Abstract
Objectives:
Endoscopic dacryocystorhinostomy (Endo DCR) is a standard treatment for nasolacrimal duct obstruction, though outcomes may be compromised by intraoperative bleeding and postoperative inflammation. This study evaluated the clinical efficacy of Novacol Fibrillar®, a collagen-based absorbable hemostatic agent, in Endo DCR.
Methods:
A retrospective review was conducted on 357 patients who underwent Endo DCR from January 2018 to September 2024. The Novacol group (n = 168) received Novacol packing, while the Nasopore group (n = 189) received Nasopore. Primary outcomes included anatomical and functional success, operative time, and intraoperative bleeding. Secondary outcomes included granulation, infection, epistaxis, postnasal drip, and revision surgery within 3 months.
Results:
The Novacol group showed higher anatomical (94.6% vs 85.2%, P = .039) and functional (93.5% vs 84.1%, P = .023) success. Intraoperative bleeding was lower (9.5 vs 12.4 gauzes, P = .022), as were rates of granulation (9.5% vs 24.3%, P = .009), epistaxis (5.4% vs 21.7%, P = .028), postnasal drip (6.5% vs 23.3%, P = .032), and revision surgery (2.4% vs 16.4%, P = .041). Infection rates were not significantly different (P = .083).
Conclusions:
Novacol Fibrillar improved surgical outcomes and reduced complications in Endo DCR, supporting its use as an effective hemostatic packing material.
Introduction
Endoscopic dacryocystorhinostomy (Endo DCR) is a surgical technique introduced in various studies for the treatment of nasolacrimal duct obstruction,1 -3 with reported success rates ranging from 54% to 96%, depending on the criteria used (eg, anatomical vs functional) and follow-up duration.1,4 Recurrence of epiphora due to obstruction at the osteotomy site has also been reported, with failure rates varying from 0.6% to 11% in studies applying stricter definitions and longer observational periods.1,5 Obstruction at the osteotomy site may result from adhesions between the ostium and middle turbinate, visible on follow-up endoscopy as fibrous bands, bridging tissue, membranes, or granulomas.1,6
Uncontrolled bleeding during ophthalmic surgery can obscure the surgical field and prolong operative time, potentially contributing to increased tissue manipulation and suboptimal outcomes, including patient discomfort and higher complication risks. 3 This risk is further elevated in elderly patients, especially those on anticoagulant therapy. 3 In Asian patients undergoing Endo DCR, specific anatomical features—such as thicker lacrimal bones and narrower nasal cavities—may predispose them to higher rates of intra- and postoperative nasal bleeding,1,7,8 which can impair healing by promoting excessive inflammatory responses and granulation tissue formation at the osteotomy site.1,9
To address these risks, adequate intraoperative and postoperative bleeding control is essential. Among various hemostatic strategies, nasal packing is frequently employed to provide tamponade, reduce mucosal trauma, and limit inflammatory membrane formation.1,3,10 -13
Several types of nasal packing materials have been used for hemostasis in surgery, including those containing collagen and hyaluronic acid. 14 Among them, collagen has been reported in various studies to facilitate wound healing, hemostasis, and epithelialization.14 -18 However, no previous studies have evaluated the efficacy of collagen-based nasal packing specifically in Endo DCR.
Novacol Fibrillar® (BioCina, Bentley, WA, Australia), a 100% natural collagen-based hemostatic agent, has been widely applied in various clinical settings. In this study, we evaluated its long-term effects in Endo DCR, focusing on its role in hemostasis and prevention of postoperative osteotomy obstruction.
Patients and Methods
Patient Selection Criteria
This retrospective study reviewed the medical records of adult patients (aged ≥18 years) who were diagnosed with unilateral primary nasolacrimal duct obstruction and underwent Endo DCR at Korea University Guro Hospital between January 1, 2018, and September 1, 2024. The study was conducted in accordance with the tenets of the Declaration of Helsinki, and ethics approval was obtained from the Institutional Review Board of Korea University Guro Hospital.
