Abstract
Objective:
Theophylline has been theorized to promote neural olfactory signaling and sensory axonal regeneration. In this study, our objective was to investigate the effect of oral theophylline on the treatment of traumatic anosmia.
Methods:
We enrolled patients who had complained of traumatic complete anosmia. Eligible patients were randomly assigned to 2 groups. In the theophylline group, oral theophylline (400 mg bid) was prescribed for 6 months and patients also underwent olfactory training (OT). In the OT group, patients performed OT only for 6 months. The bilateral and unilateral olfactory function was evaluated using the phenyl ethyl alcohol (PEA) odor detection threshold test and the traditional Chinese version of the University of Pennsylvania Smell Identification Test (UPSIT-TC), before and after treatment.
Results:
A total of 41 patients in the theophylline group and 41 in the OT group completed the study. Before treatment, the PEA threshold for all patients was −1. After 6 months of treatment, the unilateral right PEA threshold improved significantly in patients taking theophylline for 6 months (P = .018), but did not change significantly in patients who only performed OT (P = .317), and was significantly better in patients taking theophylline for 6 months than in those who only performed OT (P = .027). However, UPSIT-TC scores did not improve significantly in either the theophylline or OT group.
Conclusions:
The combination therapy of oral theophylline with OT could have a better effect on traumatic anosmia than OT alone.
Keywords
Introduction
Head trauma is one of the most common causes of olfactory loss. 1 Mechanisms of traumatic olfactory loss include shearing of the olfactory nerves on the cribriform plate and injury to the central olfactory system. 2 Although approximately one third of patients with traumatic olfactory loss may recover spontaneously, the prognosis of traumatic olfactory loss is generally poor.3,4
There is no consensus on the treatment of traumatic olfactory loss. 5 Several drugs, including steroids and zinc, have been used to treat traumatic olfactory loss; however, their effects are still controversial.4,6 Olfactory training (OT) has been used to treat various types of olfactory loss.7 -9 It has been reported to have a mild effect on improving smell thresholds when treating traumatic olfactory loss.10 -12
Cyclic nucleotides have been believed to play a role in the smell and taste function.13,14 Cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) levels in saliva and nasal mucus were found to be lower in patients with olfactory loss than in healthy subjects. 15 Low levels of cAMP and cGMP were considered to cause the appearance of hyposmia and hypogeusia. 16
Theophylline, a phosphodiesterase inhibitor, has been theorized to promote neural olfactory signaling and sensory axonal regeneration by preventing the breakdown of cAMP and cGMP. 17 Henkin et al reported in an open-label controlled trial that oral theophylline was effective in the treatment of hyposmia. 18 However, the etiology of hyposmia in their patients varied, the most common cause being postviral hyposmia, and the results of the testing method used by Henkin et al needed to be carefully interpreted. 19 The dose of theophylline ranged from 200 to 800 mg, with a greater improvement observed with higher doses. 20 In this study, our objective was to investigate the effect of oral theophylline on the treatment of traumatic anosmia.
Patients and Methods
Patients
The flow chart and design of the current study are shown in Figure 1. We collected subjects who complained of a complete loss of their smell function (anosmia) after an episode of head injury between December 2020 and December 2023 at Taichung Veterans General Hospital, Taiwan. Any patient who had a history of loss of olfactory function before head trauma or sinus surgery was excluded from the study. The nasal endoscopy was performed to exclude any patient with sinus disease. If the etiology of loss of olfactory function was suspected to not be caused by head trauma, it was also excluded. Then, patients’ general condition was evaluated. Exclusion criteria included (1) they cannot perform olfactory tests correctly, (2) they were pregnant, (3) they were breastfeeding, (4) they were allergic to theophylline, and (5) they had the history of heart disease, liver disease, or gastric ulcer. The eligible patients received right and left unilateral phenyl ethyl alcohol (PEA) odor detection threshold tests to assess their olfactory function. The definition of anosmia in this study was that the unilateral PEA thresholds for the right and left were −1. Anosmic patients were included in this study. These anosmic patients were then treated with high-dose prednisolone (1 mg/kg/day for 3 days) with tapering, along with zinc gluconate (10 mg 3 times a day) for a period of 1 month. Classic OT using 4 separate bottles of PEA, lemon, eucalyptus, and clove oil was also started. The patients were told to sniff each odorant for 10 seconds, twice a day. 21 After 1 month of treatment, all patients received unilateral right and left PEA odor detection threshold tests and the traditional Chinese version of the University of Pennsylvania Smell Identification Test (UPSIT-TC) to assess their olfactory function. Those whose unilateral PEA thresholds on the right and left remained −1 were enrolled in this study (Supplemental Material).
