Abstract

ICRP PUBLICATION 148
Approved by the Commission in May 2019
© 2021 ICRP. Published by SAGE.
Keywords: DCRL; RBE; tritium; radiation weighting
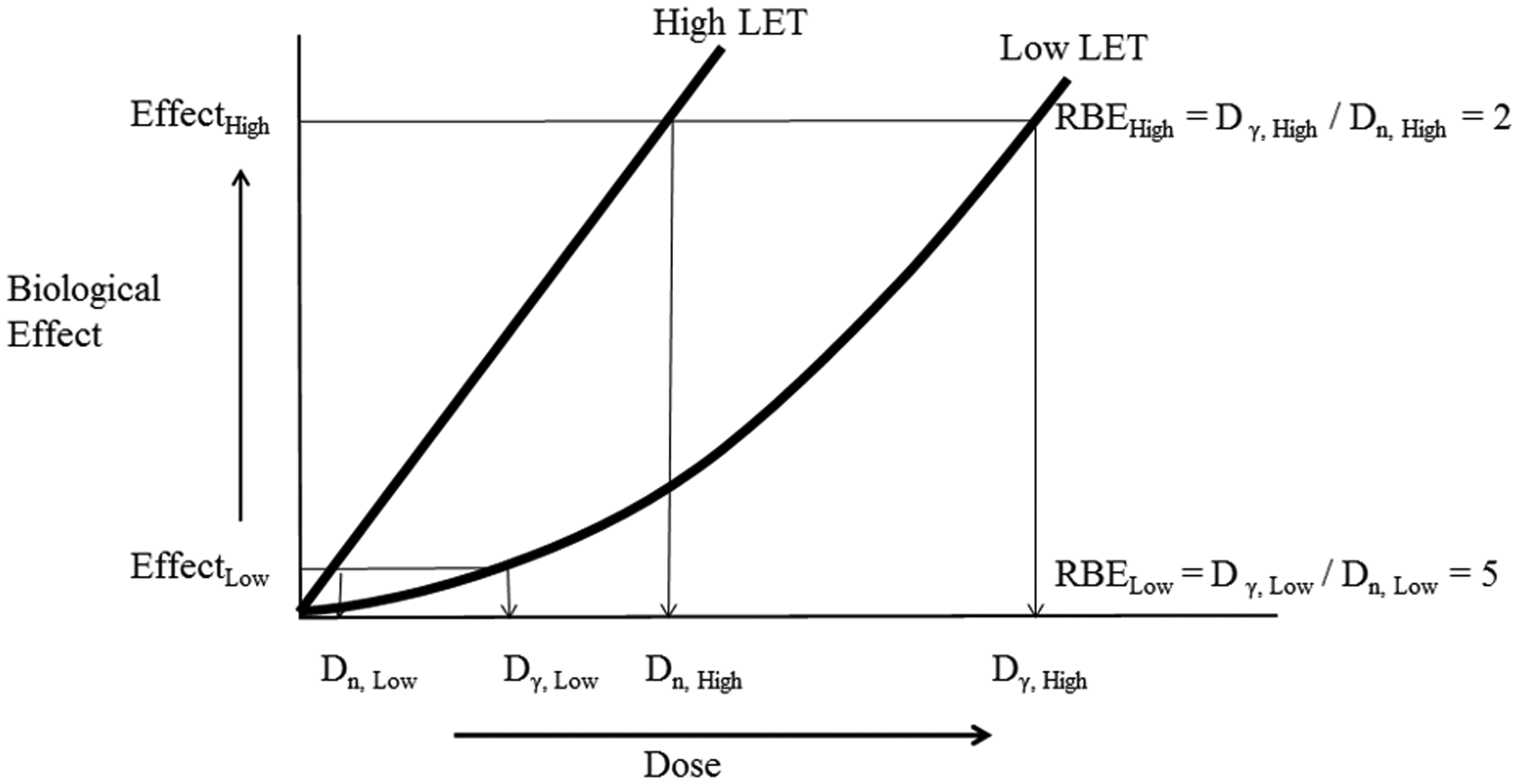
MAIN POINTS
1. INTRODUCTION
1.1. The Commission’s position on environmental protection
(1) The Commission’s environmental protection aims are to prevent or reduce the frequency of deleterious radiation effects on biota to a level where they would have a negligible impact on the maintenance of biological diversity; the conservation of species; or the health and status of natural habitats, communities, and ecosystems (ICRP, 2007). The biological endpoints of most relevance are therefore those that could lead to changes in population size or structure (UNSCEAR, 2008). Due to the immense variety of biota, and their presumed response to radiation, any credible system needs to have some key points of reference which provide some form of auditable trail that links the basic elements of the framework together – or at least could do so if further data were forthcoming, and it is feasible to obtain such data. The Commission therefore developed a small set of 12 Reference Animals and Plants (RAPs), plus their relevant databases, for a few types of organisms that are typical of the major environments (ICRP, 2008) (Table 1.1). (2) After considering relevant radiation effects to these types of biota, a set of derived consideration reference levels (DCRLs) in units of absorbed dose per day, typically reported as mGy day−1, was defined for the different types of RAPs (ICRP, 2008). The DCRL can be considered as a band of dose rate, spanning one order of magnitude, within which there is some chance of deleterious effect from ionising radiation occurring to individuals of that type of RAP. Thus, when considered together with other relevant information, DCRLs can be used as points of reference to inform on the appropriate level of effort that should be expended on environmental protection, dependent on the overall management objectives, the exposure situation, the actual fauna and flora present, and the numbers of individuals of a population thus exposed. The DCRLs considered to be most appropriate, based on the current level of knowledge, are shown in Fig. 1.1. The DCRLs as presented in Fig 1.1 do not include radiation weighting. (3) As the RAPs are, by definition, points of reference, it will also, in some circumstances, be necessary to identify Representative Organisms (ROs) relevant to the situations of exposure under consideration. The ROs may well be the same as, or similar to, the RAPs. Differences should be quantifiable in relation to their basic biology, dosimetry, and radiation effects. The extent to which differences in such factors then need to be taken into account, and the impact on the final decision on the RBE value to use, will depend on the circumstances of the assessment, as outlined in Publication 124 (ICRP, 2014). (4) Publication 136 (ICRP, 2017) provides dose coefficients for RAPs, updating the data provided in Publication 108 (ICRP, 2008). Data are provided for both internal and external exposures, as absorbed dose rates (µGy h−1 Bq−1 kg) averaged over the mass of the organism. For internal exposures, values are given separately for alpha particles, low-energy beta particles and gamma radiation (effective dose <10 keV), and all other beta and gamma radiations (effective dose >10 keV). This separation of dose contributions was done in recognition of differences between radiation types and energies in their effectiveness per absorbed dose in causing deleterious biological effects. (5) In the system of protection as applied to humans (ICRP, 2007), absorbed doses to organs and tissues from different radiation types are multiplied by radiation weighting factors (wR) before dose contributions are summed as equivalent dose in sieverts (Sv). The wR values are chosen largely on the basis of experimental data on the relative biological effectiveness (RBE) of different radiation types determined for biological endpoints related to stochastic effects (cancer, hereditary effects). RBE values are experimentally determined and are the ratio of doses of a test radiation and a low-linear energy transfer (LET) reference radiation that produce the same level of observed effect. (6) This publication provides a review of RBE data relating to exposures to tritium beta particles, as an important example of low-energy, low-LET radiation. Data on RBE for biological effects caused by alpha-particle-emitting radionuclides are also reviewed. On the basis of the analyses of these data, RBE weightings for absorbed dose are proposed for use in relation to RAPs with the dose coefficients provided in Publication 136 (ICRP, 2017). The intention is that these values will be used to calculate radiation weighted absorbed dose rates for comparison with DCRLs and corresponding data for ROs. This publication has also identified the considerable need for collection of RBE data for additional species. Identification and description of the Reference Animals and Plants as first introduced in Publication 108 (ICRP, 2008). Derived consideration reference levels for environmental protection for the Reference Animals and Plants.
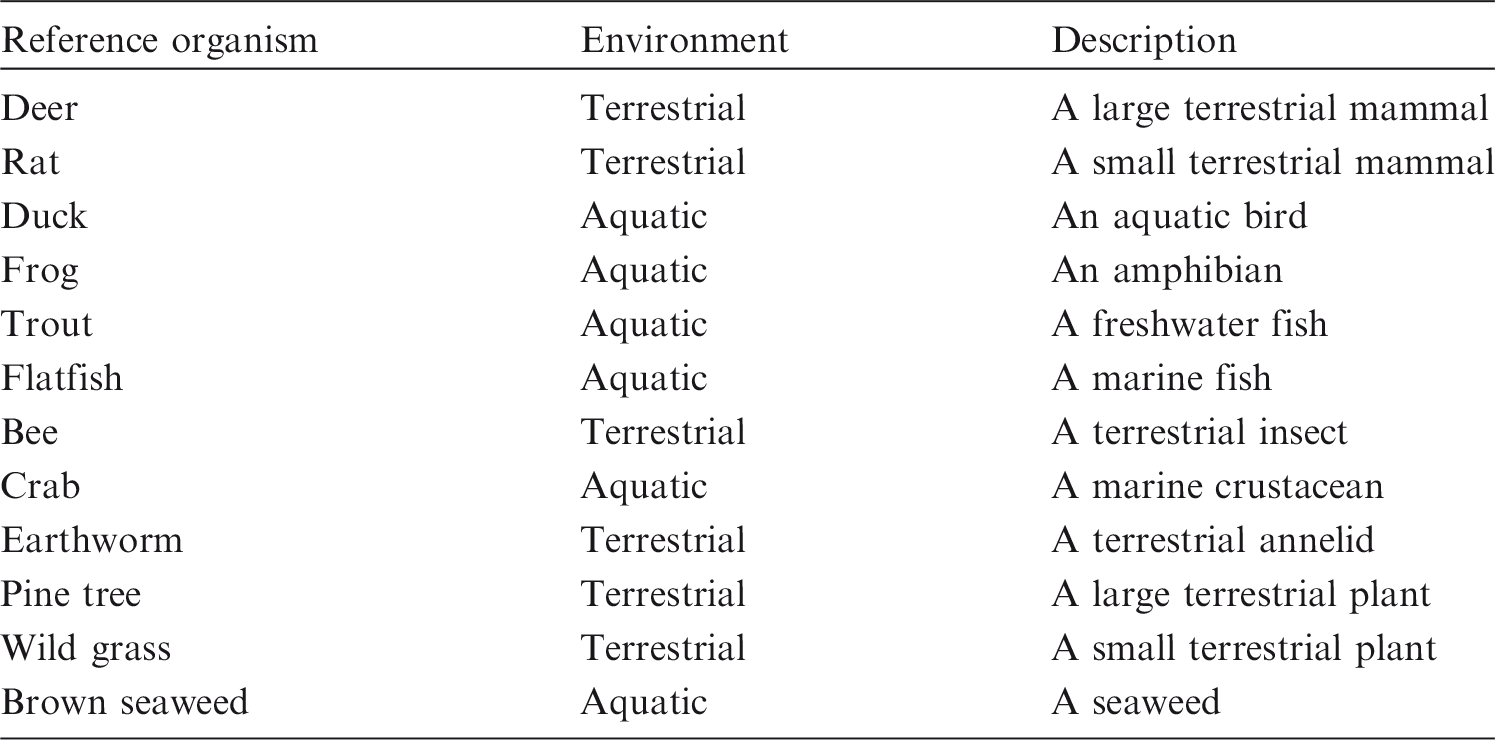
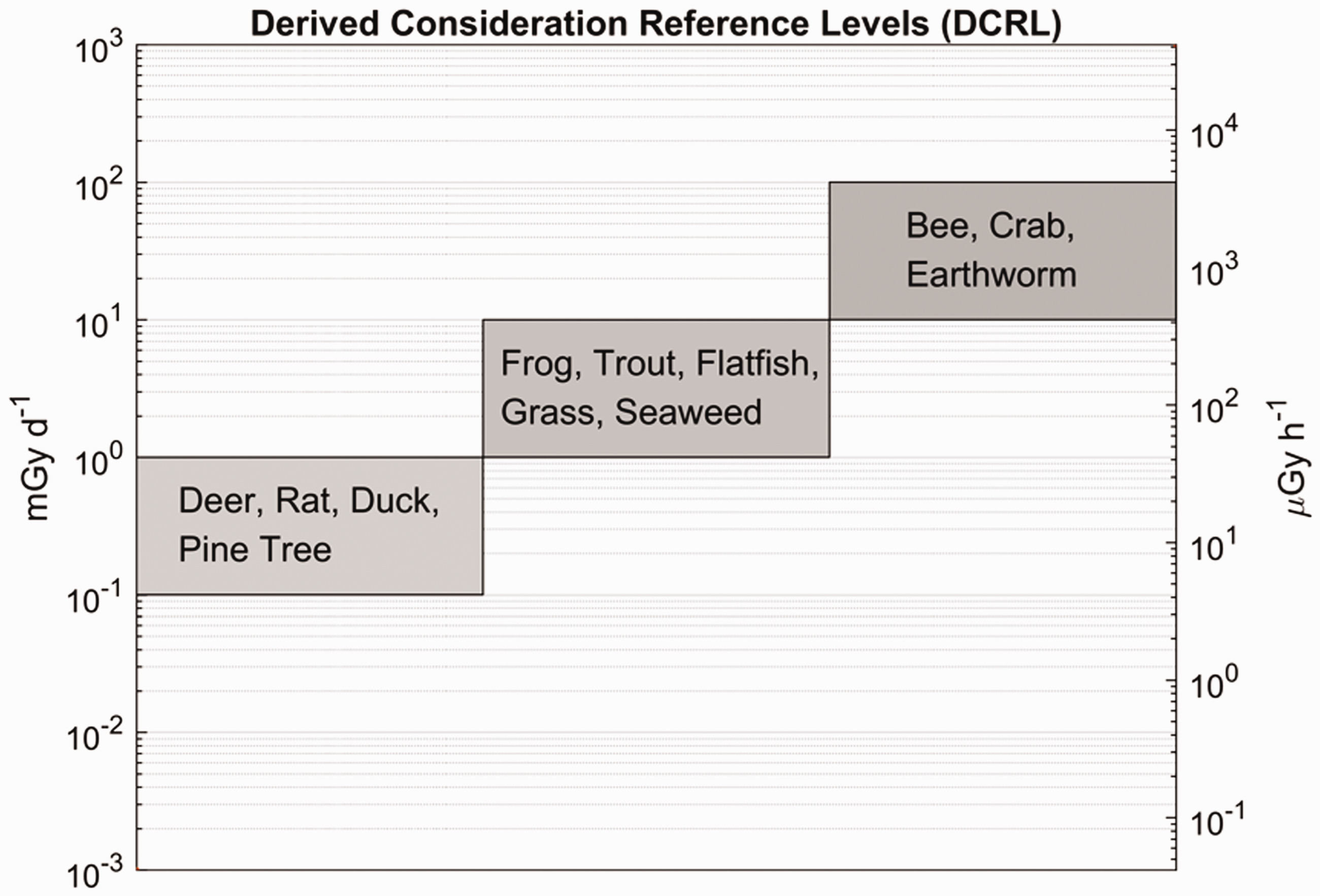
1.2. Relevance of relative biological effectiveness to Reference Animals and Plants
(7) The biological endpoints of most relevance to the protection of non-human biota are those that could lead to changes in population size or structure, including survival, fecundity, and reproductive and developmental impairments. Such effects are generally classed as tissue reactions (formerly deterministic effects) and occur above thresholds, with severity increasing with increasing dose (ICRP, 2007). Based on the current paucity of knowledge and for the purpose of protection of non-human biota, biological endpoints such as DNA damage, chromosomal aberrations, mutation, and tumour induction, which are classed as stochastic effects, are not currently assessed for their impact on population viability. Such effects are taken to occur without thresholds, and with probability (not severity) increasing with increasing dose (ICRP, 2003, 2007). Broadly speaking, effects termed ‘stochastic’ (i.e. cancer and heritable effects) are caused by non-lethal mutational events in cells, while effects termed ‘tissue reactions’ are typically caused by cell killing and other tissue abnormalities. (8) In the reviews presented in this publication, biological data are considered in four categories: mortality, reproductive dysfunction, morbidity, and chromosomal damage. Data on cancer induction are included in the morbidity category. Thus, for completeness and to allow comparisons to be made, less relevant stochastic data on cancer and chromosomal damage are included together with directly relevant data on tissue reactions. (9) ICRP has previously reviewed RBE data on stochastic effects as the basis for setting wR values for the calculation of equivalent and effective dose (Sv) for humans (ICRP, 2003, 2007). Effective dose is used to set limits, constraints, and reference levels, and in the optimisation of protection against cancer and heritable effects. ICRP also sets limits on equivalent dose to tissues to prevent tissue reactions (hand, feet, skin, lens of the eye) (ICRP, 2007), although wR values were intended to apply to stochastic effects. (10) For photons and electrons of all energies, a wR value of 1 is used (ICRP, 2007) despite recognised differences in RBE of up to a factor of 4, with higher values at lower energies. A wR value of 20 is used for alpha particles for all cancer types and hereditable effects, although the available data suggest that RBE will differ for different endpoints (ICRP, 2007), with, for example, low values for alpha-particle-induced leukaemia (RBE = 1–2) and higher values for lung and liver cancer (RBE = 10–20). The intention of wR was to balance scientific accuracy with a simple scheme of practical utility for protection purposes. (11) Similarly, in using RBE data as the basis for the choice of RBE weighting values for the calculation of radiation weighted absorbed dose rates to RAPs, a simple scheme is required to apply across radiations, species, and effects. However, there are important differences in application and, specifically for non-human species, the intention is that estimated dose rates will be compared with the most relevant DCRLs. Since DCRLs are set as order of magnitude dose rate bands of concern, the question is whether consideration of RBE of radiations will result in the DCRL being reached or breached. (12) Dose limits and dose constraints for protection of humans in planned exposure situations are set at levels where no tissue reactions occur and where inferred risks for stochastic effects are very small. Optimisation leads to actual exposures that are normally well below limits and constraints. A high level of protection is also afforded in existing exposure situations, where an appropriate reference level is selected that will inform optimisation efforts and which will be adjusted with time, as appropriate. DCRLs, however, are set at absorbed dose rates where deleterious effects may occur; the selection of an appropriate weighting factor thus has direct relevance for our understanding of likelihood of effects and need for protective measures. The relationship between optimisation (for environmental protection) and DCRLs in planned and existing exposure situations is outlined in Publication 124 (ICRP, 2014). (13) The Commission’s approach for protection of the environment is intended to be a reasonable, yet prudent, approach to understanding when there is a possibility of effects in species or populations. To that end, it may be important to take RBE into account when the radiations of concern warrant. The Commission is not, at this time, suggesting a separate protection quantity, or a weighting factor terminology, as this could be seen as adding unnecessary complexity to the scheme. Likewise, the Commission is not treating protection of the environment in the same way as protection of humans, and is therefore not specifying whether effects are deterministic or stochastic. There is much research that remains to clarify the mechanisms that may be at work in causing effects of interest. When RBE weighting is used, there should be clear documentation of the original measurements, and the value of the weighting applied, in order to ensure transparency and reproducibility of the results. (14) ICRP has also previously reviewed RBE data on tissue reactions, considering alpha particles, neutrons, and heavy ions; the data and analyses provided are referred to in Annexes A and C (ICRP, 1990). Alpha-particle-emitting radionuclides can be important contributors of dose to non-human biota, both in terms of anthropogenic sources and naturally occurring alpha-emitting nuclides. Tritium exposures can also be of concern in particular circumstances, and a range of RBE studies have been undertaken using this radionuclide. (15) The following sections provide summaries of RBE data reviewed in detail in Annexes B (tritium) and C (alpha-emitting radionuclides), and conclude by providing radiation weighting factors based on these data. Annex A provides a detailed discussion of RBE and factors that influence RBE.
2. RELATIVE BIOLOGICAL EFFECTIVENESS OF TRITIUM BETA PARTICLES
2.1. Introduction
(16) A review of the data available on RBE of tritium beta particles is given in Annex B. This section provides a summary of the main data and conclusions. Most studies have used tritiated water (HTO) as the radiation source. Information is scarce for organically bound tritium (OBT). Mammalian species have been studied the most frequently (80% of the data), either in vivo with laboratory bred animals (mainly mice) or in vitro (human cells or established cell lines). There is very limited information on RBE values for tritium beta particles that could be relevant to other RAPs: six RBE values for a fish (medaka) and single RBE values for an insect (Drosophila), a terrestrial plant, a vascular terrestrial plant (Vicia faba), and a polychaete worm (Ophryotrocha diadema). Both tissue reactions and stochastic endpoints have been analysed. (17) Regarding the reference radiation used, gamma radiation (from 60Co or 137Cs) has been used more frequently (75% of the data) than orthovoltage x rays. After critically reviewing the values of RBE when tritium was administered as HTO, in general, RBE values for tritium beta particles are almost two times higher when gamma rays are used as the reference radiation rather than x rays (Straume and Carsten, 1993; Environment Canada and Health Canada, 2003; Kocher et al., 2005; Little and Lambert, 2008; UNSCEAR, 2016). (18) Due to its low beta particle energy (mean 5.7 keV), the track average LET of tritium in water from secondary electrons is 4.70 keV µm−1. This can be compared, for example, with the 0.22 and 0.52 keV µm−1 track average LET in water generated from the 1173- and 1332-keV gamma rays of 60Co (ICRU, 1970). The net result is that the fraction of dose to tissue from tritium’s low-energy (0.1–5 keV) beta particles and/or secondary electrons is approximately 78%. This can be contrasted with the much smaller 33% contribution to dose from low-energy secondary electrons resulting from the gamma rays of 60Co (Nikjoo and Goodhead, 1991). (19) It also has to be noted that in all the studies reviewed here, the reference radiation (either x rays or gamma rays) is an external source, whereas the tritium was administered internally and the absorbed dose estimated. Although the range of tritium beta particles in tissues is low, the uniform distribution of tritium as HTO makes the comparison of averaged absorbed doses valid. (20) Despite the fact that the intakes of tritium by biota in the natural environment will be by inhalation, skin absorption, or ingestion, almost all experimental in-vivo studies have involved intraperitoneal or intravenous injection. However, in general, the different routes of exposure/administration result in similar distribution of tritium in the various organs and tissues. Regarding the irradiation schedule, this has been performed either at exponentially decreasing dose rates (single tritium injection) or at constant dose rates (multiple injections or single injection followed by ingestion of tritium in drinking water). The reference radiation (gamma rays or x rays) was administered at either a constant dose rate or an exponentially decreasing dose rate to mimic the time course of tritium beta particle irradiation. (21) Although the range of tritium beta particle doses and reference radiation doses and dose rates assayed has been wide, most studies have used doses and dose rates well above those found in the environment in planned or existing situations (but many are within the DCRL bands). Nevertheless, RBE values have been determined on the assumption that these data can be used for different biological endpoints: early mortality, reproductive dysfunction, morbidity, and chromosomal damage and mutations. (22) In the summaries provided below for the different endpoints, uncertainties on RBE values obtained from individual studies are not presented – this information is available in Annex B. Similarly, the reference radiation is not identified here but, as noted above, RBE values tend to be greater when gamma rays are used as the reference radiation than when the comparison is with x rays.
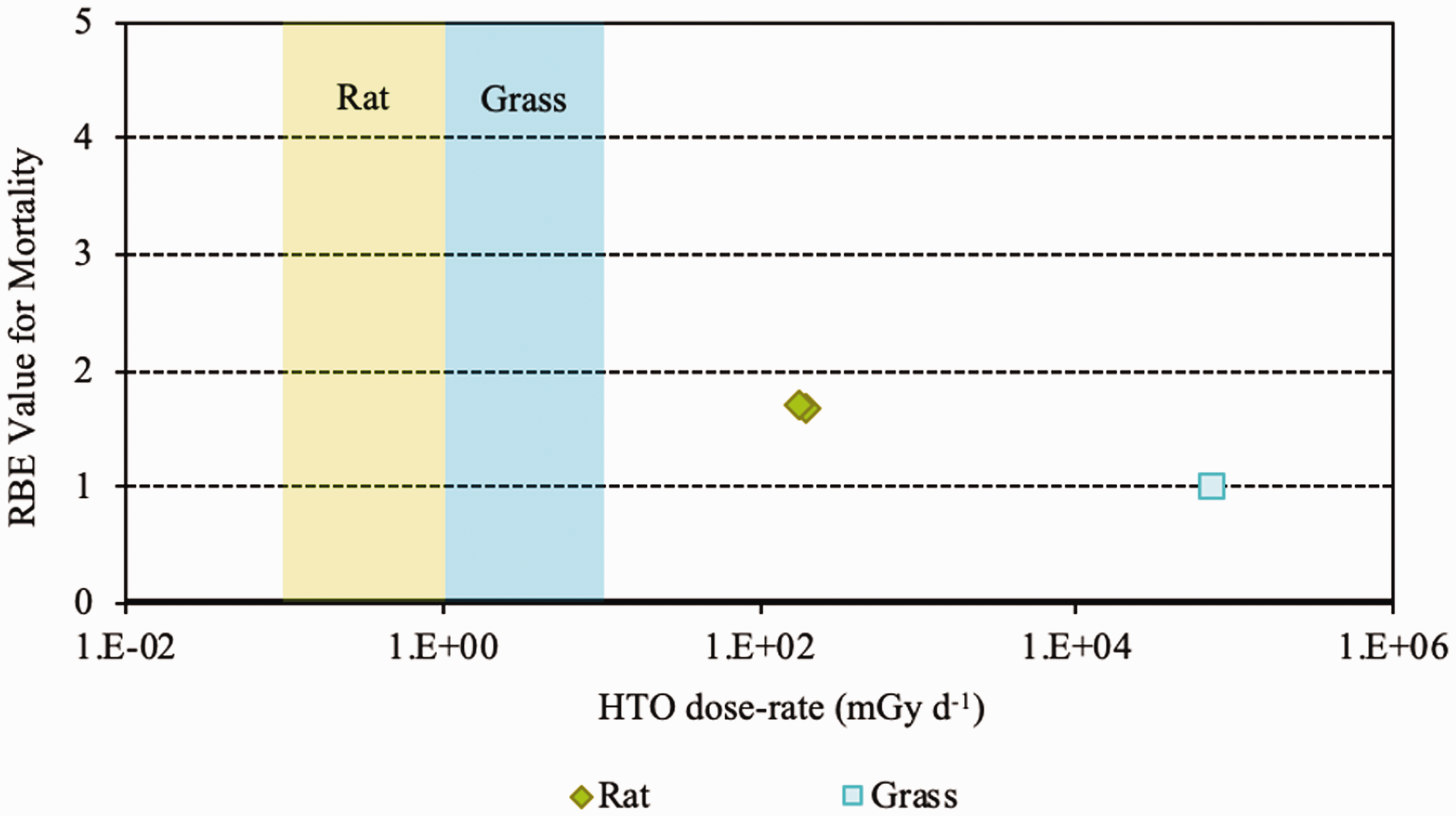
Relative biological effectiveness (RBE) as a function of dose rate from tritium beta particles [tritiated water (HTO)] for early mortality. The derived consideration reference levels (mGy day−1) for environmental protection for each Reference Animal and Plant are shown as coloured bands of green and blue.
Ranges of relative biological effectiveness (RBE) values described in the literature for tritium beta particles (tritium administered as tritiated water).
