Abstract
Background:
Laryngeal clefts are rare congenital aerodigestive tract anomalies that cause chronic aspiration, feeding difficulties, and recurrent respiratory infections in children. Types I to III clefts remain challenging because of limited exposure and technical constraints. Transoral robotic surgery (TORS) has emerged as a potential minimally invasive alternative, offering enhanced visualization and instrument articulation. However, evidence regarding its feasibility, safety, and clinical effectiveness in pediatric laryngeal cleft repair remains limited and fragmented. This systematic review synthesizes available evidence on robotic repair of Types I to III laryngeal clefts.
Materials:
A systematic search of PubMed, Scopus, Embase, ScienceDirect, the Cochrane Library, CINAHL, and Google Scholar was conducted in accordance with PRISMA 2020 guidelines. We performed study selection, data extraction, and quality appraisal using the Joanna Briggs Institute tools. Due to heterogeneity and the absence of comparator groups, a narrative synthesis was undertaken.
Results:
Four studies comprising 29 attempted robotic repairs were included, of which 26 procedures were successfully completed robotically. Robotic repair was applied to Type I (n = 19), Type II (n = 7), and Type III (n = 3) clefts using transoral robotic platforms, predominantly the da Vinci system. Early feasibility studies reported conversion due to exposure limitations, whereas later studies demonstrated 0% conversion rates. Postoperative improvement in swallowing or aspiration was reported in ~70% to 82% of Type I, 75% of Type II, and 67% of Type III clefts. Complications were infrequent, mild, and transient, with no procedure-related mortality or need for tracheostomy.
Conclusion:
Current evidence suggests that TORS is a feasible and safe adjunct for selected pediatric patients with Types I to III laryngeal clefts, particularly Type II and carefully selected Type III lesions. While routine use in uncomplicated Type I clefts offers limited advantage over standard endoscopic repair, robotic assistance may provide meaningful benefits in anatomically complex cases.
Introduction
Laryngeal clefts are rare, hereditary defects that are characterized by abnormal relationships between the larynx, trachea, and esophagus. 1 Incidences have been estimated to be between 1 in 10 000 and 1 in 20 000 live births; however, the actual prevalence is likely underestimated due to nonspecific occurrences such as aspiration, recurring pneumonia, and difficulties in feeding that delay diagnosis. 2 The results vary significantly according to the Benjamin-Inglis classification. 3 Type I clefts are in the interarytenoid region and are associated with mild symptoms, which tend to respond well to conservative management or simple endoscopic repair. Type IV clefts are able to invade the thoracic trachea, where they are at high risk of severe airway obstruction and require extensive open surgical repair, with high perioperative morbidity. 4
These clefts present heterogeneous clinical and surgical challenges across Types I, II, and III. The midline clefts run through the vocal folds into the cricoid or cervical trachea, often resulting in persistent aspiration, non-thriving, infections of the lower respiratory tract, and chronic lung damage, unless well treated. 5 Standard endoscopic approaches, which can be applied to Type I lesions, have technical drawbacks with Types II and III lesions due to limited exposure, difficult depths, multilayer repair complexities, and the need to use advanced suturing techniques. Conversely, open methods that can be applied in Type IV clefts could lead to unwarranted morbidity in Types II and III.6,7 Although Type I clefts are generally treated conservatively or with standard endoscopic repair, their inclusion is crucial for understanding the full clinical spectrum, comparing management pathways across cleft severity, and identifying whether robotic techniques have any reported or potential role in selected cases.
These restrictions have led to the growing interest in robotic surgery that has a 3-dimensional visualization, enhanced magnification, tremor filtration, and articulated instruments to perform precise suturing in tight spaces. Thus, robotic repair has evolved as an intermediate option, which is suitable for Types I to III laryngeal clefts, in which the difficulty of the surgical intervention is beyond what takes place with a standard endoscopy but does not warrant open surgery.
Despite the theoretical advantages, empirical evidence on the issue of robotic pediatric airway surgery is scanty. The existing literature is only made up of single case reports and small institutional series. The only systematic review of robotic airway surgery found only 2 suitable studies and did not differentiate the outcomes according to the type of laryngeal cleft, and, therefore, their cohort is the one that is most likely to benefit from robotic repair. The lack of subtype-specific evidence represents a significant gap in clinical knowledge, which leads to the lack of holistic, evidence-based recommendations that surgeons may need in relation to robotic surgeries in Types I to III clefts.
