Abstract
Background:
Olfactory training (OT) interventions exhibit dual therapeutic potential, enhancing both chemosensory function and multiple cognitive domains through neuroplastic mechanisms.
Objective:
To systematically review the efficacy of OT on olfaction, cognition, and neuroimaging changes across a range of populations, from healthy adults to patients with olfactory and cognitive impairments.
Methods:
A systematic review was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. A comprehensive search of PubMed, Embase, Web of Science, and Cochrane databases performed to identify studies that evaluate the effect of OT on olfaction and cognition in different populations, focusing on its potential to enhance cognition and mitigate the progression of cognitive decline.
Results:
Of the initial 412 abstracts reviewed, 24 full-text articles were included. Risk-of-bias assessment revealed 7/12 randomized controlled studies (RCTs) had high risk and 11/12 non-RCTs had serious bias. The duration of OT interventions mostly ranged from 3 to 6 months. In healthy adults (6 studies) and adults with olfactory dysfunction (13 studies), OT improved olfactory function in a population-dependent manner and exerted domain-specific cognitive benefits. Meta-analysis revealed that the pooled proportion of individuals achieving a clinically-significant olfactory improvement was 27.01% (95% confidence interval, 0.16-0.40). Improvements were observed in global cognition, verbal fluency, and working memory. In adults with cognitive impairment (5 studies), classical OT showed limited olfactory and cognitive improvements; however, intensive OT with enriched odors, longer sessions, and greater adherence demonstrated potential for enhanced cognitive and emotional outcomes in patients with dementia. Furthermore, the reviewed neuroimaging studies consistently demonstrate that OT can increase volumes in several brain regions and promote neuronal reorganization, providing a potential mechanism of OT in olfactory recovery and cognitive improvement.
Conclusions:
OT effectively improves olfaction, cognition, and brain health, particularly in cognitively-normal populations.
Introduction
A significant proportion of the population (22%) experiences olfactory dysfunction, 1 attributable to diverse factors such as sinonasal disease, traumatic brain injury, upper respiratory infections, neurodegenerative diseases, and the natural aging process. 2 Persistent olfactory dysfunction disturbs food intake and harm avoidance, impairs social and professional engagement, and is linked to higher mortality rate.3,4 Importantly, the decline in olfaction is a significant predictor of subsequent cognitive impairment. Clinical findings indicated that olfactory dysfunction correlated with impaired cognitive function, decreased hippocampal volume, greater amyloid burden, and predicted decline across various cognitive domains, especially in memory, language, and execution.5-8 Studies have also reported that odor identification impairment predicted conversion to mild cognitive impairment (MCI) among cognitively-normal individuals and conversion to dementia in patients with MCI. 9 These studies indicated a possible neurological link between olfaction and cognition, with the peripheral olfactory system differing from other sensory systems by directly projecting neuronal connections to cortical regions crucial for cognitive function, including the orbitofrontal cortex, amygdala, pyriform cortex, and entorhinal cortex.10-12 Due to this substantial neural overlap, the olfactory system could be a possible target for early interventions in cognitive decline.
Olfactory training (OT) is a non-pharmacological rehabilitation method involving systematic exposure to specific odorants to improve the sense of smell. 13 OT has been shown to effectively treat olfactory dysfunction resulting from various etiologies, including upper respiratory infections, trauma, neurological disorders, and idiopathic conditions, particularly improving odor discrimination and identification. 14 Interestingly, several studies have demonstrated that OT can reduce depressive symptoms and improve global cognition, specifically, verbal fluency, short-term memory, and verbal learning and memory.15,16 In addition, research has shown that OT leads to structural changes in several brain regions, including the olfactory bulb (OB), hippocampus, cerebellum, and thalamus, as well as alterations in functional connectivity. 17 While the precise mechanism by which OT enhances olfactory function and cognition remains unclear, it is hypothesized to relate to neuroplasticity effect of OT. 18
While extant systematic reviews have established the neurotherapeutic effects of OT, 17 critical gaps persist regarding heterogeneity in treatment responsiveness linked to underlying pathology. Interindividual variability in neuropathological burden creates differential therapeutic thresholds across a clinical spectrum encompassing neurotypical adults, adults with olfactory dysfunction but preserved cognition (with potential neural compromise), and adults with cognitive impairment exhibiting neural system damage. To address these gaps, this review aimed to (1) stratify OT outcomes according by disease status across 3 clinically-distinct populations, including healthy adults, adults with olfactory impairment, and adults with cognitive impairment, (2) synthesize evidence on the effects of OT on olfactory function, cognitive performance, and neuroimaging biomarkers within each population, and (3) evaluate the quality of the evidence through assessment of study bias and evidence levels, facilitating the translation of findings into clinical contexts.
Materials and Methods
Data Sources and Search Strategy
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis statement. The study was prospectively registered in an online database (PROSPERO ID: 655876). A comprehensive systematic literature review was performed using the PubMed, Embase, Web of Science, and Cochrane databases from inception through September 3, 2024. The comprehensive search strategy was presented in Supplementary Table 1.
Eligibility Criteria
Inclusion criteria were as follows: (1) Participants: Healthy adults, patients with olfactory dysfunction, and patients with cognitive loss. (2) Intervention: Any OT occurring over multiple sessions. (3) Comparator: Studies could utilize either a pre-post intervention design (within-subject comparison) or a control group (placebo, no treatment or other cognitive/visual training). (4) Outcomes: Olfactory testing, neuronal, or cognitive outcomes assessed before and after the OT; (5) Study Design: Case series, cohort studies, and randomized controlled studies (RCTs). We chose this broad range of designs to ensure a comprehensive synthesis of the evidence. Studies were excluded if they were non-English, nonhuman adult studies, case reports, or conference abstracts.
Data Selection, Extraction, and Management
The titles and abstracts of relevant studies were reviewed by 2 independent investigators (Y.T. and D.W.). The process of study selection was illustrated in Figure 1. Two authors independently assessed the full texts of the identified studies to confirm that the studies met the criterion. Data were extracted from each study, including details regarding sample size, patient population, the specific intervention employed (regimen and duration), outcome measures across olfaction, emotion, cognition, and neuroimaging, and the conclusions. Data extraction was also performed independently by 2 reviewers (Y.T. and D.W.) using a pre-piloted data extraction form to ensure consistency. The complete list of all extracted data items and the blank extraction template are provided in Supplementary Table 2. Disagreements were resolved through discussion. When consensus could not be reached, arbitration by a third reviewer was applied (X.L.).
