Abstract

Editorial
History is More or Less Bunk – or is It?
When the International X-ray and Radium Protection Committee [now the International Commission on Radiological Protection (ICRP)] was established back in 1928, its main concern was the occupational exposure of medical staff and the deterministic effects that were already being seen at this time. Old photographs showing doctors with missing fingers and scarred skin are a salient reminder to modern practitioners of the effects that radiation caused because the dangers were not appreciated and appropriate protection was not used. ICRP is celebrating its 90th anniversary in 2018, so it is timely that one of its publications should still have relevance in addressing occupational radiological protection of medically exposed workers (i.e. those involved in interventional procedures).
Interventional procedures using ionising radiation have revolutionised medicine in the last few decades for diagnosis, therapy, and palliation, resulting in more patients being offered treatments that would not have been possible previously. As technology continues to advance at an astonishing rate, so the interventional procedures become more complex, and some may take several hours to complete. New technologies also present new challenges for radiological protection, such as positron emission tomography-computed tomography guided interventions and selective internal radiation therapy.
Although the photographs of missing fingers are now a thing of the past, several studies have demonstrated increased opacities in the lens of the eye and, in some cases, cataracts among interventionalists. Education and training of personnel who perform and who are associated with interventional procedures cannot be emphasised enough, as good practices will reduce doses to patients and staff. A successful radiological protection programme for such procedures requires a team effort from all individuals involved, including doctors, nurses, radiographers, physicists, regulators, and administrators. In addition, staff need to be aware of the importance of wearing personal protective equipment, including dosimeters. Too often, dosimeters and protective lead glasses are not worn, either because of lack of volition or fear of recording high doses that may threaten continuing clinical practice. Interventionalists should take some responsibility for their own personal protection, and not just assume that this is the role of radiation supervisors and medical physicists. A change in behaviour is required to improve compliance with recommended personal protection, and education is key in achieving this goal.
Having worked as an interventional radiologist for nearly 30 years, I acknowledge that it takes a degree of self-discipline to constantly remember to wear several dosimeters, protective glasses, and a thyroid collar for each case, but keeping these items together and performing the same preparatory ritual is not difficult. One would hope that, nowadays, nearly all drivers setting off on a journey remember to put on their seat belt, and the start of an interventional procedure should include a similar routine with protective equipment.
Just over 100 years ago in 1916, several years before the existence of ICRP, Henry Ford gave an interview published in the Chicago Tribune in which he said:
History is more or less bunk … . We want to live in the present, and the only history that is worth a tinker’s damn is the history that we make today.
However, it can be argued that it is only by looking back at events of the past, and learning from them, that we can hope to adopt safer practices today and for the future. After all, this is at the core of ICRP’s business – to provide recommendations and guidance related to the use of ionising radiation – and, as demonstrated in this publication, medicine is a very important part of this. Some of the interventional procedures performed at the present time are likely to be obsolete in a couple of decades as they are superseded by the development of new treatments. If history had been considered ‘bunk’, we would not have the system of radiological protection that has evolved to date, but we should not forget that we are undoubtedly making history today.
C
C
Occupational Radiological Protection in Interventional Procedures
ICRP PUBLICATION 139
Approved by the Commission in October 2017
© 2018 ICRP. Published by SAGE.
Keywords: Occupational radiological protection; Interventional procedures; Exposure monitoring; Exposure of the lens of the eye; Protective garments
AUTHORS ON BEHALF OF ICRP
P. ORTIZ LÓPEZ, L.T. DAUER, R. LOOSE, C.J. MARTIN, D.L. MILLER,
E. VAÑÓ, M. DORUFF, R. PADOVANI, G. MASSERA, C. YODER
PREFACE
Over the years, the International Commission on Radiological Protection (ICRP), referred to below as ‘the Commission’, has issued recommendations and guidance on protection against the risks associated with ionising radiation. Publication 103 (ICRP, 2007a) contains the most recent update of these recommendations, and Publication 105 (ICRP, 2007b) summarises the application of the principles to medical exposures.
These publications are of a general nature, but the Commission also decided to address specific problems and difficulties that have been observed, especially in the rapidly evolving use of radiation in medicine. This has been achieved by means of concise publications focused and written in a style that is accessible to those directly involved in the procedures and who are, therefore, directly concerned in their daily work, and by making efforts to ensure wide circulation of such publications.
Examples of these concise publications are those concerned with interventional procedures, such as Publication 85 (ICRP, 2000b) on avoiding radiation injuries, Publication 117 (ICRP, 2010a) on radiological protection from fluoroscopically guided procedures outside the imaging department, and Publication 120 (ICRP, 2013a) on radiological protection in cardiology. These publications provide practical advice aimed at protecting all members of staff involved in the interventions.
However, a reader audience composed of hospital administrators, staff in charge of radiological protection of the hospital, dosimetry services staff, clinical applications specialists from the suppliers and maintenance companies, and regulators need advice on occupational exposure assessment, and tools and methods for occupational protection. They also need advice on specific issues, such as extremity and eye dose assessment (with and without eye protection), selection of protective garments (e.g. aprons, thyroid shielding, protective eye glasses), estimation of effective dose when an apron is worn, and auditing the interventional procedures when occupational doses are unusually high or low (the latter meaning that the dosimeter may not have been worn). Provision of guidance on these issues is the purpose of this publication.
Corresponding members were:
M. Doruff R. Padovani
G. Massera C. Yoder
Committee 3 critical reviewers were:
K. Applegate M.M. Rehani
Committee 2 reviewers were:
J. Hunt J.D. Harrison N. Petoussi-Henss
Main Commission critical reviewers were:
D. Cool C. Cousins
EXECUTIVE SUMMARY
1. Background
(a) Physicians in many medical and surgical specialties, assisted by nurses and radiographers (radiological technologists), perform interventions guided by radiological imaging as an alternative to conventional surgery. On average, these interventions are less invasive, their recovery periods are shorter, and – for many types of interventions – the complication rate is lower than for the equivalent conventional surgery. In addition, some patients who may not tolerate anaesthesia and conventional surgery, as well as lesions that were not previously accessible, can now be treated by less-invasive image-guided interventions. (b) The number of interventions guided by imaging is increasing greatly in both developed and developing countries. New types of interventions are also of increased complexity, require extensive use of x-ray imaging, and raise new issues of occupational protection. As well as interventional radiologists and cardiologists, other specialists, usually with little or no training in radiological protection, are now users of interventional guidance. (c) The considerable variation in occupational exposures observed for the same type of procedure suggests that radiological protection practices can be improved. Some recent ophthalmological studies described below, such as those performed under the coordination of the International Atomic Energy Agency (IAEA) programme, the Retrospective Evaluation of Lens Injuries and Dose (RELID study), have shown an increased incidence of radiation-related eye lens opacities in interventionalists when radiological protection devices were not used properly and radiological protection principles were not followed.
2. Purpose and scope of the publication
(d) In Publications 117 (ICRP, 2010a) and 120 (ICRP, 2013a), the Commission provided practical advice on occupational radiological protection for physicians and other healthcare personnel involved in interventional procedures. This publication provides guidance on exposure monitoring strategies, methods and options, radiological protection approaches and garments, their use and testing, the development of a radiological protection programme, education and training, and quality assurance of the programme implementation. The guidance is meant for medical physicists and other healthcare professionals in charge of occupational protection, personnel working in dosimetry services, clinical applications support personnel, regulators, and all those having an influence on the overall safety culture and on quality assurance and improvement. In addition, the guidance will be useful to those engaged in training, standardisation of equipment, and procedures; those with responsibilities for occupational health; hospital managers and administrators responsible for providing financial support for protection purposes; and professional bodies (interventionalists, medical physicists, nurses, and radiographers).
3. Uses of image-guided interventions, occupational exposures, and observed effects
3.1. Uses
(e) Interventions are usually guided by fluoroscopy, and radiographic cine-like series of images are taken to document both normal and abnormal conditions and the outcome of diagnosis or treatment. Interventions can also be guided by computed tomography (CT) imaging, with images taken while the interventionalist can step behind a mobile shield or out of the room, or by CT fluoroscopy, in which the interventionalist stays in the room when exposing the patient for obtaining images during device manipulation. The principal advantage of CT fluoroscopy over ordinary CT images is the real-time monitoring to access lesions that move within the body as a result of patient breathing or other motion. Its use allows interventions to be performed more rapidly and efficiently. On the other hand, CT fluoroscopy may result in relatively high radiation exposure to both the patient, the interventionalist, and other staff involved in the intervention. (f) X-ray image-guided therapeutic interventions such as radioembolisation with 90Y-labelled microspheres [selective internal radiation therapy (SIRT)] are an alternative method to treat patients with unresectable primary or secondary liver tumours. Several hospitals are exploring the use of real-time positron emission tomography (PET)-CT guidance during interventional procedures, such as for biopsies and/or radiofrequency ablations. 18F-FDG PET-CT imaging is performed within the suite to identify where an embolisation or biopsy should be performed, to check on effectiveness of interventions, and for early detection of residual disease (e.g. after radiofrequency ablation, so that ablation can be repeated, if necessary, in order to obtain the maximum therapeutic benefit).
3.2. Occupational exposures and observed effects
(g) While, with the appropriate protection, it is possible for interventionalists to keep their annual occupational effective dose below 10 mSv, and typically within a range of 2–4 mSv or less, some surveys have shown that individual occupational doses may exceed these values and have considerable variation. (h) The equivalent dose to the lens of the eye has received increased attention as evidence has become available that cataract development may have a much lower threshold for occurrence than was historically believed. The Commission’s recommendations have lowered the equivalent dose limit for the lens of the eye from 150 mSv year−1 to 20 mSv year−1, averaged over defined periods of 5 years with no single year exceeding 50 mSv. The nature of interventions guided by radiological imaging is such that, without protective measures for the eyes, personnel with a medium or high workload could receive doses to the lens of the eye that would exceed the new annual equivalent dose limit, and could result in eye lens opacities over time. (i) Several ophthalmological studies were conducted on a sample of interventional cardiologists and nurses who were attending cardiology congresses and who participated voluntarily under the coordination of the IAEA programme, the RELID study. Approximately 40–50% of interventionists and 20–40% of technicians or nurses, were found to have posterior subcapsular opacities compatible with injuries derived from exposure to ionising radiation. The incidence rate in interventionists was four to five times higher than that in unexposed individuals in the control group (approximately 40–50% vs 10%). Lifetime lens absorbed doses were estimated to reach several Gy in some cases. (j) Extremity equivalent dose may be of concern, as the dose to the interventionalist’s hand that is nearest to the irradiated patient volume can be high and requires specific hand monitoring. Values for annual lower extremity equivalent doses up to 110 mSv have been found, despite the use of a protective curtain hanging on the side of the treatment couch. This exposure is attributed to the gap between the protective curtain and the floor, the size of which is dependent on the height of the x-ray table during exposure.
4. Occupational exposure monitoring and exposure evaluation
(k) A survey performed within the IAEA Information System on Occupational Exposure in Medicine, Industry and Research (ISEMIR) (IAEA, 2014b) showed that 76% of interventional cardiologists stated that they always used their dosimeters and 45% used two dosimeters. This survey relied on self-reporting and may overestimate true dosimeter use. In addition, in some parts of the world, there is a lack of proper monitoring of radiation doses to professionals involved in interventional procedures, and individual dosimeters are often not worn regularly. (l) In addition to assessing effective dose, occupational exposure monitoring in interventions guided by radiological imaging should include an estimate of the equivalent dose received by the lens of the eye and, in some cases, the extremities.
4.1. Assessment of effective dose
(m) The combination of the readings of two dosimeters, one shielded by the apron and one unshielded above the apron at collar level, provides the best-available estimate of effective dose (as has been stated by the Commission in previous publications). The dosimeter under the apron also provides evidence that an apron that provided sufficient shielding was worn regularly.
4.2. Assessment of equivalent dose to the eye
(n) The dosimeter over the apron, at collar level on the side of the interventionalist closer to the irradiated volume of the patient, not only contributes to assessing effective dose, but also provides a reasonable estimation of the equivalent dose to the lens of the eye and the head. (o) Improved computational methodologies need to be developed to assess occupational doses, including equivalent dose to the lens of the eye, in high-dose procedures. These methods may be helpful to audit the regular and proper use of personal dosimeters and to assess the need for additional protection (e.g. protective glasses). Research programmes should pursue the development of computational technologies (not requiring dosimeters) together with personnel position sensing devices to assess personnel doses, including dose to the eye.
4.3. Equivalent dose to the extremities
(p) Assessment of equivalent dose to the hands in some specific complex interventional procedures needs more attention in the future. Finger dosimeters may be needed if the hand is very close to the direct x-ray beam. Similarly, assessment of exposure to the lower extremities, including the feet, will also require increased attention, especially when protective curtains are not available or there is a gap between the curtains and the floor. A gap may be present depending on the height of the table during the intervention.
4.4. Examples of errors with the use of dosimeters and indirect approaches to correct the situation
(q) Examples of errors include not using the assigned dosimeter, wearing a dosimeter over the apron that was intended for use under the apron, wearing a ring dosimeter on the incorrect hand, wearing a dosimeter assigned to another person, or losing a dosimeter. (r) Indirect approaches to dose assessment may be useful in identifying a lack of compliance in wearing personal dosimeters, and also in estimating occupational doses when personal dosimeters have not been used. These approaches may be based on area dosimetry of the scatter radiation near the patient (e.g. at the C-arm), together with conversion coefficients from patient-related quantities such as kerma-area product for different types of procedures and geometries to the dose to the lens of the eye of workers.
5. Guidance on occupational radiological protection
5.1. Relationship between patient and staff exposure
(s) Occupational protection in interventions guided by radiological imaging is closely related to patient protection, and most actions to protect the patient also protect the staff. There are, however, additional measures and protective devices that protect the staff alone. The use of these devices should not interfere with the manipulations of the procedure, nor increase patient exposure.
5.2. Protection by shielding devices
(t) Shielding aprons should be worn by all interventional staff working inside the x-ray room. Aprons usually contain the equivalent of 0.25 mm, 0.35 mm, or 0.5 mm of lead. Some designs overlap at the front to provide protection of 0.5-mm lead equivalence, with 0.25-mm lead equivalence elsewhere. Transmission is typically between 0.5% and 5% in the range 70–100 kV (i.e. attenuation factor between 200 and 20). Aprons shield the trunk against scattered radiation, but parts of the body including the head, arms, hands, and legs are not protected by the apron. These parts of the body need to be considered in the radiological protection programme. (u) The most important factor in protection of the head is the proper use of ceiling-suspended lead acrylic shields. They should always be included in interventional installations, as they can reduce doses to the whole head and neck by a factor of 2–10, depending on how efficiently they are positioned. (v) Staff, such as nurses and anaesthesia personnel, who need to remain near the patient may benefit from the additional protection provided by movable (rolling) shields that can be positioned between them and the source of scattered radiation. (w) As described in Point (h), under occupational exposure, the equivalent dose to the lens of the eye can exceed the new dose limit if protective measures are lacking. Over time, this could result in lens opacities. Conversely, if the interventional fluoroscopy equipment is operating correctly, procedure protocols have been optimised, the operator has been trained, and protective tools for the eyes are being used, the dose to the lens of the eye should be lower than the dose limit. (x) A close fit of leaded glasses to the facial contours, particularly around the sides and underside of the glasses, is important because the clinician is looking at the image monitor during the x-ray exposures. As a result, the eyes may be irradiated from the side and below. (y) Lead drapes attached to the bottom edge of the ceiling-suspended shield, as well as shielding drapes and pads, can be effective in protecting the hands in some procedures. This type of protection should be considered for procedures where the operator needs to be close to the source of scattered radiation (i.e. the irradiated volume of the patient). When placing disposable drapes on the patient, attention is required to avoid having the drapes in the primary beam, which might increase patient and operator exposure. (z) Staff who stand near the patient table during interventions should be aware that the radiation field is more intense in the region adjacent to the beam entrance side. This is particularly important when projections are oblique or lateral. Doses to the head, upper body, and hands of the interventionalist from fluoroscopy with the tube positioned under the table will be substantially lower than the doses received by the lower extremities. (aa) This is particularly true when no shielding curtains for the lower extremities are available, and when the table is at a higher position, so that the feet may stay unprotected even if the curtains are in place. Rolling lead shields, when available, decrease the effective dose to staff by more than 90% if used properly. (bb) In summary, all staff in the room should wear protective aprons. Wraparound aprons are desirable for individuals who may not be able to face towards the patient at all times when the beam is on. The interventionalist should be protected by ceiling-suspended screens, table-suspended curtains, and shielding drapes when feasible. Staff can also reduce doses received during the use of high-dose acquisition modes (e.g. image acquisition series and digital subtraction angiography) and during injection of contrast media using an automatic injector by stepping back and increasing the distance to the patient. Staff, such as nurses and anaesthesia personnel, who need to remain near the patient can benefit from protection by movable screens. Other personnel should increase protection by increasing their distance from the irradiated volume of the patient or, if possible, leaving the room during image acquisition.
5.3. Protection of the embryo and fetus
(cc) After a pregnant woman has declared her pregnancy, her working conditions should ensure that the additional dose to the conceptus does not exceed 1 mSv during the remainder of the pregnancy. (dd) Current data do not justify precluding pregnant woman from performing interventions guided by radiological imaging completely if they follow proper procedures. Pregnancy, in any case, requires that the employer carefully reviews the exposure conditions and other aspects of occupational hazards (e.g. back pain with use of lead aprons) of the pregnant worker.
6. Quality assurance
(ee) Quality assurance with regular documented checks to confirm that professionals involved in interventions guided by radiological imaging always wear their dosimeters and protective equipment, including eyewear, is very important. (ff) Acceptance tests for protective devices are crucial; some supplies of defective protective clothes have been documented. In addition, handling protective devices with care (e.g. avoid folding) and regular testing are required as part of the quality assurance and improvement programme, as described in Section 5.
7. Education and training
(gg) Initial and continuing education and training of professionals in occupational safety and radiological protection is required. This is especially important regarding safety culture and the proper use of imaging equipment and radiological protection tools (e.g. ceiling-suspended shields and/or leaded eyewear and shielding curtains). (hh) Use of real-time active dosimeters not only helps in optimising protection of specific high-dose procedures, but also contributes to the education of professionals on the level of doses being received. (ii) In addition to knowledge of general radiological protection, hospital staff in charge of occupational protection, dosimetry services staff, clinical applications specialists from suppliers, and regulators need knowledge of clinical practice, the x-ray equipment used in interventions, strategies for occupational exposure assessment, the protection methods, and selection and testing of protective garments.
8. Availability of key professionals for radiological protection
(jj) The role of the medical physicists or others in charge of creating and maintaining a radiological protection and training programme is crucial. They are part of the team that ultimately designs and implements optimal radiological protection and care by the interventionalists, radiographers, and nurses.
GLOSSARY
Absorbed dose (D) The quotient of the mean energy imparted to an element of matter by ionising radiation and the mass of the element.
