Abstract
Subglottic stenosis (SGS) may arise from both mechanical and inflammatory airway injury. Patients with mast cell activation syndrome (MCAS) are uniquely vulnerable, due to both
Introduction
Subglottic stenosis (SGS) is a congenital, acquired, or idiopathic condition characterized by the narrowing of the subglottis. This region is unique in that it is the only proximal airway segment completely enclosed by a cartilaginous ring, the cricoid cartilage, making it susceptible to stenosis. 1 Risk factors for acquired SGS include obesity, diabetes, gastroesophageal reflux disease (GERD), and certain autoimmune disorders. 2 The most notable precipitating scenario for SGS involves prolonged and repeated intubation, which can cause trauma and can readily progress to stenosis if underlying chronic inflammatory states are present. 3 Therefore, it is essential to identify conditions that may predispose patients to requiring endotracheal intubation more frequently.
Mast cell activation syndrome (MCAS) describes a group of disorders that result from the inappropriate release of mast cell mediators. 4 The most prominent and well-defined physiopathological role of mast cells is in the early allergic reaction. 5 Therefore, inappropriate activation of mast cells in MCAS may result in recurrent anaphylaxis, 6 leading to airway edema and bronchoconstriction, often requiring intubation. This clinical pattern suggests that MCAS and illnesses that require similar management may lead to SGS after repeated intervention, especially in the setting of baseline systemic inflammation.
Acute upper airway compromise in MCAS or anaphylaxis is typically due to laryngeal edema or angioedema, rather than fixed airway narrowing such as SGS.7,8 There are no published cases or case series describing SGS as a complication of MCAS or anaphylaxis. However, delayed recognition and management of SGS can have severe consequences, with increased risk of life-threatening airway compromise, more frequent Emergency Department visits and hospitalizations, and higher likelihood of urgent or unplanned surgical interventions, including tracheostomy.9,10 This case represents a teachable sentinel event, highlighting the consequences of delayed recognition of structural airway injury in a medically complex patient population.
Materials and Methods
Informed consent was obtained from the patient for the review of her case. Relevant details of her course were extracted from the electronic medical record, including presenting symptoms, imaging and laboratory studies, procedural interventions, postoperative course, and long-term management. Flexible laryngoscopy and operative endoscopy were reviewed for airway findings. All identifying information was removed. Approval through the Mayo Clinic Institutional Review Board (IRB) was sought, and the study was deemed not to require further IRB review.
A narrative review of the literature was performed to contextualize the presented case within the current knowledge of MCAS and SGS. Searches were performed using PubMed, Scopus, and Google Scholar with terms related to “mast cell activating/activation syndrome,” “recurrent anaphylaxis,” “traumatic intubation,” and “subglottic stenosis.” Literature search was supplemented by an AI-assisted evidence synthesis tool (www.openevidence.com; OpenEvidence) to identify additional relevant studies. All studies identified through AI-assisted search were independently reviewed and verified by authors before inclusion.
Case Report
A 34-year-old female experienced recurrent urticaria, shortness of breath, throat tightening, stridor, and facial flushing. These symptoms prompted presentation to the Emergency Department over 20 times, with 6 episodes requiring intubation of 2 to 5 days duration. For a presumed diagnosis of MCAS, the patient was initiated on high-dose fexofenadine, cromolyn, omalizumab, and montelukast without long-term relief. She carried an EpiPen and self-administered epinephrine during exacerbations with minimal improvement.
While her constellation of symptoms was most consistent with MCAS, lack of improvement on standard-of-care therapies raised concern for possible other etiologies. A thorough hematologic and immune evaluation was completed. Bone marrow biopsy revealed no mast cell aggregates, and a C-KIT mutation analysis was negative, indicating no clonal mast cell disorder. Evaluation for carcinoid syndrome was unrevealing. Autoimmune workup values, including ANA, anti-SSA/SSB, anti-dsDNA, anti-SCL70, ANCA panel, anti-CCP, and RF, were all within normal range. Testing for C4 and C1 esterase inhibitor deficiency was negative, indicating no bradykinin-induced hereditary angioedema.
One year after her initial episode, she presented again to her local Emergency Department with persistent dyspnea for 3 days. She was transferred to a large tertiary care center and admitted to the intensive care unit for continued management. She was started on Heliox, dexamethasone, and racemic epinephrine nebulizer treatments; however, she continued to have increased work of breathing. Flexible laryngoscopy revealed grade III SGS causing near-total airway occlusion.