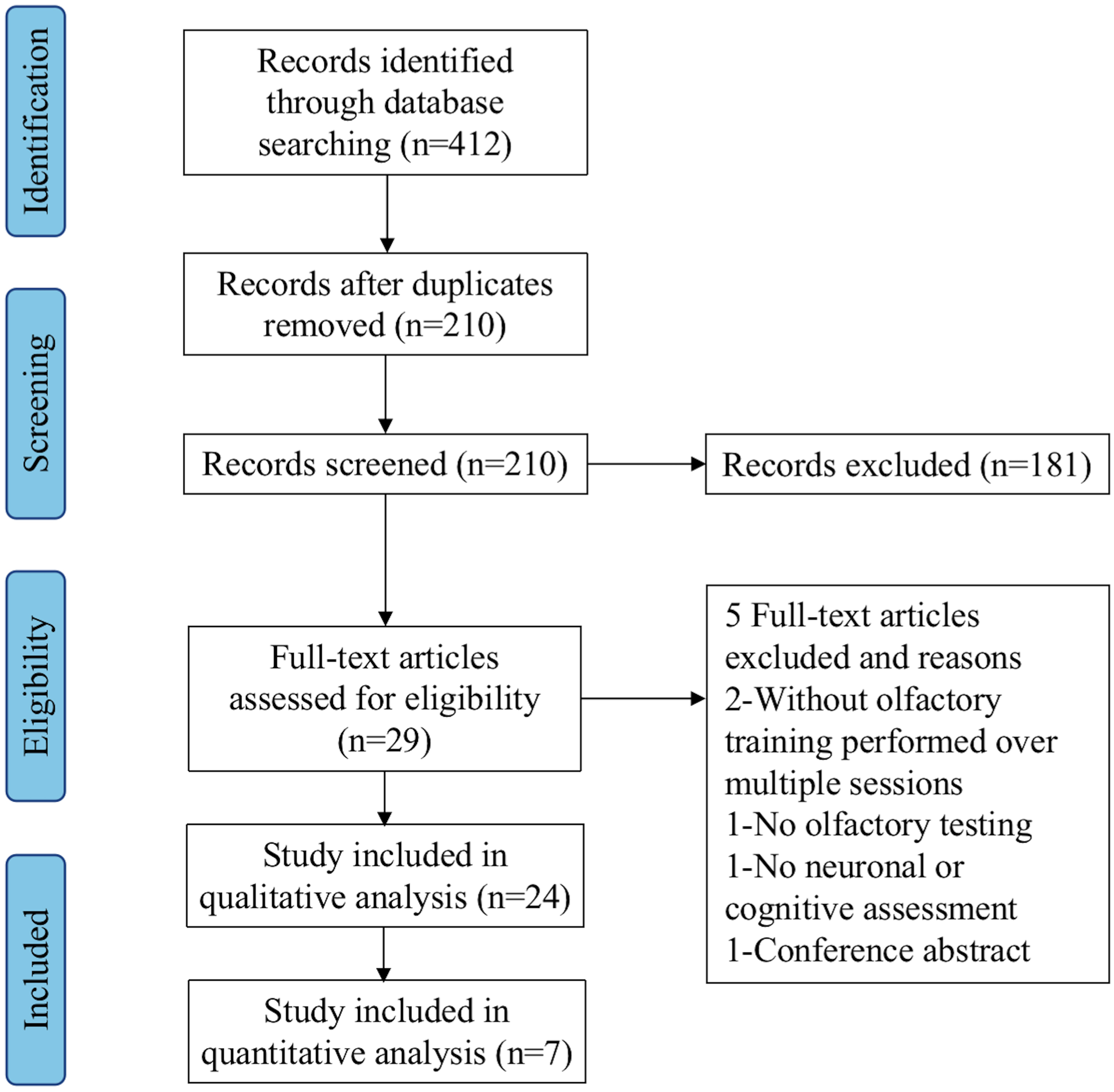
Article selection process for systematic literature review.
Risk of Bias Assessment
Two researchers (Y.T. and D.W.) assessed the potential bias in each selected study independently. The third researcher (X.L.) was consulted for resolving any difference of opinion. The Cochrane Risk of Bias Assessment Tool 2 (RoB-2) tool 19 was used for RCTs. It includes 5 domains: “randomization process,” “deviations from intended interventions,” “missing outcome data,” “measurement of the outcome,” and “selection of the reported result.” We used the Risk Of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool 20 for assessing the risk of bias in non-randomized studies. It comprises 7 domains: “bias due to confounding,” “selection of participants, classification of interventions,” “deviations from intended interventions,” “missing data,” “measurement of outcomes,” and “selection of the reported result.” Each domain is graded as “low,” “moderate,” “serious,” and “critical.”
Quality Assessment
The overall quality of evidence for each outcome (olfactory function, cognitive performance, and neuronal changes) was assessed independently by 2 authors (Y.T. and D.W.) using the Grades of Recommendation, Development and Evaluation (GRADE) approach. 21 We considered the following criteria to assess the quality of the evidence: risk of bias, inconsistency of results, indirectness of the evidence, and imprecision. We also downgraded the quality, where appropriate. These judgements are summarized in the “summary of findings” tables (Table 1).
Summary of Findings. Population: Healthy Adults, Adults With Olfactory Dysfunction, and Adults With Cognitive Impairment, Intervention: Olfactory Training, Outcome: Olfaction, Cognition, and Neuronal Changes.
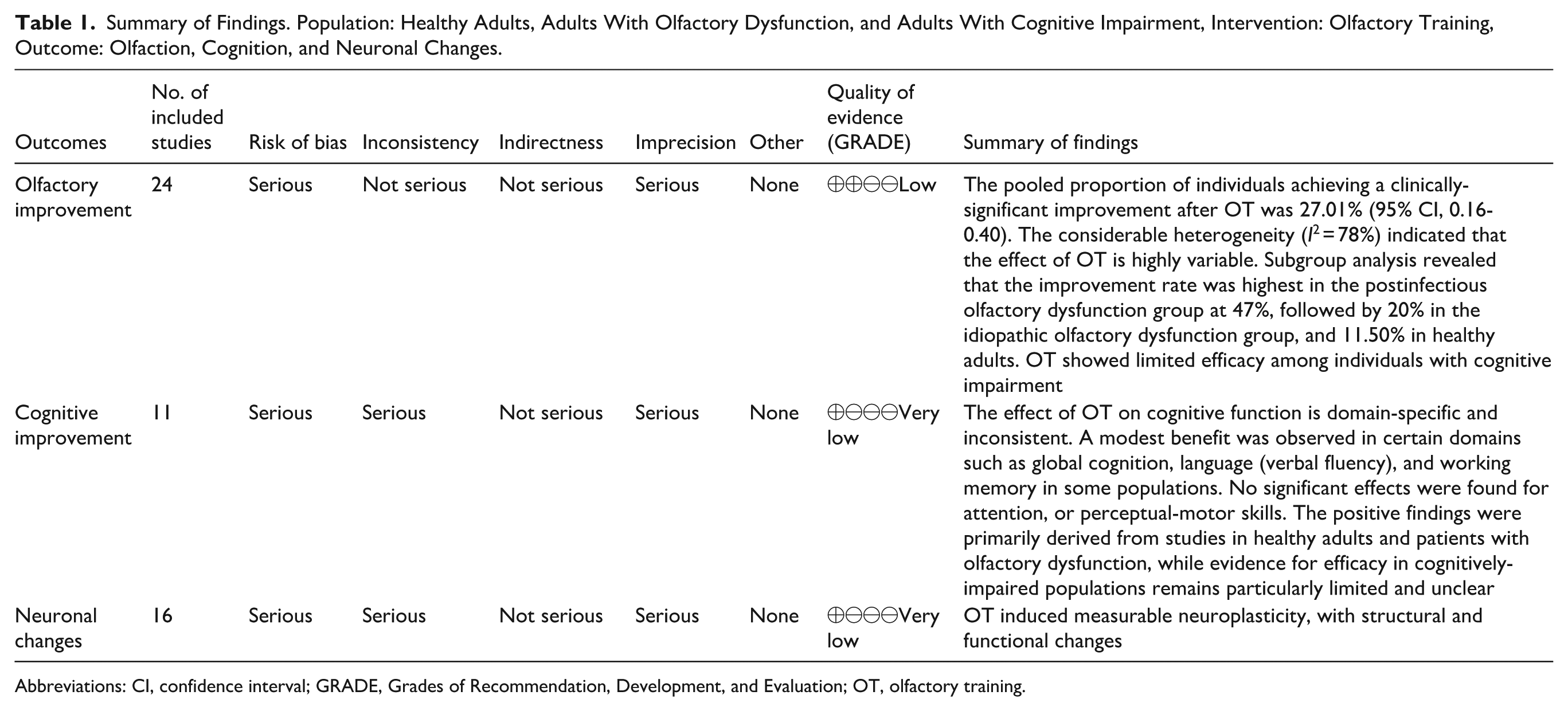
Abbreviations: CI, confidence interval; GRADE, Grades of Recommendation, Development, and Evaluation; OT, olfactory training.