All patients presented with epiphora and underwent a comprehensive lacrimal system evaluation, including slit-lamp examination, fluorescein dye disappearance test (FDDT), probing, and syringing. In addition, dry eye assessments such as the Schirmer test and tear break-up time were performed. When necessary, dacryoscintigraphy was conducted to obtain further information. Only patients diagnosed with primary nasolacrimal duct obstruction were included. Primary nasolacrimal duct obstruction was defined as an acquired obstruction of the nasolacrimal duct without secondary causes such as trauma, tumor, or prior surgery.1,19 The following exclusion criteria were applied:
presence of corneal or conjunctival disease;
punctal or canalicular abnormalities;
previous lacrimal interventions or surgeries;
history of nasal cavity disease or nasal surgery;
age under 18 years; and
follow-up period less than 3 months.
Nasal cavity diseases were defined as conditions that could distort the surgical field or impair postoperative healing, including chronic rhinosinusitis, severe septal deviation, or sinonasal tumors. All surgeries were performed by a single surgeon (S.B.). Patients were divided into 2 groups: group Novacol received intraoperative application of Novacol for hemostasis, while group Nasopore received intraoperative application of Nasopore (Polyganics, Groningen, The Netherlands). To assess the influence of nasal septal deviation, nasal endoscopy was used to classify deviation into 3 grades:
Grade 0: full visibility of the middle turbinate, including the head;
Grade 1: partial obscuration of the middle turbinate; and
Grade 2: no visibility of the middle turbinate.
Both groups were further stratified based on these grades. Patient demographics and medication history, including the use of anticoagulants, antiplatelet agents, and nonsteroidal anti-inflammatory drugs (NSAIDs), were also reviewed. The use of medications related to bleeding was discontinued prior to surgery in consultation with other specialties.
Surgical Procedure
All procedures were performed under general anesthesia. A mixture of 2 mL of 2% lidocaine and epinephrine was injected into the nasal wall after prepacking with bosmin-soaked gauze. Using a 0° 4 mm endoscope (STRYKER, Kalamazoo, MI, USA), a mucosal incision was made along the lateral nasal wall. Osteotomy (8 × 10 mm) was performed using a 45° angled 2 mm Kerrison punch (CareFusion, San Diego, CA, USA). The lacrimal sac was tented using a probe, and a posterior flap was created with a sickle knife. The lateral flap was apposed to the nasal mucosa, while the medial flap was excised.
In group Novacol, Novacol Fibrillar was applied selectively to sites of major intraoperative bleeding that could not be adequately controlled with bosmin-soaked gauze. At each application site, the material was left in place for several seconds to a few minutes until complete hemostasis was achieved, after which the procedure was resumed. The total amount of Novacol used per case was approximately 1.5 to 2.0 g, depending on the severity of intraoperative bleeding. Following bicanaliculus silicone tube intubation (Bika; FCI Ophthalmics, Pembroke, UK), Novacol Fibrillar was additionally packed around the rhinostomy site to support hemostasis in the immediate postoperative period. In the Nasopore group, Nasopore packing was placed around the rhinostomy site instead of Novacol.
Postoperative Care
All patients were hospitalized for 3 days and received systemic antibiotics during admission. Patients were discharged the day after surgery. Postoperative management included instillation of topical antibiotic drops (Tarivid®; Santen Pharmaceutical, Osaka, Japan) and steroid drops (Lotemax® 0.5%; (Bausch & Lomb inc., Bridgewater, NJ, USA)) 4 times daily for 3 months. In addition, starting from the first postoperative day, patients were instructed to perform both nasal irrigation with saline and apply a steroid nasal spray (Nasonex®; Schering-Plough Corporation, Kenilworth, NJ, USA) to support mucosal healing and minimize inflammation.
Follow-up visits were conducted at the 1st, 2nd, 4th, 8th, and 12th week postoperatively to assess tube position, FDDT outcomes, nasal wound status, and symptoms like epistaxis or postnasal drip. The silicone tube was removed at the 2nd month. Postoperative findings, including bleeding, infection, or obstruction due to granulation near the osteotomy site, were recorded via endoscopic imaging and chart review.