At that time, the patient was taken to the operating room for urgent intervention. A microdirect laryngoscopy was performed for visualization of the affected segment. A nonocclusive airway balloon with a size of 18 mm was inflated to 6 ATM twice to open the stenotic segment initially. A carbon dioxide (CO2) laser was subsequently used at a setting of 3 W to create 3 radial cuts at 6, 9, and 12 o’clock points. The balloon was inserted and inflated a third time, this time to 7 ATM. A total of 1.5 mL of a 40 mg/mL concentration of triamcinolone was injected into the stenotic area. Subglottic tissue was sent for pathologic evaluation, including keratin staining (Figure 1A and B), hematoxylin and eosin (Figure 1C and D), tryptase (Figure 1E), and CD117 (Figure 1F). Notably, keratin was negative for carcinoma, and CD117 demonstrated foci with mast cells ranging from 10 to 50 per high-power field.
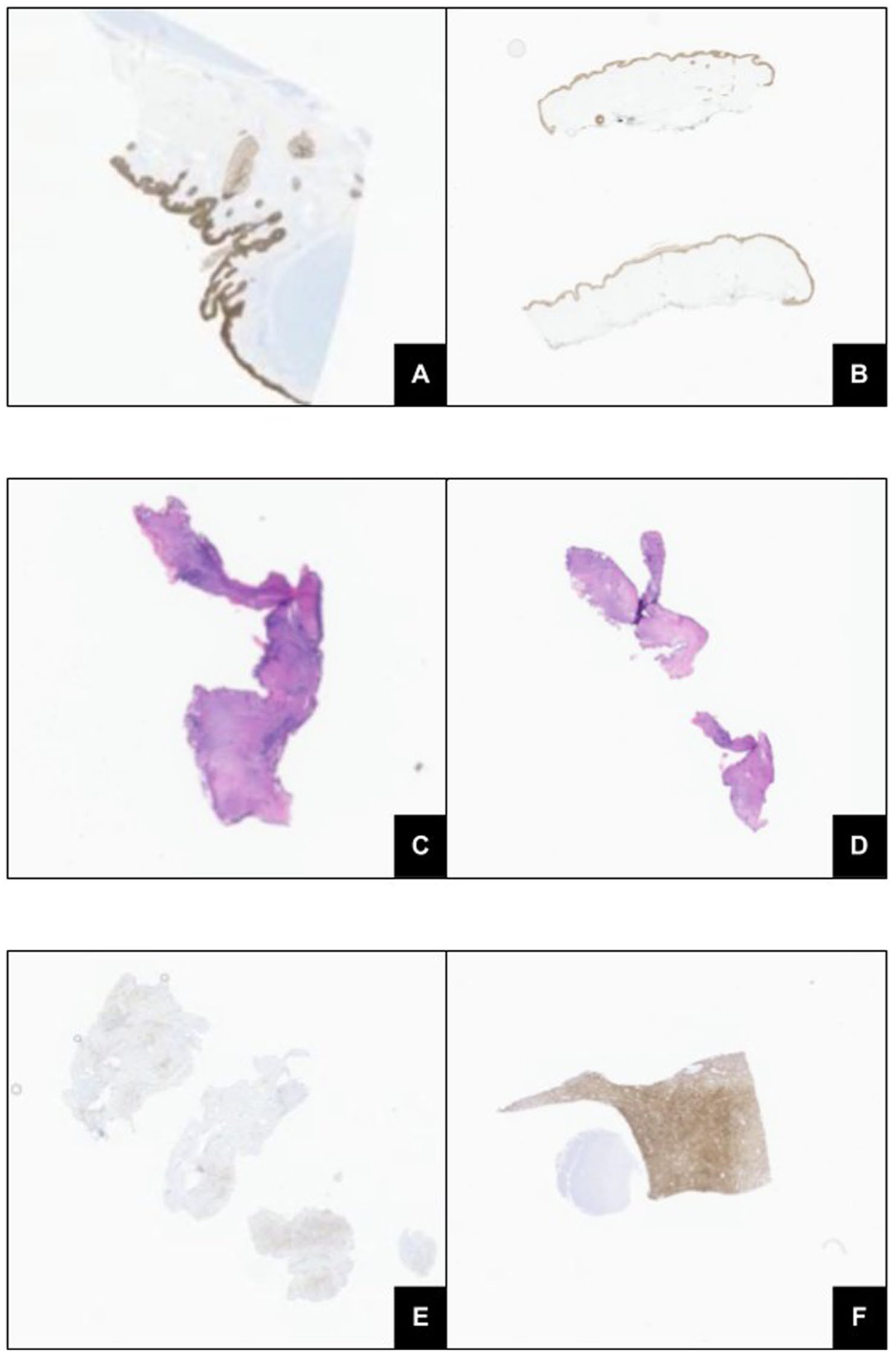
Histopathologic evaluation of subglottic tissue following dilation for grade III subglottic stenosis. (A, B) Keratin staining is negative for carcinoma. (C, D) H&E sections demonstrate submucosal fibrosis with chronic inflammatory infiltrate. (E) Tryptase staining highlights scattered mast cells within the lamina propria. (F) CD117 immunostaining confirms mast cell populations ranging from 10 to 50 cells per high-power field. H&E, hematoxylin and eosin.