Statistical Analysis
A total of 7 studies included in the meta-analysis reported the proportion of patients achieving clinically-significant outcomes following OT.15,22-27 These studies were pooled using a single-rate meta-analysis. Some studies involved mixed population, which included healthy controls and patients with olfactory dysfunction of various etiologies. Data were extracted and categorized into the following subgroups: Healthy adults, adults with postinfectious olfactory dysfunction (PIOD), posttraumatic olfactory dysfunction (PTOD), and idiopathic olfactory dysfunction (IDOD). Based on the extracted data, the total number of patients and the number of those exhibiting a clinically-significant improvement were compiled. For studies with 0 events in 1 group, a continuity correction (adding 0.5 to both the number of events and nonevents) was applied to facilitate calculation. In this analysis, the incidence of P (ie, the rate of a clinically-significant improvement) did not follow a normal distribution. Thus, the ratio type data were used to calculate the incidence with the formula:
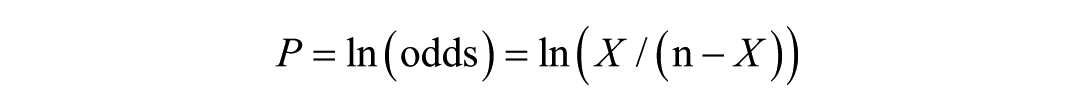
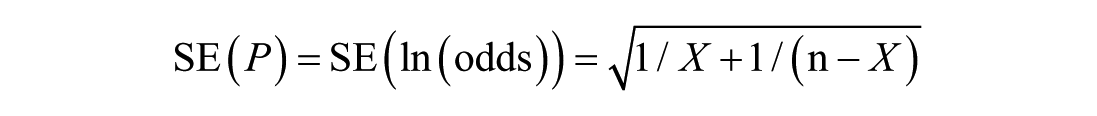
where X represented the number of patients with a clinically-significant improvement and n denoted the total number of patients. A forest plot was generated, and a random-effects model was employed to estimate the odds ratio (OR) along with its 95% confidence interval (CI). The pooled OR was then transformed into a proportion and corresponding 95% CI using the following conversions: P = OR/(1 + OR), LL = LLOR/(1 + LLOR), UL = ULOR/(1 + ULOR), where LL and UL represent the lower and upper limits of the CI, respectively. 28 Subgroup analyses were conducted based on different population to further evaluate outcomes and explore potential sources of heterogeneity. All meta-analyses were performed using the RevMan 5.4 software (The Cochrane Collaboration, London, UK).
Results
Characteristics of Studies
A total of 412 articles were retrieved in the initial database search. After removing duplicates and screening abstracts, 29 articles remained. We reviewed the full text, and 5 studies were excluded due to the following reasons: intervention without OT performed over multiple sessions (n = 2), no olfactory testing (n = 1), no neuronal or cognitive assessment (n = 1), and conference abstract (n = 1). Finally, 24 articles met the inclusion criteria for systematic review15,16,22-27,29-44 and 7 of these were included in meta-analysis15,22-27 (Figure 1).
The participants in these studies were divided into 3 distinct populations: (1) healthy adults demonstrating no cognitive or olfactory impairments (n = 6),15,16,29-32 (2) adults exhibiting olfactory dysfunction of varying etiologies (n = 13),22-27,33-39 and (3) adults with cognitive impairment due to MCI and dementia (n = 5).40-44 The olfactory dysfunction group comprised individuals with PIOD, PTOD, IDOD, and olfactory dysfunction due to laryngectomy.22-27,33-39 This study incorporated a total of 1105 participants across all included studies. The mean sample size was 46 participants, ranging from 7 to 100.
Most of reviewed studies, for example, Hummel et al. 45 , employed classical 4-odor OT strategy involving rose, eucalyptus, lemon, and cloves. The training was performed twice daily, with each odor presented for 10 to 20 seconds. Modified OT introduced innovations primarily in the number and variety of odors. For instance, Rezaeyan et al utilized a modified protocol with 4 different odors changed monthly. 33 Oleszkiewicz et al employed a set of 5 odors updated every 3 months. 27 Power Guerra et al and Woo et al employed an extended OT protocol using 7 odors.16,24 Genetzaki et al employed a protocol with 8 odors. 22 In another approach, Oleszkiewicz et al compared simple OT with single-molecule odors to a mixture OT using 9 odor mixtures. 32 Intensive OT not only increased the number of odors but also extended training duration and incorporated additional cognitive stimuli. For example, Li et al examined OT combined with concurrent video watching and sound exposure, as well as counting tasks. 30 Al Aïn et al implemented OT involving 20 to 30 minutes daily training with 3 complex tasks. 29 Similarly, Lin and Li applied 15-odor olfactory stimulation coupled with structured cognitive exercises for 30 minutes/session. 43 Cha et al used 40 odors in 15 minute sessions. 42 The duration of OT interventions mostly ranged from 3 to 6 months. However, Han et al implemented an extended training period of 9 months, 37 whereas the shortest intervention was reported by Cha et al, lasting only 15 days. 42
Our review included 12 RCTs and 12 non-RCTs. Among these, 8 studies adopted a single-arm, uncontrolled pre-post design. Three articles utilized healthy controls to compare OT effects between healthy adults and patients with olfactory dysfunction OD. Ten studies employed non-OT control groups. Additionally, a number of articles compared the efficacy of different OT protocols. The outcome measures of our study included olfactory function, cognitive function, and neuroimaging outcomes. Detailed characteristics of the included studies are presented in Supplementary Tables 3 to 5.
Risk of Bias Assessment
We employed 2 methodological tools to evaluate study quality. Among 12 RCTs assessed using the Cochrane RoB-2 tool (Figure 2), 7 demonstrated high overall risk of bias while 5 raised some concerns. Methodological limitations primarily originated from inadequately-described randomization procedures, deviations from intended interventions due to insufficient blinding, potential bias in outcome assessment, and selective outcome reporting largely attributed to absent trial registrations. For the 12 non-RCTs evaluated with ROBINS-I (Figure 3), 11 exhibited serious overall risk of bias, with only 1 study demonstrating moderate risk. Universal limitations emerged in confounding control, affecting all studies. Additionally, 2 studies showed serious concerns regarding deviations from intended interventions, 2 displayed serious risk of bias from missing data, and 2 manifested serious risk in outcome measurement. Collectively, these findings indicate substantial methodological constraints across the evidence base.
