Abstract
SMARCA4-deficient sinonasal carcinoma is a recently proposed tumor entity that is rare and associated with a poor prognosis. No standardized treatment strategy has been established. We report a case of SMARCA4-deficient sinonasal carcinoma treated at our department. A 47-year-old woman was referred for treatment of a tumor of the left nasal cavity. The biopsy at a previous institution suggested a neuroendocrine tumor, but it was finally diagnosed as olfactory neuroblastoma. Imaging revealed no lymph node or distant metastases. Endoscopic endonasal resection of the malignant sinonasal tumor was performed with resection and reconstruction of the anterior skull base, achieving complete removal of the tumor. Postoperative intensity modulated radiation therapy (IMRT, 60 Gy) was administered. During IMRT, the histopathological examination confirmed the diagnosis of SMARCA4-deficient carcinoma. Adjuvant chemotherapy following the protocol for undifferentiated carcinoma was administered using the FP regimen (5-fluorouracil 800 mg/m2 and cisplatin 80 mg/m2) for 3 cycles. At 2 years and 1 month after surgery, the patient remains alive with no evidence of disease. SMARCA4-deficient carcinoma is an aggressive and poorly differentiated tumor with a poor prognosis, and our experience suggests that a multimodal approach combining surgery, radiation therapy, and chemotherapy following the treatment strategy for undifferentiated carcinoma is effective.
Keywords
Introduction
SMARCA4-deficient carcinoma is a recently recognized tumor characterized by loss of SMARCA4 expression. Only a few cases in the sinonasal region have been reported, and few describe the full clinical course. We present our clinical experience with SMARCA4-deficient sinonasal carcinoma successfully treated by multimodal therapy and discuss its management.
Case
