Abstract
Objective:
This study aims to evaluate the surgical technique, indications, and clinical outcomes of facial nerve decompression performed via the transcanal endoscopic approach (TEA) for traumatic facial paralysis caused by temporal bone fracture. Given the anatomical constraints of TEA, this study targets patients with lesions limited to the tympanic segment and/or geniculate ganglion to provide preliminary evidence of its feasibility.
Methods:
A retrospective analysis was conducted on 6 patients with traumatic facial paralysis refractory to conservative treatment who were admitted between January 2021 and February 2024. All patients underwent facial nerve decompression via TEA, during which the surgical procedure steps were recorded. In addition, facial nerve function was assessed using the House-Brackmann (HB) grading system preoperatively and postoperatively, and pure-tone audiometry was used to compare audiological results.
Results:
All 6 patients underwent preoperative evaluation with temporal bone high-resolution computed tomography. Of these patients, 5 had injuries located at the geniculate ganglion and 1 at the tympanic segment. Preoperatively, the facial nerve function of the patients was graded as grade V in 5 patients and grade IV in 1 patient. All patients had an electroneurography value of ≥90%. At 6 months postoperatively, facial nerve function improved to HB grade I in 4 patients and grade II in 2 patients, and the overall improvement was statistically significant (P = .024). For audiological outcomes, both the postoperative air-bone gap and air-conduction thresholds improved significantly compared with preoperative values (both P = .028), whereas bone-conduction thresholds showed no significant change (P = .197).
Conclusion:
For patients with traumatic facial paralysis whose lesions are confined to the tympanic segment and/or geniculate ganglion and who fail conservative therapy, TEA may represent a feasible and minimally invasive salvage option.
Keywords
Introduction
Peripheral facial paralysis is a neuropathy characterized by the loss of innervation along the path of the facial nerve trunk, from its origin in the brainstem to its terminal branches in the face. 1 Its etiologies include idiopathic causes, viral infections, otogenic diseases, trauma, immune-metabolic factors, and so forth. Currently, the incidence rate of peripheral facial paralysis caused by trauma is continuously increasing. Studies have shown that traumatic facial paralysis accounts for approximately 17% of all peripheral facial paralysis cases. 2 Approximately 7% to 10% of temporal bone fractures may be complicated by facial paralysis. 3
The temporal bone fractures are traditionally classified into 3 types (transverse, longitudinal, and mixed) based on the relationship between the fracture line and the long axis of the petrous part of the temporal bone. 4 It is well known that the fracture type is closely associated with the occurrence of facial paralysis. For instance, longitudinal fractures are the most common, accounting for 85% to 90%, with fracture lines parallel to the long axis of the petrous part. Transverse fractures account for 10% to 15%, with fracture lines perpendicular to the long axis of the petrous part. Mixed fractures account for the smallest proportion (<5%), exhibit both longitudinal and transverse features, and are often accompanied by comminuted temporal bone injuries. Currently, temporal bone fractures are mainly classified based on whether the inner ear is involved, otic capsule-intact fractures (fracture lines do not involve the inner ear labyrinth), and otic capsule-violating fractures (fracture lines pass through the inner ear labyrinth). 5 This classification method better facilitates accurate clinical assessment of the condition, formulation of treatment plans, and evaluation of prognosis. Traditionally, facial nerve decompression has been performed through the middle cranial fossa, transmastoid, or translabyrinthine approaches, each offering access to specific segments of the intratemporal facial nerve. However, these approaches also have inherent anatomical and technical limitations, which have prompted interest in less invasive alternatives in selected cases.
In recent years, with the development of endoscopic ear surgery, the transcanal endoscopic approach (TEA) has emerged as a minimally invasive alternative for selected intratemporal facial nerve lesions. TEA provides a direct corridor to the tympanic segment and geniculate ganglion, providing potentially improved visualization of these areas in selected cases compared with microscopic approaches. 6
Given the limited evidence regarding the role of the TEA in traumatic facial paralysis, further clinical data are needed to clarify its potential value. Therefore, this study retrospectively analyzed patients with traumatic facial paralysis who underwent TEA-assisted facial nerve decompression after failing conservative treatment. The aim was to describe the surgical procedure, evaluate postoperative facial nerve and hearing outcomes, and provide preliminary insights into the indications for this approach.
