Abstract
Background:
Among the various materials used for harvesting in tympanoplasty, the temporal fascia is a commonly-used material due to its accessibility and size. Surgeons can choose to use either the superficial or deep temporal fascia, or both. This study aimed to investigate the influence of the method used to harvest the temporal fascia on postoperative pain experienced by patients.
Methods:
The medical records of 89 patients who underwent canal wall up mastoidectomy with tympanoplasty and ossiculoplasty, performed by a single surgeon between 2021 and 2024, were retrospectively reviewed. In group A, the superficial temporal fascia is harvested. In group B, both the superficial and deep temporal fasciae are harvested together, resulting exposed temporal muscle. In group C, only the deep temporal fascia is harvested and the exposed temporal muscle is sealed by suturing the superficial temporal fascia. Postoperative pain was measured using the visual analog scale (VAS) in 3 groups on postoperative days 1, 3, and 14.
Results:
The baseline characteristics are not significantly different in the 3 groups. VAS scores on postoperative days 1 and 3 showed a significant difference in 3 groups. Group B, which was supposed to have undergone most resection, exhibited the highest VAS scores, followed by group C and group A. The VAS scores on postoperative day 14 showed no difference among the 3 groups.
Conclusion:
Resection of the superficial temporal fascia is thought to be important for postoperative pain due to its rich innervation. Postoperative pain and the properties of the superficial and deep temporal fasciae can be taken into account when otologic surgeons select a harvesting method.
Introduction
Among various harvesting materials in tympanoplasty, temporal fascia is a commonly-used material, due to its accessibility and wide size. Similar to the other parts of the human body, temporal fascia divided into 2 layers, superficial and deep fascia. Otologic surgeons can choose to use either the superficial or deep fascia, or both together. All the choices might have pros and cons. A single surgeon can choose different harvesting methods on a case-by-case basis.
Superficial fascia is located under the skin and the superficial layers of the adipose tissue throughout the body. If a surgeon decides to harvest the superficial fascia, the approach will be easier and less invasive than the deep temporal fascia. This might be the reason why many otologic surgeons choose to use the superficial temporal fascia. Meanwhile, some otologic surgeons prefer the deep temporal fascia, because its histologic characteristic is different from the superficial temporal fascia. It commonly has a more fibrous consistency and is rich in hyaluronan than superficial fascia. 1 If a surgeon decides to harvest the deep fascia, the approach will be more invasive and cause more bleeding. However, due to its fibrous consistency, deep fascia is believed to be more durable than superficial fascia. The thickness and consistency of the fascia varies from patient to patient. If the superficial fascia of a patient is thick and strong enough, it will be sufficient for the graft. If the superficial fascia of a patient is too thin and weak for grafting, deep fascia (with or without the superficial fascia) will instead be harvested. This study focused on the difference in results after different harvesting methods in terms of postoperative pain.
Effective postoperative pain management is important for the patient quality of life and the achievement of a better surgical outcome. As fasciae are a deeply-innervated tissue, 2 the method of harvesting temporal fascia can affect the degree of post-tympanoplasty pain. Most studies focus on postoperative pain management. This study differs from others in that it focuses on intraoperative harvesting methods, resulting in a different degree of postoperative pain.
There are several reports about fascial innervation. These studies investigated fascial innervation using markers such as substance P, protein gene product 9.5, and calcitonin gene-related peptide.2 -7 Fede et al investigated fascial innervation of the lower limb and concluded that the superficial fascia was more densely innervated (33.0 ± 2.5/cm2), whereas the deep fascia had a nerve density of 19.0 ± 5.0/cm2. 8 To the best of our knowledge, no studies have yet been conducted on the innervation of the temporal fascia.
This study investigated the degree of postoperative pain according to different fascia harvesting methods, in an attempt to indirectly determine the innervation of the temporal fascia.
