Abstract

Editorial
Brachytherapy: A Lifesaving Treatment That Needs to Protect the Caregivers that Deliver the Therapy
In 1928, when 1000 experts in radiology from all corners of the globe gathered in Stockholm for the Second International Congress of Radiology, a key impetus was the protection of x-ray and radium workers in hospitals. This would have included the protection of workers practising brachytherapy with radium sources, which began as early as 1901 (Gupta, 1995). A ground-breaking result was the first international recommendations in this field (ICR, 1929), and the beginning of the long journey for what is now the International Commission on Radiological Protection (ICRP).
Today, the work of ICRP is much broader, covering the protection of patients, workers, the public, and the environment from all sources of ionising radiation. However, approximately one-third of ICRP’s work still focuses on radiological protection in medicine. This goes beyond the protection of patients to include medical staff, friends and family of patients, and the public who may be impacted. One reason for this is the massive use of radiation in medicine; globally, well over 100 medical examinations and treatments every second use ionising radiation, including approximately one brachytherapy treatment every minute. Another reason is that medical exposures represent approximately 98% of all artificial exposures to ionising radiation (UNSCEAR, 2008).
The use of radiation in medicine is continually evolving, and protection must adapt to the new techniques and technologies that continue to improve diagnosis and treatment. Some recent ICRP publications in this area cover radiopharmaceutical therapy, interventional procedures, medical imaging, cone beam computed tomography, and ion beam therapy (ICRP, 2014, 2015, 2017, 2018, 2019).
The present publication follows in a similar vein, focusing on the protection of medical staff for a specific treatment modality: brachytherapy. Brachytherapy involves placing small sources of radiation inside or immediately beside the part of the body to be treated. This has the advantage of delivering the radiation directly to the treatment volume, often with less radiation exposure to surrounding tissues than therapies that deliver the radiation from outside the body.
Brachytherapy is a critical modality in a modern radiation oncology department. It has demonstrated its value in our society for over a century through clinical outcomes as monotherapy and in combination with external beam radiation. The occupational hazard to radiation workers caring for a patient undergoing brachytherapy can be significant. However, this risk is minimal when appropriate quality control measures and assurance are implemented.
ICRP has made recommendations for the radiological protection of patients undergoing brachytherapy, including: prevention of accidents to patients undergoing radiation therapy (ICRP, 2000), prevention of high-dose-rate brachytherapy accidents (ICRP, 2005a), radiation safety aspects of brachytherapy for prostate cancer using permanently implanted sources (ICRP, 2005b), and overall recommendations in radiological protection in medicine (ICRP, 2007).
This publication is comprehensive for all types of brachytherapy procedures and sources used in modern clinics. The major focus is on photon-emitting sources, and high and low dose rates with manual and afterloading delivery systems. Other sources, such as beta-, neutron-, and alpha-emitting sources, are also discussed. In particular, the section on radiological protection in selective internal radiation therapy using 90Y microspheres is timely with the recent utilisation of this type of brachytherapy for liver cancer.
This publication is useful as guidance on the occupational protection of personnel involved in brachytherapy, such as radiation oncologists, medical physicists, therapists, and nurses; and also hospital administrators, radiation safety officers, those in charge of occupational protection, brachytherapy supplies vendors, regulators, and all those with an influence on the overall safety culture of a hospital. The requirements described to establish a quality assurance programme for both low- and high-dose-rate brachytherapy programmes are sound. The active participation of brachytherapy staff to develop and maintain a quality assurance programme is essential, considering ICRP’s recommendations for planned exposure situations. Routine external and internal audits will help make the clinical practice of brachytherapy safe for both personnel and patients. This publication should be used to guide new and current brachytherapy programmes to improve the radiological protection aspects. While such practices are already well defined by regulatory bodies such as the US Nuclear Regulatory Commission, this publication provides a cohesive report on all considerations for radiological protection in brachytherapy.
F
C
P
C
E
References
Occupational Radiological Protection in Brachytherapy
ICRP PUBLICATION 149
Approved by the Commission in March 2021
© 2021 ICRP. Published by SAGE.
Keywords: Occupational radiological protection; Interventional procedures; Exposure monitoring
MAIN POINTS
1. INTRODUCTION
(1) In brachytherapy, sealed radioactive sources are placed within the human body; these sources are implanted within, adjacent to, or in contact with the target tissue. As the absorbed dose (subsequently ‘dose’) falls rapidly with increasing distance from the sources, high doses may be delivered safely to a well-localised target or region over a short time period. This publication is focused specifically on brachytherapy, and brings together information relevant to occupational radiological protection in brachytherapy from the Commission’s published documents. The material and recommendations in the current publication have been updated to reflect the most recent recommendations of the Commission. (2) Parallel to the development of external radiotherapy, the use of radioactive sources inserted directly into tumours, or simply placed in contact, was explored in the early 20th Century. This technique was called ‘brachy (‘short’ in Greek) therapy’, literally ‘therapy at short distances’ by the English-speaking world, and ‘curiethérapie’, in order to honour the discoverers of radium, Marie and Pierre Curie, in France. (3) For brachytherapy, the area to be treated needs to be accessible, and the tumour or target location needs to be geometrically limited and of small to moderate size. Access generally involves some type of surgical intervention. The tumour is subjected to continuous irradiation to a total prescribed therapeutic dose for as long as the sources are present. (4) In the first decades of the 20th Century, most treatments were performed with radioactive sources inserted or placed in contact, temporarily, mainly using 226Ra tubes or needles; however, interest in permanently implanted sources dates back to the 1910s. 222Ra gas, the first daughter product (‘emanation’) of 226Ra, was felt to offer interesting advantages [i.e. small volume per unit of activity and a very short half-life (with the radioactivity becoming insignificant within weeks, so it could be implanted permanently)]. Permanent implants were initially performed using radon emanation contained in bare glass capillary pipes, approximately 3 mm in length and 0.3 mm in diameter. Apart from the problems linked to the production and implantation of such tiny glass pipes, another problem was that most of the dose was delivered by short-range beta particles (electrons), with some ‘overdosage’ of the tissues located in contact or close to the sources. To overcome this latter problem, tiny gold-encapsulated seeds were developed, with the gold casing filtering most of the electrons and the softer x rays, resulting in much better dose distribution. (5) A large number of patients, mainly presenting with gynaecological and prostatic cancers, received treatment with permanently implanted ‘radon seeds’ (sometimes called ‘gold seeds’ because of the jackets), with favourable results in some cases. Interestingly, radiographs of the pelvis after implantation of radon seeds for prostate cancer, performed in the 1920s, look rather ‘modern’, and not so different from current implantation images using 125I seeds (Aronowitz, 2002). However, this technique was progressively abandoned, mainly due to the complexity of managing the radium emanations and also because, at that time, most tumours were diagnosed at such an advanced stage that tumour extension exceeded the possibilities of cure by any type of implantation. (6) It was only in the 1950s that several groups re-activated techniques of permanently implanted sources using 198Au seeds (true gold seeds). The short half-life (2.7 days) of these sources allowed permanent implantation. 198Au seeds were used to treat a wide variety of tumours, including pelvic neoplasms. However, the use of gold seeds was progressively abandoned when 125I seeds became available in the 1970s. Approximately the same size (4 mm in length) as 198Au seeds, 125I seeds had a longer half-life (60 days), which was considered to be an advantage for slow-growing tumours such as prostate cancer, and the lower energy of its photons (∼28 keV compared with 420 keV for 198Au) allowed better radiological protection. (7) Since that time, 125I has become the standard for permanently implanted radioactive material, only challenged, more recently in some regions, by 103Pd, and most recently by 131Cs. A large variety of tumours have been implanted with 125I seeds. As examples, a number of patients had their tumour bed implanted after resection of lung carcinomas, and Memorial Hospital in New York implemented implantation for prostate cancer as early as 1970 (Hilaris et al., 1975; Aronowitz, 2012). 125I seeds have also been proposed for treating brain tumours (Marchese et al., 1984). (8) There have been no reports to date of adverse effects for medical staff, and/or the patient’s family, associated with permanent seed implantation. This shows that the technique, already applied to a significant number of patients, can be very safe. (9) In parallel, high-dose-rate [HDR; as opposed to the conventional low-dose-rate (LDR) brachytherapy described in the paragraphs above] remote afterloaded brachytherapy has gained wide acceptance, often in association with external irradiation (ICRP, 2005a). This is now used increasingly as monotherapy for early prostate cancer. (10) While external beam radiation therapy results in minimal (or no) occupational doses with an appropriately shielded facility, brachytherapy uniquely presents the possibility for doses to staff administering the treatments. In modern brachytherapy centres, radiation doses are incurred by staff (e.g. loading of seeds, sources, plaques, implants, associated fluoroscopy). A brachytherapy programme represents planned exposure situations that require active management. These planned exposure situations include operational exposures typical of such a practice (e.g. medical exposures of patients, exposures of comforters or carers, public exposures from permanent implants, and occupational exposures in applications involving source handling and image guidance), as well as potential exposures that may result from emergencies or actions following accidents. (11) There is wide variation in the practice of brachytherapy on a global scale, and facilities still practice older techniques with significantly higher staff dose potential (e.g. use of 226Ra, 131Cs, 137Cs, and 192Ir). In addition, technological developments and newer techniques present new protection concerns for staff that need to be addressed with specific recommendations for the practising medical community. (12) The Commission reviewed recent epidemiological evidence suggesting that there are some tissue reactions, particularly those with very late manifestation, where threshold doses are or might be lower than previously considered. This is the case for the lens of the eye (ICRP, 2011). Recent studies have shown that there is increased incidence of radiation-related lens opacities in some fluoroscopy users when radiological protection devices are not used properly, and radiological protection principles are not followed (Vañó et al., 1998, 2010, 2013; Ciraj-Bjelac et al., 2010; Rehani et al., 2011; Jacob et al., 2012). Fairly high radiation doses to the hands and legs of interventionalists and hair loss in the portions of the legs not shielded by a protective device have been observed (Balter, 2001). The considerable variation in operator doses observed for the same type of procedure indicates that radiological protection practices can be improved (Kim and Miller, 2009). (13) Physicians involved in brachytherapy procedures vary in their level of training in radiological protection. For example, in many countries, all radiologists receive training in radiation physics, radiation biology, and radiological protection as part of their radiology education, but physicians in other medical disciplines receive variable amounts of education in radiation-related topics, and may or may not be examined in these areas as part of the certification process. Publication 113 (ICRP, 2009) provides advice and recommendations on the minimum education and training, the professionals to be trained, objectives, contents, management approaches, approximate time needed to educate and train a wide variety of health professionals, accreditation, and certification. (14) The Commission has addressed specific patient-related radiation safety aspects associated with brachytherapy in several publications, including: Publication 86 (ICRP, 2001) on the prevention of radiotherapy (including brachytherapy) accidents; Publication 97 (ICRP, 2005a) on the prevention of HDR brachytherapy accidents; Publication 98 (ICRP, 2005b) on the radiation safety aspects of brachytherapy for prostate cancer using permanently implanted sources; and Publication 105 (ICRP, 2008) on overall recommendations for radiological protection in medicine. (15) Most common brachytherapy sources emit photons; however, in a few specialised situations, alpha-, beta-, or neutron-emitting sources are used. Intracavitary treatments employ sources placed in body cavities close to the tumour volume, while interstitial treatments employ sources implanted within the tumour volume. Intracavitary treatments are always temporary and of short duration, while interstitial treatments may be temporary or permanent. Temporary implants are inserted using either manual or remote afterloading procedures. Other forms of brachytherapy treatments include surface plaque, intraluminal, intra-operative, and intravascular applications where either gamma- or beta-emitting sources are utilised (IAEA, 2005). Recently, unique beta- (Cohen et al., 2014; Deufel et al., 2015) and alpha-emitting sources have become available (Arazi et al., 2007; Cooks et al., 2012). (16) Tables 1.1–1.4 summarise brachytherapy treatments with regard to the type of implant, duration of implant, method of source loading, and dose rate (IAEA, 2005). (17) ICRU Report 38 (ICRU, 1985) has defined numerical values of dose rate at the dose specification point(s) as a means of characterising brachytherapy by dose rate (i.e. low, medium, or high) (Table 1.5). In practice, HDR treatments are given with a substantially higher dose rate, >12 Gy h−1, than that given by the other two categories. For example, the usual dose rate employed in HDR brachytherapy units is currently approximately 100–300 Gy h−1 (Wakabayashi et al., 1971; Arai et al., 1992; Nag et al., 1999) or 1.6–5.0 Gy min−1, and some modern HDR remote afterloaders contain sources capable of delivering dose rates as high as 0.12 Gy s−1 at 1 cm distance in tissue. Medium-dose-rate brachytherapy is not in common use because of radiobiological complexity. In those few cases in which it has been used, the treatment results have been rather poor compared with LDR or HDR treatments (IAEA, 2005). Common uses of brachytherapy. Characterising brachytherapy treatments by implant type (IAEA, 2005). Characterising brachytherapy treatments by placement duration (IAEA, 2005). Characterising brachytherapy treatments by method of source loading. Characterising brachytherapy treatments by dose rate (ICRU, 1985). Note this historical characterisation of low dose rate for clinical brachytherapy differs from the typical radiological protection definition of low dose rate (i.e. <0.1 mGy min−1 for low linear energy transfer exposures, averaged over approximately 1 h) (UNSCEAR, 1993).





1.1. Purpose of this publication
(18) The purpose of this publication is to provide guidance on occupational protection of personnel involved in brachytherapy, such as radiation oncologists, medical physicists, therapists, and nurses; and also hospital administrators, radiation safety officers, those in charge of occupational protection, brachytherapy supplies vendors, regulators, and all those with an influence on the overall safety culture of a hospital. (19) This guidance includes tools and methods for occupational protection and exposure monitoring strategies; selection; use and testing of protective garments; development of a radiological protection programme; and education, training, quality management, and emergency response for implementation of the programme. (20) In brachytherapy, patients are exposed to ionising radiation from different modalities including brachytherapy, radiography, fluoroscopy, and computed tomography (CT). These modalities differ considerably in the frequency with which they are performed, in the radiation doses the patients receive, in the way that radiation is administered to the patients, and in the radiation dose to operators and staff. Radiography, fluoroscopy, and CT are not addressed specifically in this publication, but are addressed in detail in Publications 85, 117, 120, and 139 (ICRP, 2000b, 2010a, 2013a, 2018). (21) This publication does not address specific radiation therapy methodologies associated with brachytherapy, and cannot present an exhaustive discussion of brachytherapy techniques. The reader is referred to other available guidance for specific information on clinical techniques and considerations (e.g. ICRU, 1997, 2013; IAEA, 2002, 2005). This publication is intended to emphasise the radiological protection issues associated with brachytherapy for staff. (22) The guidance provided in this publication applies to all types of brachytherapy treatments that can generally be characterised by implant type, duration, method of source loading, and dose rate. (23) The biological effects of radiation have been addressed in several ICRP publications, and summarised with respect to radiological protection in medicine in Publication 105 (ICRP, 2007b). The use of dose quantities in radiological protection is discussed in detail in Publication 147 (ICRP, 2021).