Following discharge, the patient was closely followed by a multidisciplinary team, including allergy and ENT, for adjustment of her baseline regimen. She has since required repeated airway dilation for recurrent grade II SGS, including microdirect laryngoscopy with balloon dilation and steroid injection, in May 2025.
Discussion
Pathophysiology of Iatrogenic SGS in MCAS
Scar formation and SGS following intubation are driven by a cascade of pathophysiological events initiated by mechanical injury to the airway mucosa. Endotracheal tube pressure, especially at the cuff, causes mucosal ischemia and ulceration. Injury triggers an inflammatory response, mediated by transforming growth factor beta (TGF-β), hypoxia-inducible factor 1-alpha, vascular endothelial growth factor, basic fibroblast growth factor, and other growth factors important for healing. If the injury is prolonged or repeated, the healing process becomes dysregulated, with excessive fibroblast activation, increased collagen deposition, and ultimately fibrotic scar formation that narrows the airway lumen.2,11 -15
There is evidence that mast cell activation contributes directly to airway remodeling, fibrosis, and submucosal scarring in conditions like asthma and fibrotic pulmonary disease. Mast cell mediators like chymase, tryptase, TGF-β, and cytokines like IL-4 have direct effects on airway structural cells, causing subepithelial fibrosis, abnormal extracellular matrix deposition, and smooth muscle hypertrophy.16 -18 Experimental models show that mast cell stabilizers can attenuate fibrosis, supporting a direct pathogenic role.19,20
Systemic inflammation in MCAS patients can significantly alter tissue healing after airway trauma by promoting a hyperinflammatory environment that disrupts the normal phases of wound repair. In MCAS, release of mast cell mediators is excessive and dysregulated, leading to persistent inflammation, increased vascular permeability, and recruitment of additional immune cells, impairing the resolution of inflammation and delaying progression to the proliferative and remodeling phases of healing. In summary, MCAS may place patients at a greater risk for development of obstructive airway scarring not only because of its
Timing and Risk Factors for Iatrogenic SGS
Prolonged intubation is the most significant risk factor for development of SGS, with risk increasing after 48 to 96 hours of intubation in adults. Risk rises further with multiple, especially traumatic, intubations.24,25 Airway management in patients with anaphylaxis, as occurs in MCAS, is frequently complicated by rapid-onset upper airway edema, which can distort anatomy and make visualization of the glottis challenging, as well as increased urgency of the intervention. Therefore, these patients are more likely to experience traumatic intubation or require multiple attempts.26,27
Multimodal Surgical Management and Outcomes for SGS
Endoscopic procedures like CO2 laser excision and balloon dilation for SGS are considered to be safe and reliable; however, recurrence rates are high, with most patients requiring repeat procedures within 1 to 2 years. 28 Meta-analyses show long-term success in 40% to 82% of cases, depending on lesion characteristics and adjunctive therapies. 29 The addition of intralesional steroid injection, for example, appears to reduce recurrence rates and prolong the surgery-free interval in adult SGS. However, there is some conflicting evidence, and further randomized studies are needed to understand which patients benefit most from this addition.30 -32
In patients with inflamed and friable tissue, as in MCAS, rates of complications such as airway perforation, further mucosal injury, bleeding, and restenosis are elevated with these endoscopic procedures when compared against the general population. Thermal injury from a laser may cause cartilage exposure, chondritis, or airway perforation in the setting of an already-compromised mucosa. Balloon dilation in the setting of friable tissue can cause mucosal tears or submucosal dissection, further increasing the risk of scarring and restenosis. Bleeding is additionally more likely in the setting of inflammation.28,33,34
Role of Allergy/Immunology in Preventing Recurrence
