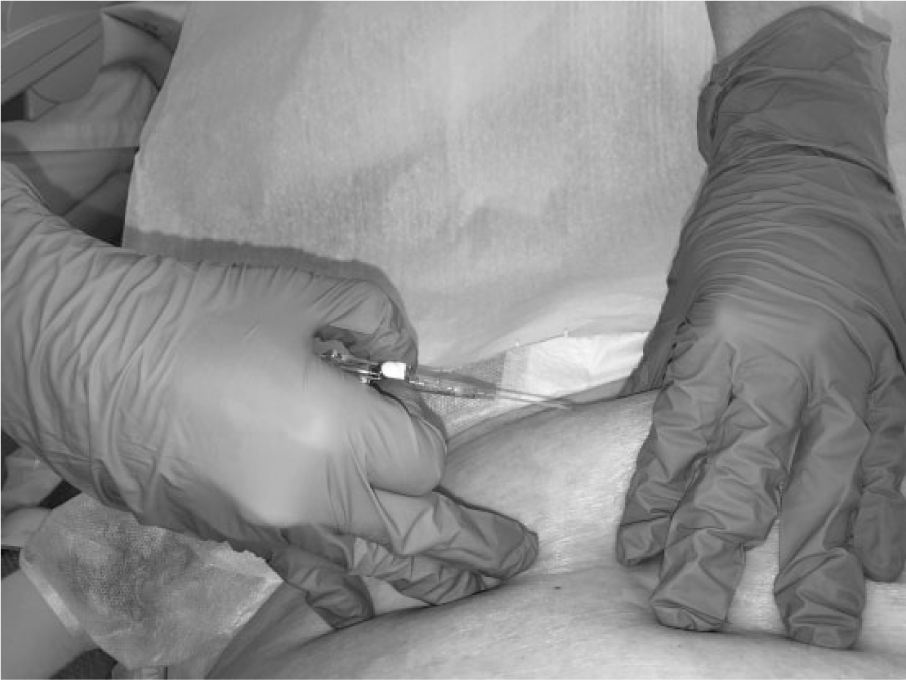
Photograph illustrating the insertion of a cannula in the abdomen for subcutaneous infusion. The cannula is inserted at a 45° angle into the subcutaneous space and aimed toward the central compartment.
Subcutaneous fluids can be delivered by gravity or infusion pump. Gravity infusion may help prevent local edema because the infusion rate naturally slows when pressure in the subcutaneous space increases. The rate of infusion should remain within the limits of tissue perfusion. Fluid is typically infused continuously over 24 hours at a rate of 62 mL/h (approximately 1500 mL in total) for an average-size adult. In our experience, up to 2000 mL can be infused per 24 hours with no or minor edema. If continuous infusion over 24 hours is infeasible, nocturnal infusion of 1000 mL/8 hours or up to 500 mL/2 hours is possible. With such infusion rates, transient local edema can be expected; however, patient discomfort is minimal. The total volume infused may be increased by concurrent infusion at multiple sites. In all cases, the development of large and/or progressive infusion site edema suggests that the rate of infusion exceeds the rate of absorption and that the infusion should be slowed or stopped.
To date, subcutaneous infusion has typically been used for short-term (≤10 days) delivery of hydration/nutrition (Tables 1 and 2); however, longer infusions are theoretically possible provided access is adequate and no complications occur.
Summary of Controlled Clinical Studies of Subcutaneous Infusion of Fluids for Hydration or Nutrition.
