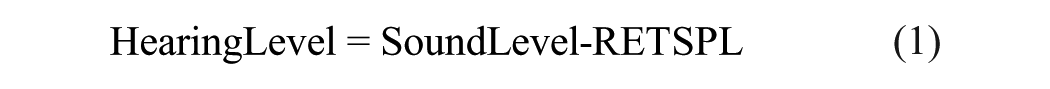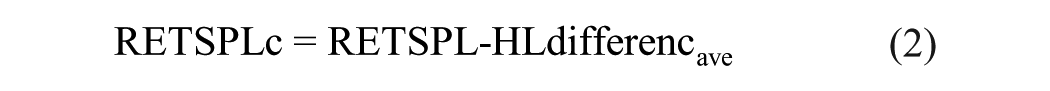Abstract
Purpose:
Hearing loss is a major global health challenge, and pure-tone audiometry remains the gold standard for assessing auditory thresholds. However, conventional clinical audiometry requires specialized sound-proof booths, which limit its accessibility in resource-constrained settings. To address this limitation, we developed and evaluated a portable audiometry system, designated as the E200, incorporating active noise control (ANC) technology with integrated supra-aural earphones.
Methods:
In this 2-phase study, reference equivalent threshold sound pressure level calibration was first performed on 183 volunteers comparing E200 to conventional clinical pure-tone audiometric testing. Subsequently, we prospectively enrolled 114 participants with diverse otologic pathologies from tertiary otology clinics to evaluate the validity of the E200 audiometric system.
Results:
Data from 228 ears were collected from 114 participants, with 7 frequencies tested/ear, resulting in 1596 sets of data. The two one-sided test equivalence analysis demonstrated that all tested frequencies met the equivalence criterion at the ±5 dB margin. Pure-tone average (PTA) was derived from the arithmetic mean of air-conduction thresholds at standard speech frequencies (0.5, 1, 2, and 4 kHz), with hearing loss severity classified according to WHO (2021) criteria: normal (<20 dB hearing level [HL]), mild-to-moderate (20-<50 dB HL), and moderate-to-severe (≥50 dB HL). Both Bland–Altman plots and Cohen’s κ statistics demonstrated good agreement with the PTA results.
Conclusions:
This ANC-integrated portable audiometer demonstrates promising potential for hearing screening outside conventional sound-proof booths; however, its long-term clinical value and practical applications require further investigation and validation.
Keywords
Introduction
The 2021 World Report on Hearing reveals that over 1.5 billion people globally experience some degree of hearing loss, representing ~20% of the world’s population. 1 With the projected increase in the prevalence of hearing loss, it poses a growing societal and economic burden.2 -4 Conventional pure-tone audiometry remains the gold standard for diagnosing hearing loss and is the primary screening method for individuals aged 4 years and above.5 -7 Typically, audiometric testing is conducted by a trained audiologist within a sound-proof booth, limiting portability. 8 Access to hearing healthcare is often limited by factors such as geographic location, availability of providers, and socioeconomic status. An emerging solution to this public health issue is to utilize portable or automated audiometry based on tablet or mobile phone apps when conventional audiometry is not available. Several studies have demonstrated that this method is effective and reliable for hearing screening in children and adults.9 -19 The development of portable audiometers addresses a persistent limitation in audiological care by enabling reliable hearing assessments outside conventional sound-proof environments.
With the advancement of noise reduction technology, it has become an increasingly critical focus in the development of portable medical audiometers. However, it is noteworthy that while active noise control (ANC) maintains a signal correlation exceeding 82% in low-noise environments (<60 dB SPL, Sound Pressure Level), its performance significantly deteriorates in high-noise scenarios (>70 dB SPL), exhibiting ~20% signal fidelity attenuation. This limitation has driven researchers to investigate hybrid noise suppression strategies. Based on the aforementioned context, Wuhan Douting Technology Co, Ltd has developed a portable audiometer, (Wuhan, China) the E200 (hereafter referred to as E200). The E200 medical audiometer integrates ANC technology, passive noise control (PNC) physical insulation, and digital signal processing (DSP) noise reduction in a nested and synergistic manner. After calibration, the device ensures effective transmission of test tones at specified frequencies while achieving robust suppression of extraneous noise outside the target frequency range. The output frequency range of E200 matches that of conventional clinical pure-tone audiometric testing (abbreviated as CA hereafter), ranging from 125 to 8000 Hz. The output hearing level (HL) range of the device extends from −10 to 105 dB HL. The E200 also includes a masking functionality to prevent inaccurate cross-hearing test results. Currently in the investigational stage, this prototype device has been provided by the manufacturer at no cost for clinical trial purposes.
Participants and Methods
Participants
This study was conducted at the Department of Otolaryngology of a tertiary referral center between March and December 2024. Patients who consented to undergo conventional and E200 audiometry were enrolled, and all participants provided written informed consent. Before the audiometric assessments, all participants underwent otoscopy to examine their tympanic membrane condition, and any excess earwax or foreign objects in the ear canal were removed. This study included participants older than 6 years who were capable of cooperating with conventional pure-tone audiometry and E200 audiometry. Individuals with acute otitis media, blocked external ear canals that could not be cleared, or those with mental illness or other conditions preventing cooperation with audiometry were excluded from this study.