2. RADIOLOGICAL ISSUES
2.1. Brachytherapy procedures
2.1.1. Practical source considerations
(24) Brachytherapy sources are usually encapsulated; this serves to contain the radioactivity, to provide source rigidity, and to absorb any alpha radiation and, for photon-emitting sources, beta radiation produced through source decay. Some brachytherapy techniques (e.g. 32P plaques or films) are not encapsulated with metal or plastic but are lightly coated with a siliconised epoxy (Cohen et al., 2014; Deufel et al., 2015), and other techniques rely on alpha-emitting nuclei ejected from wires loaded with 224Ra (Arazi et al., 2007; Cooks et al., 2012). (25) The clinically useful radiation fluence from a brachytherapy source generally consists of photons or beta particles, which can form the therapeutic component of the emitted radiation, as well as characteristic x rays and bremsstrahlung emitted incidentally that originate in the source or capsule. (26) The choice of appropriate radionuclide for a specific brachytherapy treatment depends on several relevant physical and dosimetric characteristics, including: energies, dose depth, shielding materials, half-life, half-value layer in shielding material, specific activity, and source strength. Regardless of the source used, brachytherapy is characterised by the typical steep ‘fall-off’ of dose with distance from the source. (27) The source energy influences penetration into tissue as well as the radiological protection requirements. Dose distributions in tissue, within the short treatment distances of interest in brachytherapy, are not influenced significantly by photon scattering when photon energies are >300 keV. However, tissue attenuation is highly significant for low photon energies of the order of ≤30 keV (IAEA, 2005). (28) The shielding required to protect against high-energy photons is many tens of millimetres of lead. For low-energy photons, the required thickness is much less, typically <0.1 mm of lead.
2.1.2. Physical source characteristics
(29) While the use of 226Ra and 222Rn has generally been discontinued because of safety concerns, their long history of clinical use still influences modern brachytherapy concepts. Well over a dozen radioactive nuclides have a history of use in brachytherapy. Some physical characteristics of several brachytherapy sources are listed in Table 2.1. Table 2.2 lists the radionuclides most commonly used for sealed source brachytherapy procedures. (30) Several guidance documents and publications discuss the specification of source strength for photon emitters and the determination of absorbed dose in patients, and these should be consulted for clinical applications of brachytherapy (e.g. ICRU, 1997). Physical characteristics of several isotopes used in brachytherapy (IAEA, 2005). ΓAKR, air kerma rate constant. Approximate values, depending on the source make and filtration. The use of generic values of air kerma rate constant or dose rate constant for a low-energy photon source may lead to substantial errors in dose calculations. They are therefore not given here for 125I and 103Pd. Radionuclides typically used for implantation (NCRP, 2006).


2.1.3. Mechanical source characteristics
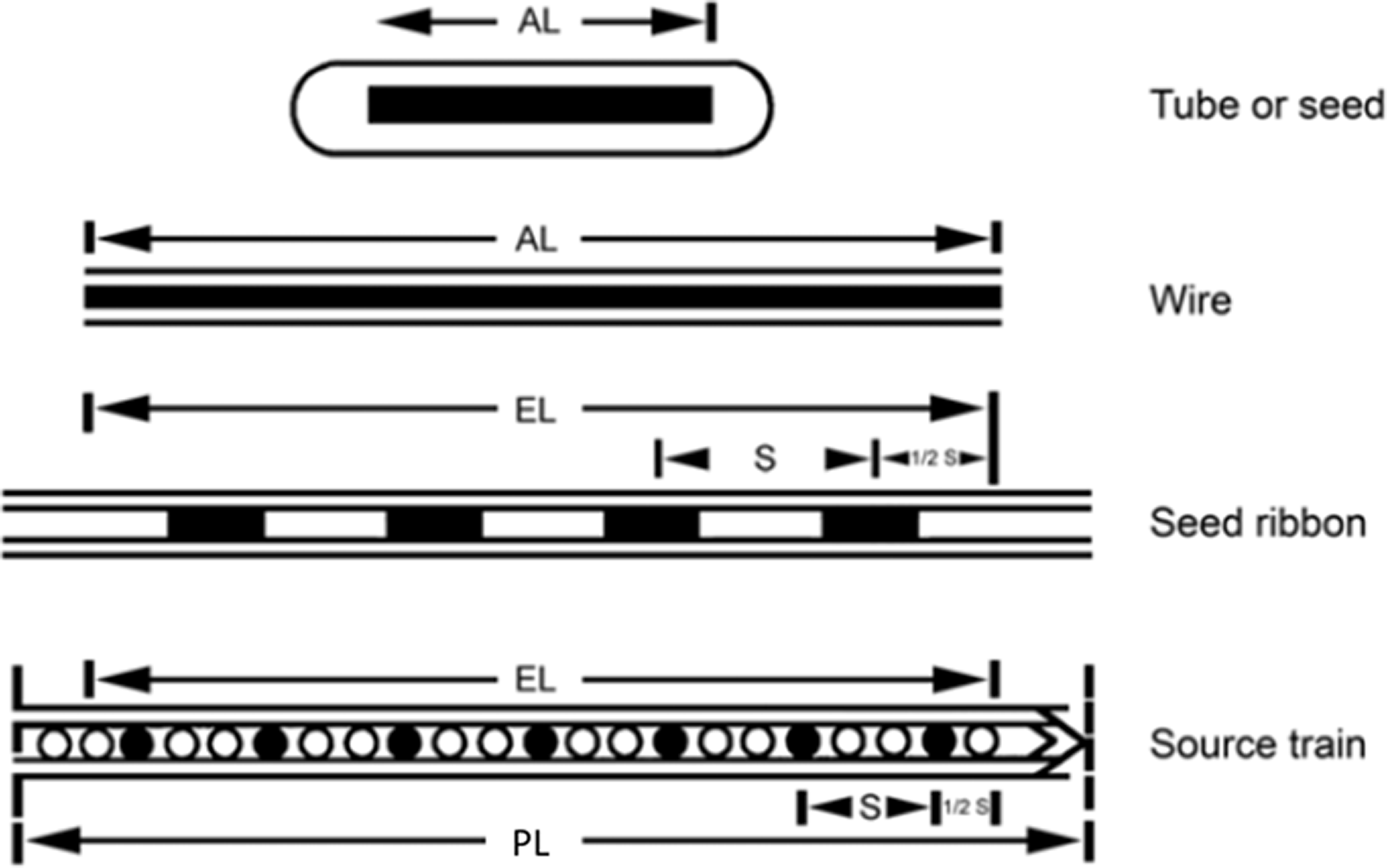
(31) Brachytherapy sources are available as seeds or plaques. Fig. 2.1 displays several mechanical forms. (32) 192Ir was historically available as wires, with the radioactive core being an iridium–platinum alloy with an outer sheath of 0.1-mm-thick platinum. LDR 192Ir sources are now available as seeds in strands of nylon ribbon. HDR remote afterloading units use specially designed 192Ir seed-like sources with typical initial activities of approximately 370 GBq. (33) 125I, 103Pd, 131Cs, and 198Au sources are available as seeds (e.g. individual, cartridge, or stranded). They are usually inserted into the tumour volume using special delivery applicators. (34) 60Co brachytherapy sources are available for HDR units with a typical initial activity of 80 GBq. (35) 90Sr is plated on the end of a rod to treat the benign disease pterygium (a non-cancerous growth over the conjunctiva of the eye) using beta radiation from the daughter product 90Y. (36) 32P plaques are planar sources where 32P is embedded in an epoxy polymer coated with silicone. (37) Novel devices have been developed (Arazi et al., 2007) consisting of needle applicators loaded with wires to which atoms of 224Ra are securely fixated. 220Rn is emitted from the wire through the alpha decay of 224Ra. 220Rn and its progeny diffuse through the surrounding tissue and deliver alpha radiation up to a few millimetres from the source. Mechanical source characteristics (ICRU, 1997). AL, active length; EL, equivalent active length; PL, physical length; S, separation between small sources.
2.1.4. Interventions for selective internal radiation therapy
(38) Less than 20% of patients with primary or metastatic liver cancers are curable at present. Therefore, palliative therapies such as interventional procedures for radioembolisation with pure beta emitter 90Y-labelled microspheres, and other loco-regional therapies have become alternative methods to treat patients with unresectable liver tumours (Camacho et al., 2015). After catheterisation of the hepatic arteries, 90Y microspheres are delivered under fluoroscopic control. The rationale for selective internal radiation therapy (SIRT) is the dominant hepatic arterial supply of malignant lesions, while the normal liver is mainly supplied by the portal vein. Some authors have suggested significant efficacy with SIRT (Bester et al., 2012).
2.2. Occupational exposure
2.2.1. Effective doses
(39) The effective dose limit for occupational planned exposure situations recommended by the Commission is 20 mSv year−1, averaged over defined periods of 5 years, with the further provision that the effective dose should not exceed 50 mSv in a single year (ICRP, 2007a). Additional restrictions apply to the occupational exposure of pregnant women. (40) Annual effective doses incurred by staff depend on their function and role in the brachytherapy team (oncologist, radiographer, nurse, anaesthesia provider, medical physicist, etc.), the type of brachytherapy procedure, the medical specifics and complexity of the cases, the patient population (e.g. paediatric patients, obese patients), and other factors, such as the skill of the team, available equipment, and relative use of associated imaging. Specific guidance with regard to monitoring is provided in Section 4. (41) Summaries and compilations of data on occupational exposure associated with concomitant fluoroscopy and interventional procedures are included in Publication 139 (ICRP, 2018), and are also available in the literature (Kim et al., 2008, 2012; ICRP, 2010a; NCRP, 2010).
2.2.2. Equivalent doses
(42) The equivalent dose limit for the skin (averaged over 1 cm2 area of skin regardless of the area exposed) for occupational planned exposure situations recommended by the Commission is 500 mSv (ICRP, 2007a). Some brachytherapy sources can deliver significant extremity doses (Tables 1.5 and 2.1), requiring special radiological protection considerations (Section 5.4). (43) The Commission issued a statement in 2011, published as part of Publication 118 (ICRP, 2012), after reviewing epidemiological evidence suggesting that there are some tissue reactions, particularly those with very late manifestation, where threshold doses are or might be lower than previously considered. For the lens of the eye, the dose threshold is now considered to be 0.5 Gy. For occupational exposure in planned exposure situations, the Commission now recommends an equivalent dose (ICRP, 2021) limit for the lens of the eye of 20 mSv year−1, averaged over defined periods of 5 years, with no single year exceeding 50 mSv. Although the dose to the lens of the eye is not typically of greater concern than the whole-body dose for general brachytherapy, some consideration should be given with regard to the use of fluoroscopy in brachytherapy procedures. Without protective eyewear, the dose to the lens of the eye may become the operationally restrictive dose for those cases with a high volume of associated fluoroscopy imaging (Lie et al., 2008; Korir et al., 2012), and the revised dose limit may be exceeded. Publication 139 (ICRP, 2018) provides additional information on equivalent dose to the lens of the eye and associated precautions.
3. APPLICATION OF THE SYSTEM OF OCCUPATIONAL RADIOLOGICAL PROTECTION TO BRACHYTHERAPY
3.1. Principles of radiological protection
3.1.1. General
(44) The Commission’s System of Radiological Protection aims primarily to protect human health (ICRP, 2007a). Its objectives are to manage and control exposures to ionising radiation so that tissue reactions (deterministic effects) are prevented, and the risks of stochastic effects are reduced to the extent reasonably achievable, societal and economic factors considered. To achieve these objectives, the Commission recommends three fundamental principles of radiological protection: justification, optimisation of protection, and limitation of individual dose (ICRP, 2007a). The principles of justification and optimisation apply to all types of exposure – occupational, public, and medical exposure – while the principle of dose limitation only applies to workers and the public, and does not apply to medical exposures of patients, carers or comforters, and subjects participating in biomedical research.
3.1.2. Justification of practices and procedures
(45) The principle of justification is that any decision that alters a radiation exposure situation should do more good than harm. This means that when introducing a new radiation source, or working to reduce an existing exposure or to reduce the risk of potential exposure, sufficient individual or societal benefit to offset the detriment it causes should be achieved (ICRP, 2007a,b). In the context of medical exposure, the aim of justification is to do more good than harm to the patient, subsidiary account being taken of the radiation detriment from the exposure of the radiological workers and other individuals (ICRP, 2007b).
3.1.3. Optimisation of protection
(46) The principle of optimisation of protection is that ‘the likelihood of incurring exposures, the number of people exposed, and the magnitude of their individual doses should all be kept as low as reasonably achievable, taking into account economic and societal factors. This means that the level of protection should be the best under the prevailing circumstances, maximising the margin of benefit over harm’ (NCRP, 1993; ICRP, 2007a,b). Optimisation of protection should be applied to the design of facilities that use ionising radiation; to the selection, set-up, and use of equipment; and to day-to-day working procedures (ICRP, 2007a,b).
3.1.4. Dose limitation
(47) The principle of dose limitation states that ‘the total dose to any individual from regulated sources in planned exposure situations other than medical exposure of patients should not exceed the appropriate limits recommended by the Commission’ (ICRP, 2007a,b). This principle applies to the exposure of medical workers. (48) For occupationally exposed workers in brachytherapy procedures, the dose limits for workers recommended by ICRP apply. In planned exposure situations, recommended dose limits for workers were established in Publication 103 (ICRP, 2007a), with an updated limit for the lens of the eye in the ICRP Statement on Tissue Reactions (ICRP, 2012). (49) The following limits apply:
whole body – an effective dose of 20 mSv year−1, averaged over defined periods of 5 years, provided that the effective dose does not exceed 50 mSv in any single year; extremities – hands and feet, an equivalent dose of 500 mSv year−1; skin – an equivalent dose of 500 mSv year−1, averaged over 1 cm2 area of skin regardless of the area exposed; and lens of the eye – an equivalent dose limit for the lens of the eye of 20 mSv year−1, averaged over defined periods of 5 years, provided that the equivalent dose to the lens of the eye does not exceed 50 mSv in any single year.
3.1.5. Dose constraints
(50) Optimisation is aided by setting a boundary on the predicted dose in the optimisation of protection (ICRP, 2007a). Such a boundary is called a ‘dose constraint’ in planned exposure situations, and is selected for planning purposes so that it effectively assists in the optimisation process, taking into account the current distribution of exposures. If it is subsequently found to have been exceeded, an investigation should be conducted to understand the circumstances, and it is unlikely that protection is optimised. Dose constraints are therefore lower than the pertinent annual dose limit. Dose constraints are established prospectively in the process of optimisation and are source related. When staff work in more than one facility, the dose limits and constraints should apply to the sum of all the individual doses incurred at the facilities. Dose constraints for the lens of the eye have been suggested by the International Radiation Protection Association (IRPA, 2017).