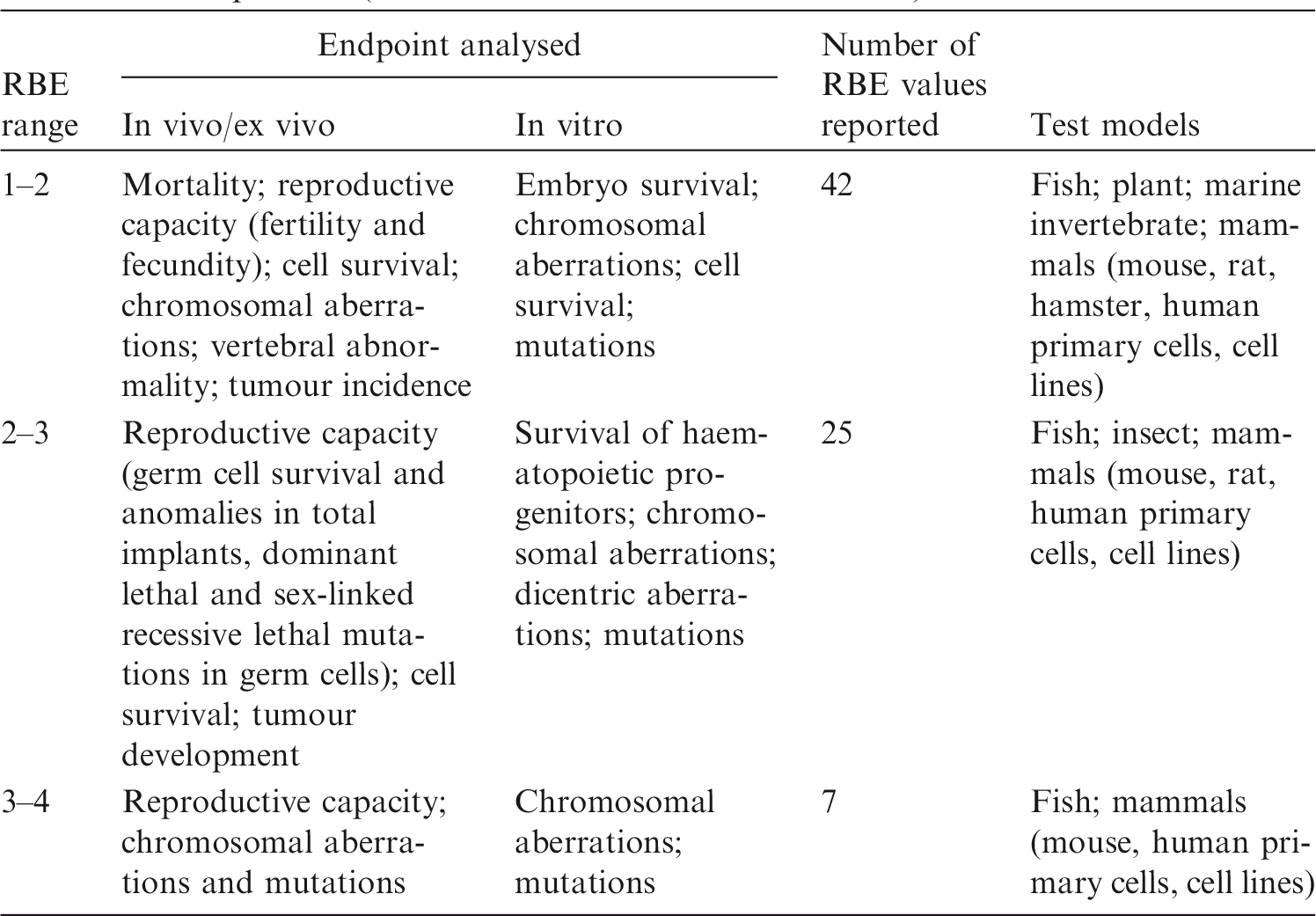
2.2. Relative biological effectiveness values for tritium beta particles for different biological endpoints
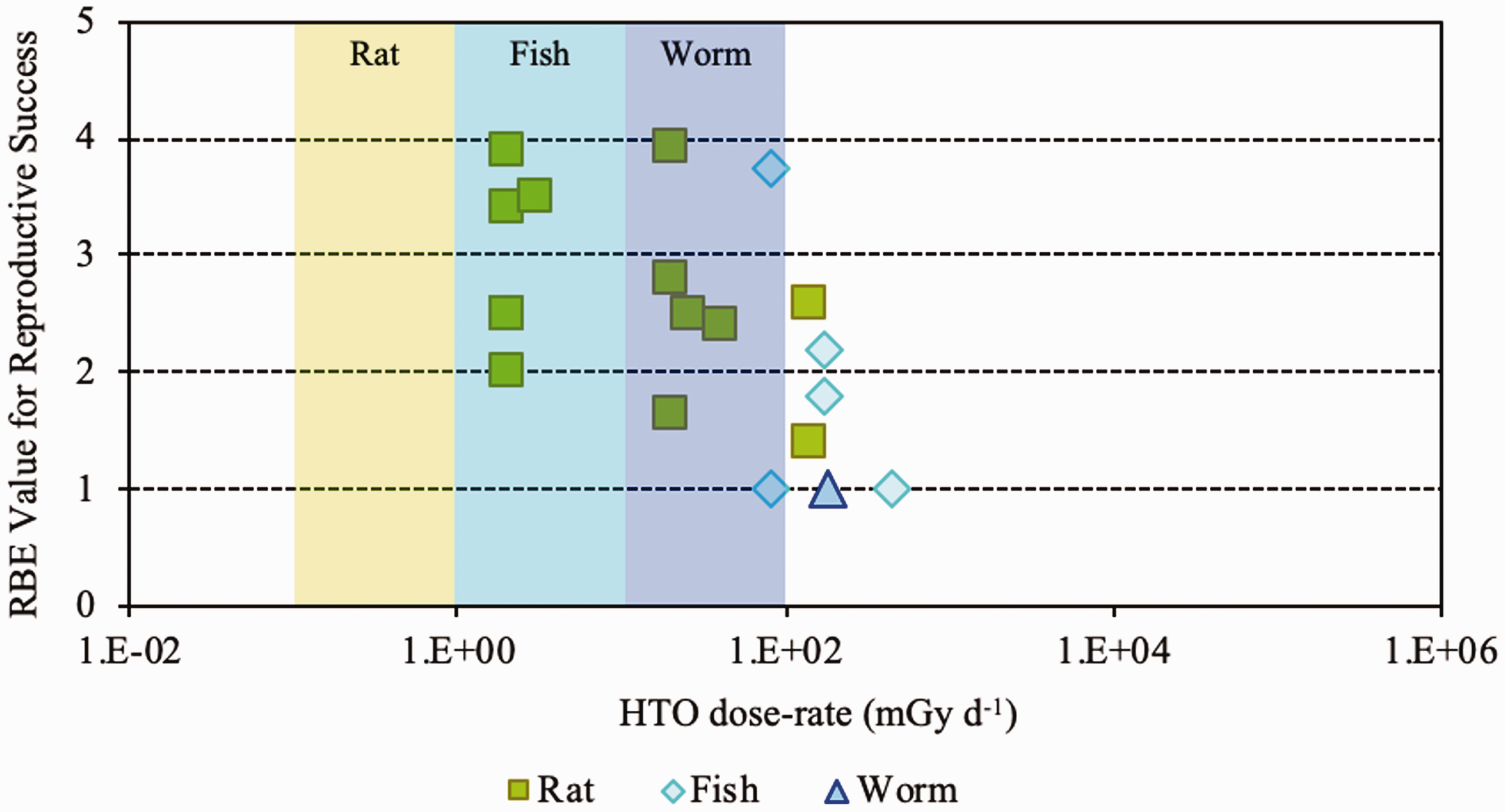
(23) RBE values for tritium beta particles for early mortality were determined to be in the range of 1.0–1.7 (three values available) for a rodent and a terrestrial vascular plant (Fig. 2.1). All relate to HTO. (24) For reproductive dysfunction, RBE values for tritium beta particles were in the range of 1.0–3.9 and relate to a rodent, a fish, and a polychaete worm (Fig 2.2). All were based on HTO. (25) RBE values available for tritium beta particles relating to morbidity showed values in the range of 1.0–2.5 (Fig. 2.3) and relate to rodents alone (rats, mice, murine leukaemia cells, hamster cells) using HTO. (26) For induction of chromosomal damage and mutations, RBE values for tritium beta particles were in the range of 1.0–3.8 (Fig. 2.4) and relate to an insect and mammals alone. All relate to HTO. It should be noted that there are substantial uncertainties in extrapolating from subcellular data, such as chromosomal damage and mutation rates, to observed effects in whole organisms. However, the data are presented for completeness. (27) Regarding RBE values for tritium beta particles following tritium administration as DNA precursors (e.g. tritiated thymidine), in relation to any of the biological endpoints of interest, it was not possible to conclude anything from the four studies available because of the experimental conditions used, the biological endpoints chosen, and the dosimetric uncertainties. Relative biological effectiveness (RBE) as a function of dose rate from tritium beta particles [tritiated water (HTO)] for reproductive dysfunction. The derived consideration reference levels (mGy day−1) for environmental protection for each Reference Animal and Plant are shown as coloured bands of green, blue, and darker blue.
2.3. Conclusions
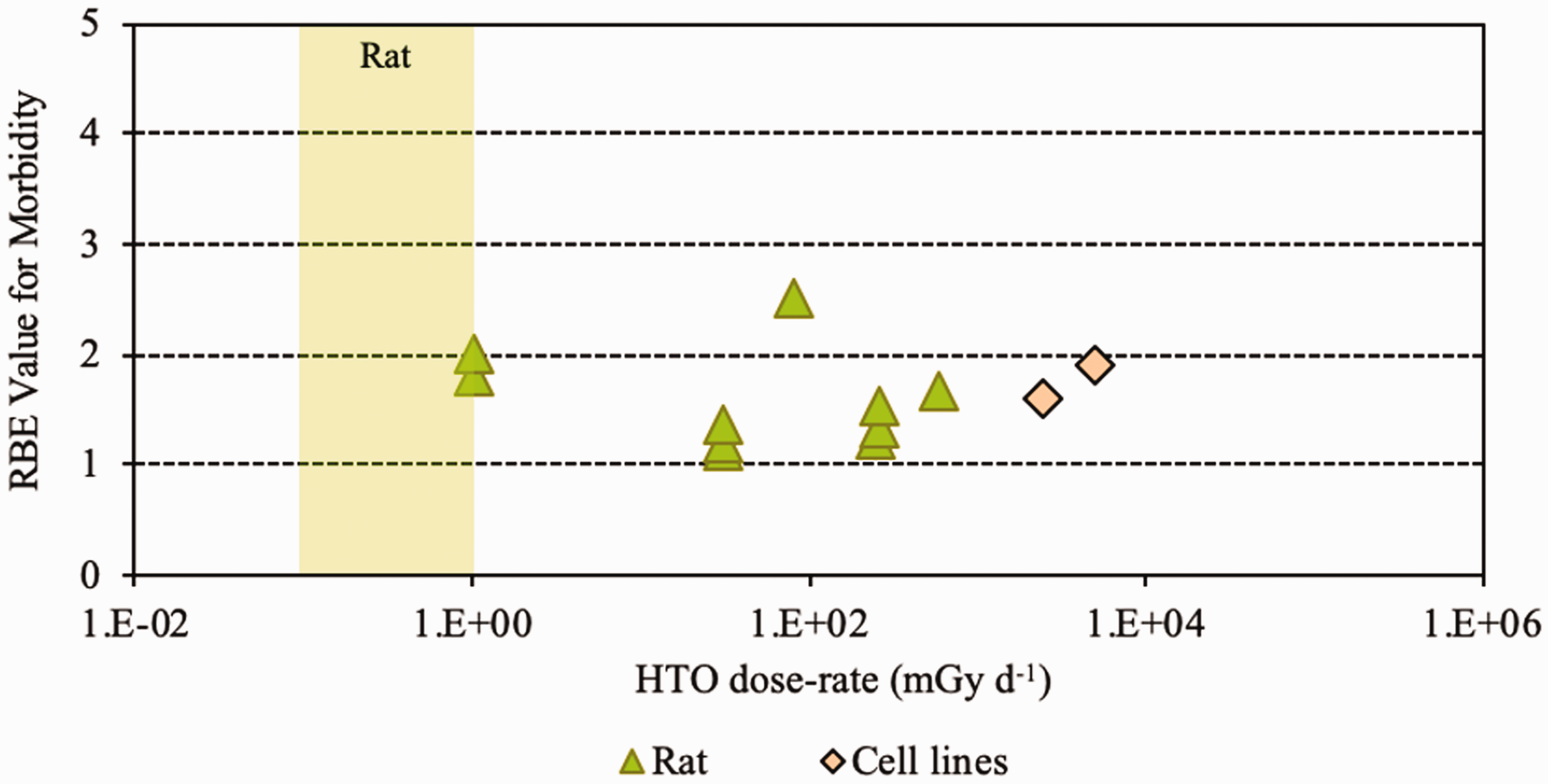
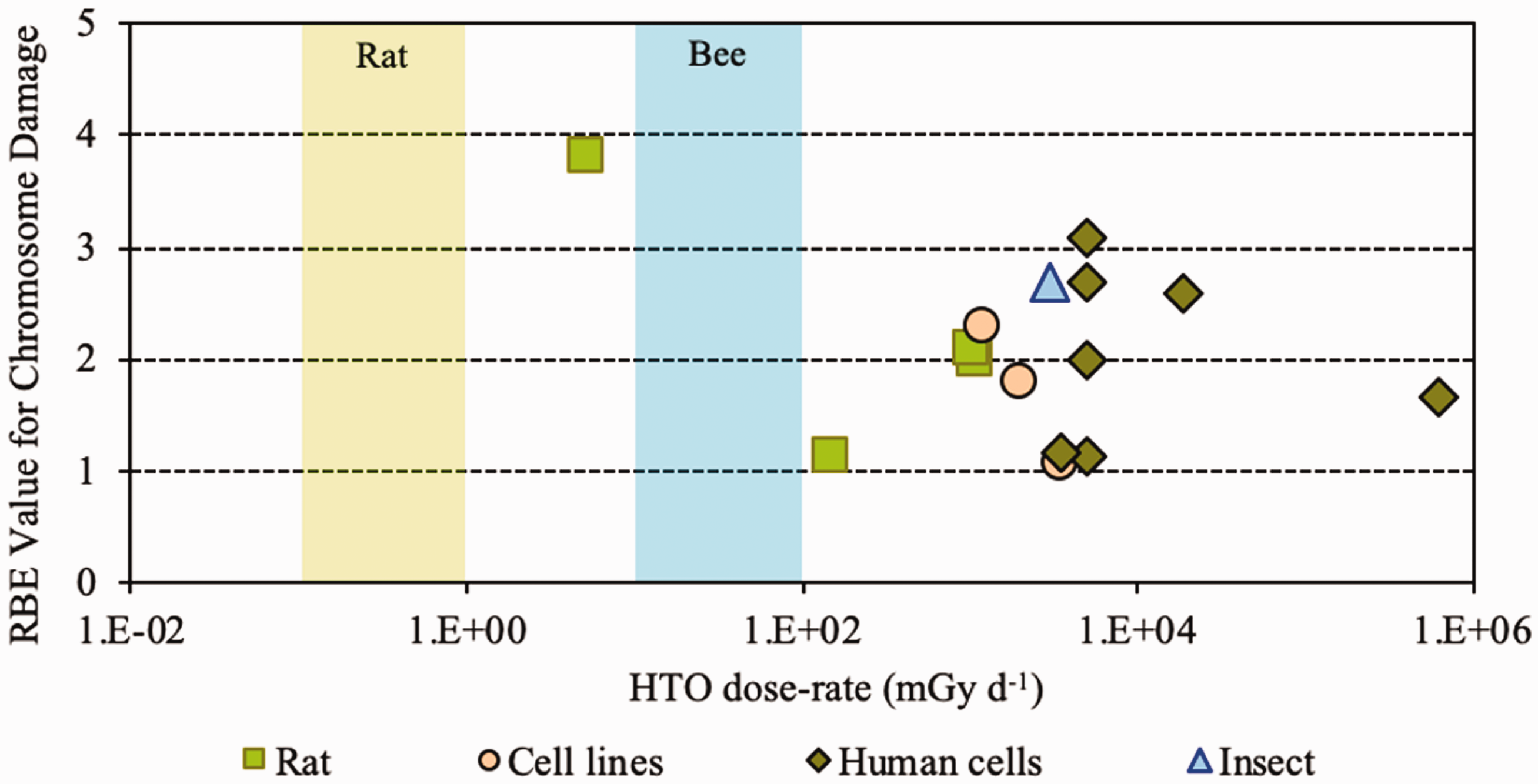
(28) Overall, the non-human biota data on RBE of tritium beta particles, summarised in Table 2.1, cover a range of endpoints and experimental conditions but relate primarily to small mammals. (29) All values were obtained at dose rates that were in or above the relevant DCRL bands. RBE has been shown to increase with decreasing dose rate. (30) In comparison with other radionuclides, the majority (approximately 78%) of dose from tritium is due to the low-energy beta and/or secondary electrons (0.1–5 keV) which generate greater density of ionisations than higher-energy electrons. (31) The spread of data for fish are from 1 to nearly 4, with a value for an aquatic invertebrate of approximately 1. The same range was seen for rats, showing consistency across species. For reduced reproductive success, RBE values were in the range of 1–3.9. (32) Overall, as concluded by UNSCEAR (2016), values centred around 1.5–2 compared with x rays and 2–2.5 compared with gamma rays (see Annex B). Relative biological effectiveness (RBE) as a function of dose rate from tritium beta particles [tritiated water (HTO)] for morbidity. The derived consideration reference levels (mGy day−1) for environmental protection for the Reference Animal is shown as a coloured band of green. Relative biological effectiveness (RBE) as a function of dose rate from tritium beta particles [tritiated water (HTO)] for chromosomal damage and mutation. The derived consideration reference levels (mGy day−1) for environmental protection for each Reference Animal and Plant are shown as coloured bands of green and blue.
3. RELATIVE BIOLOGICAL EFFECTIVENESS OF ALPHA PARTICLES
3.1. Introduction
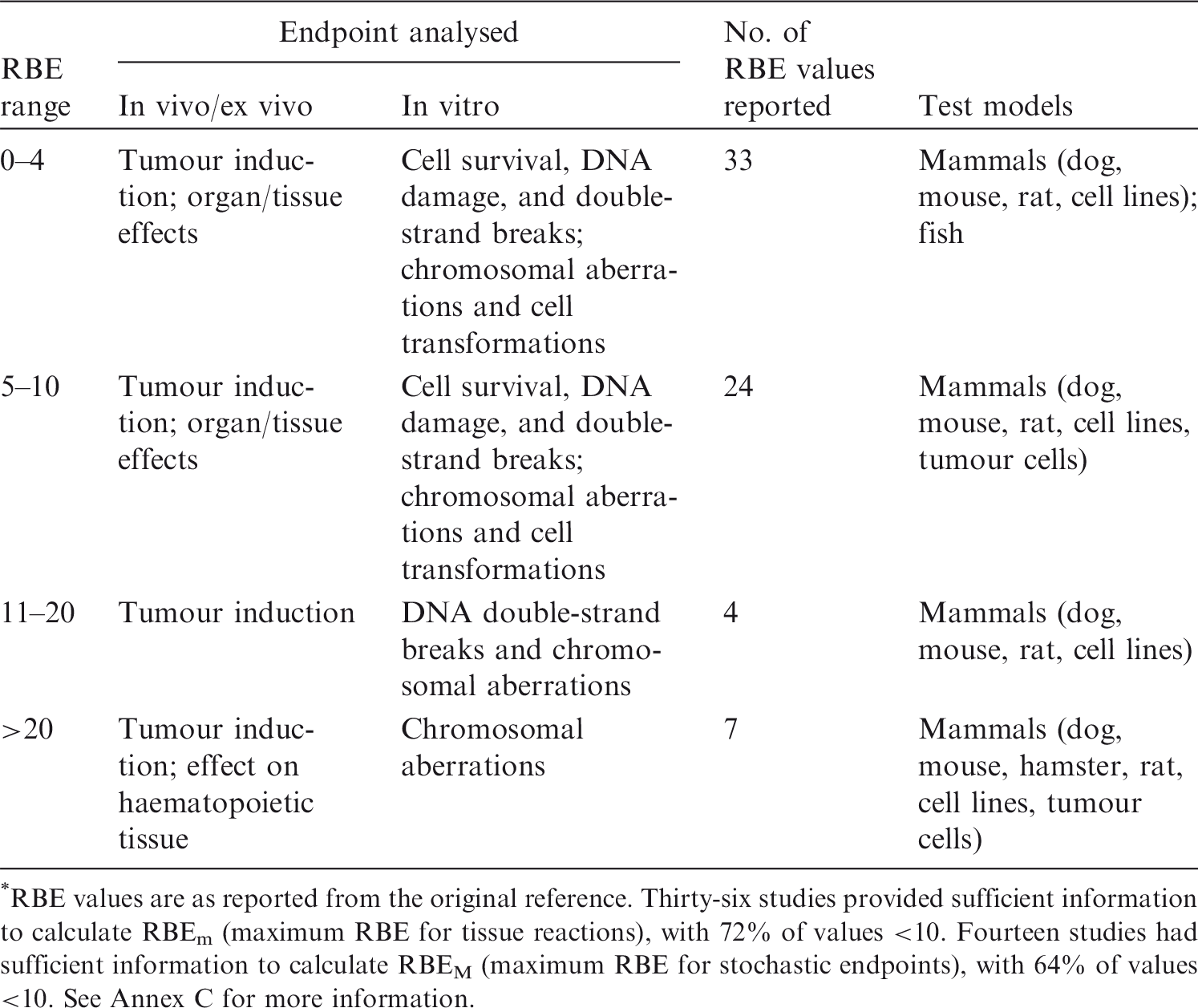
(33) A review of the data available on RBE of alpha particles and corresponding citations are given in Annex C; a summary of the main data is included here. Approximately 90 articles were found that discussed studies relevant to the RBE of alpha particles for non-human biota. Of these, 58 were reviewed in detail; the remainder were considered to have inadequate precision with regard to dosimetry, or had other limitations. Table 3.1 provides an overall summary of RBE values for internally deposited alpha particles. (34) Most of the reviewed papers either reported RBE values directly, or provided sufficient data from exposure–effect models or survival curves from which the RBE for alpha particles values could be calculated. Maximum values for RBEm or RBEM, where m and M denote values for tissue reactions and stochastic endpoints, respectively, were calculated wherever possible from the slopes of survival curves (see Annex A for discussion and Glossary for definition). These data are included in Annex C. (35) In addition to RBE values obtained from studies of internally deposited alpha emitters per se, some data on RBE derived from experimental studies involving external exposure to fission neutrons (which have similar LET to that for alpha particles for common internal emitters) have also been considered in this publication. Summary of reported relative biological effectiveness (RBE) values* for alpha particles. RBE values are as reported from the original reference. Thirty-six studies provided sufficient information to calculate RBEm (maximum RBE for tissue reactions), with 72% of values <10. Fourteen studies had sufficient information to calculate RBEM (maximum RBE for stochastic endpoints), with 64% of values <10. See Annex C for more information.
3.2. Relative biological effectiveness values for alpha particles for different biological endpoints
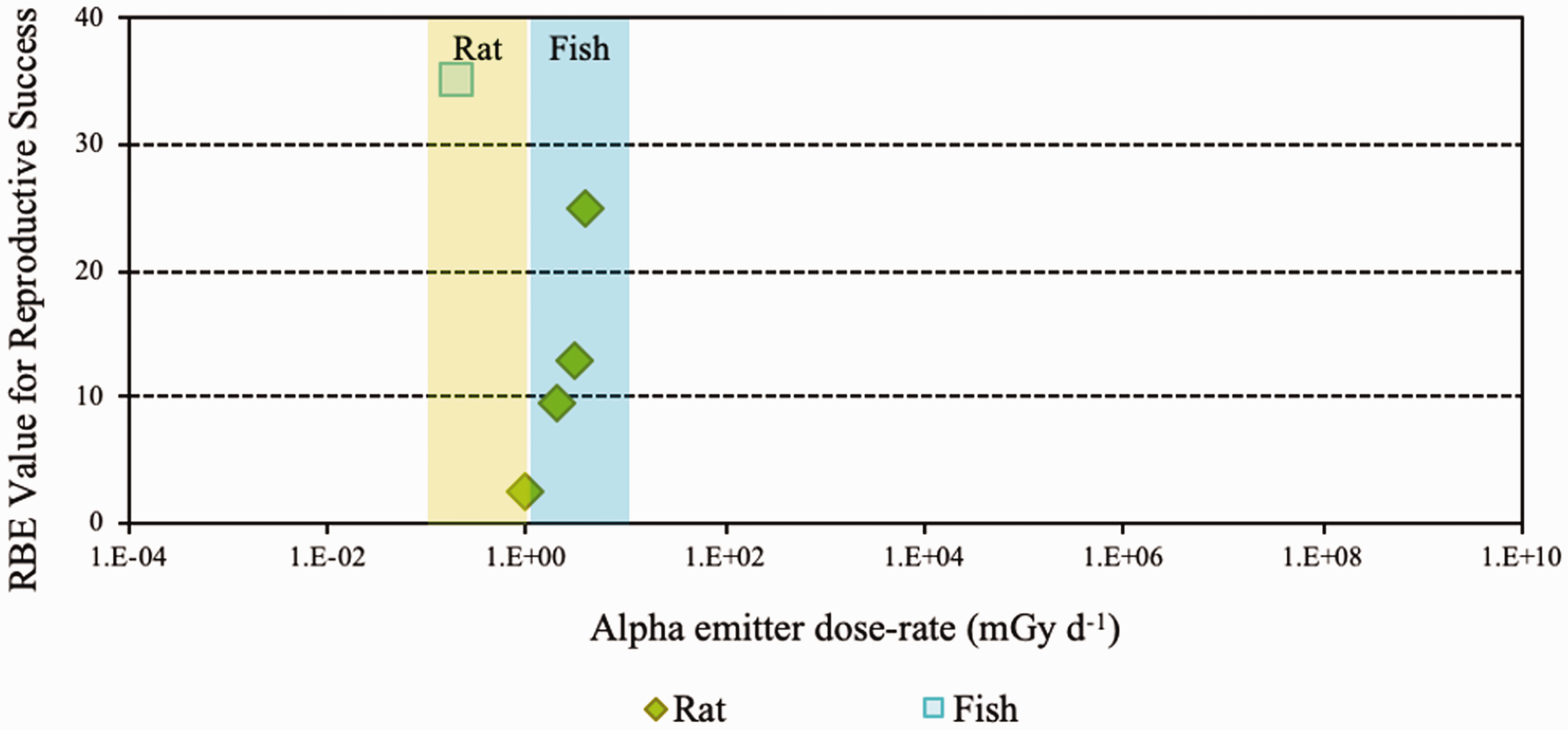
(36) In mammals, mortality is a result of extensive irradiation that causes severe cell depletion, leading to dysfunction of major organs. Death of the organism occurs due to injury of specific organs. Few RBE studies have been conducted for this endpoint. (37) Of the 58 papers reviewed, 14 examined the effects of alpha emitters on reproductive dysfunction. The reference radiations used in these studies were x rays, ranging from 60 to 120 kVp, and high-energy gamma rays from sources such as 60Co. It is important to note that RBE values obtained using x rays as the reference radiation may be up to a factor of 2 lower than those using 60Co. The alpha emitters commonly used were 238Pu, 239Pu, and 210Po. A wide range of RBE values were reported or calculated; however, most were in the range of 1–5, with very few papers reporting RBE values for alpha particles >5. Most RBE values were obtained from studies using rodents or rodent cells exposed to high doses at high dose rates. Reported RBE values vs dose rate are shown in Fig. 3.1 for studies related to reproductive dysfunction. (38) Only six publications reported RBE values for alpha particles in relation to morbidity. The reference radiations used were 60Co gamma rays and 220-kVp x rays. The alpha emitters commonly used were isotopes of plutonium and radium. A range of RBE and RBE maximum values were reported, all <11, with the majority <5 (Fig. 3.2). (39) Twenty-six articles analysed chromosomal damage and mutations caused by alpha emitters. It should be noted, however, that these effects are stochastic in nature and, at present, it is uncertain how to extrapolate such effects to relevant endpoints. The reference radiation used in these studies was 60Co gamma rays or x rays ranging from 80 to 300 kVp. Alpha emitters commonly used to irradiate cell lines, tissues, or cell cultures were 238Pu, 239Pu, 241Am, and 226Ra. Most RBE values were obtained using rodents or rodent cells exposed to high doses at high dose rates, giving values in the range of 1–10, with very few papers describing RBE values for alpha particles >20 (Fig. 3.3). As discussed in Annex A, RBE is a function of dose, with values decreasing as dose increases, and this factor must be considered in any interpretation of the data. (40) In the graphical and tabulated summaries provided below for the different endpoints, uncertainties on RBE values obtained from individual studies are not presented; this information is available in Annex C. Similarly, the reference radiation is not identified here but, as noted above, RBE values tend to be greater when gamma rays are used as the reference radiation than when the comparison is with x rays. Relative biological effectiveness (RBE) as a function of dose rate from alpha emitters for reproductive dysfunction. The derived consideration reference levels (mGy day−1) for environmental protection for each Reference Animal and Plant are shown as coloured bands of green and blue. Relative biological effectiveness (RBE) as a function of dose rate from alpha emitters for morbidity. The derived consideration reference level (mGy day−1) for environmental protection for the Reference Animal and Plant is shown as a coloured band of green. Cell lines include rodent fibroblasts and tracheal epithelium, and human skin fibroblasts. Relative biological effectiveness (RBE) as a function of dose rate from alpha emitters for chromosomal damage and mutations. The derived consideration reference level (mGy day−1) for environmental protection for the Reference Animal and Plant is shown as a coloured band of green. Cell lines include rodent fibroblasts and human lymphocytes.