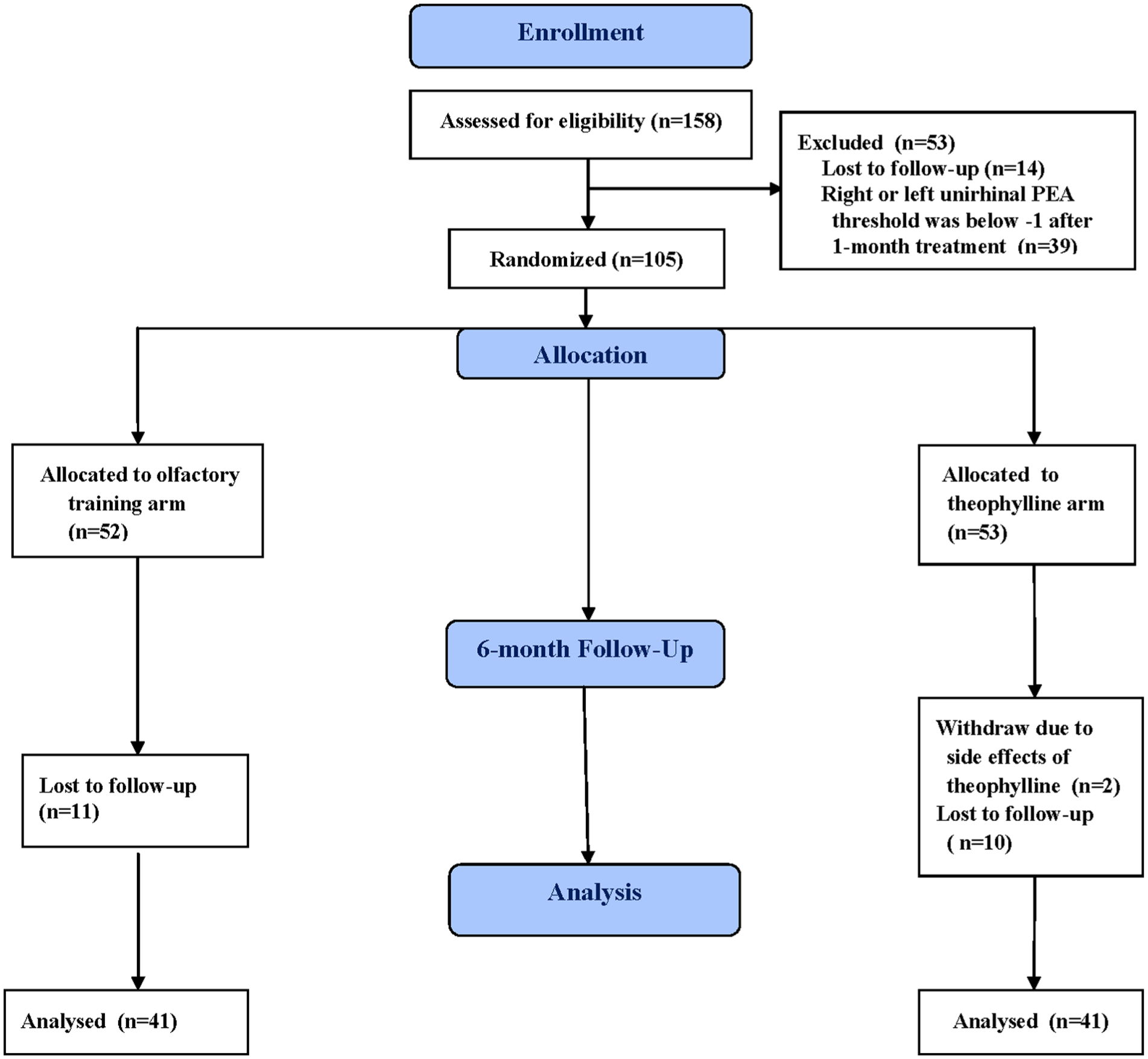
CONSORT Flow chart from enrollment to analysis.