Absorbed dose is the basic physical dose quantity and is applicable to all types of ionising radiation and to any material. Absorbed dose is a measurable quantity for which primary standards exist. In the International System of Units (SI), the unit for absorbed dose is J kg−1, and its special name is gray (Gy). Individuals, other than staff, who care for and comfort patients. These individuals include parents and others, normally family or close friends, who hold children during diagnostic procedures or may come close to patients following the administration of radiopharmaceuticals or during brachytherapy (ICRP, 2007a). See Tissue reaction. Used to express dose per unit intake of a radioactive substance, but sometimes also used to describe other coefficients linking quantities or concentrations of activity to doses or dose rates, such as the external dose rate at a specified distance above a surface with a deposit of a specified activity per unit area of a specified radionuclide (ICRP, 2007a). The value of the effective dose or the equivalent dose to individuals from planned exposure situations that shall not be exceeded (ICRP, 2007a). Dosimeter unshielded by the protective apron. Dosimeter shielded by the protective apron. The tissue-weighted sum of the equivalent doses in all specified tissues and organs of the body, given by the expression:
The sum is performed over all organs and tissues of the human body considered to be sensitive to the induction of stochastic effects. The tissue weighting factors are age- and sex-averaged, and intended to apply as rounded values to a population of both sexes and all ages. An organisation, corporation, partnership, firm, association, trust, estate, public or private institution, group, political or administrative entity, or other persons designated in accordance with national legislation, with recognised responsibility, commitment, and duties towards a worker in her or his employment by virtue of a mutually agreed relationship. A self-employed person is regarded as being both an employer and a worker (ICRP, 2007a). The dose in a tissue or organ T given by:
Procedures comprising guided therapeutic and diagnostic interventions, by percutaneous or other access, usually performed under local anaesthesia and/or sedation, with fluoroscopic or computed tomography (CT) imaging used to localise the lesion/treatment site, monitor the procedure, and control and document the therapy (ICRP, 2000b). Three-dimensional (cone beam CT) imaging using fluoroscopic equipment is also used in some interventional procedures. The special name for the SI unit of absorbed dose: 1 Gy = 1 J kg−1. The absorbed dose DT, averaged over the tissue or organ T, which is given by:
Exposure incurred by patients as part of their own medical or dental diagnosis or treatment; by persons, other than those occupationally exposed, knowingly, while voluntarily helping in the support and comfort of patients; and by volunteers. This refers to all exposures incurred by workers in the course of their work, with the exception of: (1) excluded exposures and exposures from exempt activities involving radiation or exempt sources; (2) any medical exposure; and (3) the normal local natural background radiation. However, because of the ubiquity of radiation, the Commission limits its use of ‘occupational exposures’ to radiation exposures incurred at work as a result of situations that can reasonably be regarded as being the responsibility of the operating management. Excluded exposures and exposures from exempt practices or exempt sources do not generally need to be accounted for in occupational protection (ICRP, 2007a). Quantities used in practical applications for monitoring and investigating situations involving external exposure. They are defined for measurements and assessment of doses in the body. In internal dosimetry, no operational dose quantities have been defined that directly provide an assessment of equivalent or effective dose. Different methods are applied to assess the equivalent or effective dose due to radionuclides in the human body. They are mostly based on various activity measurements and the application of biokinetic models (computational models). The process of determining what level of protection and safety makes exposures, and the probability and magnitude of potential exposures, as low as reasonably achievable, economic and societal factors being taken into account (ICRP, 2007a). In medical imaging and radiotherapy procedures, optimisation of radiological protection means keeping the doses ‘as low as reasonably achievable, economic and societal factors being taken into account’, and is best described as management of the radiation dose to the patient to be commensurate with the medical purpose. The operational quantity for individual monitoring is the personal dose equivalent Hp(d), which is the dose equivalent in soft tissue at an appropriate depth, d (in mm), below a specific point on the human body. The unit of personal dose equivalent is J kg−1, and its special name is sievert (Sv). The specified point is usually given by the position where the individual’s dosimeter is worn. For monitoring effective dose, the operational quantity Hp(10) is used, and for assessment of the dose to the skin, hands, and feet, the personal dose equivalent, Hp(0.07) is used. A depth d = 3 mm is adequate for monitoring the dose to the lens of the eye. In practice, however, in many countries, calibration of dosimeters in terms of Hp(3) has not been implemented, but Hp(0.07) can be used for the same monitoring purpose for photon radiation, which is the case in interventions guided by radiological imaging. A set of principles that apply to radiation sources and to the individual in controllable exposure situations. The principle of justification and the principle of optimisation of protection are source related and apply in all exposure situations. The principle of application of dose limits is individual related and only applies in planned exposure situations (ICRP, 2007a). A dimensionless factor by which the organ or tissue absorbed dose is multiplied to reflect the higher biological effectiveness of high-linear energy transfer (LET) radiations compared with low-LET radiations. It is used to derive the equivalent dose from the absorbed dose averaged over a tissue or organ (ICRP, 2007a). The special name for the SI unit of equivalent dose, effective dose, and operational dose quantities. The unit is J kg−1. In the context of this publication, staff are healthcare workers (see Worker) who participate in the care of a patient during a radiological procedure (e.g. physicians, nurses, radiographers) or who may be exposed to radiation from medical imaging equipment during the course of their work (e.g. equipment service personnel, janitorial staff). Malignant disease and heritable effects for which the probability of an effect occurring, but not its severity, is regarded as a function of dose without threshold. Dose estimated to result in 1% incidence of tissue reactions (ICRP, 2007a). Injury in populations of cells, characterised by a threshold dose and an increase in the severity of the reaction as the dose is increased further. Tissue reactions are also termed ‘deterministic effects’. In some cases, tissue reactions are modifiable by postirradiation procedures including biological response modifiers (ICRP, 2007a). A factor by which the equivalent dose in a tissue or organ T is weighted to represent the relative contribution of that tissue or organ to the total health detriment resulting from uniform irradiation of the body (ICRP, 1991). It is weighted (ICRP, 2007a) such that:
Any person who is employed, whether full time, part time, or temporarily, by an employer, and who has recognised rights and duties in relation to occupational radiological protection. Workers in medical professions involving radiation are occupationally exposed (ICRP, 2007).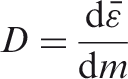
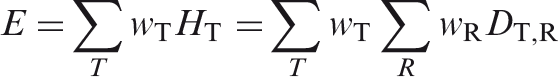
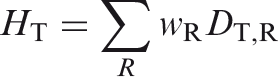
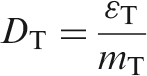
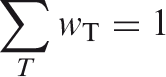
1. INTRODUCTION
1.1. Background
(1) Physicians in many medical and surgical specialties, usually assisted by nurses and radiographers, perform interventions guided by radiological imaging (NCRP, 2010) as an alternative to more complex and higher risk conventional surgery. This approach has many advantages: the interventions are less invasive than conventional surgery, recovery periods are shorter, and, for some procedures, the complication rate is lower (NCRP, 2010). (2) Some physicians perform interventions involving multiple organ systems (e.g. radiologists), and others perform procedures within one or two organ systems alone (e.g. cardiologists, gastroenterologists, and urologists). Some interventions once performed primarily by radiologists, such as endovascular procedures to treat lower extremity arterial disease, are now performed increasingly by vascular surgeons and cardiologists (Goodney et al., 2009; Harris et al., 2011). In the USA, radiologists now perform less than 20% of these procedures (Goodney et al., 2009), and less than 35% of all fluoroscopically guided interventional procedures (NCRP, 2009). (3) The increasing number, diversity, and complexity of new types of interventions guided by radiological imaging means that the benefits from these interventions continue to expand. However, they lead to an increase in exposure that appears to offset dose reductions obtained from improvements in technology (Kim et al., 2008). Moreover, occupational doses to interventionalists are among the highest observed in personnel working in medicine (Padovani et al., 2011). In a number of healthcare settings, there is a lack of proper monitoring of occupational radiation doses to professionals, and as a consequence, there is a lack of reliable data on occupational doses (Padovani et al., 2011; IAEA, 2014b). Too often, personal monitoring badges are worn intermittently, or are worn improperly (Padovani et al., 2011) or not provided. In some developing countries, no dose monitoring system is in place (Tsapaki et al., 2009). In addition, there is difficulty in comparing reported dosimetry results because of significant differences in dosimetric methods used in each study (Kim et al., 2008), as well as lack of consensus on the number of dosimeters that may be used, and where the dosimeters should be worn on the body. The fact that none of the algorithms estimate effective dose adequately for all types of procedures poses difficulties in reaching a worldwide consensus regarding which of them should be used. (4) The Commission reviewed recent epidemiological evidence suggesting that there are some tissue reaction effects, particularly those with very late manifestation, where threshold doses are or might be lower than previously considered. This is the case for the lens of the eye (ICRP, 2011). Recent studies have shown that there is an increased incidence of radiation-related eye lens opacities in interventional cardiologists when radiological protection devices are not used properly and radiological protection principles are not followed (Vañó et al., 1998, 2010, 2013a; Ciraj-Bjelac et al., 2010; Rehani et al., 2011; Jacob et al., 2012). Fairly high radiation doses to the hands and legs of interventionalists, and hair loss in the portions of the legs not shielded by a protective device (Balter, 2001) have been observed. The considerable variation in operator doses observed for the same type of procedure indicates that radiological protection practices can be improved (Kim and Miller, 2009). (5) Physicians involved in interventional procedures vary in their level of training in radiological protection. For example, in many countries, all radiologists receive training in radiation physics, radiation biology, and radiological protection and safety as part of their radiology education, but physicians in other medical disciplines receive variable amounts of education in radiation-related topics, and may or may not be examined in these areas as part of the certification process. Publication 113 (ICRP, 2009b) provides advice and recommendations on education and training, the professionals to be trained, objectives, contents, management approaches, approximate time needed to educate and train a wide variety of health professionals, accreditation, and certification. (6) Several national and international medical societies have adopted guidelines to improve occupational protection and to avoid occupational radiation injuries, such as eye lens opacities (Miller et al., 2010; Durán et al., 2013). (7) The Commission has provided practical advice regarding occupational radiological protection for interventionalists and other healthcare workers involved in x-ray-guided interventions in Publications 85 (ICRP, 2000b), 117 (ICRP, 2010a), and 120 (ICRP, 2013a).
1.2. Purpose of the publication
(8) The purpose of this publication is to provide guidance on occupational protection to personnel involved in the interventions, but also to hospital administrators, medical physicists and those in charge of occupational protection, clinical applications support personnel from supplier companies, staff from dosimetry services, regulators, and all those having an influence on the overall safety culture of the hospital. (9) This guidance includes tools and methods for occupational protection and exposure monitoring strategies, selection, use and testing of protective garments, development of a radiological protection programme, education and training, and quality assurance for the programme implementation.
1.3. Scope of the publication
(10) The guidance provided in this publication applies to interventions guided by radiological imaging, including computed tomography (CT), cone beam CT, and positron emission tomography (PET-CT), as well as selective internal radiation therapy (SIRT). However, as the vast majority of interventional procedures relate to interventions guided by x-ray fluoroscopy and image acquisition series, the text of this publication refers to x-ray imaging, unless otherwise stated. Sections related to PET-CT and SIRT are included because they are often performed in interventional suites, and in conjunction with interventional radiology. Quantities and units relevant to interventional procedures are summarised in Annex B. (11) For the purpose of this publication, interventional procedures are guided diagnostic and therapeutic interventions performed via percutaneous or other access routes, usually with local anaesthesia and/or intravenous sedation, which use ionising radiation in the form of fluoroscopy, CT, or PET to localise or characterise a lesion or diagnostic and/or treatment site; monitor the procedure; and/or control and document therapy.
2. ISSUES
2.1. Interventional procedures
2.1.1. Interventional fluoroscopy procedures
(12) There has been a large increase in the number of interventional procedures performed annually throughout the world. In the USA, interventional fluoroscopy procedures were the third largest source of medical exposure of patients in 2006, accounting for 14% (0.43 mSv year−1) of medical radiation exposure (NCRP, 2009) in terms of collective effective dose. Cardiac fluoroscopy procedures, including diagnostic cardiac catheterisation, represented 28% of all interventional fluoroscopy procedures, but accounted for 53% of the interventional fluoroscopy exposure. In 36 European countries, the frequency of all medical interventions guided by fluoroscopy ranges from 0.03% to 2.74%, with an average of 0.6% of all x-ray procedures. In terms of collective doses, medical radiation exposure in interventional procedures contributes from 0.001 to 0.34 mSv year−1, corresponding to 0.4–28.7% of total radiation collective doses (EC, 2015). Seven of 11 developing countries surveyed as part of an IAEA project demonstrated a 50% or greater increase in the number of interventional procedures performed between 2004 and 2007 (Tsapaki et al., 2009).
2.1.2. Interventional computed-tomography-guided procedures
(13) Interventions can also be performed with CT guidance. Although relatively few data are available on the number of CT-guided interventions that are performed or on temporal trends, it is clear that the numbers and types of procedures are increasing. For example, the percentage of image-guided percutaneous lung biopsies performed with CT guidance at the Mayo Clinic in the USA increased from 66% in 1996–1998 to 98% in 2003–2005 (Minot et al., 2012). The remainder were performed with fluoroscopic guidance. CT is used primarily to guide biopsy of small or deep lesions in the chest, abdomen, and pelvis that are not seen well with ultrasound or fluoroscopy, as well as to guide needle placement for other procedures. (14) CT-guided interventions can be performed by using intermittent CT scans while the physician steps behind a mobile shield or out of the scanner room, or by using CT fluoroscopy, with physician-controlled intermittent or continuous CT exposure during needle or device manipulation. CT fluoroscopy facilitates CT-guided biopsy procedures by allowing visualisation of the needle trajectory from skin entry to the target point. CT fluoroscopy is applicable to a wide variety of non-vascular interventions (Daly and Templeton, 1999). It is used for needle guidance during drainage of fluid collections and abscesses; spinal pain management; tumour ablation; and percutaneous needle biopsy in the neck, chest, spine, abdomen, and pelvis (Buls et al., 2003; Joemai et al., 2009; Hoang et al., 2011; Trumm et al., 2012). The principal advantage of CT fluoroscopy over standard CT is the ability to use real-time monitoring to access lesions that move within the body as a result of patient breathing or other motion. Its use allows interventions to be performed more rapidly and efficiently (Gianfelice et al., 2000b), and it is therefore popular. On the other hand, CT fluoroscopy also results in higher radiation doses to both the patient and the physician operator (Gianfelice et al., 2000a; Saidatul et al., 2010; Kim et al., 2011). As CT fluoroscopy images are noisier than conventional CT, this technique is predominantly used in cases of moving objects of high contrast, such as in lung biopsies.
2.1.3. Interventions for selective internal radiation therapy
(15) Less than 20% of patients with primary or metastatic liver cancers are curable at presentation. Therefore, palliative therapies such as interventional procedures for radioembolisation with the pure beta-emitter 90Y-labelled microspheres (SIRT) and other loco-regional therapies have become alternative methods to treat patients with unresectable liver tumours (Camacho et al., 2015). (16) After catheterisation of the hepatic arteries, 90Y microspheres (maximal beta-energy 2.27 MeV, half-life 64.1 h) are delivered under fluoroscopic control. Two types of 90Y microspheres are used: resin microspheres (SIR-Spheres, SIRTEX, Lane Cove, Australia; diameter 20–60 µm) and glass microspheres (TheraSphere, Nordion, Ottawa, Ontario, Canada; diameter 22 µm). The rationale for SIRT is the dominant hepatic arterial supply of malignant lesions. SIRT has demonstrated a significant increase in patient survival time (Bester et al., 2012). (17) SIRT is usually performed in two steps: in the first step, diagnostic angiography is combined with protective occlusion of non-target arteries. Then, shunting into the lung is estimated by means of a Single Photon Emission Computed Tomography (SPECT) scan of the lung and upper abdomen with 99mTc-MAA particles injected into the hepatic artery. If lung shunting is <10%, SIRT with full 90Y activity delivery is acceptable. A reduced amount of 90Y activity (20–40% less) is recommended when shunting is 10–20% (SIRTEX). When shunting is >20%, SIRT is contraindicated. The second step, usually performed 1 or more days later, is catheterisation of the hepatic artery or other arteries supplying the hepatic tumours, and administration of the microspheres. Temporary balloon occlusion of non-target arteries and antireflux catheterisation during this second step are alternatives to protective occlusion prior to microsphere delivery (Hagspiel et al., 2013; Fischman et al., 2014). (18) The second step includes dose calculation, preparation of the 90Y spheres, and delivery via a catheter into the hepatic artery. Typical activities are 2–3 GBq for resin spheres (Jakobs et al., 2007) and 3–7 GBq for glass spheres (Andrews et al., 1994). Target dose is typically 120 Gy (range 80–150 Gy). Nuclide distribution may be examined either by planar or SPECT Bremsstrahlung imaging or PET-CT. PET-CT has higher spatial resolution, and quantification of delivered activity may be more accurate (Camacho et al., 2015).
2.1.4. Use of positron emission tomography in interventional procedures
(19) PET is increasingly playing a role in image-guided interventions as it provides an image guidance technique for metabolically active targets that are inconspicuous, difficult to visualise, or not detected by CT or magnetic resonance imaging (Ryan et al., 2013a). Several hospitals are exploring, as part of their research programme, the use of real-time PET-CT guidance during interventional procedures, such as for biopsies and/or radiofrequency ablations (Purandare et al., 2011; Venkatesan et al., 2011; Ryan et al., 2013a; Aparici et al., 2014; McLoney et al., 2014), and there is current development of real-time fusion imaging using x-ray CT and PET imaging (Purandare et al., 2011; Beijst et al., 2016). The use of PET and multi-modality fusion imaging within the interventional suite can also assist in identifying the location for effective embolisation or biopsies, as well as providing immediate assessment of treatment effectiveness.
2.2. Type and energy of radiation in interventional procedures
(20) Most interventional procedures are performed with a combination of fluoroscopy and image acquisition series. Beam spectra vary with tube voltage and filtration, ranging from 50 to 125 kVp and added filtration of up to 1 mm copper (NCRP, 2010). The beam quality and operating parameters, such as tube voltage and current, pulse duration, and often beam filtration, are driven by the system's automatic exposure control (NCRP, 2010). Higher beam penetration (i.e. higher kVp and filtration) is used for fluoroscopy in low-dose-rate modes (e.g. 88–114 kVp and a half-value layer of 8–10 mm Al), while lower tube voltage is used for image acquisition mode (e.g. 68–84 kVp and a half-value layer of 3.5–4.0 mm Al) (Principi et al., 2014). In some equipment, spectral shaping for image acquisition is achieved by combining low tube voltage (for better visualisation of iodine-containing contrast media) with increased filtration (for limiting the higher patient dose associated with the lower tube voltage) (NCRP, 2010). The distribution of scattered radiation around the patient, which is most relevant to occupational exposure, is discussed in Section 5. (21) In CT fluoroscopy, the tube voltage ranges from 80 to 140 kVp. In PET-CT examinations using 18F-FDG, the photon energy of 511 keV is much higher than the energy of scattered photons in conventional interventional procedures (NCRP, 2010). The maximal beta energy from 90Y used in SIRT procedures is 2.27 MeV. As the vast majority of interventional procedures relate to those guided by x-ray imaging, the text of this publication refers to them unless otherwise stated.
2.3. Occupational exposure
2.3.1. Effective doses
(22) Summaries and compilations of data on occupational exposure are available (Kim et al., 2008, 2012; ICRP, 2010a; NCRP, 2010). While it is certainly possible for active interventionalists to keep their annual occupational effective dose below 10 mSv, and typically within an effective dose range of 2–4 mSv or less (Miller et al., 2010), surveys have shown that individual occupational doses may exceed these values (Padovani et al., 2011). (23) Annual effective doses incurred by staff depend on their function and role in the team (primary interventionalist, radiographer, nurse, anaesthesia provider), the number of interventions, the medical specifics and complexity of the cases, the patient population (e.g. paediatric patients, obese patients), and other factors such as the skill of the interventionalist and equipment as well as the use of fluoroscopic and cine times. In a review of the literature, Martin (2009) estimated that a case load of 500 cardiology procedures per year would result in an annual effective dose of approximately 2 mSv for the primary interventionalist. A maximum annual dose of 1.2 mSv [Hp(10) measured under apron] was observed for cardiologists at a Glasgow hospital (Martin, 2009). Other types of procedures that result in an effective dose per procedure greater than 10 µSv for the interventionalist might lead to annual effective doses as high as 10 mSv depending on whether thyroid shields are used. Lie et al. (2008) reported a maximum annual effective dose derived from combining the readings of two dosimeters, one under and one over the apron, of 11 mSv with a mean of 5 mSv. The outcome of a review of monthly effective doses (E), obtained during 2011 and 2012, performed by a dosimetry service provider in the USA is shown in Fig. 2.1. In total, 102,199 observations refer to workers monitored with two dosimeters (one over and one under the apron) and 196,526 observations refer to workers monitored with a single dosimeter located over the apron at the collar. The outcome revealed mean values of estimated effective dose of 0.12 and 0.27 mSv, with median values of 0.03 and 0.1 mSv, respectively (Yoder and Salasky, 2016). (24) Sánchez et al. (2012) found monthly median under-apron doses of 0.11 mSv for cardiologists and <0.01 mSv for nurses in a study of 43 workers who conducted 1467 procedures. The over-apron doses were 0.4 mSv month−1 for both cardiologists and nurses. The authors noted that perhaps as many as 50% of the cardiologists did not use their dosimeters correctly, often failing to wear the over-apron dosimeter (Sánchez et al., 2012). A multi-centre study on 39 physicians and nine assistants performing nine different types of procedures in 14 hospitals in Germany showed that the median body dose per procedure was 16 µSv for an unshielded person; the partial-body equivalent dose per procedure was 2.8 µSv to the lens of the eye, 4.1 µSv to the thyroid, 44 µSv to one of the feet, and 75 µSv to one of the hands. High exposures were measured to the hands, in some cases above the limit of 500 mSv (Häusler et al., 2009). (25) As well as the primary interventionalist, other staff may also be subject to significant exposure, such as anaesthesia providers. Kong et al. (2015) showed that radiation exposure of anaesthesia providers not only depends on their workload, but largely varies with their positions and beam projections during interventional procedures. Beam projection accounts for a factor of 10 in effective dose and 200 in dose to the lens of the eye. A position close to the patient combined with left lateral projection causes higher exposure. Optimal arrangement of the anaesthesia device was found to be useful to reduce exposure. (26) Data on occupational exposure from CT fluoroscopy guided interventions are limited. The highest doses are received by the physician’s hands, eyes, and thyroid (Saidatul et al., 2010). Use of thyroid shields provides substantial protection for the thyroid (Saidatul et al., 2010), which is especially important for younger professionals. Since average patient dose varies according to the type of procedure (Leng et al., 2011), average physician effective dose per case also varies according to the type of procedure, as would be expected; reported values measured over apron ranged from 2 to 25 μSv for Hp(10), with maximum values as high as 0.4 mSv per procedure (Paulson et al., 2001; Teeuwisse et al., 2001; Joemai et al., 2009). A variety of technical approaches and protection methods have been developed that can reduce occupational dose (Daly and Templeton, 1999; Paulson et al., 2001; Carlson et al., 2005; Hoang et al., 2011). (27) The occupational radiation exposure from transcatheter aortic valve replacement or transcatheter aortic valve implantation depends on the approach (transfemoral or transapical). Values of Hp(10) up to 0.23 mSv in a single procedure were obtained by Shatila (2015) from the over-apron dosimeter of the primary operator (median value 0.11 mSv), as well as significant exposures to eight of 10 other workers.
