Abstract
Risks of non-cancer outcomes after exposure to atomic bomb (A-bomb) radiation have been evaluated among the Life Span Study (LSS) cohort and its subcohort, the Adult Health Study (AHS). Information regarding non-cancer outcomes in the LSS is obtained from death certificates. In the AHS, members undergo clinical examinations biennially to determine their health status. Many AHS studies have been limited to participants attending the clinic over a limited period, and therefore have varying degrees of inferential utility; as such, care is required for comparison with the LSS results. Disease structure of non-cancer diseases in Japan has changed over the long follow-up period since the end of World War II. The health status of the A-bomb survivors may be associated with the hardships of living in a devastated city and impoverished country following the prolonged war effort, in addition to the direct effects of radiation exposure. Radiation-related risk of cardiovascular disease may have increased due to radiation-related increased risk of hypertension and other secondary associations, and the risk of atherosclerotic disorders has also been reported recently. These results should be interpreted with caution because of changes in disease definitions over the follow-up period. The radiation-related risk of non-cancer respiratory diseases also appears to have increased over the follow-up period, but the shapes of the dose–response curves have shown little consistency.
1. INTRODUCTION
The Life Span Study (LSS) cohort consists of approximately 120,000 atomic bomb (A-bomb) survivors who were exposed to A-bomb radiation at various distances from the hypocentres. The members of its subcohort, the Adult Health Study (AHS), were selected at random from the LSS members, stratified by distance from the hypocentres, acute radiation syndrome, and the condition that they could participate in biennial health examinations at the Atomic Bomb Casualty Commission and Radiation Effects Research Foundation (RERF) in Hiroshima and Nagasaki, Japan. The total number of candidates for the AHS was 24,000 of which approximately 20,600 had at least one examination. All members of the LSS have been followed up nationally for their vital status and cause of death since 1950, and the AHS health examination programme was initiated in 1958 (RERF, 2014). Cause of death, including non-cancer diseases, is collected for all LSS members via death certificates. The accuracy of non-cancer death diagnoses based on death certificates is sometimes considered uncertain. More detailed and precise clinical information is available from the AHS members, but the analyses have usually been limited to examinations within specific periods because the procedures and medical technologies have changed over time. Therefore, care must be taken when interpreting results from the LSS, and when comparing the AHS and LSS results.
In addition, disease structure of non-cancer diseases in Japan has changed over the long follow-up period of the A-bomb survivors. In the devastated post-war era, infectious and infection-related diseases and cerebral haemorrhage were dominant based on poor hygiene and nutrition status. In contrast, cancers, cerebral infarction, and heart disease have increased with improved economic conditions and westernisation of the Japanese lifestyle. Therefore, the overall health status of the A-bomb survivors may be associated with the poor conditions since the war, changes in disease incidence over time, and pathogenesis of the diseases, as well as the effects of radiation exposure. In analyses of LSS mortality, excess relative risk per gray (ERR Gy−1) was 0.11 [95% confidence interval (CI) 0.05–0.17] for circulatory disease and 0.21 (95% CI 0.10–0.33) for respiratory disease (Ozasa et al., 2012). The findings for these diseases are detailed below.
Tissue reaction is assumed to be induced by cell loss or injury due to radiation exposure at relatively high dose levels, typically several gray or higher, with a threshold. It is not certain whether long-term non-cancer effects of radiation exposure at relatively low dose levels, below a few gray, are induced by typical tissue reactions or other mechanisms (ICRP, 2007). A concave dose–response curve might indicate a potential threshold, and a linear dose–response might suggest mechanisms including stochastic effects. Observations in the A-bomb survivors may provide important clues.
2. CARDIOVASCULAR DISEASE
2.1. Atomic bomb survivors
In an analysis of deaths over the period 1950–2003 among the LSS cohort, the estimated radiation-related ERR Gy−1 for mortality from all heart disease was 0.14 (95% CI 0.06–0.23) with a linear dose–response, and that for stroke was 0.09 (95% CI 0.01–0.17) on the basis of a linear dose–response, although an indication of possible upward curvature was suggested. The risks varied by disease subtype (Fig. 1). Among heart disease, ERR Gy−1 was increased significantly for hypertensive heart disease, rheumatic heart disease, and heart failure, but not for ischaemic heart disease. For stroke, ERR Gy−1 was not increased for cerebral infarction, cerebral haemorrhage, or subarachnoid haemorrhage, but was increased for other or unspecified types of stroke (Shimizu et al., 2010). Therefore, increased ERR Gy−1 was observed for deaths from rather ill-defined diseases.