The proposed systematic review is aimed at summarizing, synthesizing, and framing available data regarding robotic repair of Types I to III laryngeal clefts. It will help fill a significant gap by examining feasibility, perioperative results, complication and functional improvements. This synthesis is important in guiding clinical decision-making, standardizing clinical practices, and informing future studies on airway surgery in pediatrics.
Materials and Methods
The study selection process was conducted in accordance with the Preferred Reporting Items for Systematic Review and Meta-analysis (PRISMA) 2020 guidelines and is summarized in the PRISMA flow diagram (Figure 1). 8 A total of 267 records were identified through database searching, including PubMed (n = 11), Cochrane Library (n = 6), ScienceDirect (n = 54), Scopus (n = 60), Embase (n = 13), CINAHL (n = 3), and Google Scholar (n = 120 (Supplementary File 1). Ultimately, 4 studies met all inclusion criteria and were included in the final qualitative synthesis. Eligible studies included children aged 0 to 18 years who underwent robotic laryngeal cleft repair using transoral robotic surgery (TORS) or equivalent robotic platforms. Case reports, case series, feasibility studies, and cohort studies were included, while editorials, reviews, animal studies, and studies limited to Type IV clefts were excluded. A protocol was developed in advance and registered in the International Prospective Register of Systematic Reviews (PROSPERO) under the registration number CRD420251271116 prior to manuscript submission.
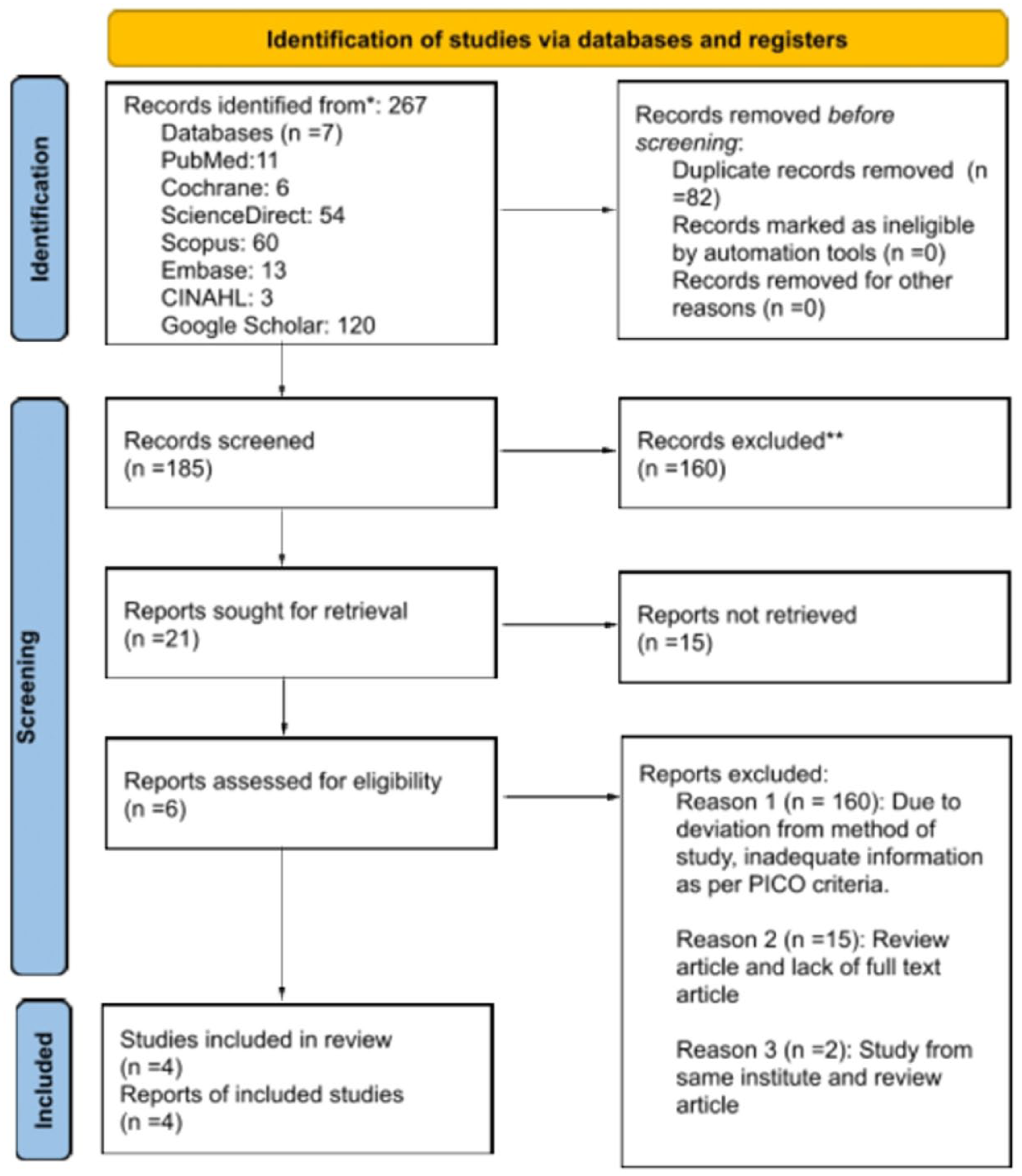
PRISMA flow diagram.