Materials and Methods
General Data
This single-center retrospective case series included patients with traumatic facial paralysis who underwent facial nerve decompression via TEA at the Provincial Hospital Affiliated to Shandong First Medical University between January 2020 and February 2024. The inclusion criteria were as follows: (1) traumatic facial paralysis secondary to temporal bone fracture; (2) failure of standardized conservative treatment for at least 3 to 4 weeks; (3) preoperative House-Brackmann (HB) grade IV to VI; (4) radiological evidence of involvement of the tympanic segment and/or geniculate ganglion; and (5) a minimum postoperative follow-up of 6 months. The exclusion criteria were as follows: (1) injury involving the mastoid or labyrinthine segments, which cannot be adequately exposed through the TEA approach; (2) otic capsule-violating fractures with profound sensorineural hearing loss; and (3) prior otologic surgery or incomplete clinical data. Preoperative assessments included otoscopic examination, audiological evaluation, high-resolution computed tomography (HRCT) of the temporal bone, electroneurography (ENoG), and HB grading. Postoperative follow-up was conducted at 1, 2, and 6 months after surgery, including assessment of HB grade, audiological evaluation, middle ear status, and documentation of perioperative complications. Written informed consent was obtained from all patients, and the study was approved by the institutional ethics committee (Approval No. SWYX2025-022).
Surgical Technique
All patients were placed in the supine position, and surgery was performed under general anesthesia with endotracheal intubation, with the head turned to the healthy side. After evaluating the integrity of the tympanic membrane under an ear endoscope, a longitudinal knife was used to make a longitudinal incision 1.0 cm away from the tympanic ring in the external auditory canal via the transcanal approach, from the inside out. For the left ear, the incision was made clockwise from the 9 o’clock position to the 6 o’clock position; for the right ear, it was made counterclockwise from the 3 o’clock position to the 6 o’clock position. A disk knife was used to make a fan-shaped incision on the posterior wall flap of the external auditory canal (Figure 1A), and the flap was separated and passed over the tympanic ring to enter the tympanic cavity (Figure 1B). The posterior wall of the external auditory canal, the scutum, and the lateral wall of the epitympanic antrum were curetted to expose the facial recess, thereby fully revealing the tympanic segment of the facial nerve. If the injury was localized to the geniculate ganglion, the incudostapedial joint was separated, the incus was removed, and the head of the malleus was excised (Figure 1C) to fully expose the epitympanic cavity and the facial nerve, allowing clear visualization of the entire tympanic segment of the facial nerve from the geniculate ganglion to the pyramidal eminence. The tegmen tympani was exposed superiorly, and the pyramidal turn of the facial nerve was exposed posteriorly using the stapedius muscle as a landmark. The geniculate ganglion of the facial nerve was localized anterior to the cochleariform process. The cochleariform process and the superior air cells were drilled away, and the greater petrosal nerve was traced to completely expose the geniculate ganglion of the facial nerve. The nerve sheath was incised, starting from the anterior space of the epitympanic cavity and ending at the pyramidal eminence. The bone surrounding the nerve that caused compression was curetted to complete the decompression (Figure 1D). A partial ossicular replacement prosthesis (PORP) was placed above the stapes (Figure 1E). The tragus cartilage was shaped such that its smooth surface faced the prosthesis, and the perichondrium-covered surface faced the detached tympanic membrane. A small amount of dexamethasone-soaked Nasopore packing was placed around the ossicles and on the surface of the decompressed facial nerve to fix the ossicles and reduce facial nerve edema. The residual tympanic membrane was repositioned and aligned completely (Figure 1F). The surface of the tympanic membrane was covered with dexamethasone-soaked Nasopore packing, the surgical cavity was packed with iodoform gauze strips, and the surgical ear was covered with a dressing.