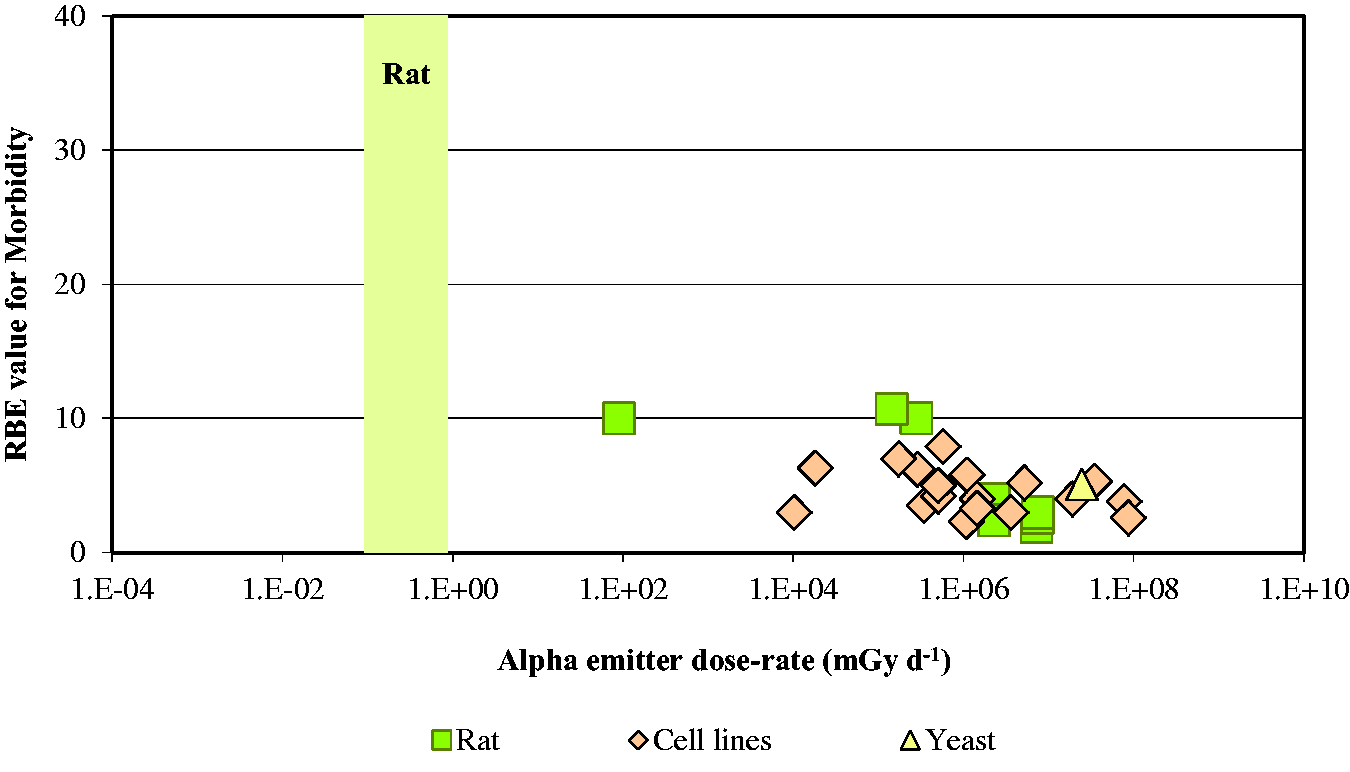
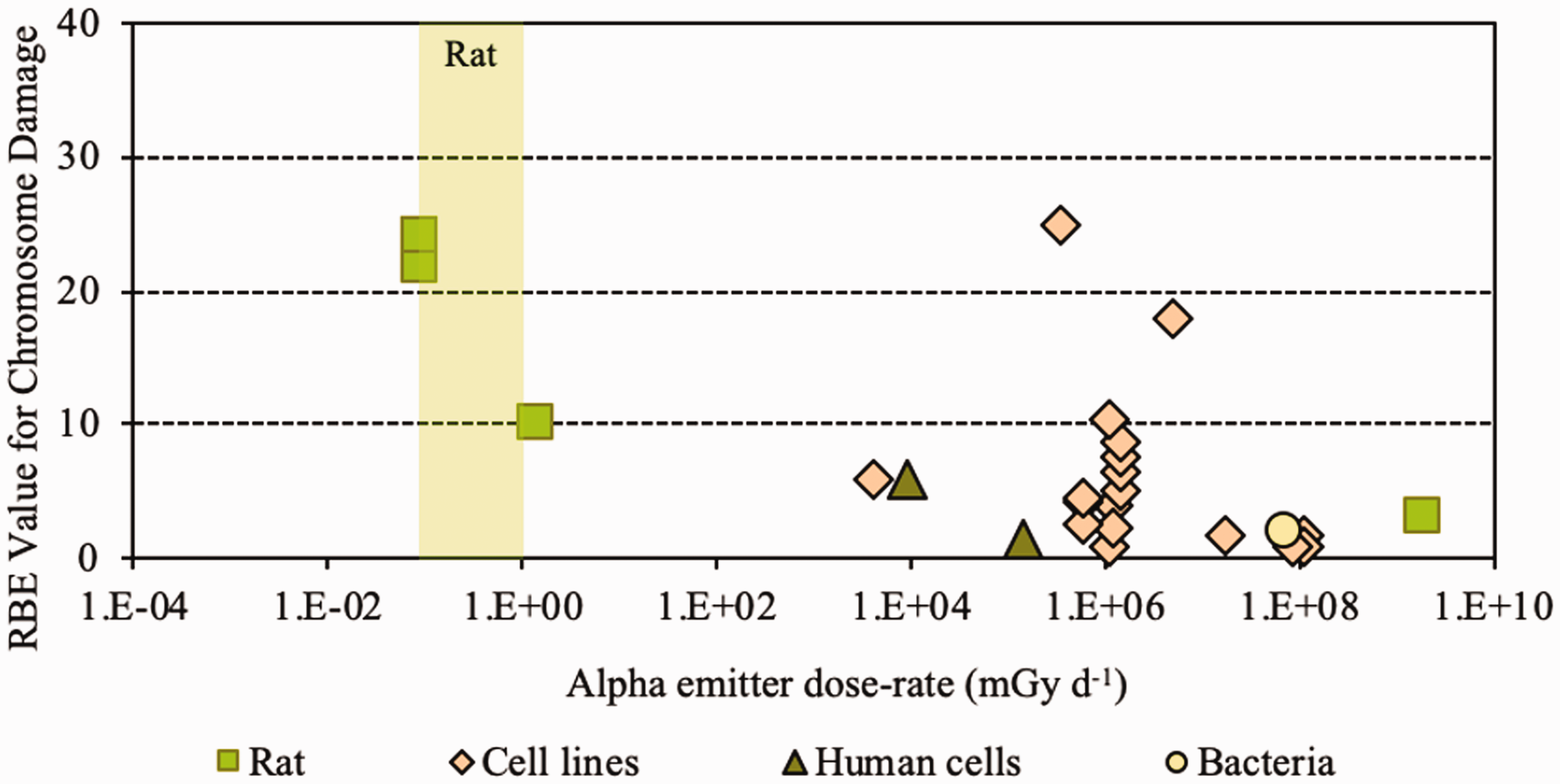
3.3. Conclusions
(41) As for tritium, it is evident that the data available for alpha emitters are primarily relevant to vertebrates – essentially to small mammals – and with respect to reproductive dysfunction and morbidity. Overall, the non-human biota data on RBE of alpha particles are limited. The single value for a fish (Fig. 3.1) is of interest, although the authors (Knowles, 2001) had reservations about the results and commented that a value <35 represented an upper limit, and that the actual value was more likely to be in the range of 7–<20. Additionally, alpha dosimetry is complex due to its heterogeneous distribution and the short range of energy deposition. Such factors should be considered in detail when assessing impacts from alpha emitters. (42) RBE values summarised here were all obtained at dose rates that were in or above the relevant DCRL bands, although the data are extremely limited in the species included. The values obtained are in a wide range but centre around values of the order of 10.
4. OVERALL CONCLUSIONS AND RECOMMENDATIONS
(43) This publication examined RBE data for tritium beta particles and alpha particles for biological effects in non-human biota to consider whether radiation weighting factors for biota should be used to modify estimates of absorbed dose rate for comparison with DCRLs. RBE values vary according to factors including the endpoint being studied, the dose and dose rates employed, and the reference radiation. However, in general, there appears to be some consistency in numerical values obtained across species and for various cell lines, as might be expected in relation to the common physical basis for differences in the effectiveness per Gy of the different radiation types. This similarity across organisms suggests that, in the absence of better information, RBE weighting can reasonably be applied to all RAPs and to ROs identified under particular circumstances of exposure (see Section 1). This publication has also identified the considerable need for collection of RBE data for additional species. (44) The available RBE data for tritium beta particles and alpha particles were obtained at dose rates at or above the corresponding DCRLs. As discussed in detail in Annex A, RBE values tend to increase to a maximum as doses and dose rates decrease. For the tissue reactions of most concern in terms of survival, these considerations are complicated by the existence of thresholds below which no effects are observed. However, it appears that extrapolated RBE values for tissue reactions are largely independent of dose below a level that may be comparable to a threshold (see Annex A). For the purposes of this publication, therefore, it is considered reasonable to base proposals for radiation weighting factors for biota on the observed RBE data without further adjustment to obtain RBEm values for tissue reactions and RBEM values for stochastic effects, although RBEm and RBEM values were calculated for some studies with alpha-particle-emitting radionuclides (Annex C). (45) Biological endpoints were considered in four categories: mortality, reproductive dysfunction, morbidity, and chromosomal damage/mutations. While the first two categories can clearly be considered as tissue reactions and relevant to survival, some of the morbidity studies and all chromosomal damage/mutation studies relate to stochastic effects, and their relevance in the context of this publication is more questionable. In general, RBE values for tissue reactions tend to be lower than values for stochastic effects. However, particularly in the case of tritium, but also for alpha particles, there was no clear difference in the ranges of RBE values observed for the various endpoints. In proposing radiation weighting for general application, therefore, it is reasonable to consider the entirety of the available data. (46) Consistent with the approach taken in specifying weighting factors used in the protection of humans, it is recommended that an RBE weighting factor of 1 should be used for all low-LET radiations, and a value of 10 should be used for alpha particles in assessments of exposures and comparison of estimated doses with the relevant DCRL. If internal exposures to tritium beta particles or other low-energy, low-LET radiations are within or close to the DCRL, additional review and possible modification of weighting of the absorbed doses might be warranted. (47) These recommendations are consistent with those of UNSCEAR (2008) for non-human biota. In Annex E of its report, the Committee recommended a nominal factor of 10 for internally deposited alpha radiation, and a nominal factor of 1 for RBE of beta and gamma radiation. The recommendations were meant to be applicable on a generic basis across all organisms and endpoints. (48) These RBE weighting factors can be used with the dose coefficients provided in Publication 136 (ICRP, 2017), which provides separate values of absorbed dose rate for internally deposited radionuclides for high-LET, and low- and high-energy, low-LET radiations.
REFERENCES
Environment Canada, Health Canada, 2003. Priority Substances List Assessment Report. Releases of Radionuclides from Nuclear Facilities (Impact on Non-Human Biota). PSL2. Environment Canada and Health Canada, Ottawa.
ICRP, 1990. RBE for deterministic effects. ICRP Publication 58. Ann. ICRP 20(4).
ICRP, 2003. Relative biological effectiveness, radiation weighting and quality factor. ICRP Publication 92. Ann. ICRP 33(4).
ICRP, 2007. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Ann. ICRP 37(2–4).
ICRP, 2008. Environmental protection – the concept and use of Reference Animals and Plants. ICRP Publication 108. Ann. ICRP 38(4–6).
ICRP, 2014. Protection of the environment under different exposure situations. ICRP Publication 124. Ann. ICRP 43(1).
ICRP, 2017. Dose coefficients for non-human biota environmentally exposed to radiation. ICRP Publication 136. Ann. ICRP 46(2).
ICRU, 1970. Linear energy transfer. ICRU Report 16. International Commission on Radiation Units and Measurements, Bethesda, MA.
Knowles, J.F., 2001. An Investigation into the Effects of Chronic Radiation on Fish. R&D Technical Report P3-053/TR. Centre for Environment, Fisheries & Aquaculture Science, Lowestoft, pp. 1–42.
Kocher, D.C., Apostoaei, A.I., Hoffman, F.O., 2005. Radiation effectiveness factors for use in calculating probability of causation of radiogenic cancers. Health Phys. 89, 3–32.
Little, M.P., Lambert, B.E., 2008. Systematic review of experimental studies of relative biological effectiveness of tritium. Radiat. Environ. Biophys. 47, 71–73.
Nikjoo, H., Goodhead, D.T., 1991. Track structure analysis illustrating the prominent role of low energy electrons in radiobiological effects of low-LET radiations. Phys. Med. Biol. 36, 229–238.
Straume, T., Carsten, A.L., 1993. Tritium radiobiology and relative biological effectiveness. Health Phys. 65, 657–672.
UNSCEAR, 2008. Sources and Effects of Ionizing Radiation. UNSCEAR 2008 Report to the General Assembly, with Scientific Annexes. Annex E. Effects of Ionising Radiation on Non-Human Biota. United Nations Scientific Committee on the Effects of Atomic Radiation, New York.
UNSCEAR, 2016. Sources, Effects and Risks of Ionizing Radiation. UNSCEAR 2016 Report to the General Assembly with Scientific Annexes. Annex C. Biological Effects of Selected Internal Emitters – Tritium. United Nations Scientific Committee on the Effects of Atomic Radiation, New York.
ANNEX A. RELATIVE BIOLOGICAL EFFECTIVENESS IN THE CONTEXT OF PROTECTION OF THE ENVIRONMENT
(A1) Studies of dose–response relationships for different types of radiation in inducing a wide variety of effects in many biological systems, ranging from cells in culture to whole organisms, have shown that knowledge of the absorbed dose is not sufficient to characterise the biological response from a given dose. It is generally observed that radiation quality, as commonly represented by LET, is important in determining the biological response from a given absorbed dose. In particular, high-LET radiations (e.g. alpha particles and neutrons) are more effective per unit absorbed dose than low-LET radiations (e.g. orthovoltage x rays and higher-energy photons) in inducing biological effects. To account for this, the absorbed dose (in Gy) is often multiplied by a modifying factor in order to account for RBE. The term ‘RBE’ applies to observations from experimental studies, and is specific to the endpoint and system studied, and environmental and exposure conditions (e.g. reference radiation, dose rate, and dose) amongst other factors. This section presents a definition of RBE and brief discussions of factors that influence RBE; extrapolation of RBE values obtained in studies at high doses to low doses of concern to radiological protection, especially extrapolation of RBE values for tissue reactions; and extrapolation of RBE values for cells to higher levels of biological organisation including whole organisms.
A.1. Relative biological effectiveness
(A2) For a specific radiation (A) of interest, RBE is a unitless quantity defined as the ratio of the dose of a reference radiation required to produce a specific level of biological response to the dose of radiation A required to produce an equal biological response, with all physical and biological variables, except radiation quality, being held as constant as possible (ICRP, 2007). RBE as so defined is a radiobiological quantity that does not depend on the dose–response relationships for the two radiations having the same functional form (e.g. a linear-quadratic relationship), or that each dose response be a proportional (linear) relationship. (A3) In most studies to estimate RBE, radiation A is a high-LET radiation and the reference radiation is a specified low-LET radiation. However, this need not be the case. For example, the radiation of interest in many studies is a lower-energy, low-LET radiation (e.g. orthovoltage x rays, lower-energy x rays such as those used in mammography, or beta particles emitted in tritium decay), and the reference radiation is higher-energy gamma rays (photons), such as those emitted in 60Co decay. Any radiation of interest and reference radiation can be chosen, provided that they differ in quality (LET). (A4) When an RBE value obtained in a study is extrapolated to other doses not included in that study using assumed dose–response relationships for the two radiations, to other biological systems, to other biological endpoints of the same type (stochastic or deterministic), or to other radiations of similar LET, the resulting inference about biological effectiveness is not strictly RBE as this term is defined above. Nonetheless, the term ‘RBE’ is widely used to describe inferred RBE that is based on specific radiobiological studies.
A.1.1. Factors that influence relative biological effectiveness
(A5) There are several factors that influence estimates of RBE obtained from radiobiological studies. Amongst others, these include the chosen reference radiation, the magnitude of the dose or dose rate and extent of dose fractionation, and the biological endpoint under study (i.e. whether the endpoint is a stochastic effect or a tissue reaction, and the particular effect of either type). Certain other factors can also be important.
A.1.1.1. Choice of reference radiation
(A6) Reference radiations used in radiobiological studies to estimate RBE are usually orthovoltage (e.g. 150–300 kVp) x rays or higher-energy photons (gamma rays). Many radiobiological studies have shown a significant difference in biological effectiveness of these two common types of reference radiation. (A7) Differences in biological effectiveness of orthovoltage x rays and higher-energy photons are especially evident in some studies of stochastic effects. For example, reviews of data for stochastic effects by NCRP (1990) and ICRP (2003) suggest that, at low doses of interest in the radiological protection of humans, the biological effectiveness of orthovoltage x rays is approximately twice (1.5–2 times) the biological effectiveness of higher-energy photons (e.g. 60Co gamma rays). This difference in biological effectiveness has also been recognised in the BEIR VII report (National Research Council, 2006). Recognition of a difference of this magnitude is important when comparing RBE values for stochastic effects that were obtained in studies using different low-LET reference radiations. This is especially the case in comparing RBE values for lower-energy, low-LET radiations, such as tritium beta particles. (A8) Differences in biological effectiveness of orthovoltage x rays and higher-energy photons appear to be less important in studies of tissue reactions. For example, early studies of tissue reactions reviewed by NCRP (1967) indicated that, at high dose rates where such effects occur, the biological effectiveness of orthovoltage x rays is only approximately 20% higher than the biological effectiveness of photons emitted in 60Co decay. Such small differences are relatively unimportant compared with uncertainties in RBE values estimated using either reference radiation. (A9) Publication 92 (ICRP, 2003) recommends that the preferred low-LET reference radiation for use in radiobiological studies is high-energy photons emitted in 60Co decay. This choice has a number of advantages including: (i) the photon energy is discrete and well defined, in contrast to the continuous and variable spectra of photons in studies using orthovoltage x rays that depend on the tube potential (kVp) and filtration (filter material and thickness); and (ii) the photon energy is closer to the average energy of photons in exposures of Japanese atomic bomb survivors, studies of which provide the primary source of data on cancer risks from exposure to ionising radiation.
A.1.1.2. Dose, dose rate, and dose fractionation
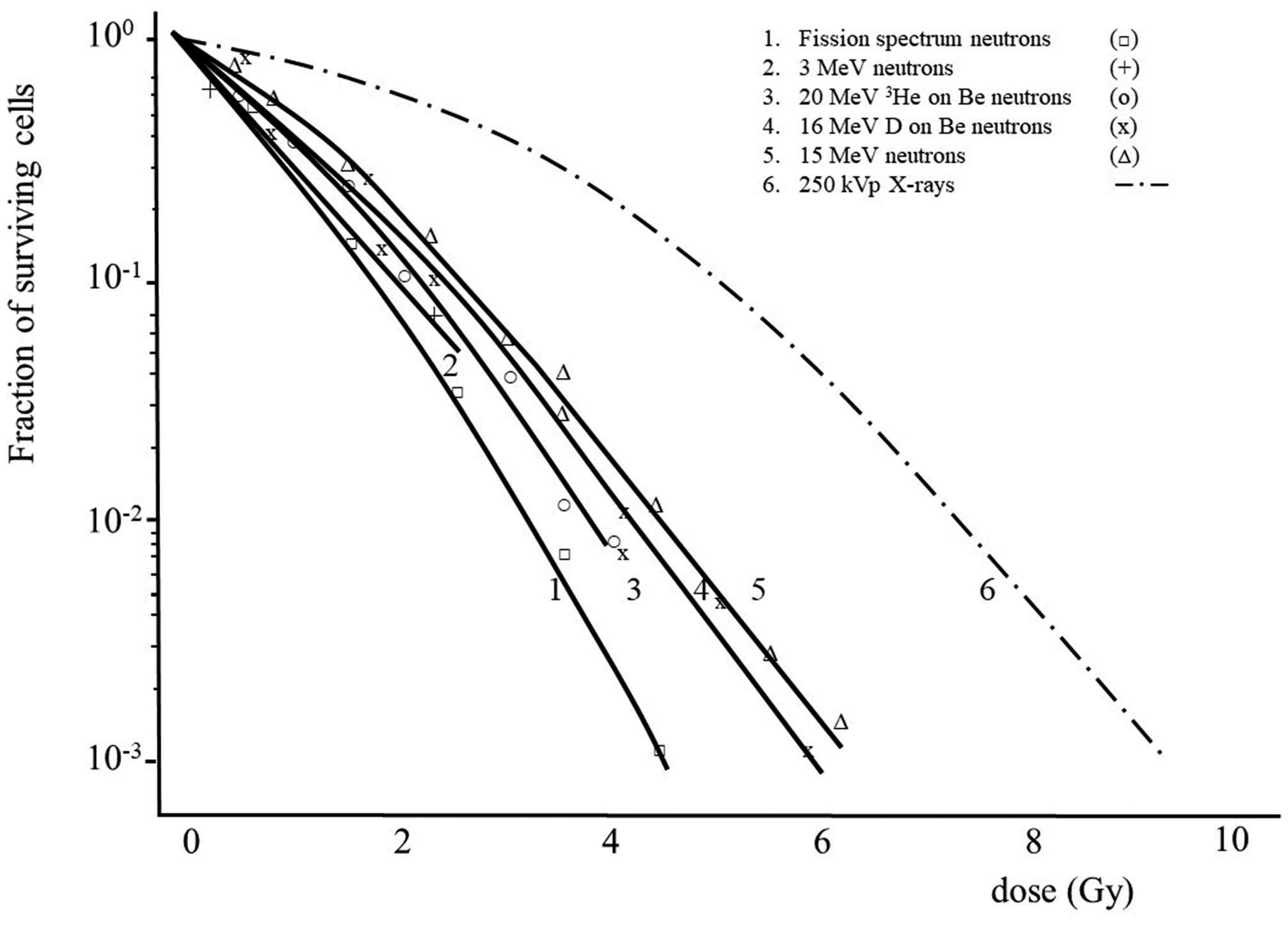
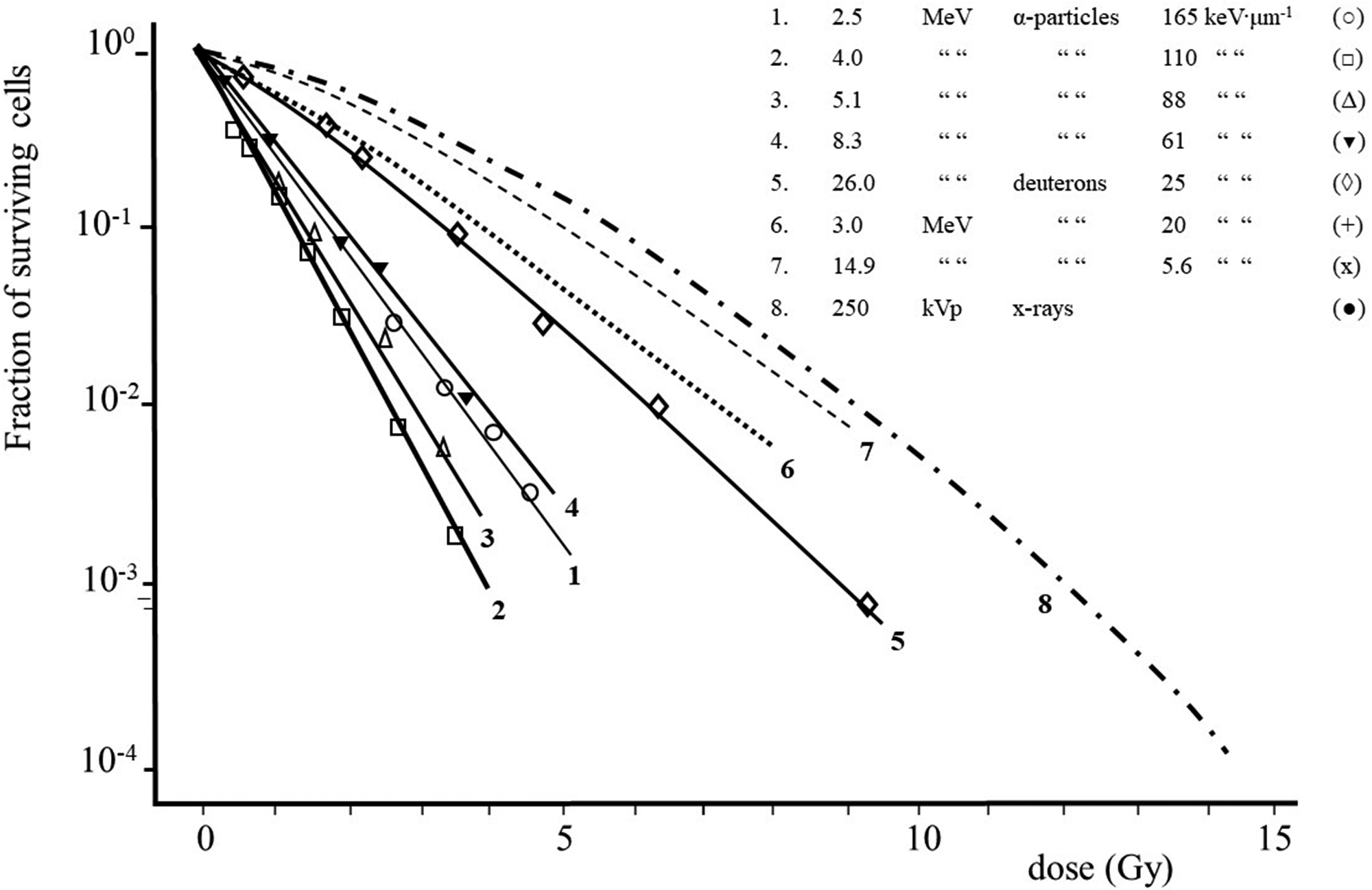
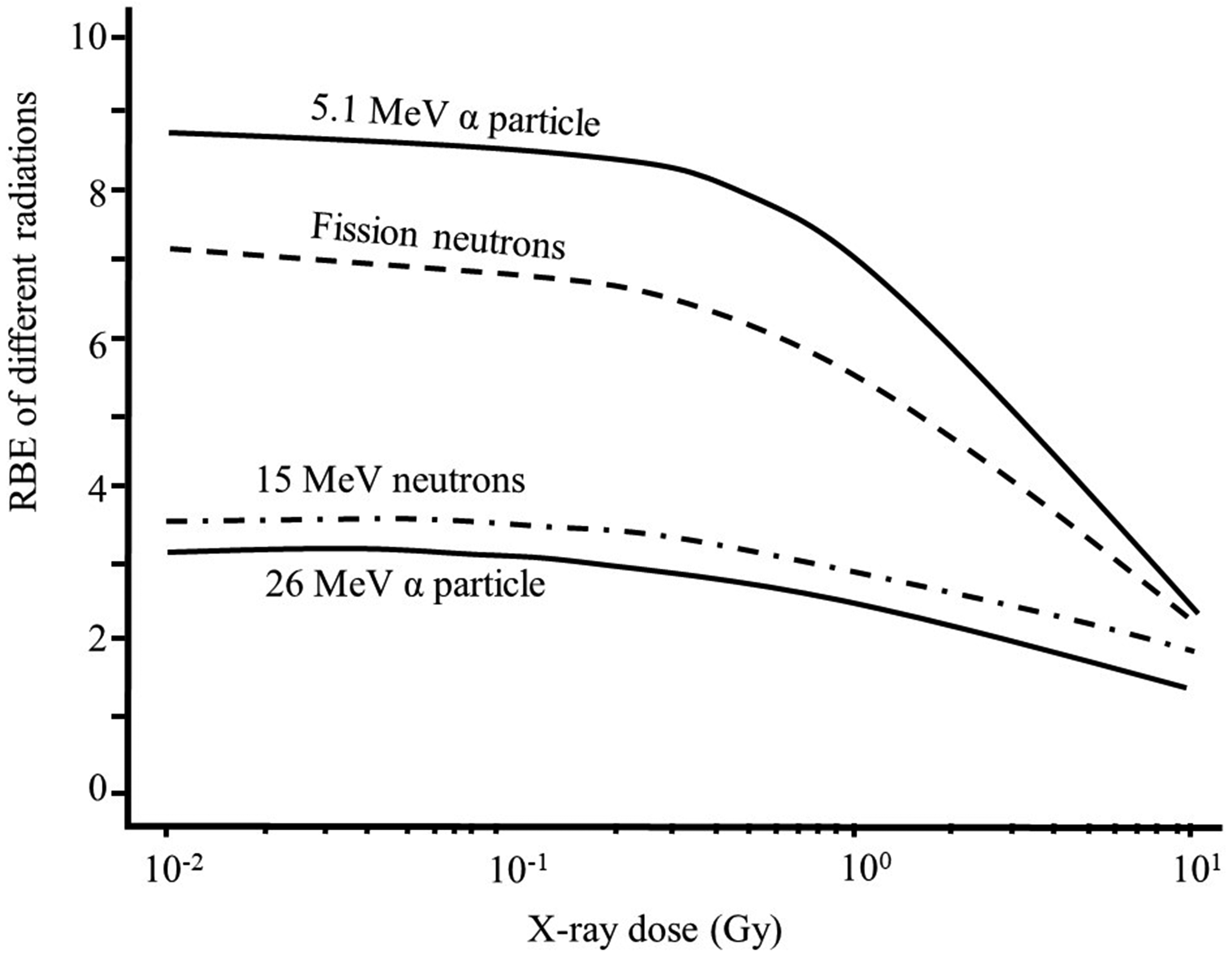
(A10) The magnitude of the absorbed dose and dose rate and the extent of dose fractionation can influence estimates of RBE obtained in radiobiological studies. RBE depends on the dose, dose rate, and dose per fraction in fractionated exposures whenever the dose–response relationship for the radiation of interest, the reference radiation, or both is non-linear. This effect is illustrated in Fig. A.1 (adapted from CNSC, 2002) which shows the response as a function of dose, both on a linear scale, for induction of a stochastic effect by a high-LET radiation and a reference low-LET radiation. As observed in many studies, the dose response for the high-LET radiation is assumed to be linear (R = αD), whereas the dose response for the reference radiation is assumed to be linear-quadratic in form (R = αD + βD2). As a consequence of this difference in the dose–response relationships, in this example, RBE for the high-LET radiation at higher doses is approximately 2 and increases to approximately 5 at lower doses. (A11) A similar dependence of RBE on dose is seen in many studies of tissue reactions. Examples of the dependence of RBE for various high-LET radiations on the dose of reference orthovoltage x rays in studies of cell survival are shown in Fig. A.2 (ICRP, 1990). The curves in Fig. A.2 are based on assumptions of a survival function for high-LET radiations of the form S = exp(–αD) and a survival function for the reference low-LET radiation of the form S = exp[–(αD + βD2)]. These survival functions are discussed further below. In these examples, the dependence of RBE on energy is most pronounced in the case of exposure to 5.1-MeV alpha particles, where RBE increases by more than a factor of 2 as the dose of the reference radiation decreases from 10 to ≤0.1 Gy. The dependence of RBE on dose is less pronounced in the cases of exposure to the two higher-energy, high-LET radiations. Dose–survival curves for cultured cells of human origin irradiated with different beams of fast neutrons and with 250-kVp x rays [Fig. 3B from ICRP (1990) and from Barendsen (1968)]. Biological effect as a function of dose for high- and low-linear energy transfer (LET) radiation. The graph illustrates how the calculated value of relative biological effectiveness (RBE) can differ based on the dose (high or low) used for the calculation (adapted from CNSC, 2002).