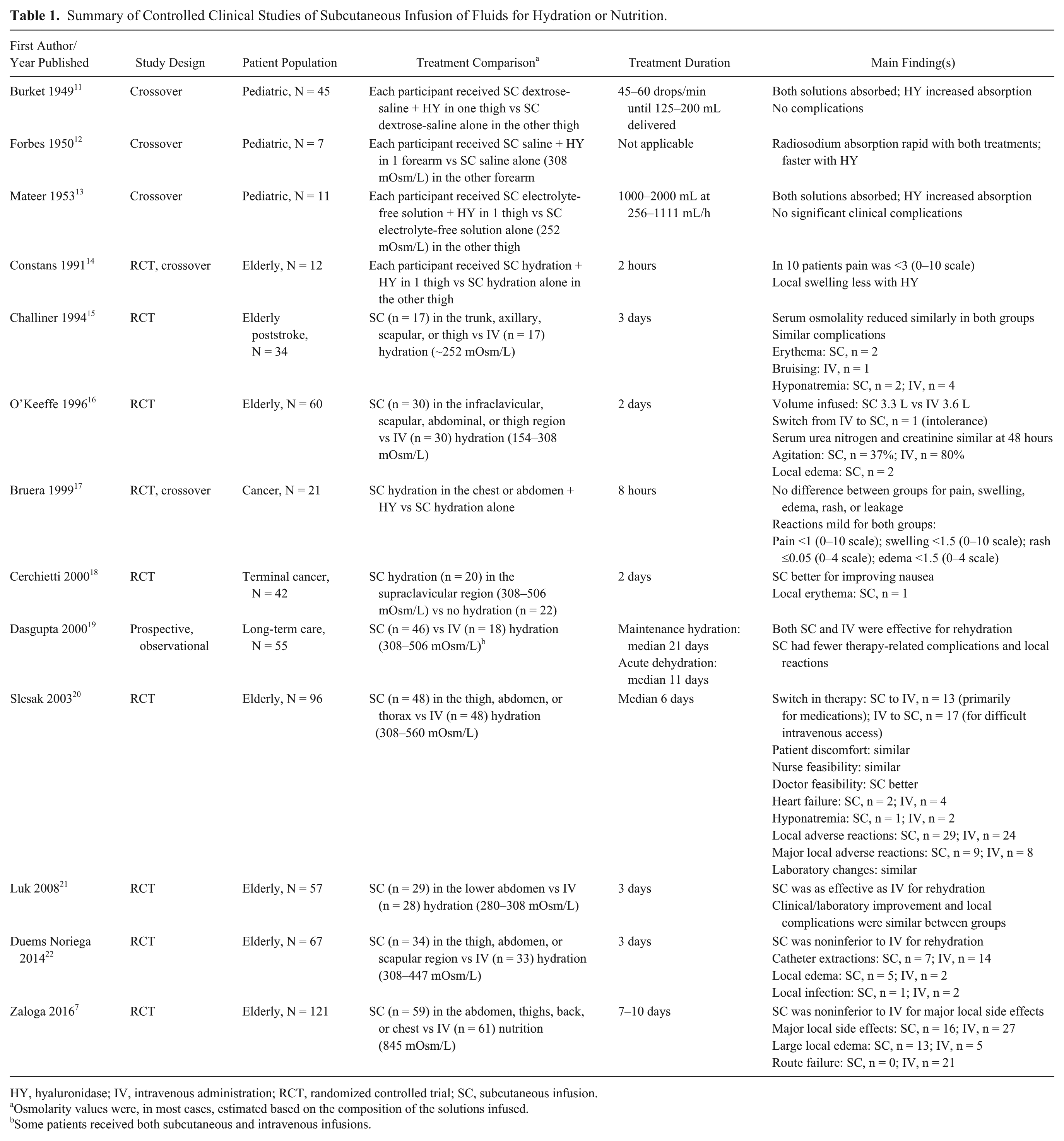
HY, hyaluronidase; IV, intravenous administration; RCT, randomized controlled trial; SC, subcutaneous infusion.
Osmolarity values were, in most cases, estimated based on the composition of the solutions infused.
Some patients received both subcutaneous and intravenous infusions.
Summary of Uncontrolled Clinical Studies of Subcutaneous Infusion of Fluids for Hydration or Nutrition.