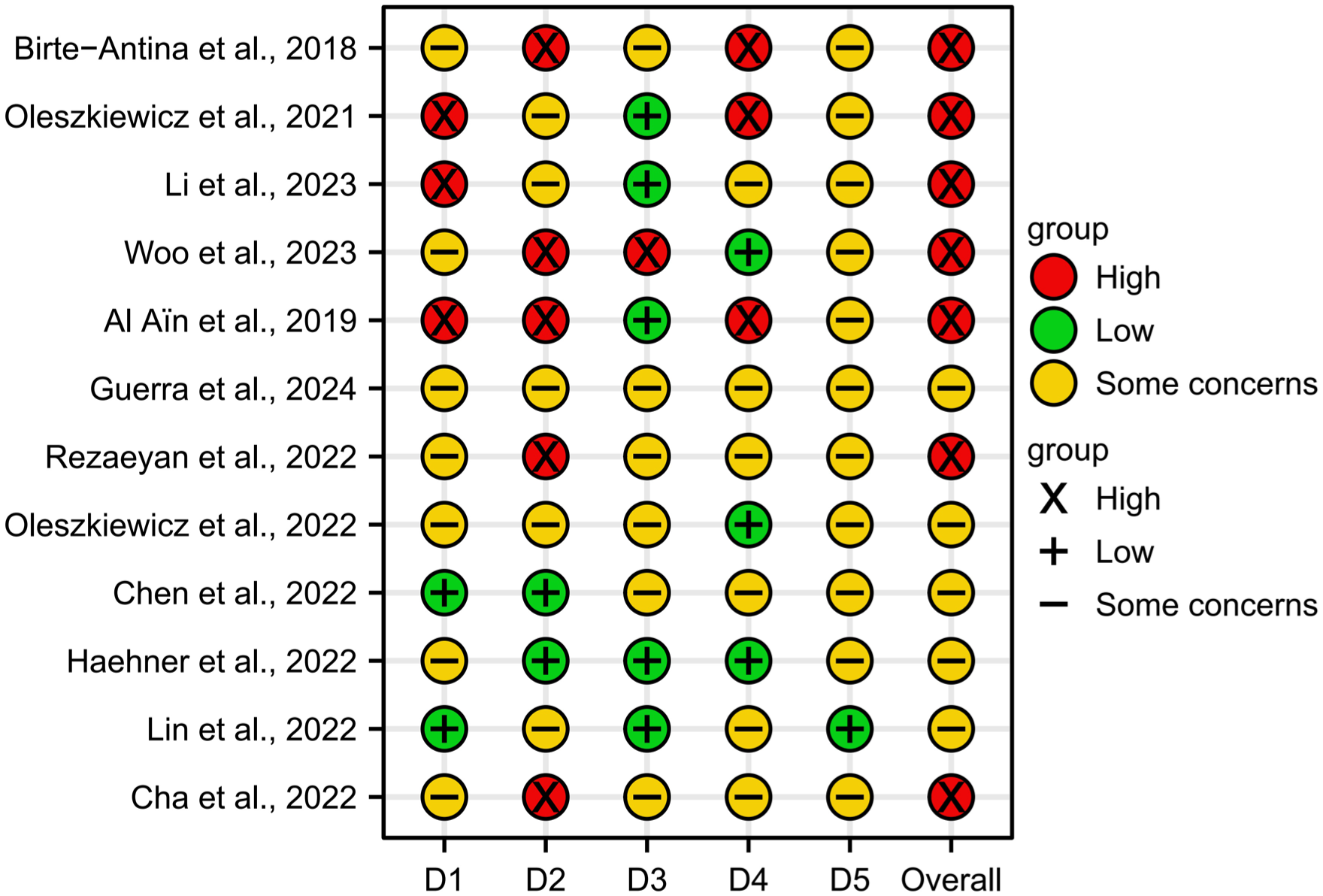
Assessment of 12 randomized controlled trials by Cochrane Risk of Bias Assessment Tool 2 tool. D1, domain 1: bias arising from the randomization process; D2, domain 2: bias due to deviations from intended intervention; D3, domain 3: bias due to missing outcome data; D4, domain 4: bias in measurement of the outcome; D5, domain 5: bias in selection of the reported result.

Assessment of 12 randomized controlled trials by the Risk Of Bias In Non-randomized Studies of Interventions tool. D1, domain 1: bias due to confounding; D2, domain 2: bias due to selection of participants; D3, domain 3: bias in classification of interventions; D4, domain 4: bias duo to deviations from intended interventions; D5, domain 5: bias due to missing data; D6, domain 6: bias in measurement of outcomes; D7, domain 7: bias in selection of the reported result.
Effects of OT on Olfactory Function
The 24 articles included in this systematic review all measured the effect of OT on olfactory function. Most studies used the Sniffin’ Sticks test battery to assess olfactory performance. This test measured odor threshold (T), odor discrimination (D), odor identification (I), and the composite TDI score. In addition, 1 study employed the University of Pennsylvania Smell Identification Test (UPSIT), 25 while another used the Connecticut Chemosensory Clinical Research Center test. 36 In the 2 studies involving participants with dementia, olfactory function was evaluated specifically using the Top International Biotech Smell Identification Test and the YSK Olfactory Function Test, respectively.42,43 These approaches was adopted because the participants faced challenges in completing other types of olfactory tests.
To quantitatively synthesize the evidence on the efficacy of OT, a meta-analysis was performed on the 7 studies that reported the proportion of individuals achieving a clinically-significant improvement in olfactory function. Among these 7 studies, 5 studies defined clinically-significant results as an increase in TDI score of ≥5.5, 1 study used a TDI increase of ≥6 and 1 defined as an increase in TDI ≥5.5 or in UPSIT ≥4. Studies were divided into 4 subgroups according to different populations: Healthy adults, adults with PIOD, PTOD, and IDOD.15,22-27 These studies were pooled using a single-rate meta-analysis, as shown in Figure 4. OR needs to be converted to get the proportion of clinically-significant results and 95% CI. The proportion of individuals achieving a clinically-significant improvement was 27.01% (95% CI, 0.16-0.40). Subgroup analysis revealed substantial differences based on etiology. The highest improvement rate was observed in the PIOD group at 47.08% (95% CI, 0.37-0.58). Lower improvement rates were observed for other subgroups. The proportion was 20.00% (95% CI, 0.09-0.36) among adults with IDOD and 11.50% (95% CI, 0.04-0.25) in healthy adults. The estimate for patients with PTOD was highly uncertain (9.90%, 95% CI, 0.01-0.67) likely due to limited data. Heterogeneity was low to moderate in the IDOD (I2 = 0%) and PIOD (I2 = 39.0%) subgroups, but relatively high in healthy group (I2 = 55%).
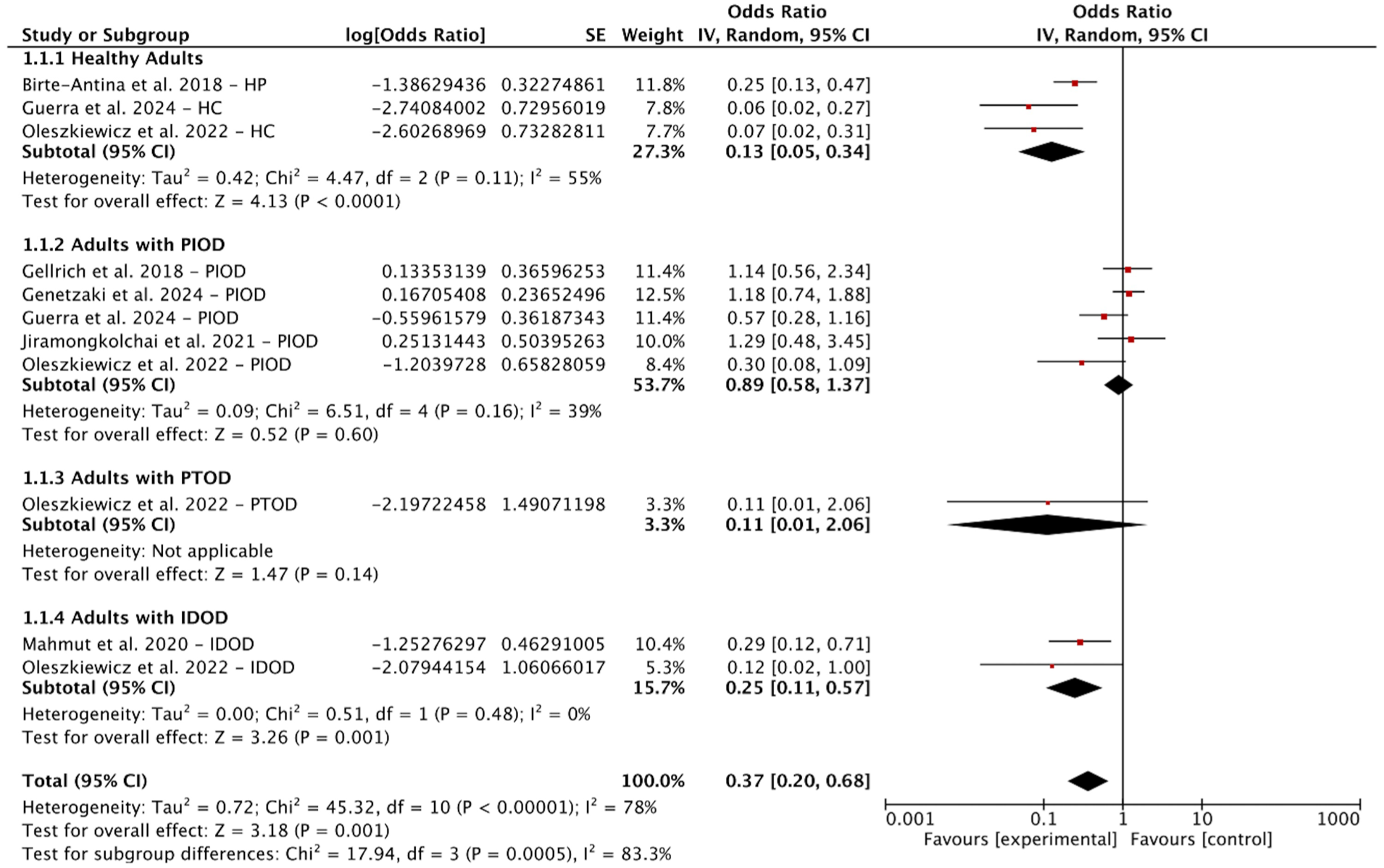
Forest plot for the proportion of clinically-significant results in healthy adults and patients with olfactory dysfunction after olfactory training. CI, confidence interval; HP, healthy population; HC, healthy control; IDOD, idiopathic olfactory dysfunction; PIOD, postinfectious olfactory dysfunction; PTOD, posttraumatic olfactory dysfunction; SE, standard error.
