Abstract
Background: It has been reported that cystine and theanine, amino acids related to glutathione synthesis, have immunomodulatory effects, such as suppressing inflammation after strenuous exercise. In this study, we examined the effects of oral administration of cystine and theanine during the perioperative period as a pilot study. Methods: Forty-three cases of distal gastrectomy for cancer conducted in our department were assigned to the cystine and theanine group (CT group) or to the placebo control group (P group), and a randomized, single-blind, parallel-group study was then performed. Cystine (700 mg) and theanine (280 mg) or a placebo was administered to participants for 10 continuous days (4 days before to 5 days after surgery). Changes in pre- and postoperative interleukin (IL)–6, C-reactive protein (CRP), albumin, white blood cell (WBC) count, neutrophil count, total lymphocyte count, resting energy expenditure (REE), and body temperature were compared and examined. Results: Ten patients were excluded, leaving 33 patients in the study. The CT group had significantly lower IL-6 values (postoperative day [POD] 4), CRP levels (POD 7), neutrophil counts (POD 4), and body temperatures (POD 5) than the P group (P < .05). In addition, REE in the P group peaked on day 1 (1.14 ± 0.16 [pre- and postoperative ratio]), whereas the CT group did not show any increase on POD 1 (0.99 ± 0.21, P < .05 vs P group). Conclusions: This study suggests that oral administration of cystine and theanine during the perioperative period may alleviate postgastrectomy inflammation and promote recovery after surgery.
Clinical Relevancy Statement
Appropriate regulation of inflammation during the perioperative period is important for achieving a stable postoperative course and early recovery and for reducing the malignancy relapse rate. For these reasons, an immune-modulating diet that includes anti-inflammatory substances such as ω-3 fatty acids has been used widely in clinical settings. The amino acids cystine and theanine are both involved in the synthesis of glutathione (GSH), a strong antioxidant, and there have been several reports that oral cystine and theanine enhance immunoreactions. This study was a pilot study to assess the effect of perioperatively administering oral cystine and theanine in gastric surgery patients and suggested that oral administration of these amino acids may reduce inflammation and promote recovery after gastric surgery.
Introduction
Preventing organ dysfunction and immune suppression by inhibiting excess inflammation is considered an important aspect of perioperative surgical management, and several studies have indicated that immunonutrition is effective for this purpose.1,2 Administering the amino acids cystine and theanine has been shown to significantly suppress increases in C-reactive protein (CRP) levels and neutrophil counts and decreases in the number of lymphocytes after excessive exercise, resulting in a reduction in excessive inflammation and immune dysfunction; similar effects are expected to result from immunonutrition. 3 Cystine is a sulfur-containing amino acid that consists of 2 cysteine molecules joined by a disulfide bond. 4 Transporters on cell membranes (including those of neutrophils, macrophages, and dendritic cells) mediate the uptake of cystine into cells, and then thioredoxin rapidly reduces it to produce cysteine. 5 Theanine is an amino acid found in green tea that is metabolized in the intestinal tract and liver to yield glutamic acid and ethylamine.6,7 Cystine and theanine are both involved in synthesizing glutathione (GSH), which is a tripeptide consisting of glutamic acid, cysteine, and glycine, and it has been confirmed that hepatic GSH concentrations increase after cystine and theanine administration in mice.4,8,9 The importance of GSH to the immune system has been reported previously. Besides having a strong antioxidant effect, GSH plays an important role, particularly in the maintenance and regulation of the thiol-redox status of the cell, and these appear to correlate with proliferation or differentiation of lymphocytes, macrophages, and dendritic cells.10-15 Studies of patients receiving elective abdominal surgery have found that GSH levels in blood and skeletal muscle decrease by 10% and 42%, respectively, within 24 hours of surgery, and preventing this reduction in GSH may reduce immune suppression during the perioperative period. 16
In this study, we postulated that cystine and theanine would reduce excessive inflammation and immune suppression during the perioperative periods of abdominal surgeries, which decreases GSH levels in blood and skeletal muscle. 16 We examined the effects of perioperatively administering cystine and theanine on the postoperative course of gastric cancer patients undergoing distal gastrectomies as a pilot study.
Methods
Patients
All patients who underwent distal gastrectomy for gastric cancer by our department between January 2009 and October 2010; who were between 40 and 75 years old; who did not have distant metastasis, undernutrition (serum albumin <3.5 g/dL), decline in performance status (≥2), or pyloric stenosis for cancer; and who agreed to participate were included in the study.
Consent and Ethics
This clinical study was approved by the institutional review board of our hospital, and informed consent was obtained from patients after the nature of the procedure had been explained.
Study Design
Patients were assigned to 1 of 2 groups using the sealed-envelope method and a randomized, single-blind, parallel-group study was performed. Either cystine (700 mg) and theanine (280 mg) or a placebo (950 mg cellulose and 30 mg glutamine) was orally administered to patients with a small amount of water from 4 days before to 5 days after surgery, including the day of the procedure. Both groups received postsurgical management that followed the clinical pathway used at our hospital (Figure 1).