A 47-year-old non-smoking woman with a history of cervical dysplasia and uterine endometriosis presented with anosmia. Initial computed tomography (CT) showed bilateral narrowing of the olfactory cleft without a mass (Figure 1A). Despite therapy, symptoms persisted. A year later, CT before olfactory cleft surgery revealed a new left nasal mass. The biopsy suggested olfactory neuroblastoma, and she was referred for treatment.
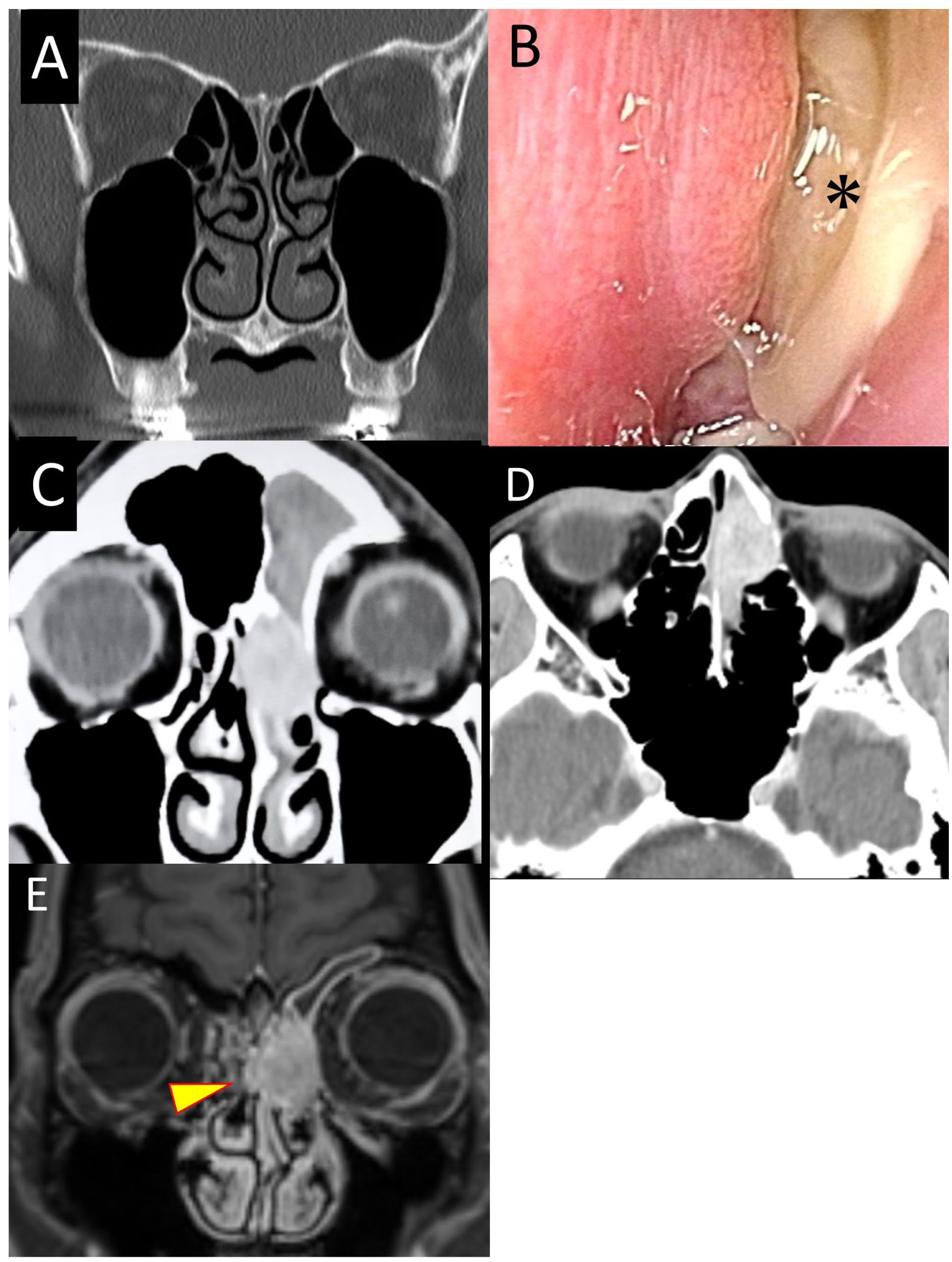
Preoperative endoscopic and imaging findings. (A) CT from 1 year before presentation. Bilateral olfactory cleft narrowing without sinonasal tumor. (B) Left olfactory cleft mass(*). (C, D) Contrast-enhanced CT showing a tumor from the left nasal cavity to the left ethmoid sinus. (E) Contrast-enhanced T1-weighted MRI showing extension across the nasal septum (yellow allowhead). CT, computed tomography; MRI, magnetic resonance imaging.
The endoscopy showed a whitish to pale reddish mass in the left olfactory cleft (Figure 1B). Contrast-enhanced CT showed an enhanced lesion that extended to the left ethmoid sinus, contacting the medial orbital wall and the nasal bone without destruction (Figure 1C, D). Magnetic resonance imaging showed a heterogeneous high-signal lesion in T2 and a heterogeneous enhancement in T1, extending across the nasal septum (Figure 1E). Positron emission tomography-CT did not reveal metastases. The histopathology of the biopsy showed a small round cell tumor positive for CD56, focally for synaptophysin and chromogranin, and calretinin, indicating neuroendocrine differentiation; olfactory neuroblastoma was diagnosed.
Under the diagnosis of olfactory neuroblastoma (Kadish B, Dulguerov T2), endoscopic endonasal resection with resection and reconstruction of the anterior skull base was performed 1 month later, followed by planned postoperative intensity modulated radiation therapy (IMRT).
