Abstract
Following the issuance of the 2007 Recommendations in Publication 103, the Commission released the adult (male and female) and paediatric (newborn, 1-, 5-, 10-, and 15-year-old male and female) reference computational phantoms in Publications 110 and 143, respectively, for use in effective dose calculations. These phantoms are voxel models represented in the form of a three-dimensional array of cuboidal voxels, which were constructed from computed tomography images of people and adjusted to be consistent with the reference anatomical parameters given in Publication 89. The voxel phantoms provide anatomical improvements over the mathematical-equation-based stylised phantoms used for the previous dose coefficient (DC) calculations prior to the 2007 Recommendations. Nevertheless, the voxel phantoms, due to the nature of voxel geometry and finite voxel resolutions, have limitations in representing small and thin organs and tissues, necessitating additional supplementary stylised models such as those defined for the respiratory tract airways, the alimentary tract organ walls and stem cell layers, the lens of the eye, and the skin basal layer. To address the limitations of the voxel phantoms, Task Group 103 was charged with developing mesh-type reference computational phantoms (MRCPs) by converting the voxel phantoms into a high-quality/fidelity mesh format with anatomical improvements for the complex organs and tissues which were not represented fully in the voxel phantoms. MRCPs for the adult male and female were then developed and released recently in Publication 145. Following the release of the adult MRCPs, the current publication describes the construction of the paediatric MRCPs, the counterparts of the Publication 143 voxel phantoms. The paediatric MRCPs, like the adult MRCPs, were developed to have all the source and target tissues required for calculation of effective dose, including the micrometre-scale regions, assimilating the supplementary stylised models. These phantoms can be used directly in general purpose Monte Carlo codes, such as Geant4, PHITS, and MCNP6, fully maintaining the high fidelity of the mesh geometry in Monte Carlo dose calculations. To investigate the impact of the paediatric MRCPs, the DCs of organ dose and effective dose and specific absorbed fractions (SAFs) for some selected external and internal exposures were calculated and compared with the values calculated using the Publication 143 phantoms, the Publication 66 and 100 mathematical models for the respiratory and alimentary tracts, and the reference values of Publication 155. While some differences in the DCs and SAFs were observed for anatomically improved organs and weakly penetrating radiations, they were found to be fairly similar, indicating that the reference DCs obtained from the Publication 143 voxel phantoms for both external and internal exposures remain valid in the current ICRP dosimetry system. The Publication 143 voxel phantoms remain the primary ICRP/International Commission on Radiation Units and Measurements reference models for the calculation of reference DCs based on Publication 103 methodology. The paediatric MRCPs will be used for all calculations of reference DCs following the next set of ICRP general recommendations, and provide a resource for wider use in radiological protection applications.
© 2024 ICRP. Published by SAGE.
MAIN POINTS
1. INTRODUCTION
(1) A system of radiological protection requires appropriate dose quantities for use in the control of radiation exposures to individuals, including workers and members of the public, to prevent tissue reactions and to optimise protection from stochastic effects. For these purposes, the latest ICRP general recommendations, published in Publication 103 (ICRP, 2007), described two protection quantities: equivalent dose (HT) and effective dose (E), both given in sieverts (Sv). These protection quantities are derived from absorbed dose (DT), which is the basic physical dose quantity in grays (Gy; J kg−1). HT is calculated from DT averaged over an organ/tissue (hereafter ‘organ’) considering the relative effectiveness of different radiation types in causing stochastic effects. E is defined as the weighted sum of the values of HT [i.e. weighted by the tissue weighting factors (wT) over all organs considered to be sensitive to the induction of stochastic effects] (ICRP, 1991a). In accordance with the definition in Publication 103 (ICRP, 2007), E is computed from the gender-averaged HT values of the reference male and female at specified ages, as defined in Publication 89 (ICRP, 2002). E is the risk-related quantity in radiation protection and is used as the central protection quantity in the optimisation of protection for workers and members of the public, the setting of control criteria (dose limits, dose constraints, and reference levels), and the demonstration of regulatory compliance. (2) For calculation of the above dose quantities, the Commission adopted adult male and female reference computational phantoms as described in Publication 110 (ICRP, 2009). The Publication 110 phantoms, to be referred to in this publication as ‘P110 phantoms’, were constructed from computed tomography (CT) data of two subjects anatomically similar to the reference person and adjusted to be consistent with the reference adult anatomical parameters given in Publication 89 (ICRP, 2002). These phantoms, coupled with Monte Carlo radiation transport codes, were used to calculate the dose coefficients (DCs) for external idealised exposures in Publication 116 (ICRP, 2010), external environmental exposures in Publication 144 (ICRP, 2020b), and specific absorbed fractions (SAFs) in Publication 133 (ICRP, 2016a). The SAFs in Publication 133 were subsequently used in computation of the DCs for internal exposures due to occupational intakes of radionuclides in a series of publications (ICRP, 2015, 2016a,b, 2017, 2019). (3) The Commission also adopted 10 paediatric male and female reference computational phantoms representing 0 (i.e. newborn), 1, 5, 10, and 15 years of age in Publication 143 (ICRP, 2020a). The Publication 143 phantoms, to be referred to in this publication as ‘P143 phantoms’, were also constructed from CT data, and adjusted taking into account the reference paediatric anatomical parameters from Publication 89 (ICRP, 2002). The P143 phantoms were used to calculate the DCs for external environmental exposures in Publication 144 (ICRP, 2020b), and are currently being used by a joint task group of ICRP Committees 2 and 3 to estimate the DCs for common diagnostic x-ray imaging examinations. In addition, these phantoms were employed to calculate the paediatric SAFs in Publication 155 (ICRP, 2023). The SAFs listed in Publications 133 and 155 are currently being used for computation of the DCs for internal exposures due to environmental intakes of radionuclides by the public, and to patients from radiopharmaceuticals administered in diagnostic nuclear medicine. (4) The P110 and P143 phantoms (ICRP, 2009, 2020a) are voxel models represented in the form of a three-dimensional (3D) array of cuboidal voxels. Such voxel phantoms, based on CT images of human bodies, provide higher realism of human anatomy than previous stylised (or mathematical) phantoms in which external body and internal organ shapes are modelled using mathematical equations such as spheres, cylinders, and ellipsoids. Prior to the 2007 Recommendations, the Commission relied on various stylised phantoms (Snyder et al., 1969, 1978; Cristy, 1980; Kramer et al., 1982; Cristy and Eckerman, 1987; Stabin et al., 1995) for calculation of the ICRP reference DCs for external and internal exposures issued in Publications 30, 53, 56, 60, 61, 66, 67, 68, 69, 71, 72, 74, 80, and 100 (ICRP, 1979, 1988, 1990, 1991a,b, 1993, 1994a,b, 1995a,b, 1996a,b, 1998, 2006). (5) While providing anatomical improvements over the stylised phantoms, the ICRP voxel phantoms have limitations in the representation of small organ structures and very thin tissue layers. The voxel phantoms, composed of millimetre-scale voxels, cannot explicitly represent the micrometre-scale radiosensitive target regions and source regions in the respiratory and alimentary tracts, skin, lens of the eye, and urinary bladder (the source regions here indicate the organs, tissues, and body regions where radionuclides reside or pass that could thus irradiate target regions). With the limitations of these phantoms acknowledged in Publications 133 and 155 (ICRP, 2016a, 2023), the SAFs for electrons and alpha particles emitted from the source regions in the respiratory and alimentary tracts were computed by employing supplementary organ-specific stylised models. For the same reason, in Publication 116 (ICRP, 2010), the DCs from external idealised exposures for the lens of the eye and local skin were computed by additional simulations with supplementary stylised models. (6) In order to overcome the limitations of the ICRP voxel phantoms associated with their intrinsic limitations of using voxels, to avoid the use of supplementary stylised models, and to provide all-in-one anatomical computational phantoms, the Commission started to develop mesh-type reference computational phantoms (MRCPs) by converting the voxel phantoms to high-quality/fidelity mesh-based phantoms. Note that at the time of writing this publication, mesh geometry, either polygon mesh (PM) or tetrahedral mesh (TM), is considered to be the most advanced geometry used to construct computational phantoms beyond voxel geometry (Kainz et al., 2019). (7) Recently, many researchers have been motivated to develop computational phantoms in the PM geometry [or sometimes combined with non-uniform rational B-spline (NURBS) geometry] for adults (Christ et al., 2009; Zhang et al., 2009; Cassola et al., 2010; Lee et al., 2010; Segars et al., 2010; Hurtado et al., 2012; Gosselin et al., 2014; Dong et al., 2015; Lombardo et al., 2018; Pi et al., 2018), children, and adolescents (Lee et al., 2007, 2008, 2010; Christ et al., 2009; de Melo Lima et al., 2011; Cassola et al., 2013; Gosselin et al., 2014; Norris et al., 2014; Ma et al., 2017; Pi et al., 2018), providing more realistic representations of the human bodies than the voxel phantoms. These phantoms, however, need to be voxelised to be used in Monte Carlo codes, which leads to recurrence of the limitations of the voxel phantoms. The aim of Task Group 103, therefore, is to develop ICRP reference computational phantoms in the advanced mesh geometry which can be used directly in Monte Carlo codes, fully maintaining the advantages of the mesh geometry in Monte Carlo dose calculations. (8) Publication 145 (ICRP, 2020c) was the first publication by Task Group 103, and describes the development of adult male and female MRCPs constructed as the mesh counterparts of the P110 phantoms (ICRP, 2009). The current publication, the second by Task Group 103, describes the development of the paediatric series of MRCPs for 0-, 1-, 5-, 10-, and 15-year-olds depicting: (i) conversion of the P143 phantoms (ICRP, 2020a) to the mesh format, including the remodelling or modification of complex organs; (2) the addition of some tissue structures, such as the target cell layers for the respiratory and alimentary tracts, skin, urinary bladder, eyes, and teeth; and (3) the impact of the paediatric MRCPs in the estimation of DCs within the ICRP system. (9) The MRCPs, while closely preserving the original topology and shape of most of the organs of the voxel phantoms, present substantial improvements in the anatomy of small organs, and include all the necessary source and target tissues defined by the ICRP system, assimilating the supplementary stylised models, such as those defined for the respiratory tract airways, the alimentary tract organ walls and stem cell layers, the lens of the eye, and the skin basal layer. The MRCPs, which are in the TM format, are no longer bounded by the limitations of voxel geometry for the representation of very small and/or thin structures. These phantoms can also be used directly in the general purpose Monte Carlo codes (e.g. Geant4, PHITS, and MCNP6) without any additional processing (i.e. voxelisation) as was needed for earlier work with mesh phantoms (Yeom et al., 2019b, 2020). (10) For calculation of reference equivalent and effective DCs based on Publication 103 (ICRP, 2007) methodology, the P110 and P143 voxel phantoms (ICRP, 2009, 2020a) remain the primary ICRP/International Commission on Radiation Units and Measurements (ICRU) reference anatomical models. The MRCPs will replace the voxel phantoms for all calculations of reference DCs following the next set of ICRP general recommendations. The MRCPs have applications beyond the calculation of reference DCs. The new mesh phantoms are highly deformable, and can be used to create phantoms of various body sizes and postures for use in retrospective emergency or accidental dose reconstruction (Lee et al., 2019; Yeom et al., 2019a; Choi et al., 2020). The deformation capability of the phantoms can also facilitate the virtual calibration of whole-body counters to account for the body size of individuals in efficiency calibration. The mesh phantoms can also be used directly to produce physical phantoms with 3D printing technology. In addition, it is relatively easy to model detailed structures in the phantoms and, therefore, the new phantoms could find applications in medicine and other areas requiring sophisticated organ models. One of the aims of this publication is to assist those who wish to implement the phantoms for their own applications; therefore, detailed data on the phantoms in the PM format as well as the TM format are provided in the supplementary electronic data that accompany the printed publication, together with some input examples of the Monte Carlo codes. (11) The present chapter (Chapter 1) explains the main background for the construction of the paediatric MRCPs. Chapter 2 focuses on the organs of the P143 phantoms (ICRP, 2020a) for which the anatomical description has been improved significantly in the paediatric MRCPs. Chapter 3 describes the general procedure for the conversion of the P143 phantoms to the mesh format, including the remodelling or modification of complex organs. Chapter 4 describes the adjustment of the converted mesh models to the reference values for the mass, density, and elemental composition of organs inclusive of blood content. Chapter 5 describes the inclusion of the thin target and source regions in the skin, alimentary tract, respiratory tract, and urinary bladder. Chapter 6 describes the general characteristics of the paediatric MRCPs. Finally, Chapter 7 investigates the impact of the improved morphology of the paediatric MRCPs on the calculation of DCs for external and internal exposures. (12) A detailed description of the paediatric MRCPs is given in Annexes A–F. Annex A presents a list of the organs/structures [list of identification (ID) numbers], together with the assigned media, densities, and masses. Annex B presents a list of the phantom media and their elemental compositions. Annexes C and D list the source and target regions, respectively, together with their acronyms and ID numbers. Annex E provides depth distributions for selected organs from the front, back, left, right, top, and bottom, along with the respective data for the P143 phantoms (ICRP, 2020a). Annex F provides chord-length distributions (CLDs) between selected pairs of source and target organs, along with the data for the P143 phantoms. Annex G presents selected transverse, sagittal, and coronal slice images of the paediatric MRCPs. In Annexes H and I, the DCs and SAFs calculated with the paediatric MRCPs for some selected idealised external and internal exposure cases are compared with the values calculated with the P143 phantoms and the Publication 155 values (ICRP, 2023). Annex J describes the contents of the supplementary electronic data that accompany the printed publication, including the detailed phantom data and examples of input files for three general purpose Monte Carlo codes (i.e. Geant4, PHITS, and MCNP6) which are widely used for dose calculation in the field of radiation protection.
2. IMPROVEMENTS IN PAEDIATRIC MESH-TYPE REFERENCE COMPUTATIONAL PHANTOMS
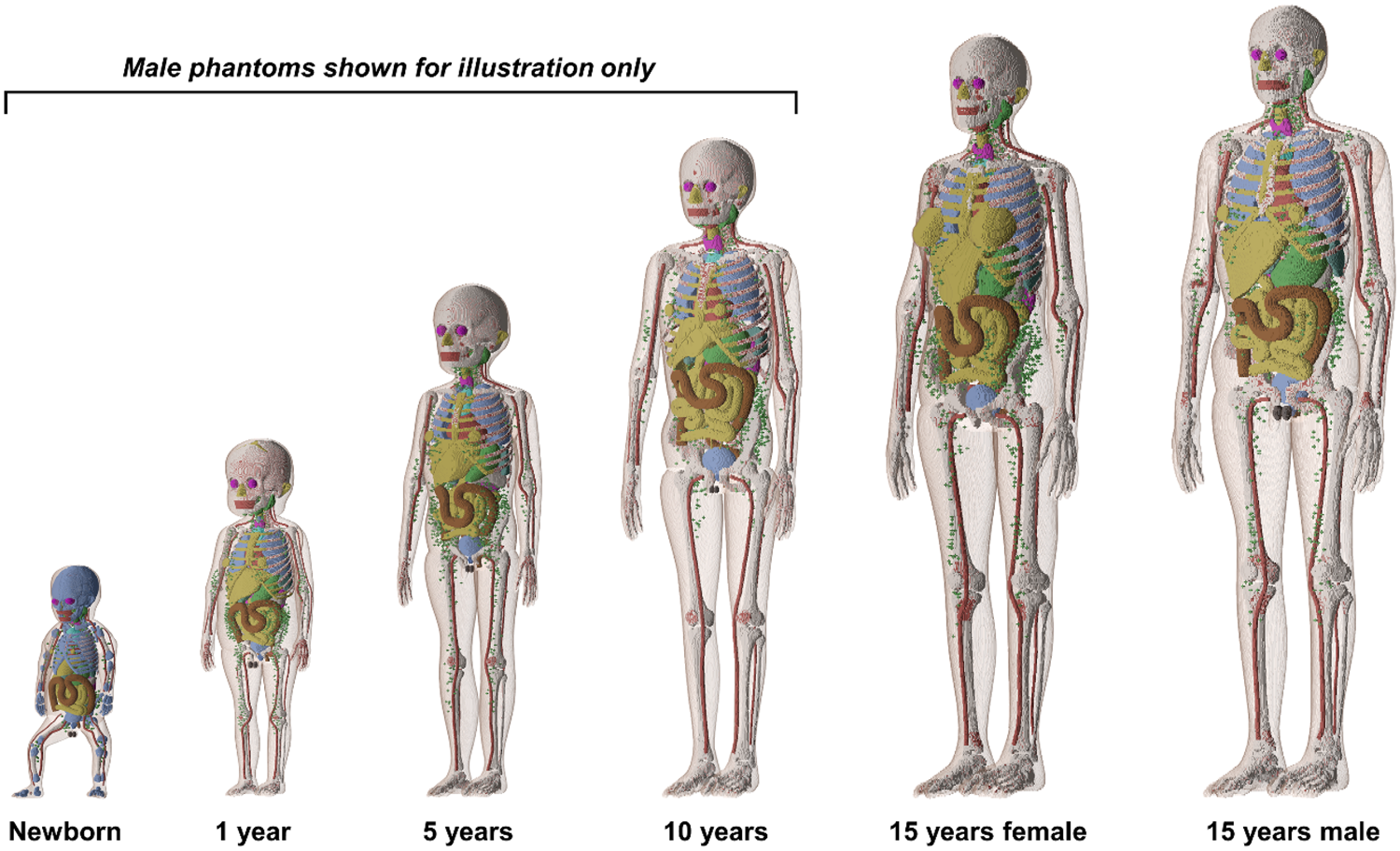
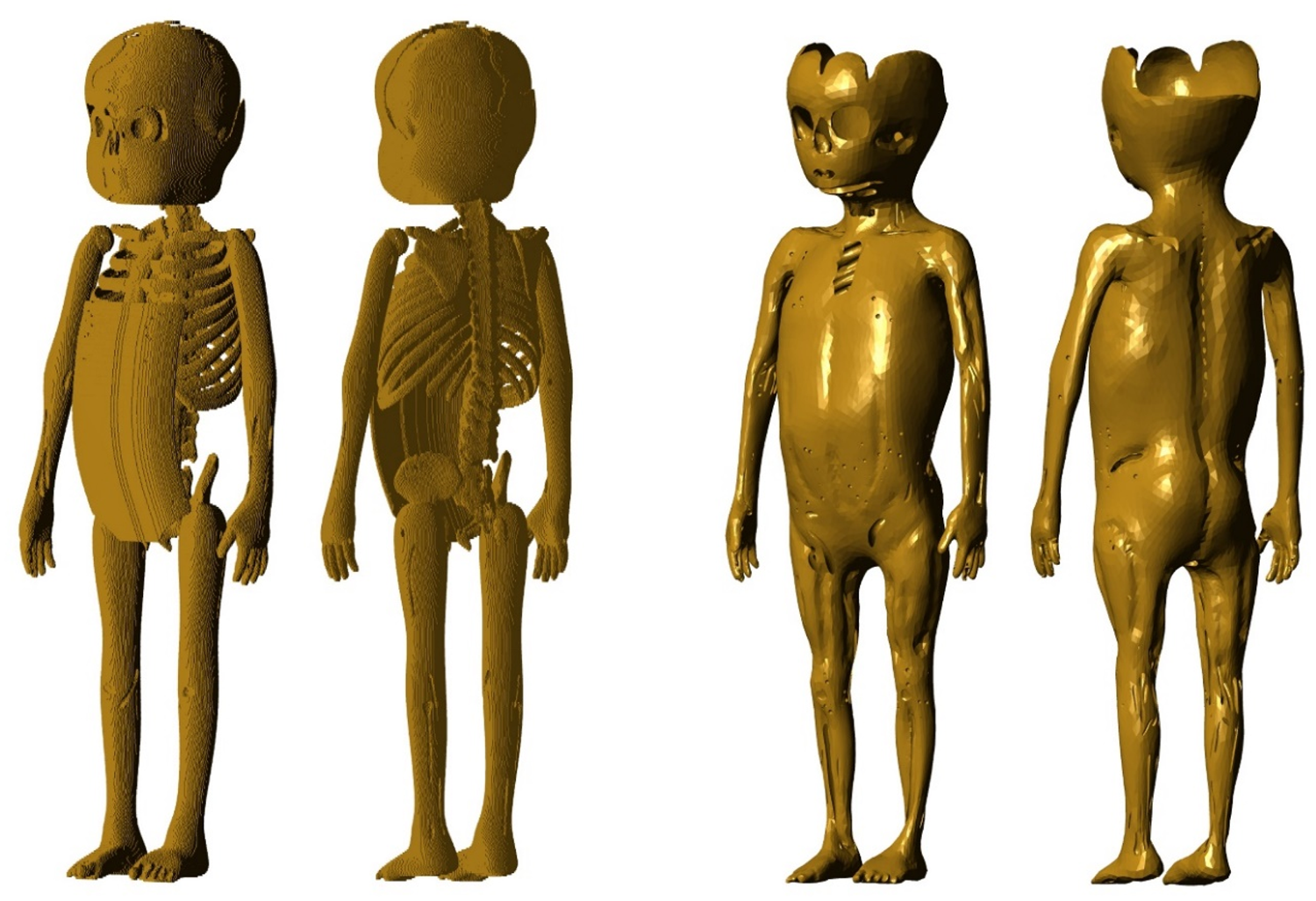
(13) Following the development of the P110 adult voxel phantoms (ICRP, 2009), the Commission released the P143 paediatric voxel phantoms (ICRP, 2020a) (see Fig. 2.1) for computation of the protection quantities for children and adolescents. The P143 phantoms comprise 10 phantoms representing the reference male and female at the five different ages defined for the reference person – 0 (newborn), 1, 5, 10, and 15 years. These phantoms were derived from a corresponding series of boundary representation paediatric phantoms with NURBS/PM surfaces based on human CT images developed in collaboration between the University of Florida (UF) and the National Cancer Institute (NCI) (Lee et al., 2010), which were adjusted to conform with Publication 89 reference data on the paediatric anatomical parameters (ICRP, 2002) and then voxelised to be used with Monte Carlo simulation codes. The male and female newborn, 1-year-old, 5-year-old, and 10-year-old phantoms are anatomically identical, except for the reproductive system organs (e.g. gonads). Their voxel resolution (for the x, y, and z axes) ranges from 0.0663 × 0.0663 × 0.0663 cm3 for the newborn to 0.1250 × 0.1250 × 0.2832 cm3 for the 15-year-old male (ICRP, 2020a).