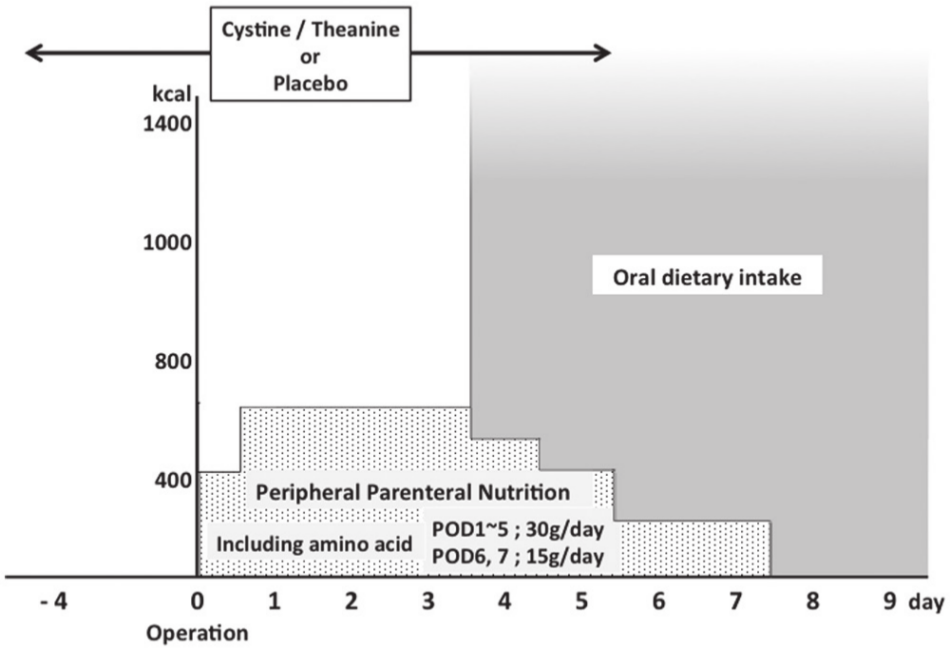
Perioperative management protocol. POD, postoperative day.
The dosage of cystine and theanine was determined based on human studies. Average daily intake of cystine from food has been reported to be 1000 mg/d with a maximum of 2200 mg/d.17,18 From the viewpoint of safety, the amount of cystine was determined to be 700 mg. Experimental analysis revealed that a weight ratio of cystine to theanine of 5:2 was the best to achieve effective increases in GSH levels. 19 According to this ratio, the dosage of theanine was decided as 280 mg. These test substances are commercially available from Ajinomoto Co, Inc (Tokyo, Japan).
Operation Method
All patients were laparotomized by upper midline incision under general anesthesia with epidural anesthesia; distal gastrectomy and regional lymph node dissection were performed according to the guideline of the Japan Gastric Cancer Association, followed by reconstruction after the Billroth-I or Roux-en Y method.
Data Collection
Variables investigated included interleukin (IL)–6 (before surgery, immediately after surgery, and postoperative days [PODs] 1, 4, and 7); CRP, albumin, white blood cell (WBC) count, neutrophil count, and total lymphocyte count (measured before surgery and on PODs 1, 4, 7, and 14); resting energy expenditure (REE, measured before surgery and on PODs 1, 4, 5, 7, and 14); and maximum body temperature (before surgery, on the day of the procedure, and every day after surgery). Changes in these variables were examined and compared between the 2 groups. REE measurements were performed on an empty stomach in the late afternoon using a small expiration gas analyzer (MedGem; HealthTech, Inc, Golden, CO).
Exclusion Criteria
Registered cases with excessive intraoperative hemorrhage (>600 mL), intraoperative complications, withdrawal of participation consent, and changes in surgical procedure were excluded from the study. The hemorrhage criterion was set as nearly twice the mean amount of bleeding during gastrectomy for cancer in our hospital. All criteria were set before starting the study.
Statistical Analyses
All statistical analyses were performed using SPSS (Statistical Package for the Social Sciences) version 17 (SPSS, Inc, an IBM Company, Chicago, IL) or Sigma Stat3.1 statistical analysis software (Systat Software, Inc, Richmond, CA). In all analyses, P < .05 was taken to indicate significance. The Student t test and Mann-Whitney U test were used to compare results between the 2 groups. A 2-way analysis of variance (ANOVA) model and the χ2 test were used to compare patient backgrounds between the 2 groups.
Results
Study Characteristics
A total of 43 cases were enrolled in this study: 22 cases in the cystine/theanine group (CT group) and 21 cases in the placebo group (P group). Of these cases, 10 were excluded due to the onset of herpes zoster before the procedure (n = 1), excessive intraoperative hemorrhage (n = 2), injury to the left hepatic artery during surgery (n = 1), changes in surgical procedure (n = 2), metastasis to the peritoneum confirmed during surgery (n = 1), or withdrawal of consent (n = 3). Therefore, the study population consisted of a total of 33 cases: 15 cases in the CT group and 18 cases in the P group. There were no significant differences in age, sex, amount of intraoperative bleeding, duration of surgery, degree of lymph node dissection and type of reconstruction, or course of disease progression (International Union Against Cancer [UICC]/TNM Classification of Malignant Tumours, seventh edition) 20 between the 2 groups (Table 1). All 33 cases in the study were managed using the clinical pathway; no cases deviated from the pathway, and no complications were observed after the procedure. No cases required blood transfusion.