3.2. Investigations of abnormal doses
(51) There is no need to wait until an annual dose limit or constraint has been exceeded to become aware that protection was not optimised. Non-optimised protection can be detected by establishing an investigation level in terms of effective or equivalent dose received in 1 month, or based on the value of a related parameter, such as the reading of an over-apron collar dosimeter. (52) Exceeding a monthly investigation level provides an alert that protection was less than optimal in that period of time, and a review of existing radiological protection is needed. An increase in a dosimeter reading may be due to a substantial increase in the number of interventions, or in the dose per procedure, which may be due to an increase in procedure complexity or to a decrease in compliance with protection measures. (53) In 2000, the World Health Organization recommended that an investigation should be carried out when monthly exposure reached 0.5 mSv for effective dose, 5 mSv for dose to the lens of the eye, or 15 mSv to the hands or extremities (WHO, 2000). Following the new annual limit of equivalent dose to the lens of the eye, the investigation levels should be lowered accordingly. An investigation level of 2 mSv month−1 (ICRP, 2018), using the reading from a collar dosimeter, may be appropriate for staff involved in brachytherapy procedures. (54) An investigation level in terms of a monthly dose should be such that when extrapolated to 1 year, it would not exceed the relevant dose limit or dose constraint. In addition, personal dosimeters are not always worn or are worn incorrectly (Padovani et al., 2011; Sánchez et al., 2012). Investigation levels can be helpful in this situation by establishing minimum dose values for over-apron and hand dosimeters, thus providing an alert for possible poor compliance with procedures for wearing dosimeters.
3.3. Classification of areas and workplaces
(55) Para. 129 of Publication 57 (ICRP, 1990) discussed the possible classification of workers in categories with regard to the need for individual monitoring, and stated that interventional radiologists and cardiologists are likely to fall into Category A. Classification of workers, however, was not supported in Publication 60 (ICRP, 1991), and Para. 184 of Publication 103 (ICRP, 2007a) stated that ‘The Commission continues to recommend the classification of areas of work rather than the classification of workers’. The assignment of individual monitoring devices should, therefore, be analysed on the grounds of workplace and duties of the workers, their location and time of exposure within the radiation field, and the shielding provided by the protection devices used.
3.4. Embryo and fetus
(56) The Commission provided advice on the management of pregnant physicians and other workers in Publication 84 (ICRP, 2000a). The early part of pregnancy (before the pregnancy has been declared) is covered by the normal protection of workers, which is essentially the same for males and females. The first responsibility for the protection of the conceptus lies with the worker herself to declare her pregnancy to her employer as soon as the pregnancy is confirmed (ICRP, 2000a). Once the pregnancy has been declared and the employer has been notified, the working conditions of a pregnant worker should be such that the additional dose to the conceptus will not exceed 1 mSv during the remainder of the pregnancy (ICRP, 2000a). (57) Unnecessary discrimination against pregnant workers needs to be avoided. The restriction on dose to the conceptus does not mean that it is necessary for pregnant workers to avoid work with radiation completely, or that they must be prevented from entering or working in designated radiation areas (ICRP, 2000a). It does imply, however, that their employer should carefully review the exposure conditions of pregnant workers. In particular, their work should be such that the probability of high accidental radiation exposure is insignificant (ICRP, 2000a). (58) As an example of a professional society guideline, a clinical practice guideline for the occupational radiological protection of pregnant or potentially pregnant workers in interventional radiology has been developed by the Society for Interventional Radiology and the Cardiovascular and Interventional Radiology Society of Europe (Blake et al., 2006). This states that excluding pregnant workers from fluoroscopic procedures solely on the basis of radiation risks to the conceptus cannot be justified on scientific grounds (Blake et al., 2006; Best et al., 2011; Dauer et al., 2015). (59) In brachytherapy procedures, although typical occupational exposures are low, some considerations for pregnant workers should be made. Declared pregnant workers should not be expected to participate in emergency response activities associated with HDR sources.
4. INDIVIDUAL MONITORING AND DOSE ASSESSMENT
4.1. Individual exposure monitoring
4.1.1. Exposure monitoring and verification of compliance with dose limits
(60) Exposure monitoring is required for demonstrating compliance with annual dose limits as well as for optimisation of protection. Monitoring compliance with dose limits requires assessment of effective dose and equivalent doses to the skin, lens of the eye, hands, and feet. Equivalent dose and effective dose cannot be measured directly in body tissues, and cannot be used directly as quantities in exposure monitoring. The protection system therefore includes operational quantities that can be measured and from which equivalent doses and effective dose can be assessed (ICRP, 2007a). (61) Occupational exposure rests on a series of assumptions regarding the relationship between what is measured by a dosimeter and the dose received by an individual. Standards include accuracy requirements and uncertainties of the dosimetry system so that these assumptions hold for the relationship between operational and protection quantities. Ensuring that workers wear the dosimeters correctly during all work time is the most important component of this series of assumptions and relationships. No dose to an individual can be estimated reasonably in highly variable radiation fields without having some type of individual monitoring on the worker during all times of exposure. Auditing compliance with procedures is important to verify that the workers wear the dosimeters regularly and correctly.
4.1.2. Exposure monitoring and optimisation of protection
(62) For prostate implantation, lower doses correlate with increased experience of the brachytherapist in the use of shielding and long-handled applicators and tools (Schiefer et al., 2009). In most experienced centres, several hundred procedures can be performed per year prior to exceeding extremity dose limits (Schiefer et al., 2009; van Haaron et al., 2011) or effective dose limits (Schwartz et al., 2003). Similarly, for eye plaque procedures, hand doses were found to be low but measurable (Laube et al., 2000; Classic et al., 2012). In endovascular brachytherapy utilising 192Ir, upper limits of whole-body dose measurements were of the order of 10 µSv per procedure (Balter et al., 2000). Although rarely utilised now, when fluoroscopy is used in brachytherapy procedures, an increase in effective and extremity dose can be expected; however, with proper use of radiological protection devices, tools, and techniques, effective doses can be maintained well below the 20 mSv year−1 limit recommended by the Commission (Tsapaki, 2004; ICRP, 2007a, 2018; Dendy, 2008; Miller et al., 2010). (63) In addition to monitoring personal exposure, the use of dosimeters helps to increase awareness about radiological protection. In the absence of formal training in radiological protection, physicians in training tend to adopt the practices of their seniors (Rehani and Ortiz-Lopez, 2006). A strict policy on the regular use of personal dosimeters should be part of any quality programme in brachytherapy. Failure to wear monitoring equipment could be a breach of the employer’s procedures and/or local regulatory or legislative requirements. (64) Verification of compliance is not typically performed by checking doses from individual brachytherapy procedures, but by integrating the doses over many procedures carried out during a prescribed monitoring period. The period is established by the regulator and is usually 1 month. While this period is adequate for checking compliance with annual dose limits, it may not be sufficient for optimisation of protection in specific procedures. (65) For associated fluoroscopic imaging, actions taken to reduce patient doses will frequently translate into reduced scattered radiation levels or the times during which elevated levels exist, thus reducing worker exposure. Separate actions may also be taken that are directed specifically at the worker. The proper use of protective shielding, and locating staff in the lower-dose-rate areas around the sources are examples of optimisation actions, the outcome of which can be verified by individual exposure monitoring. Over time, the impact of optimisation will appear through lower occupational doses for comparable workloads and types of cases performed.
4.2. Characteristics of individual dosimeters and their use
4.2.1. Types of dosimeters: passive and active dosimeters
(66) Dosimeters need to have adequate accuracy under a variety of exposure conditions, and to be small and lightweight enough to be convenient to use and not interfere with the ability of staff to execute their tasks. Passive dosimeters are typically small, lightweight, and do not require power. This makes them easy to incorporate into packages that do not interfere with the staff’s actions and comfort, thus being the most widely used option, particularly for demonstrating compliance with dose limits. However, the absence of an instant reading capability is a disadvantage of all passive dosimeters for optimisation monitoring, especially for education of workers involved in brachytherapy. (67) For monitoring of the hands, small dosimeters on rings are used due to their relative ease of fit under surgical gloves. Rings can be sized for different finger diameters; attention is required to the fact that fingers may swell during long procedures. In addition, some additional features are important such as sterilisation capability and low interference with tactile sensation in the operator’s ability to manoeuvre catheters and instruments precisely. Fingertip sachets that fit over a finger have been used as an alternative to ring dosimeters. (68) The physical construction of the dosimeter has to be compatible with the intended wearing location. Infection control is a particular concern for ring dosimeters because some ring dosimeters do not withstand sterilisation processes, and they are typically worn during procedures where infection control is essential and thus are worn under the surgical gloves. (69) Active personal dosimeters (APDs) or electronic dosimeters may be used for optimisation monitoring or for special studies that require analysis of dose by procedure or discern aspects of a procedure. Active dosimeters are able to provide immediate information about dose rate, so rapid feedback is available to staff against which they can assess changes to their behaviour that result in lower dose rates and subsequently lower accumulated doses. Active dosimeters provide information on the time of each exposure, which facilitates correlation of occupational and patient exposures, and auditing of the wearing of the personal dosimeter during brachytherapy. (70) Optimisation monitoring does not need to conform to the strict dose quantities required for compliance monitoring. Optimisation seeks to compare relative values resulting from changes in conditions in order to evaluate the effectiveness of various actions to reduce dose. Electronic dosimeters are usually calibrated to assess operational quantities without taking into account the non-uniform irradiation of the body during brachytherapy procedures. That is, electronic dosimeters, like all dosimeters, indicate the dose at a single point and make no inferences regarding effective doses or doses at some distance from the dosimeter. Conceptually, there is no technical reason why multiple electronic dosimeters could not be worn and the data combined to yield compliance-type dose information, but practical issues have tended to limit the use of electronic dosimeters to investigatory and optimisation monitoring.
4.2.2. Dosimeter specificity
(71) To generate confidence in using a measurement made externally to the body for estimating doses occurring in the body, dosimetry systems have to meet standard requirements for accuracy, precision, and reproducibility for the operational quantity of concern. While most higher-energy brachytherapy sources can be monitored adequately with standard dosimeters, low-energy sources (e.g. 125I or 103Pd) may require special considerations and low-energy dosimeters (ICRP, 2005b, Appendix B), as will beta-, alpha-, or neutron-emitting sources.
4.2.3. Dosimeter reliability and simplicity
(72) The dosimetry system must be reliable and fail-safe; in other words, it must possess a continued ability for measuring the radiation field. In addition, actions required from the user should be simple and efficient to execute. For electronic dosimeters that require the user to energise the dosimeter, an item needs to be included in the procedures as an aide-mémoire for staff when putting on dosimeters. The fewer the actions and decisions required from staff, the greater the likelihood of compliance with monitoring. Integrating passive dosimeters such as those containing film, thermoluminescence dosimeters (TLDs), optically stimulated luminescence dosimeters, and radiophotoluminescent glass are generally used in brachytherapy practices for compliance monitoring.
4.2.4. Dosimeter exchange periods
(73) Passive dosimeters provide total dose accumulated over the period of use, and must be exchanged for new dosimeters at the end of the use period. The exchange period should be on a predetermined schedule to instil a habitual routine among staff. Generally, fluoroscopic staff should be monitored for monthly periods to provide dose data with sufficient frequency that unusual events can be detected, and appropriate responses implemented. Therefore, the radiation sensing material should have the sensitivity to detect the minimally relevant dose over the shortest period of expected use, and should retain the dose information for the longest period of expected use.
4.2.5. Approaches to detect incorrect dosimeter wear in brachytherapy procedures
(74) Problems with wearing dosimeters may involve not only high-dose readings but also very-low-dose readings which may suggest misuse of, or failure to wear, dosimeters. Publication 139 (ICRP, 2018) gives examples of incorrect use, including wearing a dosimeter that was intended for use under an apron over an apron, wearing a ring dosimeter on the incorrect hand, or wearing a dosimeter issued to another person. Indirect approaches (e.g. area monitoring or historical doses) may be useful in identifying a lack of compliance in wearing personal dosimeters, and in estimating occupational doses when personal dosimeters are lost or have not been used.
4.2.6. Different scatter conditions between type-testing and calibration, and real brachytherapy procedures
(75) Monitoring to assess effective dose has been attempted using a single or two dosimeters, for example, if whole-body dosimeters are calibrated and assessed without any consideration of the effects of shielding materials. Type-test standards tend to define performance evaluations under simple conditions with dosimeters being placed on a flat surface of a tissue equivalent phantom. Assurances should be requested from the supplier to verify that the measurement of the operational quantities is within expected dosimeter performance requirements for similar conditions to normal use.
4.2.7. Dosimeters for the lens of the eye
(76) Monitoring of the lens of the eye presents special challenges due to the difficulties in placing a device to which the dosimeter can be attached near the eyes. Small dosimeters may provide opportunities for locating dosimeters near the eye and under the protective lenses. Eye doses can be assessed from a dosimeter placed over the leaded apron at the collar or level of the neck, or another dosimeter on a strip of plastic attached to a headband such that the sensor is adjacent to the temple closest to the x-ray tube. Some methods of eye monitoring use a TLD chip wrapped in an elastic band that is fitted on the head near the eye (Bilski et al., 2011). In any case, dosimeters placed near the eyes must not interfere with the wearer’s vision. For brachytherapy procedures, assessments of doses to the lens of the eye can be made to decide if specific eye monitoring is required, especially in the case of concomitant fluoroscopic imaging use (ICRP, 2018).
4.2.8. Identification of the dosimeter and the worker
(77) Individual dosimeters should have a means to let the users identify their own dosimeters. A one-to-one relationship between a dosimeter and the user is indispensable if the dosimeter results are to be applied to a specific individual. Means of identification, such as labels, need to be easily readable to prevent someone from using another person’s dosimeter. A suitable approach consists of racks on which dosimeters are stored when not needed, and visual identification on the rack and on the dosimeter.
4.2.9. Calibration of active personal dosimeters
(78) In the course of the European project ORAMED, Clairand et al. (2011) and Sánchez et al. (2014) tested the influence of dose rate as well as pulse frequency and duration on the APD responses. With the exception of Geiger–Müller (GM)-equipped APDs, which did not give any signal in pulsed mode, the APDs provided a response affected by the personal dose equivalent rate, which means that they could be used in routine monitoring provided that correction factors are introduced. Type-test procedures and calibration of APDs and area monitors should include radiation fields representative of interventional procedures, including tests in pulsed mode with high dose rates (Chiriotti et al., 2011; Clairand et al., 2011; Sánchez et al., 2014).
4.3. Assessment of occupational exposure
4.3.1. Assessment of effective dose
(79) In general, effective dose is assessed from the reading of a personal dosimeter calibrated in terms of personal dose equivalent, Hp(10). This assessment of effective dose is sufficiently accurate for radiological protection purposes provided that the dosimeter is worn in a position on the body that is representative of its exposure, under the assumption of a relatively uniform whole-body exposure (ICRP, 2007). For those rare cases where brachytherapy is performed under fluoroscopic guidance, Publication 139 (ICRP, 2018) addresses considerations of a two-dosimeter approach, algorithms for monitoring when fluoroscopy is utilised, and specific guidance for assessing equivalent dose to the lens of the eye.