Distribution of effective dose (E) assessed by two dosimeters (one over and one under the apron) (top) and one dosimeter (over the apron) (bottom) (Yoder and Salasky, 2016).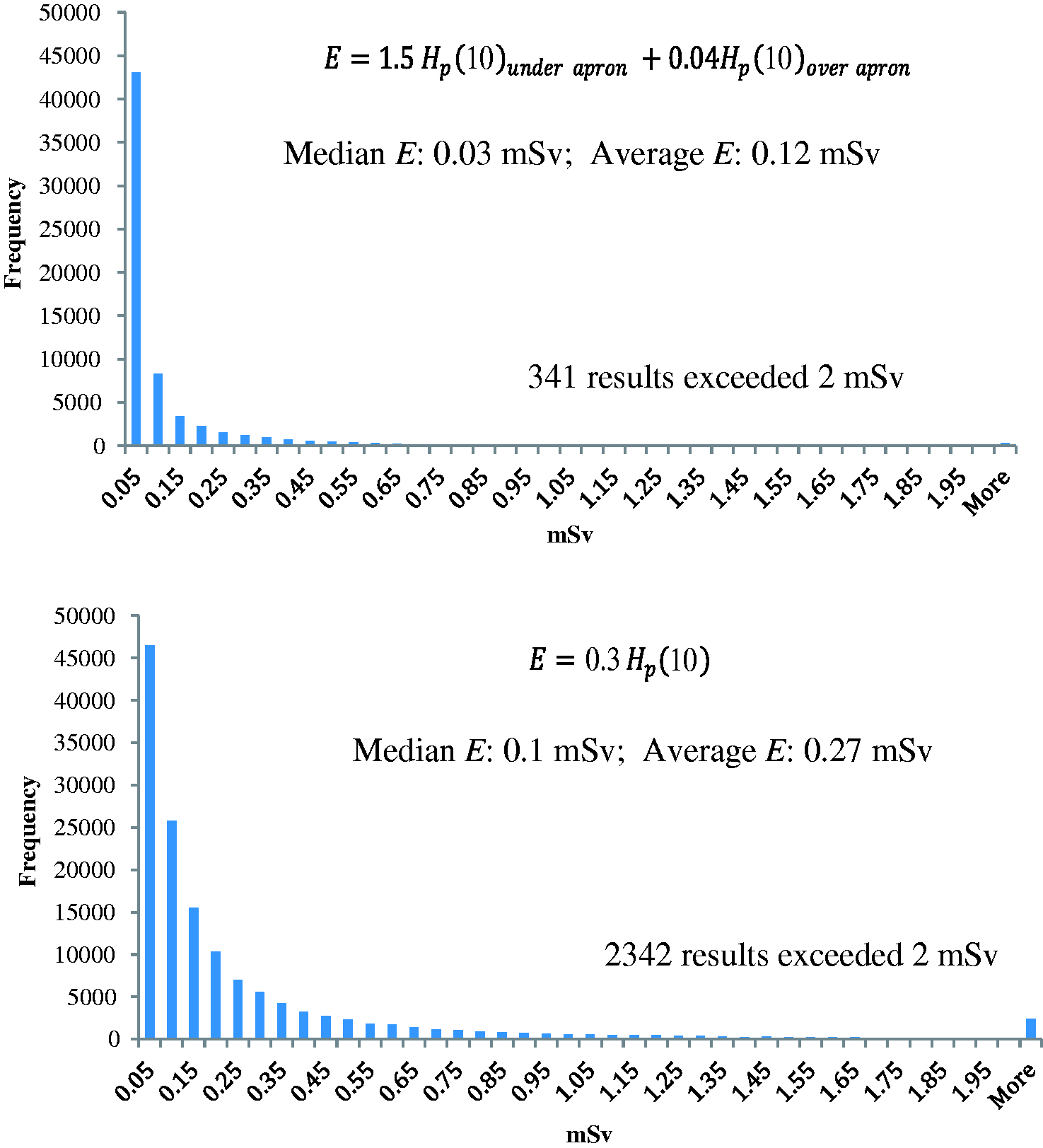
2.3.2. Equivalent dose to the lens of the eye
(28) The Commission issued a statement in 2011 published as part of Publication 118 (ICRP, 2012) after reviewing epidemiological evidence suggesting that there are some tissue reactions, particularly those with very late manifestation, where threshold doses are or might be lower than considered previously. For the lens of the eye, the threshold in absorbed dose is now considered to be 0.5 Gy. For occupational exposure in planned exposure situations, the Commission now recommends an equivalent dose limit for the lens of the eye of 20 mSv year−1, averaged over defined periods of 5 years, with no single year exceeding 50 mSv. Without protective eyewear, the dose to the lens of the eye may become the operationally restrictive dose (Lie et al., 2008; Korir et al., 2012), and the revised dose limit may be exceeded. (29) Most data on eye exposures are derived either from static experiments with phantoms or from individual monitors placed on the neck. A few studies have placed dosimeters closer to the eye on the forehead. Lie et al. (2008) compared thermoluminescence dosimeters (TLDs) placed near the left eye and between the eyes for 144 procedures, mainly cardiac. The median equivalent dose to the lens of the eye was observed to be 23 µSv per procedure, and the kerma-area product of the primary beam towards the patient was 0.4 µSv Gy−1 cm−2. The left eye dose tended to be higher than that between the eyes due to the left eye being closer to the x-ray generator. Kicken et al. (1999) assessed the absorbed dose at the forehead for under-couch and over-couch x-ray systems. They found an average absorbed dose for the operator and assistant of 8 and 6 µGy per procedure, respectively, at one hospital; 16 and 14 µGy, respectively, at a second hospital; and 43 and 28 µGy, respectively, at a third hospital. The first two hospitals used an under-couch system and the third hospital used an over-couch x-ray tube that puts the head closer to the beam entrance to the patient-irradiated volume. Vañó et al. (2016) derived dose to the lens of the eye from dose measured over the apron, and compared dose to the lens of the eye in urologists per nephrolithotomy procedure with dose received by interventional cardiologists and radiologists. The report concluded that, due to the lack of protective shields in urology, the dose to urologists per procedure was 18.7 times higher than the dose received by interventional cardiologists who used ceiling-suspended shields. (30) Within the European study on optimisation of radiological protection of medical personnel, TLD measurements and Monte Carlo simulation campaigns were performed for three cardiac and five interventional radiology procedures (Vanhavere et al., 2012). The selection was based on their potential impact on annual worker exposure (i.e. procedures with high frequency or high values of kerma-area product, or both). Operators were exposed substantially from embolisation procedures as well as from percutaneous transluminal angioplasty (PTA) of the lower limbs and renal arteries. During cerebral and carotid procedures, the doses to the operators were relatively low since femoral access is usually chosen and, therefore, the operator stands further away from the irradiated part of the patient compared with other procedures performed in the thoracic or abdominal region. Equivalent dose to the lens of the eye from digital subtraction angiography (DSA) and PTA was approximately 40 µSv, and the dose was up to 120 µSv for embolisations. Among the cardiac procedures included in the measurement campaign, higher operator doses were delivered from the implantation of pacemakers and cardiac defibrillators, despite their relatively low kerma-area product values; this is due to the fact that fluoroscopy alone is used in these interventions. The reason for the higher occupational doses from these procedures is that operators work very close to the irradiation field, and often work without any protective shielding. Average eye doses lie within the range of 40–60 μSv. (31) Other studies indicate that annual equivalent dose to the eyes of some interventional clinicians may be in the region of 50–100 mSv (Vañó et al., 2008a; Ciraj-Bjelac et al., 2010; Thornton et al., 2010; Koukorava et al., 2011; Jacob et al., 2013; Martin and Magee, 2013; IAEA, 2014b; Principi et al., 2015). Thus, radiation dose to the lens of the eye for interventional clinicians with high workloads can readily exceed the revised 20 mSv dose limit for the lens of the eye (ICRP, 2012), unless appropriate radiological protection measures are put in place.
2.3.3. Equivalent dose to the hands
(32) Dose to the extremities, particularly the hand of the physician or assistant nearest to the x-ray generator or x-ray beam path, can be substantially higher than that assessed on the torso, thereby suggesting a need to specifically monitor the hands and, in some less common situations, the feet should protective shields not extend much below the x-ray tube and to the level of the feet. Felmlee et al. (1991) compared hand doses for 30 cases at the Mayo Clinic, including transhepatic cholangiograms and biliary and nephrostomy procedures, with results from three other studies. The largest hand absorbed dose measured was 5.5 mGy with a median procedure dose of approximately 1 mGy. The other studies cited reported hand doses per procedure ranging from 0.01 mGy for neurological interventions to 0.4 mGy for peripheral vascular angiography. Whitby and Martin (2005) reviewed 18 studies that reported hand doses per procedure from less than 0.01 mGy to nearly 2 mGy. Important factors influencing the dose to the hand were the type of procedure, the x-ray equipment used, the expertise of the operator, and (particularly) the access route (antegrade access to the femoral artery can be difficult in obese patients, which may result in higher doses). Sauren et al. (2011) reported dose to the hands of approximately 2 mSv per procedure for transcatheter aortic valve replacement or transcatheter aortic valve implantation using the transapical approach. (33) In the study on Optimization of Radiation Protection for Medical Staff (ORAMED), an average equivalent dose per procedure to the left hand of approximately 240 μSv was observed for DSA/PTA of the lower limbs, approximately 320 μSv for embolisations, and approximately 60 μSv for cerebral DSA/PTA procedures. Average doses of 410 μSv have been recorded for the left finger for cardioverter defibrillator implantation, while for cardiac angiography/angioplasty and radiofrequency ablations, the respective values were 180 μSv and 60 μSv (Vanhavere et al., 2012). (34) Felmlee et al. (1991) made scatter measurements at various distances from a 12-cm × 15-cm field with a phantom entrance absorbed dose rate of approximately 65 mGy min−1 and exit dose rate of 0.7 mGy min−1. The scatter dose rates in the lateral direction ranged from 0.7 mGy min−1 at a distance of 0 cm, to 0.35 mGy min−1 at a distance of 5 cm, and 0.13 mGy min−1 at a distance of 15 cm (Felmlee et al., 1991). The variation in reported hand doses is explained by the large dose gradients near the x-ray beam, movement and placement of the hands, and whether the interventional procedure involves femoral, percutaneous, or internal jugular vein catheter insertion that places the physician in different positions relative to the patient and x-ray tube (Whitby and Martin, 2005; Martin, 2009). Hand doses also tend to be much larger for over-table x-ray units due to the greater scatter from the primary beam. (35) Poor technique in CT fluoroscopy can result in the physician’s hands being placed in the direct beam (Buls et al., 2003), reaching the annual dose limit of 500 mSv in a few minutes.
2.3.4. Equivalent dose to lower extremities
(36) Artschan et al. (2014) determined occupational effective dose from phantom irradiations, replicating exposure factors used for abdominal procedures, and from radiologists performing actual interventions on patients. They found values for annual lower extremity equivalent dose up to 110 mSv, despite the use of a protective curtain hanging on the side of the treatment couch. This exposure is attributed to the presence of a gap between the protective curtain and the floor, the size of which is dependent on the height of the treatment couch. Consequently, for procedures requiring a higher couch, such as biliary procedures, and for taller interventionalists, an increased lower extremity radiation dose may be received. (37) The group found that, without protection, the lower limb dose was frequently greater than the hand dose, with a mean leg dose between 0.19 and 2.61 mSv per procedure without any protection, and between 0.02 and 0.5 mSv per procedure with a protective curtain (Artschan et al., 2014). The ORAMED study showed leg doses of 160–250 μSv (Vanhavere et al., 2012).
2.3.5. Specific issues of occupational exposure from selective internal radiation therapy
(38) Different professionals are exposed in the different phases of SIRT:
Nuclear medicine technicians or radiopharmacists are exposed during preparation and calibration of 90Y microspheres before application. Interventional radiologists and other staff are exposed during transcatheter delivery into the hepatic artery. Nurses are exposed after the procedure until patient discharge. (39) Only a few papers on occupational doses from SIRT have been published. Occupational exposure from SIRT procedures is caused by x rays with relatively low dose rates and by direct beta radiation, especially to the hands and fingers, with high dose rates if precautions are inadequate. In addition to the dose to the hands of workers preparing the individual patient dose and to the physician implanting the microspheres, there is potential for significant contamination hazard. Specific advice to reduce this hazard is given in Section 5. Exposure data are 43.5 mSv MBq−1 h−1 skin equivalent dose due to contact with a 5-mL syringe and 1.35 mSv kBq−1 h−1 due to contamination with 50 μL on 1 cm2 (Kemerink et al., 2012).
2.3.6. Specific issues of occupational exposure from positron-emission-tomography-guided interventions
(40) 18F-FDG has a photon energy of 511 keV, which is much greater than the typical scattered photon energies from CT and fluoroscopically guided procedures (NCRP, 2010). Several studies have evaluated the radiation doses from patients receiving PET administrations (Chiesa, 1997; Benatar et al., 2000; White et al., 2000; Seierstad et al., 2006; Heckathorne and Dahlbom, 2008; Hippelainen et al., 2008; Nye et al., 2009; Demir et al., 2010; Quinn et al., 2016). These have shown that a reasonable representation of the ambient dose equivalent rate anterior to the chest of patients is approximately 0.09 μSv MBq−1 h−1 at 1 m and approximately 0.37 μSv MBq−1 h−1 at 30 cm, immediately following injection of 18F-FDG. These values can be reliably scaled to the desired time and distance for planning and prospective worker dose evaluation purposes. Lower values have been measured depending on the specific location of the measurement (Quinn et al., 2016). (41) PET-CT-guided biopsies are not common. They are performed when CT alone is not sufficient to identify the area of possible cancer (Werner et al., 2011; Aparici and Win, 2014). PET-CT-guided interventional procedures typically use 18F-FDG. Ryan et al. (2013b) quantified occupational radiation exposure and found a median effective dose per procedure of 0.02 (range 0–0.13) mSv for the primary operator, 0.01 (range 0–0.05) mSv for the nurse and anaesthesia provider, and 0.02 (range 0–0.5) mSv for the radiographer. The median extremity equivalent dose for the operator was 0.05 (range 0–0.62) mSv per procedure. Radiation exposure of the workers correlated with the duration of the procedure, and with the use of in-room image guidance. The authors concluded that operator effective dose from PET-CT-guided procedures was not significantly different from typical doses from fluoroscopically guided procedures. The major determinant of radiation exposure to the operator from PET-CT-guided interventional procedures is time spent in close proximity to the patient. As novel PET isotopes are developed, they may result in different dose profiles near the patient (Holland et al., 2010; Williamson and Dauer, 2014). (42) With regard to fingertip doses from 18F-FDG, Sánchez et al. (2015) measured dose reductions from using a full automatic system for preparing and infusing the FDG. The results show a reduction in the average skin dose to the fingertips of radiographers from 223 to 83 µSv GBq−1 (63%) from preparing the radiopharmaceutical. The average skin dose to the fingertips of nurses was reduced from 83 to 11 µSv GBq−1 (87%) from infusion to the patient. The accuracy of the delivered activity was 2%.
2.4. Reported radiation injuries to professionals involved in the interventions
2.4.1. Injuries to the lens of the eye
(43) Ocular ionising radiation exposure results in characteristic lens changes leading to opacification. While the initial stages of such opacification may not cause visual disability, the severity of such changes increases progressively with dose towards a vision-impairing lesion. The latency of such changes is inversely related to radiation dose (ICRP, 2012). During typical fluoroscopic working conditions, and if radiological protection tools are not used regularly, x-ray exposure to the eyes of interventionalists, other physicians, and/or staff working close to the patient can be high. (44) One of the first reported cases of radiation-induced opacities in interventional radiologists was in 1998, and the reason for the radiation injuries was the use of a non-optimised interventional radiology laboratory and the lack of a radiological protection programme (Vañó et al., 1998). In 2004, Haskal presented the results of a pilot study of x-ray-associated lens changes in 59 practising interventional radiologists; 37% of those screened had detectable posterior lens changes consistent with radiation exposure (Haskal, 2004; Junk et al., 2004). Although lens radiation doses were not reported, the authors noted that the frequency and severity of posterior subcapsular lens opacities increased as a function of age and years of practice, thus suggesting a possible dose–effect relationship. (45) Following these findings, in 2008, IAEA promoted a project called ‘Retrospective Evaluation of Lens Injuries and Dose’ (RELID) for interventional cardiology (IAEA, 2016), with the objectives of estimating occupational lens doses and evaluating possible lens opacities. (46) Since no personal dosimetry data were available, occupational lens doses were estimated in most cases by combining published typical scatter dose values (Vañó et al., 2008a,b) with information on the declared numbers of working years, workload, fluoroscopy and cine exposure conditions, radiological equipment used, location of the worker in the room, and use of radiological protection tools. Availability of some personal monitoring badge data helped in assessing the correlation. (47) For the ophthalmological examination of posterior subcapsular opacities, Merriam–Focht scores were used (Ciraj-Bjelac et al., 2010, 2012; Rehani et al., 2011; Vañó et al., 2010, 2013a). The scoring (i.e. 0.5, 1.0, 1.5, etc.) is done separately for each eye. In total, eight surveys were performed under the RELID study (Bogotá 2008, Kuala Lumpur 2009, Montevideo 2009, Varna 2009, Sofia 2009, Bangkok 2009, Buenos Aires 2010, and Kuala Lumpur 2011). (48) The RELID study concluded that workers in cardiac catheterisation laboratories show an increased prevalence of eye lens opacities when professionals have been working for several years without the proper use of radiological protection tools. Approximately 40–50% of interventionists and 20–40% of technicians or nurses, voluntarily attending the lens injury examination (during cardiology congresses), were found to have posterior subcapsular opacities compatible with injuries derived from exposure to ionising radiation. The incidence rate in interventionists was four to five times higher than that of the unexposed individuals in the control group (approximately 40–50% vs 10%). Estimated lens doses reached several Gy in some cases during a full professional life. However, it is still not clear if lens opacities progress to visually disabling cataracts. (49) Although a radiation-induced decrease in contrast sensitivity has not been reported in the study populations, in the last RELID study (Vañó et al., 2013a), a restricted contrast sensitivity test was performed for approximately 20% of the participants with observable lens changes on slit lamp examination. The contrast sensitivity curve for these participants resulted in a significant loss of contrast in comparison with the standardised normal data. Retrospective dose estimations are necessary to look for correlations between radiation dose and lens opacities (Vañó et al., 2013a). Comprehensive reviews of radiation effects on the lens of the eye are provided in ICRP and NCRP publications (ICRP, 2012; NCRP, 2016). (50) In many of these studies, there was irregular use of personal dosimeters and protective tools. These results point to the need for improving radiological protection, following the recommendations given in Section 5.
2.4.2. Reported incidents in selective internal radiation therapy
(51) Tosi (2003) reported an incident in a department where radioimmunotherapy with monoclonal antibodies and/or peptides was performed. 90Y was used with a concentration up to 150 GBq mL−1. The operator did not hold the vial with the special pliers provided, but held it directly in his hand, protected only with a very low-attenuation glove in lead rubber (0.1-mm Pb equivalent) covered by a disposable glove. After a few days, finger erythema was observed. Film badges, TLD finger ring dosimeter, and urine activity were normal. The estimated dose to parts of the fingers was 12 Gy (based on the energy of beta particles, attenuation by the glass of the vial and gloves, and referred total time of manipulation).
2.4.3. Reported hair loss in lower extremities
(52) Hair loss in the portions of the legs not shielded by a protective device (Balter, 2001) has been observed, and Wiper et al. (2005) reported that several senior interventional cardiologists noticed the onset of hair loss affecting both lower limbs. Dermatological advice suggested that the appearance was consistent with chronic occupational radiodermatitis.
2.4.4. Claims for an increase of brain cancers
(53) In contrast to the few small case series that have suggested a higher incidence of brain tumours in medical workers involved in interventional procedures (Wenzl, 2005; Roguin et al., 2013; Smilowitz et al., 2013), large epidemiological studies of mortality in US radiologists (Berrington de González et al., 2016) and US interventionalists (Linet et al., 2017) compared with US psychiatrists have not demonstrated evidence of increased mortality from radiation-related cancer. These studies involved more than 43,000 radiologists, 45,000 interventionalists, and 60,000 psychiatrists. Interventionalists showed a reduced risk of brain tumours, primarily malignant brain neoplasms, compared with psychiatrists. In a longitudinal study of more than 100,000 US radiologic technologists (radiographers), cumulative occupational radiation exposure to the brain was not associated with malignant intracranial tumour mortality (Kitahara et al., 2017). There was no evidence of a radiation dose–response association for the radiographers who reported working with fluoroscopically guided interventional procedures.
2.5. Challenges in monitoring exposure
(54) Challenges in monitoring exposure of workers in interventional procedures include the need for a simple, easily implemented, and consistent approach for occupational exposure monitoring that does not lead to unduly frequent investigations; estimating effective dose and equivalent dose for specific tissues from one or more dosimeter readings; and ensuring worker compliance with monitoring procedures. (55) Effective dose received by workers is estimated from dosimeters worn on the thorax or waist. Monitoring the extremities poses practical challenges related to wearing comfort and infection control of hand dosimeters. Evaluation of the radiation dose to the eye, especially when goggles are worn, is not a straightforward issue; devices for wearing the dosimeter behind the glasses have been developed and are described in Section 4.2.7. However, they are not in use worldwide and are not used regularly.
2.5.1. Incorrect and irregular use of individual dosimeters
(56) Surveys have revealed incorrect and inconsistent use of personal dosimeters. The IAEA ISEMIR (IAEA, 2014b) survey showed that only 76% of interventional cardiologists reported that they always use their dosimeters, and 45% reported using two dosimeters. Sánchez et al. (2012) indicated that as many as 50% of physicians either do not wear their dosimeters, wear them infrequently, or wear them in the wrong place on the body. Sánchez et al. (2012) reported that only 33% of monthly dosimeter readings were judged to be reliable. Physicians were less likely than nurses to use dosimeters correctly. The data for US fluoroscopic dosimeter results given by a dosimetry service provider in the USA revealed a similar lack of reliability in many of the readings. Without reliable monitoring data, radiological protection professionals may not have the information needed to offer tools and suggestions to reduce exposure or optimise protection. (57) Similarly, an important finding in ophthalmological studies (RELID study) is irregular use of personal dosimeters, and poor adherence to the ICRP recommendation to use two dosimeters, with one dosimeter located at collar level over the apron from which dose to the lens of the eye can be inferred. A study by Vañó et al. (2013b) showed that only approximately 50% of the interventionalists reported that they use personal dosimeters, and only 30% reported their use on a regular basis. Approximately 90% of nurses and technicians reported the use of personal dosimeters, but regular use was only reported by approximately 40%. Even when used, dosimeters were worn under the apron in most cases, so any retrospective evaluation of ocular radiation dose using these devices is likely to be inaccurate. In a previous study, Niklason et al. (1993) showed that half of the workers did not use their personal dosimeters regularly. (58) A retrospective study of 15 years follow-up in a cardiology department observed that 20–30% of cardiologists were not using their dosimeters routinely (Vañó et al., 2006). In surveys conducted by IAEA during various radiological protection training courses, which included cardiologists from over 56 countries, responses indicated that 33–77% of interventional cardiologists used radiation badges routinely (IAEA, 2014b). (59) If two dosimeters, meant for placement under and over the apron, show similar readings, this indicates that their placement may have been randomly reversed. Another disparity can arise when protective glasses are only used for some procedures. Therefore, a consistent deployment of dosimeters together with consistent use of the protective devices needs to be stressed. Workers need one set of instructions on how many dosimeters to use and where to place them which is specific to their most restrictive duty or risk of exposure. The Commission (ICRP, 2000b) and others (NCRP, 2010) recommend that interventional radiology departments should develop a policy and good habits for workers to wear two dosimeters.
2.5.2. Possible reasons for non-compliance with monitoring procedures
(60) Reluctance to use dosimeters may be the result of the impression that these individuals’ accumulated effective doses may approach dose limits, thereby potentially constraining them from practising their profession and treating their patients, or that time-consuming investigations may be triggered by dose readings that are high but still within occupational dose limits.
2.5.3. Assessment of effective dose
(61) There are multiple approaches for assessing effective dose from one or more dosimeters. In situations in which the dose spatial distribution varies as much as it does in fluoroscopy, dose assessment is subject to large uncertainty. A series of conservative assumptions can lead to dose estimates that are many times the true value. The personal dose equivalent, Hp(10), is recommended as a conservative estimate of the effective dose under a variety of simple exposure assumptions: anterior–posterior, lateral, rotational, isotropic, and posterior–anterior incidences on mathematical representations of the human body. When the personal dose equivalent is used to account for non-uniform exposure conditions, further conservatism is introduced. Locating a dosimeter in the area of highest photon fluence may add to the overestimation. Strategies for exposure monitoring are given in Section 4, and on the assessment of doses to the conceptus in Section 4.3.6.