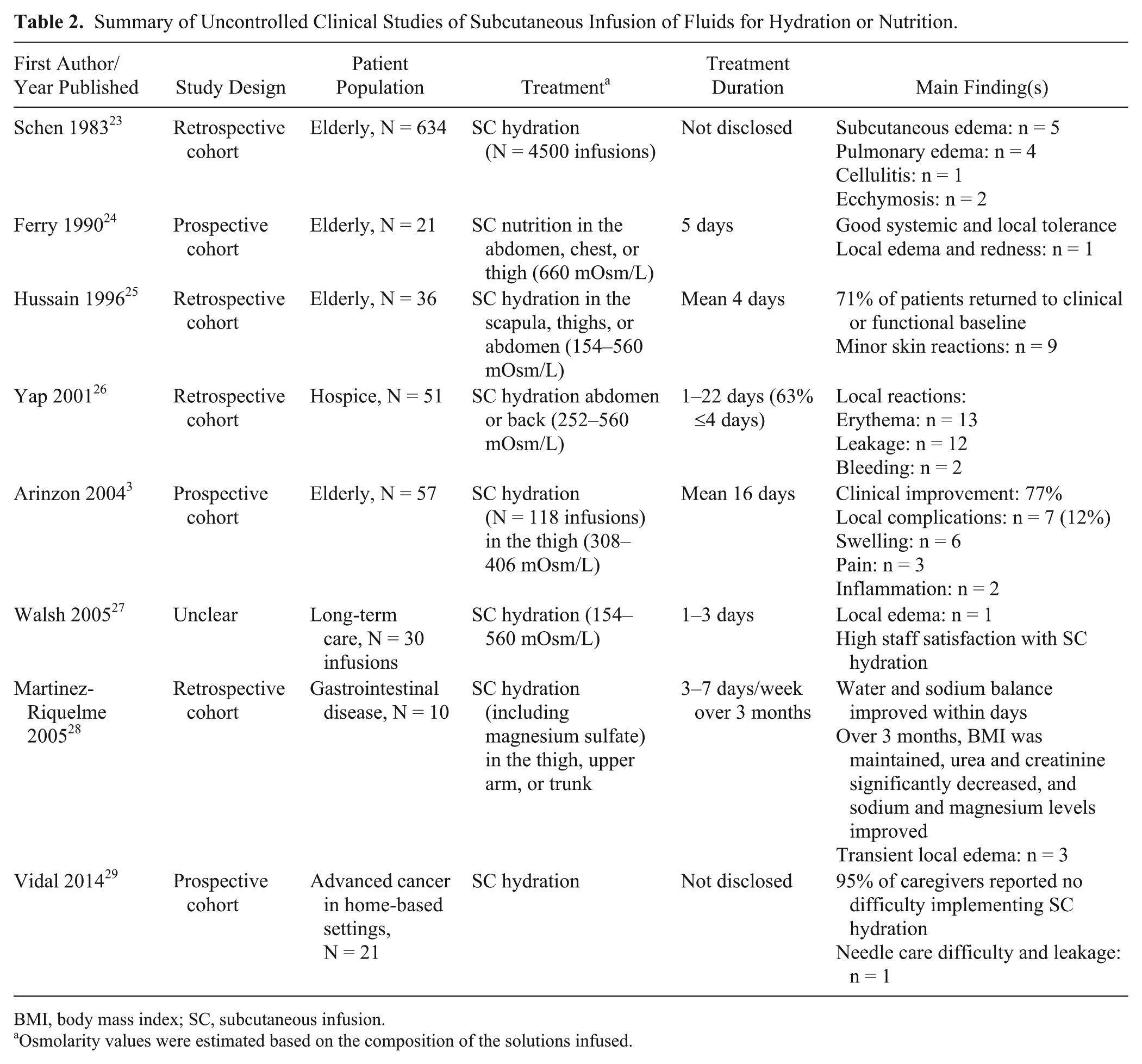
BMI, body mass index; SC, subcutaneous infusion.
Osmolarity values were estimated based on the composition of the solutions infused.
There are no published guidelines on osmolarity limits for subcutaneously infused fluids. Although infusion of fluids in the range of 154–845 mOsm/L has been reported (Tables 1 and 2), osmolarity in the range of 280–300 mOsm/L is expected to be best tolerated. However, a recent trial of subcutaneous infusion of nutrition showed that an osmolarity of 845 mOsm/L was well tolerated. 7
Hyaluronidase is an enzyme that hydrolyzes hyaluronan, the principal glycosaminoglycan of the hypodermis.9,10 Hydrolysis of hyaluronan reduces viscosity of the hyaluronic acid gel component of subcutaneous tissue and promotes permeability of the connective tissue matrix, enhancing absorption of fluid/drugs from the subcutaneous space.9,10 Because of these properties, hyaluronidase is sometimes administered before or concurrent with subcutaneously infused fluids to increase the rate of absorption; however, as discussed later in this review, the benefits of hyaluronidase appear limited at standard rates of infusion.