A.1.1.3. Type of biological endpoint
(A12) Estimates of RBE generally depend on the nature of the biological endpoint under study; that is, whether the effect is stochastic, in which case the probability of a response is a function of dose without a threshold; or a tissue reaction, in which case the severity of an effect (but not its probability) is a function of dose, and a threshold usually exists. (A13) Tissue reactions include impairment of tissue integrity and function, but also include cellular responses. Cellular reproductive death is presumed to be a significant source of tissue reactions (ICRP, 1984, 2012). Tissue reactions are presumed to have a threshold, and occur because sufficient damage has occurred that complete underlying repair is not possible. The severity of the effect therefore increases with higher doses. (A14) Stochastic radiation effects are characterised by the lack of a threshold. Conceptually, this means that a single event (i.e. radiation damage to one cell) is sufficient to cause the effect. In humans, the main stochastic effect is cancer, with the assumption of hereditary effects based on mouse data (ICRP, 2007). The frequency of the effect is related to the dose, but not its severity. However, radiation effects at chromosomal and cellular levels do not usually translate into detriment at population level, and hence RBE for stochastic effects in an individual member of the species is of limited concern for population-level effects in non-human biota. Radiological protection of non-human biota has largely focused on endpoints at individual level that could lead to impact on the maintenance of biological diversity or the conservation of species, such as reduced reproductive success, arising, for example, from effects on fertility, fecundity, growth, and early mortality. (A15) Although most biological effects can be classified as either stochastic or tissue reactions, there can be substantial variations in RBE for either type of effect, depending on the particular effect and the biological system under study. As a consequence, judgement is often required in evaluating whether an RBE value for a particular endpoint in a particular biological system is relevant to the principal concern in a system of radiological protection of non-human biota, for example maintaining the viability (reproductive capability) of the most sensitive species in radiological protection of the environment. (A16) It should also be noted that a recent ICRP report on tissue effects (ICRP, 2012) suggests that, at least for some endpoints (e.g. circulatory disease and damage to the lens of the eye), the same threshold has been proposed for acute, and either fractionated or protracted (chronic) doses, somewhat blurring the distinction between stochastic effects and tissue reactions. (A17) RBE values for high-LET radiations in inducing tissue reactions are generally lower than RBE values for those radiations in inducing stochastic effects. For example, at doses of interest in radiological protection, the reduction in RBE values for tissue reactions induced by alpha particles and fission neutrons compared with RBE values for stochastic effects appears to be approximately a factor of 2–3 (ICRP, 1990; Kocher and Trabalka, 2000). A reasonable explanation for this effect is that even at the lowest doses where significant tissue reactions are observed, occurring only in the event of severe damage to or death of a substantial fraction of cells in organs and tissues, the density of ionisation of the nominally low-LET reference radiation is relatively high and closer to the organ-averaged density of ionisation of a high-LET radiation of interest than is the case at lower doses where only stochastic effects are induced.
A.1.1.4. Other potentially important influences
(A18) Other factors can influence estimates of RBE in some studies (ICRP, 1990). Potentially important factors can include the time interval between an irradiation and observation of an effect; the conditions of the biological system under study, such as the proliferative state and cell cycle distribution; and the presence or absence of sensitising or protecting compounds, such as reactive oxidative species. Such factors can also confound an evaluation of the relevance of RBE to radiological protection of humans or the environment.
A.1.2. Extrapolation of relative biological effectiveness to low doses and dose rates
(A19) In radiological protection of humans, where limitation of the risk of cancer is the primary concern and the risk is assumed to be non-zero at any dose, it is generally accepted that quality factors and radiation weighting factors should be established on the basis of estimates of RBE values for stochastic effects at low doses and low dose rates that are obtained by extrapolation to zero dose of assumed dose–response relationships for a radiation type of interest and a reference radiation. For example, when the dose response for a stochastic effect induced by a high-LET radiation (H) is assumed to be linear (RH = αHD) and the dose response for the reference low-LET radiation (L) is assumed to be linear-quadratic (RL = αLD + βLD2), RBE at low doses and dose rates, denoted by RBEM, is the ratio of the slope of the dose response for the high-LET radiation to the slope of the dose response for the reference radiation as D → 0: RBEM = αH/αL. Given the dependence of RBE on dose discussed in Section A.1.1, RBEM is a maximum value for the stochastic effect under study. (A20) A similar approach of extrapolating observed dose–response relationships for tissue reactions induced by a high-LET radiation of interest and a reference low-LET radiation to obtain an estimate of RBE at low doses (i.e. as D → 0) for purposes of radiological protection of humans is used in Publication 58 (ICRP, 1990); RBE for tissue reactions at low doses, which is equivalent to RBEM for stochastic effects, is denoted by RBEm to indicate that this is a maximum value. Although dose–response relationships for tissue reactions are presumed to have a threshold, estimation of RBEm was judged to be ‘necessary for assessing the risk of exposure conditions where a small dose of high-LET radiation is delivered together with low-LET radiation’ (ICRP, 1990). That is, for purposes of radiological protection, use of RBEm was considered necessary to address induction of tissue reactions from exposure to mixed radiation fields in which, for example, the dose from a low-LET radiation is above a threshold dose but the dose from a high-LET radiation may be orders of magnitude below the threshold. (A21) Although the definition and use of RBEM for stochastic effects for purposes of radiological protection is relatively straightforward, there is a conceptual difficulty with use of RBEm for tissue reactions that arises from the assumption that their dose–response relationships have thresholds. However, it appears that extrapolated RBE values for tissue reactions are largely independent of dose below a level that may be comparable to a threshold. (A22) On the basis of the considerations discussed above, including that estimates of RBEm are expected to be maximum values, the practice of estimating RBEm by extrapolation of data on dose response for tissue reactions induced by a radiation of interest (e.g. alpha particles or tritium beta particles) and a reference low-LET radiation is continued in this publication. This approach is considered appropriate for the purposes of deriving weighting factors relevant to non-human biota and radiological protection of the environment.
A.1.3. Extrapolation of data on relative biological effectiveness for tissue reactions through levels of biological organisation
(A23) As indicated previously, the most common studies of RBE for tissue reactions involve irradiation of mammalian cells in culture, and most studies measured cell reproductive death. This is especially the case in studies in which the radiation of interest is alpha particles. Less common are studies of RBE for tissue reactions in whole organs, tissues, or whole organisms of direct relevance to radiological protection of the environment. (A24) The problem of extrapolating estimates of RBE obtained from studies of reproductive death in cultured cells to obtain estimates of RBE for tissue reactions in whole organs, tissues, or whole organisms is addressed in Publication 58 (ICRP, 1990) by comparing data for responses in whole tissues with data for survival of the critical cells in the same tissues. For example, in studies of early damage to the intestinal tract from irradiation by orthovoltage x rays or 15-MeV neutrons, RBE for the mean lethal dose within 4 days (LD50/4d) was similar to RBE for survival of intestinal crypt stem cells. This and other studies of exposure of various tissues and their critical cells were used to support an assumption that cell reproductive death is mainly responsible for tissue injury (ICRP, 1990). (A25) On the basis of the arguments and supporting studies discussed in Publication 58 (ICRP, 1990), it is assumed in this publication that estimates of RBE obtained from studies of cell reproductive death (cell survival) can be used to infer RBE for induction of tissue reactions in whole organs, tissues, or whole organisms.
A.2. Modelling of dose response for cell survival
(A26) As indicated previously, reproductive death of irradiated mammalian cells is the most common biological endpoint in studies to estimate RBE values for alpha particles in inducing tissue reactions. Cell killing is another frequent endpoint in studies to estimate RBE values for tritium beta particles. (A27) In Publication 58 (ICRP, 1990), analyses of data on cell survival from exposure to high- and low-LET radiations were based on an assumption that the dose–response relationship can be described by a linear-quadratic model. However, some studies have used a different description of the dose–response relationship for cell survival, which is referred to as a ‘single-hit, multi-target model’. (A28) This section discusses two models to describe the dose–response relationship for cell survival. These discussions emphasise the properties of the two models at high and low doses and use of the models to estimate RBE at low doses, RBEm, which is the quantity of interest in radiological protection. A concluding discussion compares the two models and considers the extent to which they are compatible.
A.2.1. Linear-quadratic model and its characteristic parameters
(A29) The most commonly used mathematical description of the dose–response relationship for cell survival is the linear-quadratic model. This model is based on an assumption that cell reproductive death can be caused by damage caused by a single track or by an accumulation of damage caused by two or more tracks of ionising particles (ICRP, 1990). The linear-quadratic model is a consequence of the theory of dual radiation action, which has some biological basis. This theory is used to derive the linear-quadratic model for induction of stochastic effects, in which the frequency of an effect, F, at dose D is assumed to be described by the equation:
(A30) In the linear-quadratic model to describe the dose–response relationship for cell survival, the surviving fraction, S, of cells that receive a dose D, assuming that all unirradiated cells survive [S(0) = 1], is described by the equation:
(A31) Cell-survival curves are typically displayed as plots of the natural logarithm of S as a function of dose D:
(A32) Many curves of cell survival in cases of exposure to low-LET radiation are described by Eq. (A.3). In cases of exposure to high-LET radiation, it is commonly observed that β ≈0 and ln S is essentially a linear function of dose at any dose, in a manner similar to the usual linearity in dose–response relationships for stochastic effects. Examples of survival curves for various radiations are shown in Fig. A.4 (ICRP, 1990). The survival curve for 250-kVp x rays (Curve 8) shows the influence of the quadratic term (β ≠ 0) for low-LET radiation, whereas the survival curves for alpha particles of energy typical of energies of alpha particles emitted in radioactive decay (Curves 2, 3, and 4) are essentially linear. (A33) The description of a cell-survival curve in Eq. (A.3) has two important properties. As noted previously, at low doses, where the quadratic term is negligible, the survival curve is essentially linear with a slope given by:
(A34) At higher doses where the quadratic term is not negligible, the survival curve is non-linear, with a slope that is a function of dose given by:
(A35) When the linear-quadratic model is used to describe cell survival, RBE of a high-LET radiation (H) of interest at low doses (i.e. as D → 0) is estimated as the ratio of the value of α in the survival curve for that radiation to the value of α in the survival curve for the reference low-LET radiation (L):
Dose–survival curves for cultured cells of human origin obtained with radiations of different linear energy transfer (Barendsen, 1968).




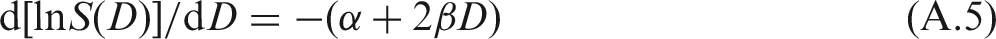

A.3. Prior reports on relative biological effectiveness
(A36) This evaluation of the biological effectiveness of alpha particles and tritium beta particles in inducing tissue reactions of potential relevance to viability of the RAPs examined previous reports by ICRP and other organisations or investigators. Most of those reports were prepared to support the development of recommendations on biological effectiveness for purposes of radiological protection of humans. Nonetheless, given that most of the available data were obtained from studies of radiation effects in biological systems other than those of human origin, portions of the previous work were directly relevant to protection of the environment. (A37) Several publications by ICRP and other advisory groups that develop recommendations on radiological protection provided information of use to the present publication. These publications include ICRU Report 40 (ICRU, 1986), Publication 58 (ICRP, 1990), Publication 92 (ICRP, 2003), NCRP Report No. 89 (NCRP, 1987), and NCRP Report No. 104 (NCRP, 1990). An earlier report by ICRP, Publication 31 (ICRP, 1980), was used to a lesser extent.
A.3.1. ICRU Report 40
(A38) ICRU Report 40 (ICRU, 1986), which was prepared by a joint task group of ICRP and ICRU, was concerned with theoretical considerations, calculations, and experimental data that could be used to develop recommendations on effective quality factors for use in radiological protection of humans. The publication is concerned mainly with RBE values at low doses for a variety of stochastic effects in biological systems ranging from cells to whole organisms. However, some information on RBE values for tissue reactions induced by fission neutrons is also presented. (A39) Several presentations in ICRU Report 40 (ICRU, 1986) were relevant to the development of the present publication. These include discussions on: (i) the potential importance of differences in biological effectiveness between high-energy gamma rays (photons of energy >∼250 keV) and lower-energy photons (e.g. orthovoltage x rays) or tritium beta particles, as indicated by calculations and available data; (ii) the weak energy dependence of the effective quality factor for alpha particles at energies of 4–9 MeV, which encompass the energies of alpha particles emitted by most potentially important radionuclides in the environment; and (iii) available data on RBE values for stochastic effects induced by high-LET radiations, mainly data for fission or other neutrons, but also including more limited data for alpha particles and heavy ions.
A.3.2. Publication 58
(A40) For the purposes of the present publication, Publication 58 (ICRP, 1990) is the most important source of information on RBE values for tissue reactions induced by high-LET radiations, including alpha particles, neutrons, and heavy ions. RBE values for stochastic effects are not discussed in Publication 58. In addition to an extensive review of studies of RBE values for high-LET radiations in inducing tissue reactions in cultured mammalian cells and whole organs or tissues of animals and humans, Publication 58 discusses basic aspects of deterministic radiation effects and the use of data on RBE for purposes of radiological protection, especially extrapolation of estimates of RBE at high doses to lower doses of potential importance in radiological protection. (A41) Discussions in the present publication make considerable use of information in Publication 58 (ICRP, 1990). Important examples include descriptions of dose–response relationships for cell survival using a linear-quadratic model, the dependence of RBE for tissue reactions on dose and extrapolation of RBE to low doses of concern for radiological protection, and reviews and evaluations of data on RBE of neutrons and heavy ions, which can be used in evaluating data on RBE of alpha particles.
A.3.3. Publication 92
(A42) Publication 92 (ICRP, 2003) presents a review of data on RBE for induction of stochastic effects by low- and high-LET radiations and recommendations on quality factors and radiation weighting factors for different radiation types for use in radiological protection of humans that were developed on the basis of the available data and other considerations. Publication 92 is not concerned with RBE for tissue reactions. (A43) Information in Publication 92 (ICRP, 2003) that was used in the present publication mainly concerns RBE of alpha particles. Given the emphasis of Publication 92 on protection of humans, much of the discussion on RBE of alpha particles focuses on estimates obtained from studies of lung cancer, bone sarcomas, leukaemia, and liver cancer in humans. However, Publication 92 also discusses RBE for those effects in animals, and RBE obtained from studies of neoplastic transformation in animal cells and dicentric chromosomal aberrations in human lymphocytes.
A.3.4. NCRP Report No. 89
(A44) NCRP Report No. 89 (NCRP, 1987) is concerned with induction of stochastic genetic effects from exposure to radionuclides that are incorporated in mammalian cells or whole organisms. The publication focuses primarily on data on genetic effects from incorporated alpha emitters, and comparisons with genetic effects from incorporated higher-energy beta emitters for the purpose of estimating the risk from alpha particles relative to the risk from beta particles. However, data on genetic effects from exposure to incorporated tritium are also presented.
A.3.5. NCRP Report No. 104
(A45) NCRP Report No. 104 (NCRP, 1990) presents an extensive review of data on RBE for induction of stochastic effects by low- and high-LET radiations, principally x rays (low-LET) and neutrons and alpha particles (high-LET). RBE for tissue reactions was not considered. A wide variety of data is discussed, including data on cytogenetic effects in plant, animal, and human cells; transformation and mutation in mammalian cells in vitro; several hereditable effects; carcinogenesis in animals from external high-LET radiation (principally neutrons but also including limited data for heavy ions); data on several endpoints in cells and whole organisms from incorporated radionuclides; and data on life shortening in mice.
Relative biological effectiveness (RBE) vs dose curves illustrating that RBE values approach RBEm (maximum RBE for tissue reactions) values at doses <10–1 Gy of x rays [Fig. 5 from ICRP (1990)].
A.4. References
Barendsen, G.W., 1968. Responses of cultured cells, tumours and normal tissues to radiations of different linear energy transfer. In: Ebert, M., Howard, A. (Eds.), Current Topics in Radiation Research, Volume IV. North-Holland Publishing Company, Amsterdam, pp. 293–356.
CNSC, 2002. Protection of Non-human Biota from Ionizing Radiation. INFO–0730. Canadian Nuclear Safety Commission, Ottawa.
ICRP, 1980. Biological Effects of Inhaled Radionuclides. ICRP Publication 31. Ann. ICRP 4(1/2).
ICRP, 1984. Nonstochastic effects of ionizing radiation. ICRP Publication 41. Ann. ICRP 14(3).
ICRP, 1990. RBE for deterministic effects. ICRP Publication 58. Ann. ICRP 20(4).
ICRP, 2003. Relative biological effectiveness (RBE), quality factor (Q), and radiation weighting factor (wR). ICRP Publication 92. Ann. ICRP 33(4).
ICRP, 2007. The 2007 Recommendations of the International Commission on Radiological Protection. ICRP Publication 103. Ann. ICRP 37(2–4).
ICRP, 2012. ICRP statement on tissue reactions/early and late effects of radiation in normal tissues and organs – threshold doses for tissue reactions in a radiation protection context. ICRP Publication 118. Ann. ICRP 41(1/2).
ICRU, 1986. The Quality Factor in Radiation Protection. ICRU Report 40. International Commission on Radiation Units and Measurements, Bethesda, MD.
Kocher, D.C., Trabalka, J.R., 2000. On the application of a radiation weighting factor for alpha particles in protection of non-human biota. Health Phys. 79, 407–411.
National Research Council, 2006. Health Risks from Exposure to Low Levels of Ionizing Radiation, BEIR VII Phase 2. National Academies Press, Washington.
NCRP, 1967. Dose–effect Modifying Factors in Radiation Protection. Report of Subcommittee M-4 (Relative Biological Effectiveness) of the National Commission on Radiation Protection. BNL5007(T-471). National Council on Radiation Protection and Measurements, Bethesda, MD.
NCRP, 1987. Genetic Effects from Internally Deposited Radionuclides. NCRP Report No. 89. National Council on Radiation Protection and Measurements, Bethesda, MD.
NCRP, 1990. The Relative Biological Effectiveness of Radiations of Different Quality. NCRP Report No. 104. National Council on Radiation Protection and Measurements, Bethesda, MD.
ANNEX B. RELATIVE BIOLOGICAL EFFECTIVENESS OF TRITIUM BETA PARTICLES
(B1) Tritium is the only radioactive isotope of the element hydrogen. Its nucleus contains one proton and two neutrons. It decays by beta particle emission, with a half-life of 12.3 years, to form stable helium (two protons and one neutron). Its atoms can replace hydrogen atoms in any molecule. Beta particles from tritium decay travel only approximately 6.0 mm in air, and they do not penetrate the dead layer of the skin. Tritium beta particles are completely absorbed by a sheet of glass, plastic, or metal. Therefore, the main hazard associated with tritium is when it is incorporated into the organism (ingestion, inhalation, absorption through the skin) and beta particles are emitted inside the body. (B2) In living tissues, tritium beta particles travel only approximately 6 µm (the average diameter of a typical animal cell is 10–20 µm and a nucleus is 6–15 µm, whereas plant cells may be 100 µm in diameter). Due to its low initial energy and short range, the average ionisation density (the linear energy transfer, LET) produced by the emitted beta particle is higher than that produced by higher-energy beta particles or photons. Tritium beta particles (mean 5.7 keV) have a track average LET in water of 4.70 keV µm−1, compared with LET values of 0.22, 0.52, and 1.7 keV µm−1 for 60Co gamma rays (1173 and 1332 keV), 90Sr beta rays, and 200-kVp x rays, respectively (ICRU, 1970). It has been calculated that the fraction of dose to tissue from tritium delivered by low-energy beta particles and/or secondary electrons (energies between 0.1 and 5 keV) is approximately 78%. This is in contrast to 33% for 60Co gamma rays (Nikjoo and Goodhead, 1991). (B3) As tritium is an isotope of hydrogen, it reacts chemically to form compounds in the same manner as hydrogen, and, thus, can be a constituent atom of a wide variety of molecules, such as water or several organic compounds. Tritium can be found in oxide form (HTO), bound to organic compounds, or as tritiated gas. Tritium in gaseous form (HT) is readily oxidised to HTO in the atmosphere, or through microbial agents near the soil surface. Therefore, HT in the environment does not generally imply an important exposure of humans or other organisms. (B4) Tritium is most commonly found in the environment as HTO. HTO has the same chemical properties as water. Once HTO is incorporated into the organism, it quickly reaches equilibrium with water in the body and is distributed uniformly among all soft tissues. For plants, tritium may label organic matter as OBT through metabolic processes, such as photosynthesis (Boyer et al., 2009). HTO is eliminated from the organism at the same rate as water. (B5) OBT refers to those forms in which tritium has been incorporated into organic molecules such as carbohydrates, fats, or proteins. Two types of OBT can be distinguished: exchangeable and non-exchangeable. When tritium atoms are bonded to oxygen, sulphur, nitrogen, or phosphorus atoms, the tritium can readily exchange with hydrogen in body water, and, therefore, is considered exchangeable. Exchangeable tritium in OBT compounds exhibits kinetics indistinguishable from HTO. When a tritium atom is bonded to a carbon atom in an organic molecule, it is non-exchangeable and can only be released by enzymatic reactions. Non-exchangeable tritium in OBT compounds exhibits kinetics characteristic of the OBT molecules concerned and their turnover in body tissues. (B6) When HTO is incorporated into animals, it will be distributed almost homogeneously throughout the body fluids within a short time after intake, as tritium exchanges easily and rapidly with other hydrogen atoms. A small proportion is incorporated non-exchangeably into organic molecules during their synthesis (becomes non-exchangeable OBT). Tritium can also be ingested by animals as OBT in foods. The biological half-time (time required for half of the activity to be physically removed from the body) in adult humans is 10 days for HTO and 40 days for non-exchangeable OBT (ICRP, 1993). Biokinetic and dosimetric models have been developed for humans of different ages, and have been used to calculate dose coefficients for intakes of tritium as HTO, OBT, or HT (tritiated gas) (ICRP, 1989, 1993, 1994, 1995, 1996). (B7) Studies of tritium exposure to plants show fast equilibrium between above- and below-ground parts and environmental concentrations in air and soil, with half-lives of the order of hours to a few days (Boyer et al., 2009). (B8) When tritium is incorporated into DNA (e.g. after administration of tritiated thymidine), the beta doses received by cells will depend on the length of their division cycles. Cells that divide rapidly will have more chance of incorporating tritiated thymidine, but they will also eliminate it more rapidly. In cells with small proliferation rates, the probability of incorporating tritiated thymidine will be much lower, but retention times will be longer. Estimation of beta doses received from OBT has many more uncertainties than estimation of the dose received from HTO (NCRP, 1979; Straume and Carsten, 1993).