Outcome Measures
Postoperative outcomes were divided into primary and secondary endpoints, reflecting surgical success, healing, and patient-reported complications. Primary outcomes included success rate (anatomical and functional), operative time, and intraoperative bleeding. Anatomical success was defined by unobstructed fluid flow on syringing and endoscopic visualization of fluorescein dye. Functional outcomes were evaluated using the Munk score, a validated subjective grading system that quantifies the severity of epiphora based on the frequency of eye wiping. 20 The scoring ranges from 0 to 4 as follows: 0 indicates no epiphora; 1, occasional wiping less than twice daily; 2, wiping 2 to 4 times daily; 3, wiping 5 to 10 times daily; and 4, wiping more than 10 times daily or continuously. A Munk score of less than 1 was considered indicative of functional success. Operative time spanned from nasal mucosal injection to the completion of silicone intubation. Bleeding was estimated by the number of bosmin gauze pieces used.
Secondary outcomes included postoperative granulation, infection, epistaxis, blood-tinged postnasal drip, and revision surgery within 3 months. Epistaxis was defined as nasal bleeding observed endoscopically or reported spontaneously by the patient. Blood-tinged postnasal drip referred to blood-stained mucus in the oropharynx, seen endoscopically or described as a metallic taste or throat sensation.
Statistical Analysis
All statistical analyses were conducted using the SPSS software version 22.0 (SPSS Inc, Chicago, IL, USA). Continuous variables (eg, patient age, operative time, intraoperative bleeding estimated by gauze count) were analyzed using Student’s t-test for univariate comparisons and linear regression for multivariable adjustment. Categorical variables (eg, sex, laterality, medication use, septal deviation grade, anatomical and functional success rates, and secondary postoperative outcomes such as granulation, infection, epistaxis, postnasal drip, and revision surgery) were compared using the chi-squared test for univariate analyses and multivariate logistic regression for adjusted models. A P-value of <.05 was considered statistically significant. A P-value of <.05 was considered statistically significant.
Results
Demographics