The eligible patients were randomly divided into 2 groups. Randomized assignment to the 2 groups was performed by an independent statistician, and the physicians were blinded to the group allocations. The statistician used the website www.randomization.com, entering the group labels, number of subjects, block sizes, and a seed value to generate the randomization sequence.
The patients in the theophylline group took theophylline (400 mg twice a day) for 6 months because Henkin et al 18 reported that improvement in olfactory function was greater at 600 and 800 mg drug doses than at 200 or 400 mg and continued OT. Those who were assigned to the OT group continued OT for 6 months without any other form of treatment. The patients in the theophylline group were mentioned about the side effects of oral theophylline such as including chest pain, dizziness, fainting, fast, slow, or irregular heartbeat, increase in urine volume, lightheadedness, vomiting, seizure, shakiness. If any side effect occurred, they were told to stop taking theophylline and came back to our clinic.
All patients returned to the clinic to receive another series of the unilateral right and left PEA threshold tests and UPSIT-TC at the end of 6 months of treatment. Patients taking oral theophylline also received blood tests to measure blood theophylline levels. Brain magnetic resonance imaging (MRI) was performed to measure the volumes of olfactory bulbs (OB) following the completion of the 6 month treatment. During the 6 month treatment period, drugs such as oral or nasal steroids, vitamin A, zinc, and antihistamines were not allowed. This study was approved by the Institutional Review Board (I) of the Taichung Veterans General Hospital (protocol code CF20317A, date of approval: December 2, 2020)., with written informed consent obtained from each patient. This clinical trial was registered at Clinicaltrial.gov (registration identifier: NCT07171021).
PEA Odor Detection Threshold Test
The PEA odor detection threshold test used in this study is also commercially available as the Snap & Sniff® olfactory test system the results of which have been shown to be well correlated with those of UPSIT. 22 The PEA threshold test uses different concentrations of a PEA odorant to measure a patient’s odor detection threshold. The PEA odorant is mixed with mineral oil in sniff bottles to prepare different concentrations of the odorant. The strongest concentration is prepared with 1 cup of PEA solution mixed with 9 cups of mineral oil to make a concentration of PEA at 10−1 log vol/vol. Then, the concentration of PEA is diluted with mineral oil in half-log steps until the weakest concentration at 10−9 log vol/vol.
A 2-alternative forced choice single-staircase procedure is used to decide the odor detection threshold. Testing procedures begin with 2 sniff bottles containing either the PEA odorant at 10−6 log vol/vol or just mineral oil alone. The 2 bottles are opened in random order and placed under the patient’s nose. The patient then needs to indicate which of the 2 bottles has the strongest odor. An assumption must be made if the patient is unable to differentiate between the 2 bottles. At the start of the test, the patient must correctly identify the PEA bottles in 5 successive trials before the PEA concentration is decreased to the next lower level; however, an incorrect answer shifts the PEA concentration to the next higher level. After 5 successive correct answers are acquired, only 2 successive correct identifications of PEA bottles are required in order to move the PEA concentration to the next lower level in the following steps. The test is completed when 7 reversal PEA concentrations are obtained, and the PEA threshold is estimated using the geometric mean of the last 4 reversed concentrations. The threshold ranges from −1 to −9.
The PEA odor detection threshold test was performed right and left unirhinally in this study. When the PEA test was performed unirhinally, 1 nostril was blocked using a pad. In this study, the unilateral test for the right and left was performed in random order. The PEA threshold was considered to be improved when the PEA threshold was lower than −1.0 after treatment.