2.5.4. Challenges in monitoring the lens of the eye
(62) In principle, as shown in Section 4, the reading of a dosimeter over the apron at collar level is a reasonable indicator of the dose to the lens of the eye when protective glasses are not worn, but when protective glasses are used, the collar dosimeter may grossly overestimate the dose to the lens of the eye. In addition, with the significant uncertainties involved in eye lens dosimetry and the fact that actual doses to the lens of the eye may be of the same order as the dose limit, assessing compliance with the dose limit represents an important challenge.
3. APPLICATION OF THE SYSTEM OF OCCUPATIONAL PROTECTION TO INTERVENTIONAL PROCEDURES
3.1. Principles of radiological protection
3.1.1. General
(63) The Commission’s system of radiological protection aims primarily to protect human health. Its objectives are to manage and control exposures to ionising radiation so that tissue reactions (deterministic effects) are prevented, and the risks of stochastic effects are reduced to the extent reasonably achievable, societal and economic factors considered (ICRP, 2007a). To achieve these objectives, the Commission recommends three fundamental principles of radiological protection: justification, optimisation of protection, and limitation of individual dose (ICRP, 2007a). The principles of justification and optimisation apply to all types of exposure – occupational, public, and medical – while the principle of dose limitation only applies to workers and the public, not to medical exposures of patients, carers or comforters, and subjects participating in biomedical research.
3.1.2. Justification of practices and procedures
(64) The principle of justification is that any decision that alters the radiation exposure situation should do more good than harm. This means that when introducing a new radiation source, or working to reduce an existing exposure or the risk of potential exposure, sufficient individual or societal benefit to offset the detriment it causes should be achieved (ICRP, 2007a). In the context of medical exposure, the aim of justification is to do more good than harm to the patient, subsidiary account being taken of the radiation detriment from the exposure of the radiological workers and other individuals (ICRP, 2007a).
3.1.3. Optimisation of protection
(65) The principle of optimisation of protection means that ‘the level of protection should be the best under the prevailing circumstances, maximising the margin of benefit over harm’ (NCRP, 1993; ICRP, 2007a). More specifically, this means that the likelihood of incurring exposures, the number of people exposed, and the magnitude of their individual doses should all be kept as low as reasonably achievable, taking into account economic and societal factors. In the context of medical exposure from interventions guided by radiological imaging, optimisation of protection implies keeping the radiation dose to patients and workers as low as possible, consistent with achieving the clinical objective of the interventions. It should be applied to the design of facilities that use ionising radiation; to the selection, set-up, and use of equipment; and to day-to-day working procedures.
3.1.4. Dose limitation
(66) The principle of dose limitation states that ‘the total dose to any individual from regulated sources in planned exposure situations other than medical exposure of patients should not exceed the appropriate limits recommended by the Commission’ (ICRP, 2007a). This principle applies to the exposure of medical workers. (67) For occupationally exposed workers in medical interventional procedures, the dose limits for workers recommended by ICRP apply. In planned exposure situations, recommended dose limits for workers were established in Publication 103 (ICRP, 2007a), with an updated limit for the lens of the eye in the ICRP statement on tissue reactions (ICRP, 2012). (68) The following limits apply:
Whole body: an effective dose of 20 mSv year−1, averaged over defined periods of 5 years, provided that the effective dose does not exceed 50 mSv in any single year. Extremities: hands and feet, an equivalent dose of 500 mSv year−1. Skin: an equivalent dose of 500 mSv year−1, averaged over 1-cm2 area of skin regardless of the area exposed. Lens of the eye: an equivalent dose limit for the lens of the eye of 20 mSv year−1, averaged over defined periods of 5 years, provided that the equivalent dose to the lens of the eye does not exceed 50 mSv in any single year. (69) The objective of the recommended limits is to ensure that the occurrence of stochastic health effects is kept below unacceptable levels, and that tissue reactions (deterministic effects) are avoided.
3.1.5. Dose constraints
(70) Optimisation is aided by setting a boundary on the predicted dose in the optimisation of protection (ICRP, 2007a). Such a boundary is called a ‘dose constraint’ in planned exposure situations, and is selected for planning purposes so that it effectively assists in the optimisation process, taking into account the current distribution of exposures. If it is later found to have been exceeded, an investigation should be conducted to understand the circumstances, and it is unlikely that protection is optimised. Dose constraints are therefore lower than the pertinent annual dose limit. Dose constraints are established prospectively in the process of optimisation, and are source related. When staff work in more than one facility, the dose limits and constraints should apply to the sum of all the individual doses incurred at the facilities. Dose constraints for the lens of the eye have been suggested by the International Radiation Protection Association (IRPA) (IRPA, 2017).
3.1.6. Investigations of abnormal doses
(71) There is no need to wait until an annual dose limit or constraint has been exceeded to become aware that protection was not optimised. Non-optimised protection can be detected by establishing an investigation level in terms of effective dose or equivalent dose received in 1 month, or the value of a related parameter, such as the reading of the over-apron collar dosimeter. (72) Exceeding a monthly investigation level provides an alert that protection was less than optimal in that period of time, and a review of existing radiological protection is needed. The increase in the dosimeter reading may be due to a substantial increase in the number of interventions, or in the dose per procedure, which may be due to an increase in procedure complexity or to a decrease in compliance with protection measures. (73) In 2000, the World Health Organization (WHO) recommended that an investigation should be carried out when monthly exposure reaches 0.5 mSv for effective dose, 5 mSv for dose to the lens of the eye, or 15 mSv to the hands or extremities (WHO, 2000). Following the new annual limit of equivalent dose to the lens of the eye, the investigation levels should be lowered accordingly. An investigation level of 2 mSv month−1, using the reading from the collar dosimeter, has been proposed for interventional cardiologists (Durán et al., 2013, endorsed by the Pacific Society of Interventional Cardiology, the European Association of Percutaneous Cardiovascular Interventions, the Latin American Society of Interventional Cardiology, and the Society for Cardiovascular Angiography and Interventions). (74) An investigation level in terms of a monthly dose should be such that when extrapolated to a year, it would not exceed the relevant dose limit or dose constraint. In addition, as described in Section 2, personal dosimeters are not always worn or are worn improperly (Padovani et al., 2011; Sánchez et al., 2012). Investigation levels can be helpful in this situation by establishing minimum dose values for the over-apron and hand dosimeters, thus providing an alert for possible poor compliance with procedures for wearing dosimeters.
3.2. Classification of areas and workplaces
(75) Publication 57 (ICRP, 1990, Para 129) discusses the possible classification of workers in categories with regard to the need for individual monitoring, and states that interventional radiologists and cardiologists are likely to fall into Category A. Classification of workers, however, was not supported in Publication 60 (ICRP, 1991), and Para. 184 of Publication 103 (ICRP, 2007a) states that, ‘The Commission continues to recommend the classification of areas of work rather than the classification of workers’. The assignment of individual monitoring devices should, therefore, be analysed on grounds of workplace and duties of the workers, their location and time of exposure within the radiation field, and the shielding provided by the protection devices used.
3.3. Embryo and fetus
(76) The Commission provided advice on the management of pregnant physicians and other workers in Publication 84 (ICRP, 2000a). The early part of pregnancy (before the pregnancy has been declared) is covered by the normal protection of workers. The first responsibility for the protection of the conceptus lies with the worker herself to declare her pregnancy to her employer as soon as the pregnancy is confirmed (ICRP, 2000a). Once the pregnancy has been declared and the employer has been notified, the working conditions of a pregnant worker should be such that the additional dose to the conceptus will not exceed 1 mSv during the remainder of the pregnancy (ICRP, 2000a). (77) Unnecessary discrimination against pregnant workers needs to be avoided. The restriction on dose to the conceptus does not mean that it is necessary for pregnant workers to avoid work with radiation completely, or that they must be prevented from entering or working in designated radiation areas (ICRP, 2000a). It does imply, however, that their employer should review the exposure conditions of pregnant workers carefully. In particular, their work should be such that the probability of high accidental radiation exposure is insignificant (ICRP, 2000a). (78) A clinical practice guideline for the occupational radiological protection of pregnant or potentially pregnant workers in interventional radiology has been developed as a joint guideline of the Society for Interventional Radiology and the Cardiovascular and Interventional Radiology Society of Europe. It states that excluding pregnant workers from fluoroscopic procedures solely on the basis of radiation risks to the conceptus cannot be justified on scientific grounds (Blake et al., 2006; Best et al., 2011; Dauer et al., 2015).
4. INDIVIDUAL MONITORING AND DOSE ASSESSMENT
4.1. Individual exposure monitoring
4.1.1. Exposure monitoring and verification of compliance with dose limits
(79) Exposure monitoring is needed for demonstrating compliance with annual dose limits as well as for optimisation of protection. Monitoring compliance with dose limits requires assessment of effective dose and equivalent doses to the skin, lens of the eye, hands, and feet. Equivalent dose and effective dose cannot be measured directly in body tissues, and cannot be used directly as quantities in exposure monitoring. The protection system includes operational quantities that can be measured, and from which equivalent doses and effective dose can be assessed (ICRP, 2007a). Operational quantities for area and individual monitoring of external exposures have been defined by ICRU, and those relevant for interventional procedures are summarised in Annex B. (80) Occupational exposure rests on a series of assumptions regarding the relationship between what is measured by a dosimeter and the dose received by an individual. Standards include accuracy requirements and uncertainties of the dosimetry system, so that these assumptions hold for the relationship between operational and protection quantities. Ensuring that workers wear the dosimeters correctly during all work time is the most important component of this series of assumptions and relationships. No dose to an individual can be estimated reasonably in highly variable radiation fields without having some type of individual monitoring on the workers during all times of exposure. Auditing compliance with procedures is important to verify that the workers wear the dosimeters regularly and correctly.
4.1.2. Exposure monitoring and optimisation of protection
(81) Verification of compliance is not performed by checking doses from individual interventional procedures, but by integrating the doses over many interventions carried out during a prescribed monitoring period. The period is established by the regulator and is usually 1 month. While this period is adequate for checking compliance with annual dose limits, it may not be sufficient for optimisation of protection in specific procedures, which may require collecting information on the same type of procedure, sometimes over multiple monitoring periods. Therefore, verification of compliance is occasionally complemented by monitoring designed for optimisation of protection, including evaluation of the effectiveness of radiological protection efforts.
4.2. Characteristics of individual dosimeters and their use
4.2.1. Types of dosimeters: passive and active dosimeters
(82) Dosimeters need to have adequate accuracy under a variety of exposure conditions, and to be small and lightweight enough to be convenient to use and not interfere with the staff’s ability to execute their tasks. Passive dosimeters are typically small, lightweight, and do not require power. This makes them easy to incorporate into packages that do not interfere with the staff’s actions and comfort, thus being the most widely used option, particularly for demonstrating compliance with dose limits. However, the absence of an instant reading capability is a disadvantage of all passive dosimeters for optimisation monitoring, and especially for education of workers involved in interventions. (83) For monitoring of the hands, small ring-shaped dosimeters are used due to their relative ease of fit under surgical gloves. Rings can be sized for different finger diameters, but attention is required to the fact that fingers may swell during long procedures. In addition, some additional features are important such as sterilisation capability and low interference with tactile sensation in the operator’s ability to manoeuvre catheters and instruments precisely. Fingertip sachets that fit over a finger have been used as an alternative to ring dosimeters, and are placed with the radiation sensor at the most proximal part of the hand where the largest doses may occur. An alternative solution that reduces interference with tactile sensation consists of wearing a TLD-type dosimeter on a finger nearest to the irradiated volume of the patient. (84) The physical construction of the dosimeter has to be compatible with the intended wearing location. Infection control is a particular concern for ring dosimeters because they may not withstand a sterilisation process, and they are typically worn during procedures where infection control is essential. (85) Dosimeters worn on the body should not create sharp pressure points that cause discomfort when placed between the heavy leaded apron and the user’s clothing. If whole-body dosimeters are placed near the neck on top of the leaded apron or over a protective thyroid shield to assess doses to unshielded areas, they should not have any edges that could irritate the neck or chin area. All methods of attachment should be strong enough to prevent dislodging during strenuous use, but not cause dislocation of protective aprons or damage to clothing in the event the dosimeter catches on a foreign object. (86) Active personal dosimeters (APDs) or electronic dosimeters are used for optimisation monitoring, or for special studies that require analysis of dose by procedure or discern aspects of a procedure (e.g. the relative dose received during fluoroscopy compared with image acquisition). APDs are able to provide immediate information about dose rate, so rapid feedback is available to staff against which they can assess changes to their behaviour that result in lower dose rates and, subsequently, lower accumulated doses. Dose rate information is needed if actions are desired within a procedure, as it can lead directly to procedural change. In addition, APDs provide information on the time of each exposure, which facilitates correlation of occupational and patient exposures, and auditing of the wearing of the APD during interventions. (87) Electronic dosimeters are useful for educating staff. For example, the large dose rate reduction when a ceiling-suspended shield is brought into place is very illustrative, and encourages clinicians to use these shields routinely. (88) Until recently, electronic dosimeters were too large to use conveniently under leaded aprons. The power requirements of older units added to the operational overhead of assuring charged batteries so that the units would not power off during a lengthy procedure. Advances in power management and wireless transmission of signals from an electronic dosimeter to a base station have overcome some of the disadvantages of using electronic dosimeters. Some manufacturers of interventional systems have included electronic monitors with wireless data transmission, so dose rates and doses can be viewed adjacent to or as part of the image video screens that the operator uses. (89) Optimisation monitoring does not strickly need to conform to the dose quantities required for compliance monitoring. Optimisation seeks to compare relative values resulting from changes in conditions, in order to evaluate effectiveness of various actions to reduce dose. Electronic dosimeters are usually calibrated to assess operational quantities without taking into account the non-uniform irradiation of the body during interventional procedures. They indicate the dose at a single point and make no inferences regarding effective doses or doses at some distance from the dosimeter. Conceptually, there is no technical reason why multiple electronic dosimeters could not be worn and the data combined to yield compliance-type dose information, but practical issues tend to limit the use of electronic dosimeters to investigatory and optimisation monitoring. (90) Electronic dosimeters have not been developed for routine use on the fingers or near the eyes. An attempt was made to place small electronic sensors on the fingers, but the electrical cables leading back to the power source and electronics were not convenient or practical for routine use.
4.2.2. Dosimeter specificity
(91) Dosimetry systems have to meet standard requirements for accuracy, precision, and reproducibility for the operational quantity of concern, for the range of photon energies between 20 and 150 keV and spectra used in interventional procedures, such as those given in IEC Standard 62387 (IEC, 2012), as well as in internationally accepted guidance (ICRP, 2010b; IAEA, 2014a).
4.2.3. Dosimeter reliability and simplicity
(92) The dosimetry system must be reliable and fail-safe (i.e. must possess a continued ability for measuring the radiation field). In addition, actions required from the user should be simple and efficient to execute. For electronic dosimeters that require the user to energise the dosimeter, an item needs to be included in the procedures for staff to remember in the process of putting on dosimeters. The fewer the actions and decisions required from the staff, the greater the likelihood of proper use. Dose-integrating passive dosimeters, such as those containing film, thermoluminescence crystals, optically stimulated luminescence crystals, and radiophotoluminescent glass, are generally used in the fluoroscopic suite to monitor compliance.
4.2.4. Dosimeter exchange periods
(93) Passive dosimeters provide total dose accumulated over the period of use, and must be exchanged for new dosimeters at the end of the use period. The exchange period should be on a predetermined schedule to instill a habitual routine among staff. Generally, interventional staff should be monitored for monthly periods to provide dose data with sufficient frequency that unusual radiation doses and events can be detected and appropriate responses implemented. Therefore, the radiation sensing material, be it thermoluminescence crystals, optically stimulated luminescence crystals, or film, should have the sensitivity to detect the minimally relevant dose over the shortest period of expected use, and should retain the dose information for the longest period of expected use.
4.2.5. Approaches to detect incorrect dosimeter wear in interventional procedures
(94) Problems with wearing dosimeters may cause not only very high dose readings, but also very low dose readings that may suggest misuse or failure to wear dosimeters. Examples of incorrect use also include wearing a dosimeter over an apron that was intended for use under an apron, wearing a ring dosimeter on the incorrect hand, wearing a dosimeter issued to another person, or a lost dosimeter. Indirect approaches may be useful in identifying a lack of compliance in wearing personal dosimeters, and in estimating occupational doses when personal dosimeters have not been used. These approaches include making use of area dosimetry to measure the scatter radiation near the patient (e.g. at the C-arm), together with conversion coefficients to enable the dose to the lens of the eye for workers to be estimated from patient-related quantities such as kerma-area product for different types of procedures and geometries. Wearing the over-apron dosimeter on a lanyard that can move in front of the body or hang down below the collar would introduce an additional difference from the radiation incident on the apron, and is, therefore, not recommended.
4.2.6. Different scatter conditions between type testing and calibration and real interventions
(95) Monitoring to assess effective dose has been attempted using one or two dosimeters. A discussion of the algorithms that adjust the dosimeter readings is presented later in this section; however, a few points should be made here. Dosimeters are calibrated and tested without any consideration of the effects of shielding materials. Type-test standards tend to define performance evaluations under simple conditions with dosimeters being placed on a flat surface of a tissue equivalent phantom. In the intervention room, dosimeters will either be placed under or over an apron containing shielding elements with high atomic number. The close proximity to the shielding materials places the dosimeter in a different scatter environment from that typically assumed during type testing. Assurances should be requested from the supplier to verify that the measurement of the operational quantities is within expected dosimeter performance and similar conditions to normal use.
4.2.7. Dosimeter for the lens of the eye
(96) Monitoring of the lens of the eye presents special challenges due to the difficulties in placing a device to which the dosimeter can be attached near the eyes. With the reduction of the dose limit for the lens of the eye, the use of protective eyewear has become more prevalent. This provides greater opportunities for locating dosimeters near the eyes and under the protective lenses. Eye doses can be assessed from a dosimeter placed over the leaded apron at the collar or level of the neck, or another dosimeter on a strip of plastic attached to a headband such that the sensor is adjacent to the temple closest to the x-ray tube. Some attempts at eye monitoring use a TLD chip wrapped in an elastic band that is fitted on the head near the eyes (Bilski et al., 2011). Others use a device that surrounds the head, with the possibility of placing the attached dosimeter inside the glasses (IRSN, 2014). In any case, dosimeters placed near the eyes must not interfere with the wearer’s vision. A dosimeter placed behind the glasses means the use of three dosimeters: one under the apron, one over the apron, and the eye dosimeter. An arrangement based on three dosimeters poses a challenge with regard to reliable and consistent use. It could, however, be used for comparison purposes during a short period of time. If leaded glasses are actually worn and the primary interventionalist uses a ceiling-suspended shield, the need for an eye dosimeter is not as critical, but quality control is necessary to ensure that the shield and the leaded glasses are actually used. The issue of when the glasses should and can be worn becomes the key issue.
4.2.8. Identification of the dosimeter and the worker
(97) Individual dosimeters should have a means to let the users identify their own dosimeters. A one-to-one relationship between a dosimeter and the user is indispensable if the dosimeter results are to be applied to a specific individual. Means of identification, such as labels, need to be easily readable to prevent someone from using someone else’s dosimeter. A suitable approach consists of racks on which dosimeters are stored when not in use, and visual identification on the rack and on the dosimeter.
4.2.9. Wearing location
(98) Visual elements should designate the intended wearing location, and help locate the dosimeter in the correct place, particularly when the shape of the dosimeter does not convey the proper placement. When two dosimeters, one over and one under the apron, are used to assess the effective dose, operators may frequently reverse the location of the over- and under-apron dosimeters so that the doses reported approximate an average of the two values. This inconsistency results in higher reported effective doses, which may frustrate the operators and discourage them from using both or even one dosimeter. Moreover, for better response reproducibility, the dosimeters should be worn in precise positions over and under the apron. Compliance with the correct location can be assured by using specific pockets on the personal apron. Icons or images of where the dosimeter is to be located combined with colours and labels have been tried to improve proper practice. A similar situation arises if both hands are to be monitored independently. The left and right rings can be reversed if distinctive features are not used. Labelling of hand or finger dosimeters is difficult given the limited space available to print all of the information needed on the ring. Different colours are an effective method to distinguish right from left. As a result of the potential for extremity dosimeters to be mixed up, the use of a single dosimeter has become common, with placement on the hand closest to the x-ray beam. This typically means the little finger on the left hand (Martin, 2009).
4.2.10. Calibration of active personal dosimeters
(99) In the European ORAMED project, Clairand et al. (2011) and Sánchez et al. (2014) tested the influence of dose rate as well as pulse frequency and duration on APD responses. With the exception of Geiger-Müller equipped APDs, which did not give any signal in pulsed mode, the APDs provided a response affected by the personal dose equivalent rate, which means that they could be used in routine monitoring provided that correction factors are introduced. Type-test procedures and calibration of APDs and area monitors should include radiation fields representative of interventional procedures, including tests in pulsed mode with high dose rates (Chiriotti et al., 2011; Clairand et al., 2011; Sánchez et al., 2014).
4.3. Assessment of occupational exposure
4.3.1. Assessment of effective dose
(100) In general, for relatively uniform whole-body exposure, effective dose is assessed from the reading of a personal dosimeter calibrated in terms of personal dose equivalent, Hp(10). This assessment of effective dose is sufficiently accurate and precise for radiological protection purposes, provided that the dosimeter is worn in a position on the body that is representative of its exposure (ICRP, 2007a). However, in interventional procedures, parts of the body are protected while other parts are unprotected. Therefore, the reading of a single dosimeter placed over the protective apron overestimates effective dose because the reading does not reflect the dose to organs of the trunk protected by the apron, while the single dosimeter placed under the apron underestimates effective dose because the reading does not reflect the higher exposure of unprotected body parts, such as the head, neck, and part of the lungs and other organs in the thorax that are exposed via the arm holes (Franken, 2002; Siiskonen et al., 2007). Thus, in order to estimate effective dose from a single dosimeter reading, a correction should be applied to the Hp(10) values. The correction factor is lower than 1 if the dosimeter is placed over the apron, and higher than 1 if the dosimeter is placed under the apron.