Clinical Backgrounds and Demographics of Patients in the 2 Groups.
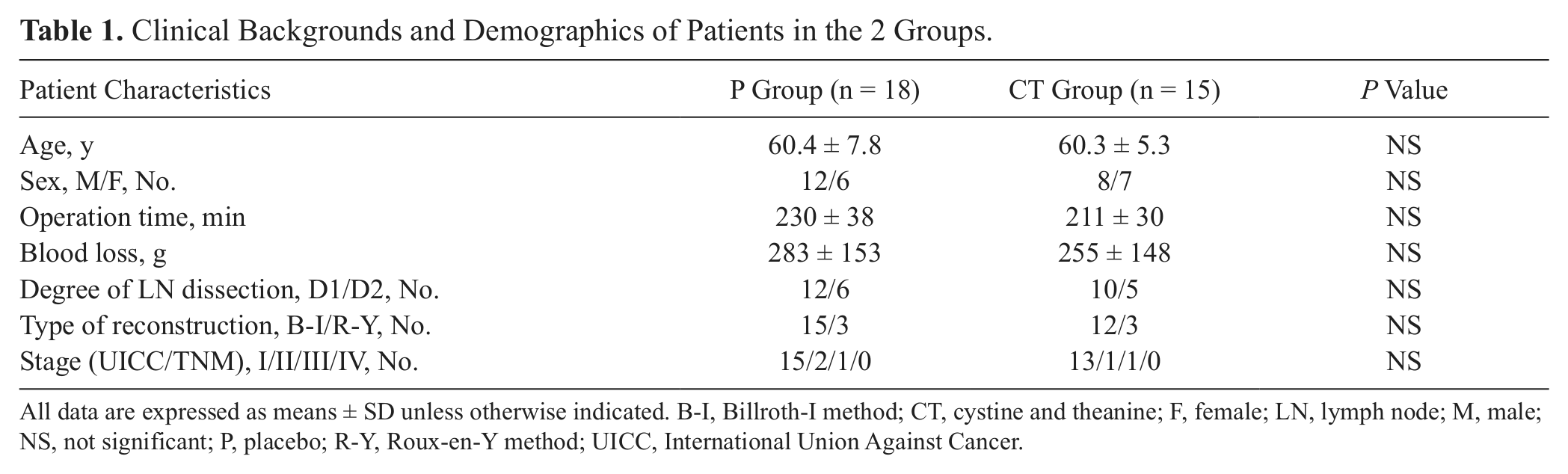
All data are expressed as means ± SD unless otherwise indicated. B-I, Billroth-I method; CT, cystine and theanine; F, female; LN, lymph node; M, male; NS, not significant; P, placebo; R-Y, Roux-en-Y method; UICC, International Union Against Cancer.
Outcomes
IL-6 levels (measured values) in the 2 groups showed a peak immediately after surgery and then decreased; however, lower values were observed in the CT group during the study period (P = .052 on POD 1 and P = .058 on POD 7) than that in the P group, and a significantly lower value was observed on POD 4 in the CT group (7.96 ± 3.50 pg/mL) than that in the P group (13.86 ± 10.21 pg/mL) (P = .048) (Figure 2a).
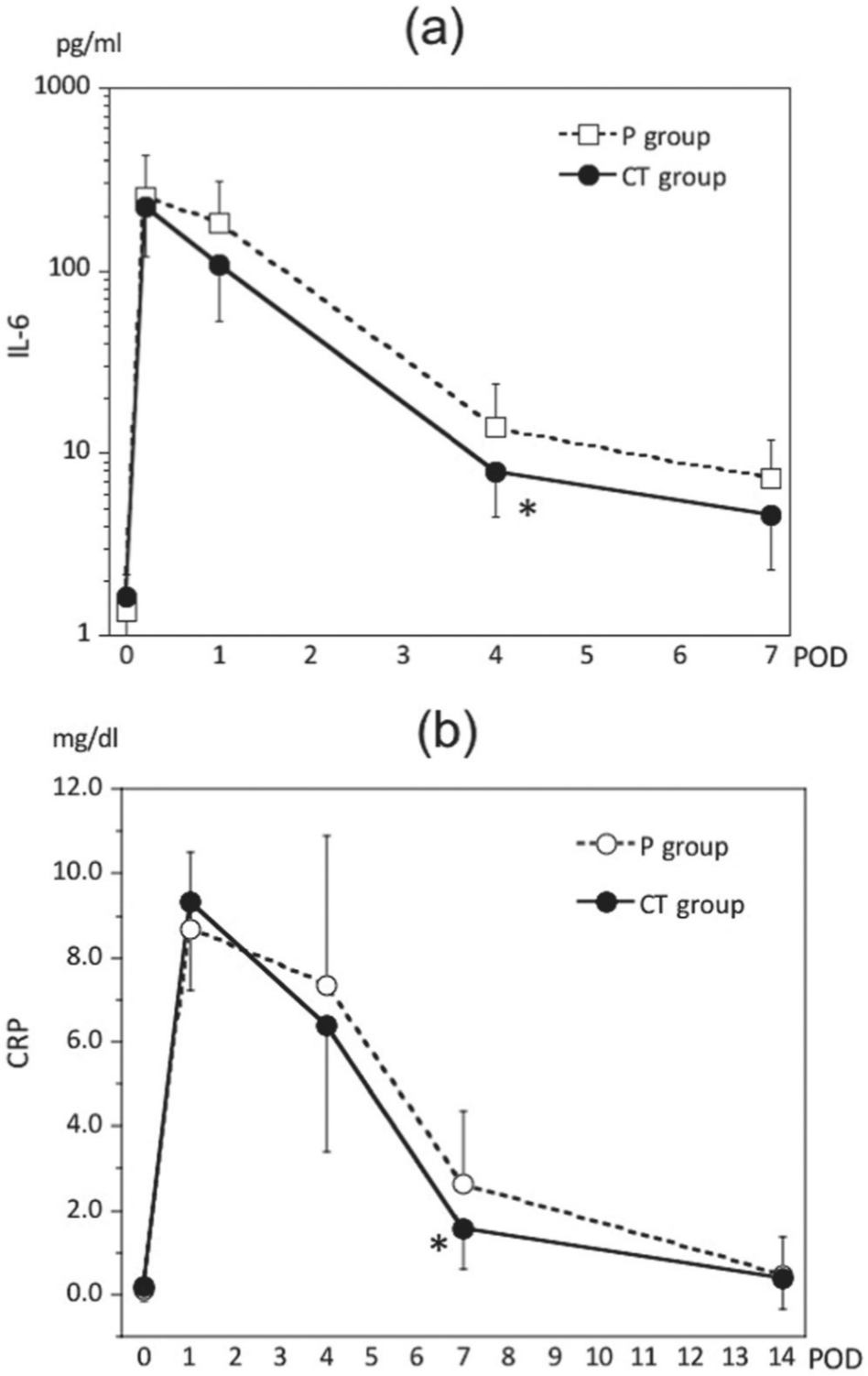
Serum interleukin (IL)–6 (a) and C-reactive protein (CRP) (b) profiles throughout the perioperative period. All data are expressed as the mean ± SD. CT, cystine and theanine; POD, postoperative day. *P < .05 vs values in the placebo (P) group.