4.3.2. Assessment of exposure in selective internal radiation therapy
(80) A difficulty when using beta emitters for SIRT interventional procedures is the finger dosimetry of staff. TLD finger dosimeters should be worn on the index finger of the hand closer to the radiation source. Due to the very small distances between the beta source and skin and the concomitantly high dose gradient, the dose can be underestimated. At some workplaces, Rimpler and Barth (2007) measured local skin doses Hp(0.07) at the fingertips due to direct beta radiation of >100 mSv up to approximately 700 mSv per working day.
4.3.3. Assessment of exposure to the embryo and fetus
(81) For pregnant workers who perform or assist in brachytherapy procedures, dose to the conceptus is usually estimated using a dosimeter placed on the mother’s abdomen at waist level, under her radiation protective garments (Miller et al., 2010; NCRP, 2010). This dosimeter overestimates actual conceptus dose because radiation attenuation by the mother’s tissues is not considered. Specific evaluations need to be made depending on the sources being used in brachytherapy. For concomitant fluoroscopic imaging, the fetal dose is typically not more than half of the dose recorded on the dosimeter worn by the worker (Dauer et al., 2015), due to the attenuation by the mother’s abdominal wall and anterior uterine wall (Trout, 1977; Faulkner and Marshall, 1993; NCRP, 2010). Therefore, when two dosimeters are used, if the dosimeter under the protective apron shows a value for personal dose equivalent, Hp(10) < 0.2 mSv month−1, the equivalent dose to the conceptus over a 9-month period would be below the limit, unless there is significant use of high-energy photon emitters. Dosimeters should be evaluated monthly. Electronic dosimeters can be used to provide rapid access to data (Balter and Lamont, 2002).
5. RADIOLOGICAL PROTECTION METHODS AND PROGRAMME
5.1. Protection of staff
5.1.1. Control of exposures (time, distance, shielding, planning)
(82) Occupational radiological protection requires planning in order to minimise time, maximise distance, and use appropriate shielding as necessary to reduce exposures. Staff radiological protection cannot be handled independently from patient protection as they correlate in many ways. Simple measures, such as standing a little distance away from the sources or patient, and planning ahead in order to be able to carry out procedures quickly, consistent with case complexity, can be very effective in reducing occupational radiation dose. (83) For brachytherapy procedures, there are four types of shielding: architectural shielding, portable shielding, equipment-mounted shields, and personal protective devices. Architectural shielding is built into the walls of the procedure room. Rolling and stationary shields that are constructed of lead, steel, leaded glass, or acrylic and rest on the floor are useful for providing additional shielding for both clinicians and associated staff. These are often particularly well suited for use by nurses, medical physicists, and anaesthesia personnel. In some cases, personal protective devices such as a lead apron, leaded glasses, a thyroid shield, and shields suspended from the ceiling can provide protection and should be evaluated for use.
5.1.2. Use of adjuvant fluoroscopic imaging during brachytherapy procedures
(84) Brachytherapy procedures using adjuvant fluoroscopic imaging often require certain staff to remain close to the patient in order to manipulate catheters, applicators, and other devices. Other staff who provide assistance may also need to be in close proximity to the patient. The higher dose rates around the patient in a fluoroscopy room result from radiation scattered back from the patient. (85) Guidance for associated fluoroscopic use has been provided in Publication 139 (ICRP, 2018). In addition, a number of professional societies, radiological protection organisations, and others have issued guidelines on practices to be followed, and made recommendations on the use of protective devices for associated fluoroscopic imaging (Miller et al., 2010; NCRP, 2010; Chambers et al., 2011; Sauren et al., 2011; Durán et al., 2013; ICRP, 2013a,b; Hiles et al., 2016).
5.2. Protection from external exposures
5.2.1. Knowledge of radiation levels around a patient
(86) Knowledge of the distribution of radiation levels around a patient, understanding how different factors influence these levels, and the effective use of protective devices is indispensable for all staff involved in interventions (ICRP, 2009). Radiation emanating from a patient and its associated occupational exposure is determined by the brachytherapy sources employed, available shielding, the complexity of the procedures, the size of the patient, the modes of operation available on the equipment, and the skills of the operator.
5.2.2. Personal protective equipment
(87) Staff such as nurses and anaesthesia personnel who need to remain near the patient may benefit from the additional protection provided by movable (rolling) shields that can be positioned between them and the brachytherapy source. Shielding effectiveness depends heavily on the source characteristics and activity employed, and should be evaluated by medical physicists and radiological protection officers. Fluoroscopic aprons can provide some protection from the radiation emitted by sources of 125I, 103Pd, 131Cs, and 90Y alone or in combination with 90Sr and 32P. For higher-energy emitters, fluoroscopic aprons provide minimal protection at best, and can actually increase the dose to the skin. (88) The hands of brachytherapy clinicians can be close to the sources or primary x-ray beam if using image guidance. For fluoroscopic guidance, if the operators’ hands stray into the beam transmitted through the patient, the dose rate above the patient would typically be 2–5 µGy s−1, so a 1-min exposure would give a dose from 100 to 300 µGy. Lead-lined gloves may be considered as protection from the fluoroscopic beam, but do not allow the dexterity necessary for manipulating radioactive sources.
5.3. Life cycle of radioactive source safety
(89) Radioactive sources used in brachytherapy require safety and control along the whole life of the source, during production, packaging, shipping, receiving, calibration, use, decommissioning, and decay or proper disposal as waste. (90) The physical plant facilities required for a brachytherapy programme include a patient treatment room or procedure room (perhaps an operating room), imaging facilities, and a source laboratory (IAEA, 2008; Papagiannis and Veselaar, 2014). For radiological protection purposes, the rooms may need to be designated according to the magnitude of expected exposure or potential for exposure as controlled or supervised areas (IAEA, 2006; ICRP, 2007). Aspects of brachytherapy facility design are reviewed in the literature (IAEA, 2001, 2006, 2008; NCRP, 2006; GEC ESTRO, 2018). (91) Access to brachytherapy sources should be limited to personnel authorised for the task at hand. It is generally limited to authorised users, radiation oncology physicians, medical physics staff, and radiation safety staff. The radiation safety officer should maintain the active list of personnel authorised access to these sources. A brachytherapy source inventory log should be maintained, and should include the number and activity of sources added to storage, removed from storage, the patient name and room number, the time and date removed, the number and activity of the sources in storage after removal, as well as the number and activity of the sources returned to storage. (92) Brachytherapy sources should be shielded appropriately and stored in a locked room, often within a locked ‘safe’ or location within a controlled room. Some short-lived sources are stored in manufacturer’s shipping containers. Rooms should be posted accordingly as radiation control areas. (93) All radioactive sources transported within the institution, for example to and from a patient’s room, should be moved in either a shielded cart or the manufacturer’s shipping container under constant surveillance and control of medical physics or radiation oncology personnel. The transportation container should be locked or securely latched to ensure that sources are not released if the container is dropped or inadvertently bumped. The container should be surveyed during commissioning to ensure adequate shielding. (94) Radiation sources used in manual brachytherapy are the most significant source of occupational radiation exposure to radiation oncology personnel (NCRP, 2006), and have the potential to contribute significant doses to medical personnel and others who may spend time within or adjacent to rooms that contain radiation sources or patients administered various types of radiation sources. Occupational and public exposure may occur during receipt, transport, and preparation of sources; loading and unloading sources in brachytherapy applicators; and care of patients during the course of treatment. Significant dose reduction can be achieved through the use of appropriate facility design associated with sources that are being prepared; are in storage; or are being administered to, or are within, hospitalised patients or outpatients. (95) Facility design should consider medical and physical well-being of the patient as well as the protection of staff, visitors, and other members of the public from actual and potential radiation hazards. (96) Every brachytherapy facility should have the following equipment: a storage container in the treatment room to serve as an emergency shielded source receptacle; long-handled forceps; a portable radiation monitor instrument; and an area radiation monitor (ICRP, 2005a). If there is an alarm from a radiation monitor, procedures need to be in place to respond, and ensure that all radioactivity is accounted for and stored properly. (97) Brachytherapy treatments may require the preparation of radioactive sources (e.g. selection, counting, calibrating, trimming of ribbons, loading of intracavitary source inserts, etc.) and should be performed in specifically designated and designed rooms. Source preparation rooms (or source laboratories) should include consideration of the following: an area where all sealed sources can be stored safely in an orderly fashion with restricted access; a method of labelling and identifying sources in a shielded location; space and facilities for receiving and returning sources, calibration of sources, assessment of homogeneity, inventory, and quality control testing; space and equipment for source preparation for specific patient treatments; area for record storage; space for treatment aids; and space for storage of short-lived sources or temporary storage of unused or spent sources. Source preparation rooms should not be shared with other functions. Rooms should be posted with radiation warning signs and equipped with a lock to secure the area from unauthorised entry. Work benches of sufficient strength to support such shielding weight and source safes should be provided. Personnel shielding that facilitates source visualisation as well as personnel protection (e.g. lead blocks with leaded windows, etc.) of sufficient thickness to reduce whole-body and eye exposures should be provided. Occupancy of the area should be limited to persons immediately involved in source preparation. (98) Source manipulation should be made using forceps or tongs and never directly by hand. Appropriate personnel shielding, such as a cave of interlocking lead bricks or a lead L-block shield, must be provided and utilised. Wipe tests for source leakage or area contamination need to be performed periodically and the results documented. (99) Room layout should be carefully evaluated and planned for optimisation of protection. The need for the use of interlocking lead blocks on benches or wall shielding should be assessed as part of the planning. An assessment of the protection afforded to the operator and surrounding areas should be performed prior to initiating use. Changes to shielding should be assessed carefully.
5.4. Radiological protection considerations in specific applications of brachytherapy
(100) For common, specific applications of brachytherapy, the following sub-sections will address radiological protection considerations, and will address the following factors: facility design and shielding; protection considerations pre-procedure, during the procedure, and post-procedure; and response readiness.
5.4.1. Manually loaded, temporary implants
(101) Manually loaded, temporary implant (e.g. LDR) brachytherapy procedures, often interstitial brachytherapy or plaque placement, are used for various tumours, especially prostate, lung, brain, and eye. The sources are placed directly into or on the tumour. Such procedures can often be performed by initial placement of applicators, followed by loading the radioactive sources as afterloading. In other cases, the radioactive sources are placed directly into or around the target volumes with or without applicators. The pre-placement of applicators helps to minimise unnecessary radiation exposures to members of the medical staff (Papagiannis and Venselaar, 2014). (102) The careful placement of these sources for optimal treatment outcome is evaluated based on various planning dosimetry systems (including the Manchester system and the Paris system) (Thomadsen et al., 2005). Several modern systems utilise reverse dose planning to evaluate optimised source placement for tumour dose coverage (Lessard and Pouliot, 2001; Dewitt et al., 2005). (103) Exposure depends on a number of factors, including the radioactive sources themselves, and others subject to optimisation, such as the number of applications/year, the number of staff performing procedures, and rotation of the nursing staff. (104) Loaded-implant techniques expose all surgical suite personnel to ionising radiation, and can result in the delivery of high doses to the hands of the radiation oncologist or others involved in the treatment. (105) Radiation surveys (using appropriate devices, e.g. ion chamber or GM probe) should be performed prior to, during, and following brachytherapy procedures. Immediately after implanting sources in a patient, staff should perform a radiation survey of the patient and the area of use to confirm that no sources have been misplaced or lost. The survey should cover the entire room, waste bins, equipment, clinical staff, and protective clothing. Nothing should be removed from the room without an appropriate survey. (106) Following an implant brachytherapy procedure, the exposure rate should be measured and recorded for locations including: at the bedside; 1 m from the bedside; in the visitor’s area; at the doorway; and in the surrounding areas. Exposure rates in adjacent uncontrolled areas must conform to the local requirements and regulations. (107) The patient’s chart should be marked or labelled as ‘Caution, Radioactive Material’ during the time the sources are associated with the patient. Doors to patient rooms should be posted ‘Caution, Radioactive Material’ while the sources are present in the room. (108) Controls on visitor locations and visit durations should be established to ensure that doses to members of the public are maintained below 1 mSv year−1 and optimised to be as low as reasonably achievable (ICRP, 2007a). At all times, visitors should remain within areas that have been established as safe for visitors. Time limits for visits should be noted in patient or nursing instructions. (109) Applicator insertion is typically performed in a separate operating or procedure room that supports the surgical procedures needed to evaluate the patient’s condition, and expose or access the implant site. For many of these procedures, an imaging system (e.g. radiographic, fluoroscopic or CT unit) is required for intra-operative examination of source placement and geometry. (110) Treatment room or area facilities should be designed such that consideration is given to proximity to required ancillary rooms and equipment, functional adequacy of floor space needed for shields, occupancy of surrounding uncontrolled areas, structural integrity of the building needed to support the weight of required structural or portable shielding, and ability to control entry into the room. (111) Normally, designated rooms should be used for brachytherapy procedures. All rooms occupied by implanted patients or containing supplies of radioactive sources should be posted as controlled or restricted areas. Adjacent rooms may be used at the discretion of the radiological protection officer after surveys. The patient’s room should be as far away from the nursing station and heavy traffic hallways as is consistent with good medical care. Ideally, this would be a corner room on top or bottom floors. (112) During treatment, patients should be housed in a private room. The entire room occupied by an implanted patient should be considered a controlled area. (113) Protection of occupationally exposed persons may be met cost-effectively by grouping treatment rooms together in one or two limited areas, rather than using individual patient treatment rooms throughout the hospital. However, in some cases, the goal of providing good-quality medical care to implanted patients may be best provided on specific floors or areas based on specialised care. For example, patients with implants of the oral cavity, tongue, and neck may need specialised wound care, and the need to respond quickly to clinical problems may demand nursing skills that are not typically found in other nursing units (NCRP, 2006). It is possible that the development of two or three specialised facilities may be considered in high-volume locations (e.g. gynaecologic oncology, otorhinolaryngology, thoracic surgery). (114) Placing rooms in the corner of a building often avoids the need to shield all walls in the designated room, especially when treatment rooms are not located at street level. Optimally, a dedicated suite of adjacent rooms on both sides of a blind-end corridor can be designated for brachytherapy (NCRP, 2006). Upper and lower floor rooms may also need floor or ceiling shielding, or avoidance of occupancy by ‘sensitive’ patients (e.g. pregnant women, children). (115) Placing brachytherapy patients in existing, unshielded hospital rooms may expose persons in adjacent areas to an effective dose that could exceed 1 mSv year−1. There may be specific local regulatory requirements for limiting the dose in unrestricted areas that need to be met. Several actions can be taken to minimise radiation exposure to persons in adjacent areas, such as evacuation of adjacent patient rooms and use of portable shielding. Radiation measurements should be made after each unshielded hospital implant to confirm that the potential dose meets requirements. The radiological protection officer should be consulted to determine whether adjacent rooms should be vacated, or whether use of portable shielding or other actions could reduce radiation exposures in adjacent areas to acceptable levels (NCRP, 2006). This use of unshielded rooms should be discouraged or only accepted in case of emergency (peak in occupancy). (116) An intercom or video monitoring system may be useful to avoid unnecessary time spent near an implanted patient and thus reduce staff exposure (Papagiannis and Venselaar, 2014). (117) Any patient who has received a temporary implant should not be released from hospital care until both a radiation survey of the patient and room, and a count of the implanted sources, trains, or ribbons confirms that all sources have been removed from the patient and have been accounted for. This check should be performed immediately after the removal of the sources. A record confirming the source count and radiation survey should be maintained. (118) In some cases, 125I seeds with high specific activity are used for temporary interstitial implants (e.g. ophthalmological treatments). Due to the low-energy photons emitted by 125I, a thin lead-foil shield, a metallic applicator, or even tissue overlying the implant site reduces ambient exposure rates dramatically, eliminating or reducing potential radiation hazards to the attending hospital staff or members of the public. (119) Some techniques rely on balloon applicators for the treatment of malignant resection cavity margins. One of the treatment options for some brain tumours, particularly gliomas, has been external radiation therapy with or without the additional implantation of 125I seeds. An alternative balloon technique relies on the installation of an organically based liquid labelled with activities up to 18.5 GBq of 125I into a balloon previously placed in the surgical cavity at the time the tumour was excised (Dempsey et al., 1998). The organic liquid and the balloon are then withdrawn after several days of treatment. The radiological protection considerations for this treatment are more typical of radiopharmaceutical therapy, and include contamination concerns and radioactive waste disposal. Another consideration is the possibility of radioiodinated molecules leaking out of a ruptured balloon, or diffusing through the balloon membrane into the cavity and being de-iodinated to liberate radioiodide which is then transported to and concentrated in the thyroid (DeGuzman et al., 2003; Strzelczyk and Safadi, 2004). Some of the solution that defuses through the balloon membrane passes into the patient’s urine, resulting in contamination (Adkinson et al., 2008). (120) The treatment room or patient’s room should be posted with signs, ‘Caution, Radioactive Material’ and ‘Radiation Area’, or similar. In addition, information for visitors should be posted at the entrance. The exposure rate, air kerma rate, or dose rate should be determined at a standard distance (e.g. 1 m, 30 cm, ‘contact’) from the centre of the implant in the patient with an appropriately calibrated survey meter, such as a portable ion chamber. (121) The total exposures to medical personnel or any unsupervised individuals, including visitors, over the life of the implant should be assessed for consistency with the facility’s programme for optimisation of protection. Any additional special precautions should be written down and included in the patient’s chart as required to satisfy dose constraints and limits for medical staff, carers, or members of the public. (122) Access to brachytherapy treatment rooms by healthcare personnel not involved in treatment (or by the public) should be controlled. Typically, nursing personnel are responsible for ensuring compliance with restrictions defined in the patient’s chart (NCRP, 2006). Nurses should notify the radiological protection officer and radiation oncologist in the event of missing or displaced sources, significant changes in implant position, or any other circumstances threatening safety. (123) A shielding container of sufficient size and shielding effectiveness to safely hold any sources that could become dislodged, and tools for the remote handling of a source, source train, or applicator containing sources should remain in the patient’s room for the duration of the implant. (124) Linens, food, utensils, rubbish, and excreta should not become contaminated; however, linens and rubbish should remain in the room until surveyed to ensure that no displaced sources are present. (125) During source removal, surgical dressings near the implanted applicators or sources should be removed carefully and checked by an appropriately trained medical staff member, taking care not to dislodge the implant. Sources should be removed using a remote handling device and placed immediately in a shielded container. A source inventory must be maintained to verify that all sources documented on the written prescription or order have been removed (visual inspection of source integrity and number). A second source-by-source (seed-by-seed) count should be performed in the source preparation area. Permanent storage locations should be adequate to provide both safeguarded inventory control and shielding. If at any time, a source appears to be lost, the radiation oncologist, medical physicist, and radiological protection officer should be contacted immediately, and the rooms secured. (126) Following verified removal of the sources from the patient’s room, a careful survey of the patient, the treatment room, and removed applicators should be performed using an appropriate survey meter (e.g. a GM detector), and the results documented. (127) Treatment rooms should not be released for cleaning and occupancy by another patient until the sources are securely removed, the source inventory is reconciled, and the radiation survey verifies that no sources remain in the room.