4.3.1.1. Considerations of the double-dosimeter approach
(101) Publication 85 (ICRP, 2000b) recommended that two dosimeters, one over the apron and one under the apron, should be used to obtain a better estimate of the effective dose. Also, the National Council on Radiation Protection and Measurements (NCRP, 2010) recommends the double-dosimeter method as it provides the best estimate of effective dose for comparison with the dose limit for stochastic effects, and a better indication (from the dosimeter worn under the protective apron at the waist or on the chest) of the shielding provided by the protective apron. (102) The readings of the two dosimeters, in terms of Hp(10), are usually combined by means of simple linear algorithms of the form:
(103) A number of pairs of α and β values have been proposed over the years, but due to the fact that no single α and β pair adequately represents occupational exposure for all types of procedures, there has been no worldwide consensus about which should be used. Without an international consensus supported by a standard, and means to facilitate the mistake-free placement of the two dosimeters, estimated values of effective dose will not be reliable nor comparable. (104) Within the European Coordinated Network for Radiation Dosimetry (CONRAD) project, dosimetry methods used in 13 European countries were compared. A single dosimeter was worn over the apron in five countries, a single dosimeter under the apron was recommended in seven countries, and two dosimeters (above and below the apron) were worn in one country (Järvinen et al., 2008). In some countries, there are no recommendations from regulatory bodies, and hospitals adopt different methods (IAEA, 2014b). (105) Also within the CONRAD study, Järvinen et al. (2008) made a comprehensive comparison of 11 different pairs of α and β values proposed by different authors for double dosimetry, and four values for the single-dosimeter approach. The study consisted of both Monte Carlo simulations and some measurements on a Rando–Alderson phantom taken for Ho correction purposes. The phantom was provided with a wraparound 0.35-mm lead apron and a separate collar for both the experiment and the Monte Carlo calculation. The specified criteria for determining the best estimate from the pairs of α and β values were that there should not be underestimation of the effective dose obtained from Monte Carlo simulations for typical irradiation geometries, and that overestimation should be minimal. (106) The CONRAD study concluded that there is no optimal algorithm for all possible geometries and that, therefore, compromises have to be made when making a choice. From all the double-dosimeter algorithms tested, two were found to be closer to the specified criteria for determining the best estimates. These were the sets of α and β values given in the Swiss Ordinance (2008) and by McEwan (2000), which are presented in Table 4.1. More recently, algorithms based on weighting factors for effective dose from Publication 103 (ICRP, 2007a) have been developed (Von Boetticher et al., 2010). These values are also presented in Table 4.1. (107) However, when the estimated effective dose is close to the annual dose limit (e.g. >15 mSv), a more accurate estimation should be made, taking into account the specific geometry and irradiation parameters, rather than using the simple approach with any of the α and β values (Järvinen et al., 2008).
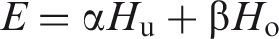
4.3.1.2. Considerations of the single-dosimeter approach
(108) Some authors have formulated objections to the generalised use of two dosimeters (Kuipers et al., 2008; Martin, 2012), and studies have been performed on the usefulness of a single dosimeter worn over the apron for assessments of dose to interventional radiologists (Stranden et al., 2008). Several studies have concluded that there is no significant difference in the accuracy of double- and single- (over apron) dosimetry algorithms (Schultz and Zoetelief, 2006; Järvinen et al., 2008; Kuipers et al., 2008; Kuipers and Velders, 2009). Although the double-dosimeter approach provides better accuracy in principle, the authors argue that it has several drawbacks: (1) the lack of international consensus on a combination algorithm renders comparison of effective doses difficult to interpret; (2) the reliability of clinicians wearing two dosimeters correctly and consistently is questionable; and (3) the cost of two dosimeters is higher. In practice, interventional clinicians may sometimes reverse the positions of the two dosimeters, and since the exposure received by the unshielded over-apron dosimeter may be 10 times that of the under-apron dosimeter, this leads to a substantial overestimate of effective dose. Clinicians may also forget to wear the second and even the first dosimeter. (109) In addition, the exposure geometry is variable, radiation is distributed non-uniformly, and parts of the body are shielded. Thus, achieving a high degree of accuracy in assessment of effective dose is not feasible. When doses are well below the respective dose limits, a pragmatic dosimetry system that is simple to implement and serves the purpose of providing a reasonable indication of dose levels is sufficient for ensuring compliance with dose limits. (110) A single dosimeter worn under the apron provides an indication of the dose received by the radiosensitive organs in the trunk, shielded by the apron. However, monthly readings of under-apron dosimeters are often below the detection level, so the accuracy of the technique is poor and the value in providing information is limited. (111) Martin (2012) suggested a pragmatic approach of using a single dosimeter placed at the collar over the apron. Only when readings of the collar dosimeter exceed an established dose level in a single year, or a shorter period to be established, is wearing a second dosimeter warranted. The reading of the collar dosimeter, corrected by a factor to take account of the organs that are protected, could provide an indication of effective dose. The collar dosimeter can also be used as an indicator of the dose to the lens of the eye. (112) Studies of the relationship between Hp(10) from the over-apron collar dosimeter and values for effective dose derived either from Monte Carlo simulations or TLD measurements in anthropomorphic phantoms suggest correction factors between 0.011 and 0.18 for situations where an apron is worn but no thyroid collar is worn, and 0.02 and 0.083 when both an apron and a thyroid collar are worn (Martin and Magee, 2013). Martin and Magee (2013) have proposed that a reasonable indication of effective dose (E) for staff involved in radiology procedures who are wearing protective aprons can be obtained from the simple relationship:
(113) This proposal of a factor of 0.1 would represent a conservative assessment of effective dose, appropriate for the majority of staff working in radiology departments, including those involved in interventional radiology and cardiology. If the Ho reading approaches or exceeds 20 mSv (effective dose ≈2 mSv in 1 month), then wearing of a second dosimeter under the protective apron and the use of a specific algorithm should be considered. NCRP (2010) also concluded that, if a single dosimeter is used, this should be worn over the apron (i.e. a single dosimeter worn under the radiation protective garments is unacceptable).
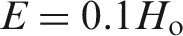
4.3.1.3. Recommended option for the assessment of effective dose
(114) While a single over-apron dosimeter at collar level provides a reasonable estimate of effective dose, and the single-dosimeter method is simpler than the double-dosimeter approach and less likely to cause errors in dosimeter location, the expression E = 0.1Ho relies on the assumption that the apron is worn during all interventions and that all aprons are alike in their attenuation. However, it cannot necessarily be assumed to be true for all countries and institutions that all staff wear aprons, nor that the aprons have similar and sufficient attenuation. Different lead-equivalent thicknesses are available, as are lighter-weight protective aprons that contain an attenuation material other than lead. The lead-equivalent thickness of the apron may also differ from the stated lead-equivalence thickness on the apron label (Lichliter et al., 2017). The Commission, therefore, maintains the principal recommendation to use the double-dosimeter approach with a simple algorithm, such as one of those found to meet the criteria and proposed in the CONRAD study (see Table 4.1). (115) If, for a given institution, it can be reliably assumed that all professionals wear protective aprons with sufficient attenuation during all interventions, the under-apron dosimeter could be omitted for the majority of staff members of the interventional team because the over-apron dosimeter will be sufficient for the assessment of their exposure. However, two dosimeters are recommended for the interventionalist performing the procedure, since interventionalists typically receive the highest occupational doses, and the actual attenuation of the interventionalist’s apron is rarely known. (116) When a member of staff works for more than one employer, cooperation among them is essential. One dosimetry option may be that the worker has different dosimeters supplied by each employer, but the sum of all the individual doses incurred at each of the facilities has to be obtained and a complete dose record has to be maintained.
4.3.2. Assessment of equivalent dose to the lens of the eye
4.3.2.1. Use of operational quantities for monitoring dose to the lens of the eye
(117) ICRP (2010b, Annex F) has considered the calculation of absorbed doses to the eye and lens of the eye using two dosimetric approaches: first, using the reference computational phantoms from Publication 110 (ICRP, 2009a), and second using the stylised model of the eye developed by Behrens et al. (2009) and Behrens (2012a). This stylised eye model was used to supplement eye lens dose conversion coefficients derived from Publication 110 (ICRP, 2009a) phantoms at low incident particle energies to capture the high dose gradients for external ocular irradiations. ICRP (2010b) also compared doses averaged over the lens with doses to the anterior epithelial cell layer, noting that this layer gives rise to the underlying anuclear lens fibre cells. Comparisons showed that for all but the lowest energy photons and electrons, similar doses were calculated for the two geometries. Bolch et al. (2015) provided details of the dosimetric models of the eye and lens of the eye, and their use to calculate dose coefficients for ocular exposures. (118) ICRU (1992) recommended the use of the operational quantity, personal dose equivalent Hp(3), for dosimetry of the lens of the eye. However, while Hp(3) is well suited to assess dose to the lens of the eye, calibration of dosimeters in Hp(3) is not available in many countries (Cantone et al., 2017). In Publications 103 (ICRP, 2007a, Annex B) and 116 (ICRP, 2010b), it is recommended that the operational quantity Hp(0.07) is adequate for monitoring the lens of the eye for photon exposures. Behrens and Dietze (2010, 2011) and Behrens (2012b) compared equivalent dose to the lens of the eye and the corresponding value of the operational quantities at the three recommended depths – 0.07, 3, and 10 mm – using realistic photon and beta radiation fields. The authors concluded that both Hp(0.07) and Hp(3) are adequate for x-ray fields calibrated on a slab phantom to simulate backscatter. Similar results were reported by Vanhavere et al. (2012) and Sánchez et al. (2014). (119) With regard to the suitability of Hp(10) for dosimetry of the lens of the eye, measurements by Sánchez et al. (2014) have shown that differences between Hp(10) and Hp(0.07) measured with optically stimulated luminescence crystal dosimeters are lower than 10% for four different spectra with mean energies higher than 44 keV, as typically used in fluoroscopy and CT-guided procedures, but increases to approximately 17% when the photon spectrum has a mean energy of 36 keV. IAEA (2013) and the International Organization for Standardization (ISO, 2015) have suggested that Hp(0.07) can be used as an approximation to Hp(3) for photon radiation in general, and that Hp(10) can also be used but only if the photon spectrum reaching the dosimeter has a mean energy above 40 keV and photons are incident mainly from the front. Monitoring procedures for the lens of the eye have been provided by ISO, IAEA, and IRPA (IAEA, 2013, 2014b; ISO, 2015; IRPA, 2017).
4.3.2.2. Assessment of dose to the lens of the eye when protective glasses are not worn
(120) State-of-the-art studies of methods for assessment of dose to the lens of the eye through experimental and computational modelling are reported in the literature (Vanhavere et al., 2012; Carinou et al., 2015). For a given tube potential and current, the resulting dose to the lens of the eye is influenced by several factors, mainly: patient size, projection angle, distance from the x-ray focus to the patient and from the patient to the interventionalist, beam collimation, and operator technique (Vañó, 2015). (121) A number of studies have investigated the position where an eye dosimeter should be worn when no eye protection is used. A dosimeter worn on the head at the left eyebrow ridge or the middle of the forehead will generally provide the best assessment of eye dose. Some studies report that TLDs positioned on the eyebrow ridge on the side adjacent to the x-ray source measured doses three to five times higher than dosimeters placed between both eyes, whereas in other studies, dosimeters in the latter location have tended to record a higher dose (Efstathopoulos et al., 2011; Vanhavere et al., 2012; Principi et al., 2014). Thus, the optimum position appears to vary with the type of procedure and the practice of the operator. (122) If a collar dosimeter is worn over the apron on the side adjacent to the x-ray tube, this should give a good indication of the level of radiation to which the eye is exposed if no eye protection is used. It is likely to overestimate the dose to the lens of the eye, although results in the literature vary (Martin and Magee, 2013). A reasonable approximation for the interventionalist (Clerinx et al., 2008; Martin, 2009) is given by Heye = 0.75Ho, and for staff further from the x-ray tube, Heye ≈ Ho where Ho is the personal dose equivalent Hp(3) or Hp(0.07) from a dosimeter worn at the collar (over apron). (123) However, the collar dosimeter is only an indicator of eye dose, rather than an accurate measurement. When the cumulated reading of the dosimeter is expected to exceed a certain value (e.g. 10 mSv) and no protective eyewear is worn, it may be advisable to improve the accuracy of assessment by wearing an eye dosimeter adjacent to the most exposed eye. (124) There are situations in which an interventionalist has not used the dosimeter regularly during interventions, and there is a need to make a moderately conservative dose estimate for this period. In exploring ways of obtaining a reasonable estimation of eye dose in these circumstances, Vañó et al. (2013b) investigated the ratio between the kerma-area product from interventional cardiology and the reading of an active dosimeter placed on the C-arm, 95 cm from the isocentre, as a surrogate for dose to the lens of the eye. The study, based on 1969 interventional procedures, resulted in a ratio of the scatter dose at the C-arm to the kerma-area product within 10.3 and 11.3 μSv Gy−1 cm−2. This ratio is just an example valid for the type of procedure investigated and the particular conditions of this facility. In the absence of any other information, radiological protection officers could use such indirect approaches for estimations, provided that the type and the approximate number of procedures are known. If the dose approaches the limit, a more detailed investigation may be required.
4.3.2.3. Assessment of dose to the lens of the eye when glasses are worn
(125) There are some attempts at eye dosimetry using devices to place the dosimeter behind the glasses to assess the dose to the lens of the eye directly (Bilski et al., 2011; IRSN, 2014). It is likely, however, that many staff worldwide will continue to be monitored by standard dosimeters at the collar over the apron. Therefore, the question arises as to when and how attenuation provided by eyewear should be taken into account in the assessment of radiation exposure of the lens of the eye. (126) Magee et al. (2014) reported measurements on 30 sets of protective eyewear made using Rando phantoms to determine the dose reduction factor (DRF; i.e. the ratio of the dose with no eyewear divided by the dose with eyewear). (127) The protection provided by lead glasses depends on the angle at which scatter from the patient is incident on the head (McVey et al., 2013; Magee et al., 2014; Van Rooijen et al., 2014). For the majority of times that an interventional radiologist or cardiologist is carrying out a procedure, he/she will not be looking towards the patient when x rays are being emitted, but will be viewing the resulting images on the monitor. Therefore, when using a DRF for assessing the eye lens factor, the x-ray beams incident from the side and below the level of the head should be taken into account. (128) Moreover, studies of lead glasses have concluded that the dose to the eyes when protective eyewear is worn results primarily from radiation scattered from surrounding tissues of the interventionalist (Moore et al., 1980; Day and Forster, 1981; Cousin et al., 1987; McVey et al., 2013; Magee et al., 2014). The size of the lenses, the use of side shields for glasses with flat lenses, and the closeness of fit to the facial contours are all important in determining the extent of protection provided. Since the scattered radiation is incident from a level below the head and to the side of the operator, then the closeness of fit and the extent of the lenses protecting regions beneath the eye and to the side of the face are crucial factors determining the DRF. (129) Magee et al. (2014) have concluded that, for most situations, the majority of lead glasses with a lead equivalence of 0.75 mm provide a DRF between 3 and 6 for exposures occurring in clinical practice. Based on these results, division by a DRF of 2 would be both a reasonable and conservative approach that could be applied routinely to account for the protection offered by lead glasses with a lead equivalence of 0.75 mm. This factor should be applied to the reading of the collar dosimeter, provided that the eyewear is of appropriate design, either with side shields or of a wraparound design, includes protection in the frames (Martin, 2016), and is worn consistently. In addition, for the DRF to be applied, there must be a quality assurance programme in place, with regular documented checks to confirm that the interventionalist concerned always wears the protective eyewear. (130) Another study using Monte Carlo simulations and measurements and considering the effect of eye equivalence and the size of the glasses was performed by Hu et al. (2017). According to the study, dose to the lens of the eye was reduced by a factor from 3 to 9 when wearing lead glasses with 20-cm2-sized lenses with an equivalent thickness ranging from 0.1- to 1.0-mm Pb. While the increase in DRF was not significant when increasing the lead equivalence above 0.35 mm, the DRF was proportional to the size of the eyeglass lens from 6 to 30 cm2 with the same lead equivalence. They also concluded that reasonable and effective protection is achieved by 0.5-mm Pb and large-sized glasses (at least 27 cm2 per eyeglass lens). (131) In institutions where a higher DRF value is considered appropriate, comprehensive measurements should be made, taking into account the direction of the primary and the scattered x rays in clinical practice including angulation in both the horizontal and vertical planes, and these measurements should be fully documented. ISEMIR has recommended that improved methodologies to assess the dose to the lens of the eye need to be developed, including when lead glasses are worn (IAEA, 2014b).
4.3.3. Assessment of equivalent dose to the hands
(132) The dose limit for the skin is applied as an average over 1 cm2 in the most exposed area, and therefore applies to the most exposed part of the hand. The hands of interventionalists can be close to the x-ray beam, and the interventionalist’s position, which is determined by the type of procedure and access route, is an important factor for estimating doses. (133) The ulnar aspect of the hand, which is side-on to the x-ray beam and closer to the irradiated volume of the patient, receives a higher dose, so dosimeters should be worn either on the little finger or the side of the wrist closest to the x-ray tube (Whitby and Martin, 2005; Vanhavere et al., 2012). (134) Proper dosimetry to evaluate dose to the hands and fingers is not easy in clinical practice. A method to estimate hand dose in interventional radiology is a wrist dosimeter but, due to the inhomogeneity of the radiation field near the patient and the potential of introducing part of the hands into the direct beam, the dose measured by wrist dosimeters could be much lower than the actual finger dose. (135) There are specific challenges in the assessment of finger dose in CT fluoroscopy. A primary beam from a CT scanner can deliver doses of hundreds of µGy in a few seconds. As indicated in Section 2, if an operator's hand is placed in the direct beam, the annual dose limit of 500 mSv could be reached in a few minutes (Buls et al., 2003; Saidatul et al., 2010). Due to the relatively narrow CT beams, of the order of 5-mm wide, a dosimeter worn at the base of the finger may fail to record the exposure of the rest of the finger to the primary beam, and in most cases, only scattered radiation would be measured. Extremity monitoring in CT fluoroscopy is therefore of limited value in this case, and may provide false reassurance. (136) The CT images should alert an operator if part of their hand enters the primary beam. In addition, the capability to review CT images provides a means of controlling these exposures and facilitating investigations. A dose assessment can be made through measurements in the actual positions where exposures have occurred for appropriate lengths of time. Since dose levels in the CT beam can be measured, dose data should also be included in the assessments.
4.3.4. Assessment of equivalent dose to the legs and feet
(137) When the x-ray tube is positioned below the couch, the primary beam is also scattered downwards from the patient and the base of the couch, so the dose received by the legs can be substantial. Where no table shield is used, the dose to the legs can be greater than the dose to the hands (Whitby and Martin, 2003). Consideration should be given to assess dose to the parts of the leg that are not shielded by the protective apron or lead/rubber drapes.
4.3.5. Assessment of exposure in selective internal radiation therapy
(138) An open problem of using beta emitters for SIRT interventional procedures is the finger dosimetry of the staff. TLD finger dosimeters should be worn on the index finger of the hand closer to the radiation source. Due to the very small distances between the beta source and skin and the concomitantly high dose gradient, the dose can be underestimated. Rimpler and Barth (2007) measured local skin doses, Hp(0.07), at the fingertips and found that the exposure of the staff can exceed the annual dose limit of 500 mSv when working at low protection. If finger tips are likely to come into contact with an unshielded vial or syringe, it may be necessary to wear finger stall dosimeters (or fingertip sachets).
4.3.6. Assessment of exposure to the embryo and fetus
(139) For pregnant workers who perform or assist in fluoroscopic procedures, dose to the conceptus is usually estimated using a dosimeter placed on the mother’s abdomen at waist level, under her radiation protective garments (Miller et al., 2010; NCRP, 2010). This dosimeter overestimates actual conceptus dose because radiation attenuation by the mother’s tissues is not considered. The fetal dose is typically not more than half of the dose recorded on the dosimeter worn by the worker (Dauer et al., 2015), due to the attenuation by the mother’s abdominal wall and anterior uterine wall (Trout, 1977; Faulkner and Marshall, 1993; NCRP, 2010). Therefore, when two dosimeters are used, if the dosimeter under the protective apron shows a value for personal dose equivalent, Hp(10), of <0.2 mSv month−1, the equivalent dose to the conceptus over a 9-month period would be below the limit. Dosimeters should be evaluated monthly. Electronic dosimeters can be used to provide rapid access to data (Balter and Lamont, 2002).
4.3.7. Computational methods for real-time monitoring
(140) Badal et al. (2013) described a dose monitoring system that uses an accelerated Monte Carlo code, detailed anatomical phantoms, and physical sensors in the imaging room. This system has the potential to provide accurate real-time dose estimations for both patients and staff during interventional fluoroscopy with higher accuracy than current dosimetry systems. These methods may be helpful in auditing the regular and proper use of personal dosimeters, and assessing the need for additional protection (e.g. protective glasses). Research programmes should pursue the development of computational technologies (not requiring dosimeters) together with personnel position sensing to assess personnel doses, including dose to the eye (IAEA, 2014b; NCRP, 2016).
5. RADIOLOGICAL PROTECTION METHODS AND PROGRAMME
5.1. Relationship between protection of the patient and that of the staff
5.1.1. Actions that reduce patient and staff exposuse
(141) Reduction of patient exposure reduces scattered radiation in a similar proportion, thus reducing occupational exposure. Therefore, the following actions protect the patient but also the workers: reduction of fluoroscopy time, number of acquisition runs and number of images per run, use of lower-dose mode fluoroscopy and acquisition, lower pulse frequency, last image hold and image loops, image receptors close to the patient, collimation to the required field of view, cautious use of steep oblique projections and wedge attenuators where appropriate, and removal of the antiscatter grid for procedures on small children.
5.1.2. Additional measures to reduce staff exposure alone
(142) The following occupational protection devices and actions reduce staff exposure without reducing patient exposure: protective apron and collar, ceiling-suspended shield, protective eye glasses, table-suspended lead curtains, shielding drapes on the patient, stepping back to increase distance from the patient whenever possible, and staying on the side of the image receptor rather than on the side of the x-ray tube.
5.1.3. Other issues of relationship between patient and staff exposure
(143) Magnification by image intensifiers increases the dose in the irradiated volume of the patient, but reduces the size of the irradiated volume. Therefore, the amount of scattered radiation and thus the radiation dose to the staff may stay similar, depending on the automatic brightness control sensor design and the algorithm used by the equipment. In the case of magnification with a flat panel, the increase in dose to tissues in the field of view is generally lower than with image intensifiers and, therefore, the scatter radiation to the staff is reduced (Srinivas and Wilson, 2002). (144) Changing beam projection angle to avoid exposing the same skin area all the time may avoid patient skin injuries in complex and long interventions, but the way it affects the staff exposure depends on the extent of gantry angulation and the position of the x-ray tube with respect to the position of the interventionalist. Staff dose from scattered radiation increases when the x-ray tube is on the same side as the interventionalist with respect to the irradiated volume of the patient.