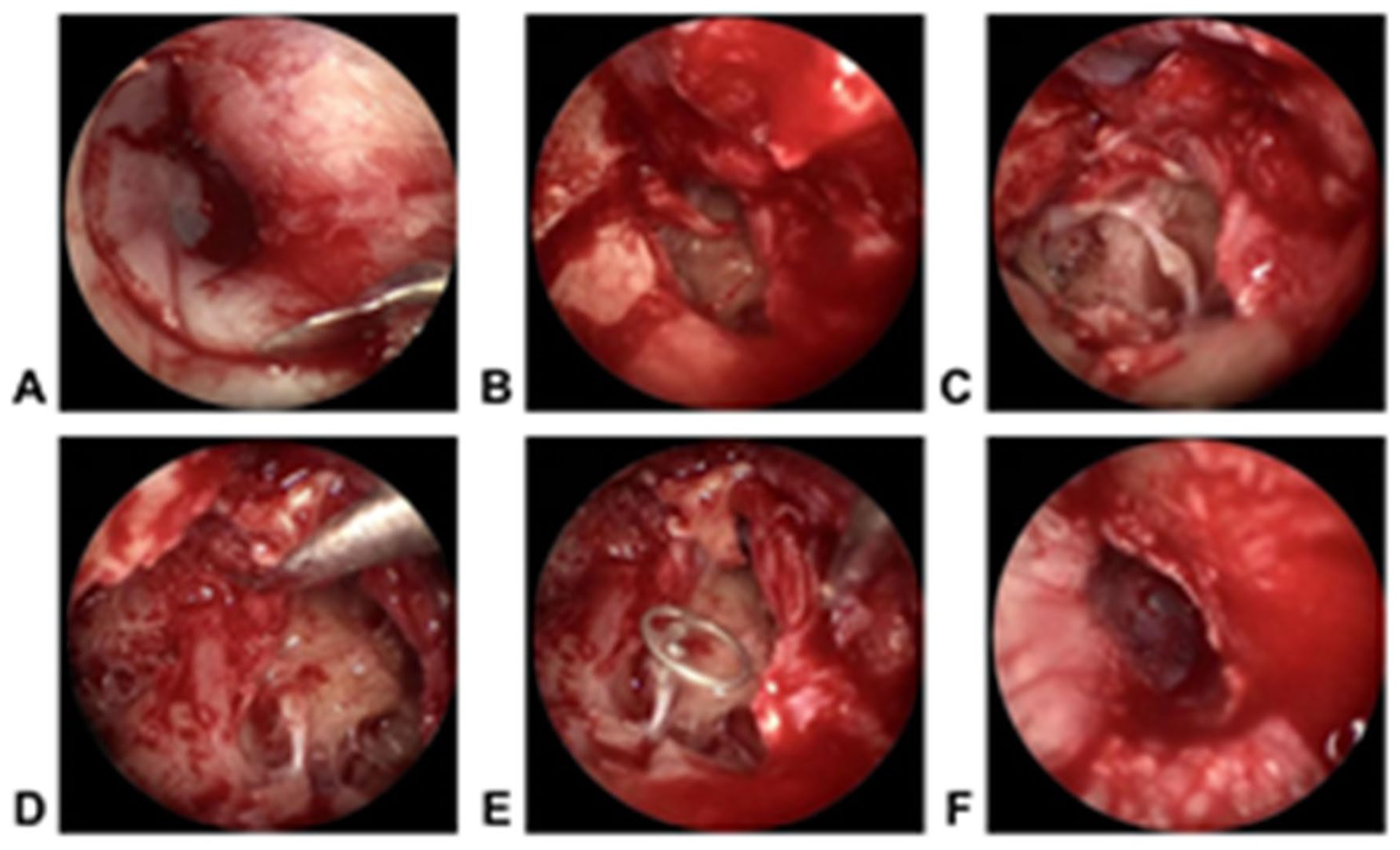
Surgical procedure. (A) A curved incision is made in the external auditory canal. (B) The tympanic membrane flap is elevated within the external auditory canal. (C) The lateral wall of the upper tympanic recess is curetted, followed by removal of the incus and malleus. (D) The facial nerve canal is removed, and facial nerve decompression is subsequently performed. (E) Artificial ossicles are implanted to restore auditory function. (F) The external auditory canal and tympanic membrane flap are restored.
Statistical Analyses
Given the small sample size (n = 6) and non-normal distribution of the data, non-parametric testing (Wilcoxon signed-rank test) was used to compare preoperative and postoperative HB grades and audiological data. Statistical analyses were performed using SPSS 27.0 (IBM Corp., Armonk, NY, USA), with the significance level set at P < .05.
Results
Patient Characteristics
A total of 6 patients met the inclusion criteria, including 5 males and 1 female, with a mean age of 35.1 years (range from 9 to 54 years). The preoperative disease duration of all patients was 1 to 11 months, with a median duration of 1.65 months (Table 1). Preoperative temporal bone HRCT findings showed that all 6 patients had longitudinal fractures, among which 5 cases had fracture lines involving the geniculate ganglion and 1 case involving the tympanic segment (Figure 2). No patient had mastoid or labyrinthine segment involvement.
Basic Information of Patients.