B.1. Review of experimental studies on relative biological effectiveness of tritium beta particles
(B9) In this publication, published data on RBE of tritium beta particles have been considered, provided that enough details on the experimental procedures used and the results obtained were reported. (B10) The experimental data on RBE of tritium beta particles have been grouped in this publication within one of four biological endpoints: early mortality, reproductive success, morbidity, or chromosomal damage and mutations; only the first three endpoints are considered to be relevant to viability (ICRP, 2008).
Data on relative biological effectiveness (RBE) of tritium beta particles for early mortality.
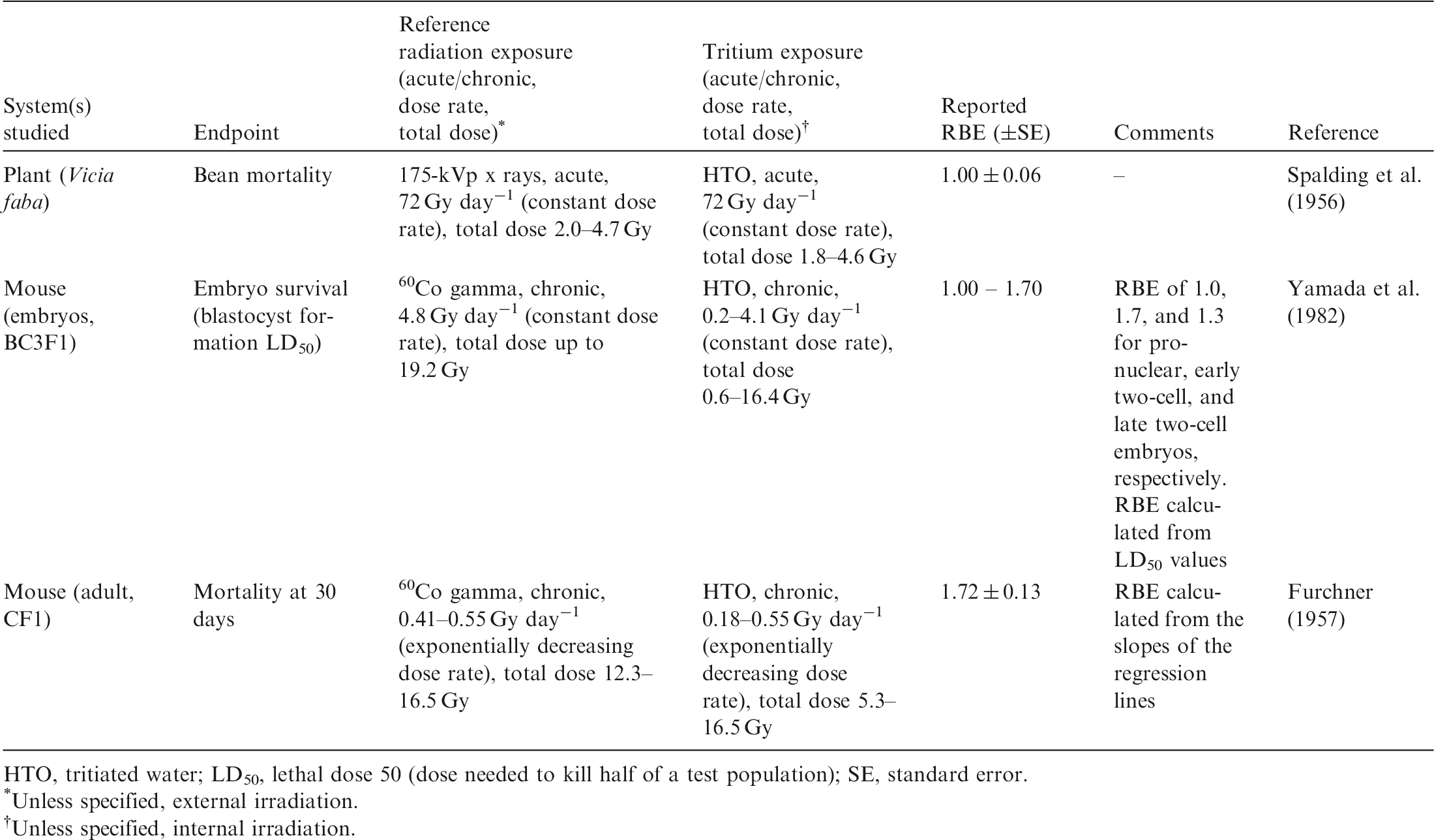
HTO, tritiated water; LD50, lethal dose 50 (dose needed to kill half of a test population); SE, standard error.
Unless specified, external irradiation.
Unless specified, internal irradiation.
B.1.1. Data on relative biological effectiveness for early mortality
(B11) RBE of tritium beta particles for lethal effects on plants (broad bean root, Vicia faba) was estimated by Spalding et al. (1956). Bean roots were exposed to HTO at cumulative doses of 1.8–4.6 Gy (dose rates of 72 Gy day−1) or were acutely irradiated with 175-kVp x rays at total doses of 2.0–4.7 Gy (dose rates of 72 Gy day−1). The mortality of the beans was quantified in both groups, and RBE of 1.0 ± 0.06 was calculated. (B12) The effects of tritium beta particles on survival of mice were studied by Furchner (1957). Adult mice (CF1 strain) received a single intraperitoneal injection of HTO, and their mortality was recorded 30 days after the injection (cumulative doses over 30 days in the range of 5.3–16.5 Gy). Mortality at 30 days was also analysed in a group of mice chronically exposed to 60Co gamma rays (reference radiation) at total doses of 12.3–16.5 Gy. Gamma irradiation was performed at decreasing dose rates (0.41–0.55 Gy day−1) to mimic the exponential decay of tritium. RBE of 1.7 ± 0.1 was calculated from the slopes of the regression lines of the dose–response curves. (B13) Yamada et al. (1982) studied the effects of in-vitro irradiation with tritium beta particles and gamma rays on mouse embryo survival. Mouse embryos [BC3F1 (C3H/C57BL)] in pronuclear or two-cell stage were cultured in vitro, and HTO was added to the culture medium at concentrations leading to dose rates of 0.2–4.1 Gy day−1 (after 3 days, the accumulated dose was in the range of 0.6–16.3 Gy). 60Co gamma rays were used as the reference radiation (chronic irradiation for 3 days at dose rate of 0.48 Gy day−1 and total dose of up to 19.2 Gy). RBE values as calculated from LD50 values (dose needed to kill half of a test population) were 1.0, 1.7, and 1.3 for pronuclear, early two-cell, and late two-cell embryos, respectively. (B14) In summary, all studies that have estimated RBE of tritium beta particles for reduced survival of individuals have used HTO as the radiation source. The species used included plants (Vicia faba) and mice (BC3F1 embryos and CF1 adult mouse). Each of the studies involved chronic irradiation at high cumulative doses administered at high dose rates. The values of RBE for increased mortality were in the range of 1.0–1.7 (Table B.1).
B.1.2. Data on relative biological effectiveness for reduction of reproductive success
(B15) Etoh and Hyodo-Taguchi published three studies on RBE of tritium beta particles for alteration of the reproductive capacity of medaka fish (Oryzias latipes) using gamma rays as the reference radiation (Etoh and Hyodo-Taguchi, 1983; Hyodo-Taguchi and Etoh, 1986, 1993). In the first study (Etoh and Hyodo-Taguchi, 1983), fertilised medaka eggs were exposed to HTO within 2 h of fertilisation at concentrations giving dose rates of 0.17–1.70 Gy day−1 (cumulative doses absorbed by the eggs in 10 days of 1.8–17.0 Gy). Another group of fertilised eggs was chronically irradiated with 137Cs gamma rays at dose rates of 0.11–2.12 Gy day−1 (total doses received by eggs of 4.3–21.2 Gy). The doses needed to reduce survival of germ cells (female and male) to 50% were 1.95 Gy for tritium beta particles and 3.5 Gy for 137Cs gamma rays, giving RBE of 1.8. The doses needed to reduce female germ cell survival to 50% were 1.4 Gy for tritium beta particles and 3.05 Gy for 137Cs gamma rays, giving RBE of 2.2. (B16) Hyodo-Taguchi and Etoh (1986) studied the effects of tritium beta particles and 137Cs gamma rays on fertility and fecundity of medaka fish. Fertilised medaka eggs were treated for 10 days with HTO at cumulative doses of 0.85–34.0 Gy (dose rates in the range of 0.085–1.70 Gy day−1) or were chronically irradiated with 137Cs gamma rays at total doses of 0.61–25.4 Gy (dose rates of 0.06–2.54 Gy day−1). The authors did not estimate RBE. However, the doses needed to reduce the female reproductive capacity to 50% were 4.0 Gy for tritium beta particles and 15.0 Gy for gamma rays, giving an estimated RBE of 3.75. No differences were seen in the capacity of tritium beta particles and gamma rays to reduce male reproductive capacity. (B17) Hyodo-Taguchi and Etoh (1993) analysed the capacity of tritium beta particles and gamma rays to induce vertebral malformations in medaka fish. The fertilised fish eggs were exposed for approximately 9 days to HTO (dose rates of 0.43–1.70 Gy day−1 and cumulative doses of 3.7–16.7 Gy) or to 137Cs gamma rays (dose rates of 0.44–1.89 Gy day−1 and total doses of 4.2–18.8 Gy). RBE of tritium beta particles to induce vertebral malformations, estimated from the regression analysis of the dose–response curves, was 1. (B18) Knowles and Greenwood (1997) studied RBE of tritium beta particles to alter the reproductive capacity of aquatic invertebrates. Mature adult polychaete worms (Ophryotrocha diadema) were irradiated continuously from immediately prior to egg laying until development into mature adult. After treatment, the reproductive output of these adults was analysed. HTO was administered at concentrations delivering a dose rate of 0.175 Gy day−1. A group of worms was chronically irradiated with 137Cs gamma rays at the same dose rate. In both experimental groups, the reproductive performance of worms (sacs/worm, eggs/worm, larvae/worm, % survival eggs to larvae, days to first egg) was studied. The authors stated that the study examined a single dose rate for tritium beta particles and gamma radiation, and that no attempt was made to estimate RBE. However, they concluded that the two radiation types produced very similar effects on the reproductive capacity of the aquatic invertebrate Ophryotrocha diadema. (B19) Chopra and Heddle (1988) studied reduction in testes weight in mice, using 250-kVp x rays as the reference radiation. Adult mice (CBA/H strain) received a single intraperitoneal injection of HTO (dose rates in the range of 0.14–0.43 Gy day−1 and cumulative doses of 1.43–4.34 Gy) with the testes weight determined after 10 days. X-ray exposures were continued for a period of 10 days (dose rates 0.13–0.33 Gy day−1 and total doses of 1.33–3.36 Gy), with the testes weight determined after irradiation ended. The estimated RBE for reduction in mouse testes weight was in the range of 1.07–1.40. (B20) Carr and Nolan (1979) studied the effects of HTO and tritiated thymidine (3HTdR) on testes mass in adult CBA mice, comparing these effects with those produced by 60Co gamma rays. Gamma radiation exposures were in 15 fractions to mimic tritium exposure (total dose of 0.578 Gy). Tritium (HTO or 3HTdR) was administered by a single intraperitoneal injection, with average cumulative doses to testes of 0.145–0.58 Gy for HTO and 0.03–0.50 Gy for 3HTdR. Testes mass was determined in each experimental group up to 24 weeks after irradiation started. RBE of tritium beta particles was calculated from the slopes of the corresponding dose–response curves (integrated fractional mass loss as a function of the calculated average absorbed dose in the testes up to 10 weeks after irradiation), and values of 1.43 ± 0.19 for HTO and 2.07 ± 0.25 for 3HTdR were obtained. It should be noted that only one dose of 60Co was used in this study, so the reported RBE applies to that dose alone. (B21) The relative effectiveness of tritium beta particles to kill resting primary spermatocytes, compared with x rays, was studied in adult DBA2 mice (Lambert, 1969). Both HTO and 3HTdR were used in this study. A group of mice received a single intraperitoneal injection of HTO at concentrations that produced dose rates in the range of 0.04–0.06 Gy day−1 (cumulative doses of 0.05–0.12 Gy). 3HTdR was also injected intraperitoneally at concentrations giving dose rates in the range of 0.06–0.11 Gy day−1 (cumulative doses of 0.084–0.19 Gy). Simultaneously, a group of mice was chronically irradiated for 72 h with x rays at decreasing dose rates in the range of 0.02–0.16 Gy day−1 (total doses of 0.05–0.50 Gy). The resting primary spermatocytes were quantified at 19 and 72 h after tritium injection (HTO or 3HTdR) or x-ray exposure. For HTO, RBE values for tritium beta particles of 2.3 and 2.4 at 19 and 72 h after exposure, respectively, were estimated, whereas estimated RBE values for 3HTdR were 1.3 and 1.6 at 19 and 72 h after exposure, respectively. In the discussion of the paper, the authors highlight that RBE values calculated in the study must be viewed with circumspection, due to the assumptions made in calculating the doses. Furthermore, the authors do not provide much detail about the experimental design (e.g. the number of animals used in each group and the statistical methods used). (B22) Zhou et al. (1989) studied the effects of tritium beta particles and gamma rays on the survival of primary oocytes and spermatogonia in juvenile mice. Two different treatments with HTO were used: (i) a single intraperitoneal injection (exponentially decreasing dose rate); or (ii) a single intraperitoneal injection followed by tritium administration in drinking water (constant dose rate). The cumulative doses received over 10 days from HTO beta particles were in the range of 0.2–1.0 Gy. Another group of mice was chronically irradiated with 60Co gamma rays over 10 days (total doses of 0.7–2.8 Gy), either at an exponentially decreasing dose rate or at a constant dose rate. For an exponentially decreasing dose rate, RBE values for tritium beta particles, as calculated from the slopes of the dose–response curves, were 1.4–2.0 for primary oocyte survival and 2.1–2.8 for spermatogonia survival. When the irradiation took place at a constant dose rate, RBE values were 1.65 for primary oocyte survival and 2.3–2.5 for spermatogonia survival. (B23) Swiss–Webster mice were used to study RBE of tritium beta particles to reduce primary oocyte survival, compared with 60Co gamma rays (Dobson and Kwan, 1976). Mice were exposed to HTO for 33 days, from conception to 14 days after birth, at dose rates in the range of 2.20–19.80 mGy day−1 (cumulative doses of 0.07–0.65 Gy). Another group of mice was chronically irradiated with 60Co gamma rays (for 33 days, from conception to 14 days after birth) at dose rates in the range of 10.08–31.92 mGy day−1 (total doses of 0.33–1.05 Gy). At 14 days after birth, the number of primary oocytes was quantified in the female offspring of each experimental group. RBE of tritium beta particles increased as the dose administered decreased, with values of 1.8 at 0.4 Gy of the reference gamma radiation and 2.5 at 0.2 Gy. (B24) In 1977, Dobson and Kwan published a more complete follow-on study (Dobson and Kwan, 1977). They used the same experimental system (non-inbred Swiss–Webster mice) and approach (exposure to HTO or 60Co for 33 days from conception to 14 days after birth), but different dose ranges. HTO was administered at concentrations giving dose rates in the range of 24.96–51.52 mGy day−1 (cumulative doses of 0.57–0.83 Gy). 60Co gamma radiation was administered at dose rates of 8.0–38.0 mGy day−1 (total doses of 0.26–1.25 Gy). RBE of tritium beta particles of 2.5 for oocyte survival at low doses was estimated. As in the previous study (Dobson and Kwan, 1976), RBE decreased at higher doses and dose rates. (B25) Satow et al. (1989a) studied RBE of tritium beta particles for murine oocyte survival. Juvenile mice (ICR strain, 14 days old) received a single intraperitoneal injection of HTO (cumulative doses for 14 days of 0.04–0.25 Gy) or chronic irradiation with 137Cs gamma rays at decreasing dose rates to mimic exposure to HTO (dose rates in the range of 0.03–0.09 Gy day−1 and cumulative doses over 14 days of 0.06–0.21 Gy). RBE of tritium beta particles, as calculated from survival curves using the single-target model, was in the range of 1.1–3.5. The authors also observed that RBE increased with decreasing doses, as described by Dobson and Kwan (1976, 1977). The highest RBE of 3.5 was seen at the lower dose used (0.04 Gy) (Satow et al., 1989a). (B26) The same group studied the teratogenic effects of tritium beta particles and 137Cs gamma rays in rats (Satow et al., 1989b). In these experiments, mature rats (Donryu strain) received a single intraperitoneal injection of HTO on day 8 or 9 of pregnancy (dose rates of 0.14–1.06 Gy day−1 and cumulative doses of 1.75–6.80 Gy). Another group of rats was chronically irradiated with 137Cs gamma rays from day 9 to 18 of pregnancy (the dose rates used were similar to those from HTO, and the total doses received were 1.75–6.80 Gy). The percentage of foetuses surviving and the frequency of foetuses with anomalies were estimated in both groups of rats. The RBEs for tritium beta particles to produce 50% and 20% anomalies in total implants were 1.8 and 2.4, respectively. RBEs to produce anomalies in surviving foetuses of 50% and 20% were also estimated; the values were 2.0 and 2.6, respectively (Satow et al., 1989b). (B27) The effects of tritium beta particles and gamma rays on the frequency of dominant lethal mutations in mice oocytes were studied by Zhou et al. (1986). Adult female mice (LACA strain) received a single intraperitoneal injection of HTO (total absorbed ovarian doses of 39–912 mGy). Another group of mice was chronically irradiated for 10 days with 60Co gamma rays at decreasing dose rates (total doses of 0.53–2.70 Gy). Twenty-one days after irradiation, females were mated with non-irradiated males. Eighteen days after breeding, females were killed to examine their ovaries for the number of corpora luteae, viable embryos, and early and late embryonic deaths, in order to estimate the frequency of induced dominant lethal mutations. The estimated RBE of tritium beta particles, as calculated from the slopes of the linear curves, was 2.5. (B28) Zhou et al. (1989) published a more complete study in which they analysed the genetic effects (dominant lethal mutations on oocytes and spermatocytes; dominant skeletal mutations in spermatogonia) of tritium beta particles and gamma rays in juvenile mice. HTO was administered in a single intraperitoneal injection (exponentially decreasing dose rate). The cumulative doses of beta particles over 10 days were in the range of 0.2–1.0 Gy. Another group of mice received chronic irradiation with 60Co gamma rays for 10 days at an exponentially decreasing or a constant dose rate (total doses of 0.7–2.8 Gy). RBE of tritium beta particles, as calculated from the slopes of the dose–response curves, was in the range of 2.8–3.4 for dominant lethal mutations in oocytes, 3.5–3.9 for dominant lethal mutations in spermatogonia, and 1.6–3.9 for dominant lethal mutations in spermatocytes (Zhou et al., 1989). (B29) In summary, most of the studies to estimate RBE of tritium beta particles to reduce reproductive success have used small mammals (rodents) (Lambert, 1969; Dobson and Kwan, 1976, 1977; Carr and Nolan, 1979; Zhou et al., 1986, 1989; Chopra and Heddle, 1988; Satow et al., 1989a,b). Three studies have been performed in fish (medaka) (Etoh and Hyodo-Taguchi, 1983; Hyodo-Taguchi and Etoh, 1986, 1993) and one in an aquatic invertebrate (Ophryotrocha diadema) (Knowles and Greenwood, 1997). Most studies used tritium administered as HTO, with two studies using 3HTdR. (B30) Several endpoints related to reproductive success have been analysed: reproductive capacity and performance, testes weight loss, germ cell (female and male) survival, and dominant lethal mutations. The high radiosensitivity of primary oocytes to beta rays from HTO has been observed, but the overall impact on reproduction may be due to shortening of the reproductive phase. There is no clear correlation between the biological endpoint studied and estimates of RBE of tritium beta particles. (B31) The vast majority of studies of RBE of tritium beta particles to reduce reproductive success have used gamma rays as the reference radiation. Only two studies have compared the effects of tritium beta particles with those of x rays (Lambert, 1969; Chopra and Heddle, 1988). In all of the studies, both tritium and reference radiation were chronically administered at dose rates ranging from 2 to 1700 mGy day−1. There is no clear correlation between the dose rate used in the study and the estimated RBE. Equal numbers of studies administered the reference radiation at constant or exponentially decreasing dose rates, with no clear influence of this parameter on estimates of RBE. (B32) For reduced reproductive success, RBE of tritium beta particles (tritium administered as HTO or 3HTdR) was in the range of 1.0–3.9. Only five of 23 RBE values for tritium beta particles were >3.0 (Table B.2).
B.1.3. Data on relative biological effectiveness for morbidity effects
(B33) RBE of tritium beta particles to induce cancer in vivo has been estimated in three studies with rodents (rat and mouse). Gragtmans et al. (1984) studied the effects of tritium beta particles and x rays on induction of mammary tumours in rats. Sprague–Dawley female rats received an initial intraperitoneal injection of HTO, which was repeated four more times at 2-day intervals to maintain a constant dose rate (cumulative doses in the range of 0.49–4.10 Gy). Another group of rats was chronically irradiated with 200-kVp x rays for 10 days with total doses of 0.3–2.0 Gy. RBE of tritium beta particles was calculated from the initial slopes of the dose–response curves (best-fit linear relationship). For cumulative tumour incidence per 100 animals at risk, RBE was 1.02 ± 0.13 when all doses from exposure to HTO were included and 1.17 ± 0.18 when the highest dose of 3.85 Gy was excluded. When the endpoint considered was the cumulative percentage of animals with tumours, RBE was 0.85 ± 0.15 when all doses were included and 1.35 ± 0.13 when the dose of 3.85 Gy was excluded. When the endpoint analysed was the time required to develop mammary tumours in 50% of the animals at risk, RBE was 0.67 ± 0.13 when all doses were included and 1.12 ± 0.18 when the dose of 3.85 Gy was excluded. None of the RBE values were statistically different from 1.0. (B34) Johnson et al. (1995) studied the effectiveness of tritium beta particles and x rays to induce myeloid leukaemia in the mouse. CBA/H mice received a single intraperitoneal injection of HTO (cumulative beta doses of 0.85–3.04 Gy). Another group was chronically irradiated with 150–200-kVp x rays for 10 days at dose rates of 0.24–0.72 Gy day−1 (total doses of 1.06–2.64 Gy). RBE of tritium beta particles was calculated considering different fits to the dose response for the incidence of myeloid leukaemia per 104 mouse-days at risk, with values ranging from 1.1 to 1.24. The best estimate gave RBE of 1.2 ± 0.3. (B35) The effects of tritium beta particles and 137Cs gamma rays on tumour development in different organs have been studied in mice (Seyama et al., 1991). Adult female mice (C57BL/6 N and BCF1) received a single intraperitoneal injection of HTO (cumulative beta doses of 0.27 or 2.7 Gy). Another group of mice was chronically irradiated with 137Cs gamma rays (total dose of 0.27 or 2.7 Gy administered at 0.08 and 0.76 Gy day−1, respectively). RBE of tritium beta particles, as calculated from the data on tumour incidence at 500 days after exposure to 2.7 Gy, was 2.5. (B36) RBE of tritium beta particles in causing splenic and thymic atrophy was studied in adult female mice (CF1) using radium gamma rays as the reference radiation (Storer et al., 1957). Mice received a single intraperitoneal injection of HTO followed by administration of HTO in drinking water in order to maintain a constant tritium concentration over 5 days (cumulative doses of 1.25–3.50 Gy). The exposure to gamma rays took place for 5 days at dose rates similar to those from exposure to HTO (total doses of 1.25–3.5 Gy). For splenic atrophy, RBE of tritium beta particles was 1.32 ± 0.12, and for thymic atrophy, RBE was 1.52 ± 0.15. The authors also studied the capacity of tritium beta particles, compared with 60Co gamma rays, to reduce 59Fe uptake by red blood cells in adult rats (Sprague–Dawley) at the same dose rates and doses as used in the experiments described above. RBE of tritium beta particles for 59Fe uptake by red blood cells was 1.64 ± 0.05. (B37) Ijiri (1989) studied RBE of tritium beta particles for cell death (apoptosis) in the crypts of adult male mice (C57Bl/6). HTO was injected intraperitoneally at concentrations giving dose rates in the range of 0.001–1.164 Gy day−1 (cumulative doses up to 2.0 Gy). Another group of mice was chronically irradiated with 137Cs gamma rays at dose rates of 0.014–11.52 Gy day−1 (total doses up to 2.9 Gy). Using estimates of the maximum number of apoptotic cells per crypt section, calculated as the mean of the data obtained at the three highest doses, RBE of tritium beta particles was calculated for the small intestine (1.6 ± 0.2) and descending colon (1.4 ± 0.1). RBE was also calculated from D0 values (doses that reduce the survival fraction to 37%) obtained from the corresponding beta particle and gamma ray dose–response curves, with values of 2.0 ± 0.2 for the small intestine and 1.8 ± 0.2 for the descending colon. (B38) RBE of tritium beta particles for cell survival in vitro has been estimated in experiments using transformed cell lines. Ueno et al. (1982) studied the effects of tritium beta particles, with tritium administered as HTO, and 60Co gamma rays on L5178Y cell survival. HTO was added to the culture medium at a concentration of 22.2–166.5 MBq mL−1 (total doses up to approximately 11 Gy). Another cell line sample was exposed to 60Co gamma radiation over a period of 4.5–100 h at dose rates of 2.9–11.5 Gy day−1 (total doses up to 11.0 Gy). RBE of tritium beta particles at 50% survival was 1.4 when linear models were used to fit the survival curves, and 1.6 using linear-quadratic models. (B39) Bedford et al. (1975) used a murine leukaemic cell line (L5178Y) and a Chinese hamster cell line (V79B) in their cell-survival studies. The cell lines were exposed to HTO or tritiated thymidine (3HTdR) at cumulative beta doses of 1.0–16.0 Gy (dose rate of 4.8 Gy day−1). The reference radiation was 60Co gamma rays at the same dose rate and total dose. The irradiations were carried out with cells held in the frozen state (to prevent cell division) or at 5℃. For 3HTdR, RBE values for tritium beta particles for L5178Y and V79B cell survival (irradiated in the frozen state) were 3.0 and 4.4, respectively. However, the authors noted uncertainty in the dose calculations; 3HTdR is incorporated into DNA and average cell dose will underestimate effects. For V79B cells irradiated at 5℃, RBE was 1.7–1.9 for the two forms of administered tritium. (B40) In summary, RBE of tritium beta particles to produce morbidity effects when tritium was administered as HTO has been studied in small mammal systems alone, either in vivo (mouse and rat) or in vitro (transformed cell lines such as murine lymphocytic leukaemia, L5178Y, or Chinese hamster V79B). Only two studies have used x rays as the reference radiation (Gragtmans et al., 1984; Johnson et al., 1995). 137Cs, 60Co, or Ra gamma rays were used as the reference radiation in the remaining studies. (B41) Several endpoints related to morbidity have been analysed in studies of RBE of tritium beta particles, including tumour induction (mammary tumours, myeloid leukaemia) (Gragtmans et al., 1984; Seyama et al., 1991; Johnson et al., 1995), tissue damage in experimental animals (splenic and thymic atrophy, descending colon and intestine cell survival, depression of 59Fe uptake) (Storer et al., 1957; Ijiri, 1989), and cell survival in transformed cell lines (Bedford et al., 1975; Ueno et al., 1982). (B42) RBE values for tritium beta particles to produce morbidity effects, when tritium was administered as HTO, were in the range of 1.0–2.5. Most RBE values were <2.0 (10 values out of 12) (Table B.3). One study using 3HTdR administered to cell lines suggested RBE values in the range of 1.7–4.4, depending on the temperature at which the cell line was irradiated and on the cell type used in the study (Table B.3).