Maintenance of subcutaneous infusion is straightforward. Needles should be changed every 24 hours to reduce risk of skin infection. Likewise, the fluid container and tubing should be changed every 24 hours as per IV infusion.
Findings From Clinical Studies
Since 1949, clinical studies of various design have reported on subcutaneous infusion of fluids for hydration/nutrition (Tables 1 and 2). Most of these studies have reported on infusion of fluids for hydration; 2 studies have reported on infusion of fluids for nutrition. Patient populations in these studies were typically the elderly (often in long-term care), although some involved pediatric populations or patients with cancer.
Early controlled studies were crossover in design and compared subcutaneous infusion of fluids with/without hyaluronidase (Table 1). More recent controlled studies were typically randomized trials comparing subcutaneous infusion of fluids with IV infusion of fluids or subcutaneous infusion of fluids plus hyaluronidase (Table 1). Most uncontrolled studies were retrospective cohort studies (Table 2).
The available evidence from controlled and uncontrolled clinical studies (see ensuing subsections and Tables 1 and 2 for further details) indicates that subcutaneous infusion can be an effective technique for administering fluids for hydration and has a favorable safety profile in various patient populations. Adverse events are relatively uncommon and are primarily local reactions, such as edema, that resolve without intervention. The subcutaneous and IV routes have similar effectiveness and safety in patients with mild to moderate dehydration.
Recent evidence suggests that subcutaneous infusion of fluids for nutrition can be effective and has a favorable safety profile as per administration of fluids for hydration. Subcutaneous infusion of fluids for nutrition may be a suitable alternative to IV infusion in some patients.
Of note, subcutaneous infusion can be provided with minimal training and can be easily applied by individuals with limited medical experience.
Hyaluronidase has no proven benefit in facilitating subcutaneous infusion when rapid absorption is not required.
Controlled Studies
Burket and Gyorgy 11 reported one of the first controlled studies of subcutaneous infusion. Pediatric patients received a 125- to 200-mL subcutaneous infusion of 2.5% glucose and 0.9% saline with/without hyaluronidase. The main findings of this study were that both solutions were absorbed, although at a faster rate with hyaluronidase (107 vs 173 min), and that there were no thermal reactions, exacerbation of infections, or evidence of renal damage. Hyaluronidase did not change phenolphthalein dye excretion, a previously used indicator of renal function, or total renal fluid excretion.
Forbes et al 12 also reported the results of a study involving pediatric patients who received 0.5 mL or 4 mL subcutaneous infusion of 0.9% saline containing radiosodium with/without hyaluronidase. The main finding of this study was that radiosodium was rapidly absorbed both with and without hyaluronidase but more so with hyaluronidase.
Mateer et al 13 reported the results of another study involving pediatric patients who received a 1000- to 2000-mL subcutaneous infusion of 5% dextrose with/without hyaluronidase. The main findings of this study were that hyaluronidase increased the rate of fluid infusion (767 mL/h with hyaluronidase vs 546 mL/h without hyaluronidase) and that there were no clinically significant changes in serum electrolytes, blood hemoglobin, or circulatory parameters.
Constans et al 14 reported the results of a study involving elderly patients who received a 500-mL subcutaneous infusion of 5% glucose (containing 2 g NaCl and 0.375 g KCl) over 2 hours with/without hyaluronidase. The main findings of this study were that there were no differences between treatments in pain, skin color, or skin temperature and that the increase in thigh circumference after infusion was significantly less pronounced with hyaluronidase (3.1 cm with vs 4.4 cm without).
In another study of elderly patients reported by Challiner et al, 15 patients with acute stroke received a 2000-mL subcutaneous or IV infusion of isotonic dextrose-saline (30 mmol/L NaCl, 40 g/L dextrose) per 24 hours. Both treatments similarly reduced serum osmolality (mean for subcutaneous group, day 1 = 299 mOsm/L, day 3 = 289 mOsm/L; IV group, day 1 = 293 mOsm/L, day 3 = 280 mOsm/L). Complication rates were similar between groups. Notably, the cost of infusion cannulae was markedly lower for the subcutaneous group compared with the IV group.
