Abstract
Objective:
To compare the effects of morning versus evening once-daily administration of intranasal mometasone furoate on nasal symptoms, sleep quality, daytime sleepiness, and quality of life in patients with moderate-to-severe seasonal allergic rhinitis (SAR).
Methods:
This prospective, randomized, single-blind study included 120 adults with SAR who received intranasal mometasone furoate (200 μg once daily) either in the morning or in the evening for 15 days. Outcomes were assessed using the Total Nasal Symptom Score, Rhinitis Quality of Life Scale, Pittsburgh Sleep Quality Index, and Epworth Sleepiness Scale. Baseline-adjusted comparisons between treatment groups were performed using analysis of covariance.
Results:
Both treatment groups showed significant posttreatment improvements in nasal symptoms, sleep quality, daytime sleepiness, and rhinitis-related quality of life. After adjustment for baseline values, no statistically significant differences were observed between morning and evening dosing for any outcome measure (all P > .05). Baseline disease severity emerged as the primary determinant of posttreatment outcomes, accounting for a substantial proportion of outcome variability, whereas dosing time showed no clinically meaningful effect.
Conclusion:
Once-daily intranasal mometasone furoate provides comparable clinical benefits regardless of whether it is administered in the morning or in the evening in patients with moderate-to-severe SAR. These findings suggest that dosing time has limited clinical relevance for long-acting intranasal corticosteroids, allowing treatment to be guided by patient preference without compromising efficacy.
Keywords
Introduction
Allergic rhinitis (AR) is a highly prevalent chronic inflammatory disease characterized by nasal congestion, rhinorrhea, nasal itching, and sneezing, frequently accompanied by ocular symptoms such as itching, redness, and tearing. In addition to nasal and ocular complaints, patients may also experience postnasal drip, cough, and palatal itching, all of which contribute to the overall symptom burden of the disease.1,2
Beyond upper airway symptoms, AR has a substantial negative impact on sleep quality, daytime functioning, and overall quality of life. Sleep disturbances are commonly reported among patients with AR, and the severity of nasal congestion has been shown to correlate directly with impaired sleep quality. Consequently, poor sleep may lead to daytime fatigue, excessive sleepiness, impaired concentration, and reduced productivity, thereby significantly affecting daily life and psychosocial well-being.3,4
Recent experimental and clinical observations indicate that allergic diseases, including AR, are closely linked to disturbances in circadian regulation, which may influence epithelial barrier function and immune responses. 5 Physiological nocturnal changes such as reduced cortisol secretion, increased vagal tone resulting in vasodilation and enhanced nasal secretions, as well as nocturnal allergen exposure, have been proposed to contribute to the diurnal variation of AR symptoms. Consistent with these mechanisms, nasal symptoms of AR are often reported to be most pronounced in the early morning hours.6,7
The well-recognized diurnal variation of AR symptoms has naturally raised the question of whether treatment timing could influence therapeutic outcomes. This concept underlies chronotherapy, a therapeutic approach that aims to optimize treatment efficacy by aligning medication administration with biological rhythms. 8 Chronotherapy has been investigated in various chronic inflammatory and respiratory diseases; however, its clinical relevance in the management of AR remains uncertain.8,9 In particular, intranasal corticosteroids—recommended as first-line therapy for moderate-to-severe AR—exert potent and prolonged local anti-inflammatory effects and are commonly prescribed as once-daily regimens.1,2
Mometasone furoate is a long-acting intranasal corticosteroid with low systemic bioavailability and a favorable safety profile. It has been shown to effectively control both daytime and nighttime nasal symptoms, as well as sleep disturbances associated with AR. 10 Despite its widespread use as a once-daily treatment, the potential influence of dosing time on its clinical efficacy has not been adequately investigated. Given its prolonged local activity, it remains unclear whether morning or evening administration provides any additional benefit in symptom control or sleep-related outcomes. 11
Although the theoretical rationale for chronotherapy in AR is compelling, its translation into clinically meaningful benefit remains unclear. Currently, there is a lack of clinical evidence addressing whether morning or evening administration of once-daily intranasal corticosteroids leads to superior improvement in nasal symptoms, sleep quality, daytime sleepiness, or quality of life. Clarifying this issue is clinically relevant, as rigid dosing-time recommendations may unnecessarily complicate treatment regimens and negatively affect patient adherence without offering additional benefit.8,12
Therefore, the aim of this study was to compare the efficacy of morning versus evening once-daily administration of intranasal mometasone furoate in patients with moderate-to-severe seasonal allergic rhinitis (SAR), with respect to nasal symptoms, sleep quality, daytime sleepiness, and quality of life.