5.2. Distribution of scattered radiation
(145) Previous sections have mentioned that medical staff working in interventional procedures can receive relatively high doses of radiation compared with other occupational groups involved with x-ray imaging (Kim et al., 2008, 2012; Martin, 2009; Koukorava et al., 2011; Vanhavere et al., 2012; Jacob et al., 2013; ICRP, 2013a,b; Vañó et al., 2015a). In addition, procedures often require the interventionalist to remain close to the patient in order to manipulate catheters and other devices. Other staff who provide assistance may also need to be in close proximity to the patient. (146) The higher dose rates around the patient in a fluoroscopic x-ray room result from radiation scattered back from the patient. If the tube is positioned below the couch, doses to the head, upper body, and hands of the interventionalist will be substantially lower because, in this geometry, the scattered radiation towards the staff has been transmitted through, and attenuated by, the patient's body (Fig. 5.1). Thus, this is the arrangement recommended for the majority of procedures (ICRP, 2013a,b). (147) When the C-arm angulation departs from the vertical, staff standing on the same side of the couch as the x-ray tube will be exposed to higher levels of radiation from x rays scattered from the side of the patient, while the radiation scattered towards staff standing on the far side is attenuated by passing through the patient (Fig. 5.2) (Balter, 1999; Whitby and Martin, 2003; Schueler et al., 2006; Morrish and Goldstone, 2008). The ratio of dose rates on the two sides of the couch will change as the angle of the tube is increased. When the x-ray beam is directed at 10° to the vertical, the dose rate on the side adjacent to the tube will be double that on the far side, and when the angle is increased to 30°, the dose rate may be five times that on the far side. Therefore, staff who stand near the couch while performing or assisting in interventional procedures should avoid the region adjacent to the x-ray tube for oblique and lateral projections. (148) As the x-ray tube angle is increased towards the lateral for examinations of the trunk, the x rays will also be passing through a greater depth of tissue. This will require a higher x-ray intensity to form an adequate image, and so further increase the dose to both patient and staff. Thus, although it is important to move the x-ray field to avoid any area of the patient’s skin receiving an excessive dose, larger gantry angulation should be used sparingly.
Air kerma rate distribution from an undercouch x-ray tube (Whitby and Martin, 2003) (reproduced with permission from British Journal of Radiology). Air kerma rate distribution around interventional x-ray unit with angled tube (Whitby and Martin, 2003) (reproduced with permission from British Journal of Radiology). Risk of exposure-induced incidence of thyroid cancer per sievert of thyroid equivalent dose based on data for the International Commission on Radiological Protection composite population defined in Publication 103 (ICRP, 2007a). Positions where the hands of operators will be manipulating catheters during (a) interventional cardiology using radial and femoral access routes, and (b) interventional radiology procedures by internal jugular vein, percutaneous, and femoral access (Martin and Sutton, 2014) (reproduced with permission from Oxford University Press). Position of the hands for manipulation of catheters for procedures undertaken with a femoral access (Whitby and Martin, 2005) (reproduced with permission from British Journal of Radiology). Examples of the positions of the hands for percutaneous and internal jugular vein access routes (Whitby and Martin, 2005) (reproduced with permission from British Journal of Radiology). α and β values [adapted from Järvinen et al. (2008)] of the algorithms that best meet the criteria of no underestimation and minimum overestimation for the typical geometries, and an algorithm based on Publication 103 (ICRP, 2007a) weighting factors for effective dose. Representative exposures for the technician or pharmacist preparing a typical patient administration, and for the physician implanting the prepared dose (adapted from SIRTEX, 2016). Typical ambient dose equivalent rates 6 h after implantation of 2 GBq 90Y activity for different distances (adapted from SIRTEX, 2016).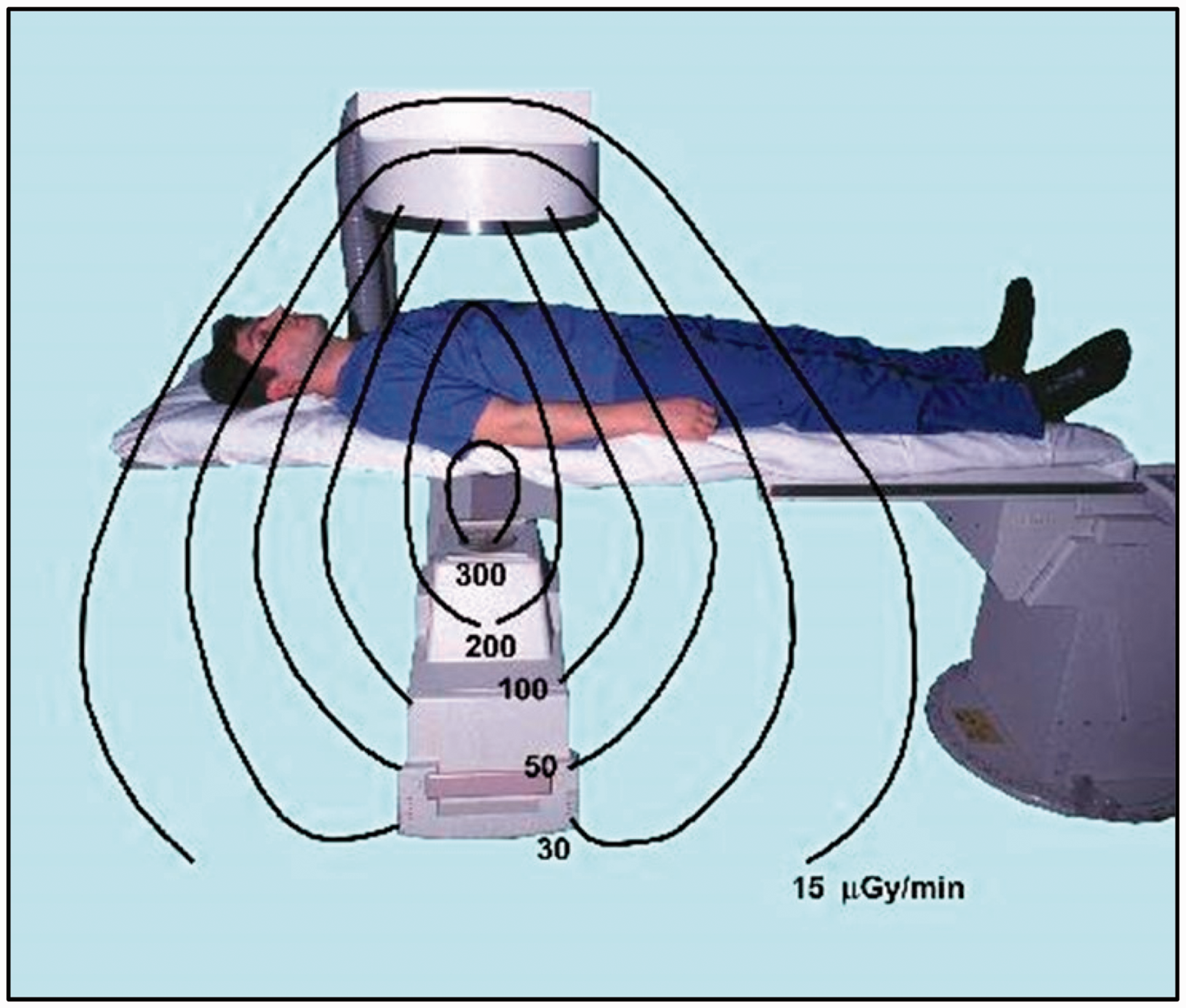
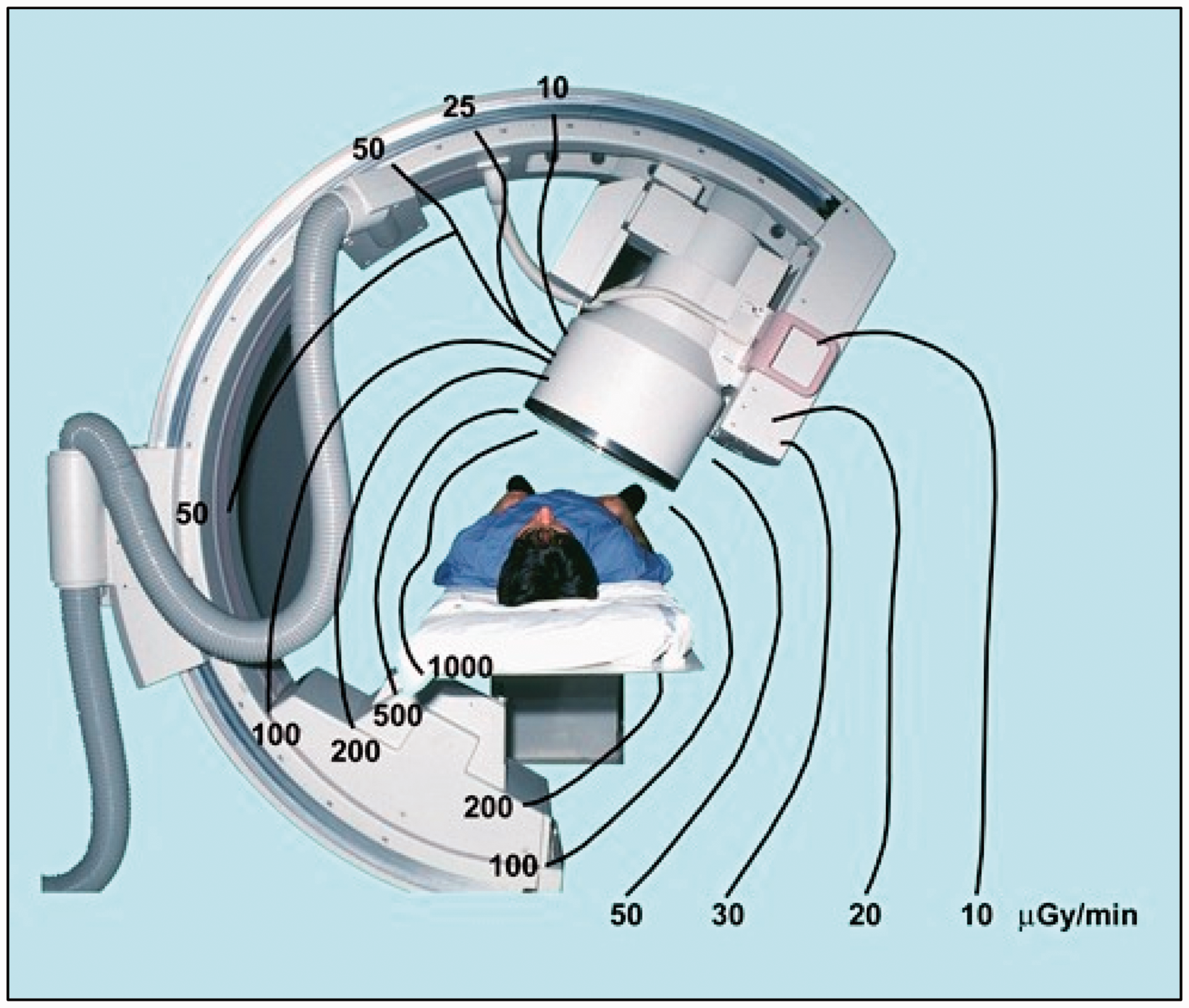
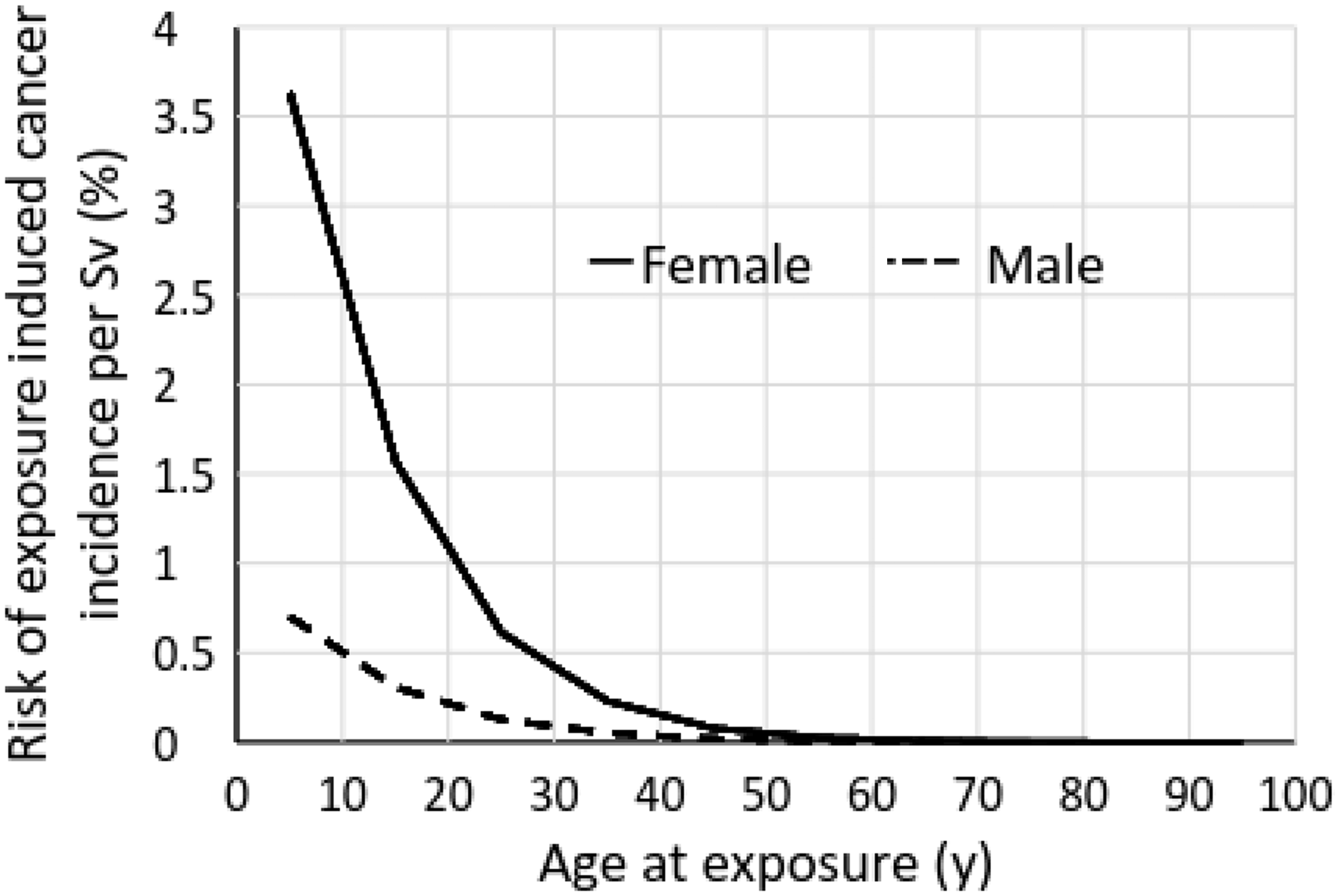
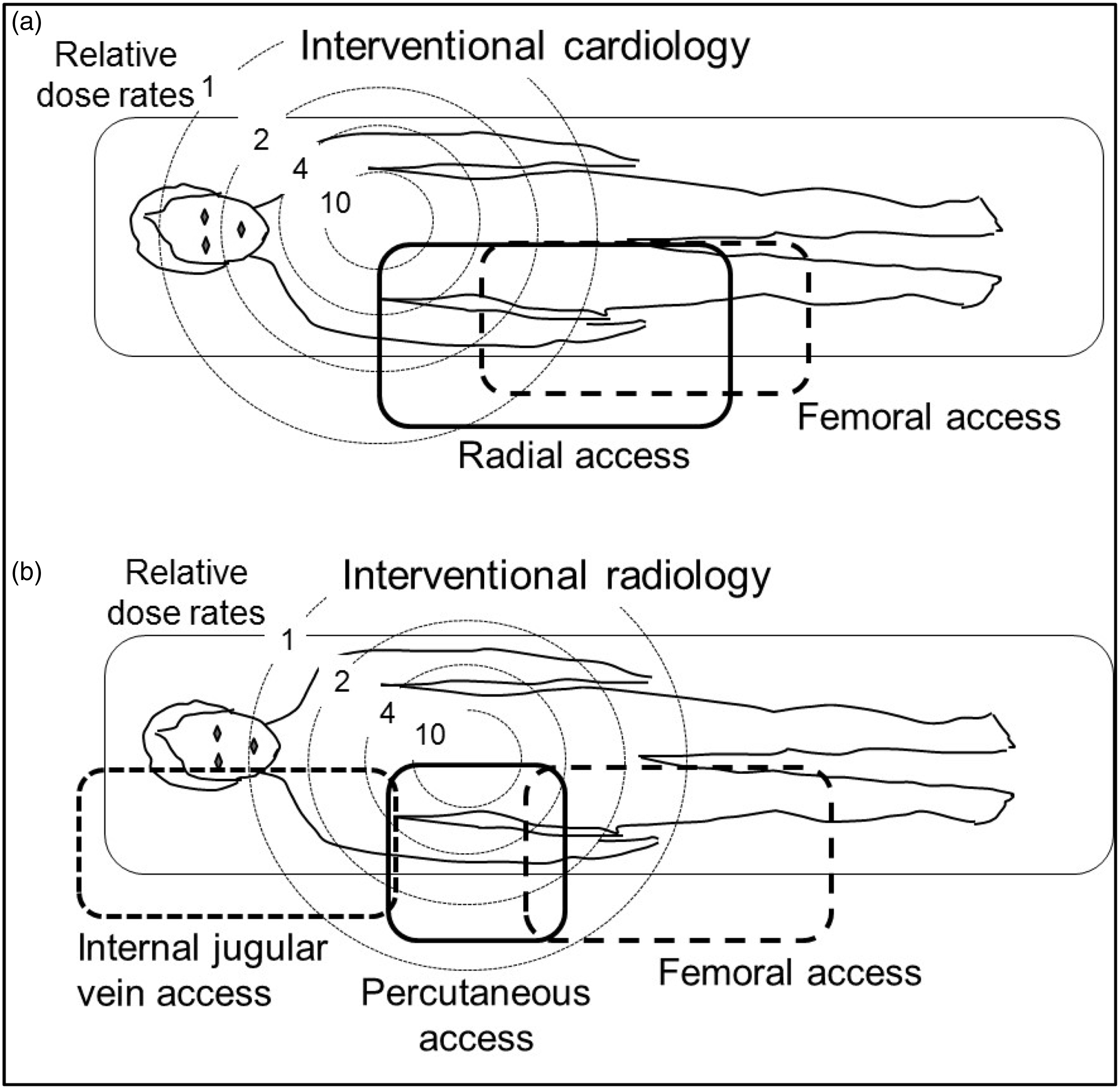
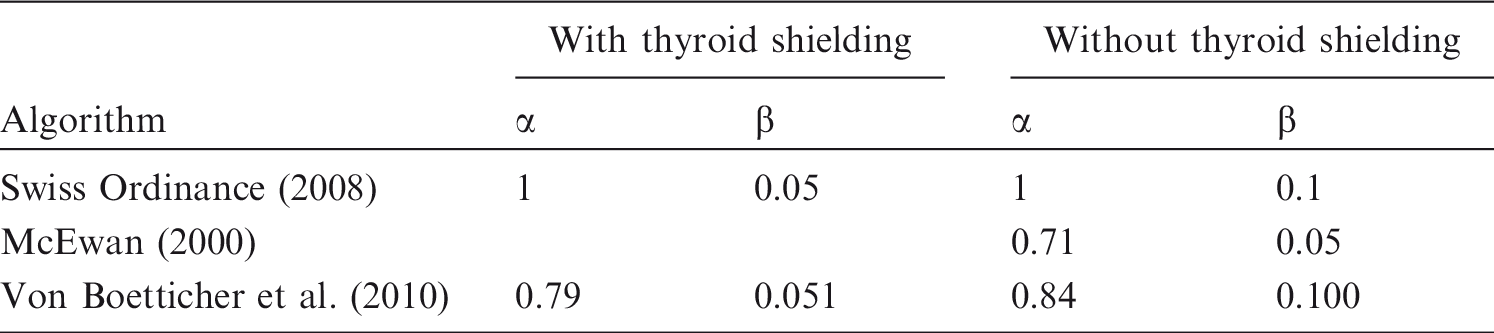
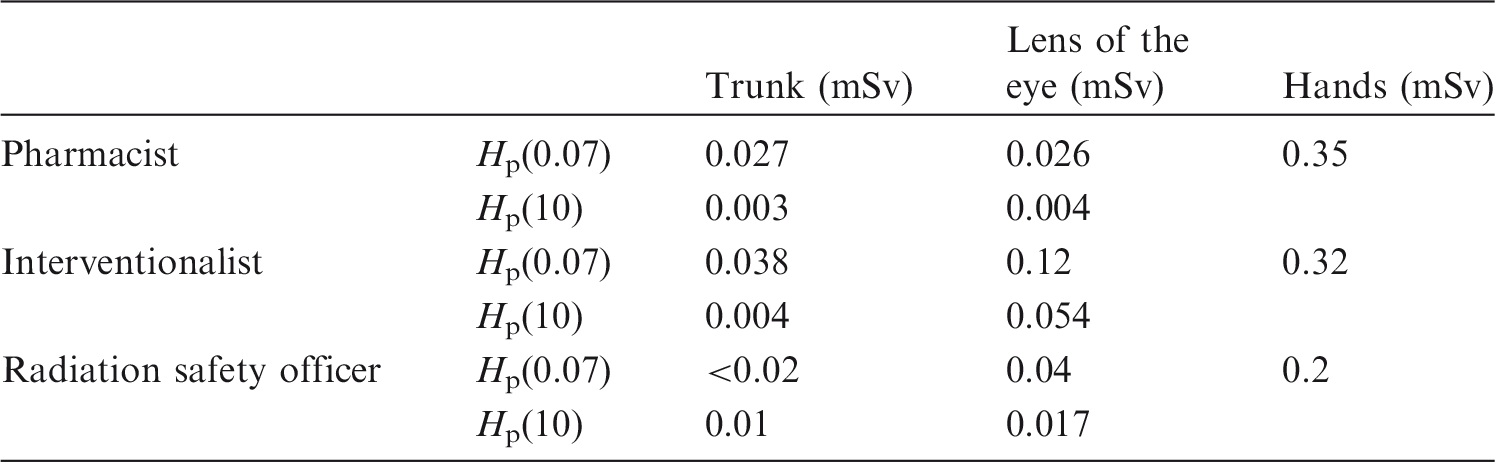
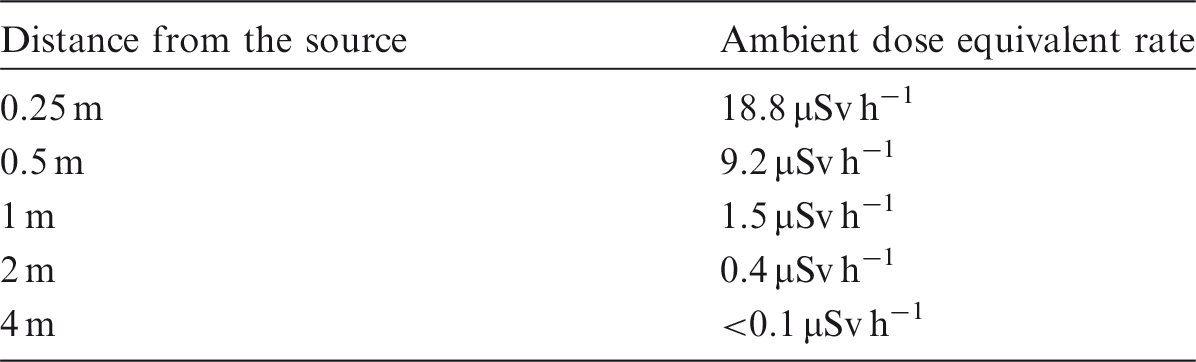
5.3. Protection of the body
5.3.1. Knowledge of scattered radiation
(149) The amount of scattered radiation and its associated occupational exposure is determined by the complexity of the procedure, the size of the patient, the modes of operation available on the x-ray equipment, and the skills of the operator (Vañó et al., 2015a). Knowledge of the distribution of scattered radiation levels around a patient, understanding how different factors influence it, and the effective use of protective devices is indispensable for all staff involved in interventions (ICRP, 2009b) in order to protect themselves. (150) A number of professional societies, radiological protection organisations, and others have issued guidelines on practices to be followed, and made recommendations on the use of protective devices (Miller et al., 2010; NCRP, 2010; Chambers et al., 2011; Sauren et al., 2011; Durán et al., 2013; ICRP, 2013a,b; Hiles et al., 2016).