O’Keeffe et al 16 studied elderly hospitalized patients with cognitive impairment who received continuous subcutaneous or IV infusion of 0.9% saline or 0.45% saline and 5% dextrose for 48 hours (maximum volume was 2000 mL/24 hours). The investigators found there were no differences in the volume of fluid infused or in serum urea or creatinine at 48 hours and that agitation was significantly (P < .005) more common in the IV group compared with subcutaneous group. One patient in the IV group was switched to subcutaneous fluids after 24 hours due to difficult IV access. Two patients in the subcutaneous group had local edema; no other complications were reported. The cost of infusion cannulae was markedly lower for the subcutaneous group compared with the IV group.
Bruera et al 17 reported the results of a study involving patients with cancer who received 500 mL of 5% dextrose and normal saline with/without hyaluronidase by subcutaneous infusion. The key findings of this study were that adverse reactions were mild in both groups and that there were no significant differences between the groups in local reactions.
Cerchietti et al 18 performed a study of terminally ill patients with cancer who received a 1000-mL subcutaneous infusion of 5% dextrose (with 140 mEq/L NaCl) at 42 mL/h or no subcutaneous infusion. After 48 hours, significant (P = .027) improvement in nausea was maintained only in the subcutaneous group. Only 1 patient in the subcutaneous group had a local adverse reaction of infusion site erythema and pain.
Dasgupta et al 19 described the outcomes of a study of patients in long-term care who received subcutaneous or IV infusion of normal saline or two-thirds 5% dextrose and one-third normal saline at a rate of 5–75 mL/h. Notable findings from this study included significantly (P < .05) fewer fluid therapy-related complications and local reactions in the subcutaneous group compared with the IV group (0.07 vs 0.21 per day and 0.05 vs 0.20 per day, respectively). Furthermore, of the 24 patients who received subcutaneous fluids for maintenance hydration, none required additional hydration therapy. Among the 37 patients who received treatment for acute dehydration, there was no significant difference between groups in the proportion who had clinical improvement (subcutaneous 57% vs IV 81%).
In a study involving elderly patients, Slesak et al 20 gave participants 500 mL subcutaneous or IV infusions of 5% dextrose and half-normal saline over 2–6 hours as necessary. Overall, the median volume administered was 750 mL/d for the subcutaneous group and 1000 mL/d for the IV group. The main findings of this study were that doctors’ procedure feasibility scores (assessed using a Likert-like scale where scores range from 1 [very good feasibility] to 6 [very bad feasibility]) were significantly (P = .011) higher for the subcutaneous group compared with the IV group (there were no between-group differences in patient discomfort and nurse feasibility scores) and that the occurrence of adverse reactions was similar between groups (there were few major adverse reactions in either group).
Luk et al 21 reported the results of a study involving elderly patients who received subcutaneous or IV infusion of 0.9% saline or 0.45% saline and 2.5% dextrose at a maximum rate of 1500 mL/d. Key findings from this study were that improvement in clinical and laboratory parameters and the incidence of local complications were similar between groups.
Duems Noriega and Arino Blasco 22 examined the effects of subcutaneous or IV infusion of 0.9% saline or 0.45% saline and 5% dextrose for 72 hours in elderly patients. The mean volume administered was 1320 mL/d for the subcutaneous group and 1480 mL/d for the IV group. The investigators found that the magnitude of improvement in urea, creatinine, and osmolarity was similar between groups; there were fewer catheter extractions in the subcutaneous group compared with the IV group; and there were few adverse reactions in either group.
Zaloga et al 7 carried out a study of hospitalized elderly patients who received subcutaneous or peripheral IV infusions of a parenteral nutrition solution (410 kcal/L; 28 g/L amino acids, 75 g/L glucose, 35 mmol/L sodium, 30 mmol/L potassium, 2.3 mmol/L calcium, 2.5 mmol/L magnesium, and 15 mmol/L phosphorus) at 1000 mL/12 hours for 7–10 days. Key findings were that nutrition and biochemical parameters, safety, and clinical outcomes were similar between groups. Notably, the subcutaneous group was noninferior to the peripheral IV group for major local side effects, including large local edema, blistering, erythema, phlebitis, cellulitis, unbearable pain, or route failure requiring a switch in route. Local edema was significantly (P < .05) more common in the subcutaneous group than the peripheral IV group, whereas route failure was significantly (P < .001) more common in the IV group.
