Abstract
Ortner’s syndrome (cardiac-vocal cord syndrome) is characterized by vocal cord paralysis resulting from underlying cardiovascular conditions. It is typically caused by compression of the left recurrent laryngeal nerve by the pulmonary artery or left atrium, as it passes between the pulmonary artery and aortic arch, leading to left vocal cord paralysis and hoarseness. We report 2 rare cases of hoarseness ultimately diagnosed as Ortner’s syndrome (ages 54 and 71). The first case involved a bicuspid aortic valve with an ascending aortic aneurysm, with a clinical course lasting over 10 years due to the rarity of the condition. The second case involved an aortic pseudoaneurysm. Both patients had resolution of hoarseness and coughing postoperatively. These cases emphasize the importance of considering rare, life-threatening cardiovascular causes in the differential diagnosis of hoarseness, highlighting the need for early diagnosis and intervention.
Keywords
Introduction
Ortner’s syndrome is a rare condition manifesting as hoarseness due to left recurrent laryngeal nerve paralysis, secondary to cardiovascular abnormalities, typically involving compression between the dilated pulmonary artery and aorta. The syndrome is primarily characterized by dysfunction of the recurrent laryngeal nerve, which regulates vocal cord movement through its innervation of the intrinsic laryngeal muscles. 1 The left recurrent laryngeal nerve is more susceptible to injury than the right, given its longer course, and can be affected at multiple anatomical sites. 2 Ortner’s syndrome is commonly associated with pulmonary hypertension, Eisenmenger syndrome, mitral valve regurgitation, patent ductus arteriosus, left ventricular aneurysm, aortic pseudoaneurysm, and aortic intramural hematoma. 3 A prospective study by Loughran et al found that Ortner’s syndrome caused left vocal cord paralysis in 11% of cases, with 27% of these attributed to left atrial enlargement.4,5 Despite its rarity, cases of Ortner’s syndrome underscore the importance of clinical presentation. We report 2 cases of left recurrent laryngeal nerve paralysis caused by an aortic pseudoaneurysm and a bicuspid aortic valve with an ascending aortic aneurysm.
Case Report
Case 1: A 54-year-old female was admitted on May 15, 2013, with a 10-year history of hoarseness, worsening over the past 3 months. The hoarseness was accompanied by mild chest tightness, shortness of breath, and fatigue, without fever or cough. She had no history of surgery, trauma, or other significant medical conditions. Over the past 5 years, she had consulted multiple hospitals, with laryngoscopy revealing left vocal cord paralysis. Despite various tests, including head and neck computed tomography (CT), thyroid ultrasound, and chest X-ray, the cause remained unclear. Treatment with corticosteroids, antibiotics, and traditional Chinese medicine had been ineffective.
On examination, her blood pressure was 121/73 mmHg, pulse 73 bpm, and she appeared alert and well. Chest examination revealed clear bilateral breath sounds, with no dry or wet rales. Heart sounds were normal with a grade I/VI systolic murmur, radiating to the neck. The laryngoscopy showed no neoplasm, but marked immobility of the left vocal cord, with inadequate abduction during inspiration and incomplete closure during phonation (Figure 1).