Publication 143 paediatric voxel phantoms (ICRP, 2020a). The figure shows male phantoms alone for ≤10 years, and does not display muscle and adipose tissue for better visualisation of internal organs. (14) While providing significant anatomical improvements over the previous stylised models, the P143 phantoms (ICRP, 2020a) have limitations, mainly resulting from the inherent nature of voxel geometry and finite voxel resolutions. Small organ structures (e.g. lens of the eye) and very thin tissue layers (e.g. stem cell targets in alimentary and respiratory tract organs) are not fully modelled, and for calculations of DCs, some organs are represented by supplementary stylised models. These limitations and some phantom characteristics were addressed in the paediatric MRCPs, as summarised in the following paragraphs. (15) In the P143 phantoms (ICRP, 2020a), the skin is represented by the outermost single voxel layer of each transverse slice, resulting in many spaces between adjacent transverse slices. Through these spaces, radiation incident at non-zero angles relative to the transverse slices can reach some radiosensitive organs (such as breasts, spongiosa, and testes) directly, leading to a significant overestimation of DCs for those tissues for weakly penetrating radiations. Furthermore, the skin masses of the P143 phantoms do not conform ideally with their reference values; the skin masses of the newborn, 1-year-old, and 15-year-old phantoms are smaller by up to approximately 33%, whereas those of the 5-year-old and 10-year-old phantoms are larger by up to approximately 20%, than the reference skin masses in Publication 89 (ICRP, 2002). In the paediatric MRCPs developed in the present work, as in the adult MRCPs of Publication 145 (ICRP, 2020c), the skin is modelled as a continuous layer and the mass exactly matches the reference value. Similarly, the limitations of other thin structures (e.g. cortical bone and alimentary tract organs) were addressed in the same way. (16) For the skin, the basal cell layer of the epidermis is assumed to be the target for radiogenic risk (ICRP, 1977, 2010, 2015). This skin target layer, however, could not be represented separately in the P143 phantoms (ICRP, 2020a) due to their voxel resolutions being limited to hundreds of micrometres to several millimetres. Consequently, the entire skin (i.e. epidermis plus dermis) is defined as a single voxel layer, and used to approximate dose to the skin target layer. Considering the small wT of the skin (0.01), this approximation is acceptable for calculation of the effective dose for most penetrating radiations. However, for external exposures to weakly penetrating radiations (e.g. alpha and beta particles, and low-energy photons), this approximation could lead to significant errors in both skin dose and effective dose calculations (Yeom et al., 2016, 2017). In the paediatric MRCPs, as in the adult MRCPs, this problem was addressed by defining the target layer in the skin. (17) The computation of SAFs for the respiratory and alimentary tracts requires modelling of the cell layers of radiosensitive tissues and similarly scaled source regions (ICRP, 1994a, 2006). However, these target and source regions were not modelled in the P143 phantoms (ICRP, 2020a) due to the finite voxel resolutions. Therefore, for computation of the paediatric SAFs in Publication 155 (ICRP, 2023), a number of mathematical-equation-based stylised models used in Publications 66 and 100 (ICRP, 1994a, 2006) to describe the respiratory and alimentary tract organs, respectively, were employed separately for charged particles. In the paediatric MRCPs, as in the adult MRCPs, the target and source regions in the respiratory and alimentary tracts were defined (Choi et al., 2022, 2023), allowing the calculation of SAFs directly using the reference phantoms without using separate supplementary models. (18) Similarly, for the urinary bladder, the basal cells of the lining epithelium are assumed to be the relevant target cells for radiogenic risk (Colin et al., 2009), and taken as the target region for calculation of the SAFs for non-penetrating radiations (i.e. alpha and beta particles), emitted from the bladder contents (Eckerman and Veinot, 2018). Therefore, in the paediatric MRCPs, as in the adult MRCPs, the target layer in the urinary bladder wall was defined explicitly to conform with following the age-dependent target depth and thickness data. (19) The complex structure of the eye was not represented in the P143 phantoms (ICRP, 2020a) due to the finite voxel resolutions. In the adult MRCPs (ICRP, 2020c), this issue was addressed by incorporating the detailed eye model of Behrens et al. (2009), which had been adopted in Publication 116 (ICRP, 2010) for calculation of the lens DCs of adults for idealised external radiation fields. However, the direct use of scaled versions of the adult eye model for children and adolescents would not adequately represent age-related changes in the dimensions of the eye and the lens. In the present work, therefore, a set of paediatric eye models was first developed following the approach used for the development of the eye model by Behrens et al., and then incorporated into the paediatric MRCPs (Han et al., 2021). (20) The teeth of the P143 phantoms (ICRP, 2020a) consist of a single homogeneous region with a simplified geometry (i.e. ellipsoid). In the paediatric MRCPs, detailed tooth models were incorporated based on existing high-quality tooth models (Shin et al., 2021). The tooth models of the paediatric MRCPs include four inner tooth structures (i.e. enamel, dentin, pulp, and cementum), and reflect the anatomical changes with age. These revised models are well suited for retrospective dosimetry based on electron paramagnetic resonance (IAEA, 2002; Shin et al., 2021). (21) The shapes and/or topologies of some organs [i.e. the thyroid, extrathoracic (ET) region, sacrum, muscle, and exterior body contours of all phantoms; the liver of the newborn phantoms; the crania of the newborn and 1-year-old phantoms; and the spine and hand/foot bones of 1-year-old and older phantoms] were modified to achieve better anatomical realism. The mass and density of the colon contents were adjusted to the reference values of Publication 89 (ICRP, 2002), and the position of the colon was also altered to provide a better representation of typical anatomy. The mass of the blood in the large vessels was increased significantly, improving the anatomical representations. (22) The organ masses of the P143 phantoms (ICRP, 2020a) are based on the reference masses listed in Table 2.8 of Publication 89 (ICRP, 2002), which represent the organ parenchymal masses alone (i.e. excluding the intra-organ blood masses). Note that in a living person, a significant proportion of the total blood is situated in the small vessels and capillaries in the organs, which should be considered in phantom construction. Therefore, in the paediatric MRCPs, as in the adult MRCPs, the organs were adjusted to the reference masses including the intra-organ blood content based on the blood distribution in Wayson et al. (2018) adopted in the calculation of the paediatric SAFs (ICRP, 2023). In the SAF calculations using the P110 (ICRP, 2009) and P143 (ICRP, 2020a) voxel phantoms, scaling of SAFs for self-irradiation geometries was performed (ICRP, 2016a, 2023). Such scaling will not be necessary with the MRCPs, the organ masses of which are consistent with the reference masses inclusive of blood content.
3. CONVERSION OF THE PAEDIATRIC VOXEL-TYPE REFERENCE COMPUTATIONAL PHANTOMS TO MESH FORMAT
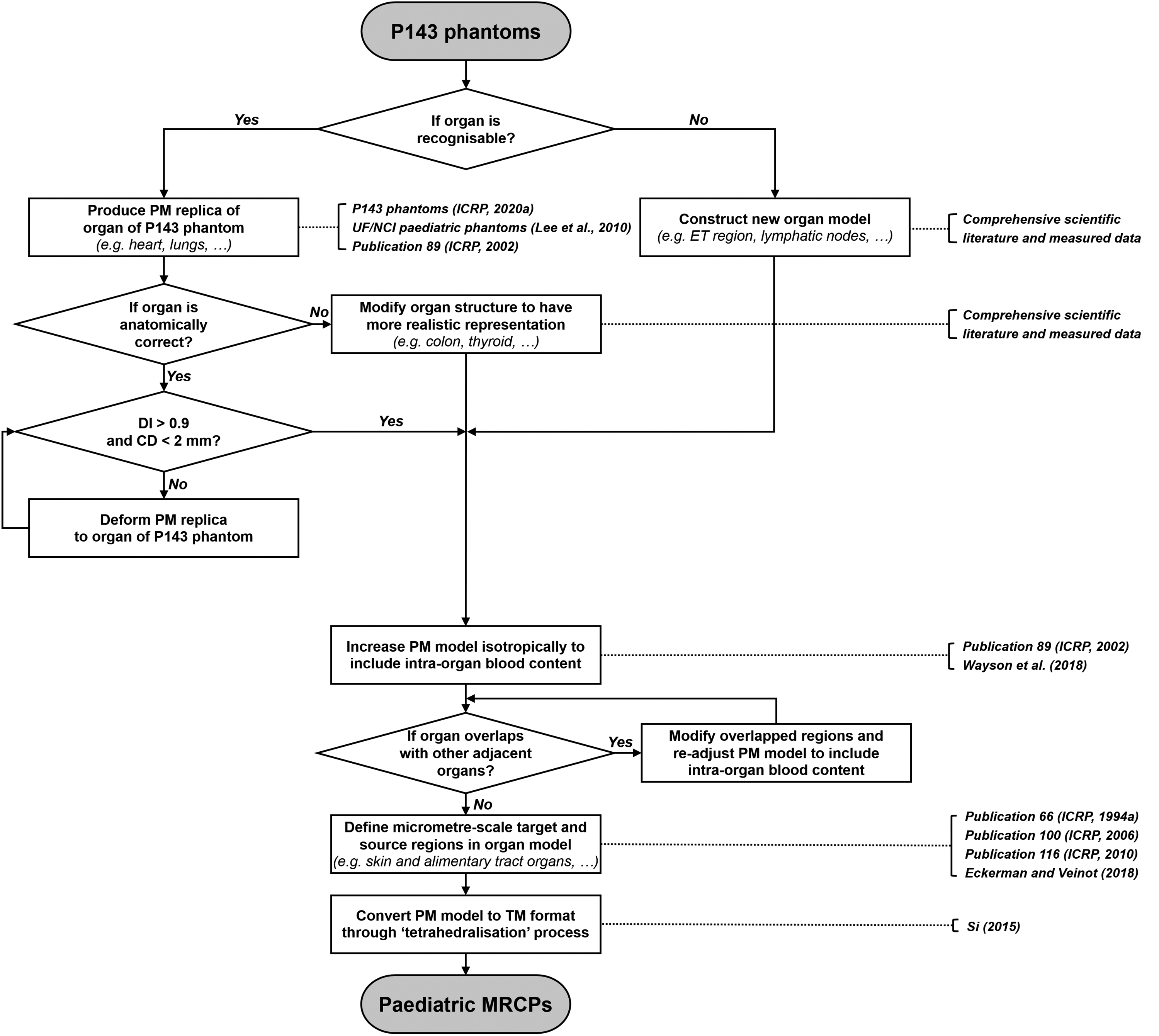
(23) Fig. 3.1 shows how the paediatric MRCPs were developed from the P143 phantoms (ICRP, 2020a). Detailed explanations for each step will be given in the following chapters. Schematic for constructing the paediatric mesh-type reference computational phantoms (MRCPs) from the Publication 143 (P143) phantoms (ICRP, 2020a). PM, polygon mesh; CD, centroid distance; DI, Dice index; TM, tetrahedral mesh; ET, extrathoracic; UF, University of Florida; NCI, National Cancer Institute.
3.1. Simple organs and tissues
(24) Most of the organs of the paediatric MRCPs [i.e. adrenal glands, trachea, main bronchi (generation 1), brain, breasts, gallbladder, stomach, small intestine, heart, kidneys, liver, lungs, oesophagus, gonads (testes or ovaries), pancreas, pituitary gland, prostate, salivary glands, spinal cord, spleen, tongue, thymus, tonsils, ureters, urinary bladder, and uterus], which have an easily recognisable and anatomically reasonable shape, were reproduced using both the P143 phantoms (ICRP, 2020a) and the UF/NCI paediatric phantom series (Lee et al., 2010) which are the source of the P143 phantoms. First, the UF/NCI phantoms were used to produce initial PM models of the simple organs, which were then refined using the approaches employed for the adult MRCPs (ICRP, 2020c). The refined models were then matched to the organs of the P143 phantoms, which are consistent with the reference values in Publication 89 (ICRP, 2002), to faithfully preserve the original organ topology. Subsequently, the models were further adjusted to allow for the intra-organ blood content. The PM models were converted to the TM format at the final stage of phantom construction with the other organ models. (25) During the conversion process, the PM models were matched to the P143 phantoms (ICRP, 2020a) by monitoring two indices which show the geometric similarity of two models: Dice index (DI), the overlapping volume fraction of two objects; and centroid distance (CD), the distance between the centroids of two objects. The matching criteria were set for the DI value to be >0.9 of the maximum achievable Dice index (MADI), and for the CD values to be <2 mm. Note that MADI was introduced to account for the intrinsic difference in the geometry format (i.e. voxel vs PM) of the two models (ICRP, 2020c). (26) Publication 89 (ICRP, 2002) provides sex-averaged reference masses for most of the organs for the ages up to and including 10 years (i.e. the same reference masses for the same age regardless of sex). Sex-specific reference masses (i.e. different reference masses depending on sex) are only provided for two organs (i.e. brain for 5- and 10-year-old phantoms, and thymus for 10-year-old phantom). However, the P143 phantoms (ICRP, 2020a) of these ages were developed using sex-averaged masses for all the organs, except for the reproductive system organs (e.g. gonads). In the present work, the paediatric MRCPs were developed using sex-specific reference organ masses where available. Therefore, the refined PM models of the brain and thymus were adjusted to match the sex-specific reference organ masses. (27) In the P143 phantoms (ICRP, 2020a), some organs and contents (e.g. brain and heart contents for newborn phantom, thymus for 1-year-old phantom, and gastrointestinal contents for all age phantoms) have masses significantly different from the reference values given in Publication 89 (ICRP, 2002), mainly due to the small space allowed for these organs and contents (Lee et al., 2010). In the paediatric MRCPs, these organs and contents were adjusted to match the reference values. During the adjustment process, these tissues were enlarged isotropically, preserving their original shapes and centroids, with slight adjustment of the adjacent organs. (28) In the P143 phantoms (ICRP, 2020a), the oesophagus contents are not defined, and in principle, estimation of the SAFs of the oesophagus wall for radiations from the oesophagus contents is not possible. This limitation was addressed in the paediatric MRCPs following the same approach used for the adult MRCPs (ICRP, 2020c); that is, the oesophagus contents were defined in the middle of the oesophagus with the volume derived from the morphological information (i.e. length and diameter) given in Publication 100 (ICRP, 2006). To maintain the oesophagus wall volume, the outer diameter of the oesophagus wall was increased slightly (i.e. by 3–7 mm). (29) The liver of the P143 newborn phantoms is unrealistically long in the vertical direction, as a result of extending the liver model downwards to match the reference mass of Publication 89 (ICRP, 2002). To address this issue, the liver was reproduced directly from the original CT image data which had been used for construction of the P143 phantoms (ICRP, 2020a). The liver was then adjusted to match the reference mass, enlarged isotropically to preserve the original shape of the liver. During this reconstruction of the liver for the newborn phantoms, the lower part of the ribs had to be moved outwards slightly (i.e. in the lateral direction); the resulting rib cage was found to be within the range of typical shapes (Devlieger et al., 1991). (30) In the present work, the organ models were mostly visualised, handled, and refined with Rapidform software (INUS Technology Inc., Seoul, Korea), with two exceptions: conversion of NURBS surfaces to PM surfaces; and generation of the blood and colon passages and the eyes, which was accomplished with Rhinoceros 5.0 software (Robert McNeel, Seattle, WA, USA).
3.2. Skeletal system
(31) The majority of the bones of the paediatric MRCPs (i.e. upper humeri, lower humeri, ulnae, radii, clavicles, cranium, upper femora, lower femora, tibiae, fibulae, patellae, mandible, pelvis, ribs, scapulae, and sternum) were produced directly from the UF/NCI phantom series (Lee et al., 2010) using the same procedure as for the simple organs described in Section 3.1. Some bones which were not represented properly in the UF/NCI phantoms were modified or remodelled using other existing high-quality models, and analysing scientific literature and measured data (Choi et al., 2021). (32) The spine models (cervical, thoracic, and lumbar regions) of the 1-year-old and older MRCPs were replaced with high-quality PM models produced from serially sectioned photographic images of adult male and female cadavers (Park et al., 2005) after several adjustments. These adult models are considered to be applicable to children and adolescents, except for the newborn, as ossification of the individual vertebrae of the spine is complete within approximately 5 months after birth (Taylor, 1975), and only the anterior–posterior curvature of the spine changes with growth. First, the adult models were scaled down in all directions, matching the spine height of the P143 phantoms (ICRP, 2020a). The individual vertebrae were then translated and rotated, matching the original spine topology (i.e. position and curvature) of the P143 phantoms. Finally, the individual vertebrae were scaled isotropically to match the reference mass of Publication 89 (ICRP, 2002). Likewise, the hand and foot bones of the 1-year-old and older MRCPs were replaced with high-quality PM models based on micro-CT images of adult male and female cadavers (http://dk.kisti.re.kr/) in a similar way as for the spine. Note that models were available for both male and female, and thus were used separately. (33) The crania of the newborn and 1-year-old MRCPs were modified according to data on fontanelle sizes and suture widths determined from the scientific literature. The anterior fontanelle sizes of the newborn and 1-year-old were taken from the data provided by Noorizadeh et al. (2015) and Duc and Largo (1986), respectively. The posterior fontanelle size of the newborn was obtained from the data of Faix (1982). Note that the posterior fontanelle is closed 2–3 months after birth (Usman et al., 2011), which was thus defined only in the newborn MRCPs. The suture widths were determined by scaling the newborn and 1-year-old values in proportion to the head circumferences (Li et al., 2015). The crania of the newborn and 1-year-old MRCPs were matched to the target values within 5% difference. (34) Some minor improvements were also made to several bones. The sacra were modified to take account of anatomical changes in the sacral crest and foramina with growth (Kim et al., 2014). The sterna of the 1-, 5-, and 10-year-old MRCPs were extended in the vertical direction to conform to the locations of the ribs. The mandibles of the 1- and 10-year-old MRCPs were adjusted slightly for normal occlusion of the teeth. (35) The bones of the paediatric MRCPs were finally divided into cortical bone, spongiosa, and medullary cavity, in a similar way as for the P143 phantoms (ICRP, 2020a). (36) The costal cartilage, intervertebral discs, and pre-osseous cartilage were produced directly from the UF/NCI phantoms. The intervertebral discs of the 1-, 5-, 10-, and 15-year-old phantoms were adjusted to their positions in the new spine models produced from the high-quality PM models (Park et al., 2005). For the newborn and 1-year-old MRCPs, the fontanelle cartilage was adjusted to the modified cranium. The other cartilages, which were not modelled explicitly in the paediatric MRCPs, were included in the residual soft tissue (RST), which will be discussed in Section 4.3. (37) Fig. 3.2 shows the skeletal system of the newborn and 10-year-old male MRCPs, including the cortical bone, spongiosa, medullary cavity, and explicitly defined cartilage. Skeletal system of newborn male (upper) and 10-year-old male (lower) mesh-type reference computational phantoms: cortical bone (white), spongiosa (red), medullary cavity (black), costal cartilage and intervertebral discs (yellow), and pre-osseous cartilage (blue).
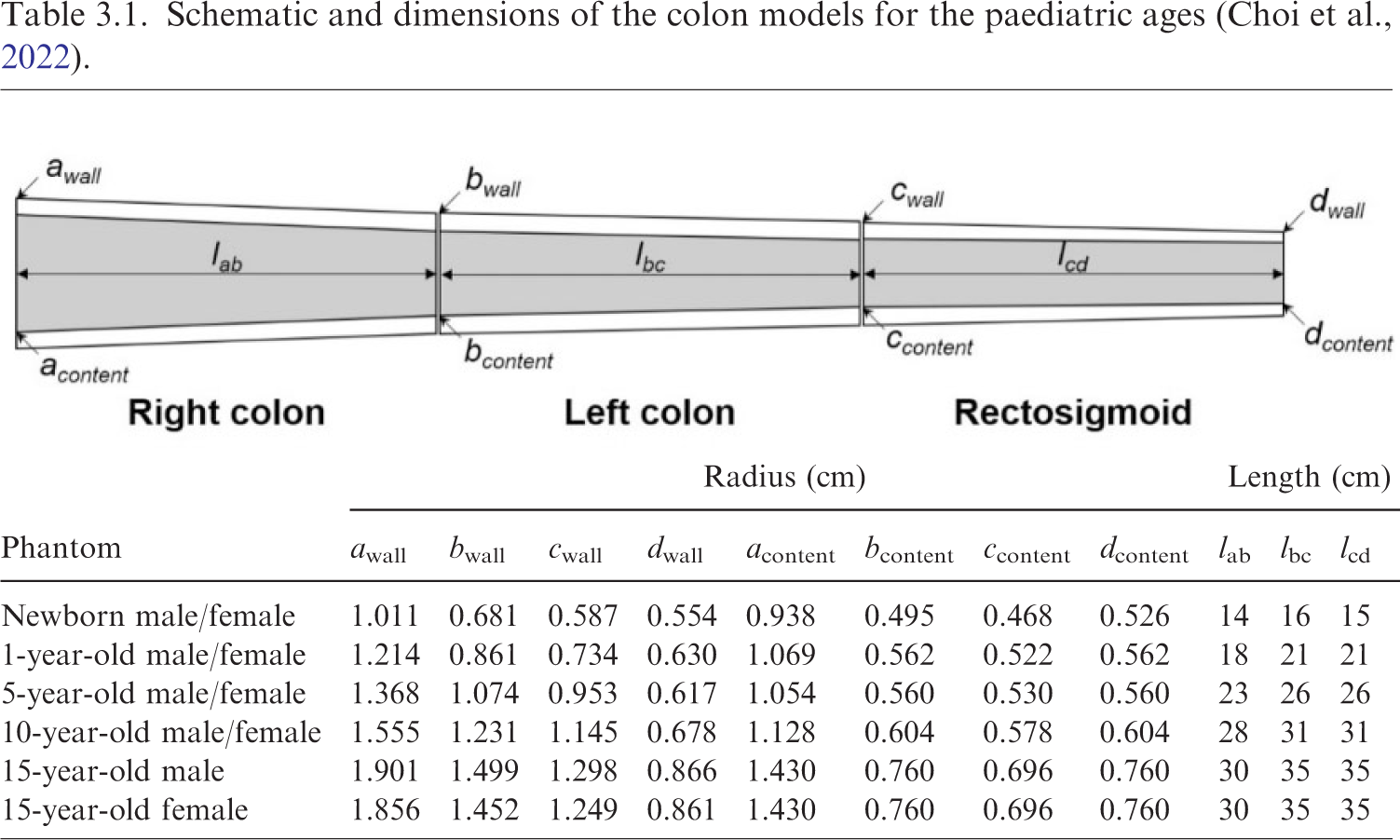
3.3. Colon
(38) In the P143 phantoms (ICRP, 2020a), the wall of the colon has the reference mass and length given in Publication 89 (ICRP, 2002). However, either the mass or the density of the colon contents was significantly different from the reference values, due to the limitation of the modelling approach using a simple cylinder (Lee et al., 2010). In addition, the middle part of the transverse colon showed significant curvature. (39) For the paediatric MRCPs, therefore, the colon was remodelled (Choi et al., 2022). First, the colon was modelled as a connection of three truncated curved cones, representing the right, left, and rectosigmoid colons, for the wall and the contents, respectively, as shown in Table 3.1. The top and bottom radii of each truncated cone were determined by a numerical approach, involving iteration of calculations, to connect the three truncated cones most smoothly while matching all the reference values (i.e. mass, length, and density of the right, left, and rectosigmoid colon, for both the wall and the contents). The details of the numerical approach can be found in Choi et al. (2022). The colon model was then subdivided into six regions based on the length data given in Publication 89 (ICRP, 2002): the right colon into the ascending and the transverse right colon; the left colon into the transverse left and the descending colon; and the rectosigmoid colon into the sigmoid colon and the rectum. Finally, the colon was produced in the NURBS format and then converted to the PM format for incorporation into the paediatric MRCPs, while addressing the issue of the transverse colon curvature. (40) Fig. 3.3 shows the developed colon of the 10-year-old male MRCP, together with the P143 phantom (ICRP, 2020a). Schematic and dimensions of the colon models for the paediatric ages (Choi et al., 2022). Colon of the Publication 143 phantom (ICRP, 2020a) (left) and the paediatric mesh-type reference computational phantom (right) for the 10-year-old male with adjacent organs.