Excess relative risk (ERR) Gy−1 for cardiovascular disease subtypes, Life Span Study, 1950–2003. Upper hierarchical categories are indicated by open diamonds. Bars represent 95% confidence intervals. Data from Shimizu et al. (2010).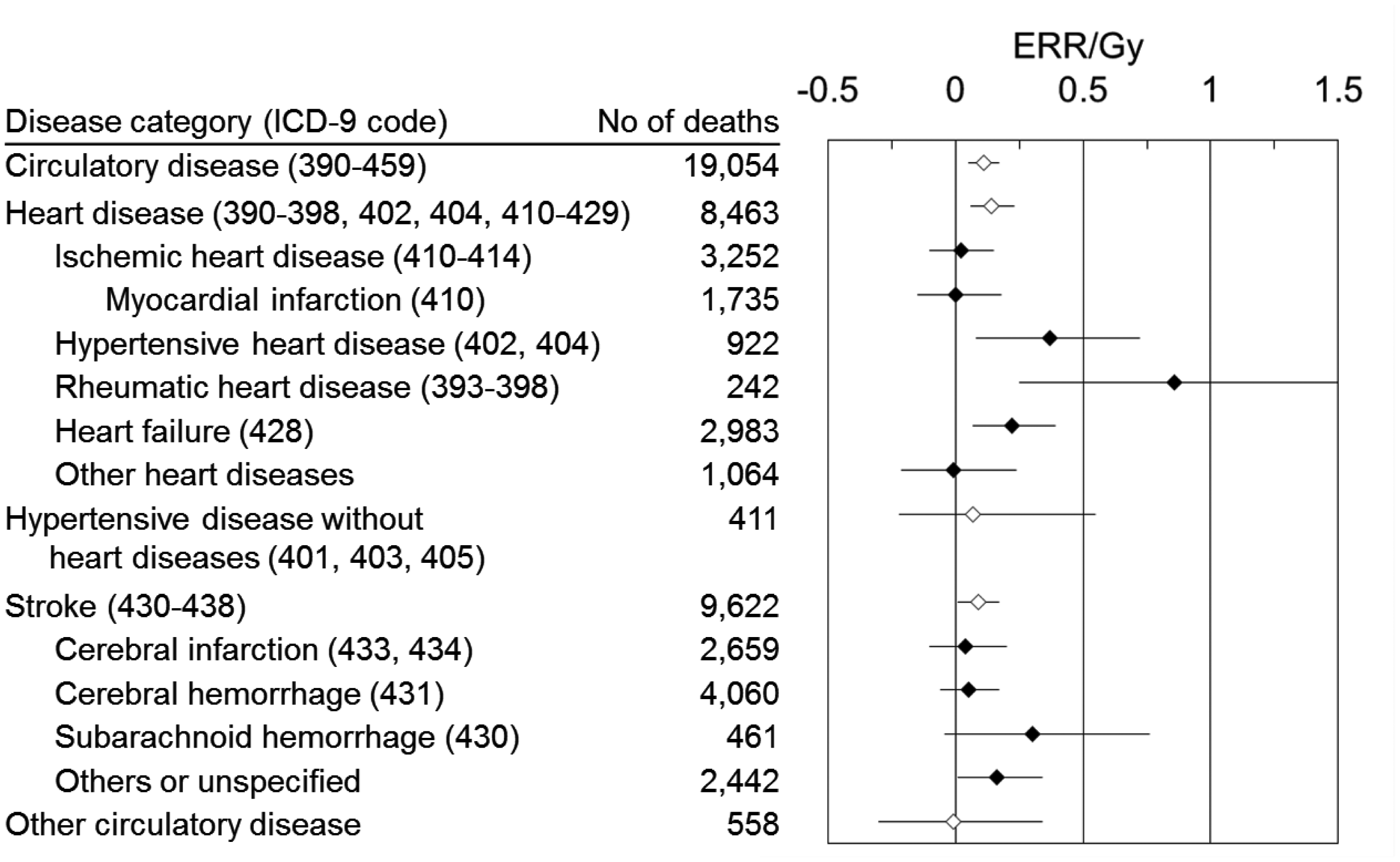
As information on non-fatal outcomes is available in the AHS, associations between radiation exposure and incident circulatory diseases were analysed. Significant quadratic dose–responses were observed between radiation exposure and incidence of non-fatal myocardial infarction between 1968 and 1998 among those exposed at less than 40 y of age [relative risk at 1 Gy (RR1 Gy) 1.25], and the incidence of essential hypertension between 1958 and 1998 was also increased among all subjects (RR1 Gy 1.03). When using a linear dose–response model, on the other hand, radiation-related risk was not increased for hypertension, hypertensive heart disease, ischaemic heart disease, myocardial infarction, vascular occlusion or stenosis, aortic aneurysm, or stroke (Yamada et al., 2004). In a recent report over the period 1980–2003 by Takahashi et al. (2012), radiation exposure was significantly associated with increased incidence of non-fatal and fatal haemorrhagic stroke in both sexes, and the effects in women were less apparent until doses exceeded a threshold at 1.3 Gy. No association was observed between radiation exposure and ischaemic stroke in either sex.
