Abstract
Objective:
This study aimed to evaluate the relationship between interleukin (IL)-38, IL-41, and allergic rhinitis (AR).
Methods:
Forty outpatients with AR and 40 healthy controls (HC) were recruited. Data such as the sex and age of the participants were collected. IgE levels were detected and 15 allergens were tested in patients with AR. Blood was collected from the participants and serum IL-38, IL-41, IL-17 and IgE levels were measured using an enzyme-linked immunosorbent assay.
Results:
Serum IL-38 and IL-41 levels in patients with AR were significantly lower than in HC (both P < .05), and serum levels of IL-17 and IgE in patients with AR were significantly higher than in HC (P < .0001 and P < .05). The levels of IL-38 and IL-41 levels in AR patients were significantly negatively correlated with IL-17 levels (r = −0.4813, P = .0017 and r = −0.4385, P = .0047, respectively); however, there was no significant correlation with IgE levels (r = −0.02976, P = .8554 and r = −0.1688, P = .2977, respectively). According to the analysis of the receiver operating characteristic curve, the area under the curve (AUC) for IL-38 was 0.6544 (P = .0175), and the AUC for IL-38 and IL-41 combined with IL-17 were 0.7919 (P < .0001) and 0.7585 (P < .0001), respectively.
Conclusions:
The decreased serum levels of IL-38 and IL-41 in outpatients with AR suggest that IL-38 and IL-41 may serve as potential indicators for the diagnosis of AR.
Introduction
Allergic rhinitis (AR) is an inflammatory disease that occurs in the nasal mucosa; it is mediated by IgE and affects other related cells after exposure to allergens. The main clinical manifestations of AR are nasal itching, runny nose, paroxysmal sneezing, and nasal congestion, accompanied by itching, rash, and other symptoms. 1 The number of patients with global AR is estimated to be more than 500 million, accounting for 10% to 20% of the world population. 2
Interleukin (IL)-36 cytokines include IL-36α, IL-36β, IL-36γ, IL-36 receptor antagonists (IL-36Ra), and IL-38. IL-36Ra and IL-38 play an anti-inflammatory role by competitively binding IL-36R to block the inflammatory signaling pathway activated by IL-36α, β, and γ through IL-36R.3,4 IL-41 is a secreted protein with a variety of functions and is widely expressed at different levels in various tissues, such as nerve tissue, adipose tissue, skin mucosal tissue, and activated macrophages. 5 IL-41 can inhibit the expression of a variety of inflammatory markers, such as IL-6, tumor necrosis factor-α and monocyte chemoattractant protein-1. 6 IL-41 can promote the expression of genes related to thermogenesis and anti-inflammatory cytokines in white adipose tissue and increase the production of IL-4 and IL-13 in adipose tissue. 7
IL-38 and IL-41 play an important role in various immune-related inflammatory diseases. However, their roles in AR occurrence and development are unclear. This study aimed to analyze changes in serum levels of IL-38 and IL-41 in patients with AR and to preliminarily explore whether IL-38 and IL-41 are related to the incidence of AR.
Materials and Methods
Subjects
Forty AR outpatients who visited the Department of Head and Neck Surgery of Affiliated First Hospital of Ningbo University from April to June 2022 were recruited, and 40 healthy volunteers without AR were selected as controls. Patients with AR were selected case by case on in order as they presented according to guidelines for the diagnosis and treatment of AR (Tianjin, 2015). The recruited AR patients were from a broader geographical zone covering Ningbo City/Haishu District, including 26 cases (65%) from urban areas and 14 cases (35%) from suburban/rural areas. The AR patients had a typical history and clinical manifestations of allergy and were either positive for the prick test or positive for both the prick test and IgE. There was no history of respiratory tract infection or related medication within 7 days and no history of antihistamines, hormones, leukotriene receptor antagonists, immunotherapy, or other medications within 30 days in patients with AR. Patients with AR had no history of autoimmune diseases (such as rheumatoid arthritis or colitis) or allergic diseases (such as allergic asthma, allergic dermatitis, or allergic purpura). All patients provided their informed consent in writing. This study was approved by the Ethics Committee of the First Affiliated Hospital of Ningbo University.