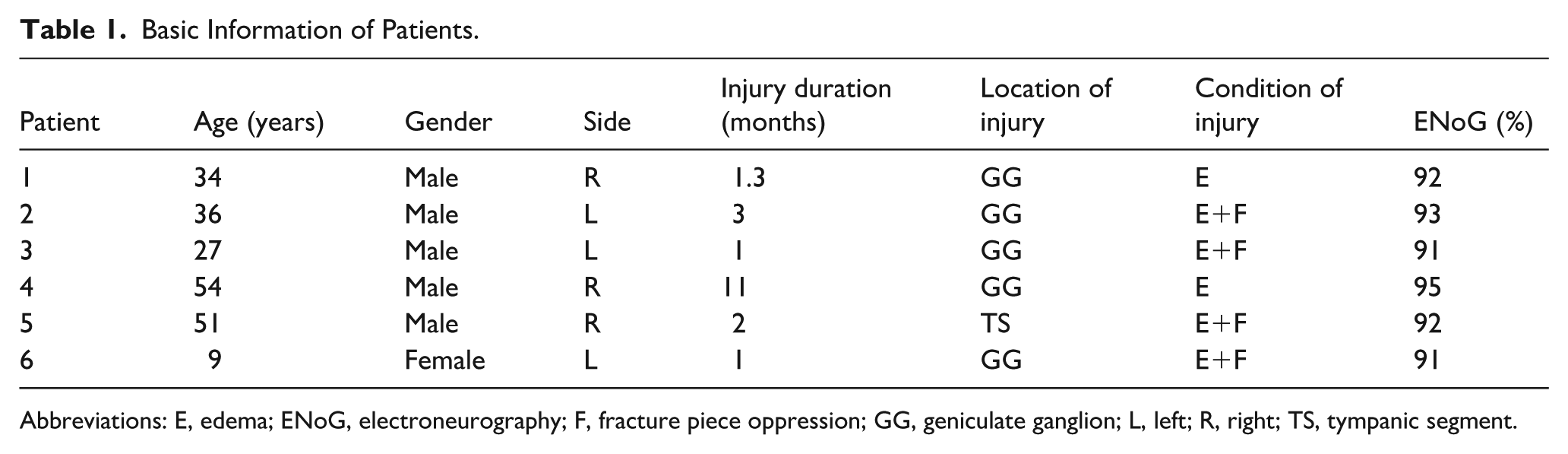
Abbreviations: E, edema; ENoG, electroneurography; F, fracture piece oppression; GG, geniculate ganglion; L, left; R, right; TS, tympanic segment.
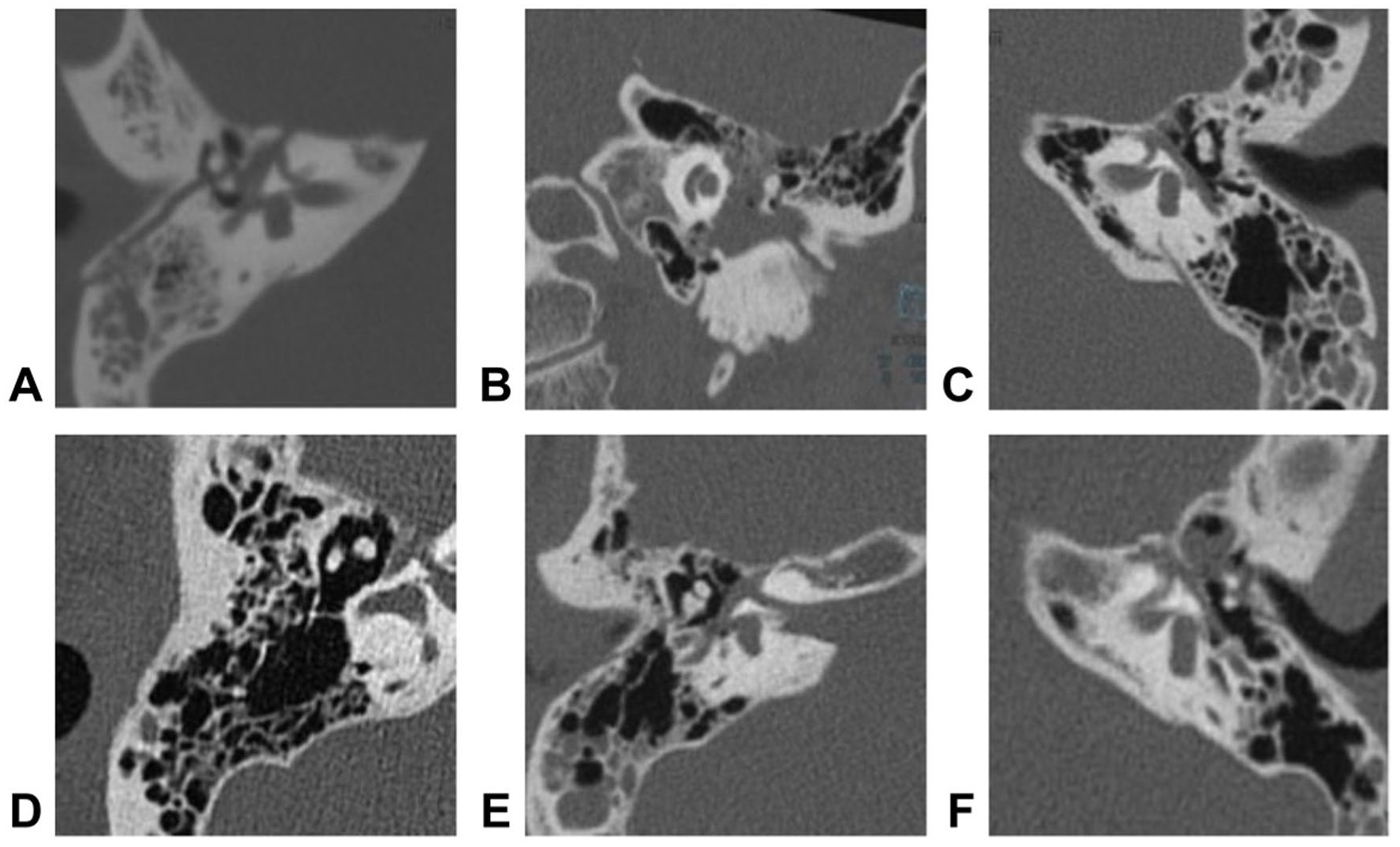
The damaged location on the temporal bone CT. (A) GG. (B) GG. (C) GG. (D) GG. (E) TS. (F) GG. CT, computed tomography; GG, geniculate ganglion; TS, tympanic segment.
Facial Nerve Function Outcomes
Preoperatively, all patients presented with HB grade IV to V facial paralysis and ENoG values ≥90%. Five patients were classified as HB grade