Under general anesthesia, the tumor extended beyond the nasal septum, invading the right superior turbinate. The margins of the frozen section were negative (Figure 2A and B). Functional endoscopic sinus surgery on the right side secured working space and reduced the risk of postoperative radiation-induced sinusitis after IMRT. The left lamina papyracea was partially resected, preserving the orbital contents (Figure 2C). After detaching the tumor from the orbit, the nasal bone and the frontal process of the maxilla were exposed and removed en bloc using a chisel and burr (Figure 2D). A left medial maxillectomy was added. The skull base was drilled circumferentially between the cecum of the foramen and the anterior sphenoid roof. The dura was incised and both olfactory bulbs and the tracts were transected, allowing removal en bloc including bilateral cribriform plates, olfactory bulbs, tracts, crista galli, left medial orbital wall, and nasal bone (Figure 2E, G). The margins were negative.
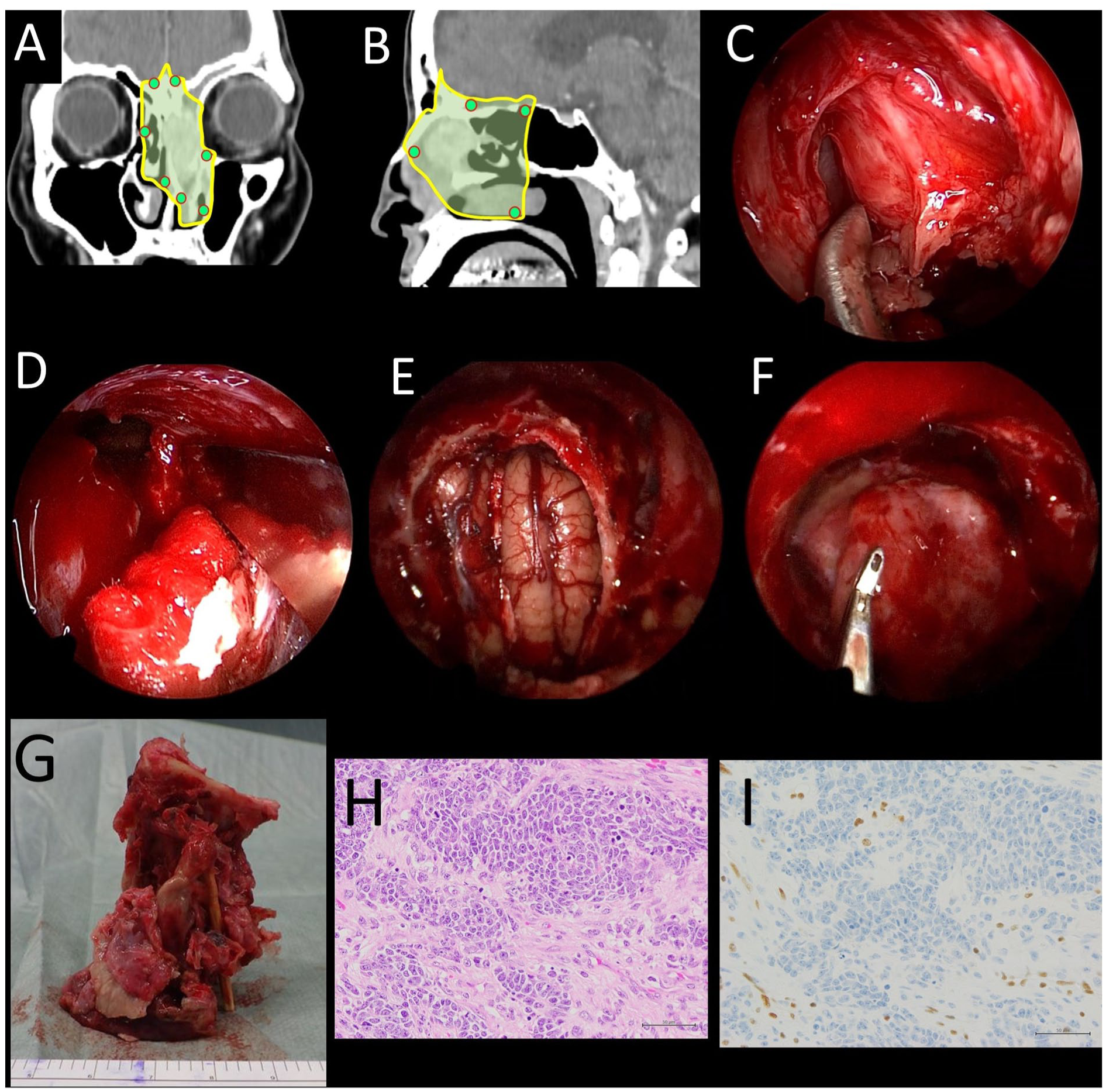
Intraoperative findings and postoperative histopathology. (A, B) Negative frozen-section margin points and resection lines. (C) Left medial orbital wall resection with orbital preservation. (D) Endoscopic removal of the nasal bone with chisel and hammer. (E) Post-en bloc tumor resection status. (F) Post–skull-base reconstruction status. (G) En bloc specimen. (H) Hematoxylin and eosin staining. Undifferentiated cells with prominent nucleoli and a high nuclear-to-cytoplasmic ratio in trabecular and sheet-like patterns. (I) SMARCA4 immunostaining: complete loss.