5.4.2. High dose rate and pulsed dose rate
(128) Radiation exposure to hospital staff responsible for source loading and the care of implant patients during treatment can be greatly reduced or eliminated by use of remote afterloading technology (Glasgow and Corrigan, 1995; Papagiannis and Venselaar, 2014). Several robotic remote afterloading systems have been developed to help minimise the radiation exposure to the medical and support staff associated with afterloading techniques. In addition, the use of remote afterloading devices offers several practical advantages over manual procedures, including increased patient treatment capacity, and consistent and reproducible treatment delivery. Such remote afterloading devices are used in both interstitial and intracavitary clinical applications. (129) The most common indications for HDR brachytherapy are treatment of cervical, endometrial, oesophageal, breast, prostate, and lung cancers; and skin and soft tissue sarcomas in adults and children. Intra-operative HDR brachytherapy is practised in some larger facilities. (130) Specific activity is an important source-selection criterion for HDR brachytherapy. The three commonly used radioactive sources in remote afterloading devices are 60Co, 192Ir, and, formerly, 137Cs (IAEA, 2005). Currently, the most commonly used source for afterloading is 192Ir because of its medium average photon energy (∼400 keV) and its high specific activity. However, its relatively short half-life is a distinct disadvantage as frequent replacement of sources is required (typically three to four times per year) (ICRP, 2005a), involving an ongoing use of resources and cost. Therefore, several facilities in certain countries are now employing 60Co sources with a longer half-life. (131) Most HDR systems use a single source of 192Ir, with a typical activity of approximately 370–500 GBq, delivering treatment dose rates at 1 cm exceeding 4 Gy min−1 (possibly as high as 8 Gy min−1). A single source (0.6–1.1 mm in diameter and 4–12 mm in length) is located at the end of a drive cable or wire which stops sequentially at each programmed treatment position, or ‘dwell’ position. This allows for technical flexibility, as each dwell position can be placed at various positions along a catheter track, and each dwell time can be programmed individually. (132) As HDR instantaneous dose rates are so high (up to 450 Gy h−1 at 1 cm), this modality requires a well-organised procedure, well-trained technical staff, and a comprehensive programme for safety, quality assurance, and emergency procedure. The need for detailed written procedures, checklists, written communication, and personnel training is especially critical in HDR brachytherapy. Various groups have developed comprehensive protocols for developing and maintaining safe treatment delivery processes (Kutcher et al., 1994; Nath et al., 1997; Kubo et al., 1998; Kaulich et al., 1999; NCRP, 2006). (133) Within the 24 h preceding any remote afterloading treatment, the correct operation of the system and its ancillary safety devices should be confirmed by performing standardised quality assurance tests. Remote afterloaders should only be operated according to written procedures, and according to a written prescription or treatment plan defining the prescribed treatment sequence. (134) HDR treatments are staffed by radiation oncology personnel, including therapists, dosimetrists, medical physicists, and radiation oncologists. Personnel need to be trained adequately on the specific model of HDR remote afterloading system used in order to avoid possible confusion leading to errors, and so they are able to promptly identify and correct any errors that may occur. (135) HDR facilities require an HDR treatment room, which can be a dedicated room, a linac room, or another room (e.g. CT scanner room normally used to plan radiation treatments – so-called ‘CT simulator’) built with shielding sufficient for HDR use (Glasgow and Corrigan, 1995). Note that CT rooms would likely need additional shielding added to the walls, and possibly ceiling and floor, to accommodate an HDR unit. Access to a radiographic imaging system for treatment verification and planning is useful for a broad range of treatment indications. An operating room or procedure room is also required to perform insertions, such as in cervical or prostate brachytherapy. (136) Inventory control and risk of the loss of individual sources is low with remote afterloading brachytherapy units. The risk of source loss is extremely small in HDR or pulsed-dose-rate (PDR) brachytherapy as there is only one source, it is housed inside the afterloader, and that is kept in a locked and controlled area. However, HDR facility design should include considerations regarding security of these areas, as the potential for portable sources used in HDR units to be stolen and placed in so-called ‘dirty bombs’ has caused many facilities to increase the level of security for these areas substantially. In addition, a secured facility helps to prevent inadvertent exposure of individuals tampering with the HDR unit itself. A security plan should be developed for HDR facilities that addresses keys, locks, cameras, and tamper indicators as deemed necessary. Access to the operator’s key for the HDR unit control console should be controlled by the radiation oncology staff, and restricted to a list of specified and trained individuals. (137) Essential components of all remote afterloading systems are a shielded compartment (or safe) to house radioactive sources (single or multiple), a local or remote operating console, a source control and drive mechanism, a source transfer guide tube and treatment applicators, and a treatment planning computer. Remote afterloaders are equipped with a timer that automatically retracts the sources when the programmed treatment time, corrected for gaps and interruptions, has been administered. HDR remote afterloaders are also typically equipped with an inert wire, mechanically identical to the wire housing the radioactive source, that is used to verify unobstructed access to the lumens before the radioactive source is deployed. (138) Overall requirements for HDR infrastructures can be found in IAEA documents (IAEA, 1998, 2001). HDR facility design should include significant engineered and administrative controls, such as: (i) a door interlock system that causes the source to retract automatically if the treatment room is entered while the source is out of its safe – this withdrawal should result from the interruption of an interlock switch located on the treatment room door; (ii) an independent radiation monitor visible at the room entrance with power back-up; (iii) appropriate radiation warning signs and ‘beam on’ light that is activated whenever the source is in the exposed position; (iv) systems for maintaining visual and aural contact with the patient during treatment (e.g. television monitoring systems and two-way intercom systems); and (v) a copy of the operator’s manual, including emergency procedures. (139) An HDR unit or facility design should include fault detection logic capable of detecting source retraction failure, separation of the source from its cable, and unscheduled displacement of the source from its programmed positions. Systems should alert users to the problem and prevent further treatment. Error detection and recovery systems located on the HDR afterloader should be tested thoroughly before implementation and at appropriate intervals thereafter. (140) Emergency procedures should be developed for rapid detection of HDR source retraction failures and bringing the source under control. These procedures should include the use of a radiation survey meter, and tools to safely manipulate the source and removal of the applicators if needed. Emergency response equipment should be present whenever the device is used, including a shielded container for source placement if it is not possible to return the source to the shielded home position. (141) A radiation monitoring system that is independent of the HDR unit should be installed inside the HDR treatment room to monitor the room radiation levels. Systems installed at the door should give both a visible and audible signal to ensure awareness. If the area monitor or treatment device indicates a source retraction failure, the responsible medical staff should respond immediately. Malfunctions of the afterloader or its ancillary safety systems should be brought to the immediate attention of the radiation oncologist and medical physicist present for the procedure. Additional emergency procedure considerations are discussed in Section 6. (142) Following completion of treatment, a careful survey of the patient, the treatment room, removed applicators, and the afterloading housing should be performed using a calibrated radiation detector (e.g. GM detector) to confirm complete retraction of the sources. Survey results should be documented in the patient’s treatment record. Treatment rooms should not be released for cleaning and occupancy by another patient until the radiation survey is complete, and is negative for an incompletely retracted source. (143) It is estimated that > 500 HDR accidents (including one death) have been reported along the entire chain of procedures from source packing to delivery of dose (ICRP, 2005a). Human error has been the prime cause of radiation events. Many accidents could have been prevented if functional monitoring equipment had been available, and if staff had paid attention to the results. Publication 97 (ICRP, 2005a) specifically addresses the prevention of such errors, and represents an important aspect of overall occupational brachytherapy radiological protection. Institutions should consider participation in the IAEA Safety in Radiation Oncology voluntary reporting and learning system in radiotherapy and radionuclide therapy incidents and near-misses with the purpose of sharing safety-related events and safety analysis for improved safe planning and delivery of treatments. (144) Although radiation exposure of personnel is almost completely eliminated (in properly shielding facilities) for HDR procedures, there are several radiological protection considerations during HDR afterloading, including the requirement for a shielding procedure suite, constant source shielding when in the retracted position, the potential for accidental high exposures and serious errors due to increased complexity (Thomadsen et al., 2003), and other problems such as failure of the source to retract. (145) For HDR treatments, survey instrumentation should be selected carefully so that the instrument does not saturate in high radiation fields. If false readings could occur in high-intensity fields, an ion chamber survey meter should be used to cover the upper extreme of the exposure rate range. Before HDR treatment, the functioning of radiation detectors should be checked. (146) PDR units have a similar design as HDR units, but the activity is approximately one-tenth of that used for HDR brachytherapy. PDR treatments are used in large implants (e.g. sarcomas, head and neck tumours, gynaecological, etc.). Treatment is over the same duration as LDR treatments in order to mimic favourable radiobiology. As such, the treatment requires hospitalisation of the patient during source administration, and a dedicated and appropriately shielded treatment room where a patient can stay for ≥ 1 day. (147) PDR devices use a single 37-GBq 192Ir source and are programmed to deliver HDR treatment pulses of short duration, with dose rates as high as 45 Gy h−1 at 1 cm, usually at hourly intervals (e.g. a source steps out for approximately 10 min h−1 and then retracts), to simulate continuous LDR treatments. Such fractions are described as ‘pulses’ and the interval between successive pulses, during which the source remains in its shielded safe, is the ‘quiescent’ period. Radiological protection considerations during PDR afterloading are similar to LDR implants because the average hourly absorbed dose rate, in Gy m2 h−1, and total reference air kerma, in Gy m2, are unchanged. However, the use of large pulse widths for several days may make it possible to exceed 1 mSv to staff over the treatment duration in uncontrolled areas. Therefore, before implementing a PDR brachytherapy treatment, the user should evaluate the average hourly and weekly exposures to determine that the proposed dwell time per pulse and cumulative dwell time will not exceed the appropriate shielding design goals. Example procedures for implementing such a requirement have been published (Williamson and Li, 1995). (148) Advantages of PDR brachytherapy include: emulation of LDR brachytherapy in terms of biological effect; optimisation of dose distribution; and visitors and staff can use the time between pulses while the source is in the safe shielded position to interact with the patient. A disadvantage of PDR brachytherapy is that the lack of stability of applicators over the course of treatment is similar to that for LDR therapy. A possible radiological protection disadvantage of PDR brachytherapy relates to the potential radiological safety hazard of a source stuck in the patient. LDR brachytherapy typically uses low-activity sources, which allows latitude in addressing sources that become dislodged. In HDR brachytherapy, the medical physicist or other staff are present during treatment to react quickly to a radiological emergency; in PDR treatments, it may be difficult to guarantee the availability of someone with sufficient training at all times. Therefore, education and continuous exercises on emergency response procedures for on-site staff are essential radiological protection programme elements for PDR (ICRP, 2005a). (149) HDR brachytherapy facilities require a properly shielded area that should be designed to limit the annual effective dose as a result of brachytherapy procedures to members of the public, including other patients, to 1 mSv year−1. For adjacent controlled areas, shielding should be designed to control occupational exposures to the annual dose values specified by an institution’s exposure control programme. For HDR brachytherapy facilities, portable shields should not be used for this purpose. The adequacy of the proposed or existing shielding design should be reviewed by a qualified expert. Before implementing HDR treatments, the dose rates in surrounding areas should be measured using properly calibrated ion chamber survey meters (NCRP, 2005). If the results indicate that the applicable effective dose values could be exceeded, the facility should limit the patient treatment workload, augment the shielding, or appropriately limit occupancy in surrounding areas to prevent the applicable values from being exceeded. (150) Radiation shielding should be designed by a qualified expert to ensure that the required degree of protection is achieved. The qualified expert should be consulted during the early planning stages as the shielding requirements may affect the choice of location and type of construction. Qualified experts should be provided with all pertinent information regarding the radiation equipment and its use, type of construction, and occupancy of nearby areas. (151) This section does not attempt to summarise the regulatory or licensing requirements of the various authorities that may have jurisdiction over such facilities. It is expected that a qualified expert will be fully aware of such matters, and will account for them in the final shielding designs. (152) The exposure rate from a point-radiation source (typical in brachytherapy) in free space varies inversely as the square of the distance from the source. When the radiation source is brought outside of the shielded housing (or ‘safe’), the radiation field will be essentially isotropic. Therefore, there are essentially no secondary barriers as all barriers may be exposed to the source as well as to radiation scattered from the patient and objects in the treatment room. (153) The exposure time involves the total time that the source is present outside of self-shielded housing. (154) The occupancy factor for an area is the average fraction of time that the maximally exposed individual is present while the sealed source is in use and outside of its self-shielded housing. For example, a waiting room might be occupied at all times during the working day, but have a very low occupancy factor as no single person is likely to spend >50 h year−1 in any given waiting room (NCRP, 2005, 2006). However, for areas where personnel are continuously present in a particular area, the occupancy factor might approach 1. In most cases, the maximally exposed individual will normally be an employee of the facility. The occupancy factor for controlled areas is usually assigned a value of 1. (155) In calculating required shielding, workloads should be estimated conservatively (i.e. including a safety margin) and should include source exposure anticipated for quality assurance, source calibration, and other measurements. For example, in HDR treatments, a moderately large workload might be estimated at 100 patients year−1 with an average between three to five treatment fractions per patient (NCRP, 2006). It is usual to assume that the workload will be distributed evenly throughout the year. (156) Source types and activities should be considered carefully in the design of shielding. Tables of half-value layers and tenth-value layers can be useful in designing appropriate shielding for brachytherapy treatment. For example, Table 5.1 lists relevant information for typical HDR sources. Lead density is typically taken to be 11.36 g cm−3 and normal concrete density is estimated at 2.3 g cm−3. (157) For a dedicated HDR treatment room, 40–60 cm of ordinary concrete or 5–7 cm of lead would typically be required to shield uncontrolled areas, depending on the location of the source relative to the areas under consideration and the occupancy of the adjacent areas. All walls, the ceiling, and the floor in the HDR treatment room should serve as primary barriers. When HDR units are placed within existing linear accelerator vaults, it is typical that no additional shielding is necessary. Primary beam teletherapy shielding is usually more than adequate for HDR treatments; however, secondary scatter shields and doors may not be adequate. The influence of HDR source position on shielding efficacy should be evaluated by a qualified expert. In those cases where shielding design restricts the source to a designated location within the room, the location should be permanently marked or fixed on the floor. In addition, such a dual-use room may need to be modified to be equipped with required interlocks for HDR procedures, as well as a method to ensure that the external beam system is locked out of use during HDR procedures. (158) Shielding of treatment rooms should be constructed so that the shielding is not compromised by joints; by openings for ducts, pipes, or other objects passing through the barriers; or by conduits, service boxes, or other structural elements embedded in the shielding barriers. Estimated shielding parameters for 60Co, 192Ir, and 169Yb (NCRP, 1976; Delacroix et al., 1998; Granero et al., 2006; Lymperopoulou et al., 2006; CNSC, 2017).