Materials and Methods
Study Design and Participants
This prospective, randomized, single-blind study included 120 patients with moderate-to severe SAR (46 males, 74 females) who applied to the Immunology and Allergy Department (Figure 1). The diagnosis and management of allergic rhinitis were established in accordance with the Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines. The study protocol was approved by the institutional Ethics Committee on Human Research (Decision No: 19/258), and written informed consent was obtained from all participants prior to enrollment.
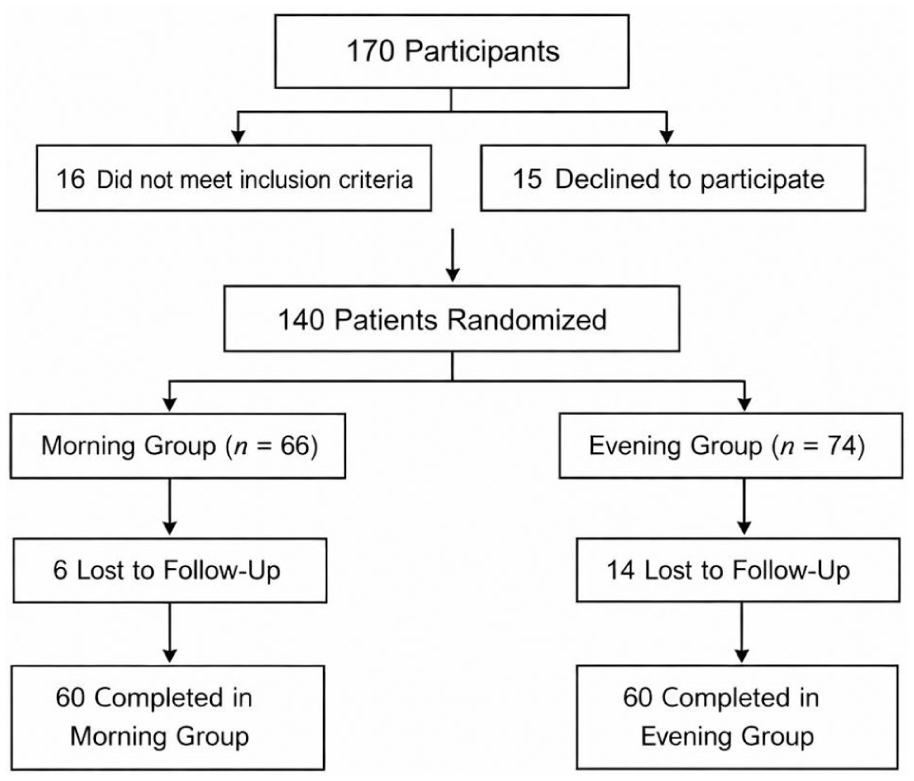
CONSORT flow diagram illustrating patient screening, exclusions with reasons, randomization, follow-up, and final analysis of the study population.
Sample size estimation was based on data from a previous similar study, assuming a 2-sided significance level of 0.05 and 90% statistical power. 13 The final sample size of 60 patients per group was considered sufficient to provide robust statistical power for detecting clinically meaningful differences.
Inclusion and Exclusion Criteria
Inclusion criteria were as follows: age between 18 and 60 years; a history of moderate-to-severe SAR for at least 1 year; and confirmation of seasonal allergy by a positive skin prick test to relevant seasonal aeroallergens (grass mix, cereals mix, weed mix, or tree mix). The classification in the ARIA guidelines was used to determine the severity of allergic rhinitis. According to ARIA guidelines moderate-to-severe allergic rhinitis should include at least one of these items; sleep disturbance, impairment of daily activities, impairment of school or work, and troublesome symptoms. 2 Patients were required to be free from any pharmacological treatment for SAR for at least 1 month prior to enrollment.
Exclusion criteria included structural or inflammatory nasal pathologies causing nasal obstruction (e.g., nasal septal deviation, nasal polyps), conditions associated with sleep disturbance (including obstructive or central sleep apnea, psychiatric or neurological disorders), unstable systemic diseases (pulmonary, cardiovascular, hematologic, hepatic, renal, neurological, psychiatric, or autoimmune disorders), and known corticosteroid hypersensitivity. Patients with a positive skin prick test to house dust mite, cat, or dog epithelia were also excluded. Additionally, patients who demonstrated poor treatment compliance or required rescue medication during the study period were excluded from the final analysis.