Traditional Chinese Version of the University of Pennsylvania Smell Identification Test
UPSIT-TC is a modified traditional Chinese version of the North American version, with 8 unfamiliar odorants replaced by other odorants. 23 The UPSIT-TC consists of 40 tests as it follows the North American version of the UPSIT. In each test, the brown strip at the bottom of the test page consists of microcapsules of an odorant that are released by scratching the strip with a pencil tip. The patient sniffs the released odorant and selects a response from 4 odor descriptor options that are used to identify the odorant. The UPSIT-TC score is the number of correct responses among the 40 tests taken. The UPSIT-TC score was considered to improve when the TC-UPSIT score increased by 4 or more points after treatment. 24
Brain MRI
Brain magnetic resonance studies were performed using a 1.5 T Excite MRI system (GEMS, Milwaukee, WI, USA) with a quadrature head coil. Routine imaging pulse sequences consisted of axial T1-weighted images, FLAIR images, along with axial and coronal T2-weighted fast spin-echo images. Contrast-enhanced T1-weighted images with axial and coronal sections were acquired. After a sagittal localization scan, 2 to 2.5 mm thick T2-weighted coronal and sagittal (both repetition time [TR] = 5000 ms, echo time [TE] = 106 ms, number of excitation [NEX] = 2, Matrix = 256 × 256) images without an interslice gap were obtained with a 12 cm field of view. Olfactory bulb (OB) volumes were calculated using Mimics Medical 21.0 (Materialise, Leuven, Belgium).
Sample Size and Statistical Analyses
The sample size was calculated using an independent t test using the power analysis program G*Power 3.1 power analysis program (Heinrich Heine University Düsseldorf, Germany). 25 The calculation was carried out according to the mean and standard deviation of the smell test score from a previous OT study. 26 The alpha value was set at .05, and the power value was set at 0.8. Therefore, the sample size was ~39 subjects in each study group.
Data are presented as mean ± standard deviation. The gender of the patients who complete the study was compared between the theophylline and OT groups using the Pearson chi-squared test. The age of the patients who complete the study, and their PEA thresholds, the UPSIT-TC scores and the OB volumes were compared between the theophylline and OT groups using the Mann-Whitney U test. The PEA threshold and UPSIT-TC scores before and after treatment were compared using the Wilcoxon signed rank test. The PEA threshold improvement rates and the UPSIT-TC score after treatment were compared between the theophylline and OT groups using the Pearson chi-squared test. All analyses were performed with IBM SPSS for Windows, version 22.0 (IBM Corp, Armonk, NY, USA). Two-tailed P < .05 were considered statistically significant.
Results
Patients
One hundred and fifty-eight patients with traumatic anosmia were enrolled in this study (Figure 1). After 1 month of high-dose prednisolone, zinc gluconate, and OT treatment, the unilateral right or left PEA threshold was reduced to <−1 in 39 patients, who were then subsequently excluded from the study. An additional 14 patients did not return, leaving the remaining 105 randomly assigned to the theophylline or OT group. After 6 months of treatment, 10 patients in the theophylline group and 11 in the OT group lost follow-up, and another 2 patients in the theophylline group who complained of palpitation after taking theophylline withdrew from the study. Therefore, a total of 41 patients in the theophylline group and 41 in the OT group completed the study.
The demographic data and head trauma profiles of the patients who completed the study are shown in Table 1. There were no significant differences in age and sex between the 2 groups. The interval periods between the head injury and the first hospital visit, the incidence of loss of consciousness, intracranial hemorrhage after the head injury, and the admission and craniotomy ratios were compared between patients taking theophylline and those who only performed OT. There were no significant differences between the 2 groups.
Comparison of Demographic Data and Profiles of Head Trauma between the Theophylline and OT Groups.

Abbreviation: OT, olfactory training.
Data presented as mean ± SD.
Olfactory Function Before Treatment
The results of the PEA test and UPSIT-TC at enrollment are shown in Table 2. The right and left unilateral PEA thresholds and UPSIT-TC scores at enrollment were not significantly different between patients in the theophylline groups who completed the study and those in the OT group who completed the study.
Comparison of Olfactory Function at Enrollment between the Theophylline and OT Groups.