5.4.3. Permanent implants
(159) For permanent implants, minimising radiation exposure to staff and the general public has greatly influenced the choice of radionuclide. Classically, high-energy radionuclides with half-lives of the order of a few days were used. 222Rn gas encapsulated in gold tubing and, later, 198Au seeds were used for permanent implants. The patient had to be confined to a controlled area until source decay reduced ambient exposures to acceptable levels. Such classical implant types delivered high doses to the hands of the radiation oncologist and exposed inpatient hospital personnel to high-energy radiation. (160) Currently, longer-lived but very-low-energy photon emitters are used for permanent implantation (i.e. 125I, 103Pd, 131Cs). A patient’s own tissues or a thin lead foil are typically sufficient to limit exposure to the hands and fingers of the radiation oncologist, and eliminate the need to hospitalise patients solely for radiological protection purposes. (161) For prostate seed implantation, a transperineal (closed) surgical procedure, with ultrasound, fluoroscopy, or CT guidance, is typically utilised and is most often carried out as an outpatient 1-day procedure. (162) Various types of single-seed, seed-train, and stranded-seed implantation instruments can be used to implant seeds. For single-seed applications, preloaded cartridges containing 10–15 seeds are placed in an applicator. By ejecting each seed at a controlled distance, a linear array of seeds can be implanted. Linear arrays of seeds contained within a semi-rigid absorbable suture material are also available. Source trains can be assembled by placing sources and non-radioactive spacers of various length in needles, with or without linkages, to allow for the prescribed source distribution throughout the prostate. (163) A study of staff exposures during LDR prostate implantation procedures found that staff received whole-body doses of approximately 90 µSv and extremity doses of approximately 600 µSv per case (Schwartz et al., 2003). Fluoroscopy time was the predominant factor, with radiation oncologists receiving whole-body doses of approximately 8 µSv min−1 and extremity doses of approximately 50 µSv min−1 during active fluoroscopy. Those groups using ultrasound guidance alone are expected to receive lower occupational doses. (164) There are several radiological protection considerations during permanent seed implantation, including: minimising exposure to the operator’s hands; inventory control; minimising large dose-delivery errors; detecting contamination or leaking seeds; and monitoring loading trays after seed handling. Many of these actions are integral elements of an overall brachytherapy quality management system (see Section 5.7). (165) Verification of the number of seeds used has proven to be a common problem with permanent implants (Stutz et al., 2003). Inventory control is essential at all points of pre-treatment planning through implementation. As the implant procedure progresses, it should be possible for a member of the implant team (e.g. medical physicist, resident, or therapist) to verify the seed count and source activity independently. If there is a discrepancy in the count, radiation safety staff should be notified and rooms should be checked for any seeds that may be lost. These checks may be performed using portable radiation detectors, such as GM counters or sodium-iodide scintillation meters. If a CT scan is performed on the patient post-implant and pre-discharge, the CT scan may be used as further verification of the seed count if a discrepancy persists. (166) Following completion of implantation procedures, a member of the implant team should measure radiation exposure from the patient on the surface of the patient’s body and at 1 m from the approximate centre of the implant. These measurements should be made using a calibrated ionisation chamber survey meter. The readings should be entered into the patient’s chart and tags or labels indicating the date on which radiation precautions are no longer necessary. (167) As permanent implants are typically performed using low-energy emitting radionuclides, the dose rates from sources and patients who have received permanent implants are also typically low (Table 5.2). Several investigators have reported direct measurements on dose rates from patients. Michalski et al. (2003) evaluated total dose to family members of 44 patients. He gave dosimeters to the patient, spouse, children, and pets, as well as monitoring four rooms frequently occupied by the patient. Low levels of exposure were found; for example, the mean lifetime dose to a spouse was 0.1 mSv for 125I implantation. Dauer et al. (2010) evaluated the potential doses received from patients following prostate brachytherapy implantation in the context of developing data-based instructions. After typical implantation with 125I, no precautions at all were required for co-workers and non-pregnant adults (even those sleeping with the patient). As an example of suggested precautions, at their median exposure rate of 5 µSv h−1 at 30 cm, the authors reported that the patient should avoid sleeping ‘in contact’ with a pregnant patient for 84 days, and should avoid holding children on their lap for long periods of time (more than 1–3 h) for 42 days. Kono et al. (2011) reported on the dose received from the implanted patients. From a series of measurements at 20, 50, and 100 cm, the authors concluded that the dose from patients following prostate brachytherapy to the general public is quite low. These evaluations and measurements show that the doses to staff will likely remain very low, certainly lower than the occupational limits to the whole body and the extremities, and potentially lower than the constraint levels set for comforters and carers of such patients. (168) The Commission has previously provided radiological protection recommendations on the release of patients following permanent implantation (specifically prostate brachytherapy) in Publication 98 (ICRP, 2005b), and readers are encouraged to consult that publication for more detailed information. A patient who has received a permanent implant cannot be discharged until it has been determined that the patient meets local regulatory requirements for such release. Direct measurements from low-dose-rate permanent implant patients (ICRP, 2005b; Dauer et al., 2010). MSKCC, Memorial Sloan Kettering Cancer Center.
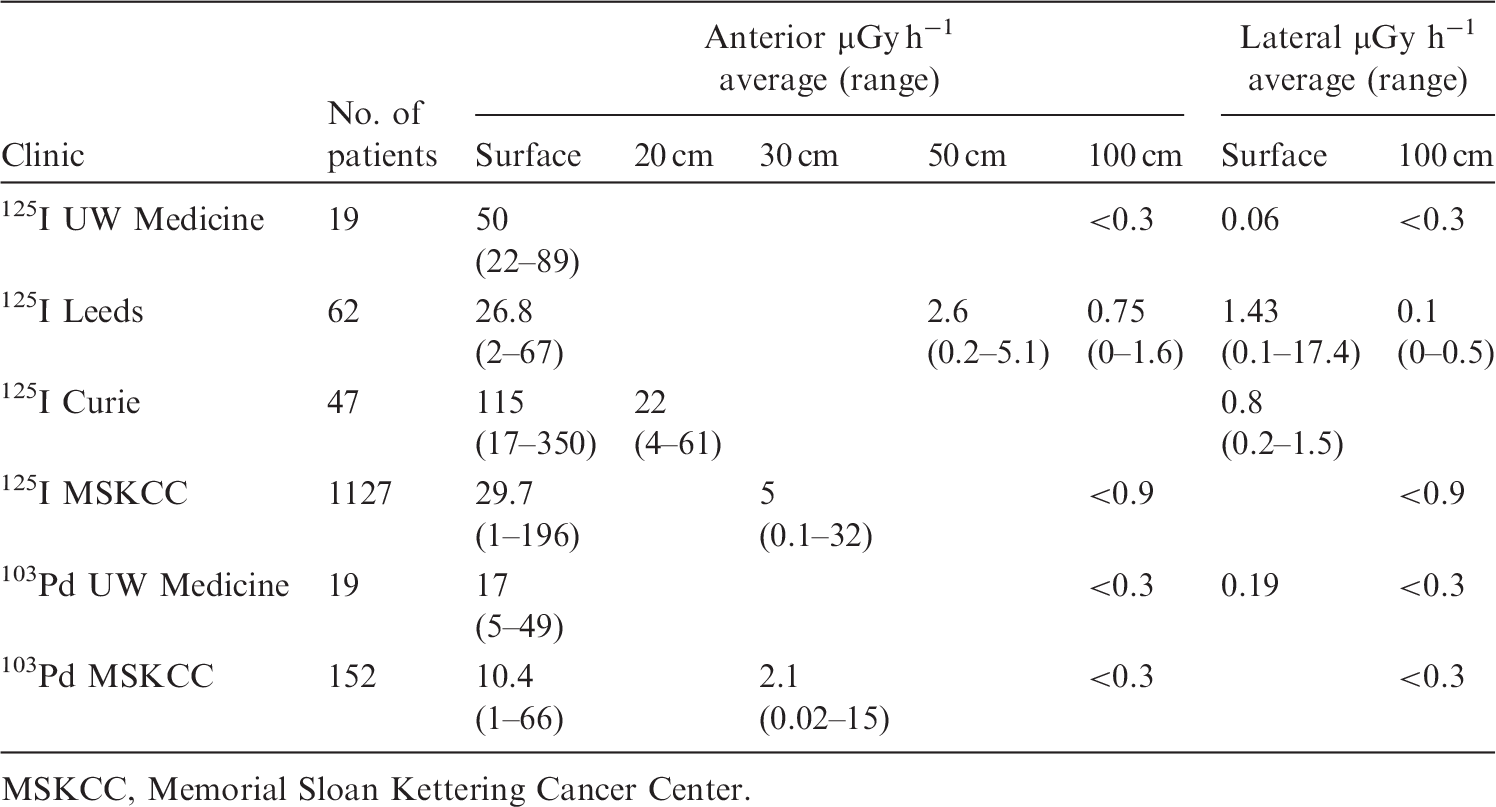
5.4.4. Intra-operative brachytherapy procedures
(169) Several institutions with large brachytherapy programmes are utilising HDR units in shielded operating rooms for intra-operative radiation therapy. These programmes combine surgery and radiation oncology. The tumour is exposed and a single fraction of radiation is delivered through the open wound. These programmes are usually only available in institutions that can commit the resources necessary to build a dedicated operating room suite for brachytherapy. The unique radiation safety issues of such facilities and associated shielding design have been discussed elsewhere (Anderson et al., 1999; Sephton et al., 1999; NCRP, 2006).
5.4.5. Intravascular brachytherapy
(170) The potential role of radiation in preventing restenosis after angioplastic treatment or stent placement has been studied using brachytherapy techniques. Pre-clinical and clinical investigations used catheter-based radiation sources or radioactive stents to deliver dose to the affected coronary artery vessel wall. Sources used include 192Ir as a medium-energy gamma-emitting source, and 90Sr/90Y, 90Y, and 32P as beta-emitting sources (Nath et al., 1999). (171) Typical intravascular dose fraction sizes range from 10 to 20 Gy at the luminal surface or external elastic lamina at a point approximately 2–3 mm from the catheter centre for coronary arteries, and as much as 5 mm from the catheter centre for peripheral arteries. 192Ir ribbons with dose rates as high as 4000 G m2 h−1 (Teirstein et al., 1997) have been used. (172) There are several radiological protection considerations during intravascular brachytherapy, including: the high level of activity needed to produce adequate dose rates carries the potential for significant personnel exposures; and catheterised patients cannot be moved to shielded vaults for treatment. (173) An evaluation of the shielding and the dose rates in surrounding areas should be performed for any intravascular brachytherapy. Evaluation should consider the number of procedures possible without exceeding the permissible dose limits in surrounding areas. Portable shielding may be needed to supplement structural shielding (Balter et al., 2000; Bohan et al., 2000; Folkerts et al., 2002). (174) The failure of the sealed source to retract into its housing has been identified as a source of abnormal incidents with intravascular brachytherapy devices (NRC, 2004). Therefore, an additional shielded container should be available in case of emergencies.