Randomization and Intervention
Participants were randomized in a 1:1 ratio using a computer-generated randomization list. The allocation sequence was kept by an investigator not involved in outcome assessments and was revealed only after participant enrollment. All patients received intranasal mometasone furoate (Nazoster®; Santa Farma) at a total daily dose of 200 μg administered once daily. Patients in the morning group applied the medication in the morning, while those in the evening group applied it in the evening, using 2 sprays per nostril.
Clinical evaluations were performed at baseline and 15 days after initiation of treatment. To minimize assessment bias, diagnosis and treatment allocation were conducted by 1 physician, whereas questionnaire administration was performed by a second physician who was blinded to treatment timing.
Skin Prick Test
Skin prick testing was performed in all patients using standardized aeroallergen extracts commonly encountered in Turkey (grass mix, cereals mix, weed mix, tree mix, house dust mite, cat fur, and dog epithelia; Laboratoires Stallergènes), as previously described. 6 Patients with positive reactions to seasonal aeroallergens were included, whereas those sensitized to perennial allergens were excluded.
Nasal Symptom Assessment
Nasal symptoms (rhinorrhea, nasal itching, nasal congestion, and sneezing) were evaluated using a 4-point Likert scale ranging from 0 (no symptoms) to 3 (severe symptoms). The Total Nasal Symptom Score (TNSS) was calculated as the sum of individual symptom scores, yielding a maximum possible score of 12.
Rhinitis Quality of Life Scale
Rhinitis-related quality of life was assessed using the Rhinitis Quality of Life Scale (RQoLS), which consists of 28 items covering daily activities, nasal and ocular symptoms, sleep disturbances, and social and emotional domains. 14 Each item is scored from 0 (no problem) to 3 (always a problem), with higher total scores indicating poorer quality of life.
Pittsburgh Sleep Quality Index
Sleep quality was evaluated using the Pittsburgh Sleep Quality Index (PSQI), which assesses sleep quality over the preceding month. 15 The questionnaire consists of 19 self-rated items generating 7 component scores. The global PSQI score ranges from 0 to 21, with higher scores indicating poorer sleep quality.
Epworth Sleepiness Scale
Daytime sleepiness was assessed using the Epworth Sleepiness Scale (ESS), which evaluates the likelihood of dozing in 8 common daily situations. 16 Each item is scored from 0 to 3, resulting in a total score ranging from 0 to 24, with higher scores reflecting greater daytime sleepiness.
Statistical Analysis
Statistical analyses were performed using SPSS software version 22 (SPSS Inc). Descriptive statistics were expressed as mean ± standard deviation. Baseline-adjusted comparisons between treatment groups were conducted using analysis of covariance (ANCOVA), with posttreatment scores as dependent variables, treatment group as the fixed factor, and corresponding baseline values as covariates. The assumption of homogeneity of regression slopes was assessed by including interaction terms between treatment group and baseline values. Effect sizes were reported as partial eta squared (η2P). A P-value <.05 was considered statistically significant.
Baseline values were included as covariates to account for interindividual differences in disease severity and to distinguish the effect of dosing time from baseline-related variability in treatment response.
Graphical representations of outcome measures were generated using R statistical software (R Foundation for Statistical Computing) to visualize changes from baseline across treatment groups.
Results
Participant Characteristics and Descriptive Statistics
A total of 120 patients were included in the study, with 60 patients assigned to the morning treatment group and 60 patients to the evening treatment group. The mean age was 30.95 ± 9.87 years in the morning group and 31.68 ± 10.30 years in the evening group, with no significant difference between groups. The study population consisted of 74 women and 46 men. Specific allergen sensitization profiles were shown in Table 1, with baseline demographic and clinical characteristics of the patients.
Baseline Characteristics of the Study Population.