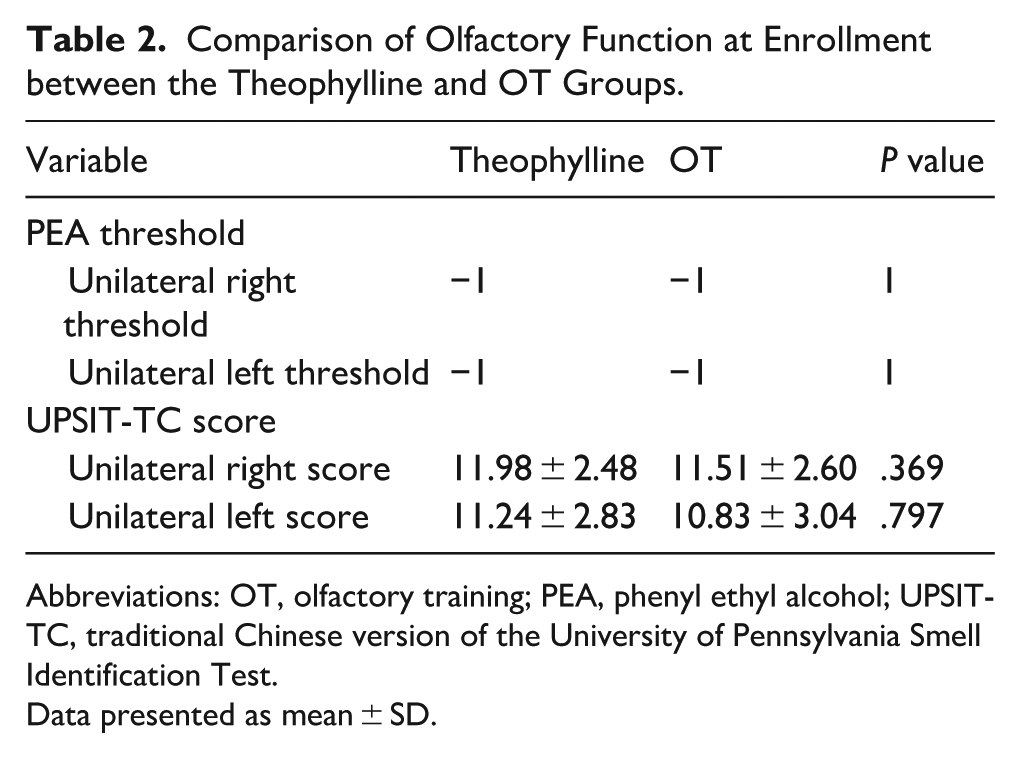
Abbreviations: OT, olfactory training; PEA, phenyl ethyl alcohol; UPSIT-TC, traditional Chinese version of the University of Pennsylvania Smell Identification Test.
Data presented as mean ± SD.
Olfactory Change After Treatment
Table 3 shows the olfactory change after 6 months of treatment in patients who completed the study. The unilateral right PEA threshold improved significantly in patients taking theophylline for 6 months (P = .018), but did not change significantly in patients who only performed OT (P = .317), and was significantly better in patients who took theophylline for 6 months than in those who only performed OT (P = .027). However, UPSIT-TC scores did not improve significantly in either the theophylline or OT group after 6 months of treatment.
Change of Olfactory Function After 6 Month Treatment.