A total of 357 eyes from 357 patients were included in this study, with 168 patients in group Novacol and 189 patients in group Nasopore. The demographics and clinical characteristics of the study population are summarized in Table 1. The mean age was 61.3 ± 12.9 years in group Novacol and 60.9 ± 10.5 years in group Nasopore, with no statistically-significant difference between the groups (P = .16; Table 1).
Demographics and Clinical Characteristics of Patients.
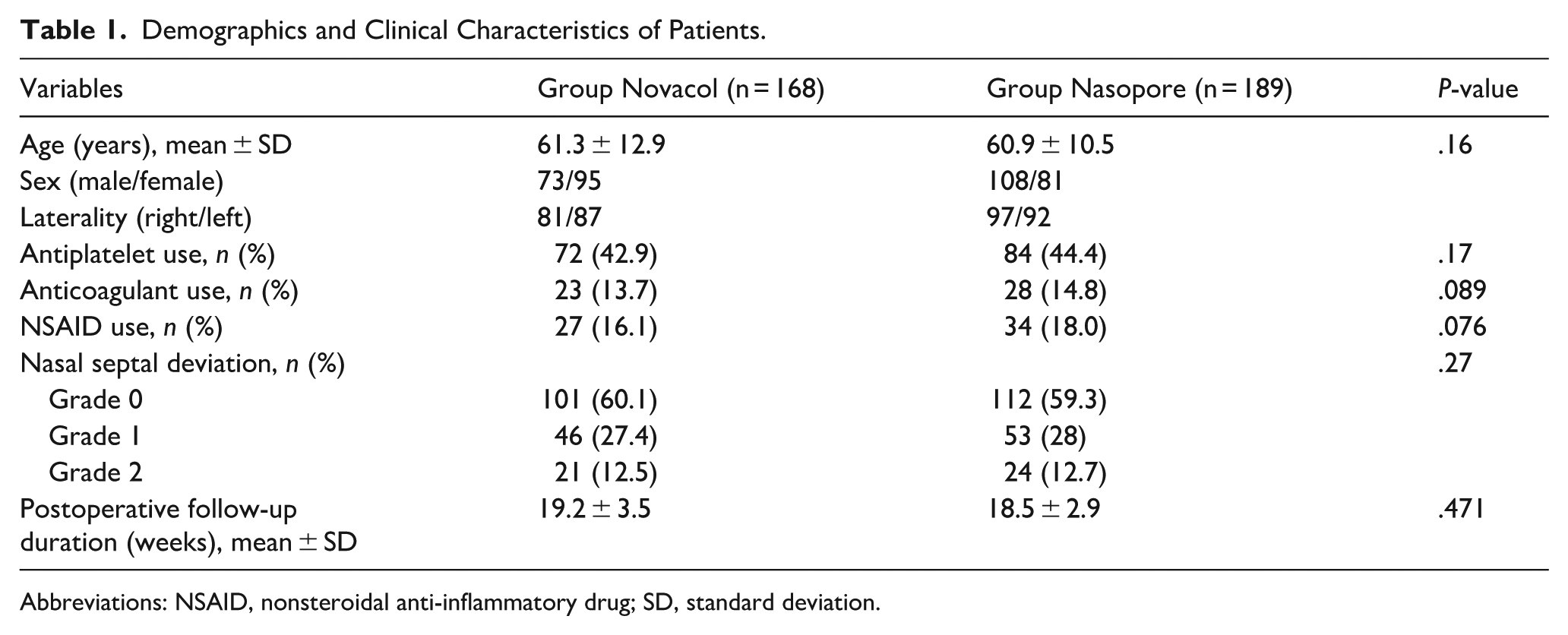
Abbreviations: NSAID, nonsteroidal anti-inflammatory drug; SD, standard deviation.
Regarding medication history, 72 patients in group Novacol and 84 patients in group Nasopore were on antiplatelet therapy. Additionally, 23 patients in group Novacol and 28 in group Nasopore were taking anticoagulants, while 27 and 34 patients, respectively, were using NSAIDs. The use of antiplatelet agents, anticoagulants, and NSAIDs did not differ significantly between the 2 groups (P = .17, P = .089, and P = .076, respectively; Table 1). Classification of nasal septal deviation (grade 0-2) did not significantly differ between group Novacol and group Nasopore (P = .27; Table 1). In addition, the postoperative follow-up period was analyzed by group, with a mean duration of 19.2 ± 3.5 weeks in group Novacol and 18.5 ± 2.9 weeks in group Nasopore, showing no statistically-significant difference (P = .471; Table 1).
Primary Outcomes
When comparing the primary outcomes between the 2 groups, the anatomical success rate was 94.6% (159 eyes) in group Novacol and 85.2% (161 eyes) in group Nasopore, demonstrating a statistically-significant difference (P = .039; Table 2). The functional success rate was also significantly higher in group Novacol, at 93.5% (157 eyes), than at 84.1% (159 eyes) in group Nasopore (P = .023; Table 2). The mean operative time was 25.7 minutes in group Novacol and 24.8 minutes in group Nasopore, with no statistically-significant difference observed between the groups (P = .13; Table 2).
Comparison of Primary Surgical Outcomes between Group Novacol and Group Nasopore.
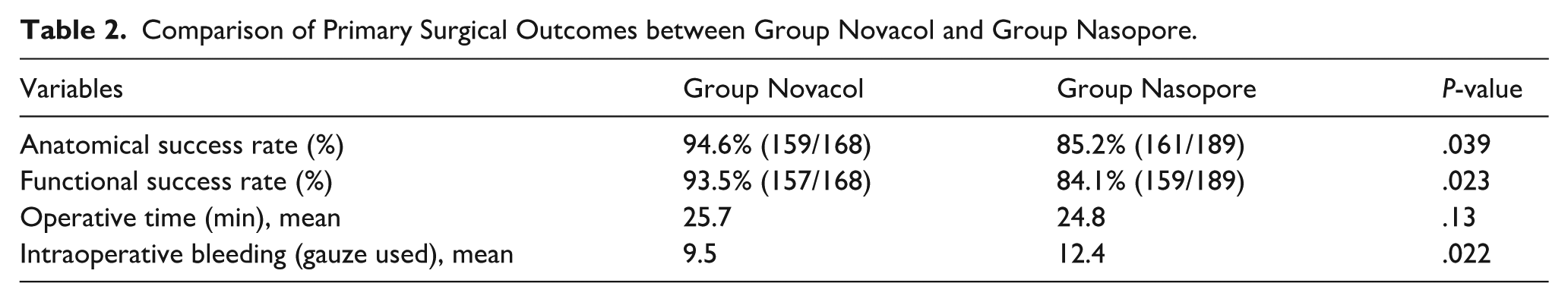
Intraoperative bleeding, estimated by the number of bosmin-soaked gauze pieces used per surgery, revealed a significant difference: Group Novacol required an average of 9.5 gauze pieces per procedure, whereas group Nasopore required an average of 12.4 (P = .022; Table 2).