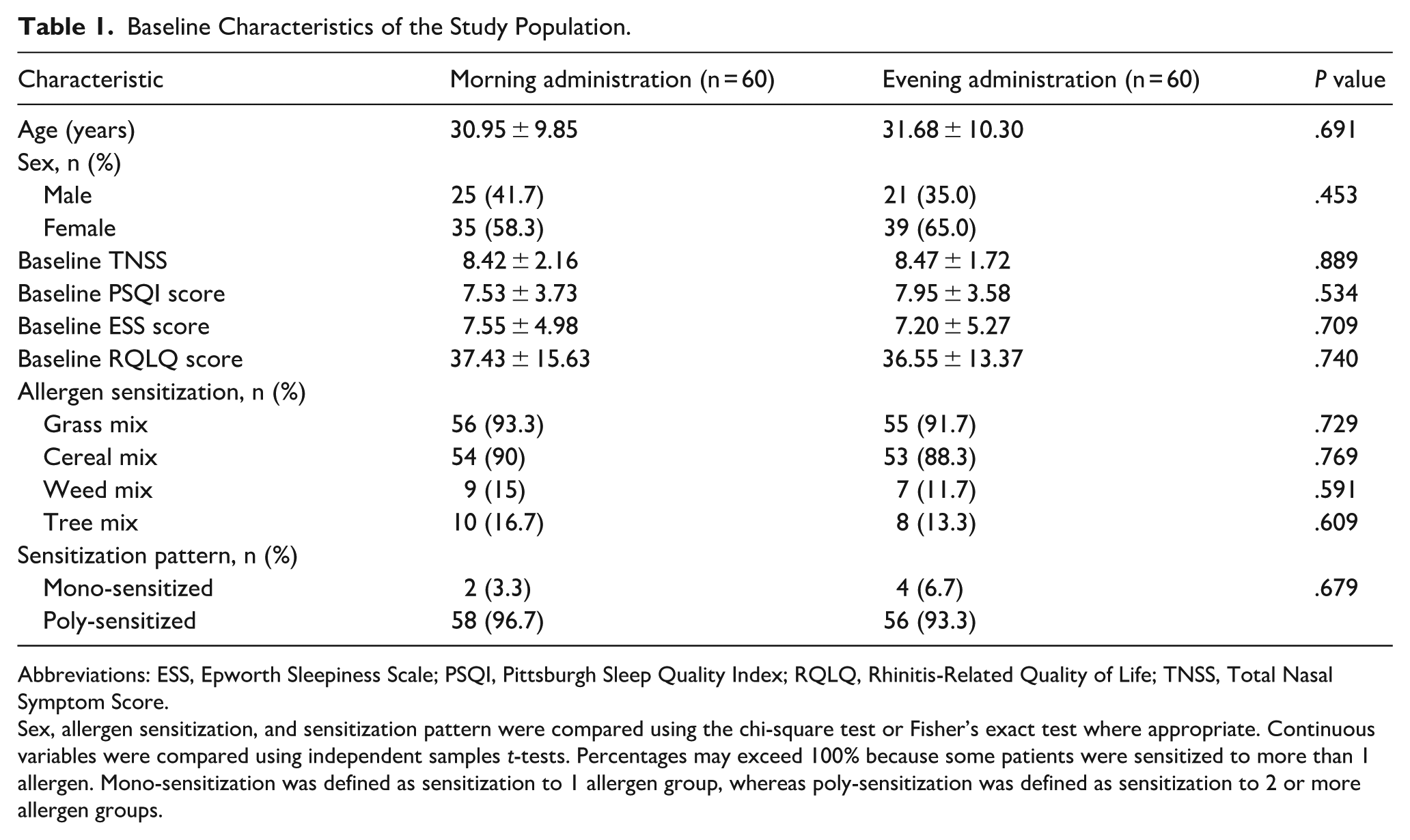
Abbreviations: ESS, Epworth Sleepiness Scale; PSQI, Pittsburgh Sleep Quality Index; RQLQ, Rhinitis-Related Quality of Life; TNSS, Total Nasal Symptom Score.
Sex, allergen sensitization, and sensitization pattern were compared using the chi-square test or Fisher’s exact test where appropriate. Continuous variables were compared using independent samples t-tests. Percentages may exceed 100% because some patients were sensitized to more than 1 allergen. Mono-sensitization was defined as sensitization to 1 allergen group, whereas poly-sensitization was defined as sensitization to 2 or more allergen groups.
Overview of ANCOVA Analyses
Baseline-adjusted ANCOVA were performed for all outcome measures to evaluate the effect of treatment timing (morning vs evening) on posttreatment outcomes while controlling for baseline values. For all analyses, no significant group × baseline interactions were observed, indicating that the assumption of homogeneity of regression slopes was satisfied. Therefore, the main effects of treatment timing were interpreted.
Pittsburgh Sleep Quality Index
After adjustment for baseline PSQI scores, treatment timing had no significant effect on posttreatment PSQI outcomes. Baseline PSQI was a significant predictor of posttreatment PSQI scores (P < .001, η2P = .432), whereas the main effect of treatment timing was not statistically significant. Baseline-adjusted mean posttreatment PSQI scores were 5.24 (95% CI: 4.87-5.62) in the morning group and 5.15 (95% CI: 4.77-5.53) in the evening group.