Skin Prick Test
The AR patients were tested for allergens using drops (S20060010, Zhejiang Wolwo Bio-Pharmaceutical Co., Ltd., Deqing, China). Skin prick test (SPT) was performed by trained allergologists following the standard procedures recommended by the International Academy of Allergy and Immunology. Standardized commercial allergen extracts were purchased from Stallergenes Greer (Lenoir, NC, USA), including:
Outdoor allergens: Grass pollen, birch pollen, ragweed pollen (dominant pollen types in the study region during spring and early summer).
Indoor allergens: Dermatophagoides pteronyssinus, Dermatophagoides farinae, cat dander, dog dander.
A positive SPT result was defined as a wheal diameter ≥3 mm larger than the negative control (saline solution). The sensitization type of each patient was classified based on SPT results: exclusive outdoor sensitization, exclusive indoor sensitization, and combined indoor-outdoor sensitization. A standard allergen reagent was used to puncture the skin on the volar side of the forearm and the results were observed 20 minutes later. Positive and negative controls were used for each test. Histamine was used as a positive control, and allergen solvent was used as a negative control. The results were determined according to the specifications of the standardized allergen reagent.
The Level of IgE
Blood (1 mL) was taken from patients with AR and healthy controls (HC). After centrifugation, serum was collected for IgE detection. IgE levels were determined using an IgE kit (Jingyuan Medical Instrument Co., Ltd., Shanghai, China).
Enzyme-Linked Immunosorbent Assay
Blood (3 mL) was collected from patients with AR and a healthy control population. After centrifugation, serum was collected and stored at ‒80°C until detection. The Enzyme-linked immunosorbent assay kit was purchased from Cusabio Technology, LLC (Wuhan, China). Serum samples were incubated at room temperature for 20 minutes. Reactions were prepared in each well with 50 μL diluted standard, serum sample, and biotin-labeled antibody added in sequence, according to the manufacturer’s instructions. The reaction holes were covered with a membrane, oscillated, mixed, and incubated at 37°C for 60 minutes. The reaction wells were washed 4 times and then mixed with horseradish peroxidase (HRP)-labeled Streptavidin 80 μL, and incubated at 37°C for 30 minutes. After washing 4 times, 50 μL substrate A and B were added to the reaction wells, and then incubated at 37°C for 10 minutes under dark conditions. The absorbance (OD) value of the sample at 450 nm was obtained by microplate meter after adding a 50 μL stop solution and the concentrations of IL-38, IL-41, and IL-17 in the 2 groups were calculated. Each sample was tested in duplicate to ensure the accuracy of the results.
Statistical Analysis
Statistical analyses and graph creation were performed using GraphPad Prism 9.0 (GraphPad Software Inc., San Diego, CA, USA). Measurement data are expressed as mean ± SD. The count data are expressed as percentages. The independent samples t test was used for comparisons between 2 groups. Comparisons between count data were performed using the Chi-square test. Pearson’s correlation analysis was performed to assess the correlation between IL-38, IL-41, IL-17, and IgE levels. P < .05 was considered statistically significant.
Results
Baseline Information
The AR group included 25 men and 15 women, with an average age of 26.23 ± 2.63 years. The HC group consisted of 23 males and 17 females, with an average age of 29.70 ± 1.87 years. There were no significant differences in sex or age between the 2 groups (P > 0.05). Among the 40 patients, 28 (70%) had exclusive outdoor sensitization (pollen), 8 (20%) had combined indoor-outdoor sensitization (pollen plus dust mites/pet dander), and 4 (10%) had exclusive indoor sensitization. This distribution is consistent with the peak pollen season (April–June) in the study region, while also covering patients with indoor sensitization, which is in line with the real-world clinical features of AR. We were unable to obtain more general and biochemical data because the patients were admitted to the outpatient department.