Secondary Outcomes
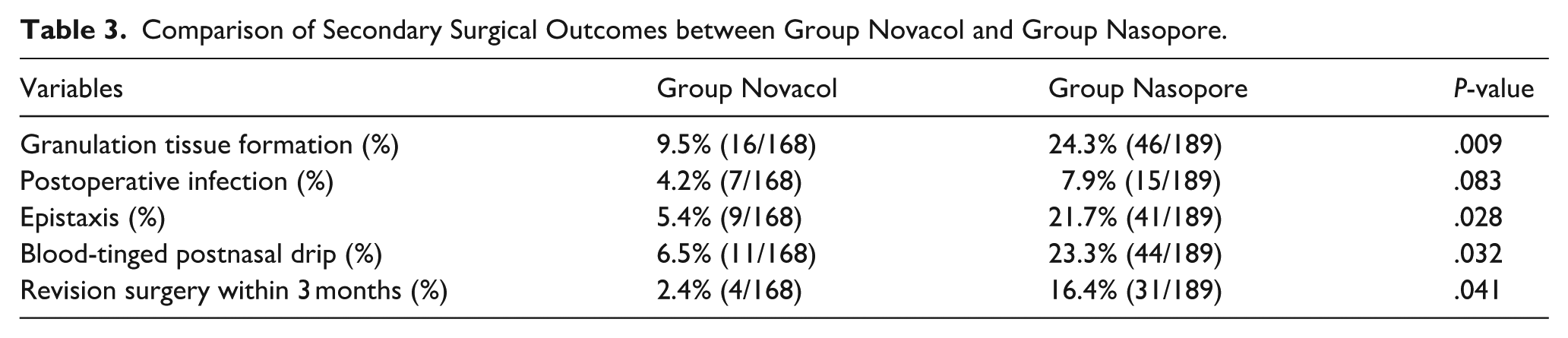
During the postoperative follow-up period, granulation tissue formation at the surgical site was observed in 9.5% (16 eyes) of group Novacol and 24.3% (46 eyes) of group Nasopore, demonstrating a statistically-significant difference (P = .009). The incidence of postoperative infection was 4.2% (7 eyes) in group Novacol and 7.9% (15 eyes) in group Nasopore, which did not reach statistical significance (P = .083).
Patient-reported postoperative epistaxis occurred in 5.4% (9 eyes) of group Novacol and 21.7% (41 eyes) of group Nasopore, showing a significant intergroup difference (P = .028). Similarly, the proportion of patients reporting blood-tinged postnasal drip was significantly lower in group Novacol (6.5%, 11 eyes) than in group Nasopore (23.3%, 44 eyes; P = .032). Lastly, the rate of revision surgery at the 3rd month postoperatively was significantly lower in group Novacol (2.4%, 4 eyes) than in group Nasopore (16.4%, 31 eyes; P = .041; Table 3).
Comparison of Secondary Surgical Outcomes between Group Novacol and Group Nasopore.
Discussion
This study assessed the efficacy of Novacol, a collagen-based hemostatic agent, during Endo DCR. At the 3rd month, Novacol demonstrated significantly-higher anatomical and functional success than Nasopore. It was also associated with reduced intraoperative bleeding, granulation, epistaxis, postnasal drip, and revision rates.