Conventional Clinical Pure-Tone Audiometric Testing
Participants underwent CA using the Interacoustics AC40 (Denmark) with TDH39 headphones (Telephonics, Farmingdale, NY, USA). We conducted CA in a standard sound-proof booth (ambient noise <30 dB) and evaluated the frequency range of 125 to 8000 Hz. Before audiometry, the participants were asked to identify which ear had better hearing, and the ear with better hearing was tested first. This study employed a modified version of the Hughson-Westlake approach, known as the “up-5 down-10 technique.” 8 The pure-tone sound level was initially introduced at an estimated higher threshold and reduced by 10 dB HL after a correct response from the participant. If the participant did not respond correctly or at all, the sound level was increased by 5 dB HL. 12 When the air-bone difference in both ears is ≥40 dB, contralateral masking was applied to correct the cross-hearing.20,21
Calibration of the E200
A key challenge in obtaining accurate HLs is calibrating the electroacoustic system when using nonstandard earphones. 22 A standard calibration procedure involves 2 steps: (i) using a standard ear simulator or acoustic coupler with a sound level meter to calibrate the output sound level of a specific earphone in dB SPL and (ii) converting the sound level (in dB SPL) to the HL (in dB HL) using the formula:
where reference equivalent threshold sound pressure level (RETSPL) represents the average SPL corresponding to the audiometric 0 level (ie, 0 dB HL) for a group of otologically normal subjects. 23 The selection of the calibrator and earphone can influence the RETSPL values. The ANC headphones (RAN-3500) used in this study differ from standard earphones. To achieve HLs consistent with those obtained from a clinical audiometer using standard earphones in a sound-proof booth, a feasible approach is to adjust the RETSPL of the RAN-3500 by compensating for the average HL difference measured with standard earphones, as shown in the following equation24,25:
where RETSPLc represents the corrected RETSPL, and RETSPL denotes the acoustically measured RETSPL of the RAN-3500. HL differenceave is the average difference in HL (in dB SPL) measured across a group of volunteers using a clinical audiometer with calibrated standard earphones. In the first phase, 183 volunteers were recruited to correct the RETSPL of the RAN-3500. Subsequently, the E200 device was sent to the Shenzhen Academy of Metrology and Quality Inspection and undergone rigorous testing and calibration (certificate no: JL2416207821). The results demonstrated full compliance with China’s national standard “JJG 388-2012 Verification Regulation of Pure-tone Audiometers” across all critical technical parameters, including Frequency Accuracy, Total Harmonic Distortion, Accuracy of RETSPL, Hearing Level Controller Accuracy, and Masking Sound Level Accuracy (complete data provided in Supplemental Material).
E200 Audiometry
This was conducted in a regular room (ambient noise: 42.6-58.7 dB, measured using a standard sound level meter: AWA6292 Multi-function Sound Level Meter) adjacent to a busy hallway without a sound-proof booth to simulate an ordinary environment. This test also utilized a variation of the Hughson–Westlake approach. The E200 only supports air conduction audiometry and does not support bone conduction. Contralateral masking was implemented when the interaural air-conduction threshold difference exceeded 40 dB.
Quality Control
The examiner has no access to the results of the other testing method until the experiment is completed, and the participants remain unaware of the results of both testing approaches until the end of the experiment. The sequence in which the participant undergoes the hearing tests is randomly determined using a random number table.
Statistical Analysis and Sample Size Calculation
All statistical analyses were conducted using IBM SPSS Statistics for Windows, Version 26.0 (IBM Corp., Armonk, NY, USA). The Spearman’s rank correlation coefficient was used to assess the relationship between the hearing thresholds obtained using the 2 detection approaches. The equivalence of the 2 audiometric methods was statistically evaluated using Schuirmann’s two one-sided tests (TOST) procedure. An equivalence margin of ±5 dB was established a priori based on clinical audiological standards. Bland–Altman analysis was employed to evaluate the agreement between pure-tone average (PTA) thresholds obtained from the 2 audiometric methods. The 95% limits of agreement (LoA) and mean differences were calculated to quantify measurement concordance. Intermethod agreement was assessed through Cohen’s κ statistics, with hearing conditions categorized into 3 grades. Based on the 69 data groups obtained in the pre-experiment, a confidence level of 0.9 (α = .1) was used, and the confidence interval width was set to 0.2. The sample sizes computed were determined to be 89 for the left ear and 107 for the right ear. Ultimately, 114 volunteers were enrolled.
Results
Demographics
Data were collected from 228 ears of 114 participants, with 7 frequencies tested/ear, resulting in 1596 datasets. Among these, 88 data points involved the use of masking during E200 audiometry. Each participant underwent CA and E200 audiometry. Among the participants, 53 were male (46.5%) and 61 were female (53.5%). The average age of the participants was 44.6 ± 16.5 years. They visited the otolaryngology department with various ear conditions (Table 1).
Demographic Characteristics of the Study Population.

Abbreviations: HL, hearing level; PTA, pure-tone average.
Mean threshold at speech frequencies: 0.5, 1, 2, and 4 kHz.