3.4. Thyroid
(41) In the P143 phantoms (ICRP, 2020a), the thyroid shows some anatomical abnormalities (e.g. the thickness of the thyroid isthmus for all phantoms, and the position of the thyroid for the newborn and 1-year-old phantoms). (42) For the paediatric MRCPs, the thyroid was therefore modified to have typical shape and position (Yeom et al., 2022). First, the isthmus thickness, width, and height were determined from the scientific literature. The isthmus thickness at each age was taken from the data provided by Sea et al. (2019). The isthmus width and height of the newborn were obtained from the data of Ozguner and Sulak (2014), and those of the 1-, 5-, 10-, and 15-year-old phantoms, due to the absence of data, were derived by linear interpolation between the newborn (Ozguner and Sulak, 2014) and adult data (Tong and Rubenfeld, 1972; Harjeet et al., 2004; Joshi et al., 2010; Ozgur et al., 2011; Won et al., 2013). The thyroid shape hardly changes after reaching adulthood (Harjeet et al., 2004; Sultana et al., 2011; Won et al., 2013) and, therefore, the adult age was set as 18 years for the interpolation. The thyroid isthmus of the paediatric MRCPs was matched to the target values within 5% difference. Next, the thyroid was placed in the typical position (i.e. in front of the second and third tracheal cartilage) (Ellis, 2007; Naqshi et al., 2016), matching the depth beneath the skin surface to the target values derived from the equations given in Likhtarev et al. (1995), again within 5% difference. (43) Fig. 3.4 shows the thyroid of the 1-year-old male MRCP, together with the corresponding P143 phantom (ICRP, 2020a).
Thyroid of the Publication 143 phantom (ICRP, 2020a) (upper) and the paediatric mesh-type reference computational phantom (lower) for the 1-year-old male.
3.5. Lymphatic nodes
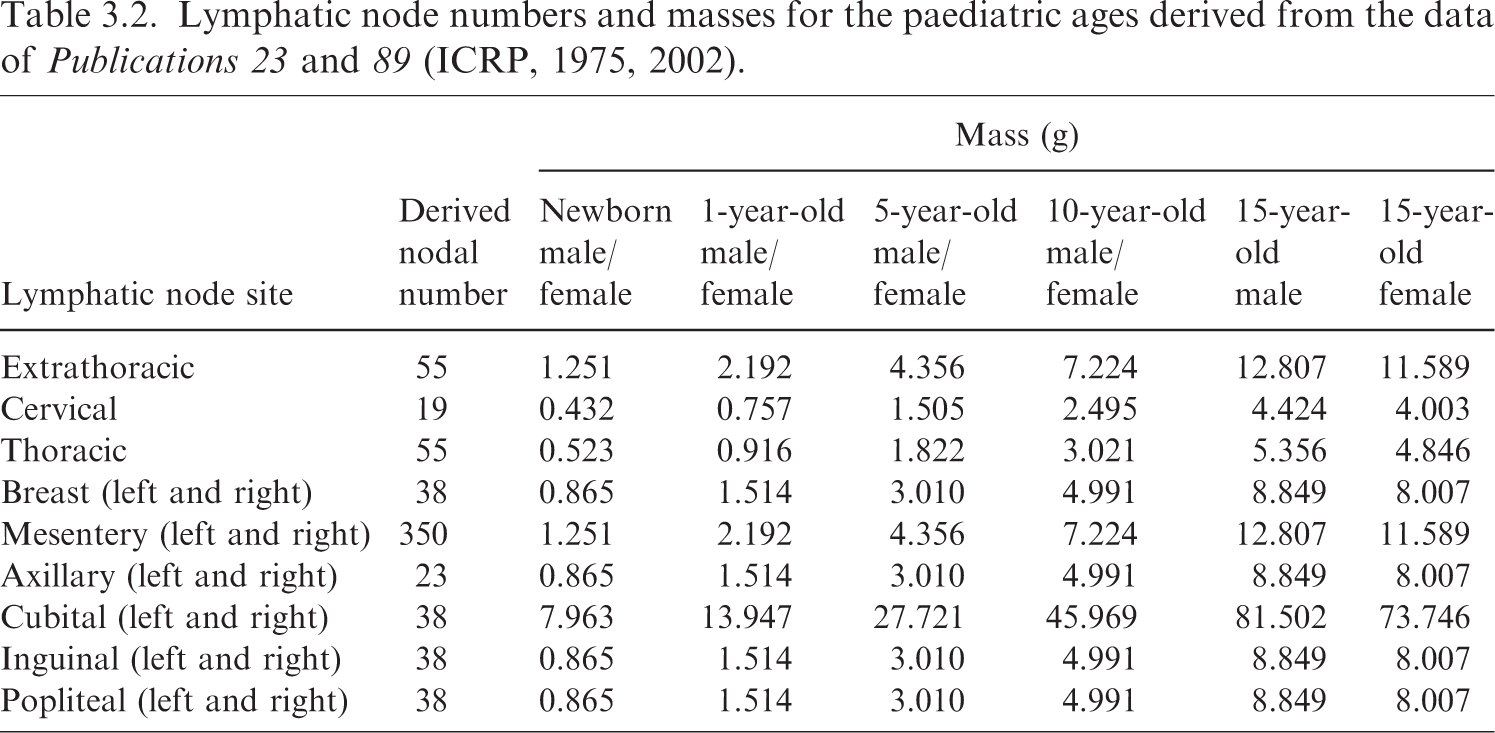
(44) Due to the complexity of their distribution, the lymphatic nodes had to be generated using a modelling approach. The lymphatic nodes of the paediatric MRCPs were generated by employing the same method used for the adult MRCPs (ICRP, 2020c), but using the paediatric lymphatic node data given in Table 3.2, which were derived from data from Publications 23 and 89 (ICRP, 1975, 2002), and were also employed for the SAF calculations in Publication 155 (ICRP, 2023). (45) Fig. 3.5 shows the lymphatic nodes of the 15-year-old male MRCP, together with the corresponding P143 phantom (ICR, 2020a). Lymphatic nodes of the Publication 143 phantom (ICRP, 2020a) (left) and the paediatric mesh-type reference computational phantom (right) for the 15-year-old male. Lymphatic node numbers and masses for the paediatric ages derived from the data of Publications 23 and 89 (ICRP, 1975, 2002).
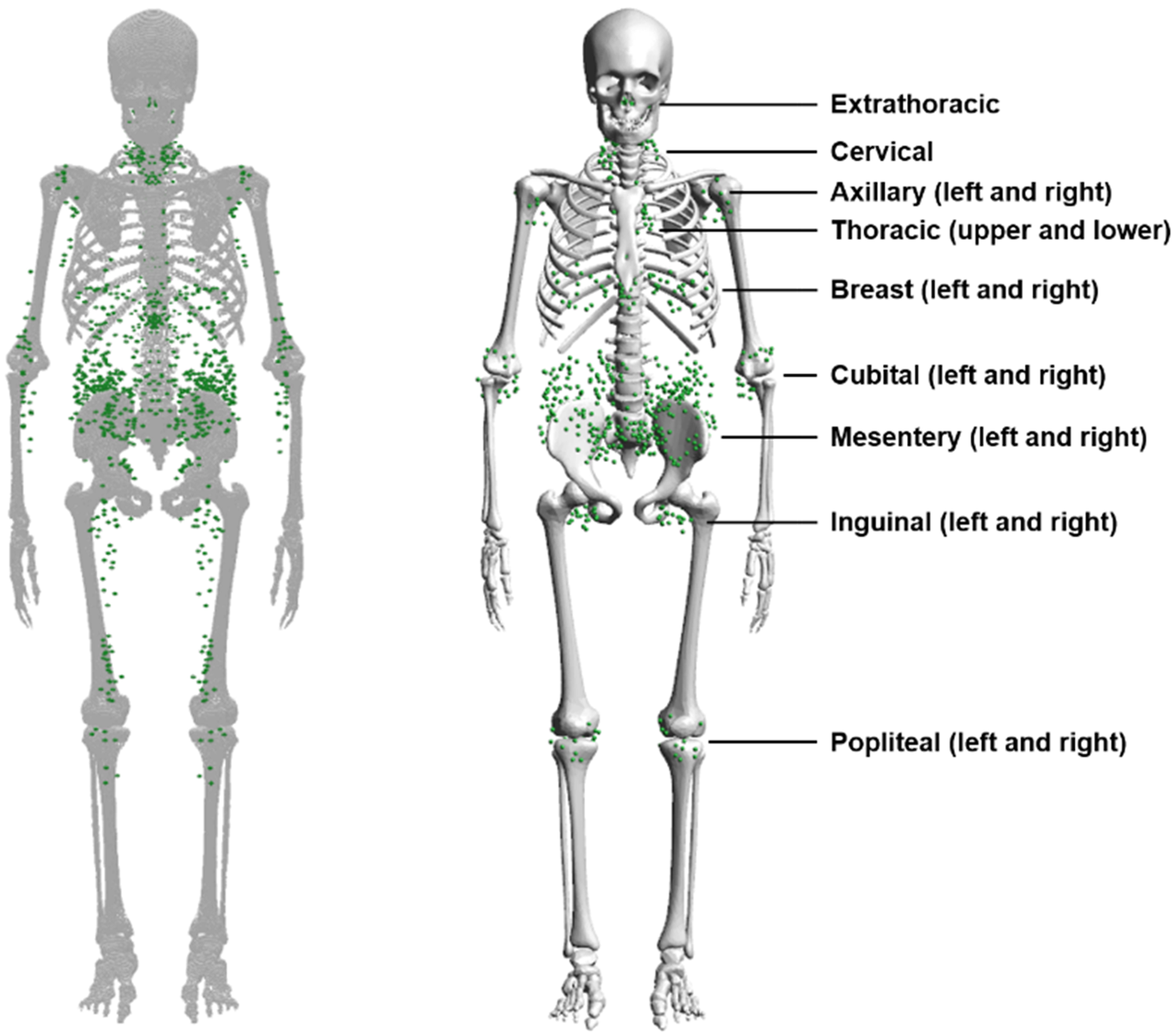
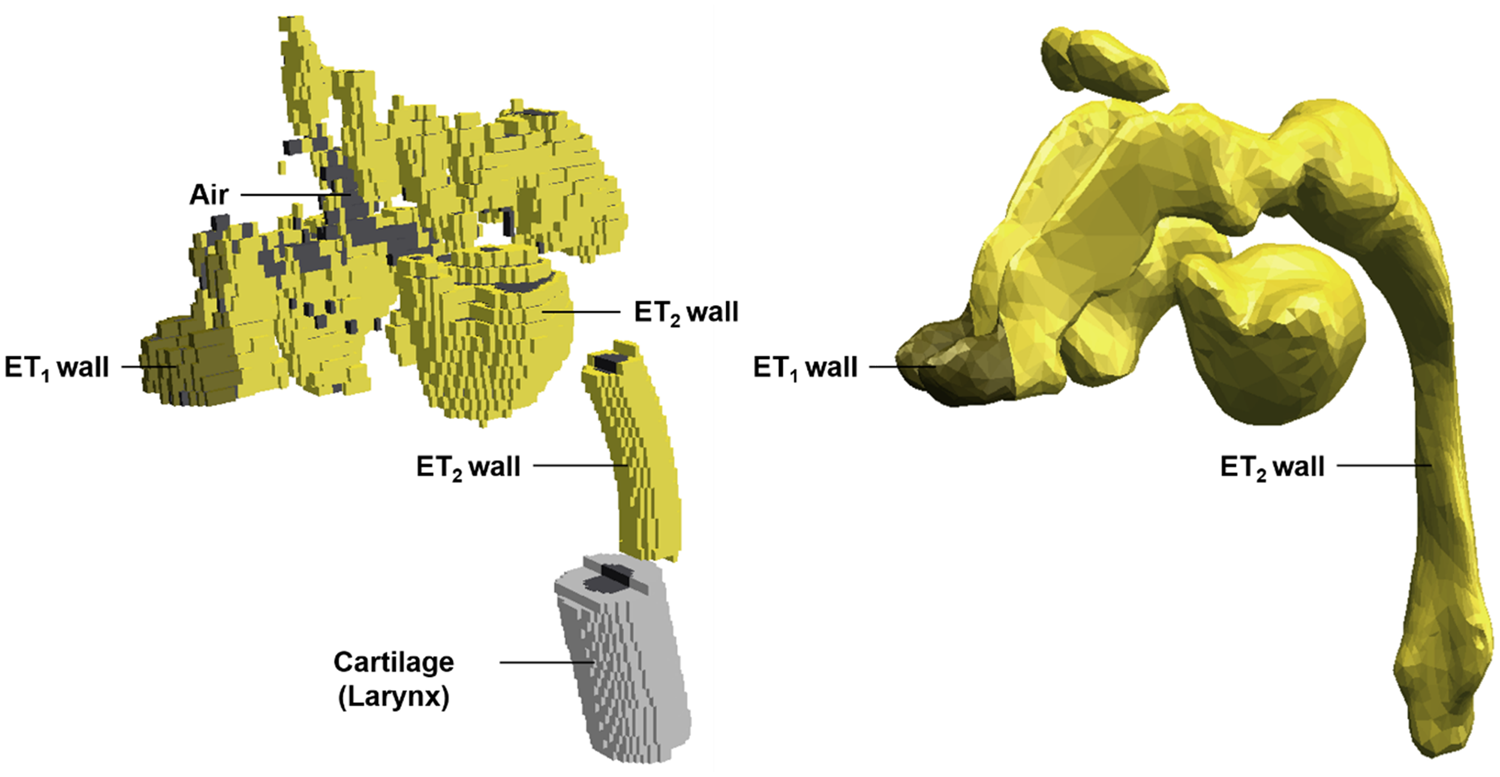
3.6. Extrathoracic region
(46) The ET region, consisting of the anterior nose (ET1) and the posterior nasal passages, pharynx, and larynx (ET2), is not represented properly in the P143 phantoms (ICRP, 2020a), mainly because its intricate and narrow conduit geometry was not clearly distinguishable in the original CT image data. In addition, the larynx was not included in the ET2 region, but was tagged as cartilage during the modification of the UF/NCI phantoms to the P143 phantoms. (47) For the paediatric MRCPs, the ET region was, therefore, remodelled (Choi et al., 2023). First, the anterior nose and the posterior nasal passages were modified manually, referring to Kozak and Ospina (2014). Next, the remaining parts (i.e. pharynx and larynx) were replaced with new models which were produced by scaling down those of the adult MRCPs (ICRP, 2020c), taking account of differences in the trachea mass between adults and children (ICRP, 1994a). Note that the adult models were available for both male and female, and thus were used separately. The details of the scaling process can be found in Choi et al. (2023). The modified regions (i.e. anterior nose and posterior nasal passages) and replaced regions (pharynx and larynx) were then connected, and were finally divided into the ET wall and inner air, matching the volume ratio of the adult models due to the absence of reference values for children and adolescents in Publication 89 (2002). (48) Fig. 3.6 shows the ET region of the 5-year-old male MRCP, together with the corresponding P143 phantom (ICRP, 2020a).
Extrathoracic region (ET1 and ET2) of the Publication 143 phantom (ICRP, 2020a) (left) and the paediatric mesh-type reference computational phantom (right) for the 5-year-old male. ET1, anterior nose; ET2, posterior nasal passages, pharynx, and larynx.
3.7. Eyes
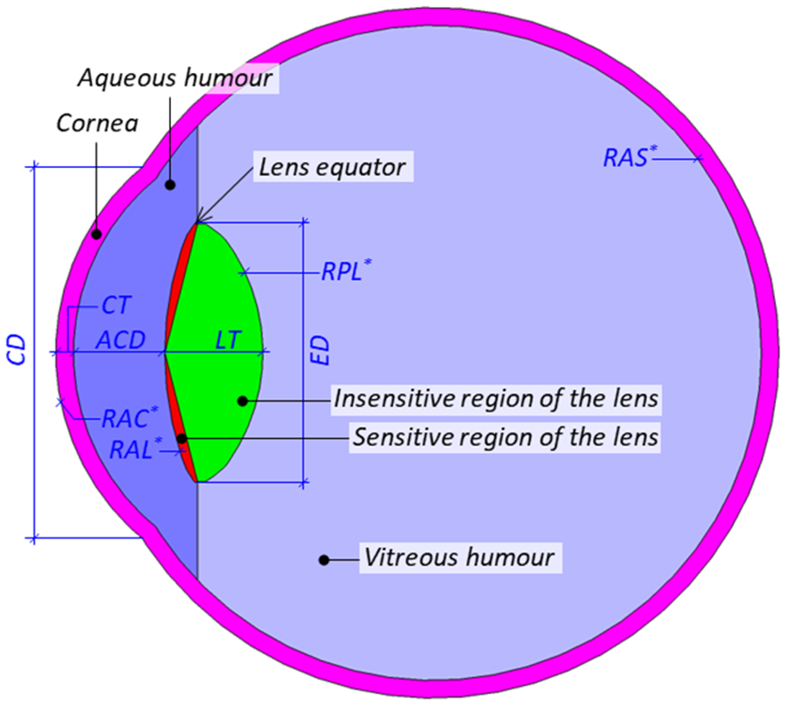
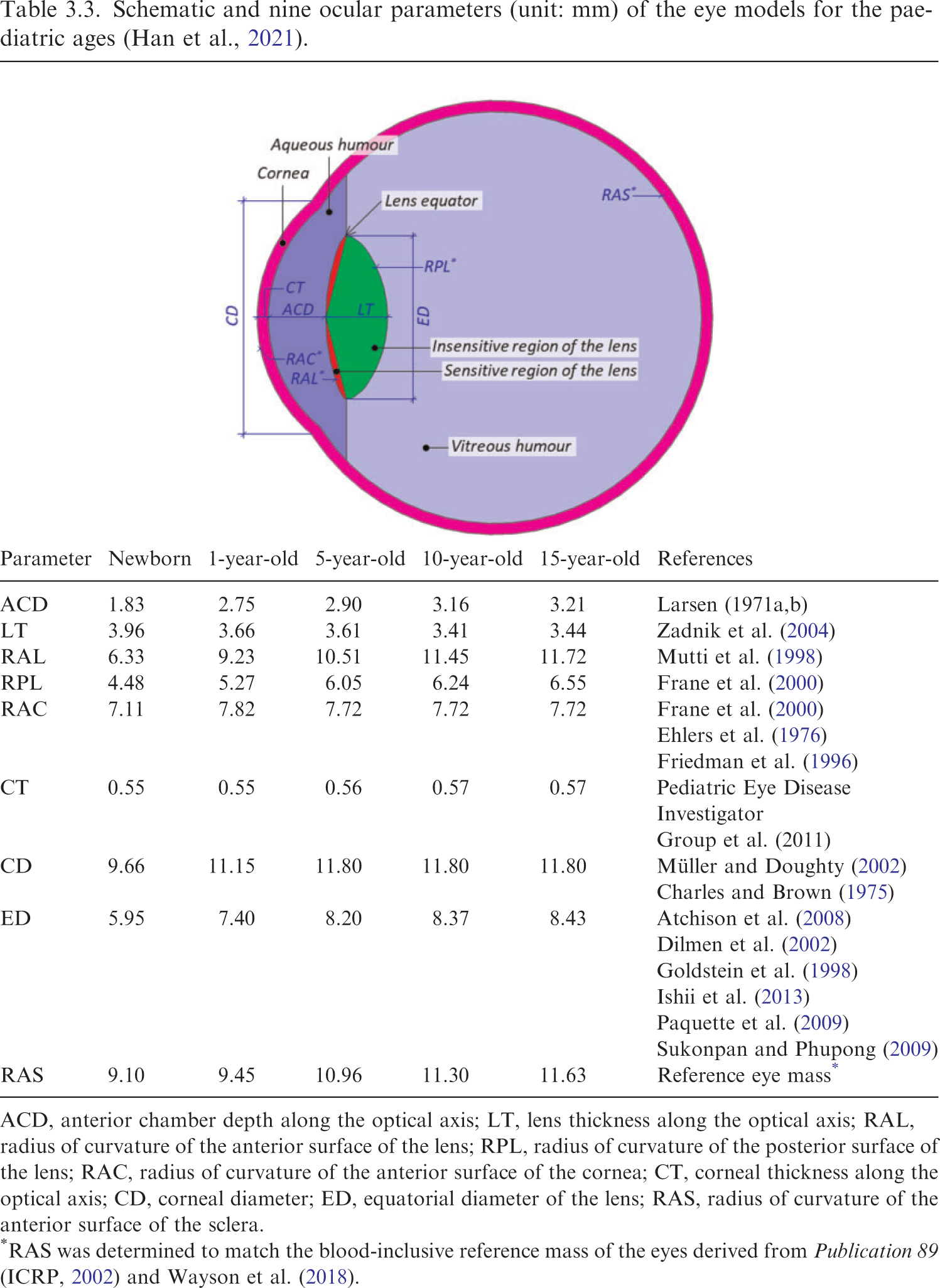
(49) Due to their finite voxel resolutions (i.e. hundreds of micrometres to several millimetres), the detailed structures of the eyes could not be modelled fully in the P143 phantoms (ICRP, 2020a). (50) For the adult MRCPs (ICRP, 2020c), this issue was addressed by incorporating the detailed eye model of Behrens et al. (2009), which was adopted in Publication 116 (ICRP, 2010) for calculation of the reference lens DCs for adults. However, this eye model is not appropriate for the assessment of dose to the lens of the eye of children and adolescents, considering that the ocular dimensions of paediatric eyes are significantly different from scaled versions of adult eyes (Ronneburger et al., 2006; Augusteyn, 2010). For example, while the lens thickness (LT) decreases until 11 years of age and then increases with age, the anterior chamber depth (ACD) of the eye increases at early ages, and then remains relatively stable or decreases with age, resulting in larger LT and smaller ACD for adults than for 15-year-olds (Shih et al., 2011; Phu et al., 2020). (51) Therefore, detailed eye models for children and adolescents at five different ages (i.e. newborn, 1 year, 5 years, 10 years, and 15 years) were developed for the paediatric MRCPs following the approach used for the development of the eye model by Behrens et al. (2009), based on nine ocular parameters of the eye: ACD along the optical axis, LT along the optical axis, radius of curvature of the anterior surface of the lens, radius of curvature of the posterior surface of the lens, radius of curvature of the anterior surface of the cornea, corneal thickness along the optical axis, corneal diameter, equatorial diameter of the lens, and radius of curvature of the anterior surface of the sclera (Han et al., 2021). Schematic and nine ocular parameters (unit: mm) of the eye models for the paediatric ages (Han et al., 2021). ACD, anterior chamber depth along the optical axis; LT, lens thickness along the optical axis; RAL, radius of curvature of the anterior surface of the lens; RPL, radius of curvature of the posterior surface of the lens; RAC, radius of curvature of the anterior surface of the cornea; CT, corneal thickness along the optical axis; CD, corneal diameter; ED, equatorial diameter of the lens; RAS, radius of curvature of the anterior surface of the sclera.
(53) According to the nine ocular parameters, the eye models were produced in the NURBS format and then converted to the PM format. The eye models in the PM format were finally refined and installed in the paediatric MRCPs, matching the centroid positions of the eyes of the P143 phantoms (ICRP, 2020a), as shown in Fig. 3.7.