Expression of Serum IL-38, IL-41, IL-17, and IgE in Patients with AR
The serum levels of IL-38 and IL-41 in the patients with AR were 39.94 ± 5.71 pg/mL and 456.10 ± 40.82 pg/mL, respectively, which were significantly lower than those of HC (70.89 ± 12.38 pg/mL and 717.10 ± 110.80 pg/mL) (P < .05, Figure 1A and B). The serum level of IL-17 in the AR group was 23.65 ± 6.99 pg/mL, which was significantly higher than in the HC group (17.22 ± 6.25 pg/mL) (P < .0001, Figure 1C). The serum IgE level in the AR group was 126.10 ± 24.57 pg/mL, which was significantly higher than that in the HC group (43.89 ± 7.34 pg/mL) (P < .05, Figure 1D).
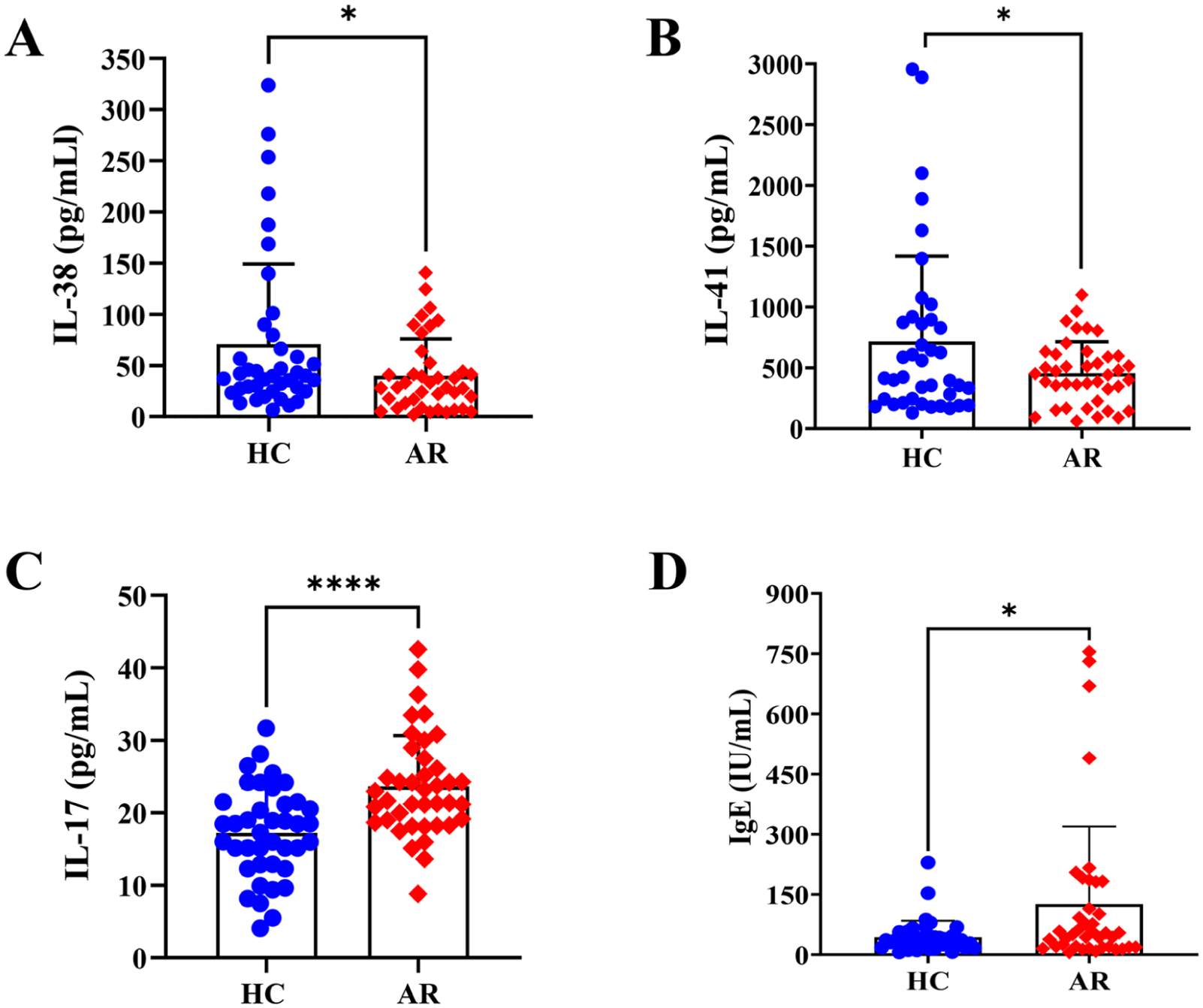
Expression of IL-38 (A), IL-41 (B), IL-17 (C), and IgE (D) in serum. AR, Allergic rhinitis; HC, Healthy controls; IL, Interleukin.