5.4.6. Electronically generated low-energy radiation sources
(175) Electronically generated low-energy radiation sources (ELS) refer to equipment utilising x-ray sources with a peak voltage of up to 120 kVp to deliver a therapeutic radiation dose to clinical targets (Devlin et al., 2017). ELS devices fall into two categories: a modern version of Grenz-ray dermatological treatment units; and devices that deliver therapy in a similar manner as intracavitary brachytherapy applications using 50-kVp x rays (electronic brachytherapy). (176) The main advantage of ELS over 192Ir HDR brachytherapy or megavoltage electrons is that the emitted and associated scatter energy is lower, allowing for much less radiation shielding. All of these devices may be used with short source-to-surface collimation, or surface applicators for treatment of tumours of the skin; electronic brachytherapy units can also be used for intracavitary treatments, such as for breast, vagina, and brain (Bhatnagar, 2013; Vaidya et al., 2014; Alvarado et al., 2015; Ouhib et al., 2015; Safigholi et al., 2015). (177) The use of low-energy radiation does not mean that ELS is without risk to patients and healthcare personnel. Some intra-operative applications of ELS have been shown to result in exposure rates to operating room staff of 2 mSv h−1 at approximately 30 cm from a treated area (Mobit et al., 2015). As in all forms of radiation therapy, ELS requires proper initial and ongoing training of the entire treatment team, with detailed attention to personnel, equipment, patient, and personnel safety (Devlin et al., 2017). (178) Techniques in risk analysis and development of a quality management programme for electronic brachytherapy have been developed by a task group of the American Association of Physicists in Medicine based on the principles of Task Group 100 (Huq et al., 2016; Thomadsen et al., 2020).
5.4.7. Selective internal radiation therapy
(179) SIRT is a technique to deliver radiotherapy for cancer or metastases in the liver. SIRT is generally implemented by interventional radiologists, nuclear medicine physicians, and radiation oncologists. Vials containing 90Y-labelled microspheres should be handled with forceps and appropriate shielding to reduce finger doses. Due to the high-energy beta emission, shielding is best provided by a material with a low atomic number, such as polymethyl methacrylate. Vendors of SIRT spheres provide advice and training material to minimise the contamination risk to staff, patients, and the room (SIRTEX Medical, 2013). This includes the use of special shielding boxes for preparation and injection. Furthermore, double gloves are recommended to allow removal of a contaminated outer glove with a gloved hand. For implantation of the microspheres, the vendor provides an acrylic delivery box and delivery set. This prevents direct contact with the 90Y vial and all stopcocks or tubes. Table 5.3 gives a representative overview of typical exposure of the different staff members for a single SIRT procedure. The actual values depend on the type of microspheres used. ICRP has developed additional guidance associated with SIRT in Publication 140 (ICRP, 2019). (180) Only a few papers on occupational doses from SIRT have been published. Occupational exposure from SIRT procedures is caused by x rays with a relatively low dose rate and by direct beta radiation, especially to the hands and fingers, with high dose rates if precautions are inadequate. In addition to the dose to the hands of workers preparing the individual patient dose and to the physician implanting the microspheres, there is potential for significant contamination hazard. Exposure data are 43.5 mSv MBq−1 h−1 skin equivalent dose due to contact with a 5-mL syringe, and 1.35 mSv kBq−1 h−1 due to contamination with 50 µL on 1 cm2 (Kemerink et al., 2012). Specific advice to reduce this hazard is given in Publication 140 (ICRP, 2019). (181) In addition to all technical measures of radiological protection, training to perform all steps of the procedure efficiently leads to a significant reduction in occupational exposure. Aubert et al. (2003) demonstrated extremity dose reduction by optimising the 90Y injection technique. They found an extremity dose reduction factor of >10 after optimisation of the procedure. (182) After SIRT, the patient requires observation, general nursing care, and accommodation. Based on 143 SIRT procedures (124 with resin spheres and 19 with glass spheres), McCann et al. (2012) reported mean equivalent dose rates of 1.1 µSv h−1 at 1 m for resin spheres and 2.4 µSv h−1 at 1 m for glass spheres. Typical dose equivalent rates 6 h after implantation of 2-GBq 90Y activity (SIRTEX Medical, 2013) are shown in Table 5.4 for different distances. Typical measured dose per patient for the technician or pharmacist preparing a typical patient administration, and for the physician implanting that prepared dose (SIRTEX Medical, 2013). Typical ambient dose equivalent rates 6 h after implantation of 2-GBq 90Y activity for different distances.
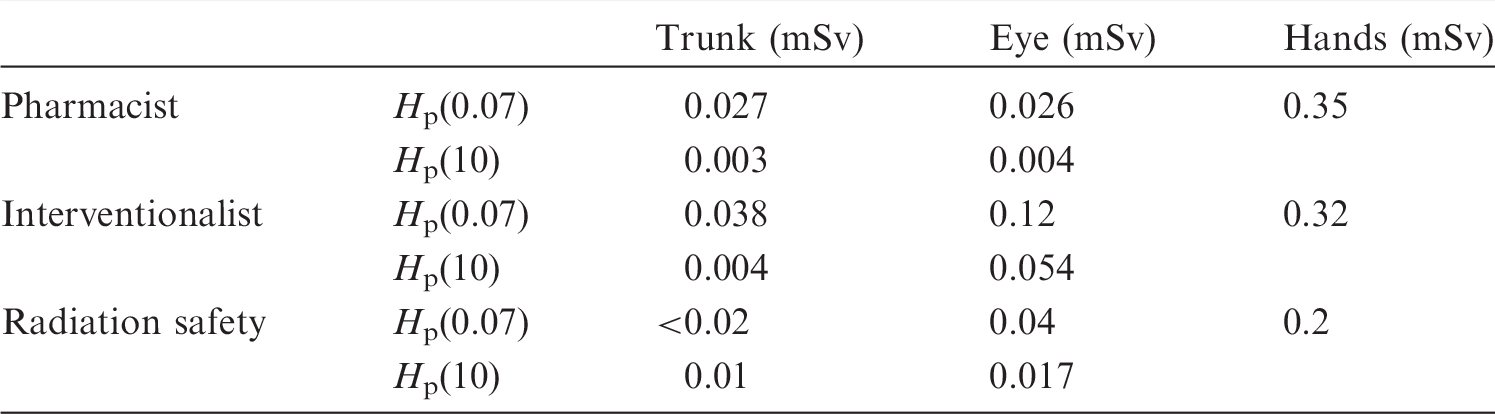

5.5. Education, training, and credentialing
(183) The Commission has addressed the specifics of minimum levels of training for interventionalists, nuclear medicine specialists, medical physicists, nurses, radiographers, and technologists, among others, in Publication 113 (ICRP, 2009). (184) Legislation in most countries requires that individuals who take responsibility for medical exposure must be properly trained in radiological protection. However, a training system and accreditation mechanism is still lacking in some countries. (185) Publication 97 (ICRP, 2005a) has provided specifications for personnel requirements and training that are applicable for all brachytherapy facilities in general and should be consulted. Clearly, a major prerequisite for the development of a safe brachytherapy facility is adequately trained staff (ICRP, 2005a). A multi-disciplinary team should be organised including, at a minimum, a radiation oncologist, a medical physicist, a technician, and a nurse. (186) All brachytherapy team members must receive adequate training (and certification where appropriate) in the brachytherapy procedures to be implemented before being authorised to perform (or take part in) the procedures. Training programmes should include initial training for all incoming staff, and regular updates and retraining. (187) Specific training in radiological protection must also be performed. Training activities should be followed by an evaluation of the knowledge acquired from the training programme to test competency before the person is awarded certification. If certification in radiological protection is required for some medical specialties, certification should be obtained before the individual is permitted to practice the specialty. Scientific and professional societies should contribute to the development of the training syllabi to ensure a consistent approach, and to promote and support education and training. Scientific congresses should include refresher courses on radiological protection, attendance at which could be a requirement for continuing professional development for professionals who use ionising radiation (ICRP, 2009). (188) Facilities performing brachytherapy should provide oral and written radiation safety instructions to all personnel involved in patient care associated with brachytherapy. Refresher training at periodic intervals should also be provided. At a minimum, the radiation safety instructions should include information regarding the size and appearance of brachytherapy sources, safe handling, and shielding instructions in the event a source becomes dislodged, as well as procedures for notifying the radiation oncologist and radiation safety officer if the patient dies or has a medical emergency. (189) In addition to general knowledge on radiological protection, all staff participating in brachytherapy procedures guided by radiological imaging need awareness of the distribution of scattered radiation levels around a patient, understanding of how different factors influence the dose distribution, and familiarity with the effective use of protective devices (e.g. shields, leaded eyewear, shielding curtains and drapes). This knowledge should be achieved by initial training, and maintained and updated through continuous education, consistent with the evolution of technology. (190) The radiation oncologist is responsible for the overall procedure, as brachytherapy is a medical treatment. He/she should be properly accredited according to each country’s regulations. Specific responsibilities of the radiation oncologist or interventional radiologist, nuclear medicine physician, and surgical oncologist include (Kutcher et al., 1994):
patient evaluation; patient selection; treatment protocol selection; treatment prescription; applicator insertion(s); imaging review; selecting tumour, target, and treatment volumes; treatment plan approval; applicator(s) removal; evaluation of tumour response and side effects; implementation of radiation safety instructions and emergency procedures; and patient follow-up. (191) Even if the radiation oncologist has experience in HDR, PDR, or LDR brachytherapy, additional training is required in the other types of brachytherapy (ICRP, 2005a). ‘Hands-on’ training is indispensable. (192) Medical physicists and radiological protection specialists providing support to brachytherapy facilities should have the highest level of training in radiological protection as they have additional responsibilities as trainers for oncologists and other health professionals involved in the procedures (ICRP, 2009). Specific responsibilities of the medical physicist include, at a minimum:
equipment life cycle, including testing equipment at the time of acceptance of new equipment or after major repairs; verification of calibration of sources; performing source exchange, if necessary; checking the treatment unit – verifying source positioning, indexing, internal gamma alarm, etc.; checking patient set-up including applicator positioning; supervising imaging; treatment planning and dose calculations; implementation of radiation safety instructions and emergency procedures; and supervising treatment administration by the technicians. (193) The medical physicist should participate in preparation of the patient after the applicator has been inserted and prior to obtaining images, as it is during such preparation that x-ray marker wires are positioned in the applicators as necessary. It is also necessary to select the angles of radiographic images or to select planes in the event of verification by CT or magnetic resonance imaging. (194) For HDR treatments, the medical physicist should be trained in the use of the HDR planning system, and should become thoroughly familiar with applicator image reconstruction. Training in equipment use, security systems, and emergency procedures are mandatory. Medical physicists should also be trained in the basic principles and procedures of radiological protection. (195) Personnel monitoring services staff need background knowledge of the clinical practice for calibrating dosimeters appropriate for brachytherapy and any associated fluoroscopic use (e.g. radiation qualities, scatter radiation fields, pulsed radiation), and for investigating abnormal dose values. (196) Technologists and brachytherapy technologists are typically in charge of the following:
checking applicators and specific accessories (alternatively, a nurse); daily checking of treatment units; assisting the radiation oncologist during implantation (alternatively, a nurse); obtaining images for localisation; using treatment planning under the medical physicist’s supervision; delivering treatment (for HDR or LDR afterloading devices, etc.); monitoring each treatment from the console; implementation of radiation safety instructions and emergency procedures; and recording treatment on appropriate documents. (197) Nurses are typically in charge of assisting the physician during each procedure. Specific responsibilities include:
daily checking of the treatment or patient rooms; ensuring supplies of disposables, gynaecological packs, etc.; scheduling of patients (alternatively, a technologist or other clinician); receiving patients and sending them home; implementation of written discharge instructions that include radiation safety instructions and emergency procedures; and assisting the radiation oncologist/interventional radiologist during implantation. (198) The role of the manufacturers is of increasing importance (ICRP, 2010c). There is a need for the design of built-in safety engineering, carefully tested software, design of informative warnings, self-test capabilities, self-explanatory user interfaces, and internal safety interlocks to prevent improper use that may lead to accidental exposures. Technology- and technique-specific training are important for users, as well as installation and maintenance engineers.
5.6. Records related to occupational protection
(199) The records to be kept are established as requirements in standards and regulations. Records of occupational exposure include information on the nature of the work in which the worker is subject to occupational exposure monitoring, including, for interventional staff: information on work for other employers that involves radiation exposure; outcomes of health surveillance; education and training on radiological protection, including refresher courses; and results of exposure monitoring and dose assessments, including results of investigation of abnormal exposure values. Employers should provide staff with access to records of their own occupational exposure. (200) Information on workload, in terms of procedures per year, is useful for optimisation of protection and for comparing and investigating unusual exposure.
5.7. Quality management system
(201) All facilities performing brachytherapy procedures should establish a comprehensive quality assurance and management programme (QAP) with well-defined objectives to ensure compliance with standard good practices. QAPs in brachytherapy should cover all of the planned and systematic actions necessary to provide confidence that optimum quality has been achieved in the entire diagnostic and treatment process. The QAP should include aims for maintaining best radiological protection practice to ensure appropriate occupational exposure control (ICRP, 2007; IAEA, 2014). Active participation of staff involved in the use of radiation is advisable, taking into account the Commission’s recommendations for planned exposure situations. The QAP should be part of the management system implemented at institutional level, including regular and independent audits (internal and external). (202) A QAP for brachytherapy includes all of the aspects of radiological protection of patients and staff in addition to the usual clinical aspects. A QAP should include physical, clinical, and organisational aspects applicable to the brachytherapy modality. The details of a full clinical QAP are beyond the scope of this publication, and the reader is referred elsewhere (e.g. IAEA, 1998, 2001; Kubo et al., 1998; NCRP, 2006). (203) Most clinical QAPs have as their main objectives: the preparation of a physician’s written directive before administration of treatment; clear identification of the patient; documentation of treatment and related calculations; compliance of each treatment with the written directive; and the identification and evaluation of any unintended deviation from the prescription. (204) A radiological protection programme must be established to ensure compliance with regulations for radiation safety and protection as promulgated by local governing agencies (IAEA, 2001, 2008). A qualified radiological protection officer, who is responsible for implementing the radiological protection programme, should be identified, officially appointed, and given sufficient administrative authority, in writing, to supervise the programme. A radiological protection committee should also be established and should include, at a minimum, an authorised user of each type of use permitted by the licence: the radiation safety officer, a representative of the nursing service, and a representative of management. (205) Two basic objectives of the radiological protection QAP are to evaluate patient radiation dose on a periodic basis, and to monitor occupational radiation dose for workers in brachytherapy facilities where radiation is used. The radiological protection component of a QAP for brachytherapy should be an independent portion of the general QAP for x-ray, radiation oncology, and nuclear medicine installations in a particular health centre. (206) The radiological protection advisor or radiation safety officer should be involved in monitoring occupational radiation dose. QAPs for brachytherapy should be reviewed at least annually to allow the opportunity for updates and periodic follow-up. Self-audit of QAPs is also advisable. (207) The design of a new brachytherapy facility, and the selection and upgrade of existing equipment are all complex and expensive processes. Planning for these processes should include radiological protection. A senior physician, a medical physicist, and a radiological protection officer should be included in this planning. Physicians representing all of the medical specialties who will be using the new facility should be involved in specifying the equipment for the room. (208) The following paragraphs discuss some of the major activities required by a QAP.