Kidney disease has been recognised as a risk factor for the development of circulatory diseases, partly through renal hypertension. A significant quadratic dose–response between radiation exposure and mortality from possible chronic renal failure (listed anywhere on the death certificate) was observed in the LSS, 1950–2003 (ERR Gy−1 = 0.09) (Adams et al., 2012). The association was similar in shape to relationships observed for radiation exposure and hypertension in the AHS, as mentioned above. In a subgroup of Nagasaki AHS members examined in 2004–2007, a significant association was found between radiation exposure and chronic kidney disease, consisting of moderate and severe renal dysfunction (odds ratio Gy−1 1.29), especially for severe renal dysfunction (odds ratio Gy−1 3.19), although the dose–response was not examined (Sera et al., 2013).
The findings described above may indicate that the shape of the dose–response curve for cardiovascular disease is non-linear, and is primarily dependent on the responses at high dose levels (>2 Gy). The associations may be mediated through hypertension rather than atherosclerosis, although it has been suggested (e.g. in studies of patients treated with radiotherapy) that atherosclerosis is a possible intermediate cause of radiation effects through endothelial cell injuries or inflammation (UNSCEAR, 2008). Based on poor hygienic conditions in Japan following World War II, the authors believe that increased radiation-related risk of rheumatic heart disease may be associated with increased risk of streptococcal infection among survivors exposed to high-dose radiation (i.e. proximal survivors). As the radiation-related mortality risk of infectious disease has not increased (Ozasa et al., 2012), the possible mechanism of increased risk of streptococcal infection among proximal survivors may not be a true radiation effect, but may be related to poorer hygienic conditions compared with distal survivors.
2.2. Background in the general population of Japan
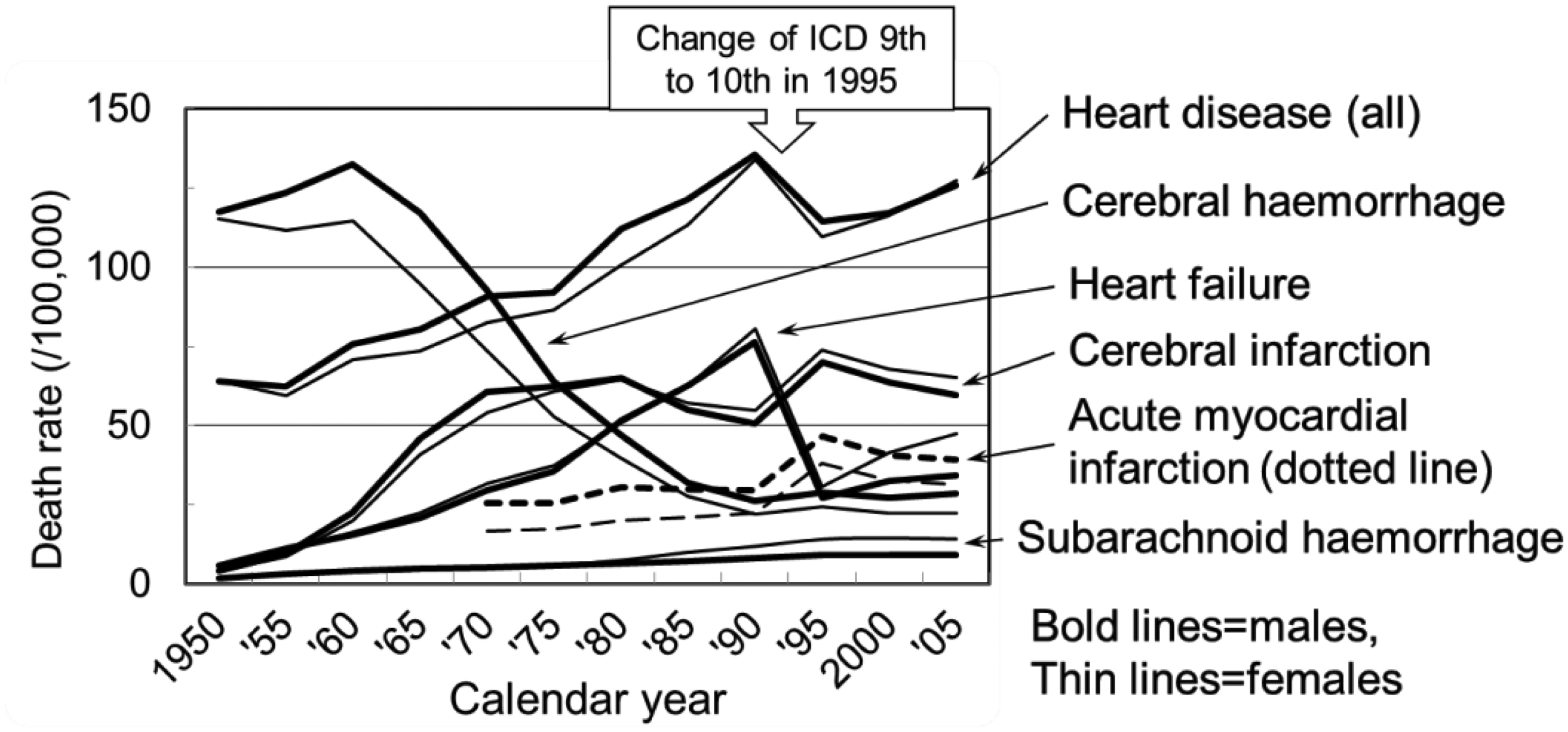
The incidence and pathogenesis of circulatory disease have been quite different between Japan and Western countries. Mortality from heart diseases, primarily ischaemic heart disease and arteriosclerotic/degenerative heart disease, has been higher in Western countries and is thought to be primarily due to atherosclerotic progression (Fig. 2). In Japan, cerebral haemorrhage was the most dominant circulatory disease in the past (Fig. 3). During the long period since World War II, which overlapped with the period of observation of the A-bomb survivors, cerebral haemorrhage decreased markedly, while cerebral infarction and heart disease, especially heart failure, increased until the 1990s.