In both groups, CRP levels (measured values) were highest on POD 1 and decreased thereafter, but this value in the CT group (1.55 ± 0.94 mg/dL) was significantly lower than that in the P group (2.62 ± 1.74 mg/dL) on POD 7 (P = .043) (Figure 2b).
Serum albumin levels (measured value) were lowest on POD 4 and subsequently increased. There were no significant differences between the 2 groups (data not shown).
Neutrophil count, neutrophil fraction, and total lymphocyte fraction (ratio relative to preoperative levels) all differed significantly between the 2 groups (Figures 3 and 4). Neutrophil count and neutrophil fraction were highest on POD 1 and tended to recover after POD 4. However, the CT group had lower values throughout the study period, and neutrophil count and neutrophil fraction were significantly different between the 2 groups on POD 4 and POD 7, respectively (P = .041 and P = .041) (Figures 3a, 4a). Total lymphocyte count began to decrease on POD 1, reached its lowest value on POD 4, and began to recover afterward; however, there were no significant differences between the 2 groups (Figure 3b). Total lymphocyte fraction in the CT group showed faster recovery after the decrease and was significantly different from the fraction in the P group on POD 7 (P = .027) (Figure 4b). Changes in WBC count were not significantly different between the 2 groups (data not shown).
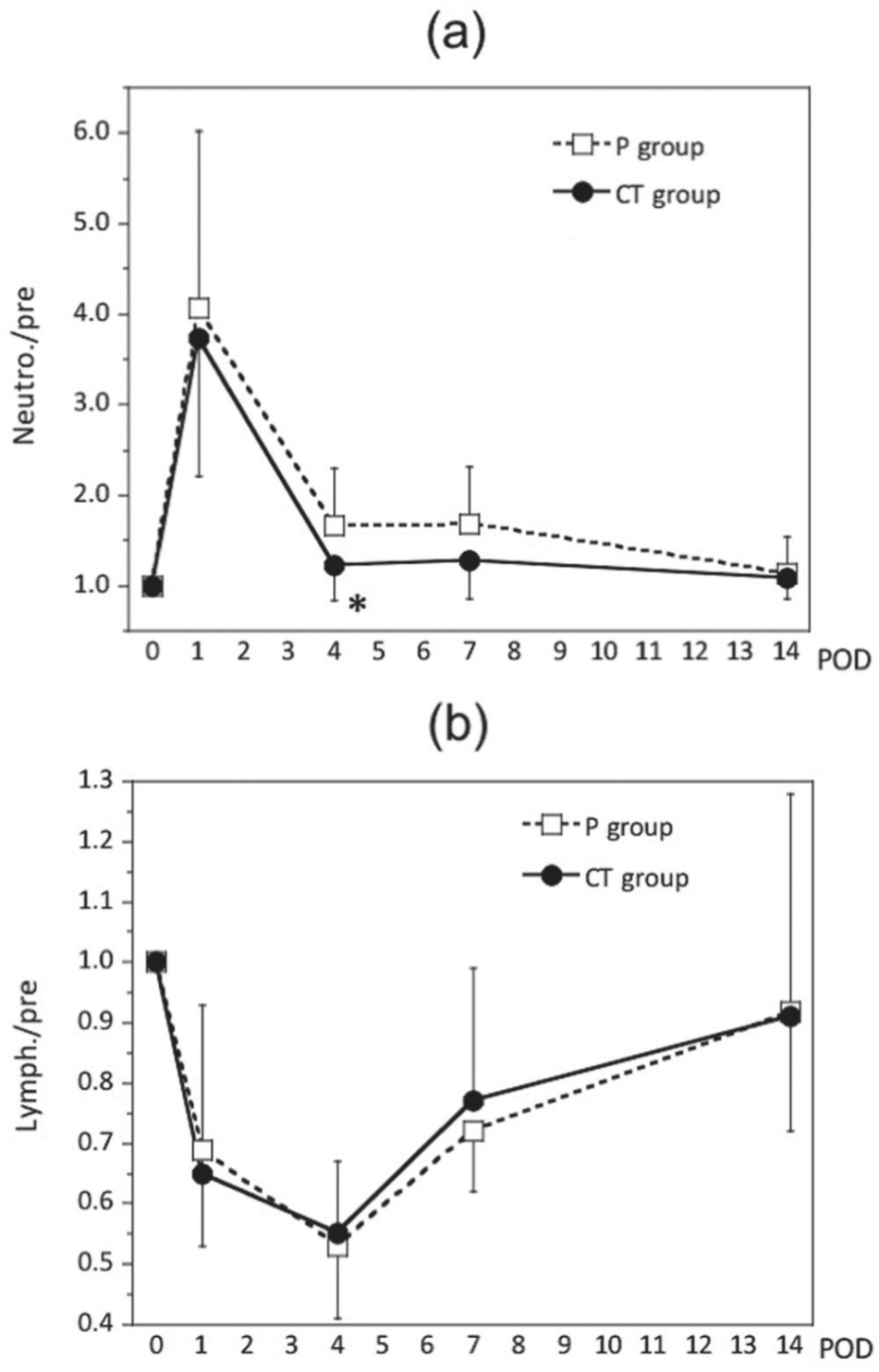
Changes in the ratios of neutrophil count (a) and lymphocyte count (b) relative to preoperative levels. All data are expressed as the mean ± SD. CT, cystine and theanine; POD, postoperative day. *P < .05 vs values in the placebo (P) group.
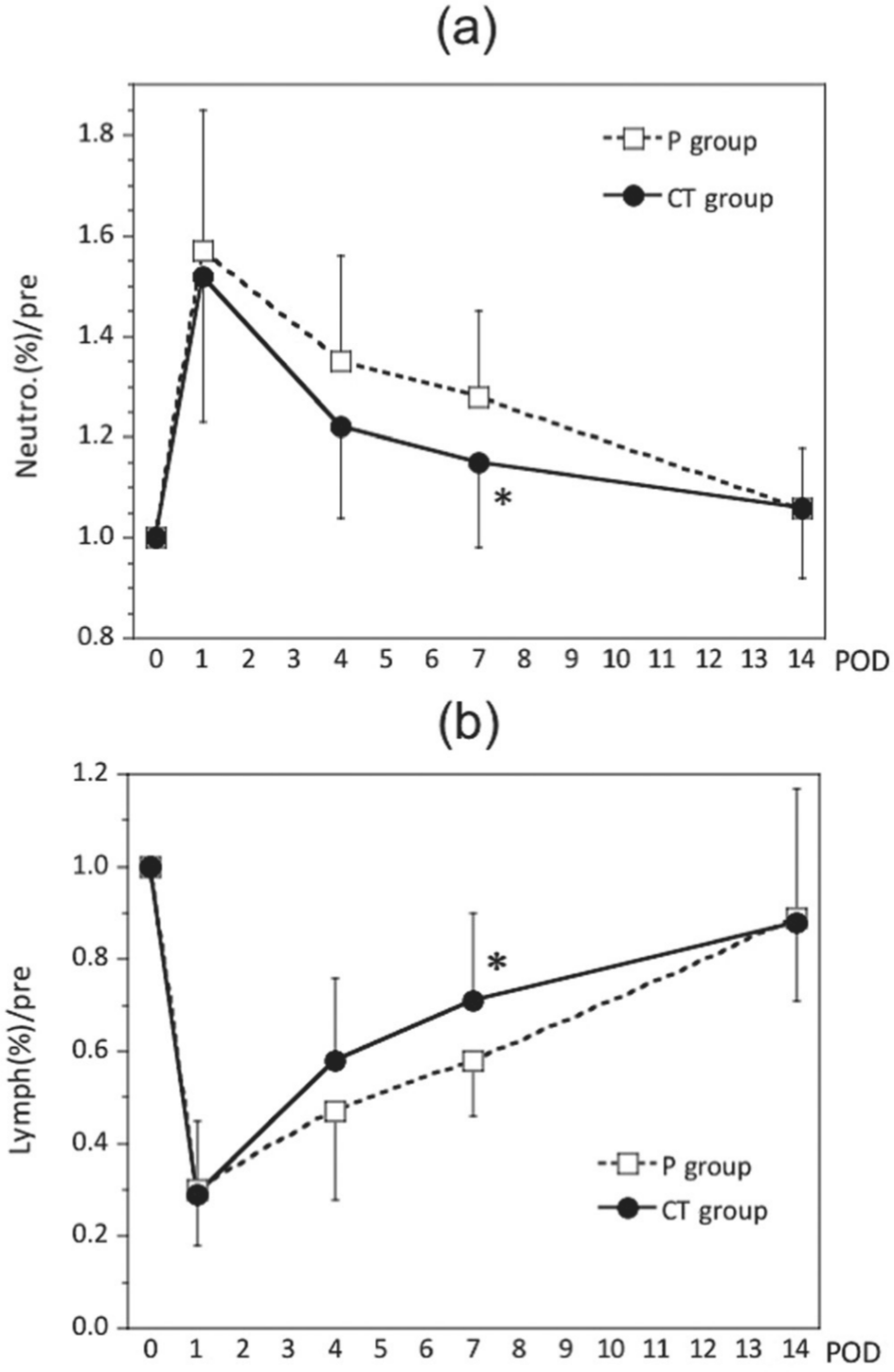
Changes in the ratios of neutrophil fraction (a) and lymphocyte fraction (b) relative to preoperative levels. All data are expressed as the mean ± SD. CT, cystine and theanine; POD, postoperative day, *P < .05 vs values in the placebo (P) group.
