Abstract
Distant metastasis occurs in 1% to 4% of patients with papillary thyroid carcinoma (PTC) and most commonly involves the lungs and bones, with skull base involvement being exceedingly rare. We present a case of a 58-year-old male patient who experienced skull-base metastasis of tall-cell variant PTC 4 years after multimodal treatment of aggressive disease. The patient presented to the otolaryngology clinic with left-sided postnasal drip, anosmia, ptosis, and proptosis. Magnetic resonance imaging of head showed a 1.9 × 2.7 × 4.4 cm mass with extensive invasion of the sinonasal cavities, cribriform plate, and left orbit, complicated by a postobstructive mucocele. In-office biopsy was consistent with metastatic PTC. He underwent endoscopic surgical debulking followed by postoperative radiation therapy. This case demonstrates how PTC with aggressive features can develop anterior skull base metastases years after initial treatment. Management required recognition of this diagnostic challenge and combined endoscopic resection and stereotactic radiotherapy, achieving local control while preserving neurologic function. The patient has remained stable at 7-month follow-up.
Introduction
Papillary thyroid carcinoma (PTC) accounts for the majority of thyroid malignancies and carries a favorable prognosis, with 10 year survival rates exceeding 90%. 1 Cervical lymph node metastases occur in up to 22% of PTC patients and have been reported to moderately impact survival. 2 In contrast, distant metastasis is rare, occurring in 1% to 4% of patients, and are most commonly seen in follicular thyroid carcinoma or aggressive PTC variants.1,3 Pulmonary and skeletal metastases are the most common sites of metastatic disease.1,3 The development of distant metastases marks a shift in clinical course, with 10-year survival dropping to 13% to 27%.4 -6
Skeletal metastases occur in an estimated 4% of all thyroid cancer, with calvarial metastases occurring in 2.5% of patients.7,8 Skull base metastases of thyroid cancer are particularly rare, with 1 study showing an incidence of 1 in 412 (0.2%).7,8 To date, only 20 prior cases of skull base metastases from primary PTC tumors have been reported in the literature (Supplemental Table 1).9 -23 Skull base metastases threaten the integrity of vital neurovascular structures, and may cause cranial nerve palsies, endocrine dysfunction, and other severe neurological complications. 24
We present a rare case of anterior skull base metastasis arising 4 years after treatment of tall-cell variant PTC, a histologic subtype associated with increased mortality. 25 This case underscores the difficulty of distinguishing metastatic disease from primary sinonasal pathology and highlights the persistent metastatic risk in aggressive PTC despite years of apparent disease stability.
Case Presentation
A 58-year-old man was referred to a tertiary rhinology clinic for evaluation of new sinonasal symptoms in the setting of a history of T4bN1bMx tall-cell variant PTC of the left thyroid lobe, for which he had previously undergone total thyroidectomy with bilateral neck dissection. Intraoperatively, bulky nodal disease involved the phrenic nerve, carotid artery, and brachial plexus, precluding gross total resection. He subsequently received adjuvant external-beam radiation and radioactive iodine (RAI) therapy. Serial neck imaging demonstrated stable residual disease without progression over the ensuing 4 years.
Four years after initial treatment, the patient developed new sinonasal and orbital symptoms that included left-sided postnasal drip, anosmia, mild ptosis, and proptosis. MRI of the skull base demonstrated a 1.9 × 2.7 × 4.4 cm enhancing mass centered in the anterior skull base, with invasion of the ethmoid and frontal sinuses, extension through the cribriform plate into the anterior cranial fossa, and erosion of the left lamina papyracea (Figure 1). A secondary postobstructive mucocele in the left frontal sinus was identified, causing orbital roof erosion and orbital mass effect.