Mortality of subtypes of heart disease in Japan and Western countries. Classification of diseases was changed in 1968 and 1995. Heart failure is included in ‘Other heart disease’. Data from Health, Labour and Welfare Statistics Association (1960, 1967, 1974, 1984, 1993, 2002), originally cited from the World Health Statistics, World Health Organization. Trend of crude mortality rates of circulatory diseases in Japan plotted with 5-y intervals, 1950–2005. Data from Ministry of Health, Labour and Welfare (MHLW, 1952–2006).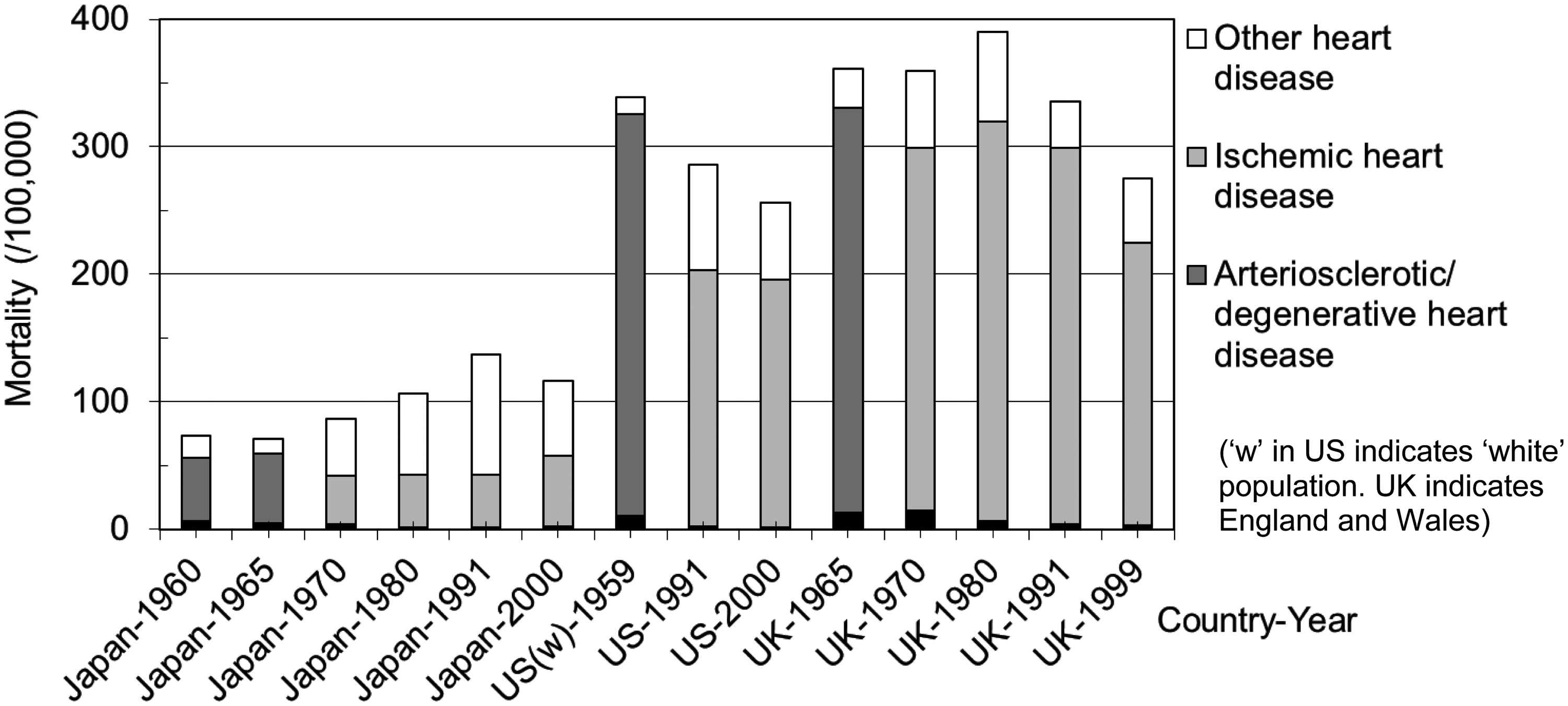
The most potent risk factor in Japan for stroke, either cerebral haemorrhage or infarction, is hypertension (Tanizaki et al., 2000; Ueshima, 2007). The Hisayama study found that the lacular type of cerebral infarction was more common than the atherosclerotic type in Japan, while the atherosclerotic and cardioembolic types were more common in Western countries. The lacunar type of cerebral infarction is based on arteriosclerosis of the cerebral parenchymatous small arteries induced by hypertension, and the latter two subtypes are based on atherosclerosis of cervical or intracranial large arteries (Tanizaki et al., 2000). Hypertension is also an important risk factor for ischaemic heart disease in Japan. Although hypercholesterolaemia can also be important, its prevalence in Japan is low in older generations (Ueshima, 2007). Therefore, the most influential risk factor for cardiovascular disease among the A-bomb survivors is thought to be hypertension. Thus, although the clinical manifestations of stroke and heart attack are similar in Western countries and Japan, the pathogenesis is thought to be different.