Detailed eye models inserted in the paediatric mesh-type reference computational phantom for the 15-year-old female.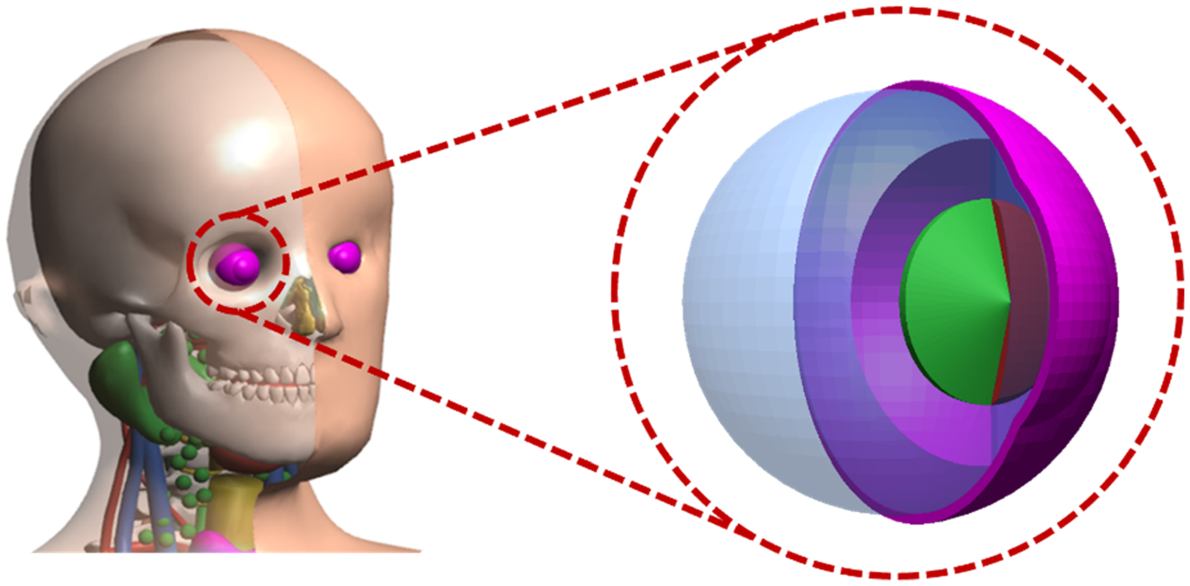
3.8. Teeth
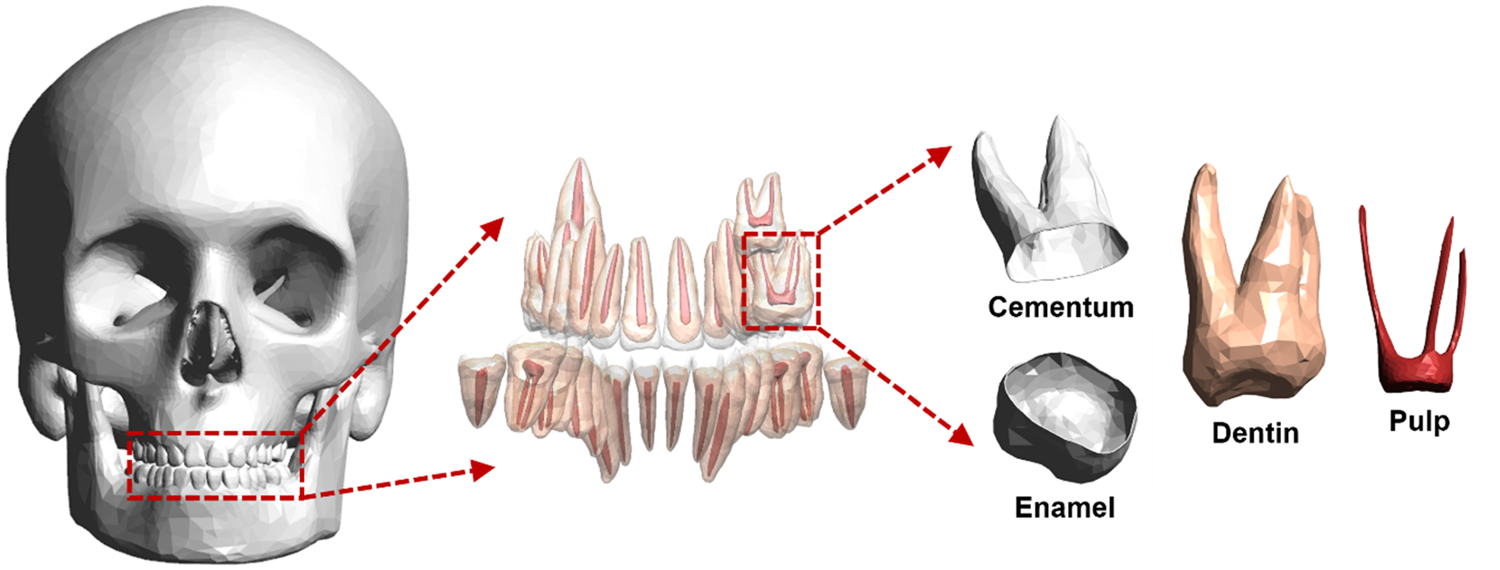
(54) In the P143 phantoms (ICRP, 2020a), the teeth are defined as a single homogeneous region with a simplified geometry (i.e. ellipsoid). A more realistic representation was considered important for applications including retrospective dosimetry based on electron paramagnetic resonance. (55) Detailed tooth models, including the inner tooth tissues (i.e. enamel, dentin, pulp, and cementum), were developed for each paediatric age and sex (Shin et al., 2021). The target masses of the tooth tissues for each age and sex were determined by analysing the scientific literature. First, the entire teeth mass was determined by adopting the reference values given in Publication 89 (ICRP, 2002). The individual tooth masses were then determined using the tooth mass fractions of the entire teeth derived from the data of Ogorelec et al. (1997). Finally, the masses of the enamel, dentin (including cementum), and pulp for each tooth were determined based on the data of Publication 23 (ICRP, 1975) and Bayle et al. (2009) for the permanent teeth and the deciduous teeth, respectively. (56) The detailed tooth models were then developed by employing existing high-quality PM tooth models as preliminary models. The preliminary models for the permanent teeth were constructed from the micro-CT images of adult male and female (http://dk.kisti.re.kr), and the model for the deciduous teeth was constructed by 3D scanning a mould of the tooth crowns of a child and modelling the roots based on image references (https://www.turbosquid.com/3d-models/primary-teeth-dentition-max/953912). The permanent tooth models, but not the deciduous tooth model, are available for both male and female, and thus were used separately. First, each tooth of the preliminary models was scaled to match the target tooth mass. In the scaled tooth model, the inner tooth tissues were modelled referring to anatomical structures represented by Schwartz (1995), matching the target tooth-tissue masses. The cementum was separated from the dentin according to the age-dependent cementum thickness (Zander and Hürzeler, 1958). In total, 332 detailed tooth models (i.e. newborn, n = 20; 1 year, n = 28; 5 years, n = 48; 10 years, n = 38; and 15 years, n = 32 for each sex) were produced individually, which were finally inserted in the paediatric MRCPs. (57) Fig. 3.8 shows, as an example, the detailed tooth models of the 15-year-old male MRCP, including the internal structures.
Detailed tooth models of the mesh-type reference computational phantom for the 15-year-old male.
3.9. Blood in large vessels
(58) In the P143 phantoms, only a small portion of the blood in the large blood vessels was modelled because of the finite voxel resolutions of the original CT image data (ICRP, 2020a; Wayson, 2012). Consequently, the masses of the blood defined explicitly in the phantoms are significantly smaller than their reference values. Note that these values are not given in Publication 89 (ICRP, 2002), but can be estimated based on the reference regional volume fractions derived by Wayson et al. (2018), and adopted in the calculation of the paediatric SAFs in Publication 155 (ICRP, 2023). (59) For the paediatric MRCPs, therefore, the blood in the large vessels was remodelled. First, the flow lines of the blood in the large vessels were generated manually, referring to the high-quality 3D blood models provided by BioDigital Human (http://www.biodigital.com). Next, the blood models were constructed in the NURBS format along the flow lines, and then converted to the PM format. Finally, the PM models were adjusted to match the reference masses. (60) Fig. 3.9 shows the blood model for the 5-year-old male MRCP, together with the corresponding P143 phantom (ICRP, 2020a). Note that the intra-organ vasculature was not modelled in the MRCPs, and the blood in the intra-organ vasculature was assumed to be distributed homogeneously with the organ parenchyma.
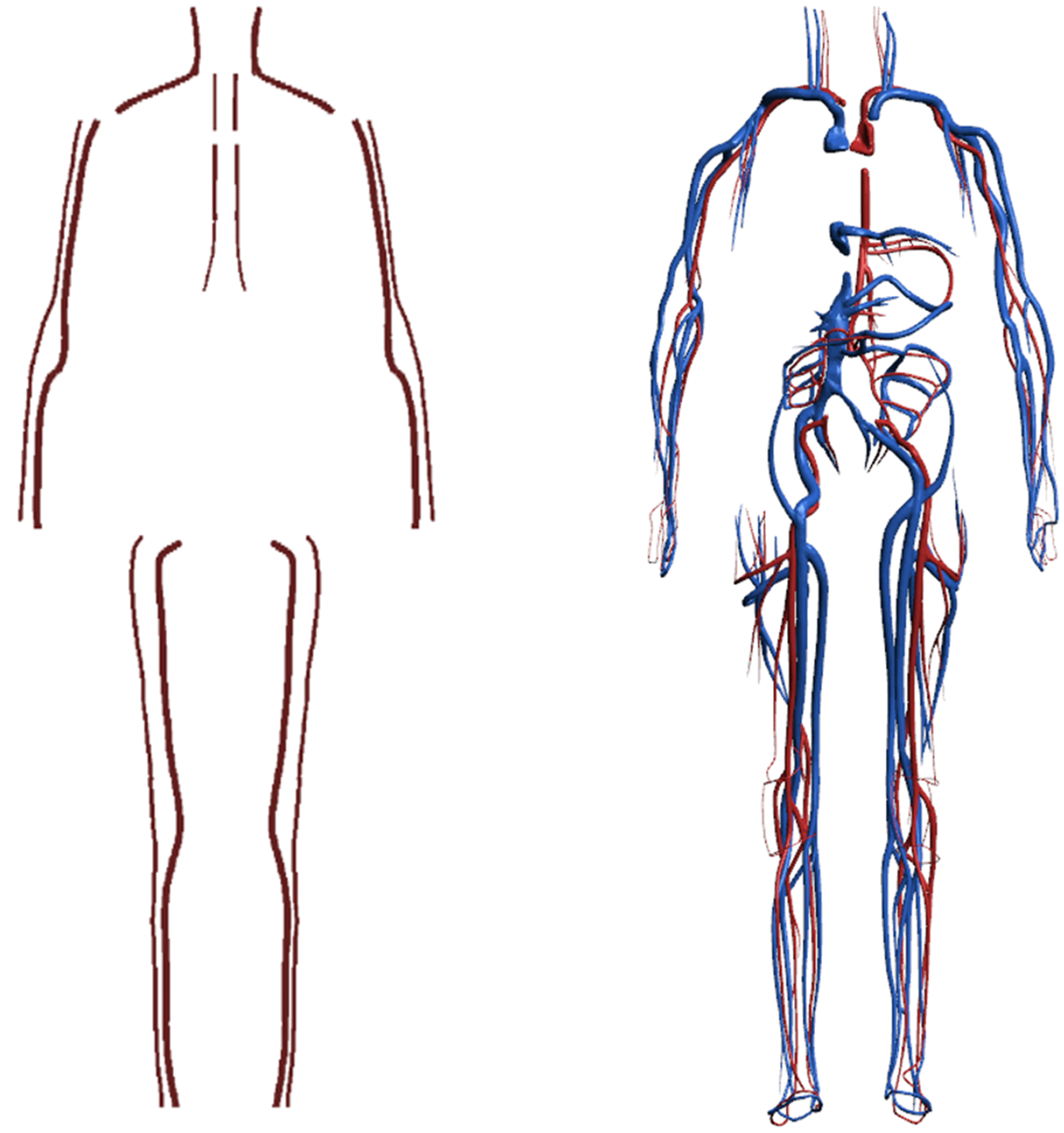
Blood in large vessels of the Publication 143 phantom (ICRP, 2020a) (left) and the paediatric mesh-type reference computational phantom (MRCP) (right) for the 5-year-old male. In the MRCP, the blood in the large arteries (red) and the large veins (blue) was modelled separately.
3.10. Muscle
(61) The muscle of the P143 phantoms (ICRP, 2020a) was developed using a voxel growing algorithm (Stepusin, 2016), by which the complex and heterogeneous anatomical structures of the various muscles of the body could not be modelled delicately. (62) In the present work, the muscle was modelled using the construction procedure described by Choi et al. (2019). First, the exterior surface of the muscle was produced by replicating and reducing the skin model, and the interior surface of the muscle was generated by producing a surface which covers all of the internal organs except for the blood in the large vessels and the lymphatic nodes. Next, a preliminary muscle model was produced by merging the exterior and interior surfaces of the muscle, and then subtracting the blood in the large vessels and the lymphatic nodes. Finally, the preliminary muscle model was adjusted to provide a more anatomically realistic representation, referring to the anatomy text of Drake et al. (2004). (63) Fig. 3.10 shows the muscle model for the 1-year-old male phantom, together with the corresponding P143 phantom (ICRP, 2020a).
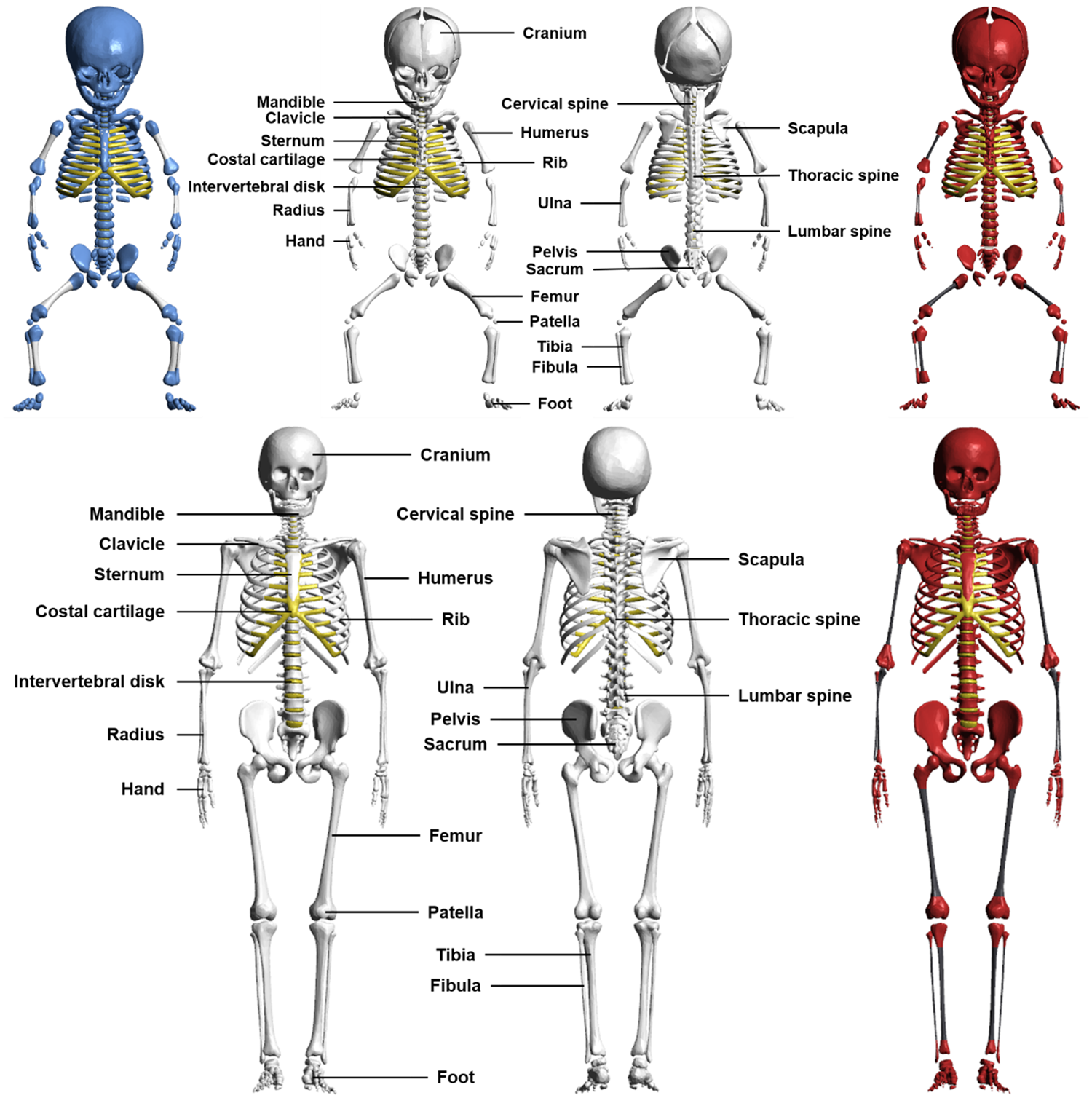
Muscle of the P143 phantom (ICRP, 2020a) (left) and the paediatric mesh-type reference computational phantom (right) for the 1-year-old male.
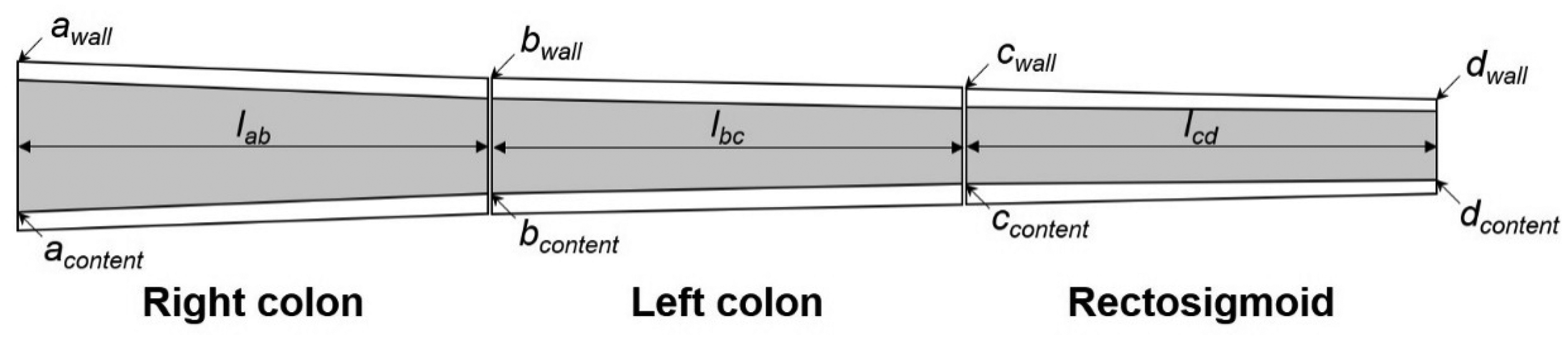
3.11. Exterior body contours
(64) The exterior body contours of the P143 phantoms (ICRP, 2020a) were produced for the head, torso, and limbs separately, matching eight reference anthropometric dimensions (i.e. standing and sitting height; arm length; biacromial breadth; and head, neck, waist, and buttock circumference), but the individual body parts (i.e. head, torso, and limbs) were not connected realistically, especially at the hip and shoulder joints. Anthropometric dimensions (unit: cm) of the paediatric mesh-type reference computational phantoms (MRCPs), along with reference values given in the Publication 143 phantom (ICRP, 2020a).
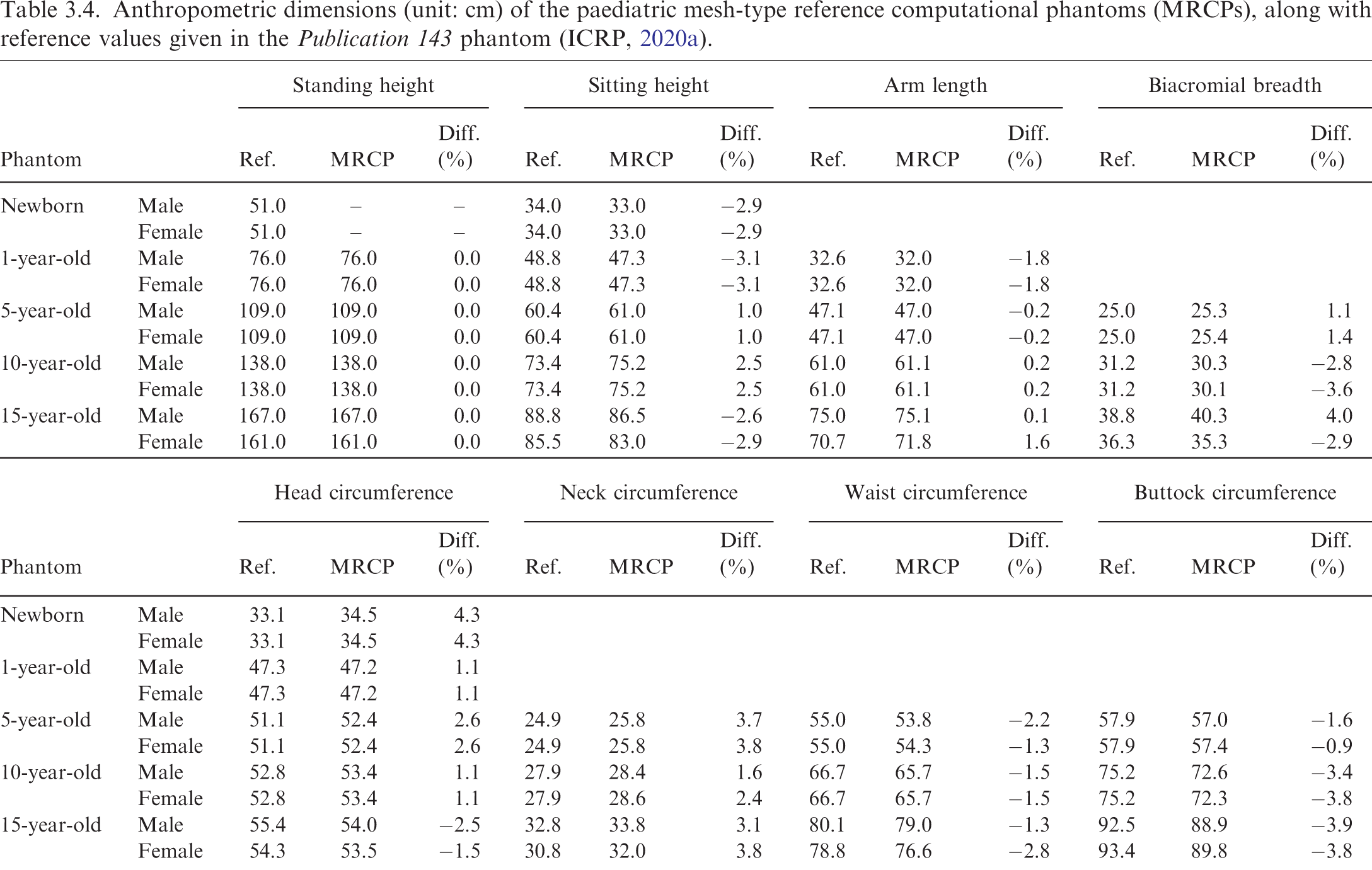
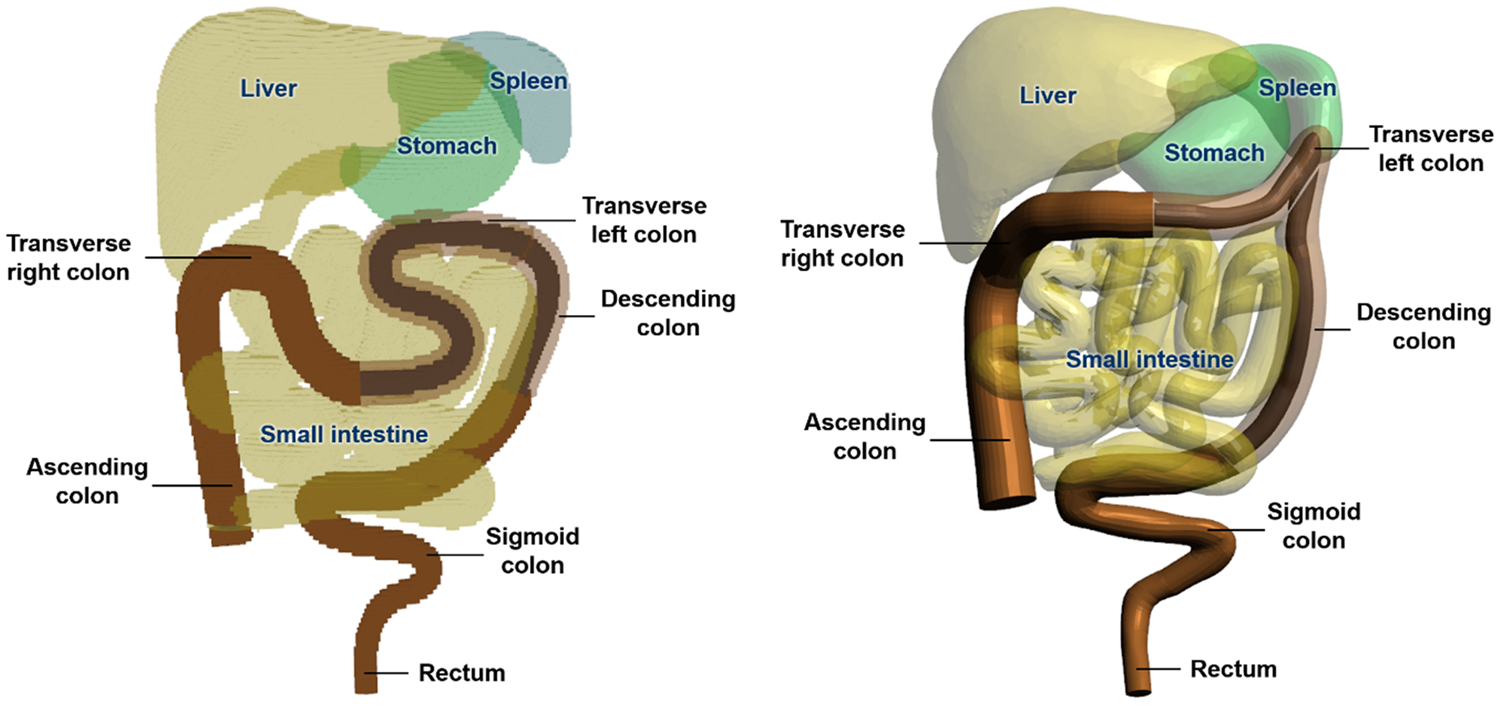
(66) Fig. 3.11 shows the exterior body contours of the 5-year-old male MRCP, together with the corresponding P143 phantom (ICRP, 2020a).