Daytime Sleepiness (ESS)
ANCOVA demonstrated that baseline ESS scores were strongly associated with posttreatment ESS outcomes (P < .001, η2P = .719). However, no significant difference was observed between the morning and evening treatment groups after baseline adjustment. Baseline-adjusted mean posttreatment ESS scores were 5.44 (95% CI: 4.95-5.94) in the morning group and 5.42 (95% CI: 4.92-5.91) in the evening group.
Rhinitis Quality of Life Scale
Baseline RQoLS scores significantly predicted posttreatment RQoLS outcomes (P < .001, η2P .316). After controlling for baseline values, treatment timing did not significantly influence post-treatment RQoLS scores. Baseline-adjusted mean posttreatment RQoLS scores were 19.80 (95% CI: 17.45-22.14) in the morning group and 19.14 (95% CI: 16.80-21.49) in the evening group.
Total Nasal Symptom Score
For TNSS, neither baseline-adjusted group differences nor baseline TNSS showed a statistically significant association with posttreatment TNSS outcomes. Treatment timing did not significantly affect posttreatment nasal symptom severity. Baseline-adjusted mean posttreatment TNSS were 4.27 (95% CI: 3.75-4.79) in the morning group and 4.51 (95% CI: 3.99-5.03) in the evening group.
Summary of Findings
Across all subjective outcome measures, including PSQI, ESS, RQoLS, and TNSS, treatment timing (morning vs evening) did not result in statistically significant differences after adjustment for baseline values. Posttreatment outcomes were significantly associated with baseline scores, whereas treatment timing showed no significant effect. Detailed ANCOVA results for all outcomes are presented in Table 2, and graphical representations of baseline and posttreatment changes are shown in Figure 2.
Baseline-Adjusted Comparison of Post-Treatment Outcomes Between Morning and Evening Dosing Groups (ANCOVA Results).
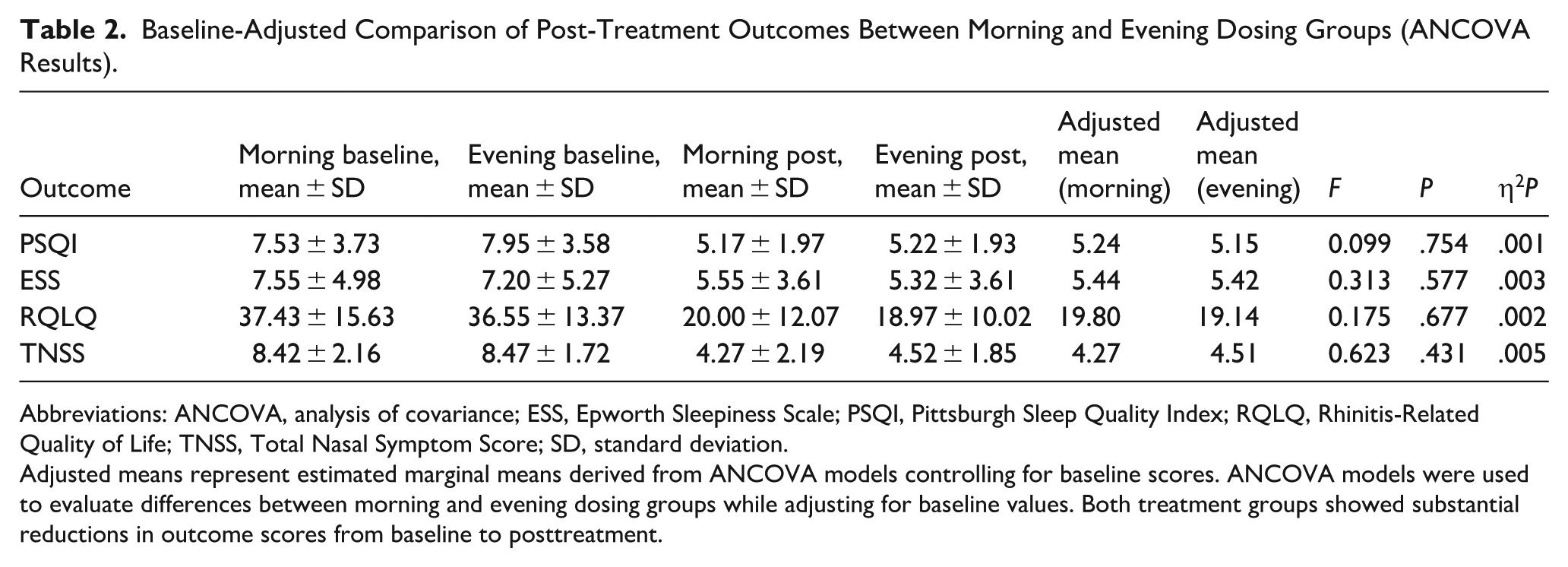
Abbreviations: ANCOVA, analysis of covariance; ESS, Epworth Sleepiness Scale; PSQI, Pittsburgh Sleep Quality Index; RQLQ, Rhinitis-Related Quality of Life; TNSS, Total Nasal Symptom Score; SD, standard deviation.