In the present case, the diagnosis of MCAS meets the American Academy of Allergy, Asthma, & Immunology (AAAAI) consensus criteria, with recurrent multisystem symptoms of anaphylaxis, objective biochemical evidence of mast cell activation, and clinical improvement with mast cell-directed therapy. 7 First-line therapies for patients with MCAS include non-sedating H1-antihistamines, H2 antihistamines, leukotriene receptor antagonists, and mast cell stabilizers. These target mast cell mediators and reduce symptom burden; however, there is a lack of large randomized controlled trials specifically addressing anaphylaxis prevention in MCAS. In addition, therapies are not targeted nor standardized for the management of MCAS, and treatment regimens are largely individualized based on patient symptomology. Most evidence for these agents is derived from case series, systematic reviews, and expert consensus.7,23,35
Omalizumab, an anti-IgE monoclonal antibody, has demonstrated efficacy in reducing the frequency and severity of anaphylaxis in patients with MCAS refractory to standard therapy. Some described cases achieved complete prevention of severe reactions that could otherwise necessitate emergency airway management.36 -39 Notably, omalizumab is not FDA-approved for MCAS with recurrent anaphylaxis, but off-label use is supported in refractory cases. 38 There is insufficient direct evidence to confirm that it specifically reduces airway emergency interventions or intubations, to this point. As seen with this patient on adequately dosed omalizumab, recurrent episodes of anaphylaxis with airway compromise can continue to occur.
Considerations for Screening in High-Risk MCAS Patients
No formal surveillance protocols exist for early detection of SGS in MCAS or similar systemic conditions, nor for asymptomatic patients with frequent intubations. In adults with idiopathic or acquired SGS, routine spirometry and pulmonary function tests have been studied as surveillance tools to monitor for recurrence after intervention, but these have not been applied to high-risk asymptomatic populations. 40 That said, in the context of post-intubation laryngeal injury, early endoscopic intervention has been shown to reduce the number of interventions required, shorten tracheostomy dependence, and restore laryngeal function without the need for open reconstruction. These findings support the value of timely diagnosis and intervention in high-risk patients, although the optimal surveillance interval and modality remain undefined. 14 The AAAAI, in its work group report on MCAS, does not address SGS surveillance or recommend airway monitoring protocols for these patients. 7
Limitations and Areas for Future Research
Major gaps in our understanding of MCAS and SGS include the lack of mechanistic clarity on how systemic mast cell activation influences local airway fibrosis after iatrogenic injury, the absence of biomarkers to predict risk or recurrence in this population, and limited translational models to study these interactions. First, while the general pathogenesis of iatrogenic SGS is attributed to aberrant wound healing, fibrosis, and persistent inflammation following injury, the precise molecular mechanisms are incompletely defined, and contributions of systemic diseases such as MCAS to these processes are not well characterized.2,11,13,41,42 Second, the AAAAI notes that the mechanisms of mast cell activation and downstream effects on tissue remodeling and fibrosis in MCAS remain poorly elucidated. 7 Finally, there is insufficient understanding of how genetic predisposition, metabolic factors, and the airway microbiome interact with both local injury and systemic mast cell activation to influence SGS behavior.7,13,43
Conclusion
This case illustrates a rare but life-threatening complication of recurrent intubation in the setting of MCAS. It highlights the dual role of mechanical trauma and systemic inflammation in the development of SGS, emphasizing the need for early airway evaluation and timely surgical intervention in high-risk patients. Greater interdisciplinary awareness and targeted research are essential to improving prevention, surveillance, and outcomes in this vulnerable population.
Footnotes
Acknowledgements
There was no additional support provided to this project. We would like to thank the patient for contributing to our improved understanding of the pathologies affecting the head and neck.
Ethical Considerations
This study was performed under the guidance of the Mayo Clinic IRB and was deemed not to need formal review. Written informed consent was obtained from the patient for the collection and publication of data. Evidence of these processes is available upon request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data underlying the results are provided as part of the article. Due to the nature of the data collected from the patient’s individual medical record, additional data are not available to external requestors.