B.1.4. Data on relative biological effectiveness for chromosomal damage and mutations
(B43) Using Drosophila germ cells, Byrne and Lee (1989) estimated RBE of tritium beta particles to induce mutations. Adult Drosophila males were exposed over 48 h to HTO or 60Co gamma rays at total doses of 6.4–25.5 Gy. In the different experimental groups, sex-linked recessive lethal mutations in germ cells were analysed. RBE of tritium beta particles, as calculated from the slopes of the dose–response curves for gene mutations transmitted to successive generations, was 2.7 ± 0.3. (B44) Matsuda et al. (1986) studied the efficacy of tritium beta particles to induce chromosomal aberrations in fertilised mice eggs compared with gamma radiation. The fertilised eggs in early pronuclear stage were treated in vitro with HTO for 2 h at dose rates of 1.02–4.08 Gy day−1 and total doses of 0.085–0.34 Gy or exposed for 2 h to 60Co gamma radiation at dose rates of 0.62–3.54 Gy day−1 and total doses of 0.05–0.30 Gy. The results showed that the dose–response curves for tritium beta particles and 60Co at doses >0.05 Gy were approximately linear. Thus, linear regression coefficients from fits at those doses were used to calculate RBE of tritium beta particles of 2.0. Using the results on chromosomal aberration frequency in fertilised mouse eggs exposed to acute doses of x rays as the reference radiation (results obtained by this group in previous studies), RBE of 1.6 was calculated (Matsuda et al., 1983, 1985a,b). (B45) Two groups have studied RBE of tritium beta particles for induction of chromosomal aberrations in murine spermatocytes. Zhou et al. (1989) studied the induction of chromosomal aberrations in juvenile mice spermatocytes. Mice received a single intraperitoneal injection of HTO, followed by tritium administration in drinking water to keep the dose rate constant. Cumulative doses of beta radiation were in the range of 0.2–1.0 Gy (dose rates of 0.005–0.05 Gy day−1). Another group of mice received chronic irradiation with 60Co gamma rays for 10 days at a constant dose rate (total doses of 0.43–2.04 Gy administered at dose rates of 0.04–0.20 mGy day−1). RBE values of 2.9–3.8 were calculated. (B46) Chopra and Heddle (1988) analysed RBE of tritium beta particles to produce chromosomal aberrations in murine primary spermatocytes and peripheral blood lymphocytes. Mice (CBA/H) received a single intraperitoneal injection of HTO or were irradiated with 250-kVp x rays for 10 days at total doses of beta radiation and x rays of 1.5–6.0 Gy. Dose–response curves for different types of chromosomal aberrations were generated and RBE was calculated from their slopes. RBE of tritium beta particles to induce chromosome translocations in primary spermatocytes was 1.21 [95% confidence interval (CI) 0.8–1.9]. RBE for induction of dicentric and centric rings in primary spermatocytes was 1.26. RBE for induction of chromosomal aberrations in peripheral blood lymphocytes was 1.14 (95% CI 0.8–1.5). The authors concluded that the different RBE values were not statistically different from 1.0. (B47) RBE of tritium beta particles to induce chromosomal aberrations in human spermatozoa was studied by Kamiguchi et al. (1990a,b). The sperm samples were treated in vitro with HTO (57–900 MBq mL−1) for 80 min. The authors argued that as it was difficult to accurately determine the absorbed dose received by the spermatozoa, doses were expressed as a range between the estimated minimum dose and the estimated maximum dose. Minimum and maximum doses were estimated to be in the range of 0.14–2.06 Gy and 0.25–3.74 Gy, respectively. Dose rates were not calculated. Other sperm samples were irradiated in vitro with 220-kVp x rays at a dose rate of 628 Gy day−1 and total doses of 0.23–1.82 Gy. After irradiation, both samples were analysed for chromosomally abnormal spermatozoa and for different types of aberrations (breakages, exchanges, chromosome and chromatid type). RBE values for tritium beta particles for the different endpoints were in the range of 1.89–3.00 (minimum) and 1.04–1.65 (maximum). The authors considered that the maximum dose estimates were more reliable (Kamiguchi et al., 1990a,b). (B48) Kozlowski et al. (2001) assessed the capacity of tritium beta particles and x rays to induce chromosomal aberrations in bone marrow cells of mice exposed in utero. Pregnant mice were treated with tritium either in drinking water or in cress, from day 1 post conception until parturition on day 20. After ingestion of HTO or tritiated cress, the accumulated doses during pregnancy were estimated to be 0.6 and 0.3 Gy, respectively. The estimated cumulative doses over the 4 weeks after birth were 0.1 Gy for both HTO and tritiated cress. Another group of female pregnant mice was irradiated acutely with 250-kVp x rays on day 7 or 14 of pregnancy at a total dose of 0.5 Gy. Chromosomal aberrations were quantified in bone marrow cells of the mothers and offspring of each experimental group. Similar levels of stable chromosomal aberrations were quantified in bone marrow of the mothers and their offspring in the three irradiated groups (HTO, tritiated cress, and x rays). The authors did not calculate RBE values for tritium beta particles, but they stated that the results were consistent with RBE in the range of 1.0–2.0. (B49) Several groups have studied RBE of tritium beta particles for induction of chromosomal aberrations in human peripheral blood lymphocytes in vitro. Bocian et al. (1978) treated blood samples with HTO for a period of 2 h at dose rates of 3.36–30.48 Gy day−1 and cumulative doses of 0.28–2.55 Gy, or irradiated them acutely with 180-kVp x rays at a dose rate of 2736 Gy day−1 and total doses of 0.5–3.0 Gy. From the dose–response curves for chromosomal aberration frequency (dicentric and centric rings) in peripheral lymphocytes after acute exposure, RBE of tritium beta particles of 1.17 ± 0.02 was calculated. In another study by Vulpis (1984), the peripheral blood samples were exposed to HTO for 20–150 min at estimated dose rates of 18.14–66.53 Gy day−1 and accumulated doses of 0.25–7.0 Gy, and the number of dicentric aberrations in lymphocytes was quantified. To calculate RBE of tritium beta particles, those investigators used the data obtained in the same laboratory, under the same conditions, for blood samples exposed acutely to 250-kVp x rays at total doses of 0.4–9 Gy. RBE was calculated from the ratio of alpha coefficients obtained by fitting the aberration yield curves with a linear-quadratic dose response. RBE of 2.6 was calculated at a dose of 0.25 Gy. RBE decreased with increasing dose, with RBE of 1.1 calculated at 7.0 Gy. (B50) Tanaka et al. (1994) studied the production of chromosomal aberrations in human peripheral blood lymphocytes and human bone marrow cells by tritium beta particles. The peripheral blood and bone marrow samples were treated with HTO at a beta dose rate of 4.8 Gy day−1 and total dose of 0.13–1.11 Gy, or irradiated with 60Co or 137Cs gamma rays at a dose rate of 28.8 Gy day−1 and total dose of 0.25–2.0 Gy for 60Co and a dose rate of 0.29 Gy day−1 and total dose of 2.0 Gy for 137Cs. In human peripheral blood lymphocytes, RBE values for tritium beta particles for induction of chromosomal aberrations and dicentric aberrations were 2.2–2.7 and 2.1–2.3, respectively, when 60Co rays were the reference radiation. RBE for induction of chromosomal aberrations was 2.0 when 137Cs gamma rays were the reference radiation. In human bone marrow cells, RBE values for induction of chromosomal aberrations and chromatid aberrations were 1.13 and 3.10, respectively, when 60Co gamma rays were the reference radiation. (B51) Dewey et al. (1965) exposed a Chinese hamster cell line to HTO or 3HTdR for a period of 10 h; dose rates and doses were not reported. Other cell samples were irradiated with 60Co gamma rays over the same period at dose rates of 3.5–20.7 Gy day−1 and total doses of 1.47–8.65 Gy. In each group, the incidence of chromosomal aberrations was quantified. RBE of tritium beta particles was calculated from the doses needed to produce two visible aberrations per cell (8.2 Gy for 3HTdR, 4.9 Gy for HTO, and 5.2 Gy for 60Co gamma rays), giving estimated RBE values of 1.06 for exposure to HTO and 1.0 for exposure to 3HTdR. (B52) Ueno et al. (1982) studied RBE of tritium beta particles, with tritium administered as HTO, to induce mutations and micronuclei in the murine lymphocytic leukaemia cell line L5178Y using 60Co gamma rays as the reference radiation. In the mutation studies, the cell lines were exposed to cumulative doses of tritium beta particles of 1.5–5.0 Gy at dose rates of 2.0–6.0 Gy day−1, or irradiated with 60Co at total doses of 2.0–6.0 Gy and dose rates of 2.40–7.20 Gy day−1. In the studies of micronuclei, the cell line was exposed to total doses of 1.0–8.0 Gy for tritium beta radiation or 2.0–9.0 Gy for 60Co gamma rays at dose rates of 1.2–9.6 Gy day−1 and 2.40–10.80 Gy day−1 for beta and gamma radiation, respectively. RBE of tritium beta particles of 1.8 for mutation induction was estimated. From the doses needed to produce 25 and 50 micronuclei per 1000 cells, RBE values of 2.3 and 1.8, respectively, were calculated (Ueno et al., 1982). (B53) In summary, the majority of studies of RBE of tritium beta particles for chromosomal damage and mutations have been performed in vitro with mammalian cells and tritium administered as HTO. One study performed with a Chinese hamster cell line used 3HTdR. The experimental systems used included fertilised mouse eggs (Matsuda et al., 1986), human cell samples (bone marrow, peripheral blood lymphocytes, sperm) (Bocian et al., 1978; Vulpis, 1984; Kamiguchi et al., 1990b; Tanaka et al., 1994), and cell lines (Chinese hamster and murine lymphocytic leukaemia) (Dewey et al., 1965; Ueno et al., 1982). One in-vitro study was performed in Drosophila (Byrne and Lee, 1989). Three in-vivo studies on chromosomal damage were performed using mice (Chopra and Heddle, 1988; Zhou et al., 1989; Kozlowski et al., 2001). (B54) The other endpoint studies were mutations and micronuclei in the murine lymphocytic leukaemia cell line L5178Y (Ueno et al., 1982) and sex-linked recessive lethal mutations in Drosophila (Byrne and Lee, 1989). (B55) All studies but two (Matsuda et al., 1986; Kozlowski et al., 2001) used tritium beta doses >1 Gy administered at constant dose rates over a range of 0.005–66.50 Gy day−1. More studies have used gamma rays (10 of 16) than x rays (six of 16) as the reference radiation. (B56) The estimates of RBE values for tritium beta particles to produce chromosomal damage and mutations varied from 1.0 to 3.8. Only two RBE estimates were >3.0 (eight values in the range of 1.0–1.9, six values in the range of 2.0–2.9) (Table B.4).
B.2. Other literature reviews of relative biological effectiveness of tritium beta particles
(B57) Several reviews of RBE values for tritium beta particles have been published in the last two decades. Most of these reviews were concerned with assessing health risks in humans. Thus, the more relevant endpoints were considered to be those directly related to induction of cancer and heritable effects at low doses and low dose rates (stochastic effects). The information analysed in each of these reviews has been almost the same: estimates of tritium in studies performed in vivo or in vitro, using either orthovoltage x rays or gamma radiation as the reference radiation. Thus, as would be expected, the conclusions of the different reviews are quite similar (Table B.5). There were some minor differences in the criteria used to select the information for review. Some authors only reviewed papers published in peer-reviewed journals (Little and Lambert, 2008), while others also considered data published in conference proceedings (Fairlie, 2007). (B58) The only published review in which non-human biota were the primary focus was from Environment Canada and Health Canada (2003). The data on RBE of tritium beta particles were analysed with emphasis on those effects related to loss of reproductive capacity due to their relevance for maintenance of populations. The aim of that review was to estimate what the investigators called an ‘ecodosimetry weighting factor’ to be applied to tritium beta particles to calculate ‘equivalent doses’ to biota. Environment Canada and Health Canada assumed that orthovoltage x rays were two times more effective in inducing biological effects than gamma radiation. Thus, the estimated RBE values when x rays were used as the reference radiation were multiplied by 2 to compare them with the estimated RBE values using gamma rays as the reference radiation. For impairment of reproductive capacity, estimated RBE values for tritium beta particles ranged from 1.7 to 3.8, while estimates for genetic endpoints were in the range of 1.5–2.9. The authors concluded that the majority of RBE values are in the range of 2.0–3.0, with a few values for ecologically relevant endpoints as high as 3.8. Accordingly, Environment Canada and Health Canada (2003) proposed an ecodosimetry weighting factor of 3.0 for calculating an equivalent dose from exposure to tritium when high-energy gamma radiation is used as the reference radiation. (B59) A more recent review was published by the Canadian Nuclear Safety Commission (CNSC, 2010). In that publication, a detailed description of each of the previously published reviews was presented, together with an analysis of the experimental studies to estimate RBE of tritium beta particles that were available in the literature. The publication focused on analysing the possibility of choosing an appropriate single value of RBE that could be applied for human radiological protection purposes. (B60) UNSCEAR (2016) Annex C provides a review of scientific information on the characteristics of tritium, including various physical and chemical forms, biokinetic models, dosimetry, and radiobiological effects associated with human exposure to tritium. UNSCEAR notes that over several decades, tens of experiments have been conducted using mammals (mostly mice) and their cells to determine RBE of tritium under various experimental conditions and considering a range of biological endpoints. UNSCEAR’s evaluation concludes from some 50 different experiments that RBE values for tritium beta in animals or animal cells have been reported as ranging from 1.0 to 5.0 (centred around 2.0–2.5) and from 0.4 to 8.0 (centred around 1.5–2) with gamma rays and orthovoltage x rays as the reference radiation, respectively. UNSCEAR also notes that there is tendency for RBE values to increase with decreasing dose, and that RBE values for stochastic effects are generally higher (centred around 2.5–3.0 compared with prolonged gamma radiation) than those obtained from studies of tissue reactions (cell killing in vivo and in vitro).
Data on relative biological effectiveness (RBE) of tritium beta particles for reduced reproductive success.
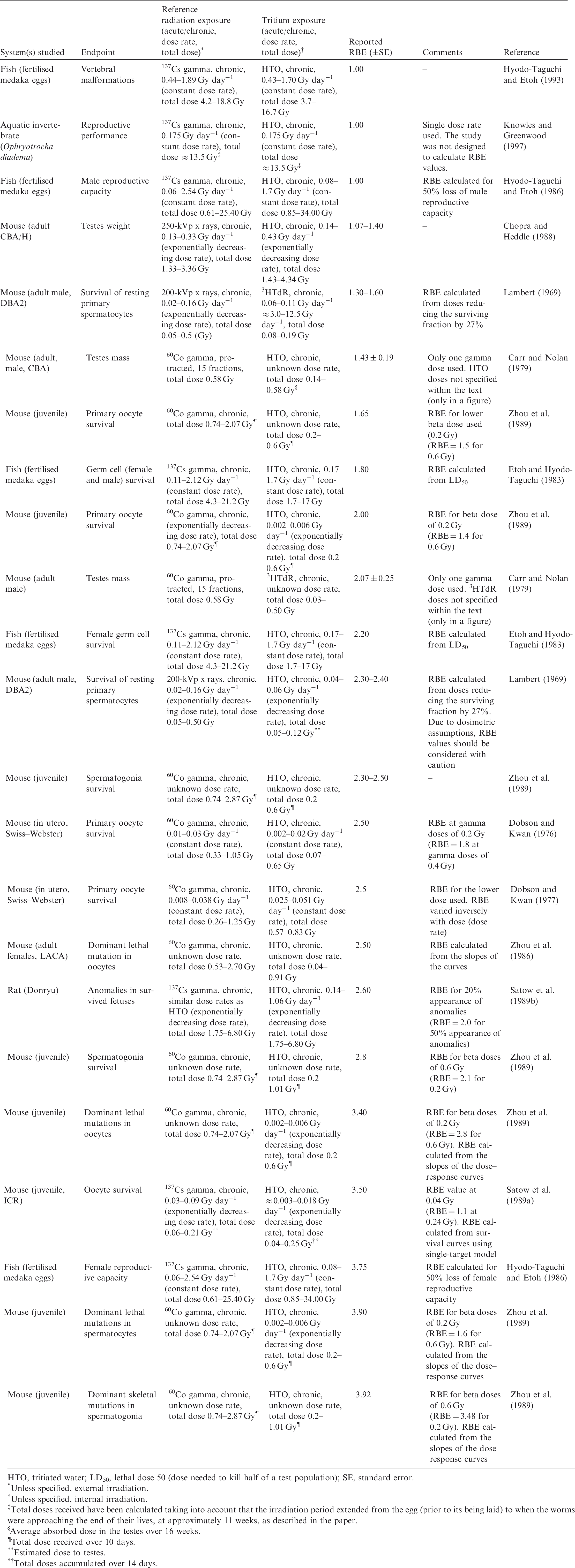
HTO, tritiated water; LD50, lethal dose 50 (dose needed to kill half of a test population); SE, standard error.
Unless specified, external irradiation.
Unless specified, internal irradiation.
Total doses received have been calculated taking into account that the irradiation period extended from the egg (prior to its being laid) to when the worms were approaching the end of their lives, at approximately 11 weeks, as described in the paper.
Average absorbed dose in the testes over 16 weeks.
Total dose received over 10 days.
Estimated dose to testes.
Total doses accumulated over 14 days.
B.3. Overall evaluation of relative biological effectiveness of tritium beta particles
(B61) RBE values for tritium beta particles in the available studies were all obtained at dose rates that were in or above the relevant DCRL bands. The calculated values were all <4, with the majority of values <3. The data for fish are from 1 to nearly 4, with aquatic invertebrates around 1; these data are consistent with the data observed for mammals.
Data on relative biological effectiveness (RBE) of tritium beta particles for morbidity effects.
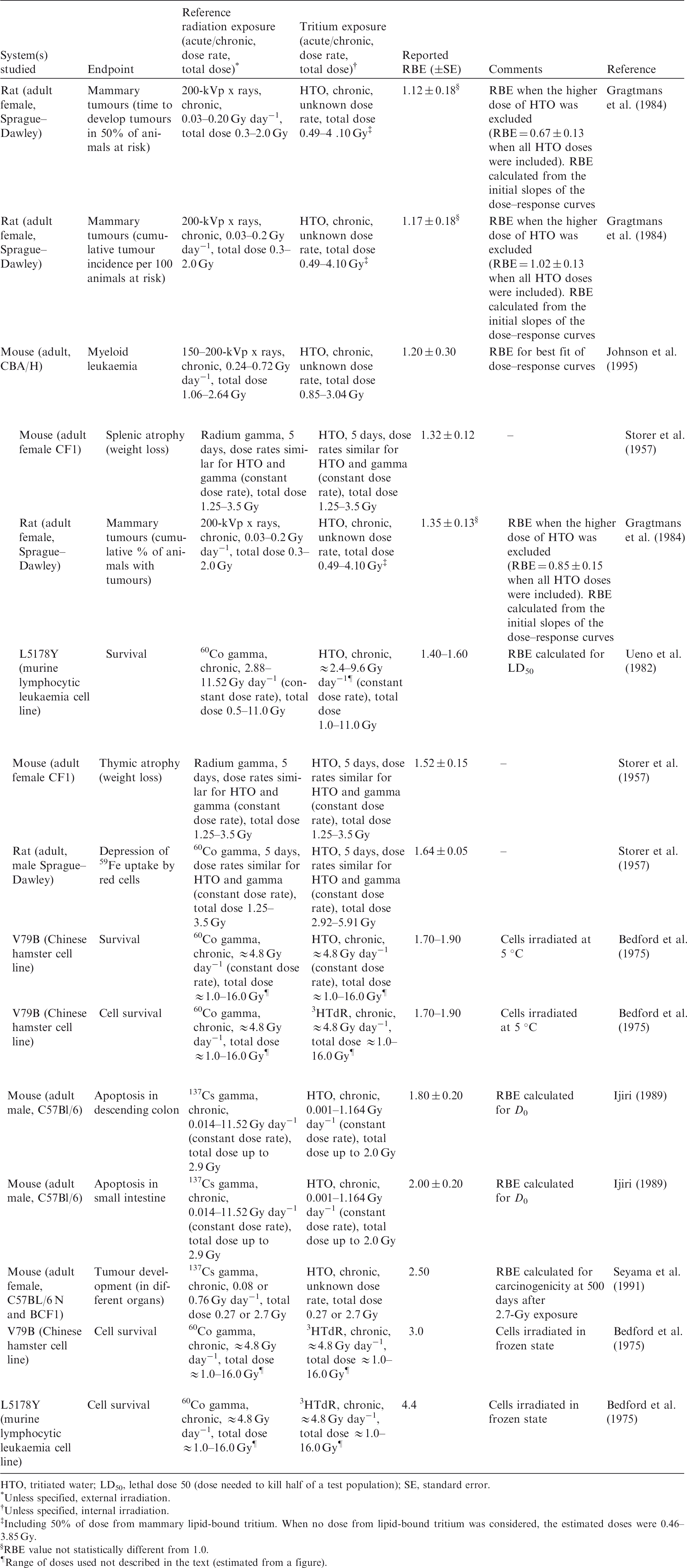
HTO, tritiated water; LD50, lethal dose 50 (dose needed to kill half of a test population); SE, standard error.
Unless specified, external irradiation.
Unless specified, internal irradiation.
Including 50% of dose from mammary lipid-bound tritium. When no dose from lipid-bound tritium was considered, the estimated doses were 0.46–3.85 Gy.
RBE value not statistically different from 1.0.
Range of doses used not described in the text (estimated from a figure).
B.4. References
Bedford, J.S., Mitchell, J.B., Griggs, H.G., et al., 1975. Cell killing by gamma rays and beta particles from tritiated water and incorporated tritiated thymidine. Radiat. Res. 63, 531–543.
Bocian, E., Ziemba-Zak, E.B., Rosiek, O., et al., 1978. Chromosome aberrations in human lymphocytes exposed to tritiated water in vitro. Curr. Top. Radiat. Res. Q. 12, 168–181.
Boyer, C., Vichot, L., Fromm, M., et al., 2009. Tritium in plants: a review of current knowledge. Environ. Exper. Bot. 67, 34–51.
Byrne, B.J., Lee, W.R., 1989. Relative biological effectiveness of tritiated water to γ radiation for germ line mutations. Radiat. Res. 117, 469–479.
Carr, T.E.F., Nolan, J., 1979. Testis mass loss in the mouse induced by tritiated thymidine, tritiated water, and 60Co gamma irradiation. Health Phys. 36, 135–145.
CNSC, 2010. Health Effects, Dosimetry and Radiological Protection of Tritium. INFO-0799. Canadian Nuclear Safety Commission, Ottawa.
Chopra, C., Heddle, J.A., 1988. Cytogenetic Measurements of the Relative Biological Effectiveness of Tritium. INFO-0287. Atomic Energy Control Board, Ottawa.
Dewey, W.C., Humphrey R.M., Jones B.A., 1965. Comparisons of tritiated thymidine, tritiated water, and cobalt-60 gamma rays in inducing chromosomal aberrations. Radiat. Res. 24, 214–238.
Dobson, R.L., Kwan, T.C., 1976. The RBE of tritium radiation measured in mouse oocytes: increase at low exposure levels. Radiat. Res. 66, 615–625.
Dobson, R.L., Kwan, T.C., 1977. The tritium RBE at low-level exposure – variation with dose, dose rate, and exposure duration. Curr. Top. Radiat. Res. Q. 12, 44–62.
Environment Canada, Health Canada, 2003. Priority Substances List Assessment Report. Releases of Radionuclides from Nuclear Facilities (Impact on Non-Human Biota). PSL2. Environment Canada and Health Canada, Ottawa.
Etoh, H., Hyodo-Taguchi, Y., 1983. Effects of tritiated water on germ cells in medaka embryos. Radiat. Res. 93, 332–339.
Fairlie, I., 2007. RBE and wR values of Auger emitters and low-range beta emitters with particular reference to tritium. J. Radiol. Protect. 27, 157–168.
Furchner, J.E., 1957. Relative biological effectiveness of tritium beta-particles and Co-60 gamma-rays measured by lethality in CF1 mice. Radiat. Res. 6, 483–490.
Gragtmans, N.J., Myers, D.K., Johnson, J.R., et al., 1984. Occurrence of mammary tumors in rats after exposure to tritium beta rays and 200-kVp X-rays. Radiat. Res. 99, 636–650.
HPA, 2007. Review of Risks from Tritium with Particular Attention to Tritiated Water and Organic Compounds Containing Tritium. Report of AGIR Subgroup on Tritium Risks, UK Advisory Group on Ionizing Radiation. Health Protection Agency, London. Available at: http://www.hpa.org.uk/radiation/advisory_groups/agir/index.htm.
Hyodo-Taguchi, Y., Etoh, H., 1986. Effects of tritiated water on germ cells in medaka. II. Diminished reproductive capacity following embryo exposure. Radiat. Res. 106, 321–330.
Hyodo-Taguchi, Y., Etoh, H., 1993. Vertebral malformations in medaka (Teleost fish) after exposure to tritiated water in the embryonic stage. Radiat. Res. 135, 400–404.
ICRP, 1989. Age-dependent doses to members of the public from intake of radionuclides: Part 1. ICRP Publication 56. Ann. ICRP 20(2).
ICRP, 1993. Age-dependent doses to members of the public from intake of radionuclides: Part 2. ICRP Publication 67. Ann. ICRP 23(3/4).
ICRP, 1994. Human respiratory tract model for radiological protection. ICRP Publication 66. 24(1–3).
ICRP, 1995. Age-dependent doses to members of the public from intakes of radionuclides: Part 4. Inhalation dose coefficients. ICRP Publication 71. Ann. ICRP 25(3/4).
ICRP, 1996. Age-dependent doses to members of the public from intake of radionuclides: Part 5. ICRP Publication 72. Ann. ICRP 26(1).
ICRP, 2008. Environmental protection: the concept and use of Reference Animals and Plants. ICRP Publication 108. Ann. ICRP 38(4–6).
ICRU, 1970. Linear Energy Transfer. ICRU Report 16. International Commission on Radiation Units and Measurements, Bethesda, MA.
Ijiri, K., 1989. Cell death (apoptosis) in mouse intestine after continuous irradiation with gamma rays and with beta rays from tritiated water. Radiat. Res. 118, 180–191.
Johnson, J.R., Myers, D.K., Jackson, J.S., et al., 1995. Relative biological effectiveness of tritium for induction of myeloid leukemia in CBA/H mice. Radiat. Res. 144, 82–89.
Kamiguchi, Y., Tateno, H., Mikamo, K., 1990a. Types of structural chromosome aberrations and their incidences in human spermatozoa X-irradiated in vitro. Mutat. Res. 228, 133–140.
Kamiguchi, Y., Tateno, H., Mikamo, K., 1990b. Dose–response relationship for the induction of structural chromosome aberrations in human spermatozoa after in vitro exposure to tritium beta rays. Mutat. Res. 228, 125–131.
Knowles, J.F., Greenwood, L.N., 1997. A comparison of the effects of long-term beta and gamma irradiation on the reproductive performance of a marine invertebrate Ophryotrocha diadema (Polychaeta Dorvilleidae). J. Environ. Radioact. 34, 1–7.
Kozlowski, R., Bouffler, S.D., Haines, J.W., et al., 2001. In utero haemopoietic sensitivity to alpha, beta or X irradiation in CBA/H mice. Int. J. Radiat. Biol. 77, 805–815.
Lambert, B.E., 1969. Cytological damage produced in the mouse testes by tritiated thymidine, tritiated water and X-rays. Health Phys. 17, 547–557.
Little, M.P., Lambert, B.E., 2008. Systematic review of experimental studies of relative biological effectiveness of tritium. Radiat. Environ. Biophys. 47, 71–73.
Matsuda, Y., Yamada, T., Tobaria, I., et al., 1983. Preliminary study on chromosomal aberrations in eggs of mice fertilized in vitro after X-irradiation. Mutat. Res. 121, 125–130.
Matsuda, Y., Tobari, I., Yamada, T., 1985a. Studies on chromosome aberrations in the eggs of mice fertilized in vitro after irradiation. I. Chromosome aberrations induced in sperm after X-irradiation. Mutat. Res. 148, 113–117.