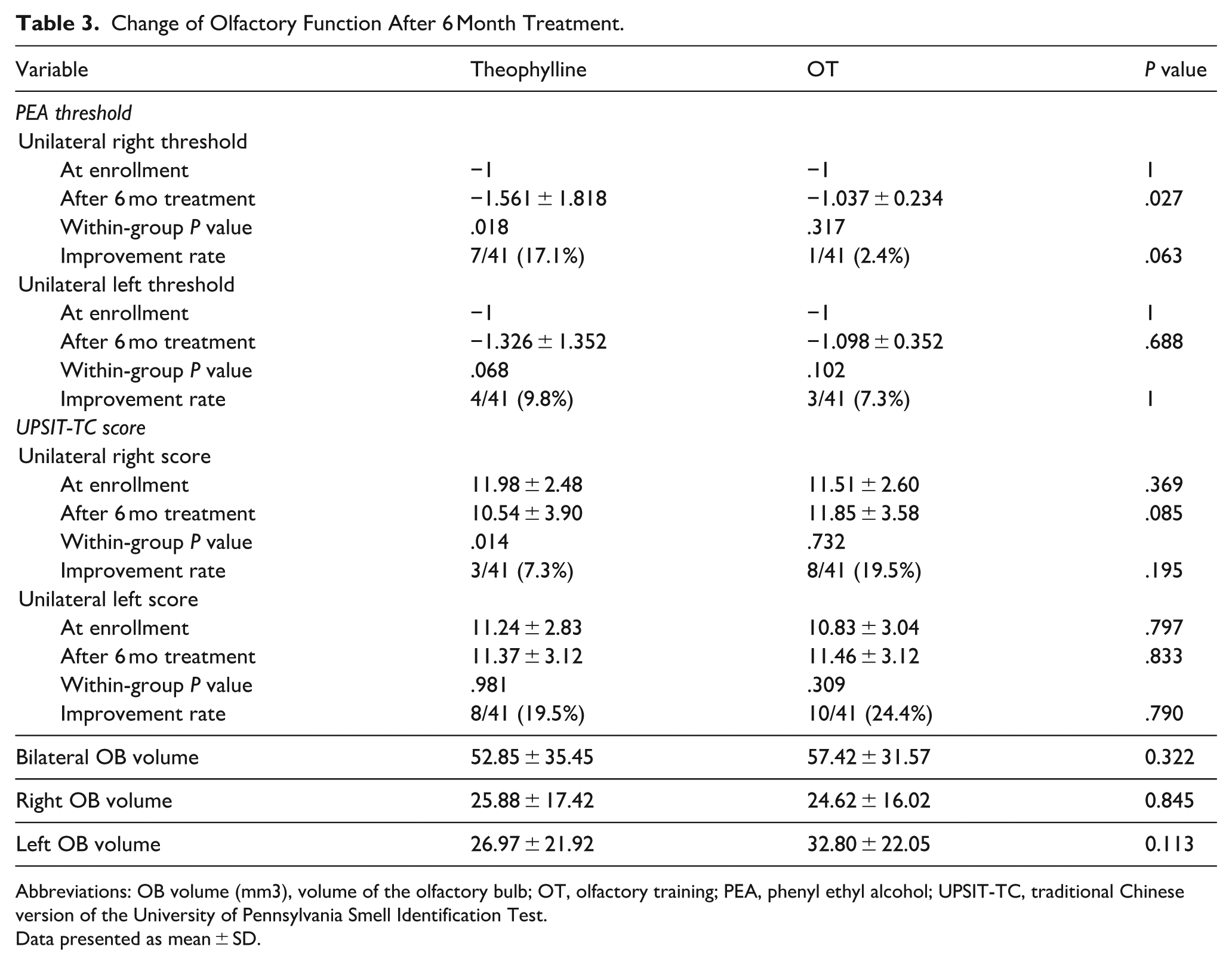
Abbreviations: OB volume (mm3), volume of the olfactory bulb; OT, olfactory training; PEA, phenyl ethyl alcohol; UPSIT-TC, traditional Chinese version of the University of Pennsylvania Smell Identification Test.
Data presented as mean ± SD.
OB Volumes and Serum Theophylline Levels
The OB volumes were measured after the patients finished their 6 month treatment program (Table 3). Bilateral, right, and left OB volumes were not significantly different between the theophylline and OT groups. Serum theophylline levels were examined after theophylline was taken by the patients for 6 months. No patient had an elevated serum theophylline level after taking theophylline for 6 months.