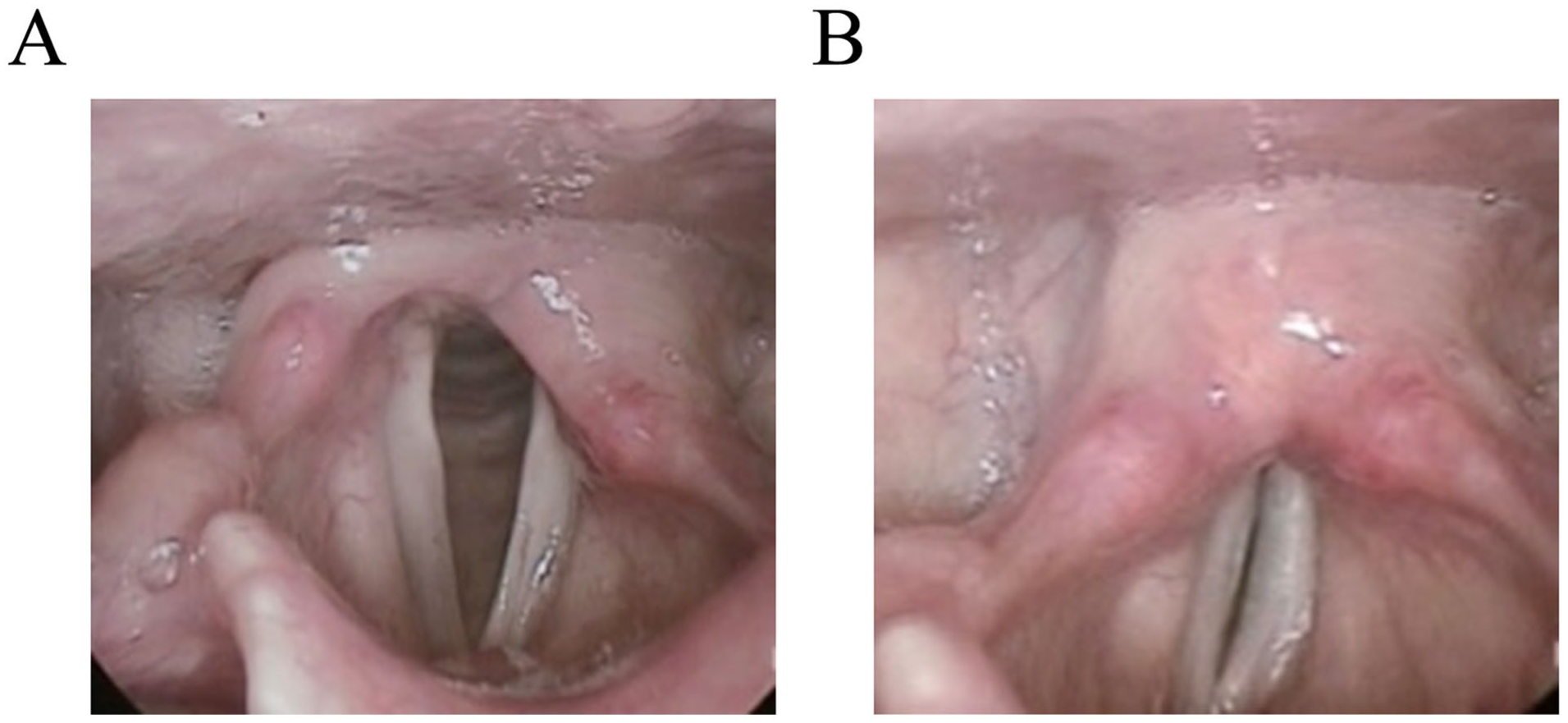
Case 1. Laryngoscopy. During inspiration, the left vocal cord fails to abduct (A); during phonation, vocal cord closure is incomplete (B).
Laboratory tests revealed mild hypertriglyceridemia (5.18 mmol/L), but other results were within normal limits. The electrocardiogram showed sinus rhythm with mild T-wave changes. Cardiac ultrasound revealed a narrowed ascending aorta with aneurysmal dilatation (41 mm), a bicuspid aortic valve with moderate stenosis, and left ventricular diastolic dysfunction. Enhanced chest CT confirmed aneurysmal dilation of the ascending aorta (Figure 2). Brain MRI and magnetic resonance angiography (MRA) showed no abnormalities. The final diagnosis was left vocal cord paralysis (Ortner’s syndrome), congenital heart disease (bicuspid aortic valve with moderate stenosis, ascending aortic aneurysm), and hyperlipidemia. The patient was referred to cardiothoracic surgery, where she underwent aortic valve replacement, ascending aortic replacement, and hemi-arch replacement on August 21, 2013. During surgery, the aortic valve was confirmed to be bicuspid with moderate stenosis, and the ascending aorta had fusiform dilation up to 55 mm. Postoperatively, the patient developed third-degree atrioventricular block and underwent pacemaker implantation. The surgery was successful, and the patient was discharged on September 17, 2013.