Reconstruction used a 3-layer technique (Figure 2F): inlay fascia lata graft sutured and sealed with fibrin glue, overlay graft, and a right extended pedicled nasal septal flap based on the posterior septal artery, ensuring watertight closure. Healing was uneventful.
Histopathology showed tumor cells with prominent nucleoli and a high nuclear-cytoplasmic ratio in trabecular and sheet-like patterns (Figure 2H) without neuroendocrine features. Immunohistochemistry showed complete loss of SMARCA4 (Figure 2I), with retained SMARCB1 and SMARCA2. The tumor was diagnosed as SMARCA4-deficient carcinoma according to the fifth edition of the WHO Classification of Head and Neck Tumors. 1 All margins were negative.
Postoperative IMRT (60 Gy/30 fractions) started on day 13 and ended without interruption. SMARCA4 deficiency was confirmed during radiotherapy. Adjuvant FP chemotherapy (fluorouracil 800 mg/m2 and cisplatin 80 mg/m2) was administered for 3 cycles. At 25 months, she remains disease-free.
Discussion
This case illustrates the diagnostic and surgical challenges of SMARCA4-deficient sinonasal carcinoma, an extremely rare and aggressive entity. The preoperative diagnosis of olfactory neuroblastoma was revised after the loss of SMARCA4, highlighting diagnostic pitfalls. The discrepancy between biopsy and final pathology highlights the need for comprehensive immunohistochemistry in undifferentiated sinonasal tumors.
The fifth WHO classification defined sinonasal carcinoma deficient in the switch/sucrose-nonfermenting (SWI/SNF) complex.1,2 The SWI/SNF complex regulates chromatin remodeling. SMARCA4 is one of its core subunits and is believed to contribute to tumorigenesis by regulating BRG1, a key tumor suppressor protein. 3 Recognition of the role of SWI/SNF complex gene mutations in oncogenesis is relatively recent compared to classical oncogenes and tumor suppressor genes such as MYC, RAS, and TP53. 4 A retrospective review of 299 cases of undifferentiated carcinoma, poorly differentiated carcinoma, neuroendocrine carcinoma, and teratocarcinosarcoma of the sinonasal tract identified only 12 cases (4%) of SMARCA4-deficient carcinoma, indicating that this tumor is extremely rare. 5 Among SWI/SNF complex-deficient carcinomas, SMARCB1-deficient carcinoma is the most common subtype, while SMARCA4-deficient carcinoma occurs much less frequently. 2 SMARCB1-deficient carcinoma generally exhibits undifferentiated morphology, and SMARCA4-deficient carcinoma shows similar cytologic features; however, it often shows an alveolar or nested architecture with incomplete rosette formation, similar to high-grade neuroendocrine carcinoma. 2 Due to these overlapping features, a definitive classification may be difficult when only a small biopsy specimen is available. 2 In the present case, an accurate preoperative diagnosis was also challenging.