The increasing trend of deaths due to heart disease in Japan is primarily due to increased heart failure rather than increases in atherosclerotic or other specific diseases (Figs. 2 and 3). In 1995, the 10th edition of the International Classification of Diseases (ICD-10) was introduced, and Japanese medical doctors were instructed to avoid diagnosing ‘heart failure’ as the underlying cause of death on death certificates. Soon after that, the mortality rate of ‘heart failure’ dropped, and diagnoses of ‘myocardial infarction’ and ‘cerebral infarction’ increased (Fig. 3 is plotted with 5-y intervals). Apparent changes are therefore artificial and not natural. As an effective coroner system with autopsy has not been introduced in most areas of Japan, doctors may indicate ‘myocardial infarction’ or ‘cerebral infarction’ as the primary cause of death without specific confirmation for deaths that would probably have been diagnosed as ‘heart failure’ prior to the new directive in 1995. ‘Heart failure’ is considered to be a ‘garbage’ category of cause of death that includes various uncertain conditions, including hidden malignancies; this practice may continue today through inappropriate use of the ‘myocardial infarction’ or ‘cerebral infarction’ categories.
3. RESPIRATORY DISEASE
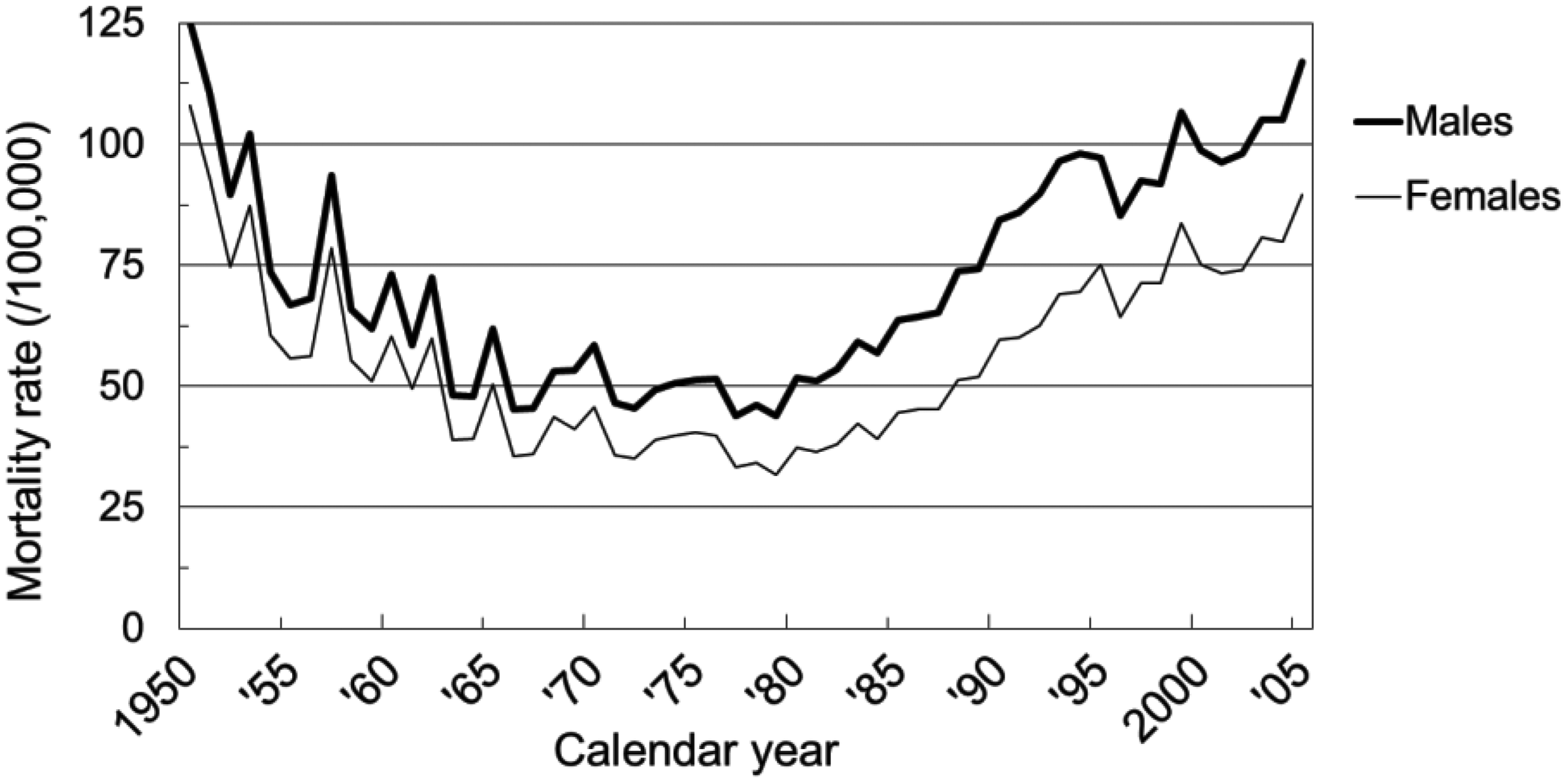
Radiation-related mortality risk of non-cancer respiratory diseases (NCRDs) increased by 17% Gy−1 (ERR Gy−1 0.17, 95% CI 0.08–0.27) with a linear dose–response between 1950 and 2005. Pneumonia and influenza were the dominant NCRD subtype (63% of deaths), and ERR Gy−1 was 0.20 (95% CI 0.09–0.34). The ERR Gy−1 of other subtypes (i.e. acute respiratory infections, chronic obstructive pulmonary disease, asthma, or others) was not increased significantly. Period-specific ERR Gy−1 of all NCRDs was 0.11 (95% CI −0.08–0.36), 0.08 (95% CI −0.09–0.29), and 0.21 (95% CI 0.10–0.34) with a linear dose–response in 1950–1964, 1965–1979, and 