5.3.2. Protective aprons
(151) Personal protective equipment, such as aprons, is worn by all interventional staff working in fluoroscopy inside the x-ray room. These aprons usually contain the equivalent of 0.25, 0.35, or 0.5 mm of lead, and some designs have an overlap at the front to provide protection of 0.5-mm lead equivalence, with 0.25-mm lead equivalence elsewhere. Transmission is typically between 0.5% and 5% in the range 70–100 kV (Marx et al., 1992). Although they shield the trunk against scattered radiation, parts of the body, including the head, arms, hands, and legs, are not protected by the apron and need to be considered in the radiological protection programme. Lead equivalence of and attenuation by the apron should be sufficient for the staff doses to meet regulatory dose limits and to optimise protection. As shown later in this section, the actual dose to the staff depends on a number of factors, and radiological protection should be optimised by taking care of all of them, not just apron thickness. In addition, the risk of musculoskeletal injuries due to the apron’s weight should also be considered in the optimisation. (152) The fit of the protective apron is often more important to reduce effective dose than the lead equivalence of the apron (Detorie et al., 2007). Thoracic organs, including the lungs and oesophagus, may receive higher levels of exposure when the operator is irradiated from the side through the armholes of the apron (Franken, 2002). This is particularly important for protection of the axillary portion of the female breast. Some facilities favour aprons with shoulder protection to reduce this exposure (Guersen et al., 2013).
5.3.3. Lighter-weight aprons
(153) The weight of lead aprons often causes discomfort to staff; fatigue and musculoskeletal problems, including those of the spine, need specific consideration (Papadopoulos et al., 2009; NCRP, 2010; Klein et al., 2015). Different designs of protective apron are available, some of which aim to reduce the ergonomic hazards in order to minimise the risk of back injury. Two-piece aprons consisting of a waistcoat and skirt allow some of the weight to be supported at the hips to reduce strain on the back (Klein et al., 2009). (154) There are lighter-weight aprons containing composite layers or bi-layers of high atomic number elements, such as tin or bismuth, as well as, or instead of, lead. Similar attenuation can be achieved with lighter-weight aprons because the alternative metals are more efficient per unit mass than lead for absorbing x-ray photons with energies between 40 keV and 88 keV. These aprons may be more effective for attenuating scattered x rays from tube voltages of 70–80 kV, but less effective for tube voltages above 100 kV (Christodoulou et al., 2003). They provide a reasonable alternative where weight reduction is required to alleviate back or neck problems. Lightweight or ‘lead-free’ aprons have a different x-ray transmission from aprons containing lead for different x-ray spectra. (155) Manufacturers often specify the attenuation properties in terms of lead equivalence (e.g. 0.5-mm lead equivalent); these data without further qualification can be misleading (Finnerty and Brennan, 2005; Schlattl et al., 2007; Eder et al., 2010) since attenuation varies significantly over the photon energy spectrum, with the largest variations occurring in the diagnostic imaging range. Manufacturers might also have tested the transmission of the protective garment in narrow-beam geometry using primary radiation, rather than broad-beam geometry using scattered radiation which is the situation that staff will be in. One study concluded that these aprons provide less lead equivalent thickness than that stated on their labels and their manufacturing certificates (Papadopoulos et al., 2009). Users and patients who use lead-free x-ray protective clothing might unknowingly be exposed to a greater dose than generally assumed. (156) The indication of the protective value of garments should be accompanied by specification of the characteristics of the radiation beams (IEC, 2014) and the geometries used to measure the attenuation, and the combination of measurements made at different beam qualities to reflect the conditions under which the garment is used. Jones and Wagner (2013) concluded that measurement of attenuation or lead equivalence at a single beam quality is inadequate, and suggested that specifications of the protective value of protective garments should accurately reflect the protection provided across the range of beam qualities for which it will be used, and these specifications should be relatively easy to verify by the end-users. They further suggest four standard beam qualities that should be selected, that penetration through protective garments should be measured for these beam qualities with the garment configured as it would be for clinical use, and that the protection index should be weighted by the distribution of workload with respect to the four beam qualities (Jones and Wagner, 2013). (157) It is important for users to understand the labelling of the lead equivalence for the particular protective garment. For example, where a manufacturer quotes ‘0.35-mm Pb’, it could be 0.35-mm Pb only when two flaps are overlapped, rather than 0.35 mm Pb through every part of the garment; users might falsely believe that when the flaps overlap, they get 0.7-mm Pb equivalence.
5.3.4. Independent support of the apron weight
(158) Reduction of the ergonomic hazards associated with leaded aprons can also be achieved by independent support of the apron in a manner such that it can be moved easily by the operator (Klein et al., 2009). One manufacturer calls this a ‘zero-gravity radiation protection device’. This might be through an independent floor-mounted frame (Pelz, 2000) or through suspension from the ceiling (Savage et al., 2009). The latest versions extend from the head to the lower extremities, and travel on rails suspended from the ceiling.
5.4. Protection of the thyroid
(159) Aprons do not protect the neck. If no thyroid protection collar is worn, the dose to the unprotected thyroid, which is sensitive to radiation in young people, may double the effective dose (Niklason et al., 1993; Mateya and Claycamp, 1997; Theocharopoulos et al., 2006; Siiskonen et al., 2007, 2008; Martin, 2009). However, as shown in Fig. 5.3 based on data from Publication 103 (ICRP, 2007a), the risk of cancer incidence is strongly dependent on age at exposure, and the risk for males above 30 years of age and for females above 40 years of age is small (NA/NRC, 2006; NCRP, 2010). NCRP (2010) suggested the use of thyroid collars, or protective aprons with thyroid coverage, for younger workers and for all personnel whose personal dosimeter readings at the collar level (unshielded) exceed 4 mSv [Hp(10)] in 1 month (Wagner and Archer, 2004). (160) Monte Carlo simulations (Marshall et al., 1992) have shown that a collar with protection of 0.5-mm of lead reduces the equivalent dose to the thyroid by a factor of 12, while a collar containing 0.35 mm of lead will reduce it by a factor of 7. However, thyroid collars can be uncomfortable when fitted tightly around the neck, so they are often worn more loosely for comfort. If the collar is at a lower level around the larynx, approximately 10 mm of upper thyroid may be unprotected. This will reduce the protection factors to 6 and 5 for the two lead thicknesses, respectively. These calculations agree broadly with measurements made on an anthropomorphic phantom during simulated patient exposures, which suggest a factor of 6 (Marx and Balter, 1995). Thus, the overall reduction in the equivalent dose to the thyroid in clinical practice resulting from wearing a collar is a factor between 5 and 10. (161) In addition to interventional radiologists and cardiologists, surgeons and other groups of clinicians also undertake diagnostic or therapeutic procedures in standard operating theatres with guidance from mobile C-arm fluoroscopy units (ICRP, 2010a). Doses to the head and thyroid might be significant in some cases, but the availability of protective devices in the operating theatre is often limited. Risk evaluations are needed to assess the techniques used, and determine whether staff should wear thyroid collars as well as protective aprons (Vañó et al., 2016). (162) Radiation doses from the majority of orthopaedic procedures are low. However, surgeons may be working close to the irradiated volume of the patient and, for some procedures, they may need to use the x-ray tube in an over-couch configuration, for which scatter dose to the head and upper body will be greater. Table-suspended lead curtains and ceiling-suspended shields are rarely available. However, for procedures involving the patient’s extremities, the dose levels should be relatively low compared with procedures where the patient’s body is in the beam. A few orthopaedic procedures, such as vertebroplasty and kyphoplasty (also performed by interventional radiologists), in which cement is injected into vertebrae to stabilise the spine, have the potential to deliver high doses to the eyes and hands of the operator (Struelens et al., 2013). (163) Freestanding adjustable over-table shields that have cutouts to allow a closer fit to the patient’s body contour can provide good protection to the interventionalist, but interventional staff often find them intrusive. Anything that interferes with necessary manipulations, thereby lengthening the procedure and increasing the dose to the patient and staff, should be avoided.
5.4.1.1. Disposable drapes
(164) Lightweight disposable lead-free drapes or pads containing tungsten/antimony or bismuth can be placed outside the field of the primary beam to reduce scattered radiation levels (King et al., 2002; Dromi et al., 2006; Thornton et al., 2010; Politi et al., 2012; Ordiales et al., 2015; Martin, 2016). Such drapes may have an aperture through which catheters can be inserted into the skin, and the shielded surround cuts down the radiation scattered from the patient. They are placed in position after the operative site has been prepared, outside the field of the x-ray beam. This type of protection should be considered for procedures where the operator needs to be very close to the irradiated volume of the patient. These drapes protect the head, hands, and upper body and have been shown to reduce dose to the eye by a factor of 5–25 (Thornton et al., 2010). Evaluation of sterile disposable lead-free drapes used for percutaneous nephrostomy procedures, as reported by King et al. (2002), concluded that their use was well worth the small amount of time and the relatively little added cost required to use them. Reusable drapes can also be fabricated from scrapped protective aprons or shielding (Miller et al., 1985). Disposable drapes can achieve a substantial reduction of operator exposure, especially when a ceiling-mounted upper body shield cannot be used effectively (Fetterly et al., 2017). However, when placing disposable drapes on the patient, attention is required to avoid placing the drapes within the primary beam, which might increase patient and operator exposure.
5.5. Protection of the head and eyes
5.5.1. Ceiling-suspended shields
(165) Eye doses are influenced by tube angulation, operator position, and beam collimation, as discussed in Section 1.1. The most important factor in protection of the head is the proper use of shields (Vañó et al., 1998; ICRP, 2013a; Vañó, 2015). Ceiling-suspended lead acrylic shields should always be available in interventional facilities as they can reduce doses to the entire head and neck by factors of 2–10 (Martin, 2016). (166) The protection to the eyes provided by ceiling-suspended shields or lead glasses can be quantified in terms of dose reduction factors (DRFs). Reports on dose reductions to the eyes achieved through use of ceiling-suspended shields give varying DRFs. A large-scale report of clinical measurements for interventional procedures gave DRFs between 1.3 and 7 (Vanhavere et al., 2012). A review comparing doses from groups at different centres performing similar procedures gave DRFs between 0.7 and 2.5 (Jacob et al., 2013), and a study comparing dose rates for periods when radiologists were using and not using shields gave a DRF of 5 when the shield was in use (Magee et al., 2014). However, phantom simulations with precise positioning of the ceiling-suspended shield yield higher values. In a phantom study, Galster et al. (2013) reported DRF values between 8.5 and 17.6 for transjugular intrahepatic portosystemic shunt (TIPSS) creation, embolisation of abdominal haemorrhage, and pelvic embolisation. Ceiling-suspended shields demonstrated a significantly higher dose reduction than lead glasses, and protect the entire head and neck, not just the eyes (Galster et al., 2013). One clinical study with careful placement of a shield for percutaneous coronary interventions observed a DRF of 19 (Maeder et al., 2006). (167) When use of a ceiling-suspended shield is possible, the level of dose reduction achieved depends on the use of the shield and how effectively it is positioned. The shield should be placed just above the patient, with the operator viewing the irradiated area of the patient through the shield. This is an important element of radiological protection training for interventionalists (Vanhavere et al., 2012). However, it is often difficult to use these shields effectively with the x-ray tube in lateral or oblique projections. Effective use of shields requires their continual repositioning as the x-ray tube and couch are moved. Thus, although the shields give good protection in principle, difficulties in their effective deployment for the range of projections used in clinical procedures may limit the overall level of protection achievable in routine use. Nonetheless, with diligence, DRFs of 2–5 should be achievable. This reduction should allow interventionalists to keep doses to the eye below the limit, and avoid eye lens opacities which may otherwise occur through the accumulation of dose over a professional working life. (168) Vañó et al. (2015b) estimated that more than 800 procedures per year and per operator would be needed to reach the new dose limits for the lens of the eye for three interventional specialties (cardiology, neurology, and radiology) using the conservative approach of estimating dose to the lens of the eye from the over-apron collar dosimeter, and assuming proper use of ceiling-suspended protective shields (Vañó et al., 2015b). (169) Some authors have shown attenuation and potential dose saving to the brain of the interventionalists (Alazzoni et al., 2015; Uthoff et al., 2013) provided by radiation-absorbing surgical caps, while others have focused on measurements of dose distributions inside the head using radiochromic films on a RANDO phantom, and have reported that the radiation absorbing surgical cap provides essentially no protection to the brain of an interventionalist (Fetterly et al., 2017). The authors have explained that this is due to the geometry of the irradiation (i.e. the fact that the scatter originates from a location inferior to the interventionalist’s head, and the cap cannot protect the brain from radiation coming from below). (170) This finding is consistent with the fact that actual doses to staff depend on a number of factors, and radiological protection needs to be optimised in a systematic approach by knowing and taking care of all of them, including the effective use of ceiling-suspended shields. The first consideration should be to find out if there is a need to provide additional brain shielding, given that the published data demonstrate no evidence of an increased incidence of brain cancers from radiation doses to the brain of interventional staff, as discussed in Section 2. Staff should have access to consultation with a medical physicist or an expert on radiological protection familiar with interventional procedures and questions related to radiological protection, including the assessment of brain radiation exposure, before undertaking additional protection measures.
5.5.2. Other movable shields
(171) Staff, such as nurses and anaesthesia personnel, who need to remain near the patient may benefit from the additional protection provided by movable (rolling) shields that can be positioned between them and the x-ray source.
5.5.3. Protective eyewear
(172) Lead glasses are an important component of the protection for the eyes against scattered radiation. The use of protective glasses has proved to reduce the dose to the lens of the eye substantially. Lead glasses are commercially available with an equivalent lead thickness of 0.75 mm that can reduce doses above 85% (Sandblom, 2012; Magee et al., 2014; Martin, 2016) for all tube potentials. (173) DRF values between 5 and 10 have been reported from experimental measurements for a variety of lead glasses when protecting against x rays incident from the front in the same horizontal plane as the eyes (Moore et al., 1980; Marshall et al., 1992; Thornton et al., 2010; McVey et al., 2013; Van Rooijen et al., 2014) and Monte Carlo simulations (Carinou et al., 2011; Koukorava et al., 2014). (174) However, in clinical practice, the DRF depends on the angle at which scattered radiation from the patient reaches the head (McVey et al., 2013; Magee et al., 2014; Van Rooijen et al., 2014), and often the interventionalists' eyes are irradiated from the side and below, at an angle between 20° and 90° with the horizontal plane. DRF values tend to be lower for irradiation from the side due to the larger spaces left between the glasses and the head for the prescription spectacles (Magee et al., 2014). For the majority of the time that interventionalists carry out a procedure when x rays are being emitted, they will be viewing the images on the monitor rather than looking towards the patient. Then, scattered radiation may be able to pass through gaps behind the lenses and through parts of the frame that are not protected to irradiate the eyes directly. (175) Also, unattenuated x rays incident on tissues that are close to the eyes and scattered from these tissues become a major source of exposure to the lens of the eye when protective eyewear is worn (Moore et al., 1980; Cousin et al., 1987; Marshall et al., 1992; McVey et al., 2013; Koukorava et al., 2014; Magee et al., 2014). For exposures from the front, differences between various categories of glasses relate to the sizes of the lenses, and the proximity of unprotected and therefore irradiated tissue. For exposures from the side, the eye dose depends on the closeness of fit to the facial contours and the extent of protection from the side. When the radiation scattered by the patient is incident towards the eye from below, it may enter directly through the gaps underneath the lenses of the glasses, without any additional scattering. (176) The majority of lead glasses have a protection equivalent to 0.75 mm or 0.5 mm of lead, and many have protection in side shields of 0.5-mm or 0.3-mm lead equivalence. The designs can be divided into a number of categories which are listed below:
(A) Purpose-designed lead glasses with large flat lenses and protective side shields. (B) Wraparound lead glasses with front lenses angled to provide more protection for radiation incident from the side. (C) Lead glasses adapted from conventional spectacles with lead glass side shields added. (D) ‘Fit-over’ glasses, similar in design to Category (A), but arranged to fit over conventional spectacles. (E) Facemasks of lower lead equivalence, held in place by a headband. (177) Custom-designed lead glasses of Categories (A) and (B), having a lead equivalence of 0.75 mm, provide protection for the eyes with DRFs between 3.5 and 6 (Cousin et al., 1987; Vanhavere et al., 2012; Koukorava et al., 2014; Magee et al., 2014; Principi et al., 2015; Martin, 2016), and 0.50-mm lead equivalence glasses might provide DRFs of 3 to 4. Wraparound lead glasses provide better protection for radiation incident from the side and below because the gaps between the frames and head tend to be smaller. Glasses based on adaptations of standard spectacles of 0.75-mm lead with added side shields have DRFs between 3 and 4, as gaps between the glasses and the head tend to be larger (Magee et al., 2014). (178) ‘Fit-over’ glasses designed to be worn over prescription spectacles are bulky, and have larger gaps underneath to allow wearing of conventional spectacles. Verification that critical parts of the frames are of protective material is important, as some models, particularly the heavier ‘fit-over’ glasses, do not use protection in the frames in order to keep the weight down. (179) For head positions behind a ceiling-suspended screen, Galster et al. (2013) reported additional DRFs for lead glasses between 1.8 and 5.8. (180) Facemasks or visors of lower lead equivalence such as 0.1 mm cover the whole of the face, and so reduce the exposure of regions of the head surrounding the eyes that would make a significant contribution to the dose to the eye from backscatter (Martin, 2016). Despite the lower lead equivalence, they provide a viable alternative to lead glasses, but are sometimes not favoured by clinicians due to their size and their tendency to fog. (181) Measurements of the protection offered by lead glasses can provide useful data from which adjustments to dosimeter reading values recorded by unshielded eye dosimeters can be made to derive a dose representing that to the lens of the eye for any interventionalists for whom it could be guaranteed that they wore the protective eyewear consistently. However, any calculations assume that lead glasses are worn for every procedure. Therefore, for an attenuation factor to be applied, quality controls should be in place with regular documented checks to confirm that the interventionalist concerned always wears the protective eyewear. (182) The factor applied could be one based on measurements with the glasses concerned, but should take account of exposure from x rays at angles encountered in clinical practice. The measurement technique and the results should be documented, and the DRF applied should not be greater than 4. (183) Where no measurements are available to confirm the DRF, but the glasses are of Category (A) or (B) and incorporate the equivalent of at least 0.5 mm of lead, a DRF of 2 represents a conservative approach to account for the protection offered by the glasses (Magee et al., 2014). (184) The protection afforded to the contralateral eye by almost all models of lead glasses is lower than that to the eye adjacent to the x-ray source, with DRFs for the contralateral eye being between 1.5 and 2 (Geber et al., 2011; Galster et al., 2013; Van Rooijen et al., 2014; Fetterly et al., 2017). This is due to penetration of radiation through the gap corresponding to the nasal cavity, when the head is at an angle to the direction of the beam, and because a substantial portion of the dose is due to scatter from within the surrounding tissues. However, the dose to the lens of the contralateral eye is still less than that to the eye adjacent to the source. (185) In summary, a variety of lead glasses are available, but care should be taken in their selection. A close fit to facial contours, particularly around the underside of the glasses, can be more important than lead equivalence, as the glasses should also provide protection against exposures from below and from the side.
5.5.4. Combined use of protective means
(186) In the framework of the ORAMED programme, Monte Carlo simulations of clinical conditions and geometries and measurements were performed to determine the effect of different protective devices on radiation doses to the lens of the eye and extremities. The results include the following: the ceiling-suspended shield can reduce the dose to the eyes two to seven times; protective glasses can reduce the dose to the eyes up to 10 times (90%); shielding curtains from the table can reduce the dose to the legs two to five times; the x-ray tube under the table can reduce the dose to the eyes two to 27 times and reduce the dose to the hands two to 50 times as compared with the x-ray tube over the table; femoral access to the arterial system can reduce the dose two to seven times as compared with radial access, when proper shielding is used; and stepping back or leaving the room for image acquisition can reduce the dose four to seven times (Vanhavere et al., 2012; Martin, 2016). (187) Thornton et al. (2010) evaluated the impact of common radiation-shielding strategies, used alone and in combination, on scattered dose to the fluoroscopy operator’s eyes. Operator phantom lens radiation dose rate was recorded with and without a leaded table skirt, non-leaded and leaded (0.75-mm lead equivalent) eye glasses, disposable tungsten-antimony drapes (0.25-mm lead equivalent), and suspended (0.5-mm lead equivalent) transparent leaded shields. Lens dose measurements were also obtained in right and left 15° anterior obliquities with the operator at the upper abdomen, and during DSA (two images per second) with the operator at the patient’s groin. Each strategy’s shielding efficacy was expressed as a reduction factor of the lens dose rate compared with the unshielded condition. Use of leaded glasses alone reduced the lens dose rate by a factor of 5–10; and scatter-shielding drapes alone reduced the dose rate by a factor of 5–25. Use of both implemented together always provided more protection than either used alone, reducing the dose rate by a factor of 25 or more (Thornton et al., 2010).