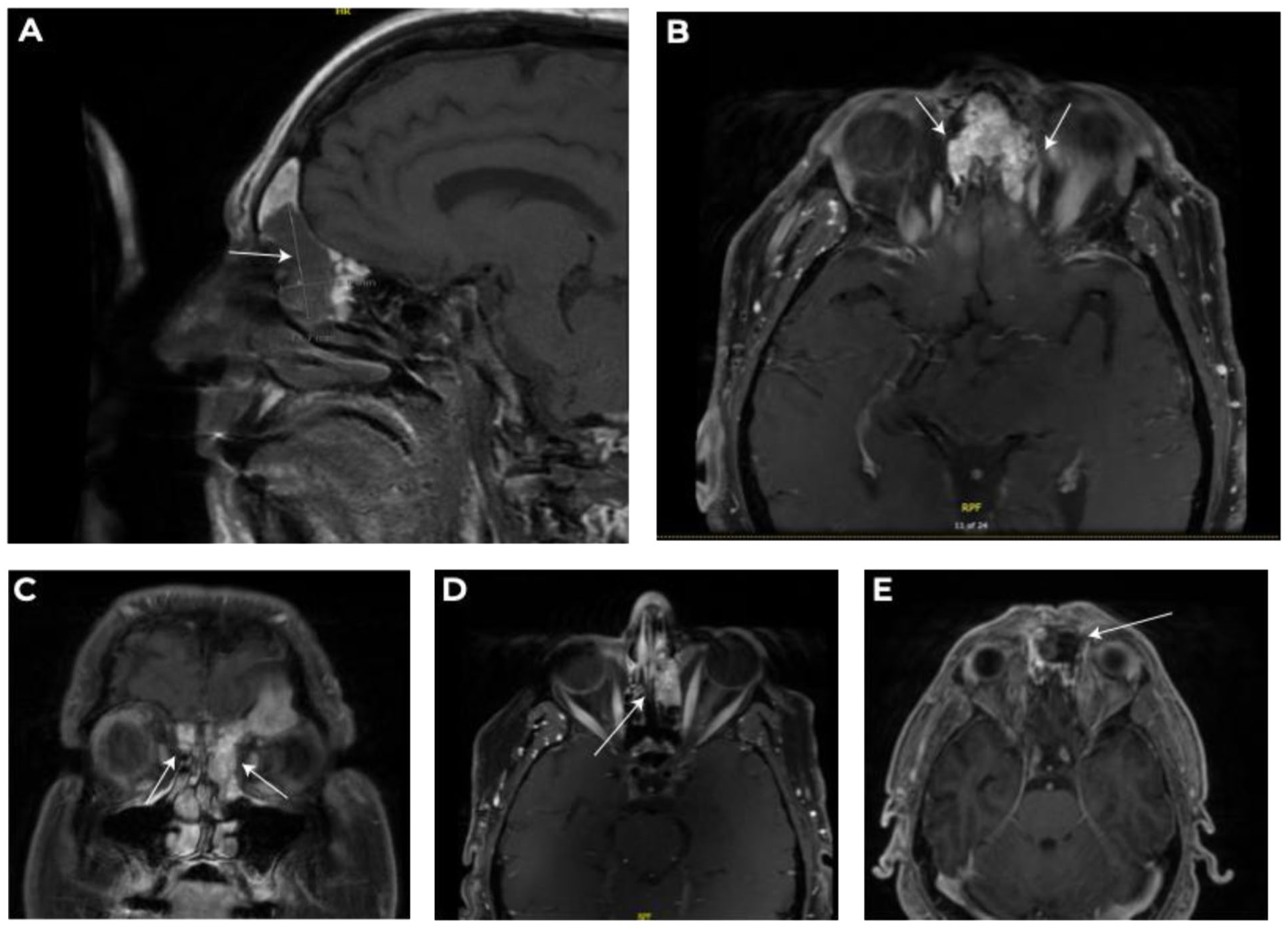
MRI imaging findings. (A) Sagittal T1 postcontrast MRI demonstrating 1.9 × 2.7 × 4.4 cm anterior skull base neoplasm (arrow) involving the frontal and ethmoid sinuses. (B) Axial T1 postcontrast MRI showing bilateral tumor extension with orbital involvement (arrows). (C) Coronal T1 postcontrast MRI demonstrating bilateral tumor extension into the medial orbital spaces (arrows). (D) Axial T1 postcontrast MRI showing extensive ethmoid involvement (arrow). (E) Axial T1 postcontrast MRI 3 weeks after surgical resection showing residual enhancing tissue (arrow).
An in-clinic biopsy demonstrated findings consistent with metastatic tall-cell variant PTC that was BRAF positive and morphologically consistent with the primary tumor. The patient subsequently underwent endoscopic debulking to decompress the orbit, evacuate the mucocele, and reduce tumor burden before adjuvant therapy. He subsequently completed 5 fractionated treatments of adjuvant stereotactic body radiotherapy. He had no new treatment-related toxicities noted on follow-up and was stable at 7 month follow-up, though longer follow-up is needed to determine durability of local control.
Discussion
Skull-base metastasis from PTC is exceedingly rare, with most reports limited to case reports. Among the 20 reported cases, no single histologic variant predominates in skull base metastases, suggesting that variant alone may not predict this specific metastatic pattern though certain variants contribute to overall high-risk biology. Beyond variant, clinical factors such as T4 stage, BRAF V600E mutation, and RAI-refractory disease appear to drive overall metastatic risk.9 -23 Our patient’s T4b PTC with neurovascular invasion precluding complete resection, BRAF V600E mutation, and RAI-refractory biology exemplifies this high-risk phenotype.