Noise Control Mechanism and Efficacy of the E200 Audiometer
By integrating PNC, ANC, and DSP technologies, the E200 achieves an overall noise suppression level exceeding 29 dB SPL, making it suitable for audiometric testing in environments with ambient noise below 60 dB (Figure 1). 26
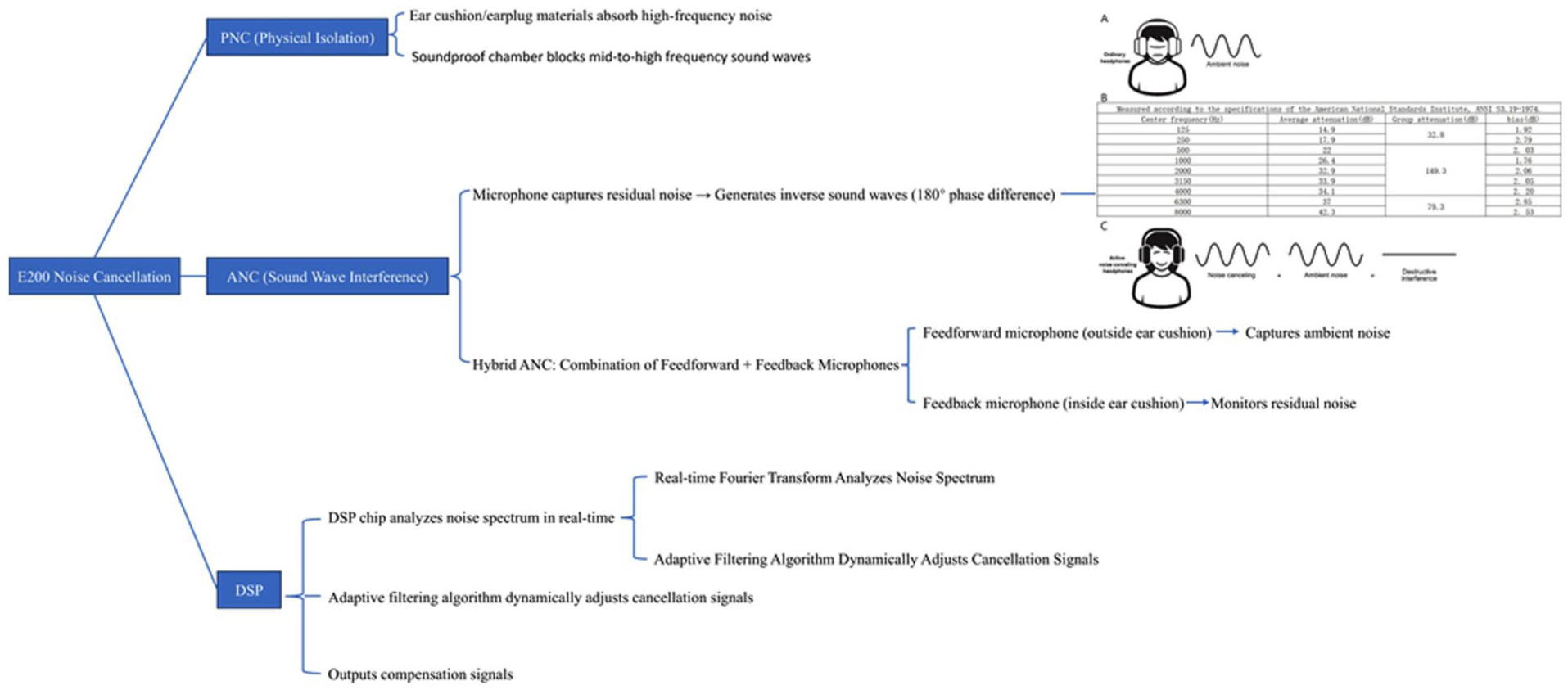
Working mechanism illustration of E200 noise control technology. The schematic illustrates the noise reduction mechanism of ANC headsets compared to ordinary headsets. Panel (A) shows ordinary headsets where ambient noise penetrates the ear cup with only passive attenuation. Panel (B) presents quantitative attenuation data measured per ANSI S3.19-1974 standards, displaying center frequencies (125-8000 Hz), average attenuation (dB), group attenuation (dB), and measurement bias (dB). Panel (C) demonstrates ANC operation: the system generates antiphase sound waves that create destructive interference with ambient noise, effectively canceling noise in the target zone. ANC, active noise control.
Correlation Between CA and E200 Audiometry