Preoperative REEs were 1414 ± 201 kcal in the CT group and 1408 ± 241 kcal in the P group. After surgery, REE in the P group peaked on POD 1 (1609 ± 330 kcal, 1.14 ± 0.16 [relative to preoperative levels]), with 2 peaks observed on POD 1 and POD 5 (1569 ± 233 kcal, 1.11 ± 0.16). No increase was observed on POD 1 in the CT group (1396 ± 277 kcal, 0.99 ± 0.21). A single peak was observed on POD 5 (1504 ± 159 kcal, 1.06 ± 0.18), and this value decreased to preoperative levels on POD 7. REE (relative to preoperative levels) was significantly lower in the CT group than in the P group on POD 1 (P = .043) (Table 2).
REE (kcal/d) During the Perioperative Period.
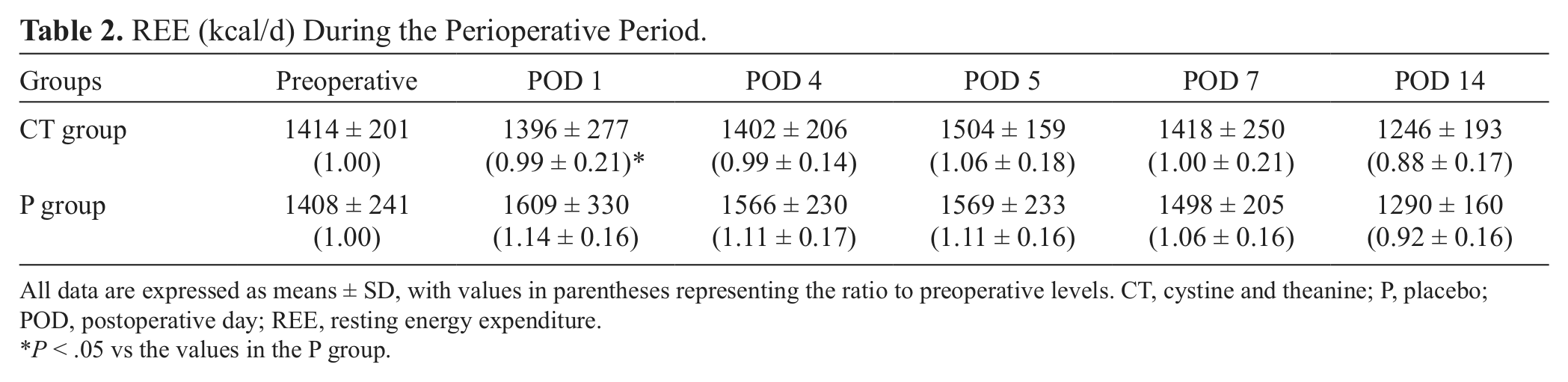
All data are expressed as means ± SD, with values in parentheses representing the ratio to preoperative levels. CT, cystine and theanine; P, placebo; POD, postoperative day; REE, resting energy expenditure.
P < .05 vs the values in the P group.
In both groups, changes in body temperature from preoperative levels peaked on the day of surgery, but a faster decrease was observed in the CT group than in the P group. A significantly lower value (P = .010) was observed on POD 5 (Figure 5). The number of times that nonsteroidal anti-inflammatory drugs (NSAIDs) were used did not differ significantly (CT group, 1.80 ± 1.78 times; P group, 2.18 ± 3.13 times).