The patient’s presenting symptoms, including postnasal drip and anosmia, closely mimicked other sinonasal pathology; however, the presence of orbital symptoms raised concerns for an underlying aggressive process. Prior to tissue biopsy and imaging, the initial differential diagnosis included inflammatory processes (allergic fungal sinusitis with orbital erosion, and obstructive frontal mucocele), benign neoplasm (inverted papilloma, osteoma, fibrous dysplasia), primary sinonasal malignancies (squamous cell carcinoma, adenocarcinoma, esthesioneuroblastoma, and sinonasal undifferentiated carcinoma), skull base tumors extending into the sinuses (meningioma, schwannoma), and metastasis from primary cancer. 24
A review of the 20 reported PTC skull base metastases (Supplemental Table 1) demonstrated that most tumors involved the middle or posterior cranial fossa (sella, clivus, cavernous sinus, and petrous bone). These cases often resulted in neurologic deficits including cranial neuropathies (CNs; cranial nerves II-VII, XI-XII) across 15 cases and pituitary dysfunction (diabetes insipidus or panhypopituitarism) across 5 cases.9 -23 To our knowledge, anterior skull base involvement presenting primarily with sinonasal symptoms has not been previously reported in the literature. Our patient’s presentation with nonspecific sinonasal complaints and absence of CNs exemplifies the diagnostic challenge when PTC metastasizes to the anterior skull base. Together, this report emphasizes the importance of ENT evaluation in high-risk PTC patients, particularly in those with T4 disease, incomplete resection, or RAI-refractory status, who develop new sinonasal symptoms.
This patient’s treatment strategy, combining surgical resection with adjuvant stereotactic radiotherapy, mirrors the multimodal approach employed in the majority of the 20 previously reported cases. A review of the documented skull base metastases from PTC (Supplemental Table 1) showed that 12 (60%) underwent surgical resection, 12 (60%) underwent radiation therapy (intensity-modulated, external beam, or stereotactic radiosurgery), and 7 (35%) received RAI, with most receiving multimodal therapy.9 -23 Outcomes were variable: 6 patients achieved stable disease at last follow-up (range 1-20 months) while 7 died of progressive disease (range 16-61 months).11 -23 The heterogeneity of outcomes and rarity of reported cases limits prognosis. In addition, a combination of systemic disease burden, completeness of local control, and treatment responsiveness likely play a role in survival outcomes.25,26 Our patient’s stable imaging at 7 months demonstrates early treatment response, though longer follow-up is needed to determine durability of local control.
Lenvatinib, a tyrosine kinase inhibitor, has demonstrated benefits in progression-free survival in the setting of RAI-refractory PTC. 27 The SELECT trial, a randomized phase-3 study examining lenvatinib versus placebo in patients with progressive radioiodine-refractory differentiated thyroid cancer, demonstrated improvements in progression-free survival (18.3 vs 3.6 months; HR 0.21). 27 In this patient, systemic therapy was deferred given his excellent response to surgery and radiotherapy with apparent stability of the skull base lesion on imaging and no evidence of additional metastatic disease. The patient remains under active surveillance with systemic therapy reserved as an option for future disease progression at the skull base or development of new metastatic sites.
Several clinical pearls emerged during this patient’s treatment: first, high-risk PTC patients with new sinonasal symptoms warrant careful evaluation regardless of disease stability duration. Second, anterior skull base metastases may present with nonspecific symptoms rather than classic CNs. Third, multimodal therapy combining surgical debulking with adjuvant radiotherapy can achieve local control while preserving neurologic function in complex skull base disease.
Conclusion
This case demonstrates how PTC carries persistent metastatic risk despite years of apparent disease stability, with skull base involvement creating diagnostic challenges by mimicking other potentially benign or malignant sinonasal pathologies. This case yields 3 key clinical implications: (1) aggressive histologic subtypes warrant long-term surveillance given persistent metastatic risk and poor prognosis after metastasis, (2) new sinonasal symptoms in high-risk PTC patients require thorough evaluation for metastatic disease regardless of disease stability, and (3) successful management of complex skull base metastases requires a multimodal approach, and surgery, radiation, and systemic therapy should all be considered to optimize oncologic control while preserving quality of life.
Supplemental Material
sj-docx-1-ear-10.1177_01455613261428524 – Supplemental material for Anterior Skull Base Metastasis From Papillary Thyroid Carcinoma: A Case Report
Supplemental material, sj-docx-1-ear-10.1177_01455613261428524 for Anterior Skull Base Metastasis From Papillary Thyroid Carcinoma: A Case Report by Mishek Thapa, Jakob L. Fischer, Jessa E. Miller and Jivianne T. Lee in Ear, Nose & Throat Journal
Footnotes
Consent to Participate
Written informed consent has been provided.
Author Contributions
M.T.: data collection, literature review, manuscript writing – original draft, manuscript revision, figure preparation, and final approval. J.L.F.: conceptualization, supervision, manuscript review and editing, critical revision for intellectual content, and final approval. J.E.M.: manuscript review and editing, critical revision for intellectual content, and final approval. J.T.L.: supervision, conceptualization, manuscript review and editing, critical revision for intellectual content, and final approval.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