V and 1 as HB grade IV. At 2 months postoperatively, 4 patients improved to HB grade II, 1 to grade I, and 1 to grade III. At 6 months postoperatively, 4 patients achieved HB grade I and 2 achieved HB grade II (Figure 3). The facial nerve function of all 6 patients was significantly improved (P = .024).
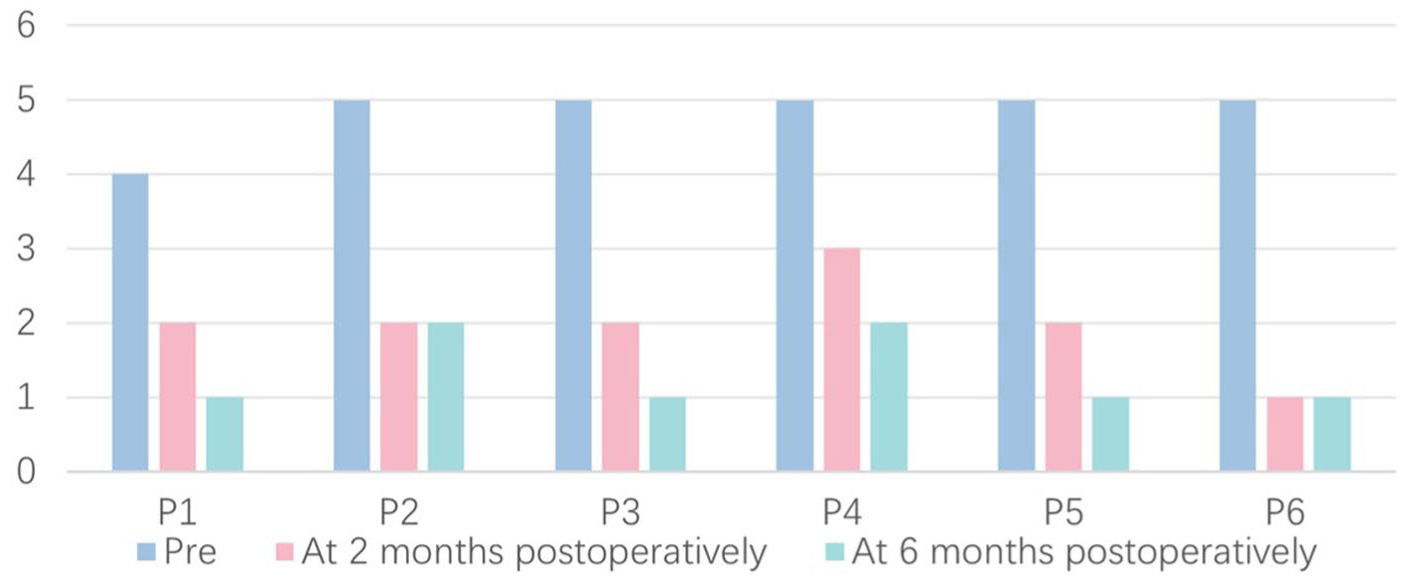
House-Brackmann facial nerve grades for all patients preoperatively, at 2 months postoperatively, and at 6 months postoperatively. A significant improvement in facial nerve function was observed (P = .024). P, patient.