A comprehensive literature search was performed in PubMed, Scopus, Science Direct, Embase, the Cochrane Library, and Google Scholar from November 2025. The search string included: (“Robotic Surgical Procedures” [MeSH] OR robotic surgery OR robot-assisted) AND (“laryngeal cleft” OR “laryngotracheal cleft” OR “laryngotracheal anomaly” OR “laryngeal clefts”). Additional records were identified through manual screening of reference lists and gray literature. Two independent reviewers screened titles, abstracts, and full texts.
Data extraction was carried out using a standardized form that captured study characteristics, demographic details, clinical presentation, robotic platforms and instruments used, operative metrics, postoperative recovery, complications, conversion rates, and swallowing or aspiration outcomes. Extraction was performed independently by 2 reviewers to ensure accuracy, with discrepancies resolved through discussion. Methodological quality and risk of bias were assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Checklists appropriate for each study design, evaluating domains such as selection methods, exposure ascertainment, outcome measurement, and follow-up completeness. 9
Given the heterogeneity of study designs, sample sizes, cleft type distribution, operative platforms, outcome measures, and due to the lack of a comparator group, a quantitative meta-analysis was not feasible. Instead, a descriptive, narrative synthesis was performed. Findings were organized around key outcomes, including the feasibility of robotic exposure, operative success, perioperative course, complication profiles, and functional improvements in swallowing and aspiration. Patterns were compared across Types I to III laryngeal clefts to identify where robotic surgery provides the most meaningful clinical benefit and to highlight remaining gaps requiring further research.
Results
Study Characteristics
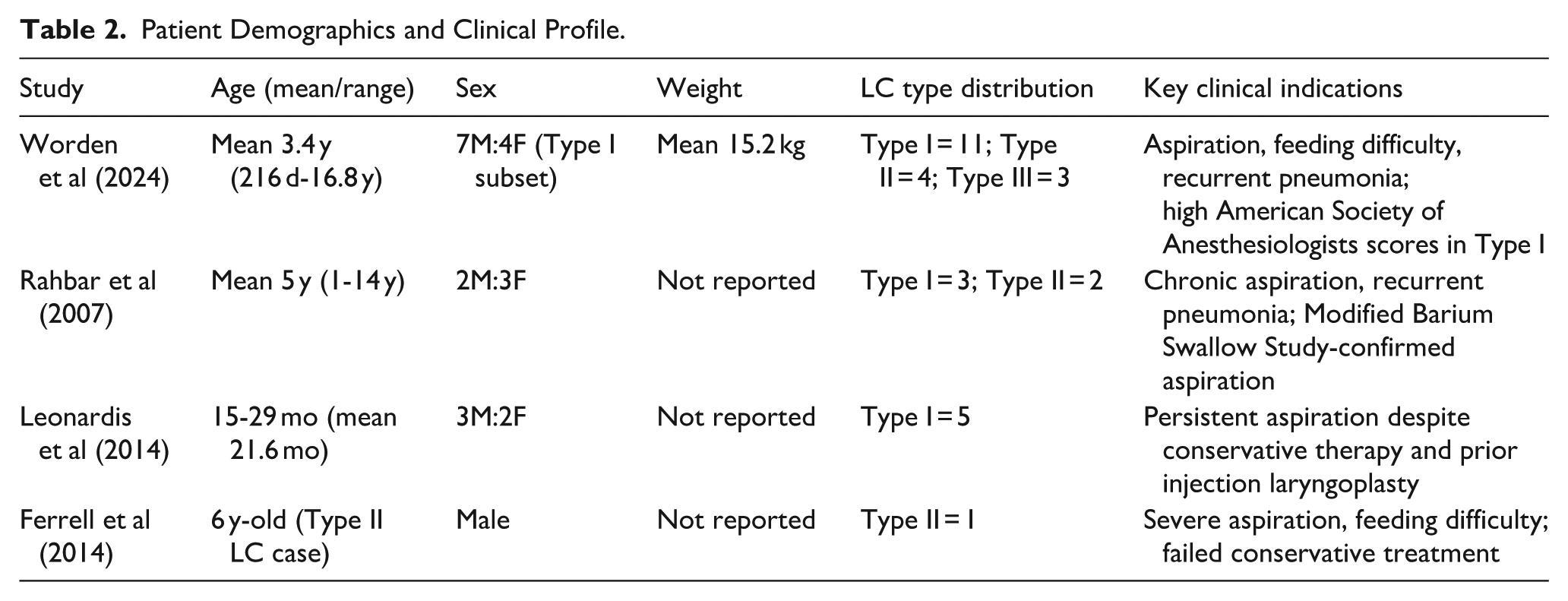
Four studies describing robotic repair of laryngeal clefts have been included, comprising a total of 29 attempted robotic surgeries, with 26 successfully executed robotically.10 -13 The data includes 1 retrospective cohort study, 1 feasibility and safety study, and 2 case series. All of these were done in tertiary pediatric airway facilities in the United States between 2007 and 2021 (Table 1). Robotic intervention was utilized for Benjamin-Inglis Type I (n = 19), Type II (n = 7), and Type III (n = 3) laryngeal clefts, excluding Type IV clefts. Only 1 study included a comparative group (conventional endoscopic repair), whereas the other trials assessed robotic repair as a singular intervention. The main reasons for surgery across studies were chronic aspiration, feeding difficulties, and recurrent lower respiratory tract infections despite conservative management (Table 2).
Study Characteristics of Included Robotic LC.
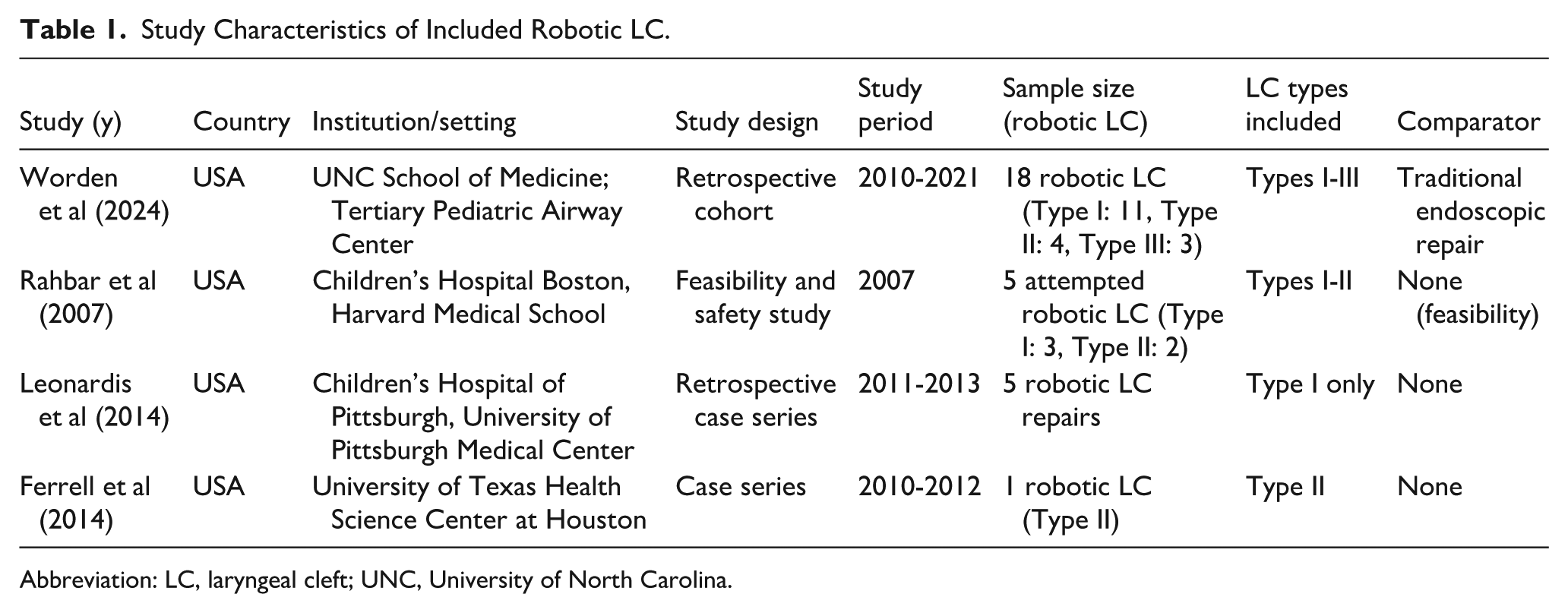
Abbreviation: LC, laryngeal cleft; UNC, University of North Carolina.