Discussion
Theophylline has been shown to be effective in treating various etiologies of hyposmia, including postviral hyposmia, allergic rhinitis, and head injury. 18 Recently, theophylline was also suggested to treat COVID-19-related olfactory dysfunction.16,27 In this study, we excluded trauma patients whose olfactory function was not anosmic or improved after treatment with steroids and zinc. Therefore, the prognosis for their olfactory function was generally very poor. Our results showed that the combination therapy of oral theophylline with OT could have a better effect on the ability to detect the threshold of smell than OT alone. When high-dose theophylline was prescribed, oral theophylline combination therapy with OT significantly improved the right unilateral PEA threshold after 6 months of treatment. However, the unilateral left PEA thresholds were not significantly between 2 groups. It implies that the effect of oral theophylline was small in the treatment of traumatic anosmia. Similar results have been reported with oral pentoxifylline. 28 Furthermore, the UPSIT-TC score did not increase after theophylline treatment. This indicates that theophylline did not improve the smell identification ability in patients with traumatic anosmia. In an open-label controlled trial, the recognition threshold improved after 800 mg of theophylline daily treatment, but the etiology of olfactory dysfunction in patients varied, with postviral hyposmia and allergic rhinitis the most common. 18
The dosage of theophylline is believed to be related to its effect. Improvement in olfactory function was greater at 600 and 800 mg drug doses than at 200 or 400 mg. 18 Therefore, we prescribed 400 mg of theophylline bid to obtain a better effect. However, oral theophylline has several side effects, such as sleep and gastrointestinal disorders, as well as tachycardia and anxiety. 16 Among our patients who took 800 mg of theophylline daily, several patients complained of palpitation and sleep disturbance, but these side effects were tolerable in most patients. Only 2 patients withdrew from the study due to the side effect of palpitation. However, several other patients were lost to follow-up. The reasons for the loss of follow-up might be due to the side effects of theophylline, but we did not reach these patients to ask for the reasons. Moreover, theophylline has a narrow therapeutic window that requires regular monitoring of blood levels to avoid the potential development of life-threatening events. 29 Serum theophylline levels were measured in all patients taking theophylline after treatment. No elevated theophylline level was found.
Recently, intranasal theophylline irrigation has been used to treat olfactory dysfunction. In a pilot study, it was claimed that intranasal theophylline irrigation was more effective and safer than oral theophylline. 30 However, only 2 patients with head injuries were included in that pilot study. In another randomized clinical trial, the effect of intranasal theophylline irrigation on COVID-19-related olfactory dysfunction was inconclusive. 17 More research is needed to determine whether intranasal theophylline irrigation has a better effect than oral theophylline in the treatment of traumatic olfactory dysfunction.
Limitations
There were several limitations in our study. First, our patients were only loss of smell function due to head trauma. Therefore, the effect of oral theophylline on olfactory dysfunction due to other etiologies such as viral infection was evaluated in this study. Second, the dosage of theophylline has been considered to affect its effect. Although we tried to use high-dose theophylline to treat patients, the optimal dosage of theophylline in the treatment of smell disorder cannot be suggested by this study. Further study is needed to determine the dosage of theophyllinete to acquire a better effect and reduce the adverse effect of oral theophylline at the same time. Additionally, in the study, we only enrolled patients with a complete loss of smell function. It is possible that oral theophylline might have a better effect on patients with partial loss of smell function.
Conclusions
Our results showed that oral theophylline could improve the smell threshold in patients with traumatic anosmia, but it did not improve the smell identification ability. Several minor adverse events occurred with the dose of 800 mg daily.
Supplemental Material
sj-pdf-1-ear-10.1177_01455613251397875 – Supplemental material for The Effect of Oral Theophylline on Traumatic Anosmia: A Prospective, Randomized Clinical Trial
Supplemental material, sj-pdf-1-ear-10.1177_01455613251397875 for The Effect of Oral Theophylline on Traumatic Anosmia: A Prospective, Randomized Clinical Trial by Yi-Fang Chiang and Rong-San Jiang in Ear, Nose & Throat Journal
Footnotes
Ethical Considerations
This study was approved by the Institutional Review Board (I) of the Taichung Veterans General Hospital (protocol code CF20317A, date of approval: December 2, 2020).
Consent to Participate
Written informed consent was obtained from each patient.
Author Contributions
The authors confirm their contribution to the paper as follows: conceptualization, methodology, validation, investigation, resources, and writing—review and editing, supervision, project administration, funding acquisition, R.-S.J.; formal analysis, data curation, and writing—original draft preparation, Y.-F.C. Both authors reviewed the results and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Taichung Veterans General Hospital (grant no TCVGH-1117310C), Taiwan, R.O.C.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Registry and the Registration Number of the Study/Trial
The clinical trial was registered at Clinicaltrial.gov (registration identifier: NCT07171021).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