Correlation Between IL-38, IL-41, IL-17, and IgE in Patients with AR
The Spearman correlation test showed that IL-38 and IL-41 levels were negatively correlated with IL-17 levels in the serum of AR patients (Figure 2A and B, P < .05). Furthermore, serum levels of IL-38 and IL-41 were negatively correlated with peripheral blood IgE levels in patients with AR; however, the difference was not statistically significant (Figure 2C and D).
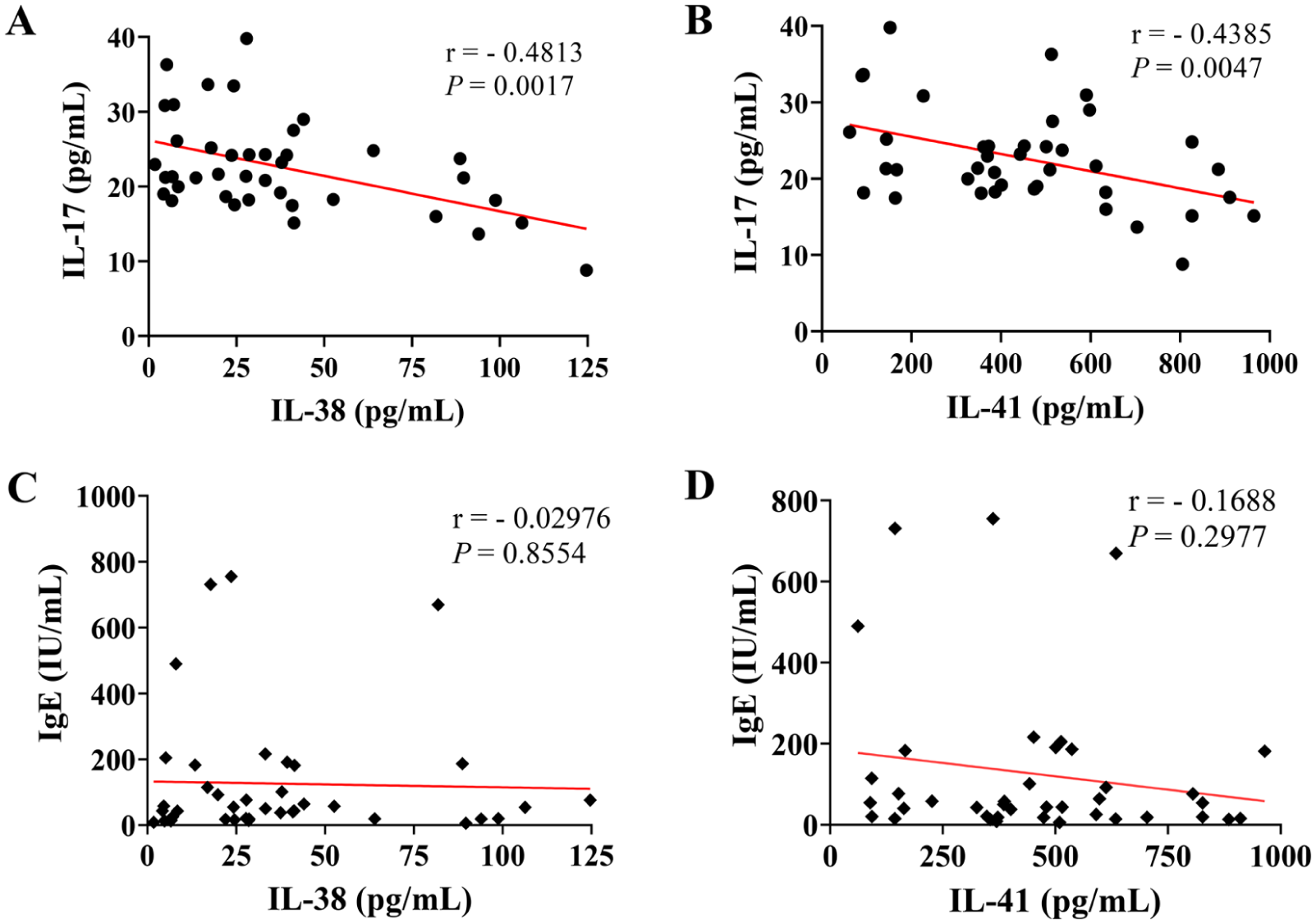
Correlation between IL-38 and IL-17 (A), IL-41 and, IL-17 (B), IL-38 and IgE (C), IL-41 and IgE (D) serum levels in AR patients. AR, Allergic rhinitis; IL, Interleukin.
The Analysis of the Receiver Operating Characteristic Curves for the Diagnosis of AR
The diagnostic values of IL-38 and IL-41 for AR were assessed using receiver operating characteristic (ROC) curves (Figure 3). The area under the curve (AUC) for the diagnosis of AR with IL-38 was 0.6544 (P = .0175, Figure 3A). The AUCs for IL-38 and IL-41 combined with IL-17 were 0.7919 (P < .0001, Figure 3B) and 0.7585 (P < .0001, Figure 3C), respectively.
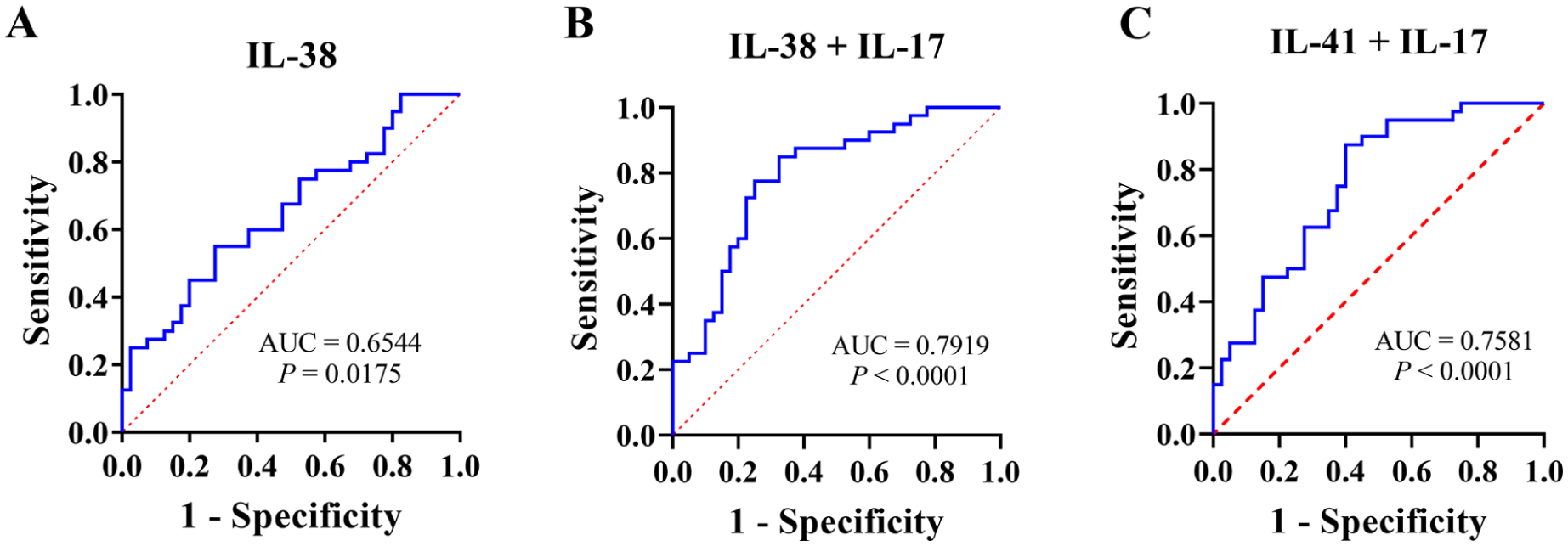