This considerable heterogeneity observed overall (I2 = 78%) indicated that the effect of OT is highly variable. Therefore, we next synthesized the qualitative findings by population subgroup to explore potential sources of this heterogeneity.
Six studies investigated OT among healthy adults who were free from olfactory or cognitive dysfunctions.15,16,30-32 In each study, the Sniffin’ Sticks test battery was used for a comprehensive assessment of olfactory function, and the collected data were analyzed through both intergroup and intragroup comparisons. Four out of 6 studies demonstrated significantly-improved olfactory function following OT, with gains observed in odor threshold (3 studies), discrimination (2 studies), and identification (1 study). Whereas the olfactory outcomes from Birte-Antina et al and Negoias et al arose from classical OT,15,31 Li et al and Oleszkiewicz et al investigated effects of OT utilizing single-molecule odorants or complex mixtures, finding that odor complexity had limited impact on improving olfaction.30,32 Li et al further demonstrated that OT together with multisensory integration and attention to daily odors could improve olfactory function while OT alone did not show this improvement. 30 Al Aïn et al examined the efficacy of a 6 week intensive OT with complex tasks in healthy young people and showed that the OT group obtained better scores in identification task than that of visual training controls. 29 However, a study conducted by Negoias et al demonstrated that following OT, the odor threshold in healthy young individuals was slightly reduced, despite an observed increase in their OB volumes. 31
Thirteen studies included adults with PIOD, PTOD, IDOD, and olfactory dysfunction due to laryngectomy (Supplementary Table 2).22-27,33-39 Improvement in olfactory function was observed across all studies. Six studies investigated OT in individuals with PIOD, with consistent findings that OT enhances olfactory functioning. The reported rates of a clinically-significant improvement ranging from 36.4% to 56.3% are consistent with the pooled estimate of 47.08% from our meta-analysis.22-25,34,35 All 6 studies investigating OT in adults with PIOD reported positive outcomes, with improvements observed in odor threshold (3 studies), discrimination (2 studies), and identification (2 studies). Two studies compared conventional OT with extended OT, which employed a wider range of odors, and found no evidence that increasing odor variety significantly improved outcomes.22,24 Three studies investigated OT in individuals with PTOD. Although all 3 reported improvements in composite TDI scores,33,38,39 none provided data on the rates of a clinically-significant improvement. Two studies explored the effects of OT in individuals with IDOD, both of which demonstrated a significant improvement in odor identification following at least 6 months of training.26,37 Oleszkiewicz et al performed an RCT comparing twice-daily versus 4-times-daily OT in healthy controls and patients with PIOD, PTOD, or IDOD. The results suggested that the classical twice-daily training protocol was more beneficial for improving olfactory outcomes than more frequent administration. 27 Additionally, 1 study by Gürbüz et al indicated that 6 months of OT could improve overall olfactory function in patients who had undergone laryngectomy due to advanced laryngeal cancer. 36
Five studies investigated OT with people with cognitive impairment (Supplementary Table 3).40-44 The findings showed limited evidence for efficacy. Among 3 studies in MCI population, only Haehner et al observed a statistically-significant improvement in odor discrimination within the placebo group following a 4 month intervention. 40 Odor identification was assessed in the 2 studies involving participants with dementia; however, neither study found a significant improvement following intensive OT with a greater number of odorants and prolonged session length.42,43 The quality rating assessed using GRADE was low (Table 1).
Effects of OT on Cognitive Function
A total of 11 studies investigated the effects of OT on cognitive function, including 4 conducted in healthy adults, 2 in individuals with OD, and 5 in individuals with cognitive impairment.15,16,24,27,29-32,40-44 The cognitive domains assessed covered global cognition as well as specific domains including language, learning and memory, attention, executive function, and perceptual-motor function. Overall, the findings present a mixed pattern, with positive outcomes observed selectively in certain domains such as global cognition, language, and working memory, while other areas like attention, planning, and perceptual-motor function remained largely unaffected. However, only 3 studies employed a comprehensive cognitive test battery,40-42 while the remaining studies included only partial cognitive assessments as outcome measures.
Of these 11 studies, all assessed global or general cognitive function using established tests for overall cognitive status, including the Mini-Mental State Examination or the Montreal Cognitive Assessment (MoCA). A significant improvement in global cognition following OT was observed in 3 studies. Oleszkiewicz et al conducted an RCT in healthy older adults and found that the OT group utilizing single-molecule odor improved on MoCA scores (increase of mean value = 0.76 ± 0.37 points, P = .046), while the control group demonstrated a significant increase in cognitive decline symptoms as measured by 8-Item Informant Interview to differentiate Aging and Dementia (increase of mean value = 0.77 ± 0.3 points, P = .01). 32 Another RCT performed by Power Guerra et al showed that OT improved significantly the mean MoCA score in patients with PIOD from 27.22 ± 2.08 to 28.16 ± 1.57 points (P < .001). 24 Lin and Li conducted an RCT in patients diagnosed with mild or moderate dementia. They provided an olfaction-based stimulation therapy that included a sequence of cognitive tasks of growing complexity. Each training session lasted 30 minutes and was performed twice a week for 3 months, with an instructor assisting with the training. Compared with baseline performance, significant cognitive improvements in the Loewenstein Occupational Therapy Cognitive Assessment test were observed in the training group, while poor progression in the level of plasma Aß1-42 was noted in the control group. This suggests that the combination of olfactory and cognitive training can delay cognitive decline. 43 In summary, only a minority of the included studies (3 out of 11) reported significant effectiveness of OT on general cognition, either through within-group pre-post comparisons or between-group analyses, while the remaining studies did not demonstrate such significant benefits.