We analyzed the data from the left and right ears separately. The hearing threshold results of conventional and E200 audiometry are shown in Figure 2. Spearman’s rank correlation analysis was performed on the results of the 2 audiometric methods at each frequency. The Spearman’s rank correlation coefficients (r) and P values are shown in Figure 3. All P < .001, indicating a significant correlation for each frequency. The results of the TOST procedure confirmed methodological equivalence across all frequencies, with the 95% confidence intervals of intermethod differences being entirely contained within the predefined ±5 dB equivalence zone (Table 2).
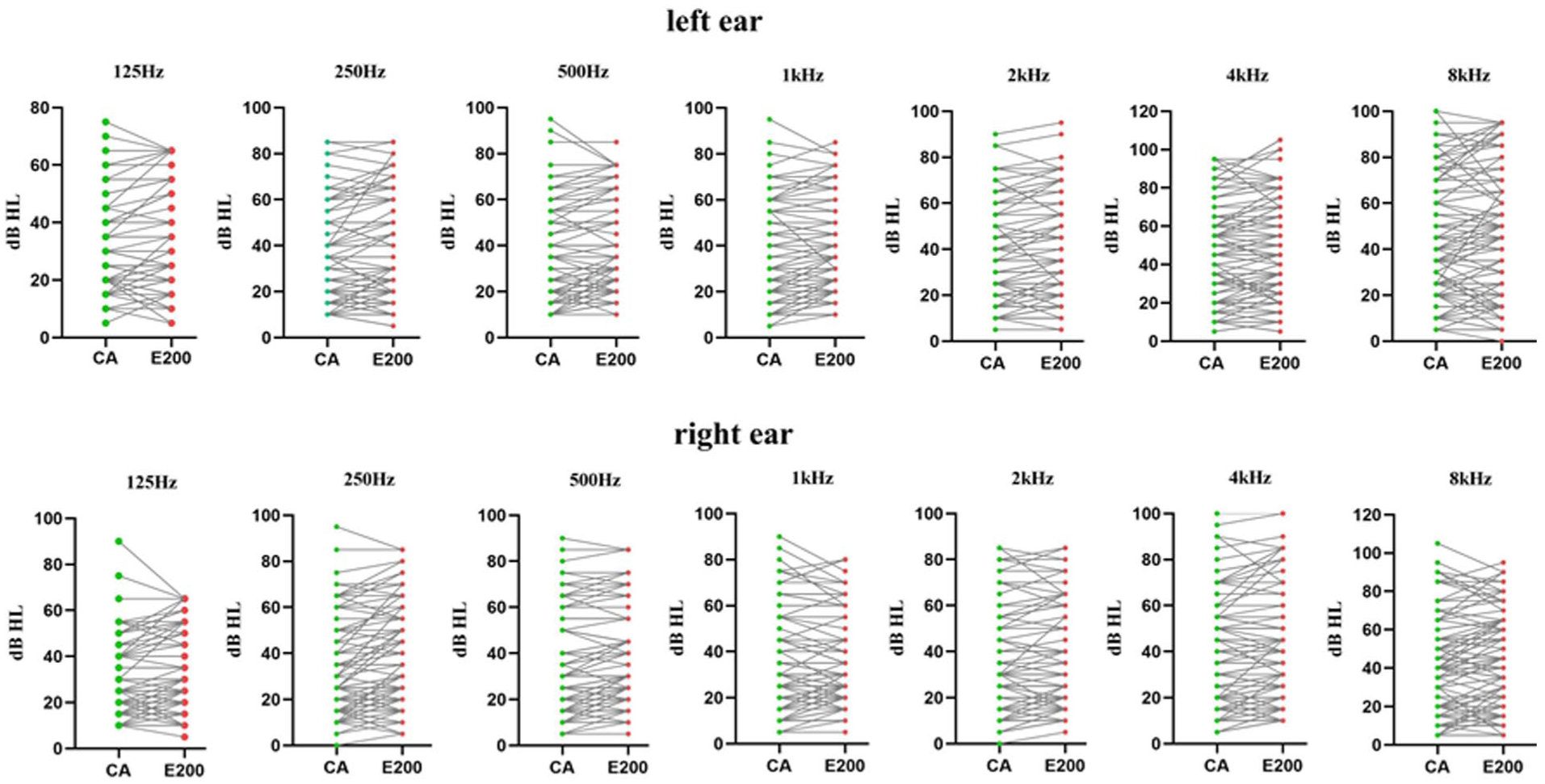
Hearing threshold results of conventional and E200 audiometry.
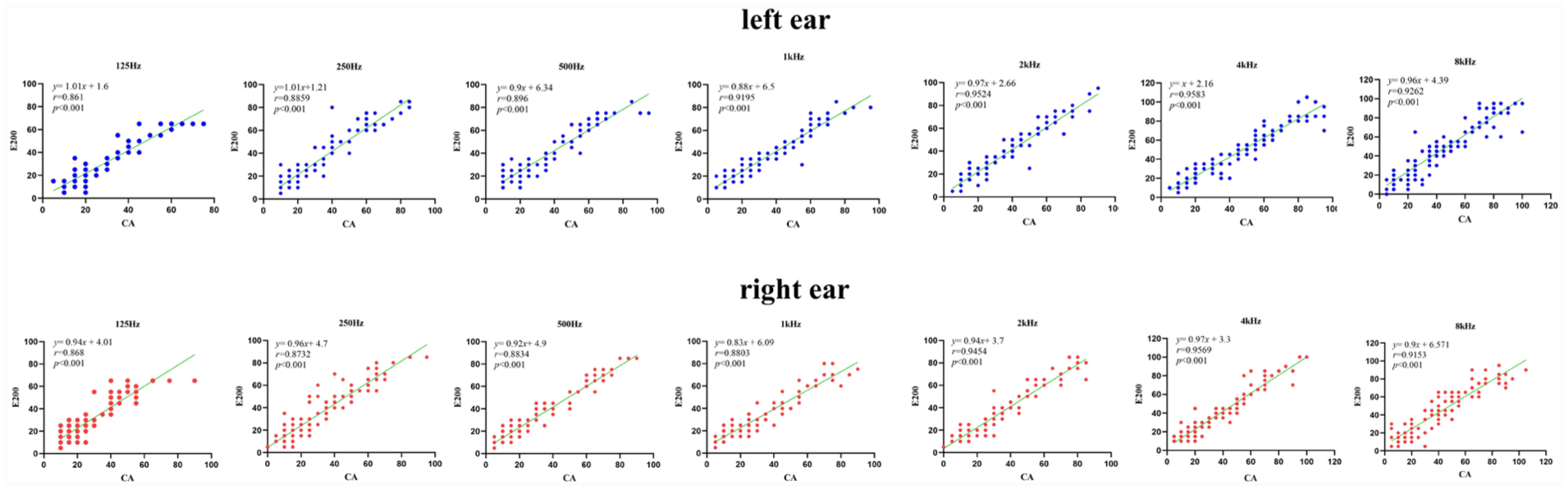
Correlation between conventional and E200 audiometry according to each frequency (dB HL). r, Spearman’s rank correlation coefficient; HL, hearing level.
Results of TOST Equivalence Testing Between 2 Audiometric Methods Across Frequencies.