Adjusted means represent estimated marginal means derived from ANCOVA models controlling for baseline scores. ANCOVA models were used to evaluate differences between morning and evening dosing groups while adjusting for baseline values. Both treatment groups showed substantial reductions in outcome scores from baseline to posttreatment.
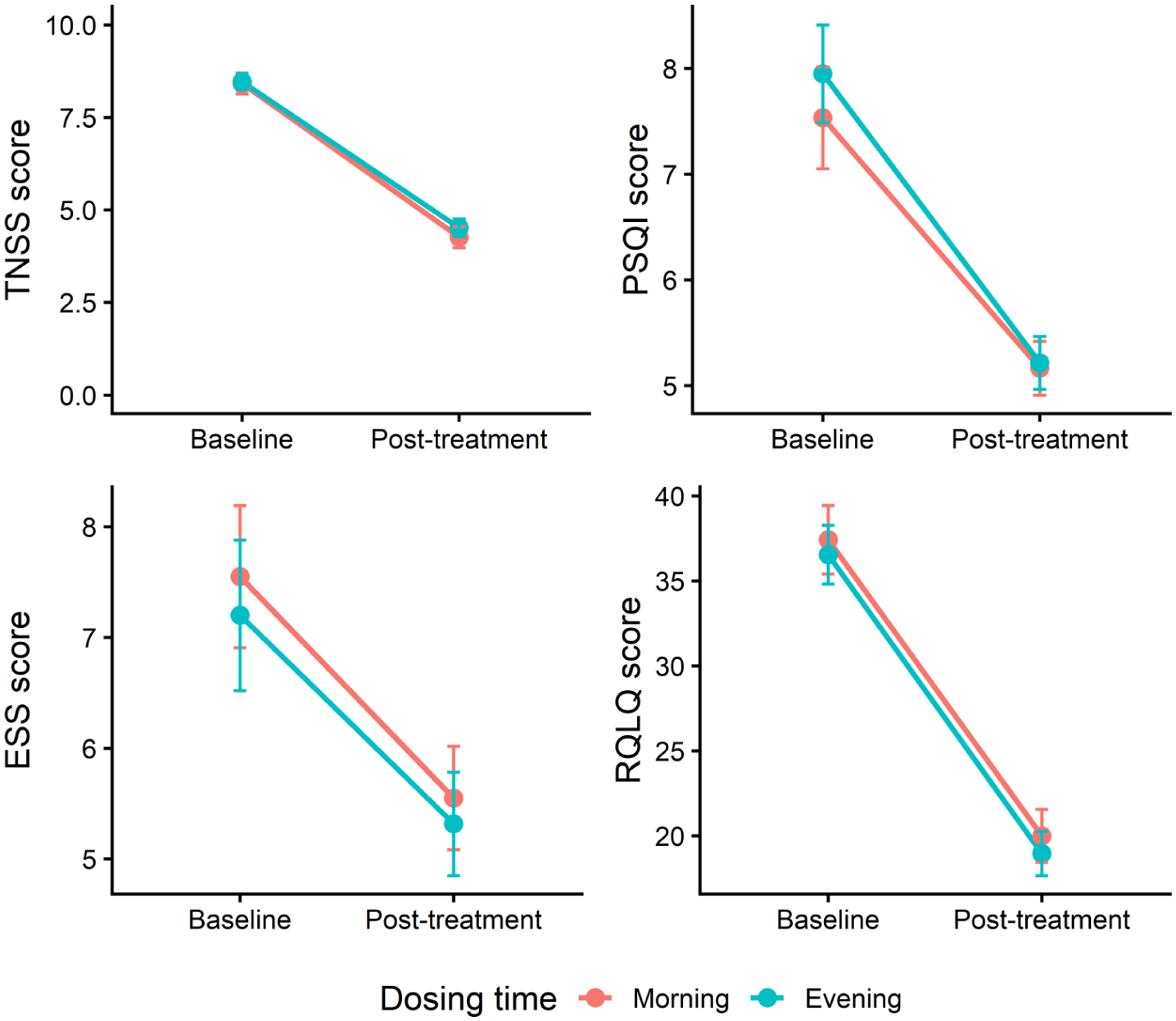
Changes in nasal symptom severity (TNSS), sleep quality (PSQI), daytime sleepiness (ESS), and rhinitis-related quality of life (RQLQ) from baseline to posttreatment in the morning and evening dosing groups. Values are presented as mean ± standard error. PSQI, Pittsburgh Sleep Quality Index; TNSS, Total Nasal Symptom Score.
