Abstract
The use of proton therapy as a treatment modality is becoming more widespread in conventional radiation therapy practice. Commercialisation and introduction of compact systems has led to embedding of proton therapy facilities in existing hospital environments. In addition, technologically, proton therapy is currently undergoing an important evolution, moving from passive scattering delivery techniques to active pencil beam scanning, adopting image guidance techniques from conventional radiotherapy and introducing various range verification techniques in the clinic. An overview is given of today’s technological evolution of proton therapy in clinical environments, and its impact on aspects of radiation protection.
1. Introduction
Over the last decade, the number of proton therapy facilities has risen at increasing speed, from approximately 20 centres in 2007 to >100 today. Although the advantageous properties of a proton beam for radiation therapy have generally been accepted since its early application in the 1960s, commercial solutions for proton therapy only became available at the end of the 1990s. The commercial availability, probably linked to the breakthrough of intensity modulated radiotherapy technology with photons at the end of the 1990s and the belief that dose distributions would be even better with proton beams, triggered greater interest in proton therapy. A high demand set into motion a process addressing a large hurdle in making this technology available to a large number of patients, namely its cost (Lievens and Pijls-Johannesma, 2013). Technological development is focusing on making proton therapy more efficient and versatile to serve a broader group of oncology patients with improved radiation therapy. Not only has the technique to irradiate target volumes with a proton beam evolved, but the layout of proton therapy facilities and their integration into existing hospital facilities has changed. In this paper, the impact of this evolution on stray radiation is considered, together with some other radiation protection aspects at the level of the patient, staff, and environment.
2. Delivery Technology and Nozzle Design
A particle accelerator, be it a cyclotron or a synchrotron, delivers a narrow particle beam, typically a few millimetres in diameter. This narrow beam is transported through a beam-line to the treatment room, where it is fed to the last part of the beam-line, the so-called ‘nozzle’. The function of the nozzle is to change the properties of the beam in such a way that a target volume of a few centimetres up to a few tens of centimetres in diameter can be irradiated with a therapeutic high dose level while sparing surrounding tissue. The nozzle is a cascade of beam-modifying devices and detectors. Its design and the production of secondary stray radiation differ depending on the technique used. Proton therapy as a treatment modality is undergoing a transition between two delivery techniques at present, moving from passive scattering to pencil-beam scanning.
Passive scattering has been the main delivery technology for the past two decades, and is applied in the majority of cases treated with proton therapy today. The technique creates a wide proton beam, making use of one or multiple scatterers designed geometrically and in composition to produce a wide beam with homogeneous intensity. A commonly used design is double scattering using a cascade of two scatterers, applying a combination of high and low Z parts, and using the differences in stopping and scattering power of protons of these materials to make optimal use of all accelerated protons, and limit beam loss in beam-shaping devices. In depth, the fine Bragg peak is modulated, typically using a modulator wheel, in a spread-out Bragg peak (SOBP) to form a plateau of high dose covering the largest dimension of the tumour in the direction of the beam axis. The beam is shaped to the target volume using treatment-field-specific apertures. These apertures are beam-portal specific and are usually made from dense high Z materials to maximise proton stopping in a short distance. Typically, solid brass is milled or a metal alloy with a low melting point, such as cerroband, is poured into a shaped mould. The combination of aperture and beam modulation is then completed with a field-specific range compensator to align the distal edge of the SOBP dose distribution with the distal contour of the target volume.
This cascade of beam modifiers makes use of certain interactions of the proton beam with the materials of the devices. As well as the useful beam modifications resulting from these interactions on which the beam modifications are based, undesired effects take place that do not serve the purpose of targeted local dose distribution with protons. This leads to secondary stray radiation, mainly neutrons and x rays, which can not only deposit energy in the patient outside the targeted tissues, but can also lead to activation of the beam-modifying devices, the environment, and the patient. In passive scattering, the beam modification is performed in the nozzle, the last part of the beam-line just before the beam enters the patient, which implies the presence of secondary radiation sources in close proximity to the patient. Research has been conducted to reduce the activation and secondary radiation levels by improving the design and replacing certain materials used in the nozzle (Moskvin et al., 2013). The presence of these devices in the proton beam is necessary in passive scattering.
Active pencil-beam scanning does not require scattering/flattening devices, compensators, or beam-shaping apparatus in the beam. The beam modifier cascade is replaced in the nozzle by a pair of scanning magnets. The scanning beam approach is applied to cover a target volume of up to tens of centimetres with a fine pencil beam, steering a large number of these beam spot dose depositions to a grid of locations in the target. The range of each beam spot is adjusted through energy switching in the energy selection system (ESS), which can be placed upstream in the beam-line at a considerable distance from the patient. Pencil-beam scanning results in a considerably lower neutron background compared with passive scattering, as the main source of secondary radiation is the interaction of the proton beam with the patient, which is obviously inevitable.