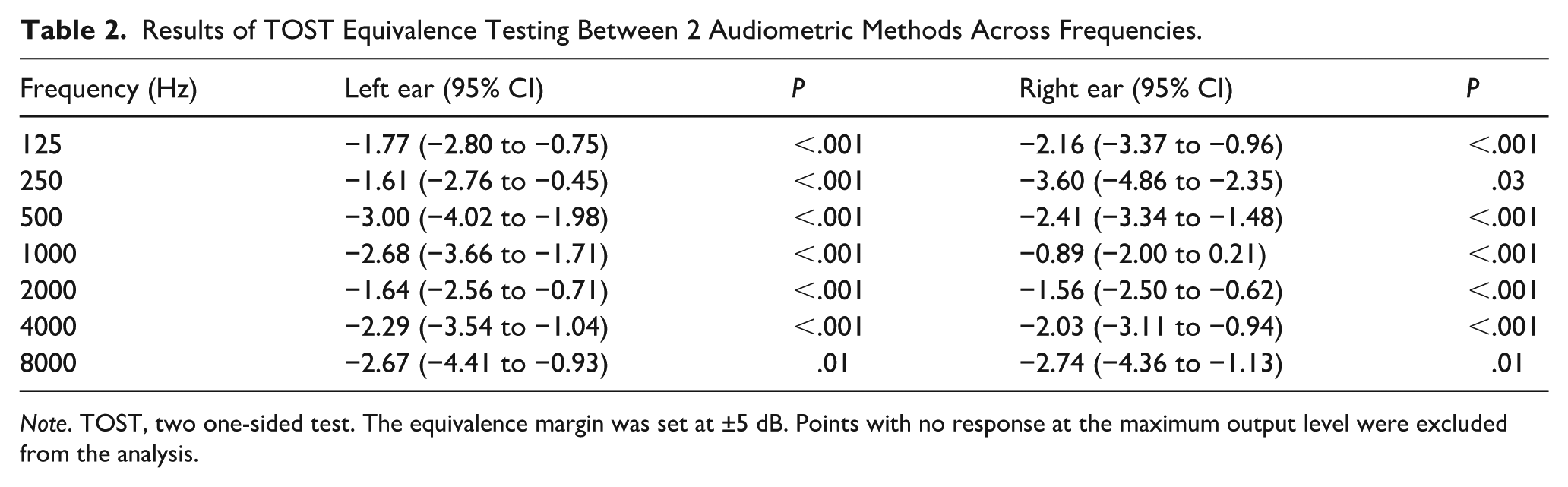
Note. TOST, two one-sided test. The equivalence margin was set at ±5 dB. Points with no response at the maximum output level were excluded from the analysis.
Accuracy of the E200 Audiometer in Hearing Threshold Measurement
|d| was defined as the absolute value of the hearing threshold difference between CA and E200 audiometry. Figure 4 shows the distribution of |d| ≤ 5 dB proportions across audiometric frequencies. Minimal values were observed at 8 kHz, reaching 60.2% (left) and 58.7% (right). Figure 5 shows the distribution of |d| values at 0, 5, 10, and ≥15 dB across all test frequencies in both ears. For most frequencies, the difference within 10 dB accounted for more than 90%.
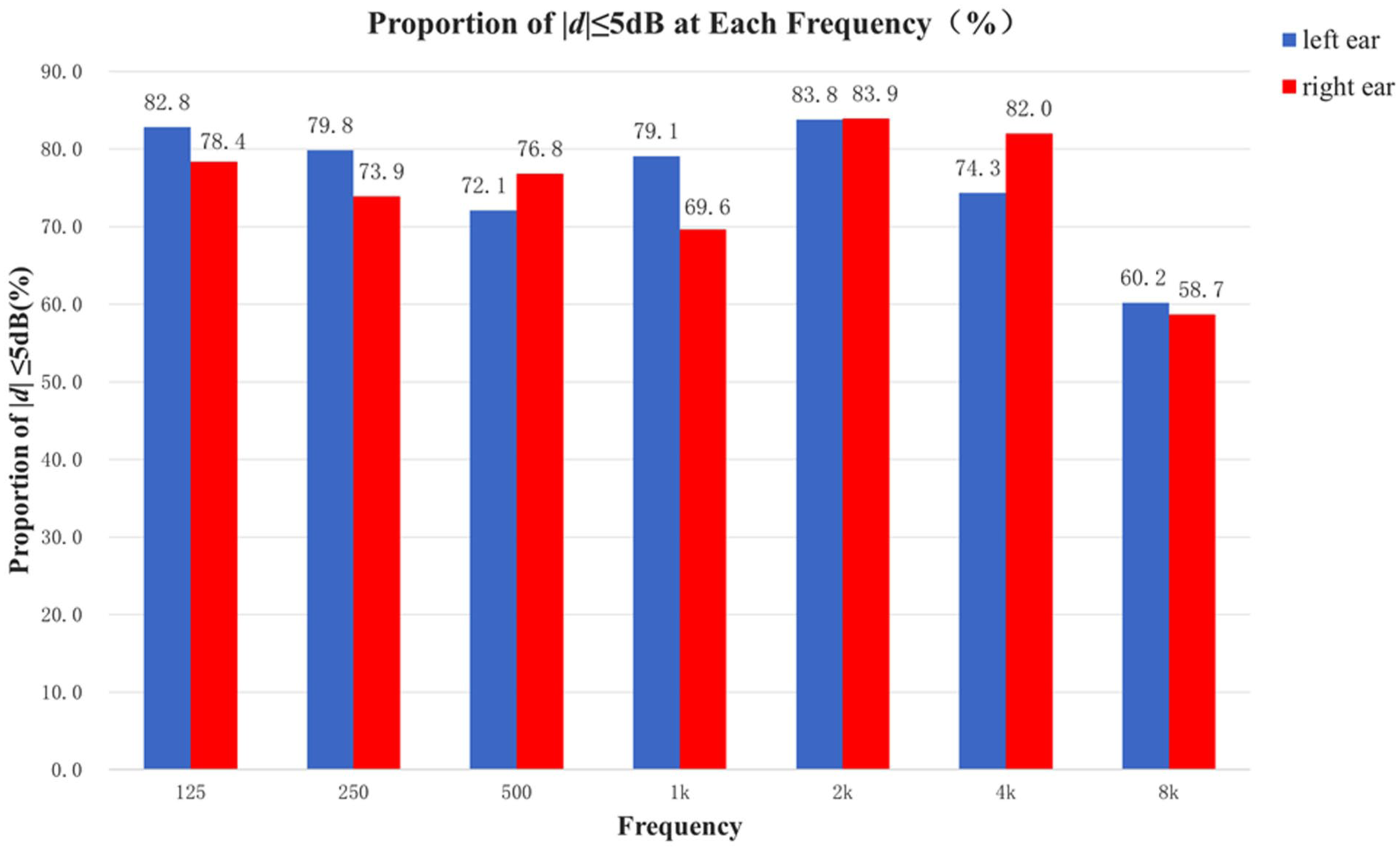
Proportion of |d| ≤ 5 dB at each frequency in both ears.