1980–2005, respectively, but a non-linear dose–response was observed over the period 1950–1964 (Fig. 4). The findings of all NCRDs reflect those of pneumonia and influenza (Pham et al., 2013). The difference in risk estimates may be associated with the different pathogenesis of NCRDs between the periods. In the early period, crude NCRD mortality consisted primarily of acute pneumonia and influenza involving people of all ages with periodical epidemics (Fig. 5). Bad hygiene, poor nutrition, and limited availability of medical care were all thought to contribute to the early high rates of mortality. Chronic obstructive pulmonary disease and asthma are exacerbated by acute infections, especially in such poor conditions. With the improvement of conditions in the 1970s, the mortality rate decreased and flattened. However, mortality increased again in the 1980s because pneumonia occurs frequently in the terminal stage of death in the elderly, such as aspiration pneumonia and infections in compromised hosts. The increase in crude mortality reflected the growing elderly population in Japan, so age-adjusted mortality did not increase after 1980 (data not shown). In Fig. 5, a drop in the mid-1990s was due to the change in the ICD edition, and a high peak in the late 1990s indicates excess deaths due to an epidemic of A/H3N2 influenza among elderly people.
Dose–response of excess relative risk (ERR) for all non-cancer respiratory diseases by period, Life Span Study, 1950–2005. The closed circles and bars represent point estimates of ERR and 95% confidence intervals for the dose categories. Solid lines show dose–response curves based on the linear-quadratic model. Data from Pham et al. (2013). Trend of crude mortality rates of all non-cancer respiratory diseases in Japan plotted with 1-y intervals. Data from Ministry of Health, Labour and Welfare (MHLW, 1952–2006).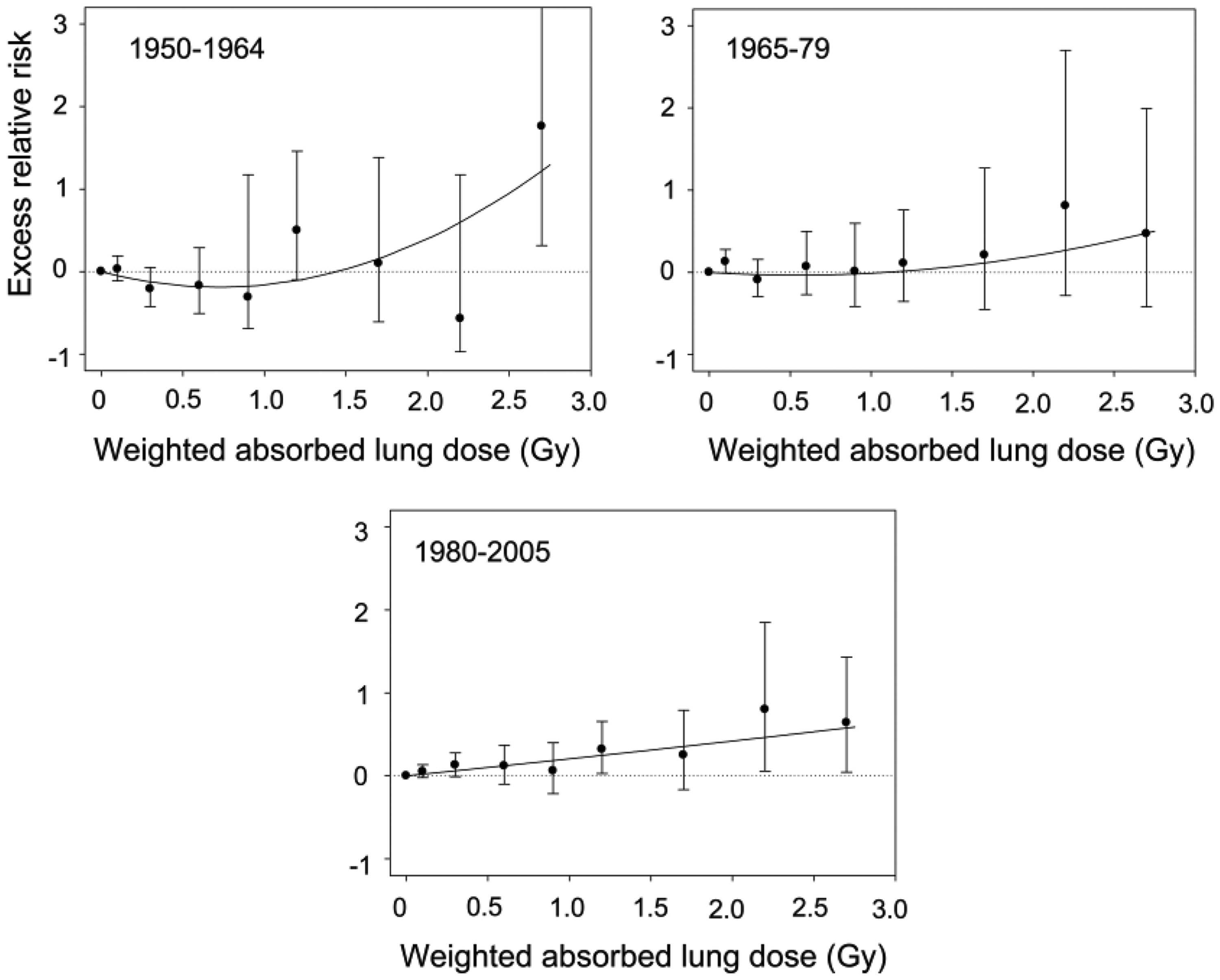