Even if passive scattering is still used for the majority of patients treated with proton therapy, most new facilities today choose pencil-beam scanning as the delivery technology. A large number of facilities that initially used passive scattering are currently upgrading their systems to pencil-beam scanning. The reason for this transition is not primarily the reduction in secondary cancer risk for patients due to lower stray neutron radiation dose in pencil-beam scanning compared with passive scattering. It is generally accepted that the major component of secondary cancer risk is from in-field radiation rather than out-of-field stray radiation (NCRP, 2011; Newhauser and Zhang, 2015). Most of the gain here is thus in the transition from photon radiotherapy to proton therapy, because of the sparing of healthy tissue, as is illustrated in Fig. 1. The difference in total risk of secondary cancer moving from passive scattering to pencil-beam scanning is estimated to be small. Nevertheless, striving towards a reduction in secondary neutron dose to the patient remains justified given our incomplete knowledge of stray neutron exposures and secondary cancer induction.
Dose distributions are shown in the axial plane. Intensity modulated photon therapy allows sparing of organs at risk, but the exit dose of the photon beams results in exposure of large parts of the body. Proton therapy does not result in exposure of tissue downstream of the targeted tumour volume, improving in-field sparing of organs at risk and resulting in no dose to these parts of the body.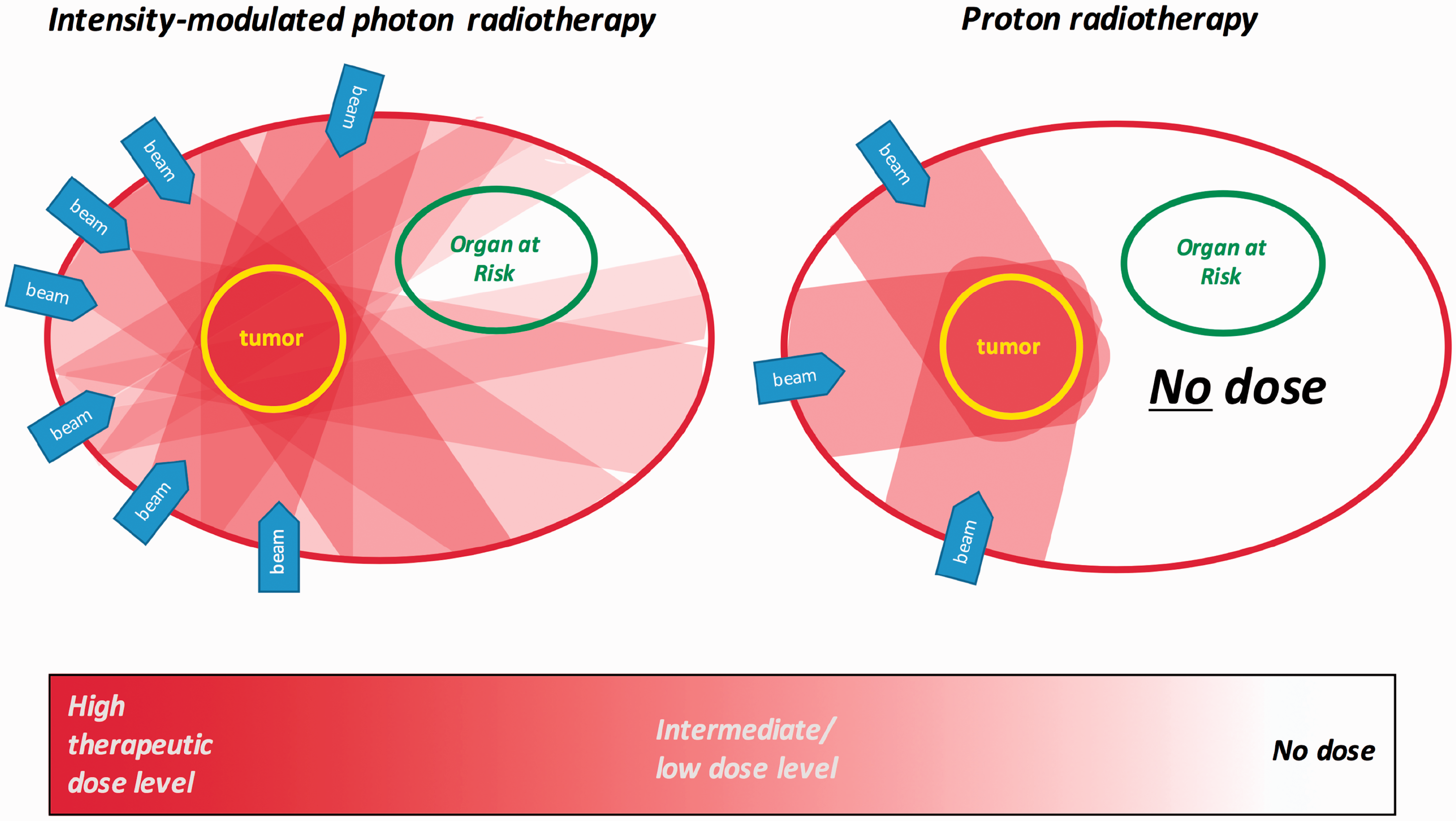
The versatility and practical advantages of pencil-beam scanning compared with passive scattering make pencil-beam scanning an attractive delivery technology for proton therapy. Pencil-beam scanning no longer requires patient-specific apertures and compensators. The patient-specific customisation of the treatment plan can be achieved fully by weighting the scanned pencil-beam spots, and no patient-specific hardware is required. Using passive scattering as a delivery technique requires setting up and maintaining a hardware workshop on-site. Even if passive scattering with its wide beam delivery is more efficient at the level of dose deposition speed, manual replacement of patient-specific hardware for every field is required, introducing delays in the workflow. As the apertures and compensators are in the proton beam, they will be activated, which complicates storage and manipulation by the therapy staff. Due to the large variety of proton therapy system designs, these activations have not been studied extensively, and very little data seem to be available on occupational exposure to clinical staff. Compensators made of Lucite or wax have been reported to decay to background in approximately 2 h, which is more than the typical treatment delivery times of 30–45 min. Activated brass apertures require longer cooling and are kept in secured storage for several months before being sent for scrap (Moskvin et al., 2013; Spitznagel, 2014).