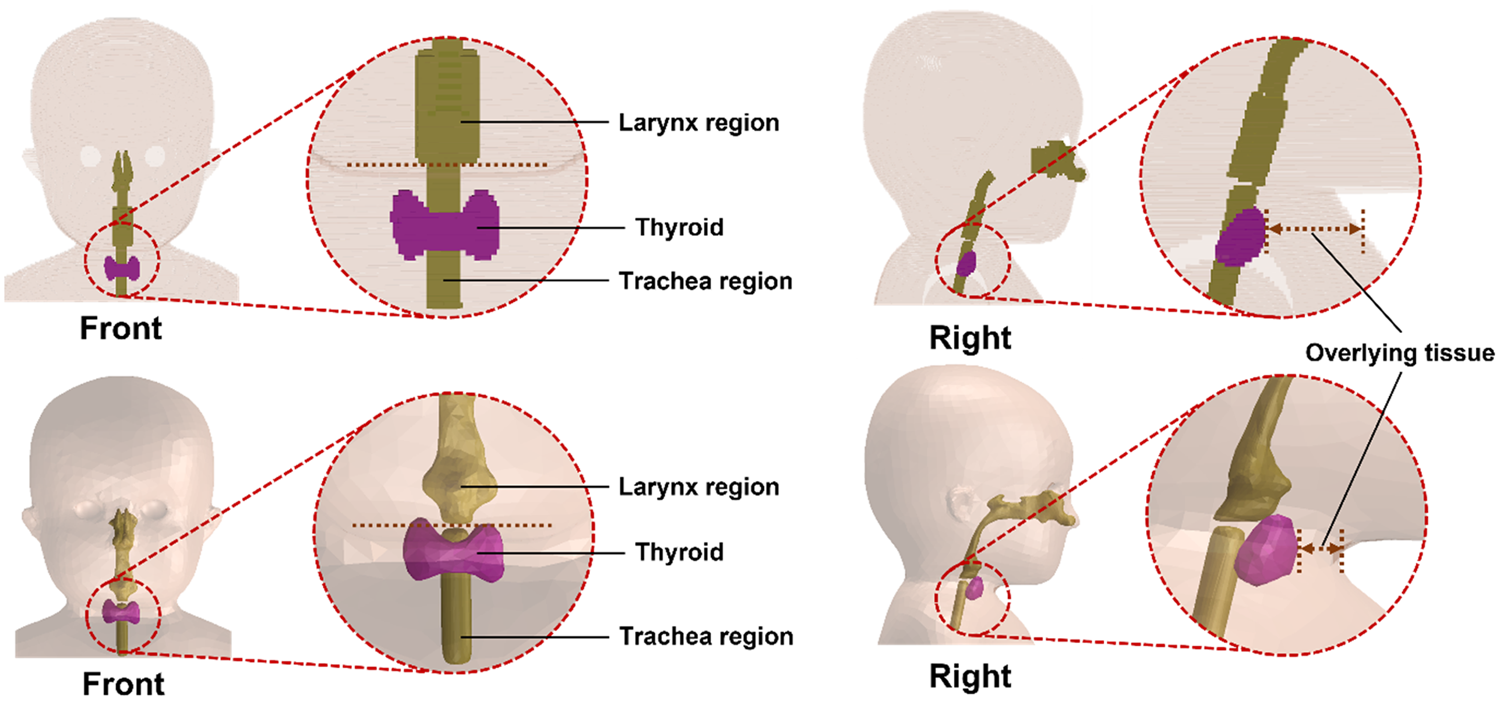
Exterior body contours of the Publication 143 phantom (left) and the paediatric mesh-type reference computational phantom (right) for the 5-year-old male.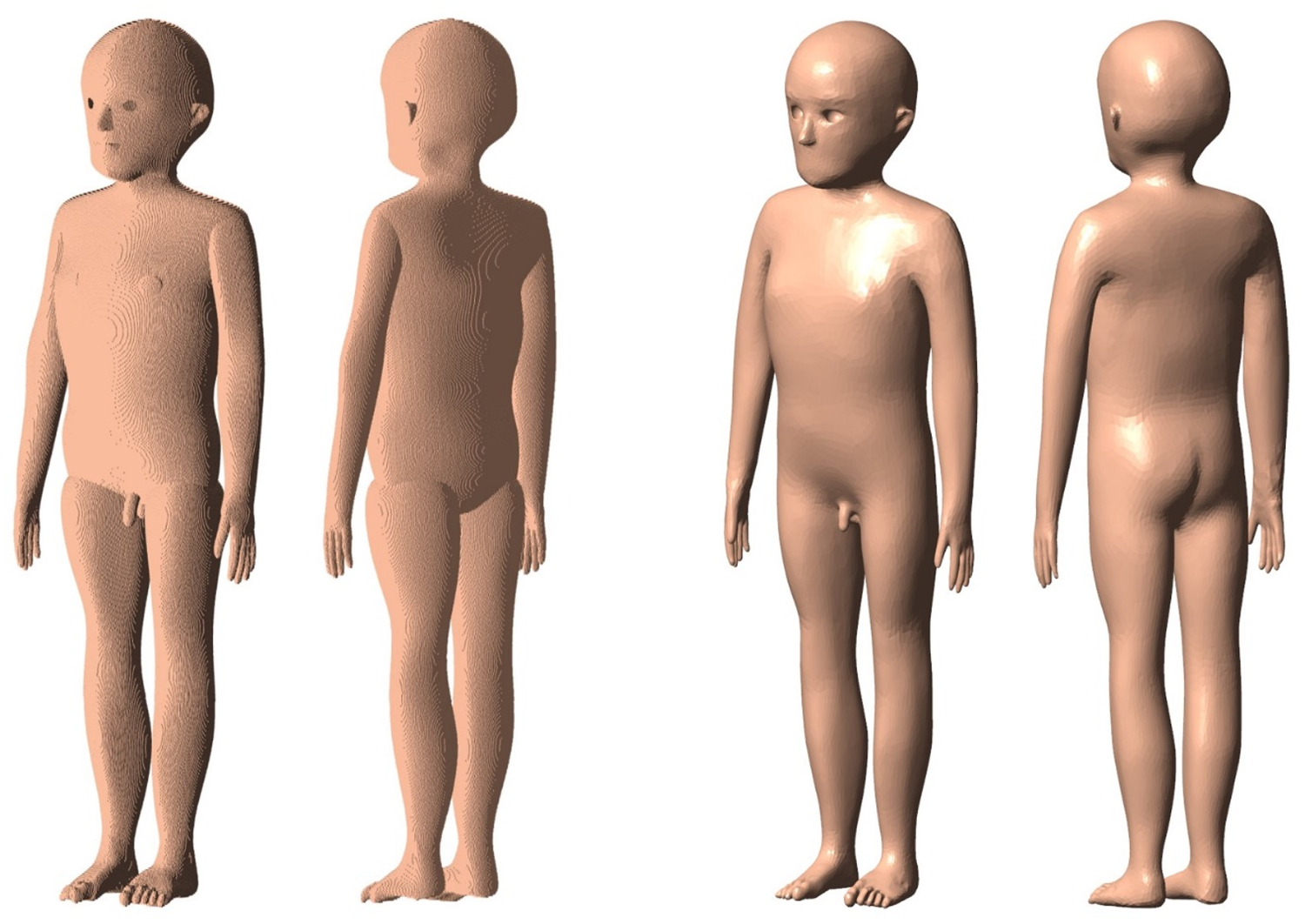
4. INCLUSION OF BLOOD IN ORGANS AND TISSUES
(67) The organs of the P143 phantoms (ICRP, 2020a) are based on the reference values for organ parenchyma (i.e. excluding the intra-organ blood content), given in Table 2.8 of Publication 89 (ICRP, 2002). In a living person, however, a considerable amount of blood is situated in the small vessels and capillaries in the organs. The most realistic phantom design would represent the organ parenchyma and intra-organ vasculature separately, but this is technically challenging due to the complexity and wide distribution of the vasculature. Consideration of the blood content is nevertheless necessary in phantom construction, especially for the purpose of internal dosimetry when the blood is considered as a source region (ICRP, 2016a, 20XX; Wayson et al., 2018). In addition, the increase in the organ masses due to the inclusion of the blood content can affect the SAF calculations, especially for the self-irradiation resulting from weakly penetrating radiations (ICRP, 2016a, 20XX). (68) For the paediatric MRCPs, as for the adult MRCPs, therefore, the organ masses, densities, and elemental compositions were recalculated to include intra-organ blood content. The organs of the MRCPs were then adjusted to the new blood-inclusive reference organ masses. (69) The reference organ parenchymal masses in Table 2.8 of Publication 89 (ICRP, 2002) were used to calculate the blood-inclusive reference organ masses, with two exceptions. The breast parenchymal masses for ≤10 years, which are not given in the table, were derived using a similar method to that used for the P143 phantoms (ICRP, 2020a); that is, the breast parenchymal masses were calculated by adopting the glandular tissue masses of the stylised phantoms (Cristy and Eckerman, 1987), and considering the glandular tissue fraction in Publication 89. The active marrow parenchymal masses were adopted, with slight adjustment, from the P143 phantoms, which differ from the reference values in Publication 89.
4.1. Calculation of mass, density, and elemental composition of organs and tissues inclusive of blood content
(70) The blood-content masses were calculated for the organs listed in Table 2.8 of Publication 89 (ICRP, 2002), based on the age-dependent regional blood volume fractions derived by Wayson et al. (2018), which are replicated in Table 4.1.
Regional blood volume fractions for children and adolescents (Wayson et al., 2018).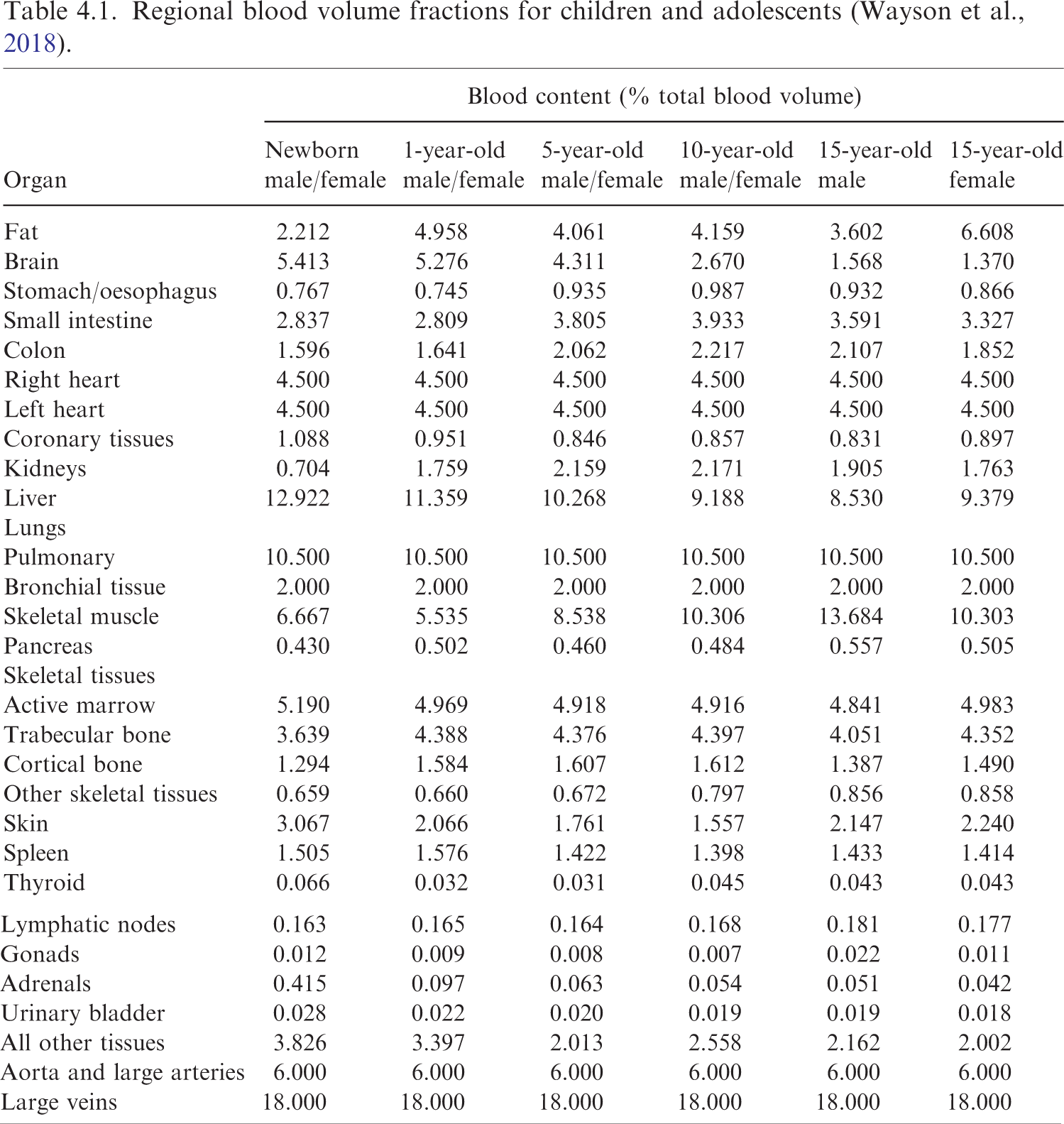
(71) For the organs for which the regional blood volume fraction is given explicitly in the table [i.e. fat, brain, stomach/oesophagus, small intestine, colon, right heart, left heart, coronary tissues, kidneys, liver, pulmonary, bronchial tissue, skeletal muscle, pancreas, active marrow, trabecular bone, cortical bone, other skeletal tissues, skin, spleen, thyroid, lymphatic nodes, gonads (testes or ovaries), adrenal glands, and urinary bladder], the blood-content masses were simply calculated as the product of their regional blood volume fractions and the total blood mass. Among them, for the organs for which the regional blood fractions are grouped into a single value in Table 4.1 (i.e. stomach/oesophagus and other skeletal tissues), the blood masses were assigned in proportion to their masses. Likewise, for the organs for which regional blood fraction is not given explicitly in the table, but listed in Table 2.8 of Publication 89 (ICRP, 2002) (i.e. tongue, salivary glands, gallbladder wall, breasts, eyes, pituitary gland, larynx, trachea, thymus, tonsils, ureters, urethra, epididymis, prostate, fallopian tubes, uterus, and remaining tissues), represented as ‘all other tissues’ in Table 4.1, the blood masses were assigned in proportion to their masses. Note that for this calculation, the mass of the remaining tissues was reduced by the mass of the lymphatic nodes, as the regional blood fraction of the lymphatic nodes is given separately, as shown in Table 4.1.
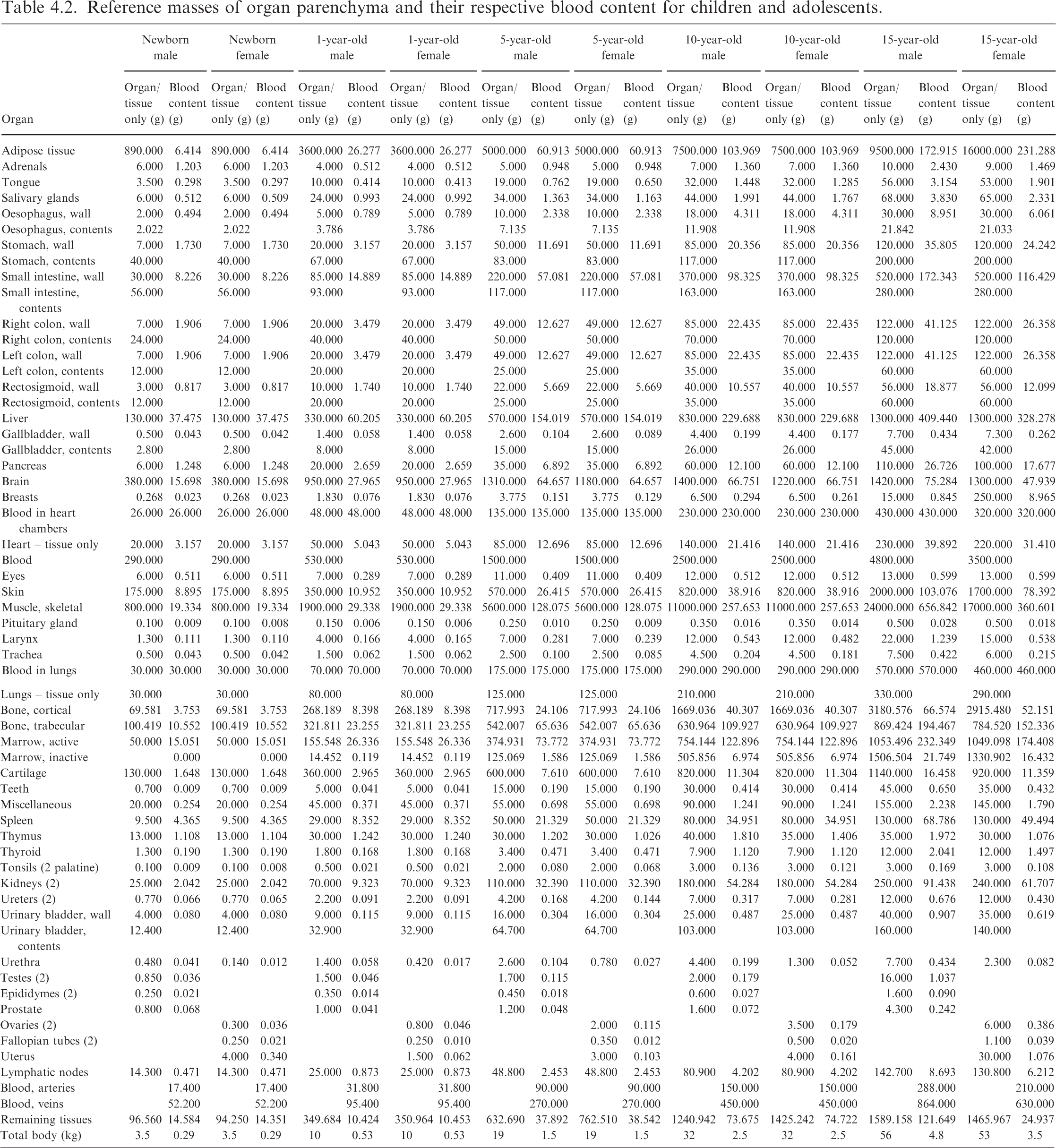
Reference masses of organ parenchyma and their respective blood content for children and adolescents.
(73) Subsequently, the densities and elemental compositions of the organs inclusive of blood content were calculated using data given in the scientific literature (White et al., 1987; ICRU, 1992; ICRP, 2002), assuming that the blood content is distributed uniformly within the organs. The density and hydrogen mass percentage of the blood-inclusive brain, for example, were calculated using the following equations:


where
4.2. Phantom adjustment for blood inclusion
(74) The volumes of the organs, produced in the PM format, were subsequently adjusted to match the blood-inclusive reference masses based on the blood-inclusive density, following the same procedure applied to the adult MRCPs (ICRP, 2020c). First, the PM models were increased isotropically, preserving the original shape and centroid of the organs. After enlargement, resulting overlaps between some of the neighbouring organs were eliminated by preferentially modifying the larger organs, rather than the smaller organs, to minimise the geometric distortion of the organs. (75) To evaluate the change of topology in the organs due to the inclusion of the blood content, the geometric similarity was investigated by calculating the DI and CD values for the organs, as discussed in Section 6.2.