Matsuda, Y., Tobari, I., Yamada, T., 1985b. Studies on chromosome aberrations in the eggs of mice fertilized in vitro after irradiation. II. Chromosome aberrations induced in mature oocytes and fertilized eggs at the pronuclear stage following X-irradiation. Mutat. Res. 151, 275–280.
Matsuda, Y., Yamada, T., Tobari, I., 1986. Chromosomal aberrations induced by tritiated water or 60Co gamma rays at early pronuclear stage in mouse eggs. Mutat. Res. 160, 87–93.
NCRP, 1979. Tritium and Other Radionuclide Labelled Organic Compounds Incorporated in Genetic Material. NCRP Report No. 63. National Council on Radiation Protection and Measurements, Washington, DC.
Nikjoo, H., Goodhead, D.T., 1991. Track structure analysis illustrating the prominent role of low energy electrons in radiobiological effects of low-LET radiations. Phys. Med. Biol. 36, 229–238.
Satow, Y., Hori, H., Lee, J.Y., et al., 1989a. Effect of tritiated water on female germ cells: mouse oocyte killing and RBE. Int. J. Radiat. Biol. 56, 283–299.
Satow, Y., Hori, H., Lee, J.Y., 1989b. Teratogenic effect of fission neutron and tritium water on rat embryo. J. UOEH 11(Suppl.), 416–431.
Seyama, T., Yamamoto, O., Kinomura, A., et al., 1991. Carcinogenic effects of tritiated water (HTO) in mice: in comparison to those of neutrons and gamma-rays. J. Radiat. Res. 32 (Suppl. 2), 132–142.
Spalding, J.F., Langham, W., Anderson, E.C., 1956. The relative biological effectiveness of tritium p-radiation with the broad bean root (Vicia faba) as a test system. Radiat. Res. 4, 221–227.
Storer, J.B., Harris, P.S., Furchner, J.E., et al., 1957. The relative biological effectiveness of various ionizing radiations in mammalian systems. Radiat. Res. 6, 188–288.
Straume, T., Carsten, A.L., 1993. Tritium radiobiology and relative biological effectiveness. Health Phys. 65, 657–672.
Tanaka, K., Sawada, S., Kamada, N., 1994. Relative biological effectiveness and dose rate effect of tritiated water on chromosomes in human lymphocytes and bone marrow cells. Mutat. Res. 323, 53–61.
Ueno, A.M., Furuno-Fukushi, I., Matsudaira, H., 1982. Induction of cell killing, micronuclei, and mutation to 6-thioguanine resistance after exposure to low-dose-rate gamma rays and tritiated water in cultured mammalian cells (L5178Y). Radiat. Res. 91, 447–456.
UNSCEAR, 2016. Sources, Effects and Risks of Ionizing Radiation. Annex C. Biological Effects of Selected Internal Emitters – Tritium. United Nations Scientific Committee on the Effects of Atomic Radiation, New York.
Vulpis, N., 1984. The induction of chromosome aberrations in human lymphocytes by in vitro irradiation with b particles from tritiated water. Radiat. Res. 97, 511–518.
Yamada, T., Yukawa, O., Asami, K., et al., 1982. Effect of chronic HTO beta or 60Co gamma radiation on preimplantation mouse development in vitro. Radiat. Res. 92, 359–369.
Zhou, X-Y., Dong, J-C., Geng, X-S., et al., 1986. Tritium beta ray and 60Co gamma-ray caused dominant lethal mutation in mice. Chin. Med. J. 99, 420–423.
Zhou, X-Y., Dong, J-C., Zhou, S-Y., et al., 1989. Experimental study on relative biological effectiveness of tritium and risk estimates of genetic damage. Chin. Med. J. 102, 872–876.
Data on relative biological effectiveness (RBE) of tritium beta particles for chromosomal damage and mutations.
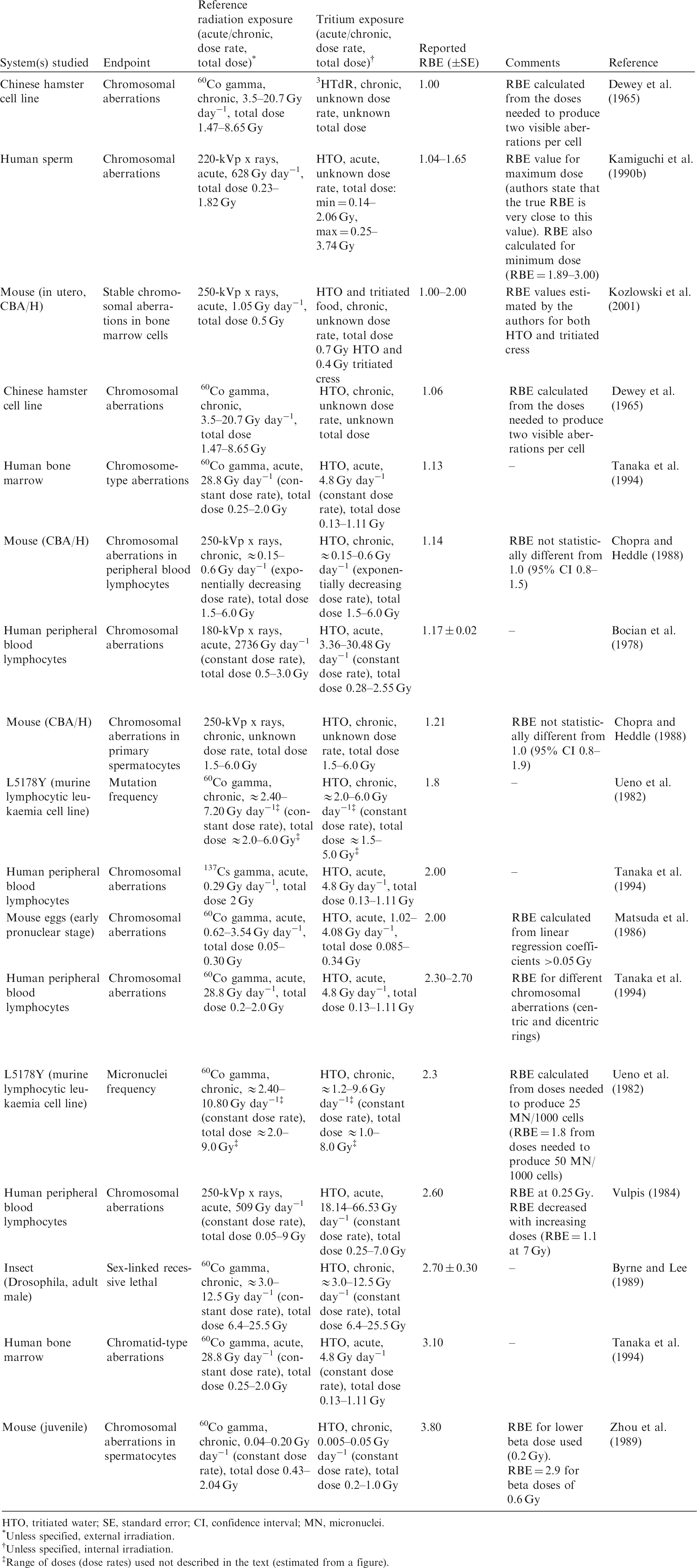
HTO, tritiated water; SE, standard error; CI, confidence interval; MN, micronuclei.
Unless specified, external irradiation.
Unless specified, internal irradiation.
Range of doses (dose rates) used not described in the text (estimated from a figure).
Summary of conclusions in several published reviews on relative biological effectiveness (RBE) values for tritium beta particles.

CI, confidence interval.
ANNEX C. RELATIVE BIOLOGICAL EFFECTIVENESS of ALPHA-EMITTING RADIONUCLIDES
(C1) An alpha particle has two protons and two neutrons, and is identical to the nucleus of a 4He atom. Alpha particles are positively charged (+2) particles with energy of approximately 4–9 MeV that are emitted by certain radionuclides in response to a low neutron-to-proton ratio in the nucleus. Radioactive isotopes that emit alpha particles include, for example, 241Am, 239Pu, and several radionuclides in the natural uranium and thorium decay chains, such as 238U, 226Ra, 222Rn, 210Po, and 232Th. (C2) Alpha particles have very low penetrative power in matter compared with gamma rays, and are primarily injurious when alpha-emitting radionuclides are deposited internally, although higher-energy alpha particles can penetrate the outer layer of dead skin and irradiate the underlying basal layer. The dosimetry of alpha emitters is complicated by a number of factors, including their non-uniform distribution in organs and tissues, and the short range of alpha particles, which can result in non-uniform localised doses to cells. (C3) In general terms, radiation quality can be specified by the fluence spectrum of the ionising particles of different charge and velocity that deposit energy in the system considered, which, in turn, determines the number of ionisations within the affected volume. To illustrate, Goodhead (1992) notes that a secondary electron track produced by an incident gamma ray that traverses the nucleus of a mammalian cell with a diameter of approximately 8 µm gives rise to 60–80 ionisations, resulting in an absorbed dose of the order of 1 mGy, whereas an alpha particle traversing the same nucleus results in some 23,000 ionisations within the same volume and an absorbed dose of the order of 400 mGy. (C4) Such differences in ionisation density or LET are the basis of observed differences in effectiveness per Gy in causing deleterious effects in cells and tissues. In general, alpha particles and neutrons can produce observable damage at much lower average absorbed doses than beta or gamma radiation.
C.1. Review of experimental studies of relative biological effectiveness of alpha-emitting radionuclides
(C5) Several criteria were used to evaluate the literature describing experiments related to RBE of alpha particles, including life stage, endpoints, LET, dose rates, total dose, and reference radiation. The papers reviewed varied greatly in the level of detail provided. (C6) Most of the reviewed papers either reported RBE values or provided sufficient data, such as the coefficients α and β linear-quadratic exposure-effect models or survival curves (see Annex A), that could be used to calculate RBE values. The reported or calculated RBE values for alpha particles were obtained in studies in which x rays, high-energy gamma rays, or, in some cases, higher-energy beta particles were used as the reference radiation. Maximum RBE (RBEm or RBEM) values were calculated where data were available, as, for example, from the slopes of survival curves provided in the reviewed papers. (C7) RBE values differ between experiments due to a number of factors, including differences in species and strains, cell lines, genetic modifications, and dose rate. It should be noted that the route of administration in many of the experimental studies was injection, whereas intakes in the natural environment will arise via ingestion or inhalation. The following sections provide a brief overview of available data from experimental studies of RBE of alpha-emitting radionuclides and fission neutrons of relevant LET (Tables C.1–C.4). Summary of publications studying relative biological effectiveness (RBE) of alpha particles to produce early mortality. RBEm, maximum value of RBE derived from tissue reaction data; RBEM, maximum value of RBE derived for stochastic endpoints; LET, linear energy transfer; CI, confidence interval; N.E., not estimated (RBEm not estimated due to lack of information). Unless specified, external irradiation. Unless specified, internal irradiation.
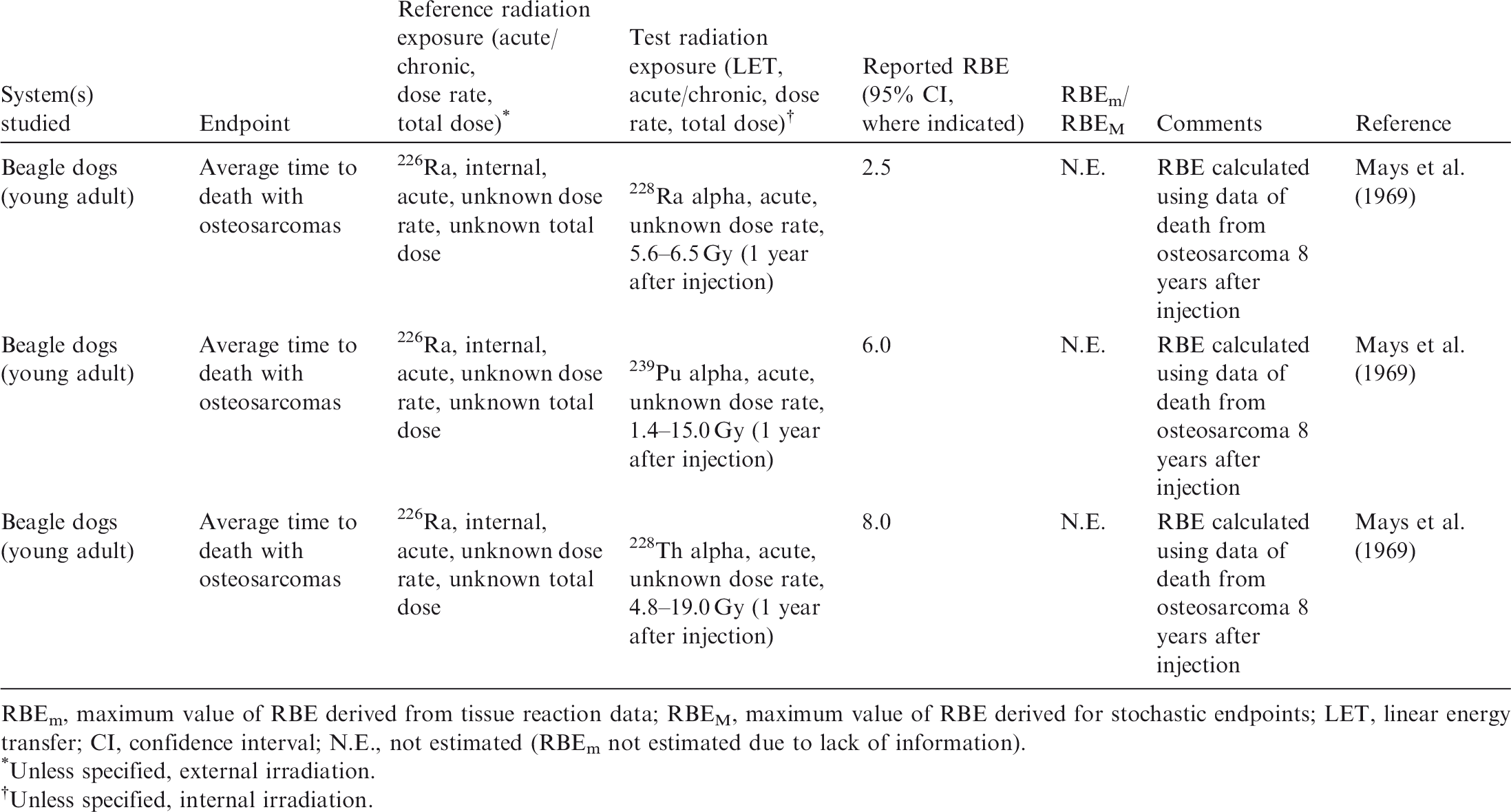
C.1.1. Data on relative biological effectiveness to produce early mortality
(C8) In mammals, early mortality is a result of extensive irradiation that causes severe cell depletion, leading to dysfunction of major organs. Death of the organisms occurs due to injury of specific organs caused by exposure to radiation. Few studies have been conducted to test this endpoint using alpha-emitting radionuclides. One study of interest but not direct relevance (Mays et al., 1969) reported RBE values of 6 (239Pu), 8 (228Th), and 2 (228Ra) relative to 226Ra as the reference radiation in a study of early mortality from radiation-induced bone cancer in beagle dogs (Table C.1). This variation in toxicity of alpha-particle-emitting radionuclides per Gy average bone dose is attributable to their different patterns of deposition in relation to the location of target cells for induction of bone cancer near to inner bone surfaces. (C9) Animals in the wild also develop cancer, but the effects of cancer morbidity on the ability to reproduce and the effect on overall mortality is not clear at environmentally relevant doses. Overall, the possibility of cancer as an endpoint is generally considered to be of relatively little interest for non-human biota compared with reproductive endpoints.
C.1.2. Data on relative biological effectiveness to reduce reproductive success
(C10) Fourteen publications were identified that considered the effects of alpha particles on reproductive success (Table C.2). The alpha-emitting radionuclides most commonly used in these studies were 238Pu, 239Pu, and 210Po, and the most common reference radiation was 60–120-kVp x rays. (C11) Depending on the species considered, a wide range of RBE and RBE maximum values were reported for endpoints related to reduced reproductive success, including numbers of surviving offspring, sperm head survival, and testes weight. Although a few papers reported RBE values of alpha particles >10 (see Section C.2), most were in the range of 1–10. Most RBE values were obtained using rodents or rodent cells exposed to high doses and dose rates.
C.1.3. Data on relative biological effectiveness to produce morbidity effects
(C12) As per Publication 108 (ICRP, 2008), all forms of morbidity in animals and plants lead to reduced fitness. This can influence an organism’s life span and reproductive capacity. There are few data on larger mammals, with most experiments conducted in mesocosms. (C13) Alpha-emitting radionuclides commonly used to irradiate cell lines, tissues, or cell cultures were 238Pu, 239Pu, and 210Po. The common reference radiation used in studies of this endpoint was 250-kVp x rays. (C14) Thirty-five publications reported RBE values for alpha particles for a variety of morbidity effects, notably, cell survival (Table C.3). RBE and RBE maximum values were calculated whenever possible from the slopes of the survival curves provided at low dose. Depending on the species considered, a wide range of RBE and RBE maximum values were reported. The majority of RBE values calculated were <5 and almost all RBE values provided for cell survival were <10.
C.1.4. Data on relative biological effectiveness to produce chromosomal damage and mutations
(C15) Thirty-three publications discussed chromosomal damage and mutations caused by exposure to alpha-emitting radionuclides (Table C.4). Alpha-emitting radionuclides commonly used to irradiate cell lines, tissues, or cell cultures were 238Pu, 239Pu, 241Am, and 226Ra. The common reference radiations in these studies were 60Co gamma rays and 80–300-kVp x rays. (C16) The majority of the reviewed publications that analysed chromosomal damage and mutations reported RBE values or provided enough data on fitted dose–response functions to allow calculation of RBE values. Most RBE values were obtained using rodents or rodent cells based on exposures to high doses and dose rates. Few papers reported RBE values for alpha particles >20, and most reported values are in the range of 1–10. (C17) Most of the studies concerned with chromosomal damage and mutation effects caused by alpha emitters were conducted on hamster cells in vitro and mice in vivo. It should be noted that while these data indicate an increase in the biological effectiveness of alpha radiation compared with the reference radiation, there are limitations to the quantitative use of these data. Moreover, the relation, if any, between chromosomal damage and mutational events at the cellular level observed in laboratories and observable level effects on environmental populations of non-human biota remains to be determined.
C.1.5. Relative biological effectiveness of alpha particles and experimental system
(C18) A wide range of experimental test systems, in vitro and in vivo, have been considered in studies of RBE. NCRP (1967) presents experimental curves of RBE vs LET for a wide variety of test organisms and endpoints, including T1 bacteriophage in broth, haploid yeast survival in air, artemia eggs hatching or emerging, various mammalian tissues, broad bean root effects on growth and survival, and others. (C19) Studies reported by Chen et al. (1984), Coquerelle et al. (1987), Edwards et al. (1980), Bedford et al. (1989), and Schmid et al. (1996) all focused on RBE of alpha particles with respect to human cells; however, these studies are considered relevant to all mammalian cells and relevant to the current evaluation. (C20) Studies of bone carcinoma induction in beagle dogs were reviewed and interpreted in terms of RBE comparing alpha-emitting 226Ra and beta-emitting 90Sr (Mays and Finkel, 1980). Amongst other observations, the data indicated that RBE approached or was greater than 20 in the lowest dose ranges, but was less at high doses. It was concluded that RBE for the alpha emitter increased as an inverse function of dose, which was attributed to be mainly due to the relatively low effectiveness per Gy of 90Sr beta particles at low doses and dose rates. (C21) The data summarised for mice show a considerable range in RBE for endpoints involving reproductive and haematopoietic systems. Rao et al. (1991) reported RBE of 245 for sperm head abnormalities from 210Po exposure, and RBE of 6.7 at 37% cell survival (Rao et al., 1989). (C22) Knowles (2001) reported studies of fish and found that there was no dose–effect relationship for zebrafish (Danio rerio) exposed to alpha particles, as none of the alpha doses were sufficiently high to result in the desired effect of cessation of egg production. Only an upper limit to RBE could be estimated, which could be a conservative upper limit. (C23) Mouse-embryo-derived fibroblastic cell lines (C3H 10T1/2 and BALB/3T3) in culture were the model systems used in several of the morbidity studies referenced in this publication. (C24) Cell lines from the Chinese hamster, V79 and CHO-K1, were the main model systems used in the in-vitro studies. Reported RBE ranged from 1 to 7, with an average of approximately 3. The calculated RBEm ranged from 1.7 to 12.8, with an average of approximately 8. (C25) Suzuki et al. (1989) reported on survival of golden hamster embryo cells and cell transformation due to exposure to heavy ions. (C26) Rats (in vivo/ex vivo): Reported experimental RBEs ranged from 1.1 to 10.7, with an average of approximately 4.
C.2. Concluding remarks on relative biological effectiveness of alpha particles
C.2.1. Other literature reviews
(C27) This section briefly reports on previous evaluations of RBE data other than those developed previously by ICRP for humans. (C28) As noted previously, NCRP (1967) provided a discussion of the concept of RBE of radiation from internal emitters, including discussions of RBE values for somatic effects in mammals and RBE data derived from dose–effect curves for a number of endpoints. It was concluded that the effects of high-LET radiations were insensitive to dose rate, while effects of low-LET radiations were dose-rate dependent. NCRP (1967) presents experimental curves of RBE vs LET for a variety of test organisms and endpoints, and suggests a maximum RBE of approximately 10 for radiation with an LET of approximately 300 keV µm−1 for human cells in culture. (C29) Thompson et al. (2002) summarised RBE values for alpha particles that were estimated in several experiments using various endpoints (Table C.5). (C30) Chambers et al. (2005) reviewed published data and summarised their conclusions concerning the range of RBE for different endpoints (Table C.6). Overall, these authors recommended a nominal (biota) radiation weighting factor for alpha particles of 5 for population-relevant endpoints, but, to reflect the limitations in the experimental data, also suggested uncertainty ranges of 1–10 and 1–20 for tissue reactions and stochastic endpoints, respectively. (C31) The reports discussed above and various other authors, including Copplestone et al. (2001), Environment Canada and Health Canada (2003), Pröhl et al. (2003), Trivedi and Gentner (2002), and UNSCEAR (2008), have provided nominal values (or ranges of values) for a radiation weighting factor, which are summarised in Table C.7. In considering these values, it is important to note that the estimates of RBE are specific to the endpoint studied; the biological, environmental, and exposure conditions (e.g. reference radiation, dose rate, and dose); and other factors. Thus, as noted in a FASSET report (Pröhl et al., 2003), it is difficult to develop a generally valid radiation weighting factor for use in an environmental risk assessment.
C.2.2. Overall evaluation of relative biological effectiveness of alpha particles
(C32) Previous evaluations of RBE data for alpha-particle-emitting radionuclides and the specification of radiation weighting factors for non-human biota include those of Pröhl et al. (2003) and UNSCEAR (2008). In order to account for the effect of radiation quality in cases of exposure to internally deposited alpha emitters, Pröhl et al. recommended that the absorbed dose should be modified by a radiation weighting factor of 10 (Pröhl et al., 2003; Larsson, 2004). In its most recent evaluation of the effects of ionising radiation on non-human biota, UNSCEAR (2008) recommended a modifying factor of 10 to reflect its judgement of the available data on RBE of alpha particles. (C33) The current evaluation considered in-vivo and in-vitro experimental data. Two significant features were evident from the in-vivo studies. Firstly, the studies were performed at relatively low doses and dose rates, and therefore, they were much closer to environmental exposure conditions than in-vitro tests, which used higher doses and dose rates. Secondly, the endpoints studied were critical from the standpoint of the maintenance of populations of organisms (reproductive performance, effects on oocytes and sperm, and immune system health). The majority of studies, notably those showing data for population-relevant endpoints, report RBE values <10.
Summary of publications studying relative biological effectiveness (RBE) of alpha particles to reduce reproductive success.

RBEm, maximum value of RBE derived from tissue reaction data; RBEM, maximum value of RBE derived for stochastic endpoints; LET, linear energy transfer; CI, confidence interval; N.E., not estimated (RBEm not estimated due to lack of information).
Unless specified, external irradiation.
Unless specified, internal irradiation.
C.3. References
Barendsen, G.W., 1989. Relative biological effectiveness for carcinogenesis. In: KF Baverstock and JW Stather (Eds), Low Dose Radiation: Biological Bases of Risk Assessment. London; New York: Taylor and Francis, pp. 467–476.
Barendsen, G.W., 1992. RBE for non-stochastic effects. Adv. Space Res. 12, 385–392.
Bedford, J.S., Goodhead, D.T., 1989. Breakage of human interphase chromosomes by alpha particles and X-rays. Int. J. Radiat. Biol. 55, 211–216.
Belli, M., Goodhead, D.T., Ianzini, F., et al., 1992. Direct comparison of biological effectiveness of protons and alpha particles of the same LET. II. Mutation induction at the HPRT locus in V79 cells. Int. J. Radiat. Biol. 61, 625–629.
Bertsche, U., Ilakis, G., 1987. Modifications in repair and expression of potentially lethal damage (A-PLD) as measured by delayed plating or treatment with ß-Ara A in plateau-phase Ehrlich ascites tumor cells after exposure to charged particles of various specific energies. Radiat. Res. 111, 26–46.
Blöcher, D., 1988. DNA double-strand break repair determines the RBE of alpha-particles. Int. J. Radiat. Biol. 54, 761–771.
Brooks, A.L., 1975. Chromosome damage in liver cells from low dose rate alpha, beta, and gamma irradiation: derivation of RBE. Science 190, 1090–1092.
Brooks, A.L., Miick, R., Buschbom, R.L., et al., 1995. The role of dose rate in the induction of micronuclei in deep-lung fibroblasts in vivo after exposure to cobalt-60 gamma rays. Radiat. Res. 144, 114–118.
Burns, F.J., Albert, R.E., Heimbach, R.D., 1968. The RBE for skin tumors and hair follicle damage in the rat following irradiation with alpha particles and electrons. Radiat. Res. 36, 225–241.
Chambers, D.B., Osborne, R.V., Garva, A.L., 2005. Choosing an alpha radiation weighting factor for doses to non-human biota. J. Environ. Radioact. 87, 1–14.
Chen, D.J., Strniste, G.F., Tokita, N., 1984. The genotoxicity of alpha particles in human embryonic skin fibroblasts. Radiat. Res. 100, 321–327.
CNSC, 2002. Protection of Non-human Biota from Ionizing Radiation. Advisory Committee on Radiological Protection. INFO-0703. Canadian Nuclear Safety Commission, Ottawa.
Copplestone, D., Bielby, S., Jones S.R., et al., 2001. Impact Assessment of Ionising Radiation on Wildlife. R&D Publication 128. Environment Agency, Bristol.
Coquerelle, T.M., Weibezahn, K.F., Lucke-Huhle, G., 1987. Rejoining of double-strand breaks in normal human and ataxia-telangiectasia fibroblasts after exposure to 60Co γ-rays, 241Am α-particles or bleomycin. Int. J. Radiat. Biol. 51, 209–218.
Cox, R., Thacker, J., Goodhead, D.T., et al., 1977. Mutation and inactivation of mammalian cells by various ionizing radiations. Nature 267, 425–427.
Durante, M., Grossi, G.F., Napolitano, M., et al., 1992. Chromosome damage induced by high-LET α-particles in plateau-phase C3H 10T1/2 cells. Int. J. Radiat. Biol. 62, 571–580.
Edwards, A.A., Purrott, R.J., Prosser, J.S., et al., 1980. The induction of chromosome aberrations in human lymphocytes by alpha radiation. Int. J. Radiat. Biol. 38, 83–91.
Environment Canada, Health Canada, 2003. Second Priority Substances List Assessment Report. Releases of Radionuclides from Nuclear Facilities (Impact on Non-human Biota). PSL2. Environment Canada and Health Canada, Ottawa.
Feola, J.M., Lawrence, J.H., Welch, G.P., 1969. Oxygen enhancement ratio and RBE of helium ions on mouse lymphoma cells. Radiat. Res. 40, 400–413.