Materials and Methods
Surgical Procedures in Groups A, B, and C
The medical records of 89 patients who underwent canal wall up mastoidectomy with tympanoplasty and ossiculoplasty, performed by a single surgeon between 2021 and 2024, were retrospectively reviewed. Twenty-three participants who received surgeon’s nonroutine surgical procedure were excluded to control for confounding factors. For example, patients from whom tragal cartilage was harvested were excluded because the surgeon routinely harvests conchal cartilage. Cases of dura injury, sigmoid sinus rupture, facial nerve injury, chorda tympani injury, and labyrinthine injury were all excluded.
A postauricular incision and dissection of the subcutaneous tissue to reveal the temporal fascia were commonly performed in all surgeries. In group A, only the superficial temporal fascia was resected and harvested; the deep temporal fascia remained intact (Figure 1). In group B, both the superficial and deep temporal fasciae are resected and harvested, under the condition that the 2 fasciae were attached. This procedure left the temporal muscle exposed (Figure 2). In group C, a minimal incision was made to the superficial and deep temporal fasciae, and the layer between the superficial and deep temporal fasciae was dissected. Then only the deep temporal fascia was harvested. The superficial fascia remained incised, and the temporal muscle was exposed below. (Figure 3A). In the following procedure, the superficial fascia that had been incised was sutured to cover the temporal muscle (Figure 3B). All surgical procedures were performed by a single surgeon to eliminate the impact of surgical skill.
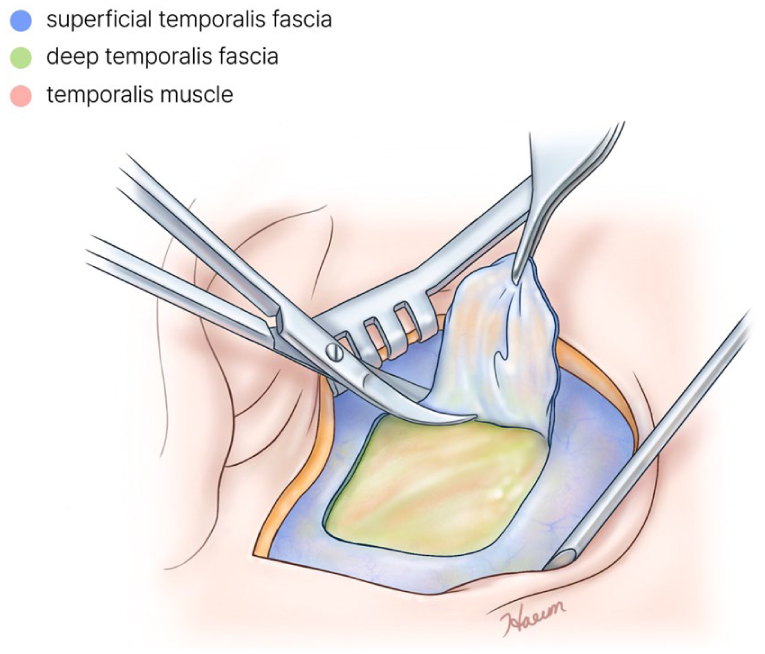
Harvesting method in group A. Only the superficial temporal fascia was resected and harvested.