Nasal packing materials play a key role in Endo DCR, with endoscopic sinus surgery literature underscoring their importance across broader nasal procedures.1,11,21 In both Endo DCR and sinus surgeries, postoperative bleeding, synechiae, and granulation at the surgical site are major causes of failure and morbidity.11,21 Nasal packing promotes hemostasis, prevents lateral nasal wall–middle turbinate adhesion, and limits inflammatory responses that hinder mucosal healing.21,22 These complications may be more pronounced in Asian populations due to narrower nasal cavities and thicker bony structures, complicating surgical access and visualization. 11 In anatomically-narrow nasal spaces, appropriate packing is essential to maintain a clear field, control bleeding, and support tissue healing.11,22 Various materials with distinct physical and biochemical properties have been developed to optimize hemostasis, patient comfort, and mucosal preservation.1,21,22 Synthetic foams, polyvinyl acetate, and carboxymethylcellulose-based dressings have been investigated in Endo DCR and sinus surgeries in recent studies.1,11,21,23
Nasopore (Polyganics), an absorbable polyurethane foam, has demonstrated favorable outcomes in several studies.1,11 In endoscopic endonasal dacryocystorhinostomy, Nasopore showed higher anatomical (90.5%) and functional (89.3%) success than Merocel (76.3% and 75.9%), with less granulation and postoperative bleeding. 1 Across endoscopic nasal surgeries including DCR and sinus procedures, Nasopore has shown better patient comfort than Merocel, with less in situ pain and bleeding upon removal, while preserving mucosal healing and synechiae prevention. 1
Merocel (Medtronic Xomed, Minneapolis, MN, USA) is known for reliable hemostasis through mechanical compression and remains a standard for managing intraoperative bleeding in endoscopic nasal surgery. 11 Its sponge-like structure provides immediate tamponade and physical stabilization of the mucosa. 11 In Endo DCR, Merocel showed a lower early-postoperative granulation rate than Nasopore, based on multivariable analysis. 11 However, due to its nonabsorbable nature, Merocel requires manual removal postoperatively, which can lead to patient discomfort, mucosal trauma, and secondary bleeding. 24 Comparisons with absorbable alternatives like Posisep show Merocel causes more postoperative discomfort, including greater pain and bleeding upon removal. 24 Patient-reported outcomes also favor Nasopore over Merocel due to less in situ pain, nasal pressure, and greater satisfaction, while maintaining comparable mucosal healing and synechiae prevention. 1 While Merocel remains effective for hemostasis, it is being supplanted by absorbable materials that improve patient comfort and maintain comparable or superior outcomes.
Guardcel (Genewel Co, Ltd, Seongnam, Korea), a carboxymethylcellulose-based bioresorbable nasal dressing, has also been introduced as a promising packing material. 23 Guardcel has been reported to reduce postoperative granulation tissue and epistaxis in Endo DCR. 23 As an absorbable material, Guardcel avoids painful removal, promotes wound healing, and has shown lower rates of epistaxis and granulation than controls in Endo DCR. 23 The study also reported favorable functional outcomes with Guardcel, indicating its potential to enhance mucosal healing and reduce osteotomy-related complications. 23
Advances in biomaterials have highlighted material-tissue interactions in regulating inflammation, supporting mucosal repair, and enhancing recovery.21,22 Ideal hemostatic biomaterials are characterized by rapid blood absorption, biocompatibility, and the capacity to modulate inflammatory pathways.21,22 Functional biomaterials such as collagen, hyaluronic acid, and carboxymethylcellulose exhibit hemostatic efficacy and aid epithelial repair, reduce postoperative discomfort, and support recovery.21,22 Natural biomaterials such as collagen, hyaluronic acid, carboxymethylcellulose, and alginate offer combined hemostatic, anti-adhesive, and regenerative effects.21,22 These properties enhance healing in nasal surgeries, such as sinus and lacrimal procedures, supporting their clinical integration.21,22 Collagen, in particular, has demonstrated intrinsic procoagulant and tissue-regenerative properties through several experimentally- and clinically-supported mechanisms.21,22 Collagen promotes platelet aggregation and thrombin generation via coagulation cascade activation, contributing to rapid hemostasis. 22 Its fibrillar structure also traps red blood cells and provides tamponade, accelerating clot formation. 22 In addition, collagen also promotes mucosal healing and reduces local inflammation by acting as a biocompatible scaffold for epithelial and stromal cell migration. 22 Evidence from nasal and sinus surgery studies indicates that functional biomaterials like collagen aid epithelial repair, reduce synechiae, and modulate inflammation.21,22 These combined effects justify investigating collagen-based materials in mucosal surgeries like Endo DCR, where hemostasis and mucosal preservation are vital for long-term success.