Hearing Outcomes
The preoperative mean air-conduction (AC) threshold was 42.29 ± 25.43 dB HL, the bone-conduction (BC) threshold was 19.79 ± 6.39 dB HL, and the mean air-bone gap (ABG) was 22.50 ± 19.81 dB HL. Among the patients, 5 underwent ossicular chain reconstruction with a PORP combined with endoscopic facial nerve decompression. One patient, whose facial nerve injury was in the tympanic segment of the facial nerve, retained the original ossicles and only received endoscopic facial nerve decompression at the injury site. At 6 months postoperatively, the mean AC threshold was 24.58 ± 8.65 dB HL, the BC threshold was 17.71 ± 8.04 dB HL, and the mean ABG decreased to 6.88 ± 1.72 dB HL. Postoperatively, both the ABG and AC thresholds showed significant improvement compared with preoperative values (both P = .028), whereas BC thresholds remained unchanged (P = .197) (Figure 4).
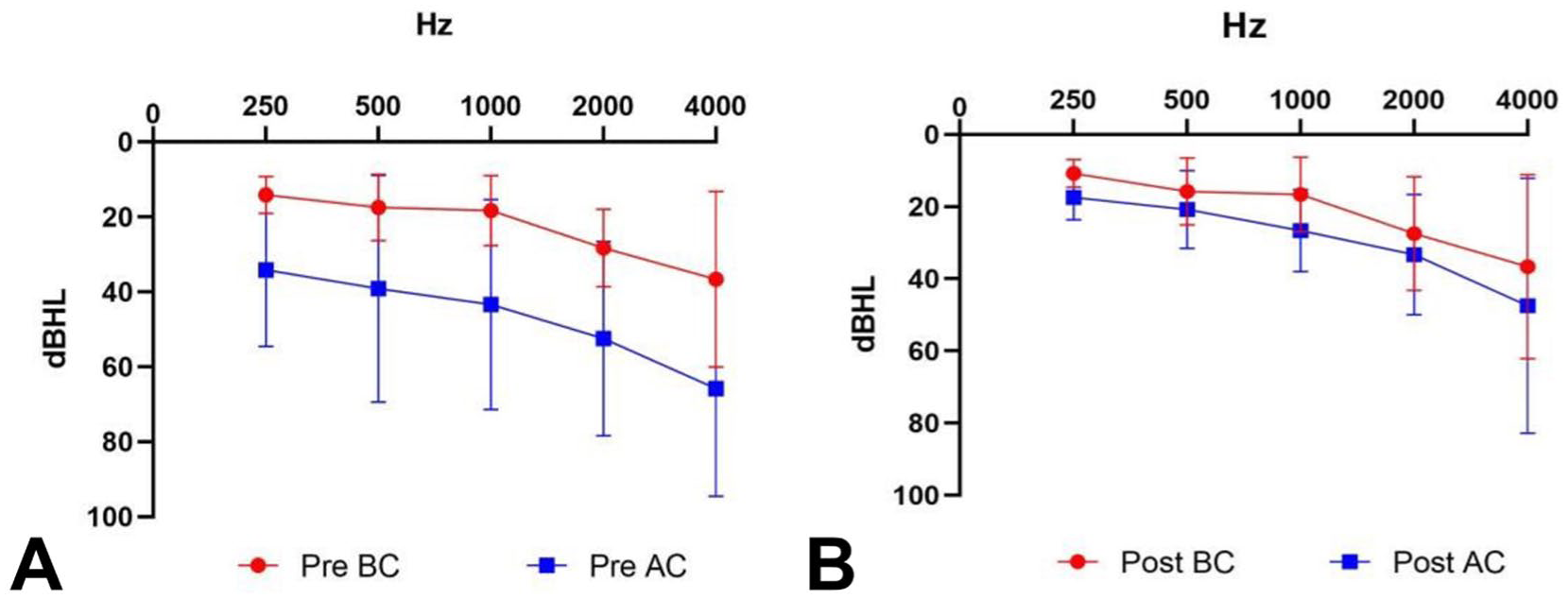
Mean hearing levels before and after surgery. (A) Mean hearing levels before operation. (B) Mean hearing levels at 6 months postoperatively. Postoperatively, both AC thresholds and the ABG showed significant improvement compared with preoperative values (both P = .028), whereas BC thresholds remained unchanged (P = .197). ABG, air-bone gap; AC, air-conduction; BC, bone-conduction.