A major advantage of pencil-beam scanning compared with passive scattering is the improved quality of the dose distribution. As the SOBP modulation is fixed per treatment field and is determined based on the largest cross-section of the target volume in that beam direction, some tissues proximal to the target will receive the same dose as the target volume itself. In pencil-beam scanning, this can be avoided and the dose to these tissues can be reduced. A step further, making optimal use of the degrees of freedom provided by pencil-beam scanning, is intensity modulated proton therapy using multi-field optimisation. Studies have shown that applying this technique, moving away from the concept of delivering a homogeneous dose to the target with each individual treatment field (single field optimisation), results in improved sparing of critical organs compared with single field optimisation and passive scattering approaches (Cozzi et al., 2001; Lomax et al., 2001; Welsh et al., 2011). To treat moving tumours with proton therapy, interplay effects of tumour motion and the pencil-beam scanning sequence can reduce the quality of dose distribution. Techniques to overcome interplay effects are being studied extensively (Knopf et al., 2010) and are finding their way into clinical proton therapy practice.
3. Compact layout systems
Further to the initial commercialisation of proton therapy systems in the 1990s, the industry is moving into the next phase by developing compact proton therapy systems. The main goal is definitely commercial: to reduce the total investment cost per proton therapy project and make it accessible to more potential customers. The reduction in project investment cost is typically from >100 M€ projects to <50 M€ projects for compact systems. Budgets of up to 50 M€ come within the reach of large hospitals and no longer require large national programmes to finance them. However, next to these financial implications, compact systems have an impact on the field of proton therapy in other ways.
The footprint of compact systems is considerably smaller than the large multi-room facilities. Using a sports field metaphor, the large multi-room facilities typically require a football field, while the compact solutions fit on a tennis or basketball court. The compact systems are developed as units including a dedicated accelerator, mostly compact cyclotrons, connected directly to a gantry. To further reduce the size of the systems, the gantries are made smaller (e.g. by limiting rotation to a 200° range instead of the full 360° range, or by placing scanning magnets upstream before the last bending magnet instead of in line with the nozzle). Multiple compact units can even form a scalable multi-room facility with each room having its own cyclotron. However, at some point, a single accelerator serving multiple treatment rooms becomes more cost-effective in terms of maintenance and resources.