4.3. Definition of residual soft tissue
(76) In the paediatric MRCPs, several organs listed in Table 4.2 (i.e. adipose tissue, urethra, epididymis, fallopian tubes, and some parts of cartilage) are not defined explicitly, while several organs included implicitly in ‘remaining tissues and regions’ [i.e. main bronchi (generation 1), spinal cord, urine, oesophagus contents, ET region, and inner air] are defined in the phantoms. Consequently, the total body masses of the phantoms are smaller than the reference masses. For this reason, as done for other ICRP reference phantoms (ICRP, 2009, 2020a,c), the total body masses of the paediatric MRCPs were matched to the reference masses by defining an additional compensating tissue, RST. RST is a homogeneous mixture of all the organs not defined explicitly in the phantoms, a commonly used approach in the field of phantom development to match the target body mass (ICRP, 2009, 2020c; Lee et al., 2010; Kim et al., 2011; Yeom et al., 2013).
5. INCLUSION OF THIN TARGET AND SOURCE REGIONS
5.1. Skin
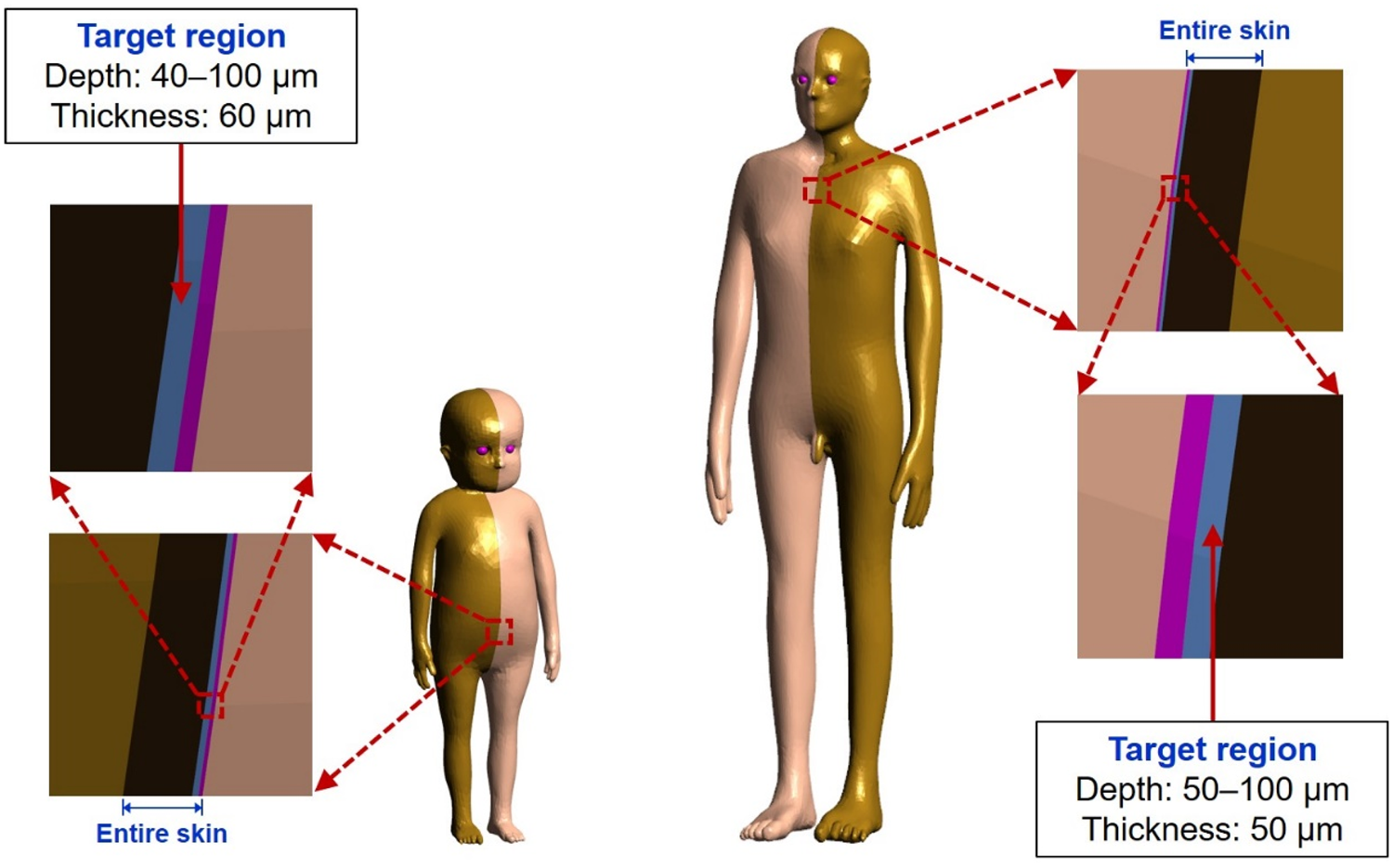
(77) In the skin, the basal cells of the epidermis and hair follicles are considered as the relevant target cells at radiogenic risk for both stochastic and deterministic effects. The target cells for adults are assumed to be at a depth of 50–100 μm below the skin surface (ICRP, 1977, 2010, 2015), and the same depth was assumed for adolescents in the present work. The skin target region for phantoms ≤10 years was assumed to be slightly wider (i.e. 40–100 μm below the skin surface), considering that the epidermal thickness is thinner but the hair follicles are more densely distributed over the skin at younger ages. Note that a substantial proportion of the stem cells that are the assumed targets are not in the interfollicular basal layer between hair follicles, but are in the hair follicles themselves which penetrate through the dermis (ICRP, 1991a, 2012). In the paediatric MRCPs, therefore, the target layer was defined at a depth of 40–100 μm for phantoms ≤10 years and 50–100 μm for the 15-year-old phantoms. (78) Fig. 5.1 shows the skin of the 1-year-old female and 15-year-old male phantoms, including the target layer.
Skin target region of the 1-year-old female (left) and 15-year-old male (right) mesh-type reference computational phantoms.
5.2. Alimentary tract
(79) The target and source regions were defined for the alimentary tract organs (i.e. oral cavity, oesophagus, stomach, small intestine, and colon) of the paediatric MRCPs (Choi et al., 2022), following the morphometric data given in Publication 100 (ICRP, 2006), which are summarised in Table 5.1. Except for the oral cavity, the target and source regions of all the alimentary tract organs were defined simply according to their depth and thickness. Note that in Publication 100, the depth and thickness of all the target and source regions for adults, with one exception, are assumed to be applicable to children and adolescents; the exception is the source region in the villi of the small intestine for children aged ≤5 years, the thickness of which is considered to be 100 μm thinner than that for adults (500 μm). This thickness variation of the villus layer with age was reflected in the paediatric MRCPs.
Information on source (upper) and target (lower) regions of alimentary tract organs from the data of Publication 100 (ICRP, 2006).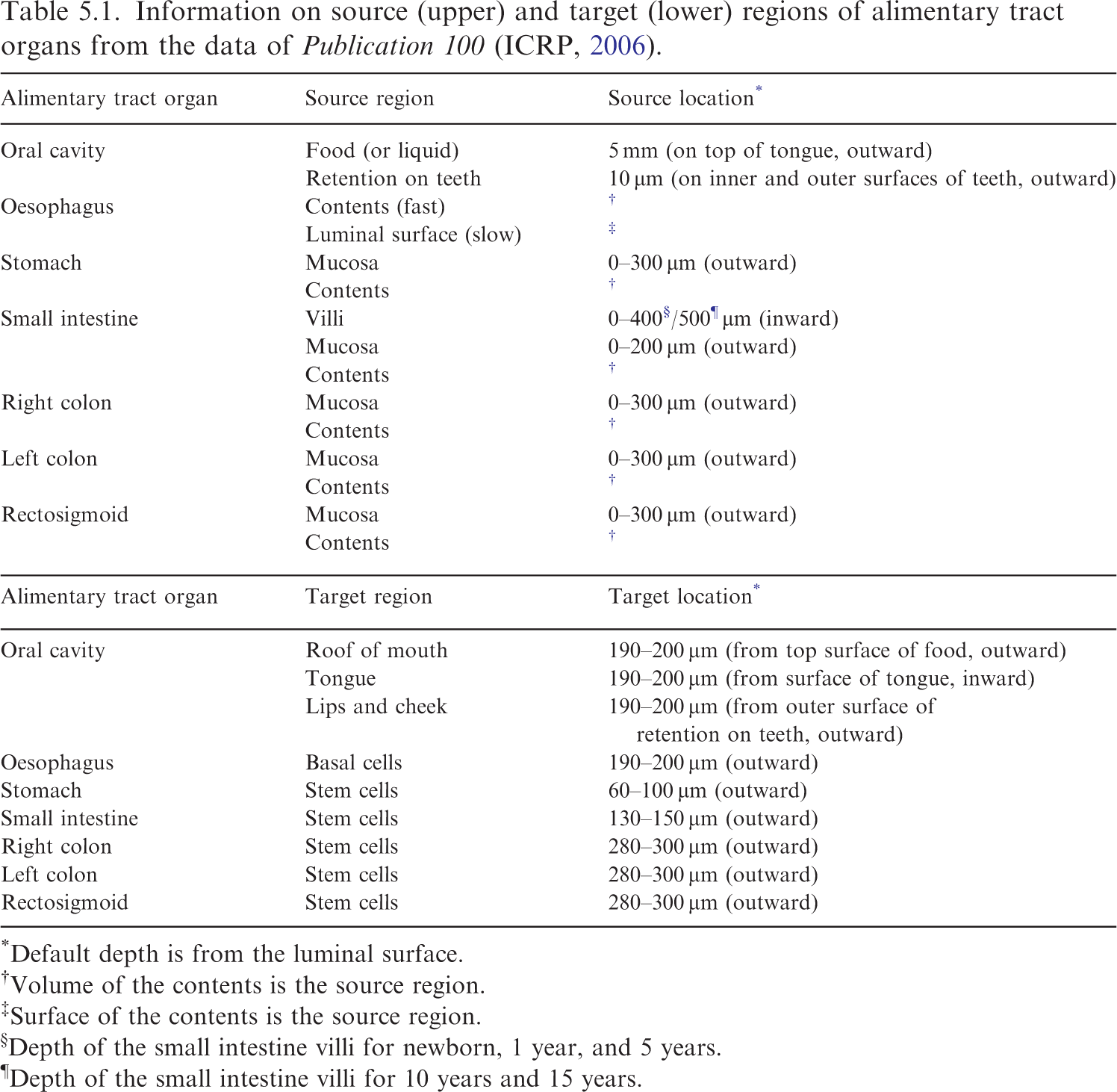
Default depth is from the luminal surface.
Volume of the contents is the source region.
Surface of the contents is the source region.
Depth of the small intestine villi for newborn, 1 year, and 5 years.
¶Depth of the small intestine villi for 10 years and 15 years.
(80) For the oral cavity, two source regions were defined in the paediatric MRCPs: food (or liquid) on the top of the tongue, and radionuclides retained on the surface of the teeth. The food volume has been estimated for adults alone (20 cm3) (ICRP, 2006); for the paediatric MRCPs, therefore, the food volume was estimated by scaling in proportion to the area of the tongue, assuming that the thickness of the food region (5 mm) is identical for all ages. The food region was then modelled in the paediatric MRCPs using the same modelling approach used for the adult MRCPs (ICRP, 2020c). The source region for radionuclides retained on the teeth was defined by adding a 10-μm-thick layer on the exposed surfaces of the teeth. The target region in the oral cavity was defined in three parts (i.e. roof of mouth, tongue and lips, and cheek) by defining a 10-μm-thick layer at a depth of 190 μm from the source regions.
(81) Fig. 5.2 shows, as examples, the target and source regions defined in the oral cavity and small intestine of the 15-year-old male phantom.
Target and source regions of the oral cavity and small intestine in the 15-year-old male mesh-type reference computational phantom.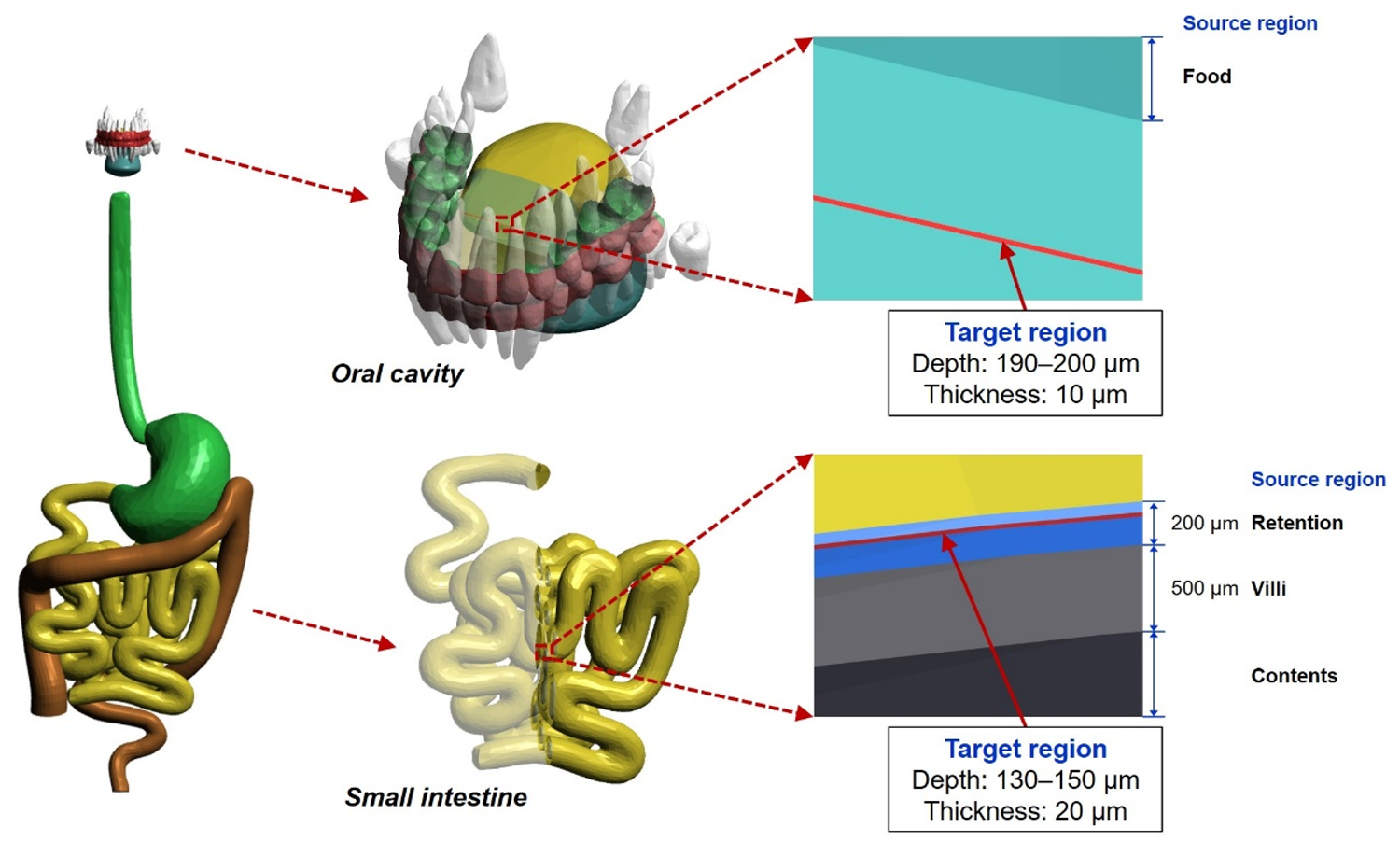
5.3. Respiratory tract
(82) For the paediatric MRCPs, as for the adult MRCPs, the target and source regions were defined in the respiratory tract tissues (Choi et al., 2023), following the morphometric data of Publication 66 (ICRP, 1994a), which are summarised in Table 5.2. The respiratory tract tissues comprise the ET region (i.e. ET1 and ET2), trachea, bronchi (BB), bronchioles (bb), and alveolar-interstitium (AI). In the present work, the AI was not defined explicitly in the phantoms, but was assumed to be distributed homogeneously in the lungs.
Information on source (upper) and target (lower) regions of respiratory tract organs from the data of Publication 66 (ICRP, 1994a).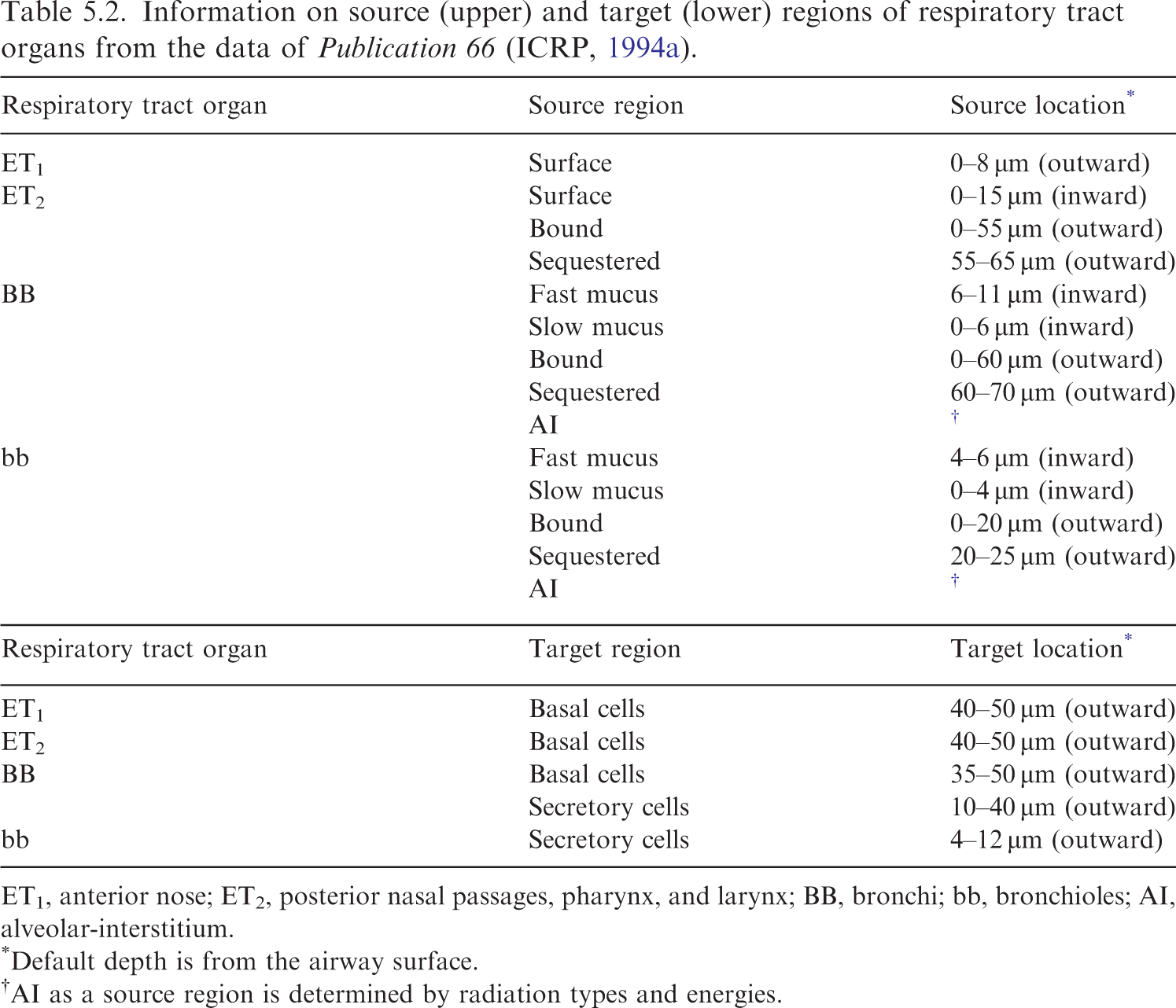
ET1, anterior nose; ET2, posterior nasal passages, pharynx, and larynx; BB, bronchi; bb, bronchioles; AI, alveolar-interstitium.
Default depth is from the airway surface.
AI as a source region is determined by radiation types and energies.
(83) For the ET1, ET2, trachea, and BB (generation 1) regions, the target and source regions were defined simply according to their depth and thickness. This approach, however, could not be used for the other generations of the BB (i.e. generations 2–8) and all the subsequent generations of the bb (i.e. generations 9–15), which are not represented in the P143 phantoms (ICRP, 2020a). These airways were modelled in constructive solid geometry (CSG) format based on airway dimensions (i.e. lengths and diameters) derived using the scaling method of Publication 66 (ICRP, 1994a), using the computer program applied for the adult MRCPs (Kim et al., 2017). The total lengths of the airway branches for each generation were matched to their reference values within 10% difference. Note that as an exception, the newborn airway dimensions were derived by scaling the adult male values in proportion to the cube root of lung volume; this is because the scaling method of Publication 66, for which limited data on newborn subjects were used, proved insufficient for the newborn.
(84) The airways generated in the CSG format could have been converted to PM format for easy incorporation into the paediatric MRCPs, but the resulting airways would require a very large number of facets and a large computer memory allocation (i.e. >50 GB) (Kim et al., 2017). In the present work, therefore, the airways in the CSG format were not converted to the PM format, but incorporated directly into the paediatric MRCPs using the overlying approach used for the adult MRCPs (Kim et al., 2017; ICRP, 2020c). Note that the overlying approach makes it possible to perform dose calculation for the airways with a minimal addition of memory usage and computation time.
(85) Fig. 5.3 shows the target and source regions of ET2 of the 5-year-old female phantom and the airway model produced in the lungs of the 5-year-old male phantom with the target and source regions of the bb.