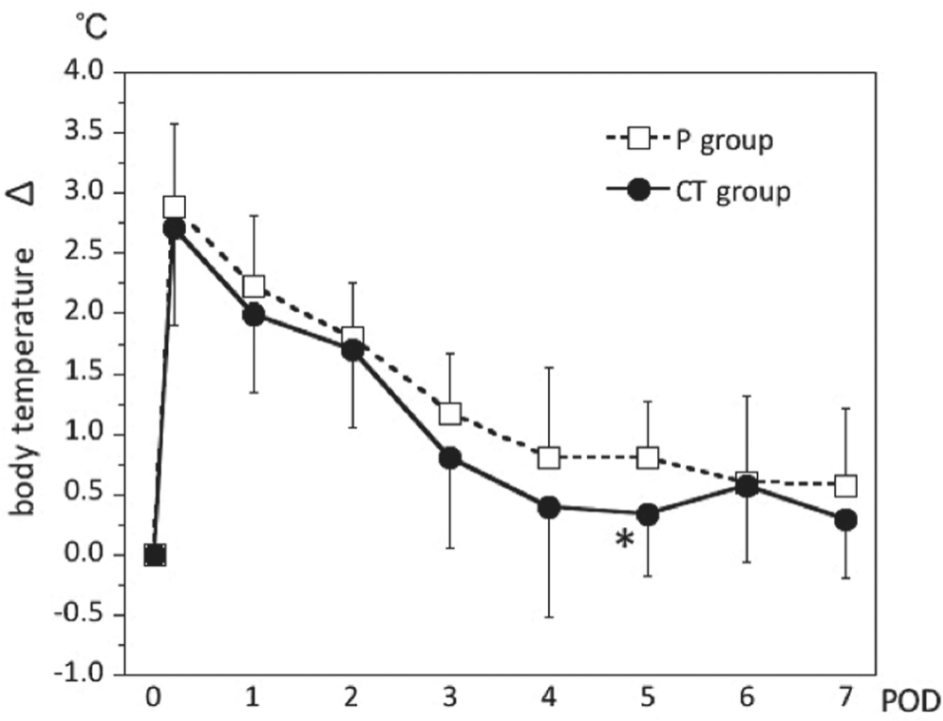
Changes in body temperature from preoperative levels over the perioperative period. All data are expressed as the mean ± SD. CT, cystine and theanine; POD, postoperative day. *P < .05 vs values in the placebo (P) group.
Discussion
The results of this study indicated that perioperative oral administration of 700 mg cystine and 280 mg theanine to gastric cancer patients treated by distal gastrectomy partly suppressed increases in REE after surgery and promoted rapid recovery of body temperature, IL-6 levels, CRP levels, neutrophil count, and total lymphocyte fraction.
In a small intestine manipulation model in mice that assumed gastrointestinal tract surgery, we also found that administering cystine and theanine for 4 days before surgery prevented GSH decreases in the intestinal tract and Peyer’s patches, as well as lowered blood IL-6 concentrations. 21 Because GSH levels in the small intestine and Peyer’s patches and IL-6 concentrations in blood were negatively correlated, GSH increases in the small intestinal wall after oral cystine and theanine were thought to play a role in suppressing increased IL-6 levels. 21 In an in vitro study using the pulmonary epithelial cells of neonatal rats, lipopolysaccharide (LPS)–induced increases in IL-6 and tumor necrosis factor (TNF)–α were suppressed in response to increased GSH induced by addition of N-acetyl cysteine. 22 This response was cancelled by depleting intracellular GSH through addition of a GSH synthesis inhibitor. 22 p38MAP kinase was shown to be related to this phenomenon. 22 In a clinical trial targeting surgery patients, cystine became essential during the perioperative period. 23 In addition, the synthetic pathway from methionine to cysteine remained inhibited in rats under surgical stress. 24 Considering these findings, suppressing GSH decreases in the small intestine with oral cystine and theanine may lead to reduced IL-6 production in response to abdominal surgery. However, further studies will be needed to elucidate the anti-inflammatory effects of cystine and theanine.
Further findings were obtained from a murine small intestine manipulation model. Locomotor activity, food intake, and weight in mice were significantly decreased after manipulation of the small intestine. 21 However, administering cystine and theanine for 4 days before surgery was associated with significantly faster recovery in these points than that of the control group. 21 Increased levels of inflammatory cytokines are known to induce various sickness behaviors, such as increased body temperature, reduced activity, and decreased appetite.25,26 Rapid decreases in elevated IL-6 levels and body temperature were observed after distal gastrectomy in the present study. These results suggest cystine and theanine may have the possibility of promoting recovery from sickness behaviors after abdominal surgery.
Five other studies on the effects of cystine and theanine administration in humans have been reported.3,27-30 The first was a study of malnourished elderly patients in whom significantly increased antibody levels after influenza vaccinations were observed with administration of cystine and theanine. 27 The second was a report of significantly increased CRP levels, increased neutrophil counts, and decreased lymphocyte counts in athletes after strenuous exercise; excessive inflammation and immune suppression after exercise were reduced by administration of cystine and theanine. 3 The third was a study of runners participating in a long-distance relay race (Eki-den) where increases in granulocytes and decreases in lymphocytes after intense endurance exercise were reduced by cystine and theanine. 28 The fourth found a significantly reduced incidence of the common cold in adults who received cystine and theanine. 29 The fifth reported that continuous administration of cystine and theanine over 2 weeks of training suppressed reductions in natural killer (NK) cell activity in bodybuilders. 30 These results suggest that cystine and theanine can suppress excessive inflammation and increase the immune response after invasive processes, such as exercise or surgery.