Postoperative Complications
No major complications were observed during follow-up, including cerebrospinal fluid leakage, sensorineural hearing loss, postoperative facial nerve deterioration, or noticeable taste disturbance. No infections, vertigo, external auditory canal stenosis, or delayed facial nerve edema were noted.
Discussion
Facial paralysis not only impairs patients’ physical function but also causes impairment of their self-image and difficulties in social interaction. 7 Therefore, implementing effective interventions for patients plays a vital role in promoting their multidimensional rehabilitation.
It has been documented that the selection of surgical approaches for facial nerve decompression should be comprehensively evaluated based on the location of facial nerve injury, hearing level, and vestibular function. 8 The middle cranial fossa approach is suitable for patients with injuries located in the geniculate ganglion or internal auditory canal segment, and with intact hearing and vestibular function. 9 This approach can preserve the integrity of the mastoid and middle ear structures without disrupting the ossicular chain. However, its limitations are also prominent; the middle cranial fossa has few anatomical landmarks. Intraoperatively, craniotomy and retraction of the temporal lobe brain tissue are required to expand the surgical field, which may lead to complications such as intracranial hemorrhage, cerebrospinal fluid leakage, vertigo, tinnitus, and cerebral edema while also causing significant impacts on patients’ appearance. 10 The labyrinthine approach is applicable to patients with complete loss of hearing and vestibular function, as it can fully expose the entire course of the facial nerve, especially when the injury is located in the labyrinthine segment and geniculate ganglion. 11 Nevertheless, for patients with residual hearing, this approach requires sacrificing the remaining hearing. The mastoid approach is the most commonly used clinical approach, 12 which is suitable for patients with hearing impairment but preserved vestibular function. It can provide a clear view of the tympanic segment and mastoid segment of the facial nerve, which enables ossicular chain reconstruction while avoiding craniotomy. However, it requires postauricular incision and extensive bone drilling, and there is a significant limitation in the visual field for exposing the labyrinthine segment and geniculate ganglion. In addition, mastoidectomy and epitympanotomy can become difficult when the mastoid is poorly pneumatized or the middle fossa dura lies low. In such cases, surgeons may need to remove the posterior canal wall to obtain adequate microscopic exposure. 13 In clinical practice, we found that the current surgical approaches still have notable shortcomings. Especially for geniculate ganglion decompression, they often rely on craniotomy, which is associated with problems such as severe trauma, insufficient surgical field exposure, prolonged postoperative recovery period, and cosmetic defects. These issues lead some patients to refuse surgical treatment and opt for conservative treatment. However, conservative treatment often yields suboptimal clinical outcomes.
With the development of ear endoscopic technology and the accumulation of clinical application experience, its advantages in facial nerve decompression surgery have become increasingly prominent. Using the natural external auditory canal as the surgical pathway, the ear endoscope can directly reach the facial nerve bony canal and adequately expose the geniculate ganglion and tympanic segment of the facial nerve. 14 However, recent evidence indicates that the TEA is suitable only for lesions confined to the tympanic segment and geniculate ganglion. 15 This cannot provide adequate exposure to the labyrinthine or mastoid segments, which are frequently involved in traumatic injuries. 16 In this study, all enrolled patients had fractures limited to the tympanic segment and geniculate ganglion, reflecting the strict anatomical indications of TEA.
This study found that when the lesion involved the geniculate ganglion, interruption of the ossicular chain with removal of the incus and/or malleus head was required intraoperatively to obtain adequate surgical space and achieve decompression. Ossicular chain reconstruction could be performed simultaneously, allowing some patients to experience postoperative hearing improvement compared with preoperative levels. In this study, both the ABG and AC thresholds showed a trend toward improvement, consistent with previous reports of facial nerve decompression performed via the TEA approach. However, endoscopic decompression of the geniculate ganglion inherently requires removal of the ossicular chain, which inevitably results in a certain degree of middle-ear trauma and hearing cost. 17 In addition, postoperative hearing outcomes are influenced by multiple factors, including the specific techniques used for ossiculoplasty, prosthesis stability, and the physiological condition of the middle-ear cavity. Middle-ear reconstruction itself also carries inherent risks and variability and therefore does not necessarily lead to hearing improvement. 18 Consequently, postoperative hearing improvement cannot be attributed solely to decompression, and decompression surgery should not be regarded as direct evidence of hearing restoration.