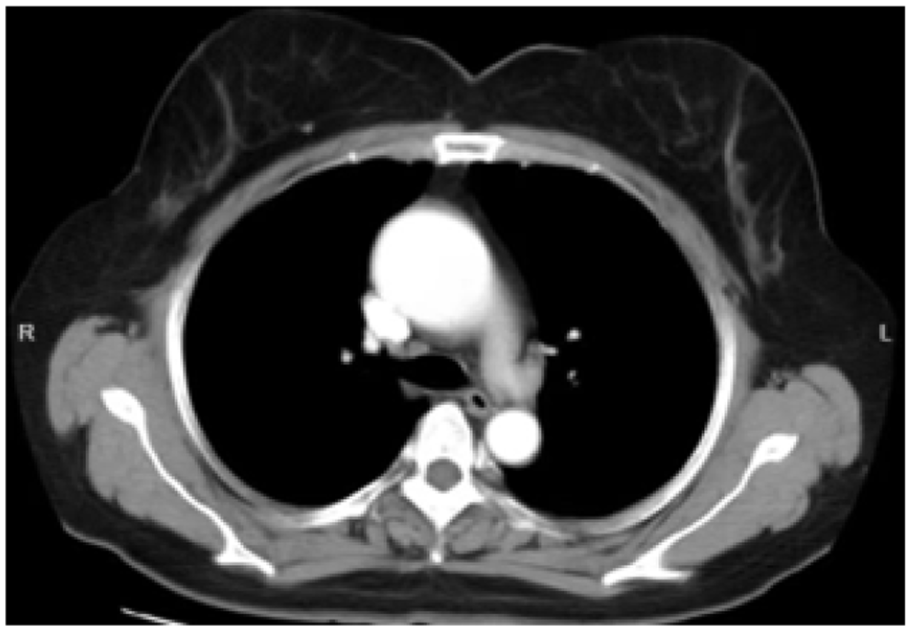
Chest CT (axial view) shows aneurysmal dilation of the thoracic aorta.
Two months post-surgery, the hoarseness gradually resolved. Laryngoscopy showed near-normal left vocal cord movement with good closure. The patient reported no further chest discomfort, shortness of breath, or palpitations. Follow-up at 10 months revealed no recurrence of hoarseness.
Case 2: A 71-year-old female presented to our clinic on May 23, 2025, with a 20-day history of hoarseness, accompanied by coughing during drinking, without fever, cough, or any surgical or traumatic history. Laryngoscopy revealed left vocal cord paralysis, prompting a chest CT scan, which identified a localized bulge in the aortic arch. A subsequent consultation with a cardiothoracic surgeon led to a chest aortic computed tomography angiography (CTA), revealing widening of the ascending aorta, a possible intramural hematoma with early organization in the aortic arch wall, and mild compression of the trachea and esophagus. Follow-up was recommended. Atherosclerosis of the thoracic aorta was also noted (Figure 3). The patient was diagnosed with an aortic arch pseudoaneurysm and admitted for treatment.