In the same way as heart failure for circulatory diseases, pneumonia as the terminal stage of death among the elderly is thought to include various uncertain conditions, including hidden malignancies. Evidence of this hypothesis was observed by results showing that the ERR of radiation for NCRDs decreased after excluding deaths with coincident cancer, using information from cancer registries and comorbidity on death certificates (Pham et al., 2013). The involvement of malignancies in the increased radiation-related risk of NCRDs may also be indicated by the linear dose–response in the period 1980–2005.
4. CONCLUSIONS
Increased radiation-related risks for some non-cancer diseases at relatively high dose levels with non-linear dose–responses have been observed. Those findings were based on mortality data from the LSS cohort, and incidence data including non-fatal outcomes from the AHS members. As heart failure (and, possibly, myocardial infarction and cerebral infarction since 1995) and pneumonia listed on death certificates are thought to include uncertain conditions at death, including hidden malignancies, the radiation-related risks of mortality/incidence of non-cancer diseases based on the information on death certificates need to be interpreted with caution. The AHS has an advantage in that the information was collected using standardised clinical measurements. In addition, background rates of non-cancer diseases have varied widely over the long follow-up period of the A-bomb survivors. Therefore, careful analysis by period, and careful interpretation of the results, are necessary regardless of whether the data were collected in the LSS or the AHS.
Footnotes
Acknowledgements
The RERF, Hiroshima and Nagasaki, Japan is a public interest foundation funded by the Japanese Ministry of Health, Labour and Welfare and the US Department of Energy. This paper was supported by RERF Research Protocols 1–75 and 2–75. The views of the authors do not necessarily reflect those of the two governments.