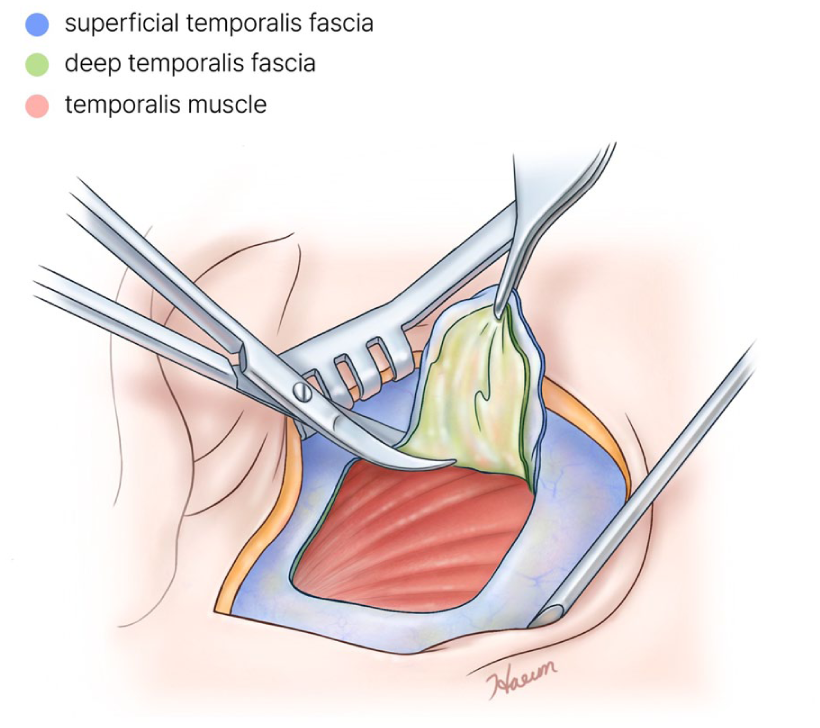
Harvesting method in group B. Both the superficial and deep temporal fasciae are resected and harvested, in condition of the superficial and deep fasciae were attached.
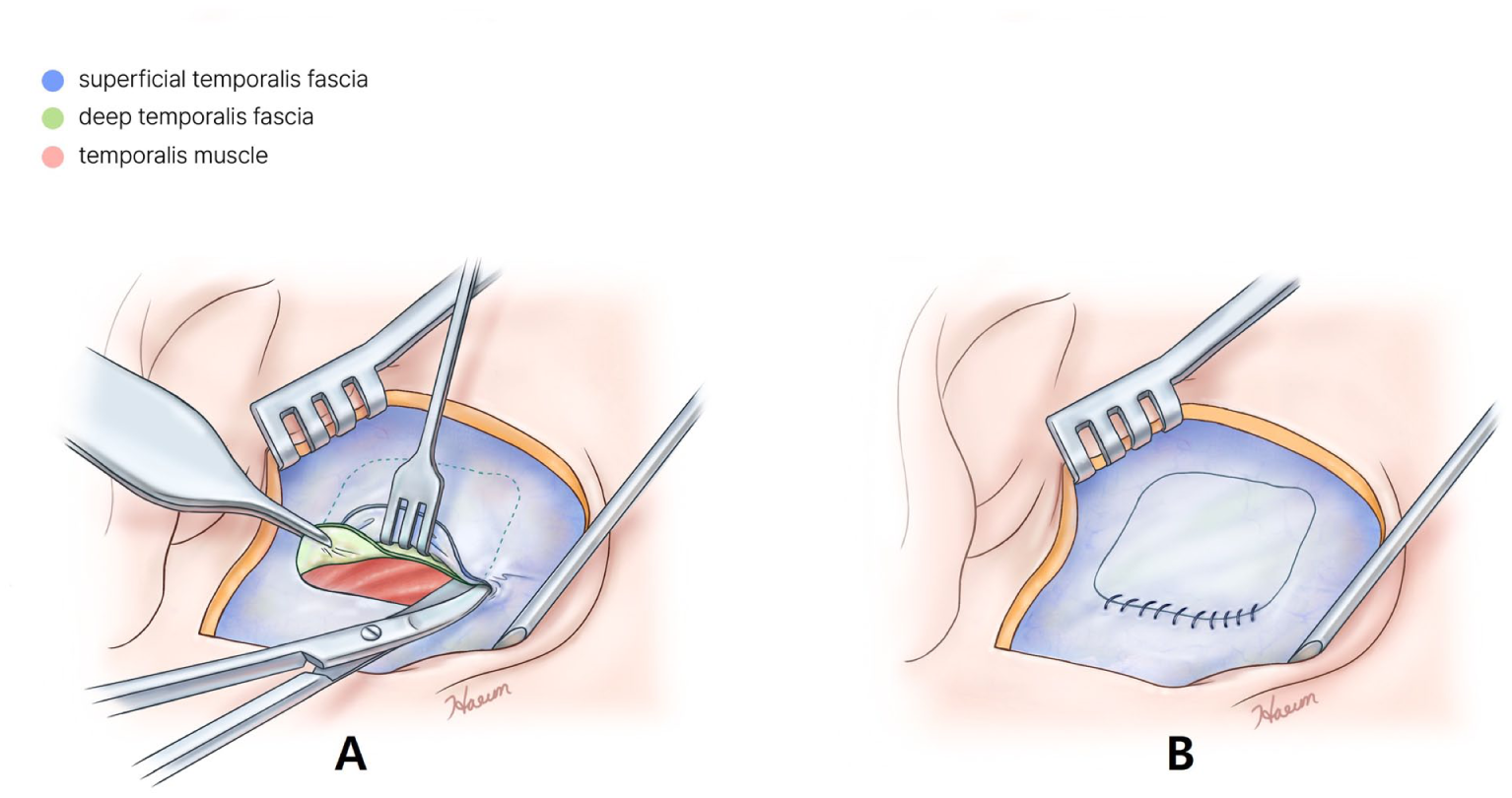
Harvesting method in group C. (A) Only the deep temporal fascia was harvested. The superficial fascia remained incised, and the temporal muscle was exposed below. (B) Superficial fascia that had been incised was sutured to cover the temporal muscle below.
Among 66 participants mentioned above, group B had the least participants, totaling 17. To ensure equal group sizes, participants from groups A to C were randomly selected to also meet a total of 17 participants each.
Postoperative Care and Evaluation for Postoperative Pain
The same analgesic protocol was applied to all 3 groups in the postoperative care setting. Additional analgesics were administered pro re nata (PRN) at the patient’s request. All patients were discharged on the third postoperative day. Subjective postoperative pain was evaluated using the visual analog scale (VAS) on postoperative days 1, 3, and 14. VAS scores were measured by patients using visual analog scale. A single physician checked all the VAS scores to control for observer error.
Statistics
All analyzes were performed using the SPSS 25.0 software (IBM Corp, Armonk, NY, USA). P < .05 was considered statistically significant. The Mann-Whitney test was used to evaluate differences in baseline characteristics, with the exception of age. The Kruskal-Wallis test was used to evaluate differences in VAS scores. Ethics committee approval was obtained from the Inje University Busan Paik Hospital (BPIRB 2022-11-033-004).
Results
Baseline Characteristics
There were no significant differences in baseline characteristics, including age, sex, underlying diseases, and the presence of cholesteatoma between the 3 groups. The incidence of mastoidectomy performed simultaneously with tympanoplasty was not significantly different between the 3 groups. The incidence of PRN analgesics was also not significantly different between the 3 groups. There was no graft failure in the 3 groups (Table 1).
Baseline Characteristics in Groups A, B, and C.