Receiver operating characteristic curves for the diagnosis of allergic rhinitis (AR). (A) IL-38 was used to diagnose AR. (B) IL-38 combined with IL-17 were used to diagnose AR. (C) IL-41 combined with IL-17 were used to diagnose AR. IL, Interleukin, AUC, Area under the curve.
Discussion
In the present study, we found that serum levels of IL-38 and IL-41 decreased and IL-17 increased in patients with AR, and that serum levels of IL-38 and IL-41 were negatively correlated with IL-17 in patients with AR. Receiver operating characteristic curve showed that IL-38 or IL-38 and IL-41 combined with IL-17 had high diagnostic significance for AR.
AR pathogenesis is a complex process that involves many factors, including environment, heredity, and immunity. Recent studies have confirmed that an imbalance between Th cells and their secreted cytokines plays a key role in the occurrence and development of AR. 8 Studies have found that abnormal activation of Th17 cells is a key factor in AR. 9 Our study also found a high expression of the Th17 effector cytokine IL-17 in peripheral blood from AR patients. This indicates that there is an imbalance in Th17 cells in patients with AR and that the immune environment lacks inhibitory factors in Th17 cells.
In recent years, there has been an increasing number of studies on the anti-inflammatory effects of IL-38. IL-38 inhibits the secretion of IL-17 in human peripheral blood mononuclear cells and the expression of IL-17 in THP-1 cells. 10 IL-38 overexpression in macrophages inhibits IL-17 secretion in CD3+ and CD4+ T cells. 11 As IL-17 is a Th17 effector, inhibition of IL-38 by IL-17 may be the key to the anti-inflammatory effect of Th17. Our study found a decrease in the expression of IL-38 in the serum of AR patients, which can weaken the anti-inflammatory effect, thus disrupting the balance between the anti-inflammatory effect of IL-38 and the proinflammatory effect of Th17 cells, leading to a subsequent inflammatory response and the appearance of AR. We also found that the expression of IL-38 in the peripheral blood of AR patients was negatively correlated with the expression of the Th17-related factor IL-17, proving that IL-38 plays an inhibitory role in the Th17 effect in a normal body environment, and its negative effect on IL-17 may be the key to its anti-inflammatory effect.