Patient Demographics and Clinical Profile.
Operative Characteristics and Feasibility
All of the procedures used TORS platforms, mostly the da Vinci robotic system. To get to the area, pediatric airway retractors such as FK, Lindholm, Crowe-Davis, and Dingman retractors were used, along with 3-dimensional endoscopic visualization and articulated 5 mm robotic instruments (Table 3). Over time, it became easier to do the surgery. In the initial feasibility study, 3 out of 5 attempted cases (60%) necessitated a transition to non-robotic methods due to insufficient laryngeal exposure and the constraints of early-generation robotic systems; however, all subsequent studies indicated a 0% conversion rate, signifying improvements in robotic instrumentation and surgical technique (Table 3). The duration of the operation consistently increased as the severity of the cleft increased. In the largest group, the average time for Type I was 149 minutes, for Type II was 168 minutes, and for Type III clefts, it was even longer. This was longer than the average time for traditional endoscopic repair of Type I lesions (111 minutes), but there was no increase in perioperative morbidity (Table 3).
Surgical Outcomes, Robotic Platforms, and Complications.
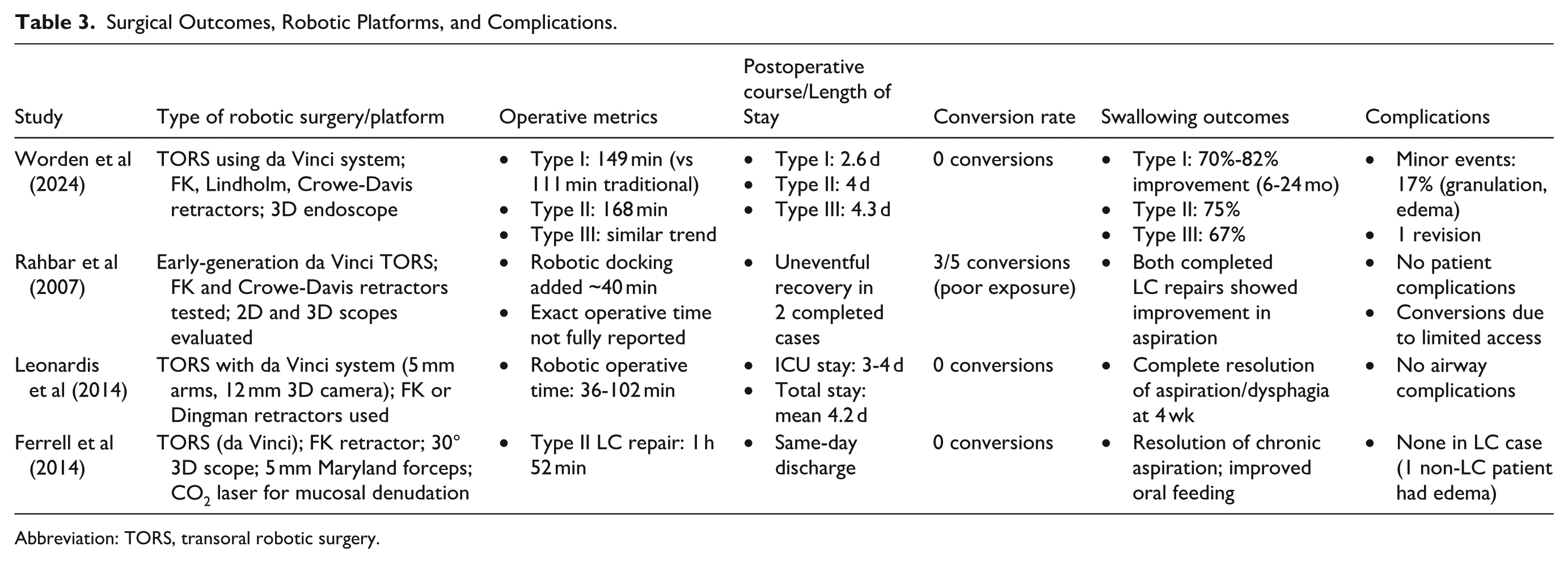
Abbreviation: TORS, transoral robotic surgery.