5.6. Protection of the extremities
5.6.1. The hands
(188) The hands of interventional clinicians can be close to the primary x-ray beam. If the operator's hands stray into the beam transmitted through the patient, the absorbed dose rate above the patient would typically be 2–5 µGy s−1, so a 1-min exposure would give a dose from 100 to 300 µGy. Doses from primary x rays scattered from the surface of the patient on the tube side of the couch will be higher, and direct exposure to the incident primary beam could be 50 times greater. (189) Employing different access routes for the intervention influences the positions of the operator's hands during procedures and has a substantial effect on the dose level (Fig. 5.3). For cardiologists, introduction of catheters via the radial artery rather than the femoral artery has advantages in achieving patient mobility more quickly, but the cardiologists’ hands are closer to the x-ray beam and so the doses they receive, particularly to the side of the hand, are higher (Mann et al., 1996). (190) In interventional radiology, femoral access is used much of the time, but percutaneous procedures such as percutaneous biliary drainage, nephrostomy tube placement, and gastrostomy placement require the operator to manipulate catheters inserted close to the area being imaged, and thus can give relatively high doses to the finger tips (Whitby and Martin, 2005). (191) In procedures such as TIPSS, in which the radiologist gains access via the internal jugular vein, the hands are located further from the area being imaged, but TIPSS procedures can be technically challenging, fluoroscopy times are long, and doses are relatively high (Fig. 5.4). (192) The hand that holds the catheter is usually closer to the edge of the x-ray beam and receives the higher dose, while the other hand performs the manipulations (Figs 5.5 and 5.6). (193) Ceiling-suspended shields provide good protection for the head and upper body, but the hands are generally positioned below the shield and so receive less protection. However, some reduction can be achieved with careful practices (Maeder et al., 2006). Lead/rubber drapes attached to the bottom edge of the shield can be effective in protecting the hands for some procedures (Vanhavere et al., 2012). (194) Freestanding adjustable over-table shields can shield the operator’s hands, but the hands may stretch underneath the shield and so receive less protection. Protective drapes and pads can also offer good protection for the hands, and have been shown to achieve a 29-fold reduction in the dose to the hands in one study (King et al., 2002). (195) Thin protective gloves are available, but reports of the protection offered are varied (15–60%). If a hand protected by a glove strays into the x-ray field, the dose rate will be increased automatically to compensate for the attenuation, thus increasing patient exposure without achieving any protection of the hand of the interventionalist (Wagner and Mulhern, 1996). New shielding materials (e.g. bismuth) have also been proposed as a hand cream for hand protection, subsequently to be covered with a surgical glove to provide containment of the cream material (McCaffrey et al., 2012). This cream has the same potential to increase dose if the hand is placed in the x-ray field. Moreover, the reduction in tactile feedback from radiation-attenuating material may lead to an increase in fluoroscopy time or CT exposure time for delicate procedures (NCRP, 2010).
5.6.2. The legs and feet
(196) When the x-ray tube is positioned below the couch, radiation from the primary beam is scattered downwards from the base of the couch, so the legs can receive a substantial dose. If no shield is available, doses to the legs can be greater than those to the hands. The dose to the interventionalist’s feet is closely related to the kerma-area product when no protection is used; procedures having a kerma-area product of 100 Gy cm2 give an absorbed dose to the legs of approximately 1 mGy (Whitby and Martin, 2003). (197) Lead curtains attached to the side of the couch, typically with a lead equivalence of 0.5 mm, provide the operator with the best protection (Whitby and Martin, 2003; Shortt et al., 2007). These drapes can reduce doses to the legs by factors of 10–20 if positioned correctly throughout a procedure (Martin, 2009), but factors between 2 and 7 are typical in practice (Vanhavere et al., 2012). Such drapes should be specified for all interventional facilities. (198) A lead curtain that is attached to the table and hangs down from it has the advantage of being as close as possible to the source, and is always in place so no conscious decision is needed to use it. For the majority of procedures, where the interventionalist stands at the side of the table, a lead drape attached to the table provides a good option. However, it rarely protects the feet fully. (199) Usually the leaded curtain attached to the table does not extend for the full length of the table, so positioning is important for protection of both the operator and assistants. Operators standing at the side of the table will be adequately protected, but when an interventionalist stands at or near the head of the table, as in the case of TIPSS procedures, the drape will only provide protection for the interventionalist if it can be moved to the head of the table. These shields may be less effective for procedures in which the interventionalist is positioned near the head or foot of the table. For such procedures, other staff may need to stand to the side of the table, and they will require leg protection. (200) Mobile freestanding shields are available for protecting the legs. A conscious decision needs to be made to put them in place before the start of the procedure in order to preserve a sterile environment. There is a risk of collision with the couch and C-arm when either is moved up and down, tilted or angled. Such shields may also be used for protecting other staff who are assisting with procedures. (201) Stepping back from the couch during radiography is an effective method to reduce occupational dose; this is rarely possible during fluoroscopy, as the operator must be close enough to the patient to perform the procedure, but it is possible during acquisition series and during injection of contrast media using an automatic injector.
5.7. Protection in positron emission tomography-computed tomography interventional procedures
(202) Personal protective equipment, such as protective aprons and glasses, for conventional fluoroscopically guided interventions are ineffective against the 511-keV annihilation energy of PET photons (Ahmed et al., 2007). Once the patient has been injected with the radiopharmaceutical, the interventionalist has minimal control over the radiation emitted from the patient, in contrast to procedures guided fluoroscopically or by CT, where the amount and quality of x rays is controlled directly by the operator. Therefore, PET-CT-guided procedures require careful design of the PET-CT suite to optimise staff and adjacent room shielding (Madsen et al., 2006; Cruzate and Discacciatti, 2008; IAEA, 2008; Elschot et al., 2010) to ensure protection. As shown in Section 2, the major determinant of radiation exposure to the operator from PET-CT-guided interventional procedures is time spent in close proximity to the patient, and reducing this time is an important occupational radiological protection factor. The same considerations apply to interventions guided by PET/fluoroscopy.
5.8. Protection in selective internal radiation therapy
(203) All vials containing 90Y activity, and all instruments and disposable items used for preparing the dose and implanting the device should be handled with forceps and appropriate shielding to reduce finger doses. Due to the high-energy beta emission, shielding is best provided with a low atomic number material such as poly (methyl methacrylate). Vendors of SIRT spheres provide advice and training material to minimise the risk of contamination to staff, patients, and the room (SIRTEX, 2016). This includes the use of special shielding boxes for preparation and injection. Furthermore, double gloves are recommended to allow removal of a contaminated outer glove with a gloved hand. For implantation of the microspheres, the vendor provides an acrylic delivery box and delivery set. This prevents direct contact with the 90Y vial and all stopcocks or tubes. It is essential to flush all tubes and catheters with water or saline for injection before manual manipulation. Table 5.1 gives a representative overview on typical exposure of the different staff members for a single SIRT procedure. (204) In addition to all technical measures of radiological protection, training to speed up all steps of the procedure leads to a significant reduction of occupational exposure. Aubert et al. (2003) demonstrated the extremity dose reduction by optimising the 90Y injection technique. They found an extremity dose reduction by a factor of more than 10 after optimisation of the procedure. (205) After SIRT, the patient requires observation, general nursing care, and accommodation. In many facilities, patients are transferred to single rooms in a nuclear medicine department, although the radiation exposure to staff, visitors, and other patients is relatively low. In 143 SIRT procedures (124 with resin spheres and 19 with glass spheres), McCann et al. (2012) determined mean equivalent dose rates of 1.1 µSv h−1 at 1 m for resin spheres and 2.4 µSv h−1 at 1 m for glass spheres. Typical dose equivalent rates 6 h after implantation of 2 GBq 90Y activity (SIRTEX, 2016) are shown in Table 5.2 for different distances.
5.9. Handling, storage, and testing of protective garments
(206) Acceptability criteria should be established and applied in the facility. Adequate resources should be allowed for the purchase, testing, and replacement of protective garments. Protective aprons should never be folded, as cracks in the protective lining can develop at the fold. Protective aprons should be inspected visually prior to each use for damage and defects, kinks, and irregularities. (207) Protective garments should be inspected with x rays for any defects in the protective material upon receipt, and annually thereafter for any deterioration. Clements et al. (2015) developed a new evaluation method using CT to reduce evaluation time, reduce staff exposure, and provide evidence that testing occurred by storing the images. Archived images are also used for future comparisons. Standardised methods for acceptance testing of protective aprons are needed due to the wide variation in actual attenuation values of aprons (CRCPD, 2001; Christodoulou et al., 2003; Finnerty and Brennan, 2005). With regard to lead-free protective aprons, transmission measurements should use broad x-ray beams, and involve x-ray spectra similar to those found in the radiation fields associated with the interventions performed in the facility where the aprons will be worn, including scattered x-ray spectra as proposed by Pasciak et al. (2015). (208) Instructions and procedures to clean protective equipment while avoiding damage of the item should be included in the quality assurance programme (Vañó et al., 2016).
5.10. Education and training
(209) Staff involved in interventional procedures need education and training in radiological protection, and in applying the quality assurance programme. The training should include distribution of scattered radiation levels around a patient, understanding of how different factors influence the dose distribution, strategies for exposure monitoring and dose assessment, and familiarity with the effective use of protective devices, such as ceiling-suspended shields, leaded eyewear, and shielding curtains and drapes. This knowledge should be achieved by initial training, and maintained and updated through continuous education, consistent with the evolution of technology. (210) Medical physicists or radiological protection specialists providing support to interventional facilities should have the highest level of training in radiological protection as they have additional responsibilities as trainers for interventionalists and other health professionals involved in the interventions (ICRP, 2009). (211) Given the close relationship between protection of patients and occupational protection in interventional procedures, personnel in charge of occupational protection, regulators, dosimetry services staff, and clinical applications specialists from suppliers need not only knowledge of general radiological protection, but also knowledge of clinical practice in interventional procedures and the radiological equipment used. Additionally, dosimetry services staff need background knowledge of radiation qualities and scatter radiation fields, including pulsed radiation, for calibrating dosimeters and for investigating abnormal dose values.
5.11. Records related to occupational protection
(212) The records to be kept are established as requirements in standards and regulations. Records of occupational exposure include information on the nature of the work in which the worker is subject to occupational exposure monitoring; results of exposure monitoring and dose assessments, including results of investigation of abnormal exposure values; information on work for other employers that involves radiation exposure; outcomes of health surveillance; initial and periodic education and training on radiological protection; and refresher courses. If eye protection is worn and adjustments are to be made to the readings of the eye dosimeter for the protection provided, a record should be kept to demonstrate that individual interventionalists have worn both their lead glasses and their eye dosimeter. Employers have to provide staff with access to records of their own occupational exposure. (213) Information on workload, in terms of procedures per year, is useful for optimisation of protection, and for comparing and investigating unusual exposure.
5.12. Need for a quality assurance system
(214) A comprehensive quality assurance programme should be established by the organisation. The programme should aim to maintain best radiological protection practice to ensure appropriate occupational exposure control (ICRP, 2007; IAEA, 2014a). Active participation of the staff involved in the use of radiation is advisable, taking into account the Commission’s recommendations for planned exposure situations. The programme should be part of the management system implemented at the institutional level, including regular and independent audits (internal and external). (215) Procedures should be in place for employment of new staff expected to be involved in interventions guided by radiological imaging to ensure the following: education and training in radiological protection; arrangements for obtaining and evaluating their previous dosimetric history; pre-employment health surveillance; and arrangements for sharing information with other employers in case the staff works in more than one place. (216) Procedures should be in place for the selection of appropriate radiation detectors and dosimetry equipment. These procedures should be developed following international recommendations, and should be in compliance with recognised quality standards. Arrangements for staff radiological protection and health surveillance should be in place, with monitoring of body, eye, and hand exposure as well as workplace monitoring, as set forth in the radiological protection programme. Personal protective devices, such as aprons, thyroid shields, and leaded eyewear, as well as ceiling-suspended shields and table-mounted curtains, should be in place and their features should be controlled regularly. (217) Results of personal exposure monitoring and workplace monitoring should be part of the programme, as well as the necessary corrective measures taken in response to unusual results. Personal dosimetry suppliers should document the accreditation and performance in dose assessment from the supplied personal dosimeters, and the information should be recorded and kept safe for the regulatory recommended time. (218) Procedures should include investigation, reporting and recording results, and audits of occupational doses, as well as corrective actions in case of incidents or accidents. (219) Procedures should address the requirement and instructions for wearing protective devices to the extent possible and compatible with the success of the interventions, including the use of ceiling-suspended shields and protective eyewear. Procedures should also include audits and recording of the wearing of protective eyewear, especially if a DRF is applied to dosimeter readings to account for the attenuation. (220) Radiological protection training and certification of interventional staff should be documented and subject to review at established periods or whenever there is a significant change. Induction training in the operation of the quality assurance system should be part of the strategy of the organisation. Administrative procedures including the assignment of responsibility for quality assurance actions and for reviewing and assessing the overall effectiveness of radiological protection measures need to be established, and be part of the quality assurance manual. (221) Since occupational protection is closely related to patient protection, the overall quality assurance programme should include quality control of the radiological equipment, acceptance testing and commissioning, full characterisation of the radiological equipment, calibration of the air kerma-area product meters, and quality control of the personal protective devices.
6. SUMMARY OF RECOMMENDATIONS
6.1. General
(222) The recommendations summarised in this section are a consolidation of the advice provided in Sections 3–5. Occupational exposure in interventional procedures is closely related to patient exposure, as most actions to reduce patient exposure also contribute to protection of workers; in addition, occupational protection requires proper use of protective garments and shielding. Actions to protect staff should not impair the clinical outcome of the intervention, and should not increase patient exposure. Therefore, occupational protection should be managed in an integrated approach with patient protection. Hospital staff responsible for radiological protection in interventional procedures should be familiar with these procedures.
6.2. Individual exposure monitoring
(223) Occupational exposure monitoring in interventional procedures has two major objectives: to verify compliance with dose limits, and to optimise occupational protection. (224) Compliance monitoring should not only include the assessment of effective dose, but also of doses that could be received by non-apron-protected organs, such as the lens of the eye and extremities. Recent studies have shown that there is a high incidence of radiation-related eye lens opacities in interventionalists, which emphasises the need for assessment of exposure of the lens of the eye. (225) The use of two dosimeters, one shielded by the apron (under apron) and one unshielded over the apron, at the collar, has been recommended by the Commission for interventional procedures as it provides not only the best available estimate of effective dose, but also a reasonable indication of the dose to the lens of the eye and the head, and confirmation that the protective apron has actually been worn. (226) Visual elements should be in place to help users place their own dosimeters in the correct position. Consistency analysis of the two readings allows an indication of the proper use of the dosimeters, making the monitoring system more robust. (227) Optimisation monitoring evaluates the effect of protective actions to reduce staff doses without impairment of the success of the procedures. Over time, the impact of optimisation will appear through lower occupational doses. APDs have proven to be useful for optimisation purposes, for studies of radiation exposure by type of procedure or for specific aspects of a procedure, and for educational purposes. (228) Type-test procedures and calibration of APDs and area monitors should include radiation fields representative of those encountered in interventional procedures, including tests in pulsed mode with high dose rates. (229) Improved technology and methodology is needed to assess dose to the lens of the eye when lead glasses are worn. (230) The Commission recommends that proper dosimeters should always be worn and in the right position defined by procedures, and that audits of compliance with procedures should be performed. In addition, ambient dosimeters are useful to assess scatter radiation fields continually, and to provide backup to personal dosimetry. Comparing individual dosimeter readings with those of an ambient dosimeter near the patient (such as on the C-arm) may be helpful in discovering non-compliance with procedures for wearing individual dosimeters, as the ambient dosimeter can provide a reasonable estimate of occupational exposure, especially doses to the unshielded lens of the eye. For managing optimisation of protection, investigation levels are required to provide an alert when radiation exposure is higher than normal, and a review of the working conditions is needed. In addition, a low-dose investigation level for the reading of over-apron and hand dosimeters can also be used to trigger a review of whether dosimeters are worn consistently and properly when the readings of these dosimeters are lower than expected. (231) The operational quantity Hp(0.07) can be used as an approximation to Hp(3) for photon radiation of all energies used in radiology in general; Hp(10) can also be used for the same purpose, but only if the photon spectrum has a mean energy above 40 keV. (232) Wrist dosimeters may not be able to reflect actual finger doses, if parts of the hands are very close to or even introduced into the direct x-ray beam. (233) Consideration should be given to assess doses to the parts of the legs that are not shielded by the protective apron or table-suspended curtains. (234) Research efforts should pursue the development of computational technologies (not requiring dosimeters) together with personnel position sensing to assess personnel doses, including dose to the eye. (235) The radiological protection programme should include audits of occupational doses, investigation of abnormal exposure, reporting and recording results, and corrective actions if appropriate.
6.3. Occupational radiological protection methods and devices
(236) Actions for patient protection generally protect staff in a similar proportion. In addition, the following means and actions are applicable specifically for occupational protection: protective apron and collar; ceiling-suspended shield and leaded eye glasses; table-suspended leaded curtains; stepping back to increase distance from the patient; and staying on the side of the image receptor rather than on the side of the x-ray tube. (237) There are lighter-weight aprons that contain composite layers or bi-layers of high atomic number elements such as tin or bismuth, instead of lead. Characterising attenuation properties only in terms of ‘lead equivalence’ can be misleading, since photon attenuation varies significantly over the photon energy spectrum, with the largest variations occurring in the imaging range. Attenuation factors should be specified with information on the radiation beam qualities used to measure the attenuation and the weighting of measurements made at different beam qualities, in order to reflect the conditions under which the garment is used. (238) If no protective measures for the eyes are used, personnel with a typical workload will receive doses to the lens of the eye that could exceed the dose limit, and could result in lens opacities over time. Interventionalists should make use of ceiling-suspended shields whenever possible while working. The effectiveness of these shields depends on their positioning and proper use. (239) When protective leaded eye glasses are worn, dose to the eye results primarily from radiation backscattered from surrounding tissues in the interventionalist’s head. In addition, during fluoroscopy, the interventionalist is usually looking at the image monitor, and so the lens of the eye is exposed by radiation coming from the side and from below the level of the head. Leaded glasses should, therefore, fit closely to the wearer’s facial contours. Dose to the lens of the eye can be reduced by a factor of 2–7 by the use of leaded glasses. (240) The hand of the interventionalist that is closer to the x-ray beam and to the irradiated volume of the patient receives the higher dose. Leaded drapes attached to the bottom edge of the ceiling-suspended shield, and drapes and pads applied on the patient can be effective in protecting the operator’s hands for a number of procedures. Such drapes may have an aperture through which catheters can be inserted. (241) The operator’s feet may be exposed even when lead curtains suspended from the table are in place, due to the presence of a gap between the curtains and the floor. This is especially true when the couch is in a higher position. Interventionalists should step back from the couch during cine or DSA acquisition whenever possible, as well as during injection of contrast media when an automatic injector is used. (242) The specification of the protective value of garments should be accompanied by an indication of the characteristics of the radiation beams used to measure the attenuation. The combination of measurements made at different beam qualities should reflect the conditions under which the garment is used.
6.4. Protection of pregnant workers
(243) The early part of pregnancy (before the pregnancy has been declared) is covered by the normal protection of workers. Once the pregnancy has been declared and notified to the employer, additional protection of the fetus should be considered. The working conditions of a pregnant worker, after the declaration of pregnancy, should be such as to make it unlikely that the additional dose to the conceptus will exceed approximately 1 mGy during the remainder of the pregnancy. (244) Unnecessary discrimination against pregnant workers should be avoided. Currently available data do not justify automatically precluding pregnant interventionalists or other workers from performing procedures in the intervention room. (245) When two individual dosimeters are used, the under-apron dosimeter should be worn on the abdomen for monitoring the dose to the conceptus. If this dosimeter shows a value for personal dose equivalent [Hp(10)] of <0.2 mSv month−1, the equivalent dose to the conceptus would be below the dose limit.
6.5. Storage and quality control for protective garments
(246) Adequate resources should be allowed for the purchase, testing, and replacement of protective garments. Acceptability criteria should be established and applied in the facility. (247) Protective aprons should never be folded, as cracks in the protective lining can develop at the fold. Protective aprons should be inspected visually prior to each use for damage and defects, kinks, and irregularities. They should also be inspected with x rays for any defects in the protective material upon receipt, and annually thereafter for any deterioration. (248) Written procedures to clean protective equipment while avoiding damage to the item should be included in the quality assurance programme, and followed carefully.
6.6. Quality assurance programme
(249) A comprehensive quality assurance programme should be established by the organisation. The programme should aim to maintain best radiological protection practices to ensure appropriate occupational exposure control. The programme should include appropriate audits to ensure that personnel adhere to procedures, especially related to wearing of dosimeters, protective devices, and methods to optimise occupational protection.
6.7. Education and training
(250) Staff involved in interventional procedures need initial and periodic education and training in applying the quality assurance programme, including strategies for exposure monitoring and dose assessment, and protection methods and garments. (251) Given the close relationship between protection of patients and that of the staff involved in interventional procedures, personnel in charge of occupational protection, dosimetry services staff, clinical applications specialists from suppliers, and regulators need not only knowledge of general radiological protection, but also knowledge of clinical practice in interventional procedures and the characteristics of x-ray equipment used. (252) Medical physicists and radiological protection specialists who provide support to interventional facilities should have the highest level of training in radiological protection, as they have additional responsibilities as trainers for interventionalists and other health professionals involved in interventions (ICRP, 2009). Dosimetry services staff need background knowledge of clinical practice in order to calibrate dosimeters (e.g. radiation qualities, scatter radiation fields, pulsed radiation) and to collaborate with the user in investigating abnormal dose values.
6.8. Records
(253) Records on occupational exposure should include information on the nature of the work, exposure from work for other employers, outcomes of health surveillance, education and training on radiological protection (including refresher courses), and results of exposure monitoring and dose assessments, including results of investigation of abnormal exposure values. Employers must provide staff with access to records of their own occupational exposure.