IL-41 is a novel immunoregulatory cytokine. Jung et al 12 reported that IL-41 can reduce lipid-induced inflammation and insulin resistance, and induce fatty acid oxidation through signals from adenosine monophosphate-activated protein kinase or peroxisome proliferator-activated receptor (PPAR)δ signals in skeletal muscle. Zuo et al 13 found that IL-41 promotes the function and differentiation of adipocytes in mesenteric adipose tissue by partially activating signal transducer and activator of transcription 5/PPARγ signaling pathway, thus alleviating mesenteric injury and improving Crohn’s disease such as colitis in mice. Ushach et al 14 observed abnormal cytokine production, altered IgG production, and high sensitivity to lipopolysaccharide in the sepsis mouse model with IL-41 knockout. Serum levels of IL-41 were elevated during chronic obstructive pulmonary disease exacerbations and smoking, which may be important for balancing inflammatory responses. 15 Our study found that serum IL-41 levels were significantly decreased in outpatients with AR, suggesting that IL-41 may also be involved in the development of inflammation in AR. In this study, the subjects were patients with mild disease admitted to the outpatient department. Therefore, although IL-41 levels were reduced, there was no correlation with IgE levels in these patients. Subsequently, we collected serum and nasal mucosa samples from patients with severe AR to explore the expression of IL-41 in the systemic and local nasal mucosa.
Furthermore, our findings regarding the reduced serum levels of IL-38 and IL-41 in AR patients may have implications for the characterization of specific rhinitis subtypes, particularly local allergic rhinitis (LAR). LAR represents a distinct clinical entity characterized by local nasal allergic responses without systemic atopy, posing significant diagnostic challenges that require enhanced cooperation between allergology and otorhinolaryngology specialties. 16 We hypothesize that IL-38 and IL-41 could serve as potential serum biomarkers to differentiate LAR from classical AR and non-allergic rhinitis, given their distinct immunological profiles. Specifically, the differential expression patterns of these cytokines might reflect the absence of systemic sensitization observed in LAR patients. However, it is important to note that our current study did not include an LAR subgroup, limiting our ability to validate this hypothesis. Future investigations specifically comparing cytokine profiles across classical AR, LAR, and non-allergic rhinitis cohorts are warranted to establish the diagnostic utility of IL-38 and IL-41 in this context.
Several limitations of this study should be acknowledged. First, the single-center, case-control design with a modest sample size (40 patients per group) may limit the generalizability of our findings and the statistical power to detect subtle subgroup differences. Second, our cohort did not include patients with LAR or pediatric populations, restricting the applicability of our findings to these clinically important subgroups. Third, while we measured IL-17 as a Th17 marker, we did not assess key Th2 cytokines such as IL-4 and IL-13, which would have provided more comprehensive insight into the type 2 inflammatory status of our patients. Finally, we relied solely on serum measurements without concurrent analysis of local nasal biomarkers (eg, nasal lavage fluid or tissue biopsies), which may better reflect the mucosal immunopathology of AR.
In conclusion, our data confirmed that serum levels of IL-38 and IL-41 were decreased in patients with allergic rhinitis, while IL-17 levels increased; Furthermore, serum levels of IL-38 and IL-41 were negatively correlated with IL-17 levels. ROC curve analysis showed that IL-38 or IL-38 and IL-41 combined with IL-17 had a clinical significance for AR. Although changes in the concentrations of IL-38 and IL-41 cannot be used as diagnostic indicators for AR, these interleukins can serve as important biological indicators reflecting the immune pathological status of AR, and help clarify the regulatory roles of different immune cells in the disease process. In the future, further studies with larger sample sizes and wider cytokine combinations (including Th2 cytokines IL-4/IL-13 and correlation analysis) are needed to elucidate the interaction between IL-38/IL-41 and the classical Th2 axis in AR pathophysiology.
Footnotes
Ethical Considerations
The study protocol followed the ethical guidelines of the Declaration of Helsinki of 1975 and was approved by the Ethics Committee and Institutional Review Boards of The First Affiliated Hospital of Ningbo University. Informed consents were signed by all patients.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Medical Health Science and Technology Project of Zhejiang Province (2024KY1542 and 2025KY1358), Key Project of Ningbo Natural Science Foundation(2024J040).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting the findings of this study can be obtained from the corresponding author upon reasonable request.