Previous studies found that the combination of temporal bone HRCT and ABG assessment enables more accurate determination of the ossicular chain status. 19 Preoperatively, the ossicular chain is mostly disrupted when ABG ≥35 dB HL, while it is mostly intact when ABG <35 dB HL. In this study, 1 patient had preoperative ABG >35 dB HL. Meanwhile, temporal bone HRCT revealed incudostapedial joint dislocation, which was also confirmed intraoperatively. The above results indicate that preoperative evaluation of the ossicular chain status using temporal bone HRCT combined with ABG can prepare for intraoperative ossicular chain reconstruction.
Previous studies have generally emphasized the timeliness of facial nerve decompression, highlighting the core role of early surgery.20-23 These timing criteria, based on strict electrophysiological thresholds (such as performing surgery within 14 days or requiring an ENoG value >90%), have notable limitations in clinical practice and are often not applicable to patients who miss the early intervention window due to ineffective conservative treatment, delayed referral, or concomitant severe injuries. Recent studies have indicated that when facial nerve degeneration >90% and HB grade is ≥V, early surgery is the first choice. 24 However, in the present study, all patients underwent delayed decompression only after initial conservative therapy failed to achieve satisfactory improvement, and 1 patient with preoperative HB grade IV even underwent surgery after the indication threshold was relaxed at the patient’s strong request. Although all patients in this study demonstrated postoperative improvement in facial nerve function, delayed decompression should be regarded as a salvage option rather than a substitute for early intervention. Moreover, a proportion of patients with delayed-onset or incomplete traumatic facial paralysis may experience spontaneous recovery under conservative management. 16 Therefore, the postoperative improvement observed in this study may partially reflect natural recovery rather than being solely attributable to surgical decompression.
This study used the HB grading system rather than the more detailed Sunnybrook Facial Grading System, which may have reduced the sensitivity of facial nerve function assessment. The decision not to use the Sunnybrook scale was based on several considerations. First, because this was a retrospective study, the available clinical data were derived from routine medical records collected at different time points. The Sunnybrook system requires standardized photographic or video documentation and consistent scoring procedures, which were not uniformly available in the existing records, making complete and reliable scoring impractical. 25 Second, the HB scale is the most widely used clinical tool for evaluating facial nerve function and is routinely incorporated into the diagnostic and follow-up workflow for traumatic facial paralysis.1,24 All patients in this study had HB grading at baseline and during follow-up, ensuring good data availability and comparability. Future prospective studies with standardized follow-up protocols may incorporate the Sunnybrook system to enhance the granularity and sensitivity of facial nerve outcome assessment.
In addition, cosmetic outcomes are also an important consideration when selecting a surgical approach. Compared with traditional postauricular or craniotomy approaches, TEA utilizes the natural external auditory canal and requires no additional skin incision, theoretically reducing postoperative scarring and visible deformity and thus offering theoretical cosmetic advantages. 26 However, this study did not include an objective assessment of cosmetic outcomes. Future research may consider incorporating standardized cosmetic evaluation scales or patient-reported outcome measures to more comprehensively assess the aesthetic impact of TEA.
This study has several limitations. First, the sample size was relatively small, which may limit the robustness of the statistical analysis. However, as an exploratory study, the primary aim was to evaluate the feasibility and preliminary effectiveness of this technique. Second, this was a retrospective case series without a control group, making it impossible to directly compare outcomes with conservative management, early decompression, or other surgical approaches such as the transmastoid, translabyrinthine, or middle cranial fossa routes. Consequently, it remains difficult to determine whether facial nerve recovery exceeded the natural course or alternative treatments. Third, due to the anatomical indications of TEA, only patients with injuries involving the geniculate ganglion and tympanic segment were included, which may limit the generalizability of the findings. Larger, prospective studies with appropriate control groups are needed to further clarify the role of TEA in the management of traumatic facial paralysis.
Conclusion
The TEA is a feasible, minimally invasive option for decompression of traumatic facial nerve injuries confined to the tympanic segment and geniculate ganglion. Used as a salvage procedure in this study, TEA resulted in postoperative improvement in facial nerve function in all patients. However, because ossicular manipulation is required and spontaneous recovery may also contribute, the extent to which this improvement reflects a surgical effect remains to be clarified. Larger prospective studies are needed to further clarify the role of TEA in the management of traumatic facial paralysis.
Footnotes
Ethical Considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Shandong Provincial Hospital Affiliated to Shandong First Medical University.
Consent to Participate
All participants provided informed consent to participate in this study.
Consent for Publication
All participants provided consent for publication of anonymized data.
Author Contributions
All authors contributed significantly to the work, have seen and approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