A total of 8 studies evaluated changes in language function, half of which (n = 4) reported beneficial effects of OT on language outcomes.15,27,30,32,39-42 Verbal fluency was primarily assessed using tests such as the Controlled Oral Word Association Test (COWAT) and other verbal fluency tests, while naming ability was evaluated with the Boston Naming Test. Birte-Antina et al performed classical OT with healthy old people and compared the effect of OT with daily Sudoku tasks. Significant enhancements in the semantic verbal fluency portion of the COWAT (P < .001) were reported after 5 months of OT. 15 Power Guerra et al reported a significant improvement in verbal fluency test in both healthy adults (P < .001) and patients with PIOD (P < .019). 24 Oleszkiewicz et al performed an RCT comparing the effects of 2 different OT regimens (twice daily vs 4 times daily) in healthy controls and patients with PIOD, PTOD, or IDOD. The group that trained twice daily displayed significant enhancement of semantic fluency. 27 Cha et al conducted an RCT in people with dementia. Compared with the non-OT group, OT with 40 aroma oils twice daily for 15 days improved verbal fluency and naming ability. 42
Learning and memory were evaluated in 7 studies, only 2 of which reported positive outcomes following OT intervention.15,16,30,40-42,44 Assessments included standardized instruments such as the Auditory Verbal Learning Test, California Verbal Learning Test, Word List Recall Test, Word List Recognition Test, and the verbal memory subtests from the Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) neuropsychological battery. One study conducted by Woo et al observed a significant improvement in Auditory Verbal Learning Test performance, specifically in the last learning trial A5 (P = .02), among healthy older adults. 16 Additionally, Cha et al demonstrated notable enhancements in Word List Recall Test (P = .031) and Word List Recognition Test (P < .001) in individuals with dementia after IOT. 42
Attention was assessed in 3 studies using the d2 Test of Attention and the Letter-Number Sequencing subtest from the Wechsler Adult Intelligence Scale-III. Results from these studies indicated that OT did not produce significant improvements in attention among either healthy adults or individuals with PIOD.15,16,39
Executive function was evaluated across multiple domains, including general execution, working memory, and planning. With regard to working memory, 3 out of 5 studies reported positive outcomes. Birte-Antina et al observed a significant improvement in the short-term memory section of the MoCA (P < .001) among healthy older adults. 15 Chen et al found significant enhancement in the Wechsler Memory Scale—Revised (P < .05) in individuals with MCI, 41 and Cha et al reported improvement in the Word List Memory Test (P < .001) in patients with dementia. In contrast, Woo et al did not identify a significant change in the Digit Span test among healthy older adults, 16 and Haehner et al also reported no significant improvement in Wechsler Memory Scale-Revised scores among MCI patients.40,42 Furthermore, Chen et al and Haehner et al found no significant change in the Nuremberg Age Inventory in subjects with MCI, indicating a lack of OT effect on planning ability.40,41 Tests such as the Trail Making Test, the 5-Words Test, and the Stroop Color and Word Test were used to assess overall executive function; however, none of these measures yielded significant results following OT intervention.40-42
Perceptual-motor function was evaluated in only 1 study. Cha et al conducted an RCT in individuals with dementia, in which IOT was administered using 40 different aroma oils twice daily for 15 days. However, the intervention did not yield significant improvements in visuospatial abilities, as measured by the Cognitive Performance Test and Constructional Recall Test. 42
In conclusion, the current evidence suggests that the efficacy of OT is domain-specific, showing relatively-consistent positive effects in global cognition and language function, and promising but less consistent benefits in aspects of learning, memory, and working memory. In contrast, attention, executive functions, and visuospatial abilities appear unresponsive to OT based on available studies. The quality rating assessed using GRADE was very low (Table 1).
Effects of OT on Brain Structure and Function
Neuroimaging was assessed in 16 of the studies, including 3 in healthy adults, 11 in individuals with olfactory dysfunction, and 2 in patients with cognitive impairment. The findings are summarized below in terms of structural and functional changes.
Structural Changes were assessed in 10 studies. Regarding OB volume, findings were inconsistent and population-dependent. Significant increases were observed following OT in healthy adults, individuals with IDOD, and individuals with post-laryngectomy.26,36 In contrast, no significant change in OB volume was reported in patients with PIOD or PTOD.22,23,39 Beyond the OB, OT was associated with increased gray matter volume and cortical thickness in various regions. Healthy adults exhibited increase in cortical thickness in the inferior frontal gyrus, inferior temporal gyrus, fusiform gyrus, and entorhinal cortex. 29 Additionally, individuals with PIOD showed volume increases in the thalamus, cerebellum, and hippocampus, 23 while those with PTOD exhibited cortical thickening in frontal regions and the cerebellum. 33 In IDOD patients, gray matter volume increases were noted in the cerebellum, thalamus, precentral gyrus, and several prefrontal areas. 37 In patients with MCI, increased cortical thickness was observed in specific hippocampal subfields. 40
OT also induced widespread functional reorganization, primarily characterized by a shift toward enhanced connectivity and activation within olfactory and cognitive networks. In healthy adults, Woo et al reported increase in the mean diffusivity of the left uncinate fasciculus after OT, indicating improved integrity of a specific brain pathway, that is, crucial to learning and memory. 16 In individuals with PIOD, OT led to increased functional connectivity within olfactory, somatosensory, and integrative networks, alongside a reduction in connectivity to non-olfactory regions such as the prefrontal and premotor cortices. This suggests a refinement of neural resources toward smell processing.25,34,35 Similarly, in PTOD patients, OT modulated effective connectivity, strengthening the connection from the cingulate cortex to the insula and altering self-inhibitory connectivity in the orbitofrontal cortex. 38 Altered activation patterns were also found in the anterior cingulate and superior frontal gyri. 39 In MCI patients, OT enhanced activation in frontal areas critical for cognitive control, including the left middle frontal gyrus and orbitofrontal cortex, and these changes were correlated with improvements in olfactory function. 41
In summary, neuroimaging evidence indicates that OT induced measurable neuroplasticity, with structural changes demonstrating population-specific patterns and functional changes consistently reflecting a reorganization of brain networks toward more efficient olfactory and cognitive processing. The quality rating assessed using GRADE was very low (Table 1).
Discussion
This is the first review to separately summarize the effects of OT across different stages of the continuum of cognitive decline, including healthy adults, adults with olfactory impairment, and adults with cognitive impairment. Our findings demonstrate that OT improved olfactory function across populations, with effect strength varying by group. The most robust response was seen in PIOD (47% clinically improved), followed by IDOD (20%) and healthy adults (11.5%). Furthermore, OT improved cognitive performance, particularly in the domains of global cognition, language, verbal learning, and working memory. In individuals with cognitive impairment, while classical OT showed limited olfactory benefits, intensive OT showed promise in delaying cognitive decline (Figure 5). Neuroimaging evidence further confirmed that OT induced measurable neuroplasticity, encompassing both structural and functional reorganization within olfactory and cognitive networks.