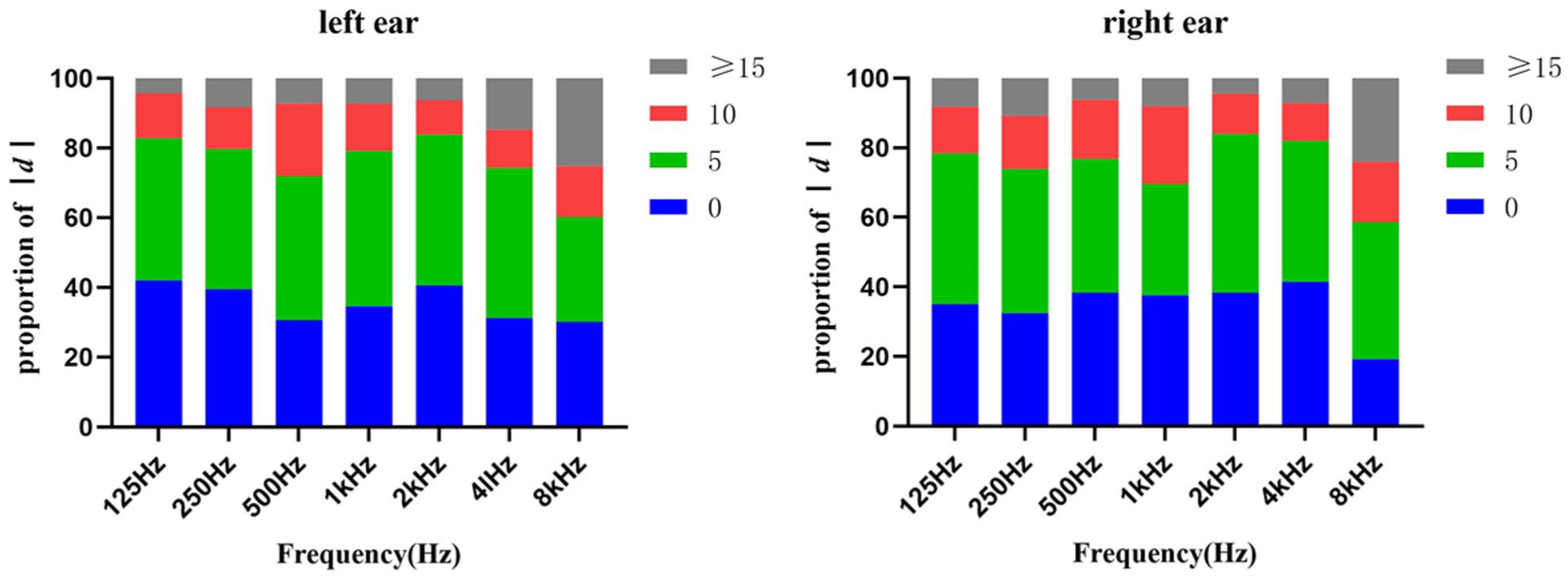
Distribution of |d| values at 0, 5, 10, and ≥15 dB across all test frequencies in both ears.
Accuracy of the E200 Audiometer for Grading Hearing Loss Severity
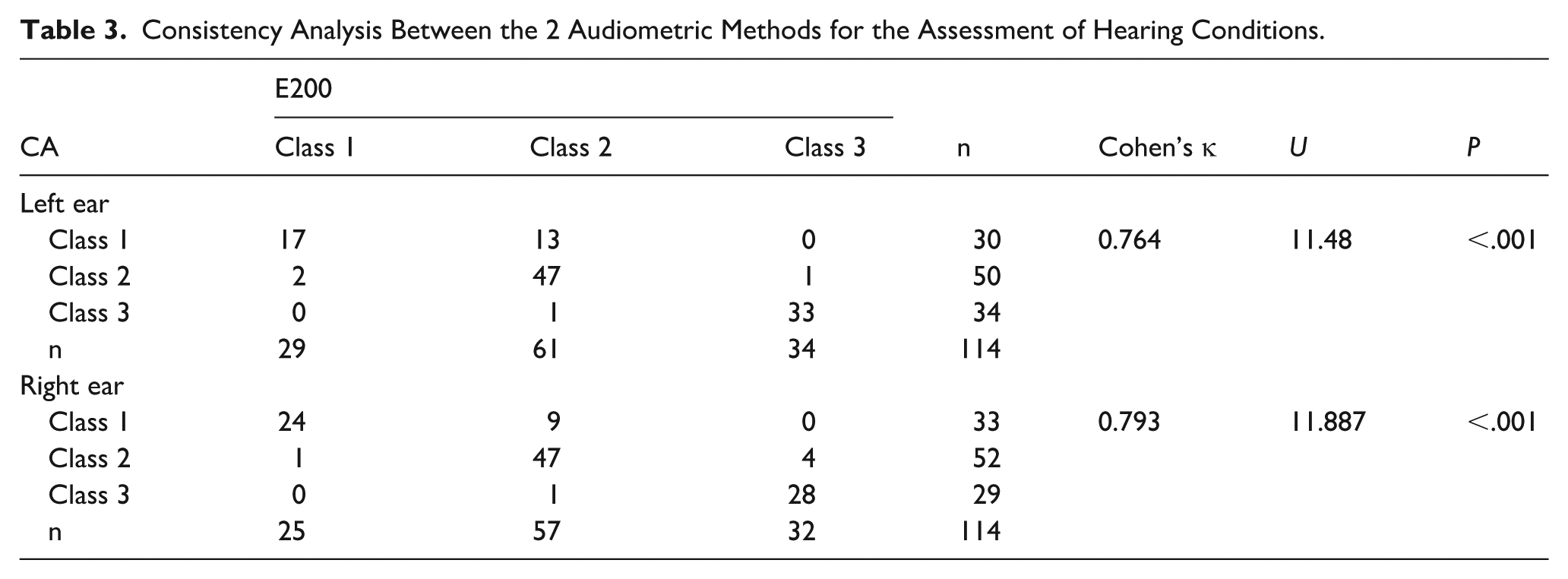
PTA datasets were obtained from both left and right ears (n = 114). After excluding 5 nonresponding left ears and 3 nonresponding right ears, the Bland–Altman plots showed 4.6% (5/109) and 4.5% (5/111) of points beyond 95% upper and lower limits for left and right ears, respectively, meeting the <5% criterion for excellent clinical agreement, 27 demonstrating strong concordance between the 2 audiometric methods in PTA results (Figure 6). Intermethod reliability was evaluated using Cohen’s κ statistics after categorizing hearing conditions into 3 grades based on PTA thresholds. According to the Landis and Koch interpretation criteria (0: poor; 0.01-0.2: slight; 0.21-0.4: fair; 0.41-0.6: moderate; 0.61-0.8: substantial; 0.81-1.0: almost perfect), 28 the analysis demonstrated substantial agreement for both ears: left ear κ = 0.764 (P < .001) and right ear κ = 0.793 (P < .001; Table 3).
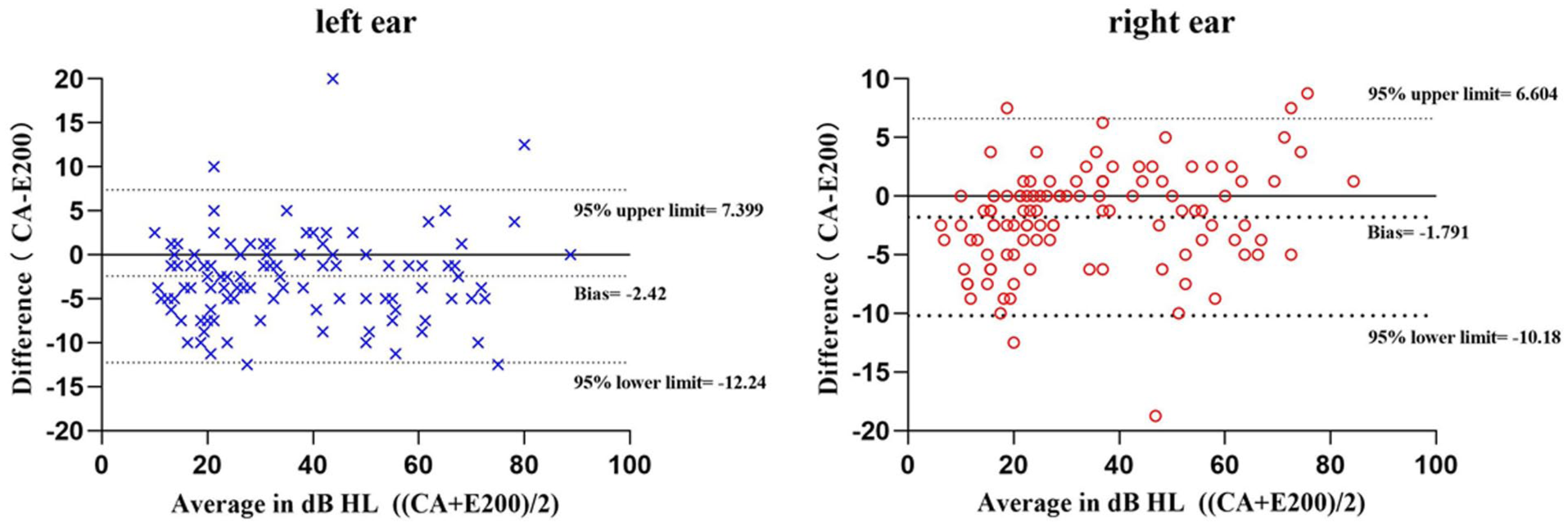
Bland–Altman plots of PTA results obtained using the 2 audiometric methods. The dotted lines in the plots represent the upper and lower limits of the 95% confidence intervals. PTA, pure-tone average.
Consistency Analysis Between the 2 Audiometric Methods for the Assessment of Hearing Conditions.