To our knowledge, this is the first study to evaluate a collagen-based packing material in Endo DCR. Unlike Merocel, Novacol is fully absorbable, avoiding discomfort and trauma associated with removal. Compared with Nasopore, Novacol achieved comparable or better outcomes in anatomical success, granulation, and revision rates, aligning with previous evidence on collagen-based agents in other surgical contexts.21,22 Collagen has been shown to promote hemostasis, reduce inflammation, and support epithelial regeneration.21,22 Specifically, collagen scaffolds promote platelet aggregation, stabilize the coagulation cascade, and limit foreign-body inflammation through their biocompatibility. 22
Collagen-based hemostatic agents are well-documented for their rapid onset of hemostatic activity in diverse clinical and surgical settings. In general surgical applications, absorbable collagen sheets have achieved effective hemostasis within an average of 2.2 minutes, even in patients with coagulopathies. 25 Similarly, in percutaneous interventions such as femoral artery closure, bovine collagen plugs have demonstrated complete hemostasis in approximately 2.3 minutes. 26 Notably, in a preclinical rat liver hemorrhage model, a collagen fiber composite sponge achieved hemostasis within approximately 25 seconds, 27 underscoring its prompt coagulative efficacy in highly-vascularized tissues. These findings support the clinical plausibility of using collagen-based materials like Novacol for efficient intraoperative bleeding control in Endo DCR, where precise hemostasis must be achieved rapidly within the limited confines of the nasal cavity.
These findings support the hypothesis that collagen-based packing, such as Novacol, improves Endo DCR outcomes by reducing bleeding and postoperative inflammation, thereby promoting regeneration and lowering complication rates. Rapid hemostasis may also slightly reduce operative time.
This study has several limitations. First, its retrospective design may introduce selection bias and reduce control over confounding factors. Second, the 3 month follow-up may be insufficient to assess long-term outcomes or delayed complications. Third, while Nasopore served as the control, other packing types such as HA-based gels or collagen-HA composites were not included, limiting generalizability. Thus, further randomized trials with longer follow-up are needed to validate the comparative efficacy of Novacol. Furthermore, the absence of a control group without nasal packing precludes assessment of whether any form of packing is truly necessary in mucosa-sparing Endo DCR procedures. In light of growing interest in the potential benefits of avoiding nasal packing—such as improved patient comfort and accelerated mucosal recovery—future randomized controlled trials are warranted to directly compare outcomes between packing and non-packing strategies. Lastly, while the number of bosmin-soaked gauze pieces was employed as a semi-quantitative indicator of intraoperative bleeding, this method has inherent limitations. In endoscopic procedures, blood is frequently aspirated via suction before accumulation can be visually assessed, and the constrained operative field—particularly in Asian patients with narrower nasal anatomy—further complicates accurate estimation of blood loss. 28 Accordingly, while gauze count constitutes a practical and consistently-recorded intraoperative parameter, it may reflect an estimated rather than exact blood loss volume, particularly in the context of endoscopic surgery.
In conclusion, this study provides initial evidence supporting collagen-based packing in Endo DCR. Novacol yielded favorable surgical outcomes, improved bleeding control, and fewer complications, with the advantage of absorbability and no need for removal. Further research is warranted to confirm these results and compare with other nasal packing materials.
Footnotes
Ethical Considerations
This study was approved by the Institutional Review Board of Korea University Medical Center in Seoul, Korea. All data collection and analysis efforts were conducted in accordance with the tenets of the Declaration of Helsinki.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Author Contributions
H.K.L.: Conception and design of the study; conduct of study; collection and management of data; data analysis and interpretation; preparation, review, and approval of the manuscript. S.B.: Preparation, review, and approval of the manuscript. S.B.: contributed to the manuscript as a corresponding author.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