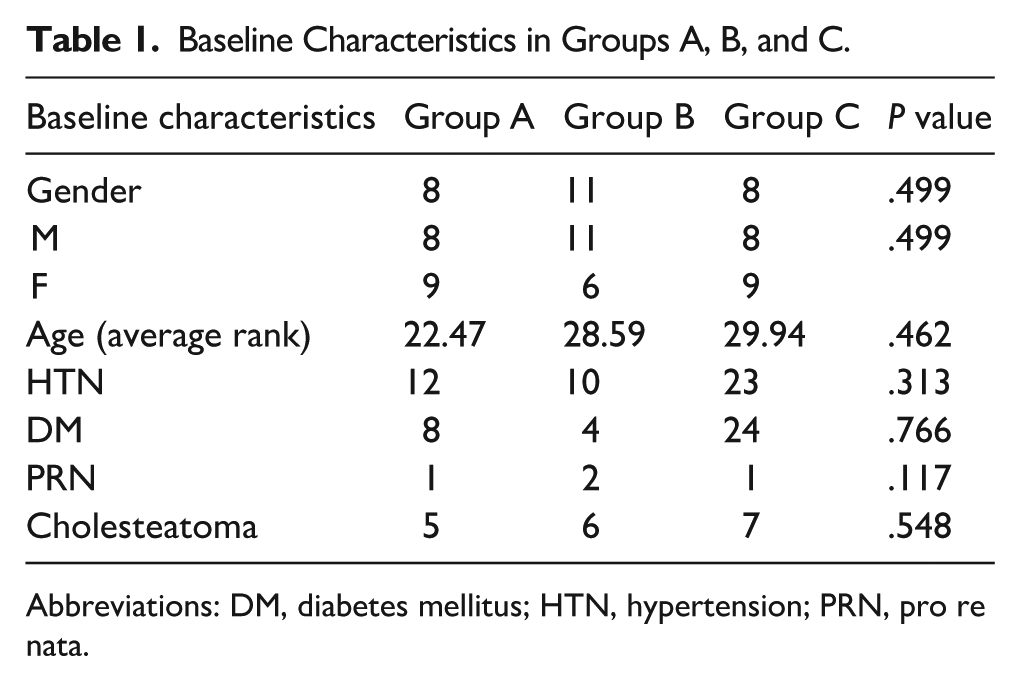
Abbreviations: DM, diabetes mellitus; HTN, hypertension; PRN, pro re nata.
VAS Scores on Postoperative Days 1, 3, and 14
VAS scores on postoperative days 1 and 3 differed significantly between the 3 groups (Table 2). VAS scores on postoperative day 14 showed no difference in 3 groups. Group B experienced the highest level of subjective postoperative pain, followed by groups A and C. This pattern was evident on days 1 and 3. The results of the Mann-Whitney test for the post hoc comparison showed a significant difference between groups A and B on postoperative day 1 (Table 3). The results of the Mann-Whitney test also revealed a significant difference between groups B and C on postoperative days 1 and 3 (Table 4). There was no significant difference between groups A and C on postoperative days 1, 3, and 14 (Table 5).
Kruskal-Wallis Test for Comparing Groups A, B, and C.

Abbreviations: POD, postoperative day; VAS, visual analog scale.
The Kruskal-Wallis test result showed significantly-different VAS scores in postoperative days 1 and 3.
*Statistically significant.
Mann-Whitney Test Result for Post Hoc Comparison Comparing Groups A and B.
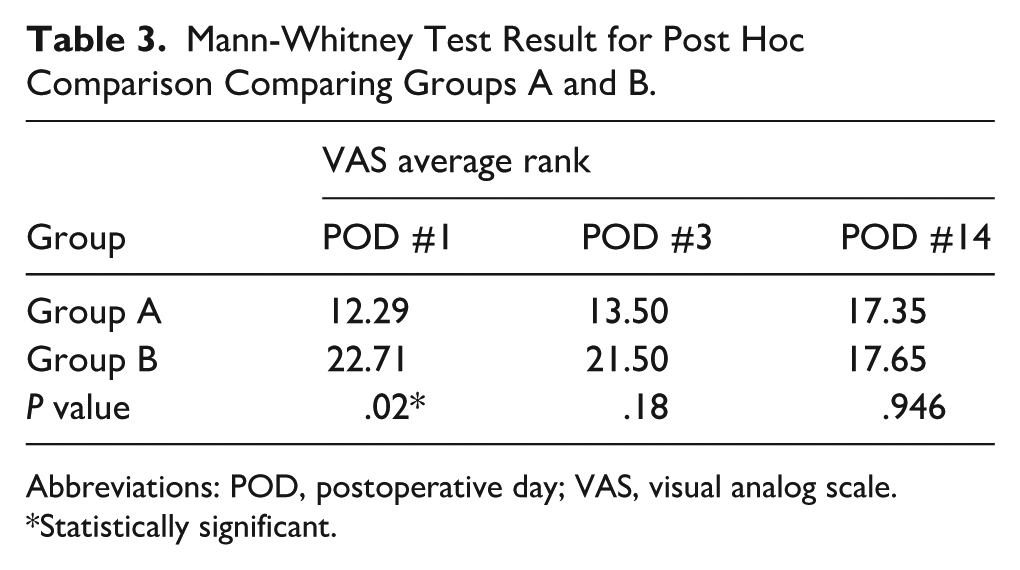
Abbreviations: POD, postoperative day; VAS, visual analog scale.