Postoperative Course
Postoperative recovery was generally favorable and correlated with cleft severity. The average length of stay in the hospital went up with the cleft grade: 2.6 days for Type I, 4.0 days for Type II, and 4.3 days for Type III clefts in the comparable group (Table 3). Reports of ICU admissions varied, and it seemed that institutional practice, not postoperative instability, was the main reason for them. For example, some patients were discharged the same day after an isolated Type II robotic repair. No study reported prolonged postoperative ventilation, airway obstruction requiring tracheostomy, or procedure-related mortality associated with the robotic approach.
Functional Outcomes
Functional outcomes exhibited clinically significant enhancement subsequent to robotic repair. In the biggest cohort with longitudinal follow-up, improvement in swallowing or aspiration was observed in 70% to 82% of Type I, 75% of Type II, and 67% of Type III clefts throughout follow-up periods ranging from 6 to 24 months (Table 3). Smaller case series indicated total cure of aspiration and dysphagia, frequently within 4 weeks postoperatively, including patients who had previously unsuccessful conservative treatments or injection laryngoplasty.
Complications
The rates of complications were minimal, with only around 17% of patients having mild postoperative events. These events were mostly temporary laryngeal edema or granulation tissue formation that was treated conservatively. In all investigations, there was only 1 revision surgery (Table 3).
Methodological Quality of Included Studies
Using the JBI techniques to assess methodological quality showed that the overall quality was moderate. The recent studies were more rigorous, but the main problems were small sample sizes, retrospective designs, and a lack of randomized comparisons (Table 4).
JBI Critical Appraisal Summary for Included Studies.
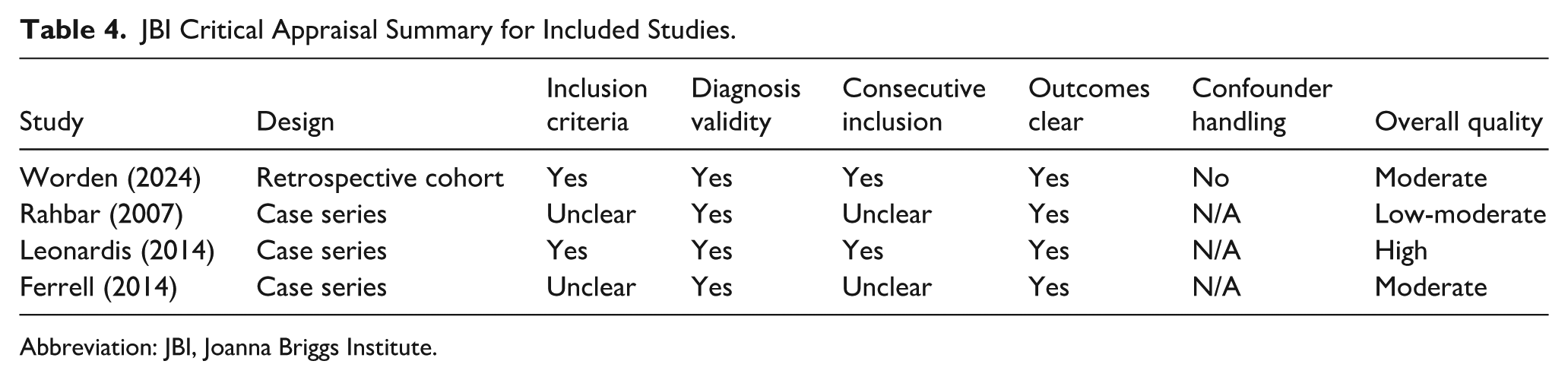
Abbreviation: JBI, Joanna Briggs Institute.
Discussion
Laryngotracheoesophageal clefts are a rare continuum of airway malformations that have still been one of the most difficult cases in childhood otolaryngology. 14 The technical difficulty of their surgery is due to the deep midline defect, a limited endolaryngeal working space, and incessant exposure to aerodigestive secretions, as well as the necessity to close the defect with a multilayer repair and spare the adjacent neurovascular and phonatory structures. 5 The Benjamin-Inglis classification reflects not only increasing anatomic severity but also escalating surgical difficulty, particularly for Types II and III clefts, where standard endoscopic techniques may be limited by exposure, depth, and suture control. 15 Except for Type IV clefts, which typically require open reconstruction, Types I to III clefts are theoretically amenable to endoscopic approaches; however, the constrained operative corridor and demand for meticulous suturing provide a compelling rationale for robotic assistance. 14 The present systematic review synthesizes available evidence on robotic repair of Types I to III clefts and demonstrates that robotic surgery has been selectively applied across this spectrum, with feasibility and outcomes closely linked to both cleft severity and technological evolution.
Pediatric airway surgery robotic surgery uses began to be implemented in the mid-2000s, with the wider adoption of the da Vinci system in adult surgery. Robotic laryngeal cleft repairs were first described in Rahbar et al, 10 and it was the earliest effort to apply the benefits of robots, including 3-dimensional visualization, wristed instrumentation, and tremor filtration to pediatric airway surgery. 11 Although it was initially enthusiastically adopted, widespread adoption was not achieved, especially because of the constraints of first-generation systems, such as the massive size of instruments, inability to reach into small airways, long docking time, and uneven exposure. 16 Such limitations are manifested in the initial feasibility investigations with a large fraction of conversion and technical complexity. 10 Over time, however, refinements in robotic platforms, improved optics, and growing surgeon experience have reshaped feasibility, as evidenced by the absence of conversions and improved outcomes in more recent series. 17
The studies included all TORS via da Vinci platforms, with a developmental shift of the initial systems to advanced configuration. The technological improvements, such as better endo-wrist articulation, better 3-dimensional imaging, and compatibility with pediatric retractors, seem to have a determining effect on enhancing operative feasibility. 16 Previous research by Rahbar et al reported an increase in docking time and exposure failures, whereas subsequent cohorts by Worden et al exhibited steady exposure, stable operations, and zero rates of conversion.10,11 These improvements in feasibility and safety likely reflect a combination of factors, including increasing surgical experience, better patient selection, and gradual refinement of robotic techniques, rather than technological evolution alone. 18 Given the relatively small airway dimensions in pediatric patients and the limited differences in instrument size across robotic platforms, the learning curve and case selection likely played a substantial role in the improved outcomes reported in later series.
When looked at by cleft type, robotic repair showed different levels of value. In Type I laryngeal clefts, existing evidence indicates that conventional endoscopic techniques, particularly endoscopic division and repair, achieve high rates of symptom improvement and aspiration resolution, with pooled improvement rates exceeding 80% in contemporary meta-analyses. 19 Within this context, robotic repair does not appear to confer a uniform advantage over standard endoscopic approaches in uncomplicated Type I lesions and is associated with longer operative durations and increased resource utilization. However, comparative data suggest that robotic repair may offer superior postoperative swallowing outcomes in selected Type I cases, particularly those with recurrent clefts, failed prior endoscopic repair, or anatomically complex interarytenoid defects, despite longer surgical times.6,19 Importantly, available studies report no increase in major complications with robotic repair, indicating that the approach is safe when performed in experienced centers, although its incremental benefit over standard endoscopic repair in routine Type I clefts remains uncertain and should be weighed against cost and operative efficiency.
On the other hand, Type II clefts seem to be a case where robotic surgery may have clearer benefits. These lesions penetrate further into the cricoid area and necessitate meticulous dissection and secure multilayer closure. 4 In the studies that were included, robotic repair of Type II clefts was linked to high rates of improvement in aspiration, controlled handling of tissue, and no major complications. The articulated robotic instruments probably made it easier to do traction, countertraction, and precise suturing in tight spaces, which may have lowered the risk of mucosal trauma and suture failure.11,20 However, the early learning curve effects and longer surgery times show how important it is to choose the right cases and have the right level of expertise in the hospital. 2 From a practical standpoint, Type II clefts may represent the subgroup in which robotic assistance offers the greatest incremental value. Type I lesions are often managed successfully with standard endoscopic techniques, while Types III and IV clefts may require highly specialized or open approaches. Type II clefts occupy an intermediate position, where the defect extends into the cricoid region and demands precise multilayer suturing within a confined operative field. 12 In this context, the improved visualization and instrument articulation provided by robotic platforms may offer meaningful advantages over conventional endoscopic instruments, 14 particularly in anatomically challenging cases. However, direct comparative studies are still limited, and further research is needed to define the relative benefits of robotic versus standard endoscopic repair in this subgroup.