5.7.1. Source commissioning – wipe tests
(209) A package containing a shipment of radionuclides must be monitored immediately upon receipt for any physical damage or excessive radiation levels. Wipe tests for any contamination should be carried out on the package surface. Radiation levels should be measured and recorded both at the surface and at 1 -m distance, and compared with the shipping label. Individual large encapsulated sources should be wipe tested for possible leakage or contamination. This should be performed at the time of receipt of new sources and every 6 months for sources with a long half-life that are kept in the permanent inventory. A source is considered to be leaking if approximately 200 Bq of removable contamination is measured. The measurement is usually performed using a sensitive scintillation well counter or a liquid scintillation counter. For permanent implants with seeds which are delivered sterile (usually in sterile cartridges), it is not realistic to test (dosimetry and wipe test) all the seeds to be implanted. In such cases, it is recommended to test the outside of the vial containing the seeds at delivery, and the inside of the cap of the vial when the vial is opened.
5.7.2. Calibration chain
(210) Brachytherapy sources should have their source strength calibrations traceable to a national standards laboratory. In some instances, it may be necessary to establish a second level of traceability by comparison with the same type of calibrated source. Guidelines for the number of sources in a sample that should be measured can be found in Butler et al. (2006).
5.7.3. Constancy check of calibrated dosimeter
(211) The constancy response of the calibrated dosimeter system may be checked by periodic measurement of a long half-life source, such as 137Cs (or other acceptable source) in the case of a well-type chamber. Reproducible positioning is important. Such periodic measurements also provide a good quality assurance check of the entire measuring system.
5.7.4. Regular checks of sources and applicators
(212) Mechanical integrity of a long-lived source must be checked at regular intervals by visual inspection, leak testing, and activity measurement. Source strength and wipe tests should be performed on a periodic frequency, such as on a semi-annual basis. Visual inspection and radiographic evaluation of all applicators should be performed at some established frequency, including checks for structural soundness, and ensuring that all clamps, screws, and retaining devices are functioning properly and that the source insert carriers seat correctly. (213) An important aspect of a QAP is a description of the roles and responsibilities of personnel. There should be enough staff to avoid an excessive number of procedures per specialist, and sufficient nursing and technologist support. Support by network specialists (for new digital systems), maintenance and service personnel, and medical physics specialists is advised. Medical physicists should be active in brachytherapy departments. They should work with radiation oncologists to ensure that proper equipment is purchased and utilised. (214) Procedures should be in place for new staff expected to be involved in interventions guided by radiological imaging to ensure the following: their education and training in radiological protection; arrangements for obtaining and evaluating their previous dosimetric history; pre-employment health surveillance; and arrangements for sharing information with other employers in case the staff members work in more than one place. (215) Procedures should be in place for selection of appropriate radiation detectors and dosimetry equipment. Arrangements for staff radiological protection and health surveillance should be in place, with monitoring of body, eye, and hand exposure as well as workplace monitoring, as set forth in the radiological protection programme. Personal protective devices, such as aprons, thyroid shields, and leaded eyewear, as well as ceiling-suspended shields and table-mounted curtains, should be made available when appropriate. (216) Analysis of staff radiation dose should be included in the QAP. Calibrated dosimeters for staff must be available. Personnel working in fluoroscopy laboratories should wear appropriate dosimeters, and a strict policy for their use should be implemented. Additional electronic dosimeters may also be useful, especially for radiological protection training of students and inexperienced personnel. The QAP should ensure the regular use of personal dosimeters and include a review of all abnormal dose values. Results of personal exposure monitoring and workplace monitoring should be recorded, as well as the necessary corrective measures taken in response to unusual results. Personal dosimetry suppliers should document the accreditation and performance in dose assessment from the supplied personal dosimeters, and information should be recorded and kept safe for the regulatory recommended time. Procedures should include investigation, reporting and recording results and audits of occupational doses, as well as corrective actions in case of incidents or accidents. (217) It is extremely important that there is immediate local reporting and analysis of all accidental exposures of staff, as well as unexpected events. This should be followed by the identification of causes, contributing factors, and extent of conditions, all of which should result in corrective measures. Responses to such situations should be followed by rapid and widespread circulation of the relevant information to avoid similar problems being reproduced in another centre. Institutions should consider participation in the IAEA Safety in Radiation Oncology voluntary reporting and learning system in radiotherapy and radionuclide therapy incidents and near-misses with the purpose of sharing safety-related events and safety analysis for improved safe planning and delivery of treatments. (218) Procedures should address the requirement and instructions for wearing protective devices to the extent possible and compatible with the success of the interventions, including the use of ceiling-suspended shields and protective eyewear. Procedures should also include audits and recording of the wearing of protective eyewear, especially if a dose reduction factor is applied to dosimeter readings to account for the attenuation. (219) Radiological protection training and certification of brachytherapy and interventional staff should be documented and subject to reviews at established periods, or whenever there is a significant change. Induction training in the operation of the quality assurance system should be part of the strategy of the organisation. Administrative procedures including the assignment of responsibility for quality assurance actions and for reviewing and assessing the overall effectiveness of radiological protection measures need to be established, and be part of the quality assurance manual. (220) For fluoroscopy and CT systems, acceptance tests should be made by the company supplying the equipment in the presence of technical personnel from the centre buying the system, or by centre technical personnel. This should include tests to determine the functionality of the radiation safety features of the equipment. Commissioning of the new equipment before its clinical use should be the responsibility of the personnel of the centre. (221) Periodic quality control, including dosimeter calibration, should be planned, taking into account international standards, local regulatory requirements, local recommendations, and the recommendations of the x-ray system manufacturer. These should also include practical results to assist the radiation oncologist in appropriate management of patient doses (e.g. dose rate in different fluoroscopy modes, CT scan protocols). (222) For fluoroscopy and CT systems, periodic evaluation of image quality and procedure protocols should also be included in the QAP. Image quality should be measured with test objects during the acceptance and constancy tests. With digital imaging detectors, it is possible to select a wide range of dose values to obtain the required level of quality in the images. Radiation oncologists, in cooperation with radiographers/technologists, the medical physicist, and the industry engineer, should set the fluoroscopic or CT system protocols to achieve the appropriate balance between image quality and dose needed during brachytherapy planning, treatment, and follow-up. (223) For each imaging modality they use, radiation oncologists should learn the dose required to obtain an adequate level of diagnostic information. (224) As occupational protection is closely related to patient protection, the overall QAP should include quality control of the radiological equipment, acceptance testing and commissioning, full characterisation of the radiological equipment, calibration of the air kerma area product meters, as well as quality control of the personal protective devices.
6. EMERGENCY PLANS AND RESPONSE IN BRACHYTHERAPY
6.1. Need for emergency plans and response readiness
(225) Accidents associated with brachytherapy procedures have been reported, and some of them have had significant impacts on staff safety, especially those involving HDR brachytherapy (ICRP, 2005a). Indeed, more than 500 HDR brachytherapy accidents (including one death) have been reported along the entire chain of procedures from source packing to delivery of dose. Human error has been the prime cause of radiation events. Many accidents could have been prevented if staff had had functional monitoring equipment, paid attention to the results, and responded quickly and appropriately. (226) Such events have different origins (ICRP, 2005a), including:
incorrect measurements leading to erroneous source positioning; handling and transport of the sources; inadequate shielding; inadequate handling of sources in transit (sources remaining in HDR safe, in the patient, or along transfer tubes); treatments given to wrong patients; treatments given to wrong cavities or orifices; incorrectly prescribed or delivered doses, or repeated treatments to the same patient; sources placed outside the transport safe and not secured; damage in transit with failure to survey shipping packages; inadequate shielding of brachytherapy procedure rooms; source exchange accidents for HDR machines; mechanical events associated with cables, kinks in applicators, and stuck sources; dislodged applicators; treatment planning software errors; failure of a retraction system; and failure to adequately calibrate or recalibrate brachytherapy systems or sources. (227) A collaborating team of specifically trained personnel following quality assurance procedures is necessary to prevent accidents (see Section 5.7). Maintenance is an indispensable component of a QAP. External audits of procedures reinforce good and safe practice, and identify potential causes of accidents. QAPs should include peer review of cases. Accidents and incidents should be reported, and the lessons learned should be shared with other users to prevent similar mistakes (ICRP, 2000c, 2005a). (228) Publication 97 has addressed such exposures, events, and accidents for HDR brachytherapy in some detail, and should be consulted for the identification of prevention methodologies (ICRP, 2005a).
6.2. Emergency procedures during and after treatments
(229) Emergency procedures need to be developed for each type of brachytherapy procedure (IAEA, 1998, 2001). Written procedures must be available on site, and important information should be displayed prominently in the treatment room and control room, as needed. These procedures should be practised periodically to ensure emergency preparedness of staff members. It is essential that the items required to perform emergency procedures are available and remain in the brachytherapy suite, procedure room, or operating room prior to and during all cases. (230) The dose potentially received by both the patient and staff may be very high in case of accidents if emergency procedures are not adapted – or worse, do not exist. Often, ‘immediate’ (and not just ‘quick’) detection, and presence of the responsible radiation oncologist, medical physicist, and therapist, is required, especially in HDR or PDR brachytherapy, where high doses can be delivered to patients or staff in seconds. For both patient and staff safety, there may only be a few minutes to recognise an event, react, and resolve issues (Kaulich et al., 1999), and a very high standard of quality assurance is mandatory. It is estimated that in such a case, staff must react to correct the problem within 1–2 min. This minimal opportunity for mitigation, by necessity, requires specific organisation and emergency response training (Kaulich et al., 1999). The swiftest possible rescue of a patient in an emergency and minimisation of staff exposures demands an unequivocal definition of responsibilities. It is advised that (as the organisational structure of the clinic allows) the emergency-responding physician should preferably be the physician who placed the applicator. Clearly, well-practised emergency management can be of life-saving importance for the patient, and can serve to minimise and mitigate potential exposures to staff. (231) One of the most significant accidents in HDR brachytherapy occurred in 1992 in the USA during treatment of a patient with anorectal cancer (USNRC, 1993; ICRP, 2005a). The source (HDR 192Ir) became detached from the drive mechanism at the moment of planned retraction of the source (which therefore remained in the patient). Unfortunately, the physicians in charge had to deal with conflicting signals as the area radiation monitor actually detected the radiation, but the equipment (irradiator) indicated that the source had been shielded. In addition, radiation monitor malfunctions in the months leading up to the accident encouraged misinterpretation and induced staff not to trust the indications. Consequently, the wrong indication (‘source shielded’) of the equipment was accepted, and the patient, clothes, and room were not subsequently checked with another radiation monitor or survey instrument. The HDR source remained within the patient for 4 days, delivering a total tumour volume dose of approximately 16,000 Gy (of note, the prescription was only for 18 Gy). The patient died on day 4. The catheter with the source went unrecognised, although it was removed from the patient along with necrotic tissues. This material was subsequently disposed of in a waste container, without identification of the source at that time. The waste container was picked up by a commercial medical waste disposal company 5 days later. It was then taken to an incinerator where the radiation monitor detected the source, and the facility tracked the source back to the clinic, and the medical physicist was contacted. During the days the source remained in the patient or in the waste container, it irradiated 94 staff to various external dose levels (usually in only a few minutes). (232) External audits of procedures re-enforce good and safe practice, and identify potential causes of errors.
6.3. Emergency surgery or death of a radioactive, LDR-brachytherapy-implanted patient
(233) If surgery is being contemplated for a patient receiving an LDR brachytherapy treatment, the radiation oncologist, medical physicist, and radiation safety officer should be notified immediately. Temporary brachytherapy implants should be removed prior to any surgery needed by a patient. The radiation oncology team should give guidance to the surgical team during operations on patients with permanent implants. (234) If the patient should die while the radioactive sources are in place, the radiation oncologist, medical physicist, and radiation safety officer should be notified. The body should not be moved until these individuals arrive. If an autopsy is to be performed, it should be carried out only after the radioactive sources have been removed. (235) Publication 98 (ICRP, 2005b) lists additional considerations with regard to cremation of a body containing LDR permanent implant sources, noting that cremation can be allowed if 12 months have elapsed since the implantation (some countries set this delay at 3 years). If cremation is to be considered before that time, specific measures must be undertaken.
6.4. Preventing emergencies and accidents
(236) An emergency plan should be prepared and practised with commencement of any brachytherapy procedure or operation. A list of emergency procedures (both medical and radiation) should be displayed prominently within the brachytherapy suite. All necessary emergency equipment items should be present. Training for all personnel should be repeated regularly, especially when new personnel are introduced to the team. (237) The person responsible for performing an emergency procedure should remain in the brachytherapy suite during the entire treatment. In some countries, it is a requirement that both a clinician and a medical physicist remain. (238) General recommendations on preventing emergencies and accidents that could result in overexposure of patients, members of the public, or occupational staff include several considerations, such as a written comprehensive QAP (see Section 5.7) and compliance with quality assurance procedures. While not necessarily required by regulation, a hospital radiation safety committee (and perhaps a quality assurance committee) is needed and should interact with regulatory and health authorities. Maintenance of equipment is extremely important. External audits of procedures should be performed to reinforce good and safe practice, and identify potential causes of error. All significant steps from prescription to final delivery of treatment should be checked and verified by a second competent person. The objective is to ensure that the correct patient receives the correct dose at the correct site. Peer review of each case improves quality. Every incident or accident should be reported as required to the appropriate authority. (239) For HDR units, if a source comes away from the drive cable and lodges in a catheter in the patient, the catheter should not be removed with an open end passing through the patient as the source could leave the catheter and end up in the patient’s tissues. Approaches to such events must be planned by the facility before patients are treated and emergency steps mut be practised. (240) Training at a centre with experience in specific brachytherapy modalities (e.g. LDR or HDR) should commence prior to machine acquisition, and should include the specific techniques to be used. (241) Training should be directed towards ensuring a team approach involving a radiation oncologist, a medical physicist, a technician, and a nurse. Emergency plans should be exercised on a regular basis pursuant to the QAP. (242) Source transportation should adhere to all applicable regulations. On site, shipping containers should be inspected for damage. For HDR brachytherapy sources, removal of old sources, their transfer to the container, and installation of new sources into appropriate shielded safes should be performed with care and by factory-trained and certified operators. (243) So-called ‘false alarms’ and interlock ‘failures’ should be taken seriously and investigated thoroughly, and appropriate action should be taken. Failure to do so may encourage staff to ignore valid alarm signals. (244) Survey of the patient by a portable radiation monitor is essential after each treatment. (245) For HDR machines and sources, particular attention should be paid if the facility or machine is decommissioned to prevent the source from ending up in a junk yard or included in scrap metal. (246) Immediate local reporting and analysis of all accidents is extremely important. This should be followed by the identification of causes, contributing factors, and extent of conditions, all of which should result in corrective measures. Responses to such situations should be followed by rapid and widespread circulation of the relevant information to avoid similar problems being reproduced in another centre. (247) All procedures should undergo risk analysis and assessment of the quality management procedures in place to prevent events (Huq et al., 2016).