IL-6 is a representative proinflammatory cytokine that is produced by macrophages and lymphocytes in response to invasion, and its level indicates the strength of a systemic inflammatory reaction. In addition, IL-6 itself promotes the production of acute-phase proteins in the liver, thereby acting as a mediator and promoting further inflammatory reactions.31,32 It also acts on the hypothalamus-pituitary-adrenal axis; in recent years, IL-6 has been shown to induce immune suppression at a considerable level by stimulating secretion of glucocorticoids and catecholamines.1,31-35
CRP is an acute-phase protein that is produced by the liver in response to a stimulus by IL-6, and its levels reflect the strength of a systemic inflammatory reaction to surgical stress. In this study, both IL-6 and CRP levels recovered faster from their postsurgical increases in the CT group than in the P group, suggesting reduced systemic inflammation and reduced immune suppression.
During the perioperative period, neutrophils are released into the blood in response to the degree of inflammation, and increases in their numbers reflect a systemic inflammatory reaction to surgical stress.36-40 In addition to IL-6 and CRP levels, neutrophil counts also showed early recovery in the present study, and oral administration of cystine and theanine appeared to reduce the inflammatory reaction.
In contrast to neutrophils, lymphocytes decrease with surgical stress.40,41 In the present study, changes in total lymphocyte count did not differ significantly between the 2 groups. The blood lymphocyte fraction was significantly higher in the CT group on POD 7, but the effect of the lymphocyte fraction on the immune system during the perioperative period remains unclear. Lymphocytes play an important role in the immune system, but a detailed assessment of their function requires measuring both the cell count and cell types and activities. Therefore, further studies are necessary.
With regard to the daily dosage and duration of cystine and theanine treatment, the daily dosage used in the present study was the same as in other studies, but the duration of treatment varied from 10 to 35 days between studies.3,27-30 In the present study, these amino acids were administered for 4 days prior to surgery, over a total of 10 days; the duration of this presurgical treatment was shorter than that in other studies. Extending the dosing period before and after the procedure may clarify its efficacy for reducing excessive inflammation and lymphocyte decreases; this issue should be examined in future studies.
REE has also been reported to increase according to the degree of operative invasion and the strength of inflammation.33,42,43 The REE value 1 day after surgery was significantly lower in the CT group than in the P group, suggesting that the biological reaction against surgical stress was reduced in the CT group. Mochizuki et al 44 developed a burn injury model using guinea pigs and reported that early enteral nutrition (EN) suppressed increases in REE after injury, which emphasizes the importance of early EN during invasive procedures. Their report was the first to indicate that early EN suppresses increased REE after injuries. Other studies have reported suppression of increases in REE after gastrectomy managed with a fast-track surgery program for the first time.45,46 Results of our study suggested that administration of cystine and theanine may also reduce REE increases after gastrointestinal surgery in humans, as described in the study with fast-track surgery.45,46 There have been no previous reports on the relationship between these amino acids and REE after surgery.
Inflammation is a biological response that helps to maintain homeostasis, but excessive inflammation following excessive stress causes organ damage and compromised immune function; thus, it has a negative influence on short- and long-term prognoses.1,34-36 Local inflammation due to surgery becomes systemic within 4–8 hours and induces systemic inflammatory response syndrome (SIRS), which results in increases in proinflammatory cytokine levels, a condition known as hypercytokinemia.1,47 The effects of this excessive production of proinflammatory cytokines, such as IL-6, include propagation to other organs and organ damage (eg, tissue damage in the lungs due to neutrophil infiltration).1,47-49 In addition, there have been several reports that reduced immune function, especially reduced cellular immunity during the perioperative period induced by the hypothalamus-pituitary-adrenal system, is related to metastatic relapses of malignant tumors.1,50-52 Appropriate regulation of inflammatory reactions during the perioperative period is important in preventing the onset of organ damage and infectious complications, achieving a stable postoperative course and early recovery, and reducing the relapse rate from malignant tumors. From this perspective, an immunomodulating diet, including ω-3 fatty acids and arginine, that aims to reduce excessive inflammation during the perioperative period has been widely used in clinical settings recently.2,53-56 We could not reveal the beneficial effects of cystine and theanine on the morbidity of postoperative complications in this study because no complications were observed after surgery. We also did not assess the length of hospital stay because hospital stay was designated to be longer than 14 postsurgical days by our clinical pathway. More detailed large-scale clinical studies using an intention-to-treat analysis with more invasive operative procedures that assess complication rates, lengths of hospital stays, and long-term prognoses should be conducted in the future. Also, more research is needed to determine if there is any interaction (synergistic or antagonistic) between common immune-modulating ingredients (including ω-3 fatty acid and arginine) and cystine/theanine. However, because it has been shown that oral administration of cystine and theanine tends to reduce inflammation after uneventful gastric cancer surgery, cystine and theanine may be used for immunonutrition.
Conclusions
This study suggested that oral administration of cystine and theanine during the perioperative period may alleviate postgastrectomy inflammation and promote recovery after surgery. Future studies are expected to define the efficacy and mechanisms of cystine and theanine.
Footnotes
Financial disclosure: none declared.
This article originally appeared online on September 12, 2012.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