Target and source regions of the ET2 region (posterior nasal passages, pharynx, and larynx) in the 5-year-old female mesh-type reference computational phantom (MRCP) (upper) and lung airway of the 5-year-old male MRCP including the target and source regions (lower). AI, alveolar-interstitium.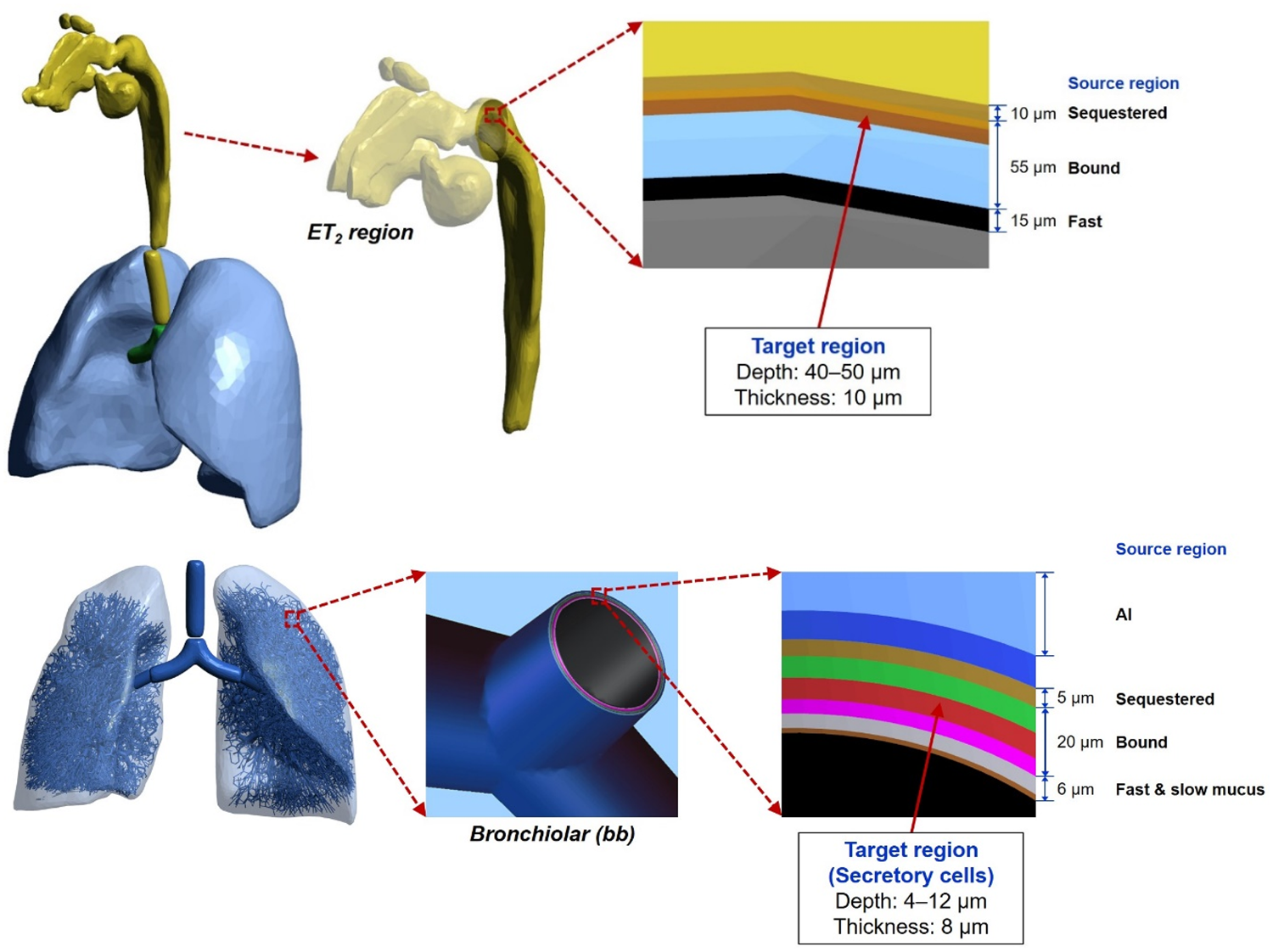
5.4. Urinary bladder
(86) The basal cells in the epithelium of the urinary bladder are considered to be the relevant target cells for carcinogenesis (Colin et al., 2009). Eckerman and Veinot (2018) derived the depth and thickness of the target layer of the urinary bladder for adults, which were used to define the target layer of the urinary bladder in the adult MRCPs (ICRP, 2020c). In their subsequent study, the values for children and adolescents were also derived, as shown in Table 5.3. In the paediatric MRCPs, these values were adopted to define the target layer of the urinary bladder.
Depth and thickness of target layer of urinary bladder for children and adolescents.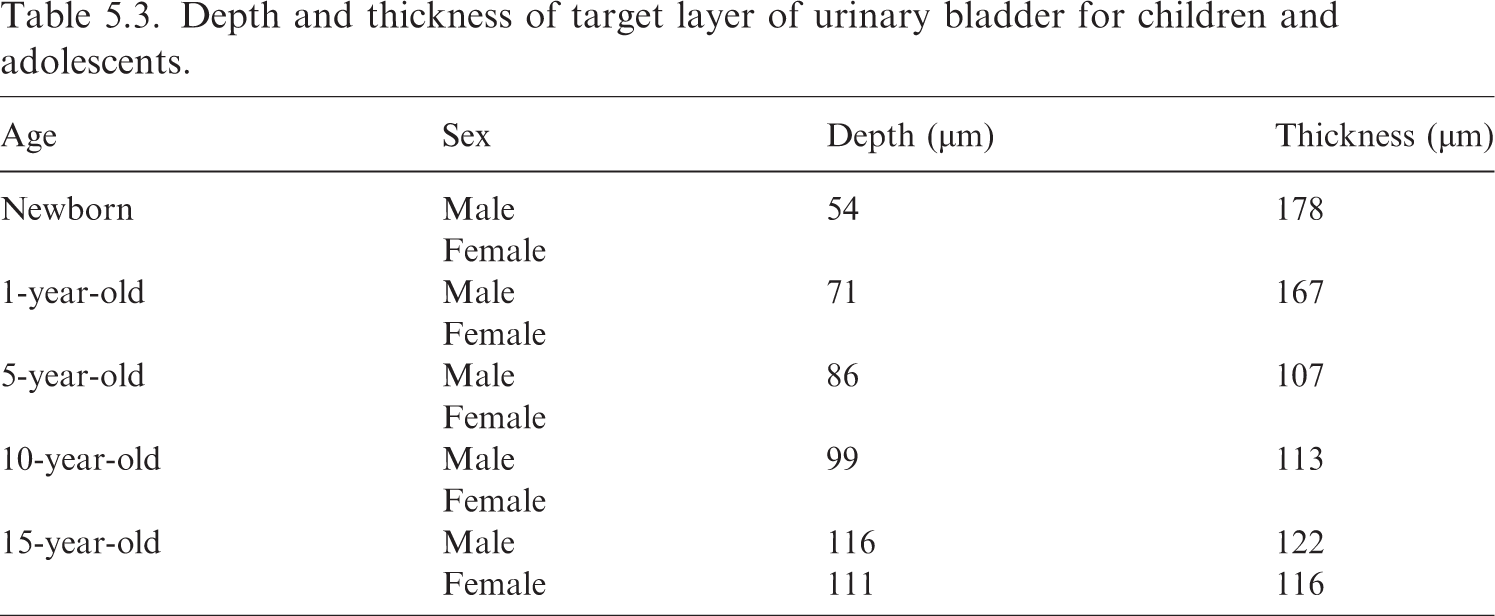
(87) Fig. 5.4 shows the urinary bladder of the 10-year-old male phantom including the source and target regions.
Source and target regions of the urinary bladder in the 10-year-old male mesh-type reference computational phantom.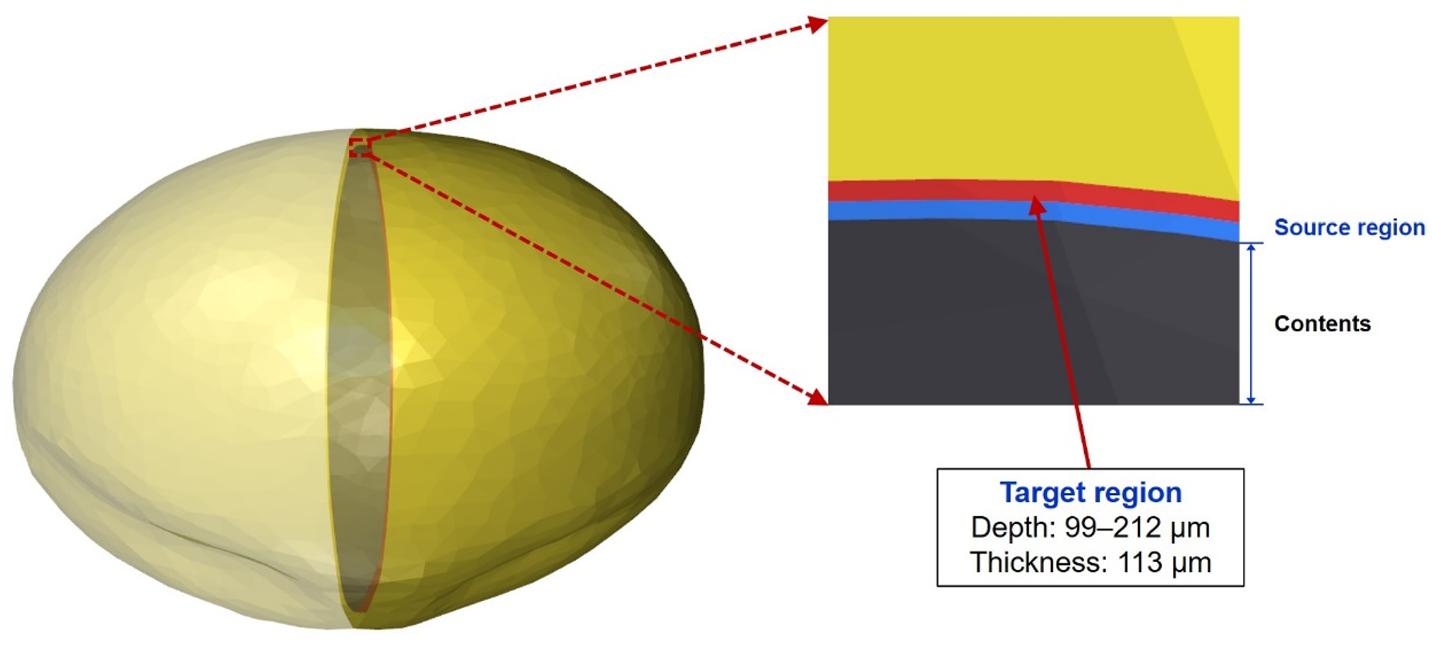
6. DESCRIPTION OF THE PAEDIATRIC MESH-TYPE REFERENCE COMPUTATIONAL PHANTOMS
6.1. General phantom characteristics
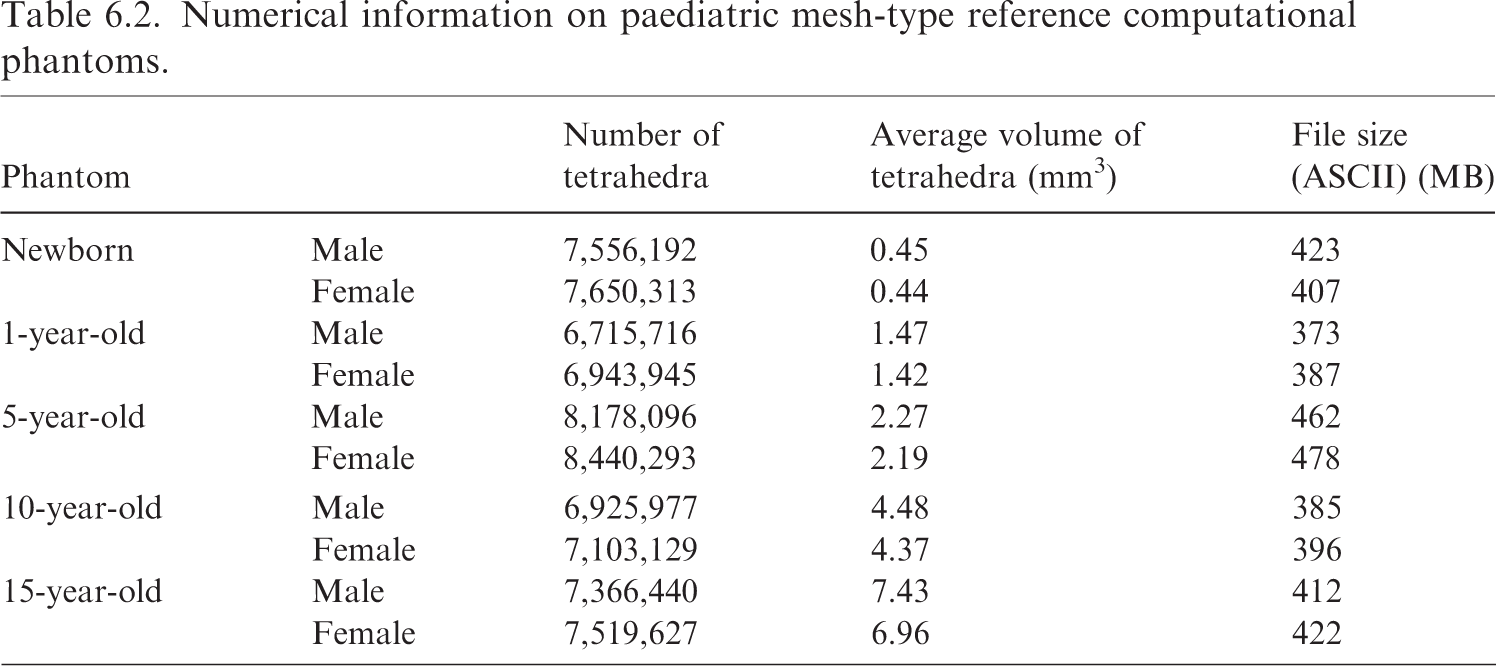
(88) Fig. 6.1 shows the paediatric MRCPs, and Table 6.1 gives the standing height and total body mass of these phantoms, which are consistent with the reference values in Publication 89 (ICRP, 2002).
Paediatric male (M) and female (F) mesh-type reference computational phantoms. Standing height and total body mass of the paediatric mesh-type reference computational phantoms. (89) The paediatric MRCPs include all the radiosensitive organs required for dose assessment from ionising radiation exposures for radiological protection purposes (ICRP, 2007). These phantoms also include the micrometre-scale target and source regions in the respiratory and alimentary tracts, skin, lens of the eye, and urinary bladder, assimilating the supplementary organ-specific stylised models. The new phantoms at ≤10 years, unlike the P143 phantoms (ICRP, 2020a), have different masses and/or shapes for several organs, besides the sex-specific organs, due to the adoption of individual sex-specific organ masses (i.e. brain for 5- and 10-year-old phantoms, and thymus for 10-year-old phantom) and high-quality organ models (i.e. spine, hand/foot bones, ET region, and teeth). Note that the complex microstructures of skeletal target tissues [i.e. red bone marrow (RBM) and endosteum] in the trabecular spongiosa and medullary cavity are not modelled explicitly; thus, skeletal dosimetry should be performed by employing the approximation techniques, namely simplified approach and fluence-to-dose response functions, which are described in Section 3.4 and Annex D of Publication 116 (ICRP, 2010), respectively. Numerical information on paediatric mesh-type reference computational phantoms.

(91) The TM-format phantoms, when compared with the PM-format phantoms, show much faster computation speed in Monte Carlo dose calculations (i.e. by a factor of tens to hundreds for photons, electrons, neutrons, and protons in the energy range of 10
(92) The masses of the organs of the paediatric MRCPs are in accordance with the reference values inclusive of blood content (see Table 4.2) within 0.1% deviation. Tables A.1 and A.2 list the organ ID numbers, medium, and mass for each organ of the paediatric MRCPs. Tables B.1–B.10 list the elemental composition and density for each organ. Table C.1 lists the anatomical source regions, their acronyms, and corresponding ID numbers. Table D.1 lists the anatomical target regions, their acronyms, and corresponding organ ID numbers.
(93) For dose calculations for the organs in which micron-scale target regions are defined explicitly (e.g. alimentary and respiratory tract organs), due to their small target volumes, longer computation times are generally required to achieve an acceptable statistical precision when compared with calculations for other organs. To save computation time, entire regions instead of the thin target regions can be used in dose calculations for cases where the entire regions of the organs provide dose values similar to those calculated using the target regions (Yeom et al., 2019b, 2020; ICRP, 2020c). For example, for external exposures to penetrating radiations (e.g. photons and neutrons), where low dose gradients are generally observed in the organs, the absorbed doses to the thin target regions tend to be close to those to the entire regions, and thus the entire regions can be used instead of the thin target regions; exceptions are the skin and lens of the eye, for which the thin target regions should be considered, because charged-particle equilibrium (CPE) is not always established in these superficial tissues. For external exposures to weakly penetrating radiations (e.g. alpha particles and protons), the radiosensitive regions should be used for dose calculation because significant spatial dose gradients could be observed, depending on the particle energies and organ topologies. The dose discrepancies between the thin target regions and the entire regions for external exposures are discussed by Yeom et al. (2019b, 2020). For internal exposures to penetrating radiations, dose calculations for entire regions can replace those for thin target regions to estimate cross-fire irradiation doses (e.g. lungs ← liver).
(94) However, in cases where subregions of the same organs are considered as source regions (e.g. source region: BB bound region; target region: BB secretory region), the thin target regions should be used in dose calculation, again due to the lack of CPE. For internal exposures to weakly penetrating radiations which could establish steep dose gradients in the organs, it is recommended that doses should be calculated using the thin target regions.
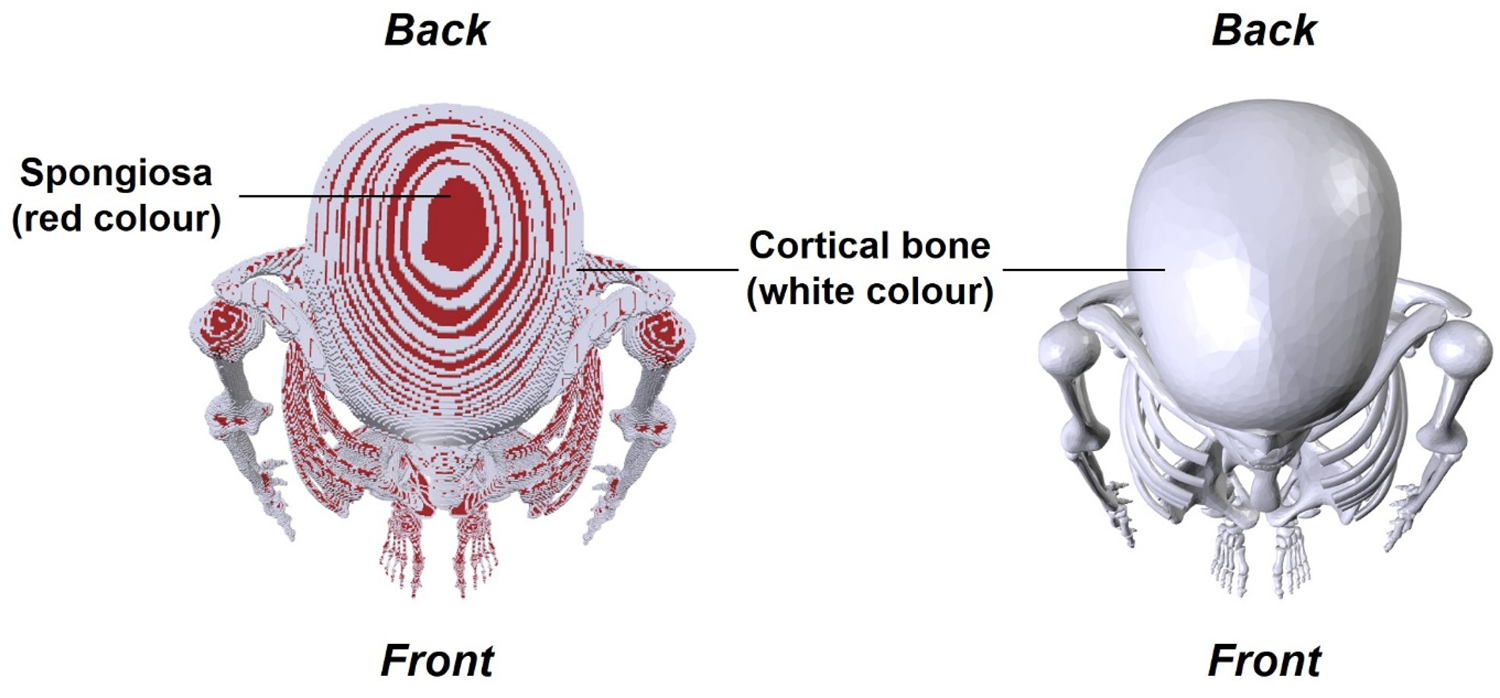
(95) The paediatric MRCPs overcome the limitations of the P143 phantoms (ICRP, 2020a) resulting from the inherent nature of voxel geometry and finite voxel resolutions. Fig. 6.2 shows, as an example, the 15-year-old female MRCP, along with the 15-year-old female P143 phantom, viewed in superior–inferior direction. It can be seen that the organs of the P143 voxel phantom are represented with stair-stepped surfaces, whereas those of the MCRP are represented with smooth surfaces. Moreover, in the voxel phantom, several radiosensitive organs (e.g. breasts and muscle) are not covered fully by skin voxels and are, thus, exposed directly to the air. This limitation is addressed in the MRCPs, preventing significant overestimations in dose calculations for these organs for external exposures to weakly penetrating radiations. Similarly, in the voxel phantoms, the spongiosa is not covered fully by the cortical bone, which is also addressed in the MRCPs (see Fig. 6.3).
Publication 143 (P143) phantom (ICRP, 2020a) (left) and paediatric mesh-type reference computational phantom (right) for the 15-year-old female viewed in superior–inferior direction: spongiosa (red), muscle (brown), and breasts (yellow) in P143 phantom. Skeletal system of Publication 143 phantom (ICRP, 2020a) (left) and paediatric mesh-type reference computational phantom (right) for the 15-year-old female viewed in superior–inferior direction: spongiosa (red) and cortical bone (white).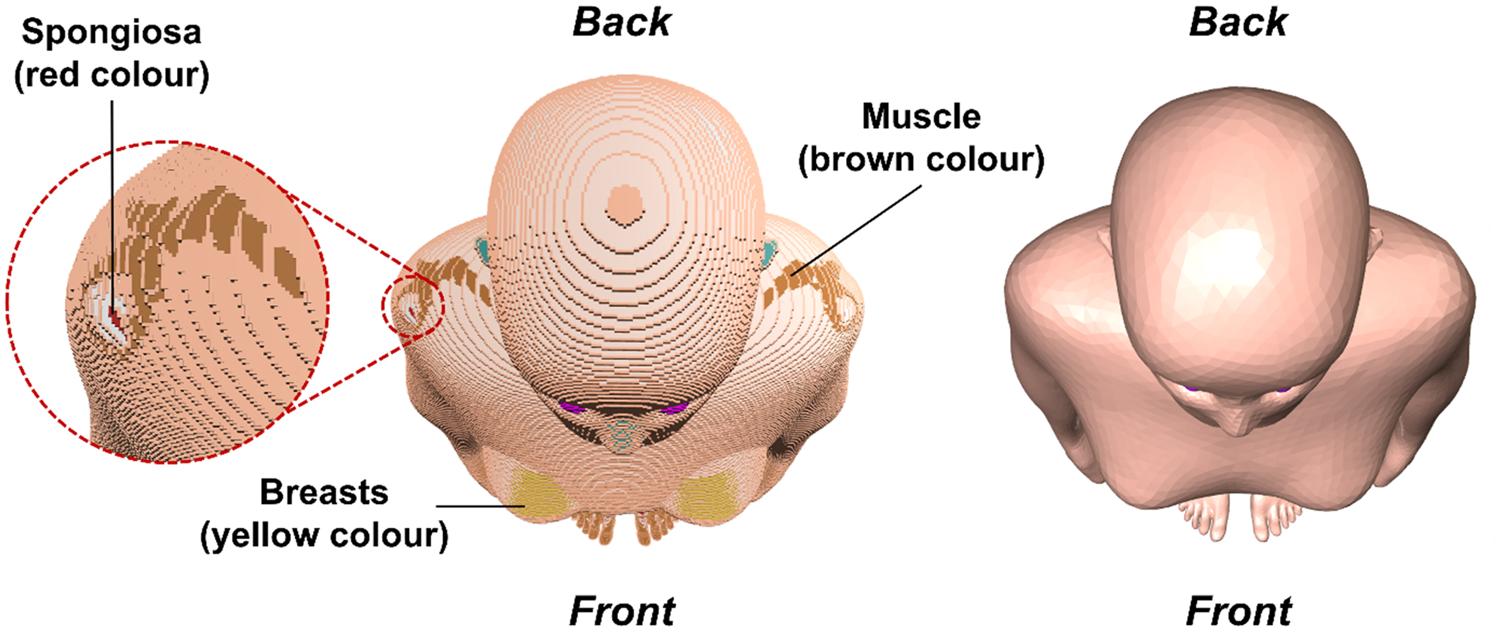
6.2. Geometric comparison with the P143 phantoms
(96) In order to investigate the geometric similarity between the paediatric MRCPs developed in the present work and the P143 phantoms (ICRP, 2020a), DI and CD of the organs were calculated as shown in Table 6.3.