Uncontrolled Studies
Schen and Singer-Edelstein 23 reported the results of a study involving elderly patients who received a subcutaneous infusion of isotonic saline or dextrose. The main finding of this study was that there were few adverse reactions; the most common was fluid overload in the form of subcutaneous edema (typically in the pelvic and genital regions) or heart failure.
Ferry et al 24 carried out a study involving elderly malnourished patients who received a 500-mL subcutaneous infusion of amino acids (70 g/L) and 500 mL of 5% dextrose with NaCl and KCl for 5 days. The infusion, which delivered 35 g/d of protein, was found to be well tolerated, both locally and systemically, and there were no generalized reactions. Furthermore, prealbumin levels increased over the treatment period.
Hussain and Warshaw 25 conducted a study of elderly patients in nursing homes who were given a subcutaneous infusion of 0.9% or 0.45% saline (both could be given with 5% dextrose) at a rate of 1000–2000 mL/d for a mean of 4 days. Hyaluronidase was given in 78% of infusions. Of note, nearly three-quarters of patients returned to clinical or functional baseline after treatment (only 2 patients were hospitalized for further treatment), serum sodium was improved after treatment, and serum urea nitrogen/creatinine ratios were maintained. No significant complications were reported; minor skin reactions reported included local inflammation, infusion site pain, bruising, edema, and local swelling.
In a hospice care study performed by Yap et al, 26 patients received a subcutaneous infusion of dextrose and saline or 5% dextrose at a rate of 1500 mL/d for 1–22 days. Local reactions were generally infrequent; erythema, leakage, and bleeding were the most common. No patients developed infection.
Arinzon et al 3 examined the outcomes of a study of elderly patients in long-term care who were given subcutaneous infusions of 0.9% saline, 3.3% dextrose in 0.3% saline, or 5% dextrose in 0.45% saline at a mean rate of 1100 mL/d for 5–60 days. Key findings were that more than three-quarters of patients had clinical improvement and that no patient developed fluid overload. After hydration, serum urea and creatinine significantly (P < .001) decreased, potassium significantly (P < .001) increased, and sodium and hematocrit significantly (P < .05) decreased. Local complications included swelling, pain, and inflammation.
In a study of patients in long-term care, Walsh 27 examined the effects of subcutaneous infusions of 0.45% or 0.9% saline with or without 5% dextrose at a rate of 40–125 mL/h. Adverse reactions were found to be rare (there was just 1 case of local edema), and staff and facility management reported high levels of satisfaction with the technique.
Martinez-Riquelme et al 28 described the results of an investigation that included patients with short bowel or gastrointestinal failure who self-administered a 500- to 1000-mL subcutaneous infusion of 5% dextrose or 0.9% saline and MgSO4 every 6–12 hours, 3–7 days/week. All patients found the technique easy to learn and reported good tolerance. Furthermore, clinical improvement in sodium and water balance was found to be rapid, while there were no instances of infection, pulmonary edema, or vascular incidents. Approximately 30% of patients experienced transient local edema, which resolved with a reduced rate or volume of fluid administration. Patients with hypomagnesemia and magnesium deficiency normalized their magnesium status during treatment.
Vidal et al 29 reported the results of a study of patients with advanced cancer in home-based hospice care settings given subcutaneous infusions for hydration by caregivers who had received training. Notable findings from this study were that caregivers were able to successfully implement subcutaneous infusion in home settings and that adverse effects were rare.
Hyaluronidase Studies
A number of studies and case reports/series involving different patient populations (pediatric, elderly, cancer, palliative care, healthy) have reported on the use of hyaluronidase to facilitate subcutaneous infusion.11–14,17,25,30–48 Taken together, the findings from these studies suggest that hyaluronidase can increase the rate of fluid absorption and therefore may be of benefit when fluid is infused at high rates or is given as large bolus injections, when large volumes of fluid are required, or when local edema limits fluid administration. However, when rapid absorption of fluid is not required, as per a typical subcutaneous infusion, hyaluronidase has no proven benefit. Indeed, many clinicians do not routinely use hyaluronidase with subcutaneous infusion.6,49–51 In addition to having no benefit in most cases, hyaluronidase increases the complexity, risk of allergic reaction to hyaluronidase (especially with animal-derived preparations), risk of fluid overload, and cost of subcutaneous infusion.