A smaller proton therapy system also better fits and integrates in existing hospital facilities (examples in Fig. 2). The early radiotherapy with protons was usually a by-product of large nuclear research facilities providing particle beams. After the commercialisation of proton therapy in the 1990s until a few years ago, the leading trend was to build large stand-alone multi-room facilities. The main purpose of these facilities was the medical application of proton therapy, although some of them were not in close proximity to a hospital, and had on-site medical imaging facilities for treatment planning and follow-up. The compact proton therapy systems can more easily be placed adjacent to existing hospitals and radiation oncology departments, which allows an elevated level of integration as a ‘fully embedded’ facility (Fig. 3). In this way, proton therapy will be seen as an additional modality for the radiotherapy clinic rather than a separate facility. Part of the preparatory workflow can be shared between proton therapy and state-of-the-art photon radiotherapy. Having more of these combined facilities, where the same medical team has access to both modalities, is important in the process of seeking the right balance between the justified application of proton therapy and using the more cost-effective advanced photon treatments, by applying objective evidence-based methods (Langendijk et al., 2013). Many hospital-embedded proton therapy centres provide research facilities for physics and biology which, in a way, is the reverse of how proton therapy originally started as a research facility by-product (Fig. 3).
Ongoing construction sites of two compact proton therapy (PT) systems embedded in existing hospital facilities: (left) ParTICLe at UZ Leuven, Leuven, Belgium and (right) ZONPTC at MAASTRO Clinic, Maastricht, The Netherlands. The evolution of proton therapy (PT) centres from a by-product of large nuclear physics research facilities over large stand-alone facilities towards facilities embedded in existing hospital environments.

The smaller footprint of compact proton therapy systems does have implications for the shielding design. A compact gantry with limited gantry rotation range of (typically) 200° will require more asymmetric shielding in the allowed beam direction than a 360° gantry, where the beam orientation is distributed evenly between both sides. The wall thickness might also be larger in compact systems than in large facilities to reach public area limits at the outer shell, because the distances between the radiation sources and the walls are smaller and thus there is no benefit from the dose reduction with distance for radiation protection. As the walls can be much closer to the cyclotron than in a large facility, the neutron fluence reaching the wall surface is higher. Thus, the specific activity of the long-lived isotopes such as 152Eu or 60Co will be larger after 20 years of operation. As the characterisation of nuclear waste is based upon the specific activity, a larger amount of potential low-level nuclear waste results from a compact system than from a large multi-room system, even if the beam usage is lower. In order to address this issue, proton therapy vendors are looking into ‘low-activation concrete’ solutions to solve the problem in the compact systems, while the problem is less of an issue for the large footprint facilities.
In compact proton therapy systems, some radiation sources are closer to the patient than in the larger multi-room systems. An important source of secondary radiation, the ESS is located in the treatment room while multi-room systems have this component further upstream at larger distances. The principle of an ESS is to let the beam interact with a low Z material of variable water equivalent thickness to reduce the energy of the protons by inelastic interactions. To re-establish a narrow energy spread after a widening of the spectrum due to range straggling, the beam is bent in a magnetic field followed by passage through a movable momentum slit. Again, the proton beams interact with the materials of the ESS and result in neutron production. Some increase in neutron exposure can be expected when parts of the ESS, the divergence and momentum slits, are located inside the treatment room, which is the case for the IBA ProteusONE compact system. Secondary neutron scatter from the walls in the smaller vaults of compact facilities could potentially lead to an increase in the neutron dose for the patient compared with larger vaults in multi-room facilities.
The commercial solution of the company Mevion is to have a small cyclotron mounted directly on the gantry, not requiring any bending to point the pencil beam directly at the gantry rotation isocentre. The energy selection is performed without bending the beam, based on energy degradation in a cascade of carbon slab range shifters in the nozzle from 230 MeV to the desired energy (down to 70 MeV), in close proximity to the patient. It is claimed, however, that this set-up without beam-lines can be run at lower proton beam currents, because of the lower beam losses in energy selection, which reduces secondary neutron source intensity. This method of energy selection, however, can impact the proton beam characteristics. Howell et al. (2016) and Chen et al. (2013) have reported similar secondary neutron dose levels using this compact system compared with other proton beam-lines for passive scattering. Stichelbaut et al. (2014) performed a Monte Carlo simulation study investigating secondary neutron dose levels comparing the Mevion solution for pencil-beam scanning mode with the IBA ProteusONE compact system. They concluded that the secondary cancer risk did not increase for ProteusONE, but did increase by a factor of 3.74 for the gantry-mounted cyclotron with range-shifter-only energy selection, compared with a large multi-room layout facility using pencil-beam scanning.