Fisher, D.R., Frazier, M.E., Andrews, T.K. Jr, 1985. Energy distribution and the relative biological effects of internal alpha emitters. Radiat. Prot. Dosim. 13, 223–227.
Ford, J.R., Terzaghi-Howe, M., 1993. Effects of 210Po alpha particles on survival and preneoplastic transformation of primary rat tracheal epithelial cells irradiated while in suspension or in the intact tissue. Radiat. Res. 136, 89–96.
Fox, J.C., McNally, N.J., 1990. The rejoining of DNA double-strand breaks following irradiation with 238Pu α-particles: evidence for a fast component of repair as measured by neutral filter elution. Int. J. Radiat. Biol. 57, 513–521.
Goodhead, D.T., 1992. Track structure considerations in low dose and low dose rate effects of ionizing radiation. Adv. Radiat. Biol. 16, 7–44.
Goodhead, D.T., Belli, M., Mill, A.J., et al., 1992. Direct comparison between protons and alpha-particles of the same LET: I. Irradiation methods and inactivation of asynchronous V79, HeLa and C3H 10 T½ cells. Int. J. Radiat. Biol. 61, 611–624.
Grahn, D., Frystak, B.H., Lee, C.H., et al., 1979. Dominant lethal mutations and chromosome aberrations induced in male mice by incorporated Pu-239 and by external fission neutron and gamma irradiation. In: Biological Implications of Radionuclides Released from Nuclear Industries. International Atomic Energy Agency, Vienna, pp. 163–184.
Hei, T.K., Komatsu, K., Hall, E.J., et al., 1988. Oncogenic transformation by charged particles of defined LET. Carcinogenesis 9, 747–750.
Hieber, L., Posnel, G., Roos, H., et al., 1987. Absence of a dose-rate effect in the transformation of C3H 10T1/2 cells by alpha-particles. Int. J. Radiat. Biol. 52, 859–869.
Howell, R.W., Azure, M.T., Narra, V.R., et al., 1994. Relative biological effectiveness of alpha-particle emitters in vivo at low doses. Radiat. Res. 137, 352–360.
Howell, R.W., Goddu, S.M., Narra, V.R., et al., 1997. Radiotoxicity of gadolinium-148 and radium-223 in mouse testes: relative biological effectiveness of alpha-particle emitters in vivo. Radiat. Res. 147, 342–348.
IAEA, 1992. Effects of Ionizing Radiation on Plants and Animals at Levels Implied by Current Radiation Protection Standards. Technical Reports Series No. 332. International Atomic Energy Agency, Vienna.
ICRP, 2008. Environmental protection – the concept and use of Reference Animals and Plants. ICRP Publication 108. Ann. ICRP 38(4–6).
Jenner, T.J., Belli, M., Goodhead, D.T., et al., 1992. Direct comparison of biological effectiveness of protons and alpha-particles of the same LET. III. Initial yield of DNA double-strand breaks in V79 cells. Int. J. Radiat. Biol. 61, 631–637.
Jenner, T.J., deLara, C.M., Oneill, P., et al., 1993. Induction and rejoining of DNA double-strand breaks in V79-4 mammalian cells following gamma and alpha irradiation. Int. J. Radiat. Biol. 64, 265–273.
Jiang, T.N., Lord, B.I., Hendry, J.H., 1994. Alpha particles were extremely damaging to developing hemopoiesis compared to gamma irradiation. Radiat. Res. 137, 380–384.
Kadhim, M.A., Macdonald, D.A., Goodhead, D.T., et al., 1992. Transmission of chromosomal instability after plutonium α-particle irradiation. Nature 355, 738–740.
Khan, M.A., Cross, F.T., Jostes, R.F., et al., 1994. Micronuclei induced by radon and its progeny in deep-lung fibroblasts of rats in vivo and in vitro. Radiat. Res. 139, 53–59.
Knowles, J.F., 2001. An Investigation into the Effects of Chronic Radiation on Fish. R&D Technical Report P3-053/TR. Centre for Environment, Fisheries & Aquaculture Science, Lowestoft, pp. 1–42.
Kozlowski, R., Bouffler, S.D., Haines, J.W., et al., 2001. In utero haemopoietic sensitivity to alpha, beta or X-irradiation in CBA/H mice. Int. J. Radiat. Biol. 77, 805–815.
Kugel, C., Tourdes, F., Poncy, J.L., et al., 2002. RBE of α-irradiation for in vitro relative transformation of rat tracheal epithelial cells. Ann. Occup. Hyg. 46, 285–287.
Larsson, C.M., 2004. The FASSET framework for assessment of environmental impact of ionising radiation in European ecosystems – an overview. J. Radiol. Prot. 24, A1–A12.
Lord, B.I., Mason, T.M., 1996. On the relative biological effectiveness of alpha-particle irradiation with respect to hemopoietic tissue. Radiat. Res. 145, 510–518.
Lücke-Huhle, C., Pech, M., Herrlich, P., 1986. Selective gene amplification in mammalian cells after exposure to 60Co gamma-rays, 241Am alpha particles, or UV light. Radiat. Res. 106, 345–355.
Lundgren, D.L., Gillett, N.A., Hahn, F.F., et al., 1987. Effects of protraction of the alpha dose to the lungs of mice by repeated inhalation exposure to aerosols of 239PuO2. Radiat. Res. 111, 201–224.
Manti, L., Jamali, M., Prise, K.M., et al., 1997. Genomic instability in Chinese hamster cells after exposure to x-rays or alpha particles of different mean linear energy transfer. Radiat. Res. 14, 22–28.
Martin, S.G., Miller, R.G., Geard, C.R., et al., 1995. The biological effectiveness of radon-progeny alpha particles. iv. Morphological transformation of Syrian hamster embryo cells at low doses. Radiat. Res. 142, 70–77.
Mason, T.M., Lord, B.I., Molineux, G., et al., 1992. Alpha-irradiation of haemopoietic tissue in pre- and postnatal mice. 2. Effects of mid-term contamination with 239Pu in utero. Int. J. Radiat. Biol. 61, 393–403.
Mays, C.W., Dougherty, T.F., Taylor G.N., et al., 1969. Radiation induced bone cancer in beagles. In: Delayed Effects of Bone Seeking Radionuclides. University of Utah Press, Salt Lake City, UT, pp. 387–408.
Mays, C.W., Finkel, M.P., 1980. RBE of alpha-particles versus beta-particles in bone sarcoma induction. In: Radiation Protection: a Systematic Approach to Safety: Proceedings of the 5th Congress of the International Radiation Protection Society, Jerusalem. Pergamon Press, Oxford.
Miller, R.C., Geard, C.R., Brenner, D.J., et al., 1989. Neutron-energy-dependent oncogenic transformation of C3H 10% 1/2 mouse cells. Radiat. Res. 117, 114–127.
Miller, R.C., Brenner, D.J., Randers-Pehrson, G., et al., 1990. The effects of the temporal distribution of dose on oncogenic transformation by neutrons and charged particles of intermediate LET. Radiat. Res. 124, S62–S68.
Miller, R.C., Marino, S.A., Brenner, D.J., et al., 1995. The biological effectiveness of radon-progeny alpha particles. II. Oncogenic transformation as a function of linear energy transfer. Radiat. Res. 142, 54–60.
Muller, W.A., Luz, A., Schaffer, E.H., et al., 1983. The role of time-factor and RBE for the induction of osteosarcomas by incorporated short-lived bone-seekers. Health Phys. 44, 203–212.
Nagasawa, H., Robertson, J., Little, J.B., 1990. Induction of chromosomal aberrations and sister chromatid exchanges by alpha particles in density-inhibited cultures of mouse 10 T½ and 3T3 cells. Int. J. Radiat. Biol. 57, 35–44.
Nagasawa, H., Little, J.B., 1992. Induction of sister chromatid exchanges by extremely low doses of α-particles. Cancer Res. 52, 6394–6396.
NCRP, 1967. Dose-effect Modifying Factors in Radiation Protection. BNL 50073 (T-471). National Council on Radiation Protection and Measurements, Bethesda, MD.
NCRP, 1991. Effects of Ionizing Radiation on Aquatic Organisms. NCRP Report No. 109. National Council on Radiation Protection and Measurements, Bethesda, MD.
Nikjoo, H., Munson, R.J., Bridges, B.A., 1999. RBE-LET relationships in mutagenesis by ionizing radiation. J. Radiat. Res. 40, 85–109.
Oghiso, Y., Yamada, Y., 2003. Comparisons of pulmonary carcinogenesis in rats following inhalation exposure to plutonium dioxide or X-ray irradiation. J. Radiat. Res. 44, 261–270.
Petin, V.G., 1979. Effect of gamma and alpha irradiation on survival of wild-type and sensitive mutants of yearst. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis, 60(1), pp.43–49.
Petin, V.G., Kabakova, N.M., 1981. RBE of densely ionizing radiation for wild-type and radiosensitive mutants of yeast. Mutat. Res. 82, 285–294.
Prise, K.M., Davies, S., Michael, B.D., 1987. The relationship between radiation-induced DNA double-strand breaks and cell kill in hamster V79 fibroblasts irradiated with 250 kVp X-rays, 2.3 MeV neutrons or 238Pu α-particles. Int. J. Radiat. Biol. 52, 893–902.
Pröhl, G., Brown, J., Gomez-Ros, J.M., et al., 2003. Dosimetric models and data for assessing radiation exposure to biota. Deliverable 3 to the Project “FASSET” Framework for the assessment of Environmental Impact, contract No. Contract No. FIGE-CT-2000-00102. Stockholm: Swedish Radiation Protection Authority.
Rao, D.V., Sastry, K.S., Gnmmond, H.E., et al., 1988. Cytotoxicity of Some Indium. J Nucl Med, 29, pp.375–384.
Rao, D., Roger, V., Howell, W., et al., 1989. In-vivo radiotoxicity of DNA-incorporated 125I compared with that of densely ionising alpha-particles. Lancet 334, 650–653.
Rao, D., Venkateswara, V., Narra, R., et al., 1991. Induction of sperm head abnormalities by incorporated radionuclides: dependence on subcellular distribution, type of radiation, dose rate, and presence of radioprotectors. Radiat. Res. 125, 89–97.
Roberston, J.B., Koehler, A., George, J., et al., 1983. Oncogenic transformation of mouse Balb/3T3 cells by plutonium-238 α-particles. Radiat. Res. 96, 261–274.
Roberts, C.J., Goodhead, D.T., 1987. The effect of 238Pu α-particles on the mouse fibroblast cell line C3H 10T1/2: characterization of source and RBE for cell survival. Int. J. Radiat. Biol. 52, 871–882.
Samuels, L.D., 1966. Effects of polonium-210 on mouse ovaries. Int. J. Radiat. Biol. 11, 117–129.
Schmid, E., Hieber, L., Heinzmann, U., et al., 1996. Analysis of chromosome aberrations in human peripheral lymphocytes induced by in vitro α-particle irradiation. Radiat. Environ. Biophys. 35, 179–184.
Schwartz, J.L., Rotmensch, J., Atcher, R.W., et al., 1992. Interlaboratory comparison of different alpha-particle and radon sources: cell survival and relative biological effectiveness. Health Phys. 62, 458–461.
Searle, A.G., Beechey, C.V., Green, D., et al., 1976. Cytogenetic effects of protracted exposures to alpha-particles from plutonium-239 and to gamma-rays from cobalt-60 compared in male mice. Mutat. Res. 41, 297–310.
Searle, A.G., Beechey, C.V., Green, D., et al., 1980. Comparative effects of protracted exposures to 60Co γ-radiation and 239Pu α-radiation on breeding performance in female mice. Int. J. Radiat. Biol. 37, 189–299.
Suzuki, M., Watanbe, M., Suzuki, K., et al., 1989. Neoplastic cell transformation by heavy ions. Radiat. Res. 120, 468–476.
Thacker, J., Stretch, A., Goodhead, D.T., 1982. The mutagenicity of α-particles from plutonium-238. Radiat. Res. 92, 343–352.
Thomassen, D.G., Seiler, F.A., Shyr, L.J., et al., 1990. Alpha-particles induce preneoplastic transformation of rat tracheal epithelial cells in culture. Int. J. Radiat. Biol. 57, 395–405.
Thompson, P.A., Macdonald, C.R., Harrison, F., 2002. Recommended RBE Weighting Factors for the Ecological Assessment of Alpha-emitting Radionuclides and Tritium Beta Particles. Third International Symposium on the Protection of the Environment from Ionising Radiation, 22–26 July 2002, Darwin, Australia.
Trivedi, A., Gentner, N.E., 2002. Ecodosimetry Weighting Factor (eR) for Non-human Biota. Paper T-1-5, P-2 a-114. In: IRPA-10. Proceedings of the International Radiation Protection Association, 14–19 May 2000, Hiroshima, Japan.
Ullrich, R. 1984. Tumor Induction in BALB/c Mice after Fractionated or Protracted Exposures to Fission-Spectrum Neutrons. Radiation Research, 97(3), 587–597. doi:10.2307/3576150
UNSCEAR, 1996. Sources and Effects of Ionizing Radiation. Report to the General Assembly, with Scientific Annex. United Nations Sales Publication E.96.IX.3. United Nations Scientific Committee on the Effects of Atomic Radiation, New York.
UNSCEAR, 2008. Sources and Effects of Ionizing Radiation, Volume II, Annex E. Effects of Ionizing Radiation on Non-human Biota. Report to the General Assembly with Scientific Annexes. United Nations Sales Publication E.11.IX.3. United Nations Scientific Committee on the Effects of Atomic Radiation, New York.
Yang, T.C., Craise, L.M., Mei, M.T., et al., 1985. Neoplastic cell transformation by heavy charged particles. Radiat. Res. 104, SI77–S187.
Zyuzikov, N.A., Prise, K.M., Zdzienicka, M.Z., et al., 2001. The relationship between the RBE of alpha particles and the radiosensitivity of different mutations of Chinese hamster cells. Radiat. Environ. Biophys. 40, 243–248.
Summary of publications studying relative biological effectiveness (RBE) of alpha particles to produce morbidity effects.
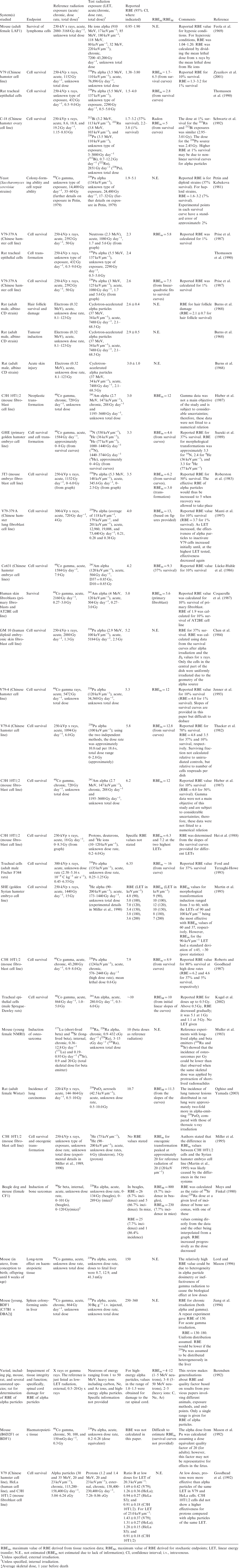
RBEm, maximum value of RBE derived from tissue reaction data; RBEM, maximum value of RBE derived for stochastic endpoints; LET, linear energy transfer; N.E., not estimated (RBEm not estimated due to lack of information); CI, confidence interval; i.v., intravenous.
Unless specified, external irradiation.
Unless specified, internal irradiation. ‡Average skeletal dose, 1 year before death
Summary of publications studying relative biological effectiveness (RBE) of alpha particles to produce chromosomal damage and mutations.
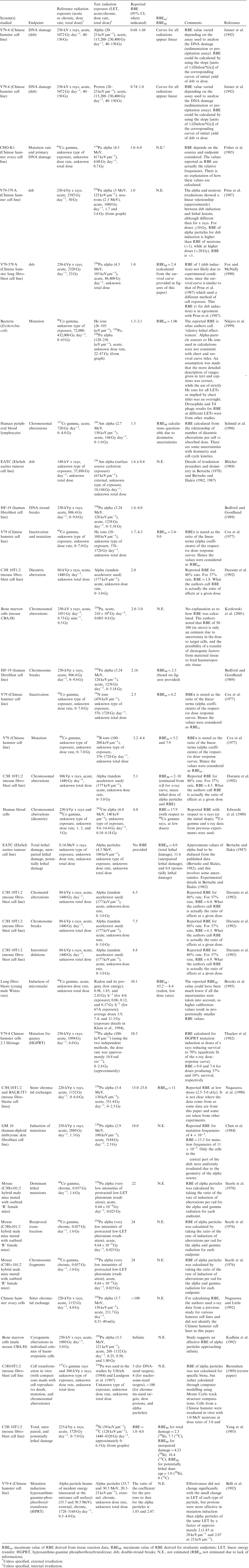
RBEm, maximum value of RBE derived from tissue reaction data; RBEM, maximum value of RBE derived for stochastic endpoints; LET, linear energy transfer; HGPRT, hypoxanthine-guanine phosphoribosyltransferase; dsb, double-strand breaks; N.E., not estimated (RBEm not estimated due to lack of information).
Unless specified, external irradiation.
Unless specified, internal irradiation.
Relative biological effectiveness (RBE) of alpha particles (Thompson et al., 2002).
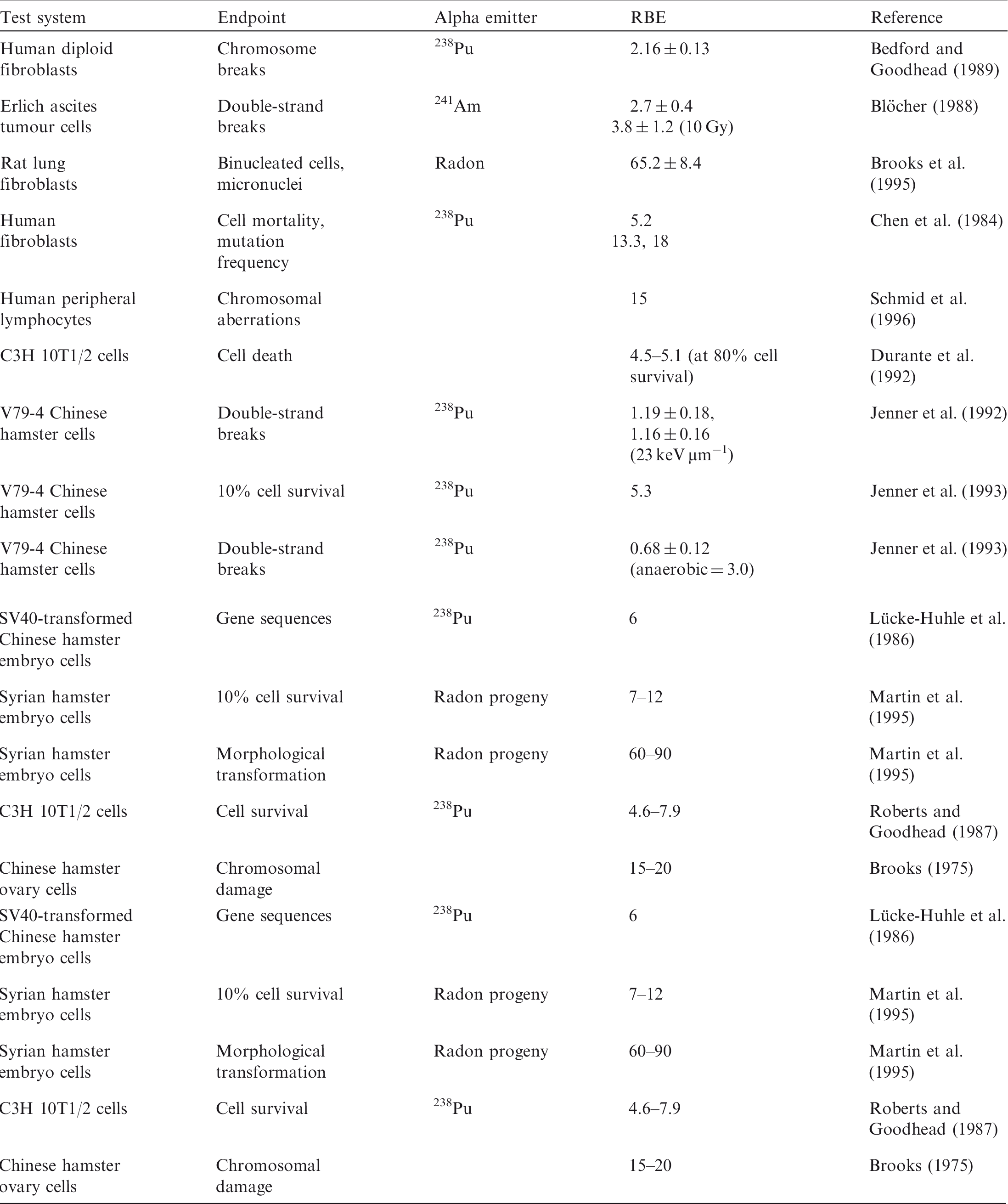
Range of relative biological effectiveness (RBE) values for alpha particles reported by Chambers et al. (2005).

Radiation weighting factors for alpha particles in non-human biota (relative to low-linear energy transfer radiation).
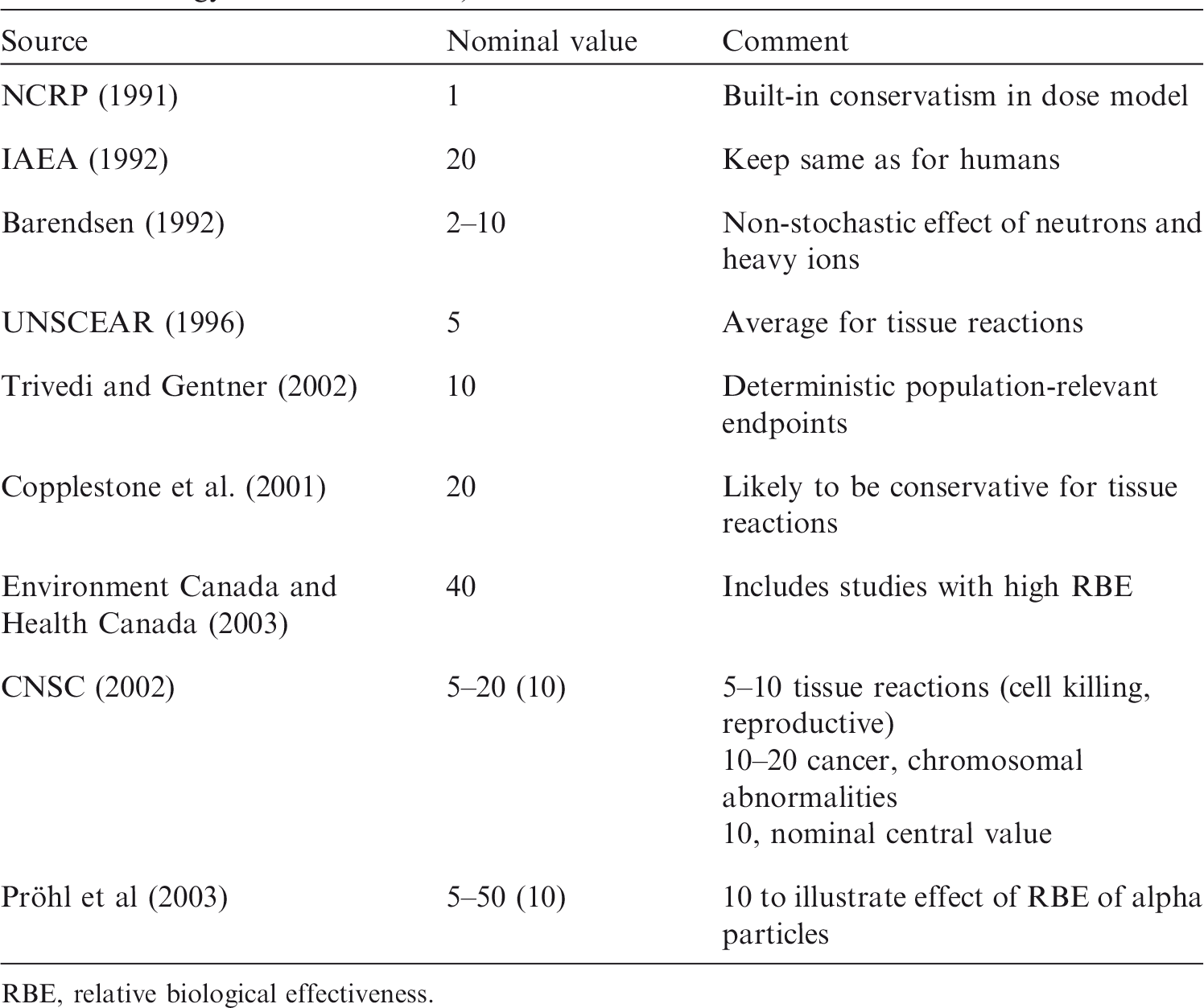
RBE, relative biological effectiveness.
GLOSSARY
Natural environment A collective term for all of the physical, chemical, and biological conditions within which wild animals and plants normally live. The factor characterising the biological effectiveness of a radiation, based on the ionisation density along the tracks of charged particles in tissue. Q is defined as a function of the unrestricted linear energy transfer, L∞ (often denoted as L or LET), of charged particles in water:
Q has been superseded by the radiation weighting factor in the definition of equivalent dose, but is still used in calculating the operational dose equivalent quantities used in monitoring. A practical method (function or numerical value) used to represent relative biological effectiveness for a specific type of radiation, based on existing scientific knowledge and adopted by consensus or via recommendations. Within the system of human radiological protection, it is used to define and derive the equivalent dose from the mean absorbed dose in an organ or tissue. The ratio of a dose of a low-LET reference radiation (usually of 60Co gamma rays or kilovoltage x-ray quality) to a dose of the test radiation considered that gives an identical biological effect. RBE values vary with the dose, dose fractionation, dose rate, and biological endpoint considered. Maximal value of RBE derived from tissue reaction data. There is a dose dependence to RBE, which reaches a maximal value as the dose drops below approximately 0.1 Gy of x rays. RBEm is the calculated ratio of slopes of the dose–effect curves at zero dose. Maximal value of RBE derived for stochastic effects, e.g. carcinogenesis. There is a dose dependence to RBE, which reaches a maximal value as the dose drops below approximately 0.1 Gy of x rays. RBEM is the calculated ratio of slopes of the dose–effect curves at zero dose.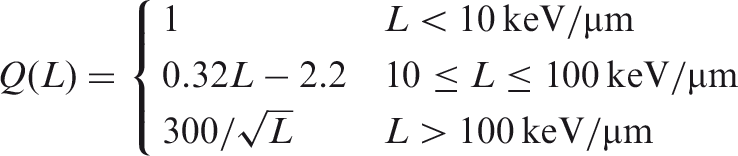
ACKNOWLEDGEMENTS
This publication provides a review and summary of studies that allow the derivation of radiation weighting factors for alpha-emitting radionuclides and low-energy beta emitters, such as tritium, for application in dose assessment for RAPs, taking into account different endpoints that are relevant for protection of the maintenance of biological diversity and the conservation of species of biota (mortality, fertility, and morbidity). The use of the proposed values is discussed.
ICRP thanks all those involved in the development of this publication for their hard work and dedication over many years.


C.H. Clement (Scientific Secretary and Annals of the ICRP Editor-in-Chief)
H. Fujita (Assistant Scientific Secretary and Annals of the ICRP Associate Editor)




Chair: C. Cousins, UK
Vice-Chair: J. Lochard, France

†Although formally not a member since 1988, the Scientific Secretary is an integral part of the Main Commission.
ICRP and the members of Task Group 72 thank R.J. Pentreath for his helpful comments to this publication.
Finally, thank you very much to all organisations and individuals who took the time to provide comments on the draft of this publication during the consultation process.