Safety of Subcutaneous Infusion
As already noted, adverse reactions of subcutaneous infusion are generally infrequent; however, further discussion of potential reactions, including local reactions, infection, fluid overload, and vascular collapse, is warranted.
Potential local adverse reactions of subcutaneous infusion include edema, erythema, pain, and ecchymosis. Some local edema, particularly in the genital area, is normal and typically resolves within hours of stopping the infusion. Pain is rare but may occur, particularly if the needle is inserted into the muscle or due to cutaneous tension from large volumes of fluid or use of potassium. 52 There is a small risk of blood vessel puncture; however, bleeding is rare in patients with normal coagulation. Furthermore, as products approved for subcutaneous infusion are also approved for IV infusion, any inadvertent IV infusion due to blood vessel puncture should not pose a safety concern. Sloughing (necrosis) of tissue may result if inappropriate fluids (ie, markedly hypertonic or hypotonic, or those containing high concentrations of KCl) are administered. 33 Infections (ie, abscess, cellulitis) at the site of infusion can occur but are rare.4,23,52,53 Appropriate skin preparation and wound dressing are recommended to help reduce the risk of infection, 1 as is changing the site of infusion every 24 hours.
Fluid overload is a potential risk of any fluid administration that may lead to local and regional edema, heart failure, and pulmonary edema. The use of hyaluronidase, which increases the rate/volume of fluid absorption, may increase the risk of fluid overload with subcutaneous infusion; however, the risk of fluid overload with standard subcutaneous infusion is lower than the risk with IV infusion due to the lower rate of infusion.50,54,55 Nevertheless, monitoring to minimize risk is important, particularly in light of evidence that subcutaneously infused fluids may be absorbed at a similar rate to intravenously infused fluids.36,56
Subcutaneous infusion of large volumes of electrolyte-free solutions or hypertonic solutions has been rarely associated with vascular collapse.4,6,52,57–60 These solutions may not be as well absorbed as electrolyte-containing solutions and may draw water from the vascular space, contributing to decreased intravascular volume and hypotension. 57 Slower infusion and infusion of smaller volumes may ameliorate any risk. This being said, the danger of vascular collapse with isotonic dextrose may be overstated; several reports note no increased risk with 5% dextrose.13,16,23,33,52
In addition to these adverse reactions, blood exposure accidents are possible with subcutaneous infusion. For instance, agitated or confused patients may dislodge metal needles inserted into the subcutaneous space by pulling on the tubing. Staff treating such patients could experience needlestick injury and/or blood exposure, although, given the location of the catheter, the likelihood of blood exposure is clearly less than that associated with IV infusion. Nevertheless, the use of nonmetal catheters may ameliorate these risks.
Advantages of Subcutaneous Infusion
Subcutaneous infusion offers several advantages over IV infusion (Table 3). Notably, subcutaneous infusion is straightforward, is economical, and can provide an alternative means of infusion for patients with difficult IV access. Specialized expertise is not required for setup, and the time required to place the catheter is less than that for IV infusion.1,9,50,54,61 Furthermore, the ease of this technique means it may be used in multiple settings, including home, nursing home, hospice care, and other settings where skilled nursing care is not available, as well as in hospitals.9,52 As such, subcutaneous infusion may help prevent hospitalization (and the associated costs) for rehydration.4,54,61,62 The cost of placing catheters and the number of catheters used are less than for the IV route. 1 An additional cost benefit of subcutaneous infusion is the lack of requirement for costly electronic infusion pumps. Other important advantages of subcutaneous infusion over IV infusion include increased patient mobility and comfort.9,50,52 This comes from the lack of requirement for limb immobilization, which also reduces the risk of pressure sores and deep vein thrombosis. 1 Agitated patients, who frequently pull out IV catheters, are also less likely to be bothered by the less restrictive subcutaneous infusion lines.1,6,9 For similar reasons, subcutaneous infusion is particularly well suited for use in nursing homes (ie, lines can be placed out of reach of the patient, reducing the risk of dislodgement). 1 Increased patient comfort with subcutaneous infusion is also reflected by less pain and fewer systemic complications (including thrombophlebitis, sepsis, pulmonary edema, and hyponatremia) than IV infusion.50,54
Advantages and Disadvantages of Subcutaneous Infusion (Without Hyaluronidase).