Discussion
It is known that allergic rhinitis has a pattern of symptoms that change throughout the day. Sleep disturbances associated with allergic rhinitis are commonly reported, and nasal symptoms are often worse in the early morning; however, the mechanisms driving this temporal variation are not fully elucidated. 6 Several factors have been suggested to influence the chronobiology of allergic rhinitis. These factors include nasal congestion related to sleeping position, nocturnal exposure to indoor allergens such as mites, molds, or animal dander, as well as reduced cortisol secretion at night. Elevated vagal tone during the night may further contribute by promoting vasodilation and increased nasal secretions. 9 Together, these physiological and inflammatory processes offer a plausible explanation for the time-dependent variation in allergic rhinitis symptom patterns. In addition, the inflammatory response of the respiratory mucosal epithelium to allergen exposure involves early and late phases, during which differences in the predominant symptom profile of allergic rhinitis have been observed. 7 In the present study, however, baseline disease severity consistently emerged as the main determinant of clinical improvement, while the timing of once-daily intranasal corticosteroid administration did not appear to modify treatment response.
Given the well-documented diurnal variation in allergic rhinitis symptoms, the timing of medication administration has been proposed as a potential factor influencing treatment outcomes. 6 In the present study, mometasone furoate has been found to have a significant effect on nasal symptoms (nasal congestion, rhinorrhea, nasal itching, and sneezing). Treatment was well tolerated, and no significant adverse effects were observed in patients due to intranasal mometasone furoate administration. The study did not show any statistically significant difference between morning and evening administration of intranasal mometasone furoate in patients with moderate-to-severe SAR in terms of nasal symptoms (nasal congestion, rhinorrhea, nasal itching, sneezing), sleep disturbances, daytime sleepiness, and quality of life.
To date, no studies have directly compared morning and evening once-daily administration of intranasal corticosteroids; however, chronotherapeutic effects have been explored in allergic rhinitis using oral H1 receptor antagonists. One such study evaluated morning versus evening dosing of mequitazine, a long-acting antihistamine with a reported half-life of approximately 45.5 hours. In that study, improved control of allergic rhinitis symptoms was observed when two-thirds (7.5 mg) or the full dose (10 mg) was administered in the evening. 8 In contrast, Haye et al compared the efficacy of morning and evening administration of once-daily oral desloratadine (5 mg), another H1 receptor antagonist, and found no significant differences between dosing times with respect to nasal and ocular symptoms, although both groups showed clinical improvement. Desloratadine, which has a reported half-life of approximately 27 hours, was effective regardless of dosing time. 12
Chronotherapeutic effects related to the timing of allergen immunotherapy administration have also been investigated. Li et al evaluated the chronotherapeutic impact of subcutaneous immunotherapy in patients with allergic rhinitis and reported that afternoon administration was associated with significantly greater improvement in rhinorrhea and nasal congestion compared with morning administration. 17 In another study, evening administration of sublingual immunotherapy was associated with better asthma control outcomes than morning administration in pediatric patients with allergic asthma. 18 In contrast, in an ovalbumin-induced allergic rhinitis model, mice that received sublingual immunotherapy during the morning (resting phase) demonstrated a greater reduction in total and ovalbumin-specific IgE levels compared with those treated in the evening (active phase). 19 Importantly, mice are nocturnal animals, and therefore, these findings do not contradict the results observed in human studies of sublingual immunotherapy.
Beyond allergic rhinitis, chronotherapeutic effects of corticosteroid administration have also been explored in bronchial asthma, another chronic respiratory disease in which corticosteroids constitute the cornerstone of treatment. Studies comparing morning and evening once-daily administration of inhaled corticosteroids in asthma have yielded inconsistent findings. While some investigations, such as the study by Zetterström et al, reported no meaningful differences between morning and evening dosing of once-daily mometasone furoate with respect to daytime symptom scores, 20 others have suggested a potential advantage of evening administration for selected pulmonary function parameters. For example, Noonan et al observed greater improvements in lung function following evening dosing of once-daily mometasone furoate compared with morning administration. 21 Similarly, studies evaluating fluticasone furoate and ciclesonide have demonstrated modest timing-related differences in certain physiological measures, such as peak expiratory flow, while showing no consistent differences in asthma symptoms or adverse effects.22-24 More recently, mid-afternoon once-daily dosing of inhaled beclomethasone dipropionate was associated with more favorable lung function and inflammatory biomarker profiles compared with morning or twice-daily regimens. 25 Taken together, these findings indicate that while regular corticosteroid use remains the primary determinant of clinical efficacy, the impact of dosing time may vary depending on the pharmacological properties of the agent and the outcome measures evaluated.