Type III laryngeal clefts represent a rare and anatomically complex subgroup that has traditionally been managed with open reconstruction or staged endoscopic approaches because of their depth and extension toward the cervical trachea. Over the past 2 decades, endoscopic CO2 laser-assisted techniques have demonstrated favorable outcomes in carefully selected patients, with reported aspiration resolution rates ranging from ~68% to 87% in small retrospective series. 21 While these data support endoscopic management as an effective option, they also highlight the technical challenges associated with exposure, multilayer suturing, and precision closure in Type III defects.4,22 Within this context, robotic-assisted repair has emerged as a novel and evolving strategy, with early reports demonstrating technical feasibility and encouraging functional outcomes in highly selected Type III cases. The enhanced visualization, instrument articulation, and suturing precision offered by robotic platforms suggest a potential role in anatomically favorable Type III clefts. 1 Accordingly, robotic repair should be viewed as an emerging modality under investigation, warranting further evaluation through larger, multicenter studies to better define its efficacy, safety profile, and optimal position within the management algorithm for Type III laryngeal clefts. 23
According to the included studies, complications associated with robotic laryngeal cleft repair were infrequent, predominantly minor, and showed a clear temporal improvement. In our synthesis, exposure-related difficulties, including limited working space, instrument collision, prolonged docking time, and conversion to conventional endoscopic surgery, were observed almost exclusively in early feasibility studies, whereas later cohorts reported zero conversion rates and a substantially reduced complication burden. These complications occurred primarily because early robotic systems were not designed for pediatric airway dimensions, with instrument calibers disproportionately large relative to pediatric laryngeal anatomy, restricted lines of sight within narrow supraglottic spaces, and a steep procedural learning curve, particularly for airway suturing.23,24 Mucosal injury and transient airway edema were the most reported adverse events, resolving with conservative management and without progression to major airway compromise, tracheostomy, or mortality. Importantly, despite longer operative times compared with traditional endoscopic repair, robotic approaches in our review did not demonstrate higher rates of clinically significant complications, indicating that early technical limitations and operator experience, rather than inherent procedural risk, accounted for most reported difficulties. 11
The evidence indicates that robotic surgery is a viable and secure supplementary approach for the repair of specific Types I to III laryngeal clefts, particularly for Type II and certain Type III lesions where traditional endoscopic methods may be technically constrained. Improvements in robotic technology have made it much easier to do and have lowered the number of complications over time. However, present evidence does not endorse the routine replacement of standard endoscopic techniques, especially for uncomplicated Type I clefts. Future multicenter studies employing standardized outcome measures and extended follow-up are crucial to delineate the specific role of robotic surgery in the management algorithm for laryngotracheoesophageal clefts.
It is important to recognize a few limitations. The evidence base consists solely of small observational studies characterized by variability in outcome definitions, follow-up duration, and reporting standards. There was only 1 study with a comparison group, which made it hard to say for sure that the new technique was better than standard endoscopic techniques. Also, robotic surgery was only done in high-volume tertiary centers, which makes it hard to say how well it works in other places. Cost, access to robotic platforms, and the steep learning curve make it even harder to use them more widely.
Conclusion
TORS is a feasible and safe option for repairing Types I to III laryngeal clefts in carefully selected pediatric patients. The technique achieves good functional outcomes, particularly improvement in aspiration and swallowing, with low rates of complications. Its clinical benefit appears greatest in Type II and selected Type III clefts, where conventional endoscopic approaches may be technically limited. For uncomplicated Type I clefts, standard endoscopic management remains sufficient, and routine robotic use does not show a clear additional advantage. Overall, robotic repair should be considered a selective adjunct rather than a replacement for established endoscopic techniques.
Supplemental Material
sj-docx-1-ear-10.1177_01455613261434142 – Supplemental material for Transoral Robotic Surgery for Laryngeal Cleft Types I to III: A Systematic Review and Narrative Synthesis of Techniques, Outcomes, and Safety
Supplemental material, sj-docx-1-ear-10.1177_01455613261434142 for Transoral Robotic Surgery for Laryngeal Cleft Types I to III: A Systematic Review and Narrative Synthesis of Techniques, Outcomes, and Safety by Bigyan Raj Gyawali and Bibek Shrestha in Ear, Nose & Throat Journal
Supplemental Material
sj-docx-2-ear-10.1177_01455613261434142 – Supplemental material for Transoral Robotic Surgery for Laryngeal Cleft Types I to III: A Systematic Review and Narrative Synthesis of Techniques, Outcomes, and Safety
Supplemental material, sj-docx-2-ear-10.1177_01455613261434142 for Transoral Robotic Surgery for Laryngeal Cleft Types I to III: A Systematic Review and Narrative Synthesis of Techniques, Outcomes, and Safety by Bigyan Raj Gyawali and Bibek Shrestha in Ear, Nose & Throat Journal
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data analyzed in this study are derived from published literature and are available within the included articles.
Supplemental Material
Supplemental material for this article is available online.
Research Registry
PROSPERO with unique identifying number: 1271116.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