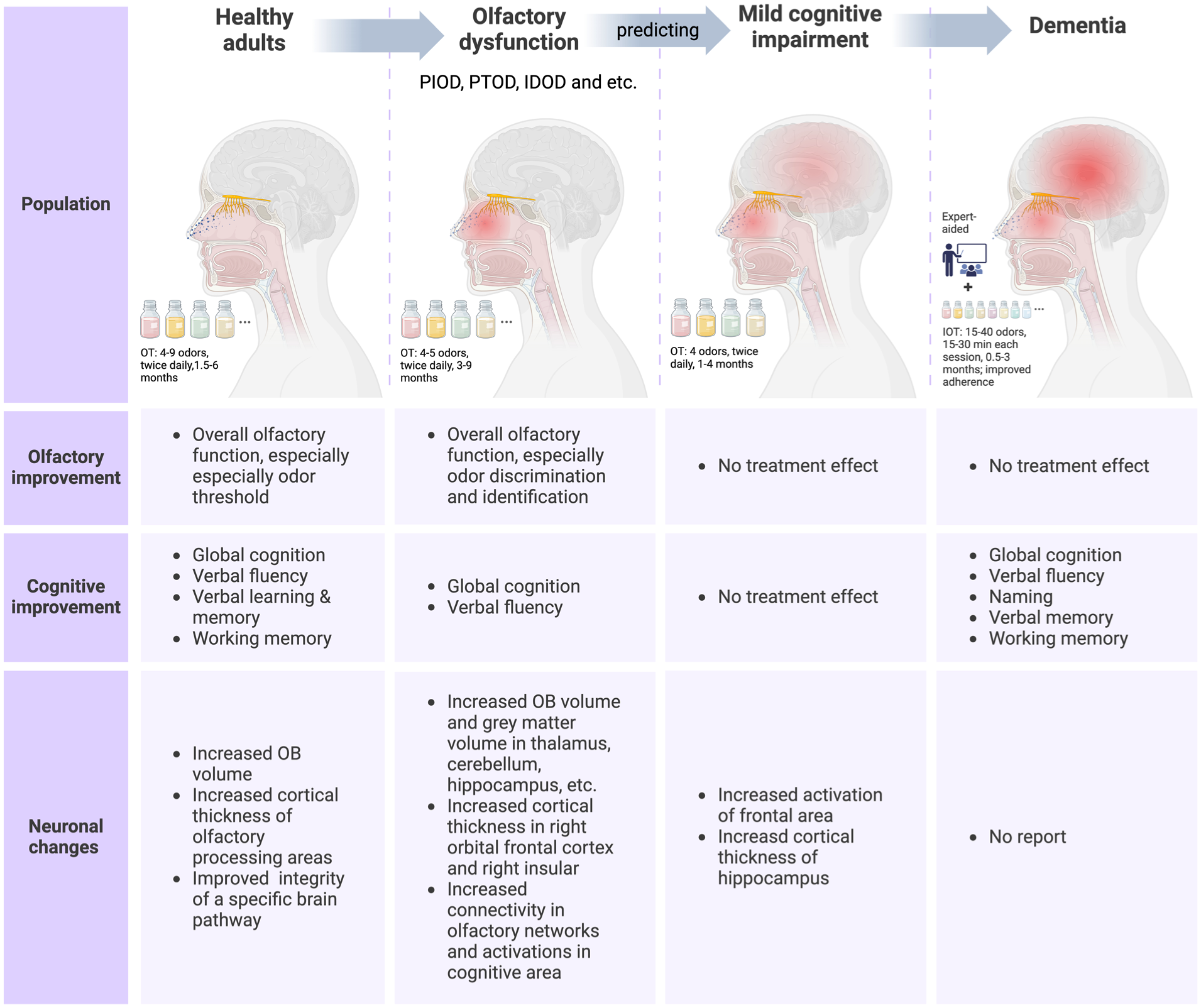
Effects of OT on olfaction and cognition across different populations. The review involved 3 distinct populations: healthy adults, individuals with olfactory dysfunction, and individuals with cognitive impairment, including MCI and dementia. These 3 populations align with specific stages along the trajectory of cognitive impairment: Healthy adults may develop olfactory dysfunction, which can be predictive of progression to MCI, and MCI may eventually progress to dementia. The efficacy of OT as a potential intervention was investigated across different stages of cognitive decline. The olfactory, cognitive, and neuronal outcomes of OT were separately summarized in the figure. IDOD, idiopathic olfactory dysfunction; IOT, intensive olfactory training; OB, olfactory bulb; OT, olfactory training; PIOD, postinfectious olfactory dysfunction; PTOD, posttraumatic olfactory dysfunction.
Previous studies suggest a potential neurological connection exists between olfaction and cognition and olfactory dysfunction is an earliest discernible symptom in neurodegenerative diseases.5,46 The neuroanatomical overlap between olfactory and cognitive systems indicates that the olfactory system is a potential target for cognitive intervention. Previous reviews have provided converging evidence that OT not only improves olfactory function but also enhances brain function, as evidenced by improved cognitive performance, increased volumes in several brain regions, and enhanced neural connectivity.13,17 However, OT’s population-specific effects remain unassessed, and the quality of supporting evidence lacks systematic evaluation.
This review first investigated the effects of OT on healthy individuals, assessing its impact on olfactory function, cognition, and brain structure to establish a baseline for comparison with studies in clinical populations. We found that OT resulted in the most significant improvements in olfactory thresholds. This differs from previous studies focusing on patients with olfactory loss, which found OT exhibited larger effects on cognition-related olfactory domains such as identification and discrimination and had smaller to moderate effects on odor detection thresholds. 14 Whereas odor identification and discrimination rely on higher cognitive abilities, 47 thresholds are more closely related to the peripheral olfactory system. The observed increase in odor threshold in our review supported the hypothesis that OT may enhance the regenerative capacity of the olfactory mucosa in the normosmic population, which showed a bottom-up process. It can be hypothesized that the mechanism of olfactory regeneration during OT involves an initial phase of olfactory receptor stimulation, subsequently activating neurotrophic factors that promote neuronal plasticity. 48 However, this hypothesis requires further validation through electrophysiological and histological studies at the level of the olfactory epithelium. Furthermore, our study population consisted largely of elderly individuals who experienced a more significant decline in odor threshold during the aging process as compared to odor identification and discrimination. 49 Therefore, changes in threshold scores were more readily-detectable post-training. However, the relatively-stable odor identification and discrimination among healthy individuals, with the existence of the ceiling effect, might require an extended version of OT with multiple cognitive stimulations to elicit observable improvements.
We further explored the impact of OT on cognition and brain morphology in healthy adults. Existing studies on the effects of OT on cognition present a mixed picture. While some studies have demonstrated positive effects on global cognition, verbal fluency, verbal learning and memory, and working memory, others have failed to replicate these findings. This inconsistency may stem from the heterogeneity of cognitive assessment tools and variations in OT interventions across studies, as well as methodological limitations in existing RCTs. Assessments of risk of bias in these trials reveal high risk, frequently due to issues such as inadequate randomization and incomplete blinding. Therefore, further research with standardized assessment and rigorous methodological designs that minimize bias is needed to examine OT’s effect on specific cognitive domains. Studies utilizing neuroimaging techniques demonstrated structural brain alterations following OT in healthy adults, offering a potential neural mechanism for olfactory and cognitive improvements. The observed increases in cortical thickness in regions such as the inferior frontal gyrus, an important region involved in language comprehension, speech production, and semantic processing, 50 may explain the improvements in verbal fluency. 29 Similarly, the changes reported in the uncinate fasciculus and medial entorhinal cortex structures critical for learning and memory 51 align with the enhanced performance on learning and memory tasks. 16 Interestingly, these studies did not find a significant correlation between changes in olfactory test scores and brain measures.29,31 A possible explanation is that OT-induced changes in brain structure are mediated by higher level cognitive processes, as supported by a recent study that showed that changes in brain gray matter volume following OT were significantly correlated with a sense of accomplishment and training interest. 52 Further research is needed to elucidate the clinical implications of neuroimaging findings, specifically addressing how structural brain changes relate to improvements in olfaction and cognition. In addition, more microscopic-level research is crucial to explore underlying mechanisms responsible for OT-induced neuroplasticity.
We then summarize the efficacy of OT among patients with olfactory dysfunction and provide further evidence of the neuroplasticity effects of OT. Our findings are consistent with a previous review, reporting a significant improvement in olfaction in patients with olfactory dysfunction, especially in PIOD. 13 We also confirmed that OT may not only cause change at a cognitive level, as evidenced by improved verbal fluency and global cognition scores,24,27 but also induce structural and functional reorganization of brain regions, including the OB, piriform cortex, amygdala, insular cortex, orbitofrontal cortex, entorhinal cortex, hippocampus, and the thalamus. Olfactory disorders involve structural damage across the olfactory system, including peripheral disruption in the olfactory epithelium and central nervous system damage affecting the OB and various cortical areas. The loss of sense input might also lead to reduced sensory-specific connections and reduced connectivity in global brain networks. 18 OT may compensate for these alterations and thus lead to olfactory recovery. At a peripheral level, increased electrophysiological activity at olfactory mucosa has been observed in individuals undertaking OT. 53 Neuroimaging studies also linked OT to structural and functional modifications in the brain. Increased OB volume after OT was observed in patients with IDOD, laryngeal cancer postoperative patients, and healthy individuals,26,36 reflecting the neural plasticity of the primary olfactory system. Structural changes were also detected in brain regions less directly involved in olfactory processing, for example, the thalamus, cerebellum, and hippocampus, indicating that exposure to odor influences the processing and evaluation of olfactory input.23,33,37 For instance, the thalamus is associated with odor attention, the hippocampus is involved in processing odor-related memories, and the cerebellum is activated during the motion of sniffing.54-57 In addition to structural changes, functional neuroanatomical changes were also observed not only within olfactory-related areas but also within higher order olfactory pathways connected to cognitive processes.25,34,35 Increased activation in semantic processing areas was found in patients with PTOD who underwent OT, explaining the improved verbal fluency reported in several studies. 39 This indicates that OT may stimulate extensive neural reorganization in more than just the olfactory areas, and strengthen olfaction as well as cognition, as olfactory and cognitive systems appear to share neural substrates and associations. Our study reaffirms the efficacy of OT for treating olfactory dysfunction. Furthermore, as olfactory impairment is a common precursor to cognitive decline, the cognitive benefits of OT suggest its clinical potential in preventing cognitive decline among individuals at high risk.