Dice index (DI) and centroid distance (CD) comparing the paediatric mesh-type reference computational phantoms and the Publication 143 phantoms (ICRP, 2020a).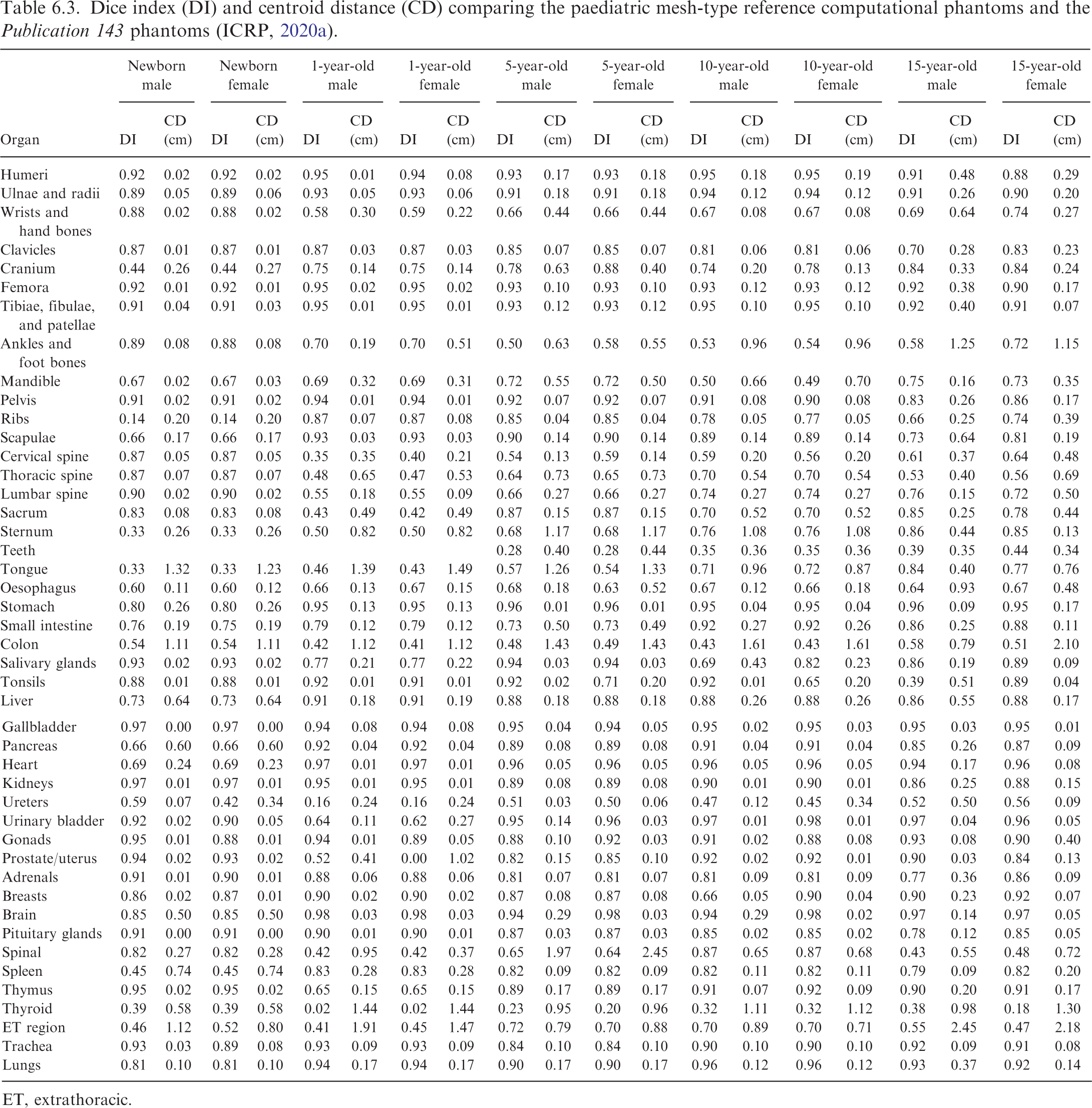
ET, extrathoracic.
(97) The DI and CD values are generally >0.7 and <10 mm, respectively, with some exceptions. For the spine models, except for the newborn phantom, relatively large dissimilarity is found, which is due mainly to the fact that the spine models of the paediatric MRCPs, except for the newborn, were not converted directly from the P143 phantoms (ICRP, 2020a), but constructed based on the PM models of Park et al. (2005). For the colon models, the relatively large dissimilarity is attributed to the fact that the colon models were reproduced with different colon shapes. Large dissimilarity is also found for the organs which were modelled in the present work (i.e. thyroid and ET region models). The large dissimilarity observed for the ureters is mainly due to the shape of the organ; the ureters are very thin and a slight shift in the location results in a very small DI value. Large dissimilarity is also found for some small organs due to the shift of their positions caused by the changes in the adjacent large organs. For example, the low DI values for the spleen of the newborn phantoms and the uterus of the 1-year-old female phantom can be attributed to their shifting due to the change of the colon models. The other large dissimilarities in specific phantoms generally reflect the procedures used in the phantom construction process.
(98) The organ depth distributions (ODDs) and CLDs of the paediatric MRCPs were compared with those of the P143 phantoms (ICRP, 2020a), as shown in Annexes E and F. ODD represents the distance from the body surface to the organ, and CLD represents the distance from the source organ to the target organ, which mainly influence the doses from external and internal exposures, respectively. Although the organs of the paediatric MRCPs were adjusted for blood inclusion, the comparison results show that the ODDs and CLDs of the paediatric MRCPs are generally in good agreement with those of the P143 phantoms for most organs.
(99) The results of the geometric similarity investigation show that, overall, the paediatric MRCPs preserve the shape and location of the organs in the P143 phantoms (ICRP, 2020a), and thus they are expected to provide similar dose values for penetrating radiations in both external and internal exposures. For weakly penetrating radiations, however, they will provide significantly different (more accurate and reliable) dose values, especially for the organs with large dissimilarities (e.g. colon and thyroid).
6.3. Computational performance in Monte Carlo codes
(100) The TM geometry of the paediatric MRCPs can be implemented directly in general purpose Monte Carlo codes, such as Geant4 Version 8.0 and later versions, PHITS Version 2.82 and later versions, and MCNP6 Version 1.0 and later versions (ICRP, 2020c). The features of these codes that enable implementation of the TM geometry have been improved significantly in the last few years (Allison et al., 2016; Furuta et al., 2017; Martz et al., 2017). In this section, as an example, the computational performance of the 5-year-old male MRCP and P143 phantom (ICRP, 2020a) were compared in terms of run time and memory usage. (101) Computational performances were measured for Geant4 Version 10.06.p01, PHITS Version 3.10, and MCNP6 Version 2.0 on a single core of the Intel Xeon CPU E5-2698 v4 (2.20 GHz and 512 GB memory). Run time was measured for photons, electrons, and neutrons in the left-lateral (LLAT) irradiation geometry by simulating 105 primary particles with energies of 10−2–104 MeV for photons and electrons and 10−9–104 MeV for neutrons. The run time results were obtained by averaging values from multiple measurements to achieve relative errors <5%. (102) For Geant4, the physics library of G4EmLivermorePhysics was used for the transportation of photons and electrons, and the physics models and cross-sections of NeutronHPThermalScattering, NeutronHPElastic, ParticleHPInelastic, Neutron-HPCapture, and NeutronHPFission were used for the transportation of neutrons. A range cut-off of 1 μm was applied for the production of secondary particles. For PHITS, the EGS5 physics library was used for the transportation of photons and electrons, and the JENDL-4.0 physics library and event generator mode Version 2 were used for the transportation of neutrons. For MCNP6, the default physics libraries based on Lawrence Livermore National Laboratory evaluated data were used for the transportation of photons and electrons, and the ENDF70 physics library was used for the transportation of neutrons. For the energy cut-off values, considering the range cut-off value of 1 μm used for Geant4 calculations, the equivalent energy cut-off values were applied in the PHITS and MCNP6 codes. Variance reduction techniques were not used. (103) Fig. 6.4 compares the run times measured for the 5-year-old male MRCP and P143 phantom (ICRP, 2020a) implemented in Geant4, PHITS, and MCNP6. The results of Geant4 show that, for photons, the run time of the MRCP is longer than that of the P143 phantom when the energy is <1 MeV, by up to 7.0 times, and shorter when the energy is higher, by up to 4.3 times. For electrons, similar differences were found: the run time of the MRCP is longer when the energy is <10 MeV, by up to 5.2 times, and shorter when the energy is higher, by up to 3.2 times. For neutrons, it can be seen that the run time of the MRCP phantom is similar in the energy range of 10−5–1 MeV, and longer than that of the P143 phantom in the other energy ranges, by up to a factor of approximately 4 and approximately 2 at the lower and higher energies, respectively.
Run time measured for the 5-year-old male mesh-type reference computational phantom (MRCP) and Publication 143 (P143) phantom (ICRP, 2020a), implemented in Geant4, PHITS, and MCNP6, for transporting 105 source particles in the left-lateral irradiation geometry. (104) For PHITS, the run time of the MRCP is generally shorter than that of the P143 phantom (ICRP, 2020a) for all three radiation types. Unlike Geant4, the run time of the MRCP is shorter at low energies; the maximum difference is approximately four times for both photons and electrons at 0.01 MeV, and approximately two times for neutrons at 10−9 MeV. For photons and electrons with energies >1 MeV, on the other hand, the run time of the MRCP is similar to that of the P143 phantom. This high computation speed of the MRCP at low energies is due to the fact that the computation speed of the TM geometry in PHITS is accelerated by using the octree decomposition technique (Furuta et al., 2017). (105) For MCNP6, the run time of the MRCP is significantly longer than that of the P143 phantom (ICRP, 2020a) for all three radiation types. The differences are between three- and 11-fold, except for high-energy (≥ 100 MeV) photons and electrons for which even larger differences (20–27-fold) are found. Such slow computation speeds for the MRCP are due mainly to the fact that MCNP6 uses features dedicated to unstructured mesh geometry; that is, MCNP6 is overly sophisticated for implementing simple TM geometry, although it can also implement pentahedral and hexahedral mesh geometry (Martz, 2017). Note that Geant4 and PHITS have features dedicated to TM geometry. (106) The run times of the MRCP were compared between Geant4, PHITS, and MCNP6. Geant4 shows the shortest run time of the three codes for photons and electrons. For photons, PHITS and MCNP6 take 1.8–7.8 and 6.3–24.1 times longer than Geant4, respectively. For electrons, PHITS and MCNP6 take 1.4–7.0 and 6.3–26.0 times longer than Geant4. On the other hand, for neutrons, except for the highest energy point, PHITS shows the shortest run time of the three codes; Geant4 and MCNP6 take 1.7–3.3 and 1.6–3.4 times longer than PHITS. At the highest energy point (i.e. 104 MeV), the run time of MCNP6 is shorter than that of Geant4 and PHITS by a factor of 3.0 and 2.4, respectively. Memory usage required to implement the 5-year-old male mesh-type reference computational phantom and Publication 143 (P143) phantom (ICRP, 2020a) in Geant4, PHITS, and MCNP6.
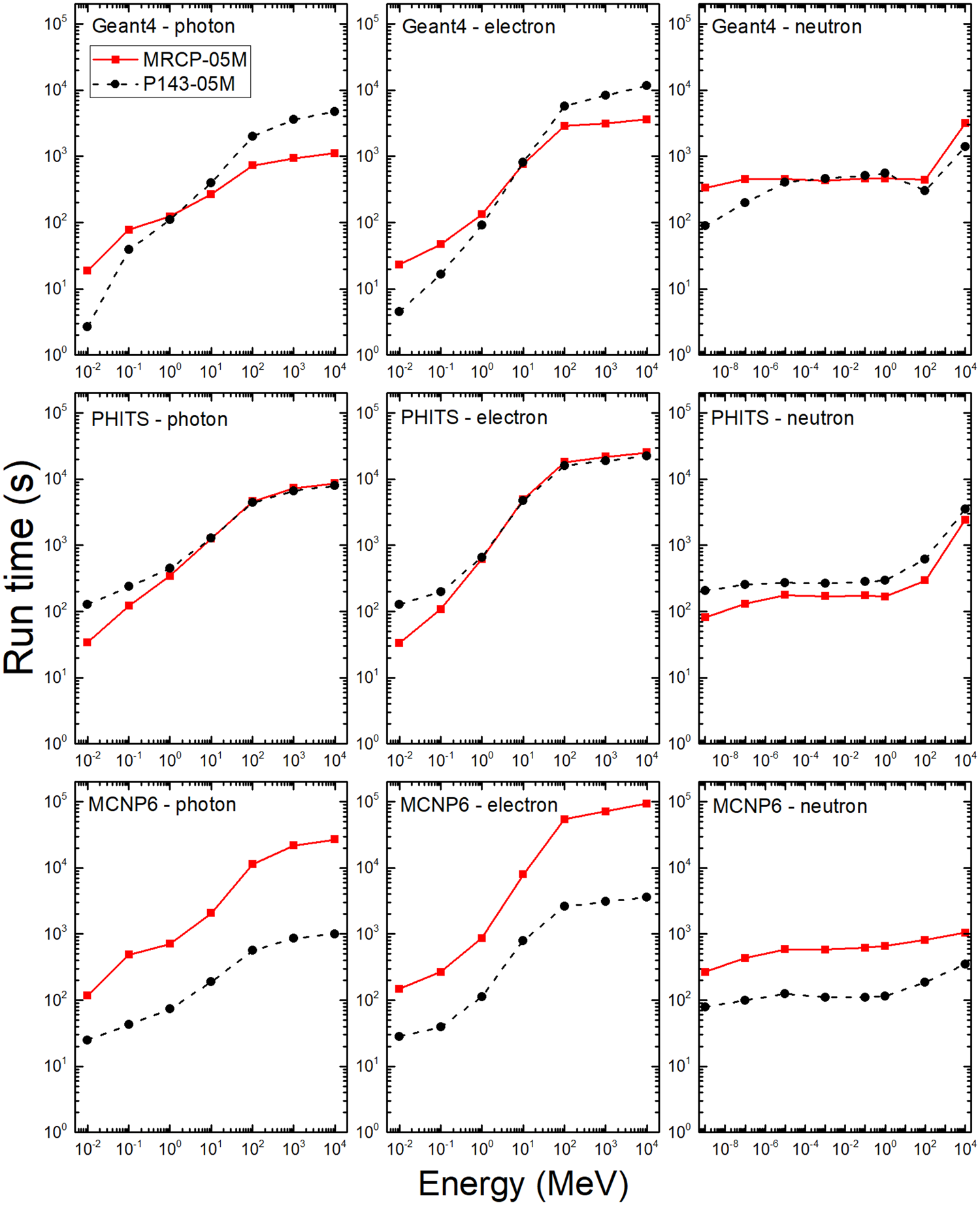

7. DOSIMETRIC IMPACT OF THE PAEDIATRIC MESH-TYPE REFERENCE COMPUTATIONAL PHANTOMS
(108) This chapter discusses the dosimetric impact of the paediatric MRCPs for external and internal exposure geometries compared with the P143 phantoms (ICRP, 2020a) and the Publication 66 and 100 mathematical models for the respiratory and alimentary tracts (ICRP, 1994a,2006) which have been used to estimate reference DCs under the current ICRP dosimetry system (ICRP, 2007). In the present work, Geant4 Version 10.06.p02 (Allisons et al., 2016) was used for all calculations, and MCNP6 Version 2.0 (Martz, 2017) and PHITS Version 3.10 (Furuta et al., 2017) were used for some limited cases for spot checking purposes.
7.1. External exposures
(109) Organ DCs were calculated for idealised external exposures, in terms of mean absorbed dose per fluence (pGy cm2), for six organs (i.e. RBM, colon, lungs, stomach, breasts, and skin) and four radiation types (i.e. photons, neutrons, electrons, and helium ions), and then compared with values calculated using the P143 phantoms (ICRP, 2020a) (see Annex H for calculation details). All the selected organs have the highest wT ( = 0.12) except skin; skin, despite the small wT (0.01), could significantly affect the effective dose calculation for external exposures to weakly penetrating radiations of low energies (Yeom et al., 2016, 2017), and its DCs themselves are important in the current ICRP dosimetry system (ICRP, 2007). (110) The comparison of the values shows that for uncharged particles (i.e. photons and neutrons), the DCs calculated using the MRCPs tend to be close to the values obtained using the P143 phantoms (ICRP, 2020a), and the differences are <10% for most cases. Exceptions are observed for photons at low energies (<20 (111) For charged particles (i.e. electrons and helium ions), the DCs calculated using the MRCPs are close to the values of the P143 phantoms (ICRP, 2020a) for electrons ≥20 MeV (≥2 MeV for the skin) and helium ions ≥500 MeV u−1 (≥50 MeV u−1 for the skin). At lower energies, on the other hand, significant differences are observed for the breast, RBM, and skin. The breast DCs of the 10- and 15-year-old MRCPs, for example, are significantly smaller (i.e. up to four orders of magnitude) than those of the P143 phantoms, which is due to the fact that the breasts are covered fully by skin in the MRCPs but not in the P143 phantoms. The RBM DCs of the MRCPs tend to be significantly smaller, which is because, as mentioned earlier, the spongiosa region is covered fully by cortical bone in the MRCPs. The skin DCs of the MRCPs and the P143 phantoms are significantly different (i.e. up to five orders of magnitude), which is due to the consideration of the micron-scale radiosensitive skin target layer in the MRCPs which was not possible for the P143 phantoms due to the resolution of the voxel geometry. (112) The effective DCs were also calculated using the MRCPs, in terms of effective dose per fluence (pSv cm2), and then compared with the values obtained using the P143 phantoms (ICRP, 2020a). For uncharged particles, the DCs calculated using the MRCPs are very close to those obtained using the P143 phantoms, and the differences are <5% for most cases. Relatively large differences were only found for photons at low energies (<50 (113) The DCs for the entire lens of the eye were calculated for photons using the paediatric MRCPs, and the calculated values were compared with those obtained using the P143 phantoms (ICRP, 2020a) (see Annex H for calculation details). For the antero-posterior (AP) geometry in which the dose to the lens of the eye is of primary concern, significant differences are observed by up to 2.5 times, except for the newborn, due to the different depth of the lens between the MRCPs and P143 phantoms. In other irradiation geometries, except at very low energies (<20 (114) In the paediatric MRCPs, the subregions of the teeth (i.e. enamel, cementum, dentin, and pulp) were modelled explicitly, in particular to support electron paramagnetic resonance retrospective dosimetry. Shin et al. (2021) calculated DCs for the enamel of the teeth, in terms of dose per air kerma, for the AP and right-lateral (RLAT) geometries using the MRCPs and then compared the calculated values with DCs for the entire teeth obtained using the P143 phantoms (ICRP, 2020a). The results of the study show that the differences in DCs are very large; that is, the differences are a few tens of times for most phantoms at 10 and 20
7.2. Internal exposures
(115) SAFs were calculated for photons and electrons, and then compared with values calculated using the P143 phantoms (ICRP, 2020a), as well as the Publication 155 values (ICRP, 2023) which were calculated using the P143 phantoms and MCNPX Version 2.7 (Pelowitz, 2008) (see Annex I for calculation details). Note that the SAFs obtained from the P143 phantom for self-irradiation were scaled to account for blood content, as were the Publication 133 and 155 values (ICRP, 2016a, 2023). The calculated SAFs cover four source organs (i.e. liver, lungs, cortical bone, and thyroid) and four target organs selected for each source organ considering contributions to effective dose. (116) For most cases, the SAFs calculated using the P143 phantoms (ICRP, 2020a) in the present work are in good agreement with the P155 values (ICRP, 2023); the differences are generally <10%, which mainly result from the differences in physics models or cross-section data between the Geant4 code used in the present work and the MCNPX code used in Publication 155. For photons and electrons at low energies (<20 (117) The comparison of the values for photons shows that for the liver, lungs, and cortical bone as source organ, the SAFs of the MRCPs and P143 phantoms (ICRP, 2020a) are marginally different, except at energies <50 (118) The opposite tendency is also observed. For example, the RBM ← liver SAFs of the MRCPs are generally smaller than those of the P143 phantoms (ICRP, 2020a) at the lowest energy considered (i.e. 10 (119) Photon SAFs obtained using the MRCPs and P143 phantoms (ICRP, 2020a) show relatively large differences when the thyroid is the source organ, which is because the thyroid was relocated to its typical position in the neck in the MRCPs. The ET ← thyroid SAFs of the MRCPs tend to be greater than those of the P143 phantoms; this is because the larynx, the location of which is very close to the thyroid, was modelled as part of the ET region in the MRCPs. (120) Electron SAFs calculated using the MRCPs are close to those obtained using the P143 phantoms (ICRP, 2020a) for the self-irradiation cases (e.g. thyroid ← thyroid), while for the cross-fire-irradiation cases, the differences are very large (i.e. up to more than four orders of magnitude) in some cases. The pattern of differences is similar to that for low-energy photons for most cases, but the differences are much larger for most cases. For example, the muscle ← cortical bone SAFs of the newborn MRCPs are smaller than those of the P143 newborn phantoms by up to a factor of approximately 2 for photons, but by up to a factor of approximately 80 for electrons. Nevertheless, the committed effective DCs are not expected to be very different, which is due to the fact that the self-irradiation SAFs, not cross-fire-irradiation SAFs, mainly affect the calculation of committed effective DCs due to the limited range of electrons for most radionuclides. (121) Electron SAFs calculated using the MRCPs for the alimentary tract were compared with the P155 values (ICRP, 2023) calculated using the Publication 100 (ICRP, 2006) stylised models (Choi et al., 2022). Although the absorbed fractions (AFs) are in good agreement for most cases, some differences in SAFs are observed, mainly due to differences in target masses between the MRCPs and the stylised models. However, for some cases (i.e. oral mucosa ← food; oral mucosa ← teeth surface; small intestine ← contents; and large intestine ← contents), significant differences in AFs, as well as in SAFs, are observed, which are attributed to the differences in the interorgan distances and organ dimensions. For example, for the small intestine as target region and its contents as source region, the AFs obtained using the MRCPs are larger than the P155 values, resulting in even larger SAFs with the smaller target mass. These differences result because the lumen diameter of the MRCPs is smaller than that of the stylised models, leading to less energy being absorbed in the contents (i.e. self-absorption), and thus more energy being deposited in the target region. (122) Electron SAFs calculated using the MRCPs for the respiratory tract were compared with the P155 values (ICRP, 2023) calculated with the Publication 66 (ICRP, 1994a) stylised models (Choi et al., 2023). The SAFs of the MRCPs are generally larger than the P155 values, mainly due to the smaller target masses of the MRCPs. For the bb region, when compared with the P155 values, the SAFs of the MRCPs are increased by not only the smaller target masses but also the electron cross-fire from other branches. For the AI source region, rather complex differences are observed, mainly due to differences in geometries and densities between the MRCPs and the stylised models.