Deficient carcinomas in the SWI/SNF complex show highly aggressive clinical behavior, with more than 50% of patients dying within 2 years of diagnosis.2,6 Some reports suggest that SMARCA4-deficient carcinoma has an even poorer prognosis than SMARCB1-deficient carcinoma.5,6 In non-small cell lung carcinoma, loss of SWI/SNF complex function has been associated with tumor dedifferentiation. 7 Similarly, in the sinonasal region, loss of components of SWI/SNF, including SMARCA4, has been implicated in the development of poorly differentiated and undifferentiated carcinomas. 8 Our case also exhibited an undifferentiated morphology without any specific differentiation pattern and, given the presumed poor prognosis, a multimodal treatment strategy similar to that used for SNUC was considered. At our institution, we have previously treated 4 cases of SNUC. Although 2 patients died of distant metastases, local control was achieved in all cases. 9 Based on these experiences, we consider that combining surgery, radiation therapy, and chemotherapy as extensively as possible is important to achieve local control. 9
In this case, since the preoperative diagnosis was olfactory neuroblastoma, transnasal endoscopic surgery was performed, resulting in a successful en bloc tumor resection. Furthermore, the affected nasal bone was also successfully resected under endoscopic guidance. Although nasal bone involvement is listed as one of the contraindications to a purely endoscopic approach, 10 we consider that endoscopic resection is feasible when the lesion is localized and does not extend beyond the nasal bone to the subcutaneous tissue. By detaching the outer margin of the tumor from the orbital side and mobilizing it medially, the area of contact with the nasal bone becomes visible, allowing the bone to be removed with a chisel and hammer and dissected free from the subcutaneous tissue for en bloc resection. Given the proximity of critical structures such as the brain and orbit, even a negative margin in endoscopic surgery can be considered ‘close’. Therefore, postoperative IMRT was performed with a total dose of 60 Gy.
No standardized chemotherapy regimen has been established for SMARCA4-deficient carcinoma or undifferentiated carcinoma, and comparative data between regimens are lacking. Although some reports involving thoracic SMARCA4-deficient tumors have suggested the efficacy of taxane and platinum combination therapy, 11 no consensus has been reached. For SNUC, platinum-based regimens are commonly employed. At our institution, cisplatin and fluorouracil (FP regimen) is administered to patients with good systemic tolerance, resulting in favorable local control. 9 In the present case, the FP regimen was administered according to the SUNC treatment strategy. Because the final pathological diagnosis was confirmed near the end of IMRT, concurrent chemoradiation was limited to 2 days. Given the rarity and unknown clinical course of this disease, but considering its high-grade undifferentiated nature and poor prognosis, intensive therapy was pursued, and 3 cycles of chemotherapy were completed within the systemic tolerance of the patient. The patient remains disease-free despite the high-grade histology, probably reflecting the benefit of this aggressive multimodal approach.
Conclusions
SMARCA4-deficient sinonasal carcinoma is a rare and highly aggressive tumor lacking an established treatment strategy. Our case demonstrates that long-term disease control can be achieved through an aggressive multimodal approach combining endoscopic skull base resection, postoperative IMRT, and adjuvant chemotherapy. Furthermore, this case illustrates that even tumors involving the nasal bone can be safely and completely removed en bloc using a purely endoscopic transnasal approach with appropriate surgical planning and reconstruction, thus broadening the scope of endoscopic skull base surgery.
Footnotes
Acknowledgements
The authors declare that there are no acknowledgements to disclose.
Ethical Considerations
This study was approved by the Clinical Research Ethics Review Committee of Mie University Hospital (approval no. H2020-054). Written informed consent was obtained from the patient for publication of this report. The authors declare no conflicts of interest.
Consent to Participate
Not applicable.
Consent for Publication
The patient provided us informed consent for the publication of this case report.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are included within the article. Additional details are available from the corresponding author upon reasonable request.