*Statistically significant.
Mann-Whitney Test Result for Post-Hoc Comparison Comparing Groups B and C.
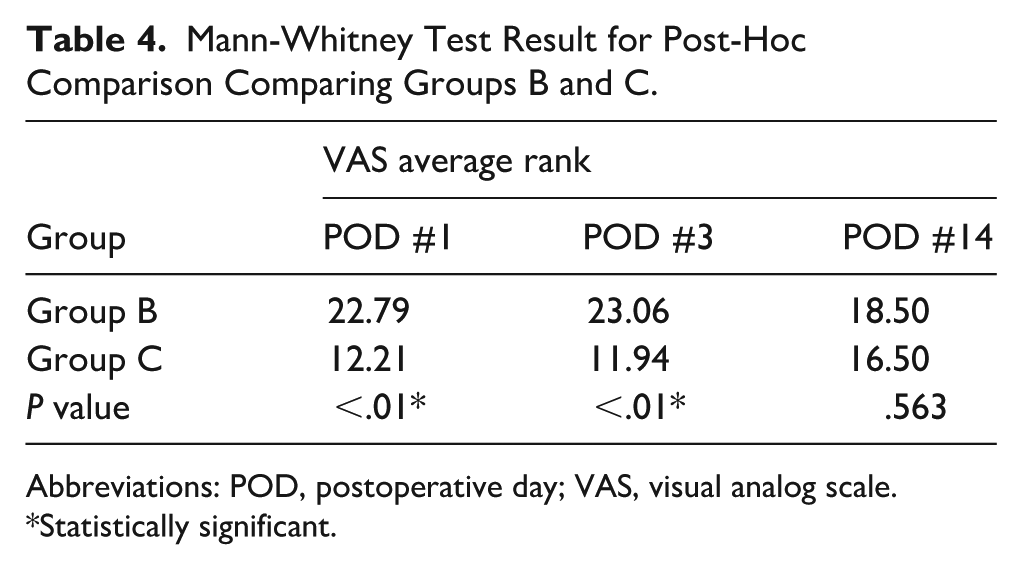
Abbreviations: POD, postoperative day; VAS, visual analog scale.
*Statistically significant.
Mann-Whitney Test Result for Post Hoc Comparison Comparing Groups A and C.
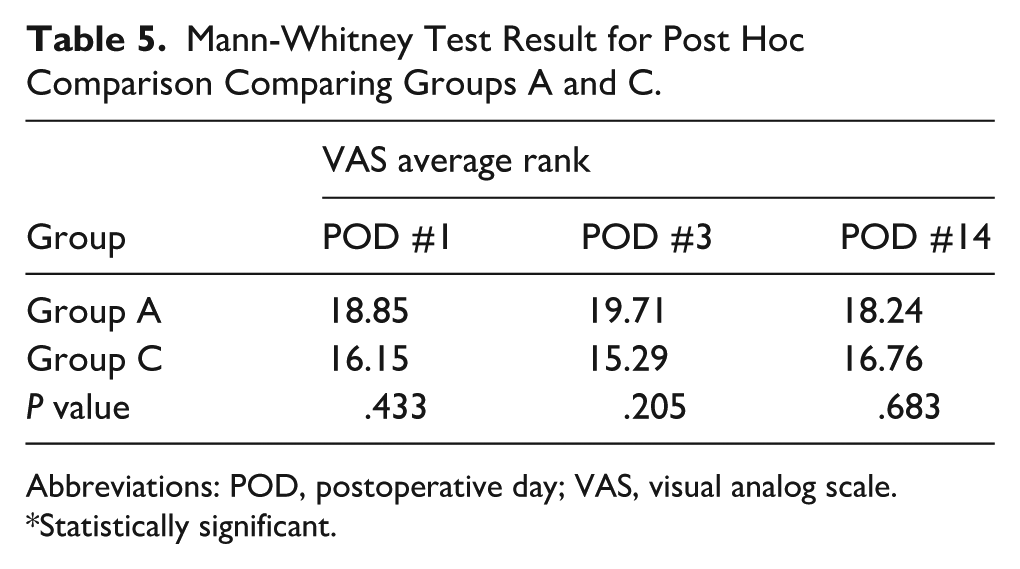
Abbreviations: POD, postoperative day; VAS, visual analog scale.