Discussion
Medical audiometers are essential tools for hearing assessment; however, they typically require controlled acoustic environments. Conventional pure-tone audiometry relies on sound-proof booths. With the rapid advancement of noise reduction technologies—including passive noise control (PNC), ANC, and DSP—the E200 integrates all 3 to achieve superior noise reduction performances. For device calibration, 183 participants underwent bilateral hearing assessment using both conventional sound-proof booth audiometry (reference standard) and the E200 audiometry. The device parameters were iteratively adjusted to minimize the differences in threshold between the 2 methods. Subsequently, 114 additional participants were enrolled in a reliability study to validate the performance of the calibrated E200 against the reference standard. The E200 audiometry was performed in a regular room (ambient noise: 42.6-58.7 dB) adjacent to a busy hallway. Significant correlations were observed between the 2 audiometric methods across all test frequencies. According to ANSI S3.6 standards, the 5 dB step size in pure-tone threshold measurements establishes ±5 dB as the clinical tolerance threshold. All frequency comparisons satisfied the ±5 dB equivalence criterion in TOST testing. Regarding accuracy, the proportion of thresholds within ≤5 dB was 60.2% for the left ear and 58.7% for the right ear at 8 kHz, whereas all other frequencies exceeded 68.1%. This may suggest greater variability at the high-frequency of 8 kHz during audiometric testing. The Bland–Altman plots demonstrated good agreement between the 2 audiometric methods in PTA measurements, with only 4.6% (5/109) and 4.5% (5/111) of data points exceeding the 95% LoA for the left and right ears, respectively—well below the 5% threshold for clinical acceptability. Further supporting this, Cohen’s κ statistics demonstrated substantial intermethod agreement for both ears (left: κ = 0.764; right: κ = 0.793) in the 3-category classification based on PTA results, confirming strong concordance in hearing loss severity grading.
Conventional pure-tone audiometry, while considered the gold standard, is inherently constrained by its reliance on sound-proof booths, which creates substantial barriers to care in resource-limited settings such as primary care clinics, community health centers, and remote or rural areas, where the high cost and physical space for such infrastructure are often unavailable.11,29 The E200 system demonstrates that reliable hearing thresholds can be obtained outside traditional sound-proof booths, enabling earlier detection and intervention for hearing loss in underserved populations. Beyond improving geographical accessibility, this technology enhances clinical efficiency and continuity of care by offering a practical solution for postoperative monitoring following otologic surgeries (such as tympanoplasty, stapedectomy, or cochlear implantation), allowing for convenient in-office checks without the need to schedule scarce booth time.
The final sentences of the Lancet editorial 5 are as follows: “Global multidisciplinary and collaborative efforts are urgently needed to address the health needs of children and adults with hearing loss” and “hearing loss must not remain a silent epidemic.” While hearing rehabilitation technologies become increasingly accessible in low- and middle-income countries (LMICs), the WHO recommends prioritizing primary and secondary prevention through enhanced education and screening initiatives. 5 By integrating ANC technology, the E200 system achieves clinical-grade audiometry without sound-proof environments, enabling decentralized hearing screening programs crucial for LMICs. This audiometric device can be operated by nonmedical personnel after undergoing simple training. Expanding its use across diverse settings and involving nonmedical personnel may alleviate the burden caused by global hearing loss.
Limitations
The E200 system only supports air conduction audiometry and does not support bone conduction testing. Consequently, it may miss cases requiring masking and cannot differentiate between types of hearing loss (sensorineural, conductive, or mixed). In this study, when conducting hearing tests on the participants, each participant underwent the 2 audiometric methods, but no repeated measurements were conducted, so the reliability of the test-retest could not be evaluated. A test–retest design will be implemented in subsequent studies to evaluate measurement consistency.
Conclusions
This study evaluated the feasibility and reliability of pure-tone audiometry using the E200 system integrated with ANC technology. This portable ANC-integrated audiometric system demonstrates considerable potential for hearing screening beyond traditional sound-proof booth; however, its long-term clinical value and practical applications require further investigation and validation.
Supplemental Material
sj-pdf-1-ear-10.1177_01455613251385770 – Supplemental material for Reliability Evaluation of a Portable Active Noise Control-Integrated Audiometer for Clinical Hearing Assessment
Supplemental material, sj-pdf-1-ear-10.1177_01455613251385770 for Reliability Evaluation of a Portable Active Noise Control-Integrated Audiometer for Clinical Hearing Assessment by Qianjin Gao, Cuncun Ren, Xiaoqin Fan, Minjiao Wang, Yongshou Liu, Xiaogang An, Xinyu Dong, Xiaocheng Wang, Lihui Zhou, Jianhua Qiu, Jun Chen and Dingjun Zha in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
Acknowledgments are extended to Prof Yongjun Hu (Hubei University of Economics) for his scholarly advice regarding active noise control (ANC) techniques, and to Douting Technology Co, Ltd (Wuhan) for their instrumental technical and equipment support. The authors additionally acknowledge with deep appreciation the essential contributions of all participants involved in this study.
ORCID iDs
Ethical Considerations
The study was approved by the Medical Ethics Committee of the First Affiliated Hospital of the Air Force Medical University and was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines (Ethics Committee File No: KY20242145-F-1).
Consent to Participate
All participants were informed of this study and signed the informed consent form.
Author Contributions
Data analysis, visualization, and manuscript drafting were performed by Q.G. Data collection was conducted by C.R., X.F., M.W., X.D., and Q.G. Experimental supervision and quality control were carried out by X.W. and Y.L. Study design and manuscript revision were accomplished by L.Z., J.Q., J.C., and D.Z. All authors performed data interpretation and approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Interdisciplinary Integration Program of Air Force Medical University (2024JC016), Shaanxi Key Industrial Chain Project (2024SF-ZDCYL-01-16), Innovative Talent Promotion Plan of Shaanxi Province (2023-CX-TD-70), Shaanxi Health Commission Research Innovation Platform Development Plan (2024PT-07), Shaanxi Provincial Science and Technology Research Fund (2024SF-YBXM-343), Key Research and Development Plan of Shaanxi Province (2022ZDLSF02-10), National Natural Science Foundation of China (82301333).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The E200 devices were provided free of charge by Wuhan Douting Technology Co, Ltd for this clinical trial. The authors declare no financial or non-financial competing interests with the company, and the authors declare that they have no competing interests.
Data Availability Statement
The datasets are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.