IV, intravenous administration.
The disadvantages of subcutaneous infusion are also summarized in Table 3 and are discussed in further detail elsewhere in this review (see Safety of Subcutaneous Infusion and Indications sections).
Indications
Based on the available evidence, subcutaneous infusion may be indicated for patients
with mild to moderate dehydration or mild to moderate malnutrition when oral/enteral intake is insufficient;
when placement of an IV catheter is not possible, tolerated, or desirable (eg, for patients with a systemic infection who do not have an IV line or for patients recovering from injury/illness who require nutrition supplementation but do not have an IV catheter);
at risk of dehydration when oral intake is not tolerated;
as a bridging technique for patients with difficult IV access while IV access is being attempted;
as a bridging technique for patients with catheter-related bloodstream infection while infection control treatment (eg, systemic antibiotic therapy, antibiotic lock therapy) is being attempted;
in multiple settings, including emergency department, hospital, outpatient clinic, nursing home, long-term care, hospice, and home.
Subcutaneous infusion is not primarily indicated for patients with
severe dehydration or malnutrition, shock, or any condition requiring rapid or large-volume administration of fluids;
severe electrolyte disturbances;
decreased tissue perfusion;
compromised skin integrity or evidence of skin infection;
bleeding or coagulation disorders;
generalized edema.
Note: Products for subcutaneous infusion are available in non-U.S. countries, some for more than 30 years, and there have been no safety concerns. Despite the availability of extensive safety/pharmacovigilance data, U.S. regulations require clinical trials to be conducted in the United States before approval can be considered.
Conclusions
In summary, the available evidence suggests that subcutaneous infusion can be an effective technique for the administration of fluids for hydration or nutrition, with minimal complications. Consistent with the findings of a recent Cochrane Review, 63 which compared different methods of parenteral access for administering fluids or medications, our review also highlights that subcutaneous infusion can provide an alternative means of administering fluids when IV access is difficult or not possible. Furthermore, subcutaneous infusion has several advantages over IV infusion, including ease of application in multiple settings, low cost, and the lack of potential serious complications, particularly infections. Nevertheless, as much of the evidence on subcutaneous infusion to date comes from relatively small randomized controlled trials or observational studies, high-quality randomized controlled trials are warranted. Studies examining the use of subcutaneous infusion for providing nutrition or drugs in different patient populations (ie, geriatrics, palliative care, neurology) would be particularly welcome.
Footnotes
Acknowledgements
We thank Dr Julianna Roth Jakubowski for her contribution to the literature search and review of the literature.
Financial disclosure:
The study reported by Zaloga et al was sponsored by Baxter Healthcare Corporation, the manufacturer/licensee of CLINIMIX parenteral nutrition products. Medical writing assistance was provided by Luke Carey, PhD, and Tania Dickson, PhD, of ProScribe–Envision Pharma Group and was funded by Baxter Healthcare Corporation. ProScribe’s services complied with international guidelines for Good Publication Practice (GPP3). Baxter Healthcare Corporation was involved in the collection of information/data, interpretation of information/data, and preparation of the manuscript.
Conflicts of interest:
RC has received research funding from Baxter Healthcare Corporation. RC and TC have served as consultants and/or on advisory panels for Baxter Healthcare Corporation. RC and PC have participated in speakers’ bureaus for Baxter Healthcare Corporation. GPZ is a former employee of Baxter Healthcare Corporation. AP is employed by Baxter Healthcare Corporation. GPZ and AP own shares in Baxter Healthcare Corporation.
Statement of Authorship
R. Caccialanza, T. Constans, P. Cotogni, G. P. Zaloga, and A. Pontes-Arruda interpreted the data from the literature, participated in the drafting and critical revision of the manuscript, and read and approved the final manuscript. All authors agree to be fully accountable for ensuring the integrity and accuracy of the work.