*Statistically significant.
Discussion
By comparing the VAS scores in groups A and C, we can infer the impact of superficial temporal fascia resection on postoperative pain. In group A, only the superficial temporal fascia is resected; in group C, the deep temporal fascia is resected with a minimal incision in the superficial temporal fascia. Although more incisions and resections were performed in patients in group C, VAS scores in groups A and C did not reveal significant differences. This result is thought to be due to the fact that superficial fascia resection has a greater impact on postoperative pain.
The result of the present study provides information about the innervation of the temporal fascia. The results of the present study are consistent with those of Fede et al, who studied innervation of the fascia of the lower extremities rather than fascia of the temporal muscle. As Fede et al reported that the superficial fascia was found to be more densely innervated than the deep fascia, the results of this study conclude that the superficial temporal fascia is more important in postoperative pain due to its rich innervation.
The superficial temporal fascia lies under the skin and the subcutaneous tissue over the temporal fossa. It is continuous with the superficial musculoaponeurotic system that is inferior to the zygomatic arch. It is 2~3 mm thick. The deep temporal fascia is known to divide into a deep and superficial layer before connecting to the superior part of the zygomatic arch. The superficial temporal fat pad is believed to divide these two layers. 9 However, the two layers of deep temporal fascia have been the subject of debate recently. Li et al investigated 130 cases operated on for zygomaticofacial fractures and concluded that there might not be a “deep layer of the deep temporal fascia.” 10 Plastic surgeons, including Hwang and Kim and Zhao et al specified the deep and superficial layer of the deep temporal fascia during the explanation of temporal anatomy.11,12 The author of the present study agrees with the suggestion of Li et al based on personal surgical experience that the deep layer of the deep temporal fascia could not be identified in most cases. This opinion may differ from surgeon to surgeon and is left as an open question. More studies by many surgeons are needed to reach consensus on the deep layer of the deep temporal fascia.
This study has several limitations. First, the present study included small number of participants. Participants had to meet the criteria of a single surgeon and adhere to the same postoperative analgesic protocol to control for potentially-influential variables, resulting in a small sample size. Second, this result indirectly implies information about the innervation of the temporal fascia without pathological confirmation. Therefore, the difference in postoperative pain does not directly indicate a difference in innervation, since innervation is made up of various nerve receptors, including nociceptors. Third, not all confounding factors responsible for postoperative pain, such as individual susceptibility, preoperative status of middle ear, use of electrocoagulation devices during surgery, could be perfectly controlled. However, these results can provide information on postoperative pain according to the different methods of harvesting the temporal fascia.
Otological surgeons can select the harvesting material and method according to the histological characteristics of the individual patient, the surgeon’s preferred approach, and the available tissue remnants in cases of revision. When selecting the harvest material and method, postoperative pain is an additional factor that this study tentatively suggests should be carefully considered. If the deep temporal fascia is harvested, the suturing method presented in group C may be advisable if the surgeon takes into account postoperative pain.
Conclusion
Group B, which was supposed to have undergone more resection than groups A and C, presented the highest VAS scores on postoperative days 1 and 3. By comparing groups A and C, it can be inferred that the resection of the superficial temporal fascia is important for postoperative pain due to its rich innervation.
Footnotes
Acknowledgements
This research was not supported by any external funding.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