4. In-room imaging and treatment verification
Limitation of volumes exposed to high therapeutic levels of dose outside the actual tumour is an aspect of patient-related radiation protection. Uncertainties in the dose delivery process and anatomical/positional changes of the patient have traditionally been addressed in advanced photon therapy by including safety margins around the volume irradiated at full therapeutic dose levels. Even if the value of the margin concept, as for pencil-beam scanning, is currently a debated issue, a level of robustness of the treatment plan delivery is usually accomplished by a exposure of surrounding tissues to assure coverage of the target volume. This additional exposure can be reduced by minimising the uncertainties in the treatment delivery process. Proton range uncertainties (Paganetti, 2012) and anatomical changes during the course of treatment are identified as the main components of uncertainty.
Proton therapy is considerably more sensitive to anatomical changes of/in the patient than intensity modulated radiotherapy or volumetric arc x-ray therapy. Nevertheless, image guidance in proton therapy was not at the level of state-of-the-art photon therapy. The fact that proton therapy is moving gradually into mainstream radiation therapy also results in more cross-talk with state-of-the-art photon radiotherapy. Up to a few years ago, x-ray images and image registration based on bony structures were generally used. Most indications typically treated with proton therapy in the past were targets where bony structures could be used as fiducial surrogates for tumour position. For more than a decade, image-guided radiation therapy in advanced photon therapy has been performed using the soft tissue information of on-board cone-beam computed tomography (CT) volumetric imaging. The first clinical application of on-board cone-beam CT in proton therapy dates from 2016. The additional patient dose of the volumetric imaging acquired with cone-beam CT or an in-room CT on-rails is typically several cGy. Similar to advanced photon therapy, in proton therapy, the balance between the value of daily volumetric imaging to reduce uncertainties in the process and the additional imaging exposure has to be found. Probabilistic planning (Unkelbach et al., 2009) and adaptive radiotherapy (Yan, 1997), where anatomical changes are compensated for by modifying the treatment plan regularly to re-establish clinical goals, will most probably become the preferred methodologies to reduce the impact of uncertainties in proton therapy.
The inevitable secondary radiation and activation generated in the patient during the delivery of proton therapy have also led to useful applications in treatment delivery verification (Knopf and Lomax, 2013). As a consequence of inelastic nuclear interactions of the traversing protons, a range of short-lived isotopes are created. In human tissues, 15O and 11C, with half-lives of 2 and 20 min, respectively, are produced predominantly and emit a characteristic positron through β+ decay. The annihilation 511-keV gamma ray pair can be captured using a positron emission tomography (PET) camera (Vynckier et al., 1993; Parodi and Enghardt, 2000). The captured signal intensity is related to nuclear interactions and does not directly show dose deposited by the proton beams as such, but the in-vivo proton range can be inferred. The nuclear interactions with protons also leave many nuclei of patient tissues in an excited state. They decay very rapidly by emitting neutrons or prompt gammas, with half-lives of 10−19–10−9 s. Prompt gammas have a wide energy spectrum, mainly between 0 and 7 MeV, with a few characteristic rays of the nuclei that are present or produced in the target. Stichelbaut and Jongen (2003) proposed to measure those gamma rays leaving the patient to deduce the proton range in vivo. Both prompt gamma imaging and PET in vivo have been demonstrated in clinical proton therapy practice (Nischwitz et al., 2015; Xie et al., 2017).
5. Discussion/Conclusion
Further to its commercialisation in the 1990s, proton therapy technology is moving into the next phase by becoming a mainstream radiation therapy modality. State-of-the-art delivery systems have become compact units making use of a versatile delivery technology. Pencil-beam scanning reduces secondary stray radiation in comparison with passive scattering, and does not require beam shaping and modification devices that can be activated. For radiation protection of the patient and staff, this can be considered an advantage, even when knowledge of stray neutron exposures and secondary cancer induction is incomplete, and few data on occupational exposure to clinical staff are available. Standardisation of technological solutions, such as pencil-beam scanning compact system units, will not only reduce the cost of proton therapy, but will also facilitate the acquisition of more generally usable radiation protection data. Compact proton therapy systems show differences with the large multi-room facilities in terms of activation and stray radiation, depending on the technology used. Further investigation is required to quantify the impact of these differences on radiation protection. Compact proton therapy systems can be embedded in existing hospital and radiation therapy facilities, which can be important in finding the balance for justified use of proton therapy compared with conventional and more economical radiotherapy techniques. Technology to reduce the impact of delivery uncertainties and anatomical changes are currently being developed, implemented, and, to some extent, adopted from image-guided radiation therapy.