We finally explored the therapeutic potential of OT in improving olfactory function as well as delaying or reversing the progression of cognitive decline in patients with cognitive dysfunction, including MCI and dementia. Across all studies, OT failed to demonstrate any significant improvement in olfactory function in both MCI and dementia populations. Our findings suggest that the olfactory deficits may represent irreversible neuronal damage, beyond the potential reparative effects of OT among individuals with MCI and dementia. One hypothesis is that the accumulation of abnormal proteins in the brain contributes to both anosmia and cognitive dysfunction in many individuals experiencing cognitive impairment. This protein deposition may impair migration and proliferation of stroma cells and ultimately limit the regenerative capacity of olfactory neuroepithelium. 58 However, it is important to note that the number of studies investigating OT in populations with cognitive dysfunction limited, and the existing evidence often carries a risk of bias assessed as “some concerns” or “high risk.” Therefore, these findings require cautious interpretation and necessitate further high-quality research for validation.
We next summarized cognitive outcomes following OT in patients with MCI. Global cognition, language, executive function, learning, and memory were assessed, and no treatment effect of OT across these domains was reported. There are several possible explanations for this result. First, amnestic difficulties, prevalent among individuals with MCI, could have led to inconsistent completion of training. Second, the heterogeneity of patients may have weakened the therapeutic effect. Third, improvements in olfaction and cognition may manifest later than months of training (delayed effect) or may occur earlier and subsequently diminish (temporary effect). However, the current follow-up assessment did not capture this potential dynamic change. Although there was no significant improvement in cognitive scores, neuroimaging studies reported local changes in brain structures and function in MCI patients. These encouraging neuroimaging outcomes imply that OT may possess potential cognitive benefits, though more sensitive, multi-timepoint evaluations may be required to detect these subtle alterations. Future studies should also investigate OT effects across etiology-stratified MCI subgroups. Furthermore, more intensive and higher adherence training protocols appear to be essential for individuals with clinical-level cognitive impairment.
Studies in dementia patients, though limited in number, have demonstrated promising outcomes, particularly in enhancing attention, language, and memory function as well as reducing depressive symptoms. Previous reviews reported that passive exposure to odors can trigger autobiographic memory and positive emotions in dementia patients,59,60 consistent with neurological evidence of activation in memories and emotional processing areas including the amygdala, hippocampus, temporal gyrus, and temporal pole during odor exposure.61,62 In addition, the constituents of essential oils may influence mood states by acting on the central nervous or endocrine systems. 63 Emotional benefits of OT were reported in studies of healthy older adults and patients with dementia.15,42 It should be acknowledged that some studies have reported no significant effects of OT on emotional well-being. This discrepancy may result from a ceiling effect, as many studies have excluded participants with preexisting psychiatric disorders. However, given the strong neuroanatomical relationship between mood and cognition, further investigation into the effects of OT on emotion is of significant importance. OT, unlike simple odor exposure, involves a structured program with specific tasks and goals in an interactive setting. This structured and targeted approach may explain its observed benefits in specific cognitive domains, such as language or attention. It should be noted that these encouraging findings of OT stem from the effectiveness of high-intensity training regimens, which are characterized by enriched odors, and extended session length. The experimenters also facilitated training as opposed to the other studies that relied on self-administration, which improved adherence. Furthermore, these studies utilized outcome measures suitable for individuals with dementia, employing simplified or modified versions of assessments to enhance the detection of olfactory and cognitive alterations. Plasma biomarkers linked with cognitive decline and neurodegenerative diseases were also incorporated alongside neuropsychological tests, enabling a more convenient and objective evaluation of OT’s potential to slow disease progression in patients with dementia. Therefore, future research must incorporate simple and sensitive tests for individuals with cognitive impairment to further examine the effects of OT in this population. From a clinical standpoint, our findings indicate that intensive OT is effective in slowing cognitive decline and improving emotion in individuals with dementia, representing an effective and accessible intervention that can enhance their lives.
The limitations of this study included the language bias arising from English-only inclusion and the omission of unpublished studies. Furthermore, the small number of studies included in the quantitative meta-analysis precluded a formal assessment of publication bias. Additionally, the heterogeneity in OT protocols combined with the frequent absence of long-term follow-up and blinded assessments in most trials introduced significant risks of bias. The variability in cognitive assessment tools across studies also limited the quantitative evaluation of cognitive changes. To address these issues, future studies should conduct adequately-powered RCTs with standardized cognitive batteries and validated OT protocols. We further recommend preregistering OT protocols and incorporating multi-timepoint imaging to track neuroplasticity mechanisms.
Conclusion
Accumulating evidence supports the view of OT as a multimodal neurotherapeutic intervention that modulates the chemosensory-cognitive axis through distinct neurobiological pathways, particularly in cognitively-normal populations. OT elicits improvements in olfactory function across diverse populations, including individuals with olfactory dysfunction and healthy adults. Cognitive performance is also enhanced following OT, with notable gains observed in global cognition, language abilities, verbal learning, and working memory. Although classical OT yielded a limited improvement in cognitively-impaired individuals, intensive protocols demonstrated potential to slow cognitive decline. Furthermore, neuroimaging studies consistently showed that OT increases volume in several cognition-related brain regions and promotes neuronal reorganization, suggesting a potential mechanism for OT-mediated olfactory recovery and cognitive improvement. Importantly, our structured evidence synthesis enhances the reliability of these conclusions and provides a robust foundation for future clinical applications.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251384121 – Supplemental material for Effects of Olfactory Training on Olfactory and Cognitive Function: A Systematic Review
Supplemental material, sj-docx-1-ear-10.1177_01455613251384121 for Effects of Olfactory Training on Olfactory and Cognitive Function: A Systematic Review by Yangzi Tan, Xiangxian Liu, Yankun Li, Xiaolu Liu, Yali Du, Danhua Zhao and Dawei Wu in Ear, Nose & Throat Journal
Footnotes
Ethical Considerations
This article does not contain any studies with human or animal participants.
Author Contributions
Yangzi Tan: conceptualization, data curation, formal analysis, methodology, investigation, writing – original draft, writing – review and editing, visualization. Xiangxian Liu: conceptualization, formal analysis, methodology, investigation, writing – review and editing. Yankun Li: methodology, writing – review and editing. Xiaolu Liu: methodology, writing – review and editing. Yali Du: methodology, writing – review and editing. Danhua Zhao: methodology, writing – review and editing, supervision. Dawei Wu: conceptualization, data curation, formal analysis, methodology, investigation, writing – review and editing, visualization, supervision, project administration.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work is financially funded by the key clinical projects of Peking University Third Hospital (BYSYZD2023029), Natural Science Foundation of China (82000954), Beijing Science and Technology Nova Program (Z201100006820086), Beijing Hospitals Authority Youth Program (QML20190617), and Beijing Hospitals Authority Clinical Medicine Development of Special Funding (XMLX202136).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, DW, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