Previous studies demonstrating time-dependent effects have largely involved treatments such as oral H1 antihistamines or allergen immunotherapy, whose efficacy more closely follows circadian fluctuations in mediator release and immune activity.8,12,17,19 To our knowledge, there are no prior studies directly evaluating chronotherapeutic differences in intranasal corticosteroid use in allergic rhinitis, limiting direct comparison with the existing literature. However, evidence from bronchial asthma indicates that findings regarding inhaled corticosteroids remain inconsistent.20-25 This discrepancy suggests that the observed lack of difference in our study may be related to the pharmacological properties of intranasal corticosteroids within a chronobiological framework. Long-acting intranasal corticosteroids exert sustained anti-inflammatory effects through genomic mechanisms, providing continuous suppression over the 24-hour cycle and thereby attenuating potential timing-related differences. 26 Although diurnal variation in allergic rhinitis can serve as a basis for chronotherapy, the possibility of finding a difference between morning and evening administration may decrease for long-acting, once-daily intranasal corticosteroids. Also, the relatively short duration of follow-up and reliance on patient-reported outcomes may have limited the ability to detect subtle circadian-related differences. Since some studies in asthma have reported timing-dependent effects for inhaled corticosteroids,21,24,25 further research is needed to better elucidate the role of chronotherapy in the use of intranasal corticosteroids in allergic rhinitis.
Overall, the clinical efficacy of once-daily intranasal mometasone furoate appeared to be comparable regardless of dosing time. In this context, interindividual differences at treatment initiation may be more clinically relevant than dosing time in shaping short-term treatment outcomes. This finding allows for greater flexibility in clinical practice, with dosing time tailored to patient preference.
A potential limitation of the present study is its relatively short duration. While the follow-up period was sufficient to assess nasal symptom control, as the maximal therapeutic effect of intranasal corticosteroids is typically achieved within 1 to 2 weeks, it may have been insufficient to fully evaluate changes in sleep quality. In this regard, patients completed the PSQI based on the preceding week rather than the past month. Nevertheless, a reduction in PSQI scores was observed in both treatment groups. Another limitation is that TNSS assessments were performed at baseline and at 15 days after treatment initiation, rather than on a daily basis. This approach was chosen to evaluate potential differences between groups at the time point when intranasal mometasone furoate was expected to reach its maximal therapeutic effect. Also, data on disease duration, and previous treatments were not systematically recorded and therefore were not included in the baseline comparison.
Conclusion
In patients with moderate-to-severe SAR, once-daily intranasal mometasone furoate provided significant improvement in nasal symptoms, sleep quality, daytime sleepiness, and rhinitis-related quality of life, regardless of whether it was administered in the morning or in the evening. After adjustment for baseline disease severity, no clinically meaningful differences were observed between dosing times across any of the evaluated outcome measures.
These findings suggest that, despite the well-recognized diurnal variation of allergic rhinitis symptoms and the theoretical rationale for chronotherapy, treatment timing may have limited clinical relevance for long-acting, once-daily intranasal corticosteroids such as mometasone furoate. From a practical perspective, allowing flexibility in dosing time may help simplify treatment regimens and support patient adherence without compromising therapeutic efficacy.
Longer-term studies incorporating objective sleep measures and different intranasal corticosteroid formulations may help to better define whether certain patient subgroups derive benefit from time-dependent treatment approaches. Based on the present findings, however, once-daily intranasal mometasone furoate appears to offer comparable efficacy irrespective of dosing time, allowing treatment to be scheduled according to patient preference.
Footnotes
Acknowledgements
The authors thank Dr Ceren Karaçaylı for statistical consultation and analysis. Dr Karaçaylı has advanced training in biostatistics and research methodology.
Ethical Considerations
The study protocol was approved by the institutional Ethics Committee on Human Research (Decision No: 19/258)
Consent to Participate
Written informed consent was obtained from all participants prior to enrollment.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data are not publicly available due to privacy or ethical restrictions.