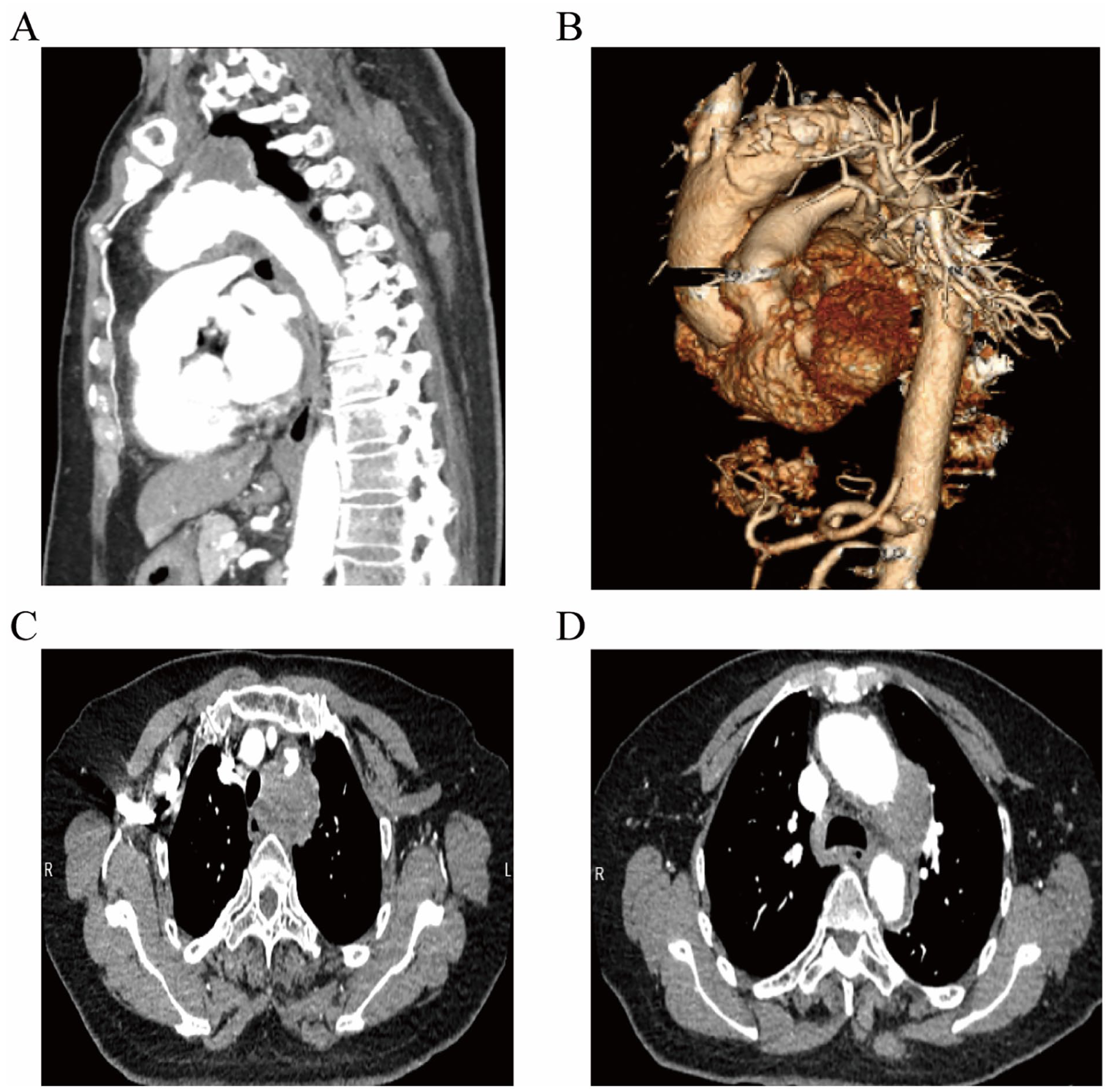
Chest aortic CTA: thickening and irregularity of the aortic arch and descending aorta wall, with inward displacement of calcified intima and surrounding patchy filling defects. (A) Sagittal computed tomographic imaging. (B) Volumetric rendered imaging. (C, D) Axial computed tomographic imaging.
On examination, her blood pressure was 142/92 mmHg (1 mmHg = 0.133 kPa), pulse rate 103 bpm, and she appeared alert and well. Lung examination revealed clear bilateral breath sounds with no rales. Heart sounds were normal, with regular rhythm and no murmurs. Abdominal, limb, and neurological examinations were unremarkable. Laryngoscopy confirmed no neoplasms on either vocal cord, but there was significant reduced mobility of the left vocal cord, with inadequate abduction during inspiration and incomplete closure during phonation (Figure 4).
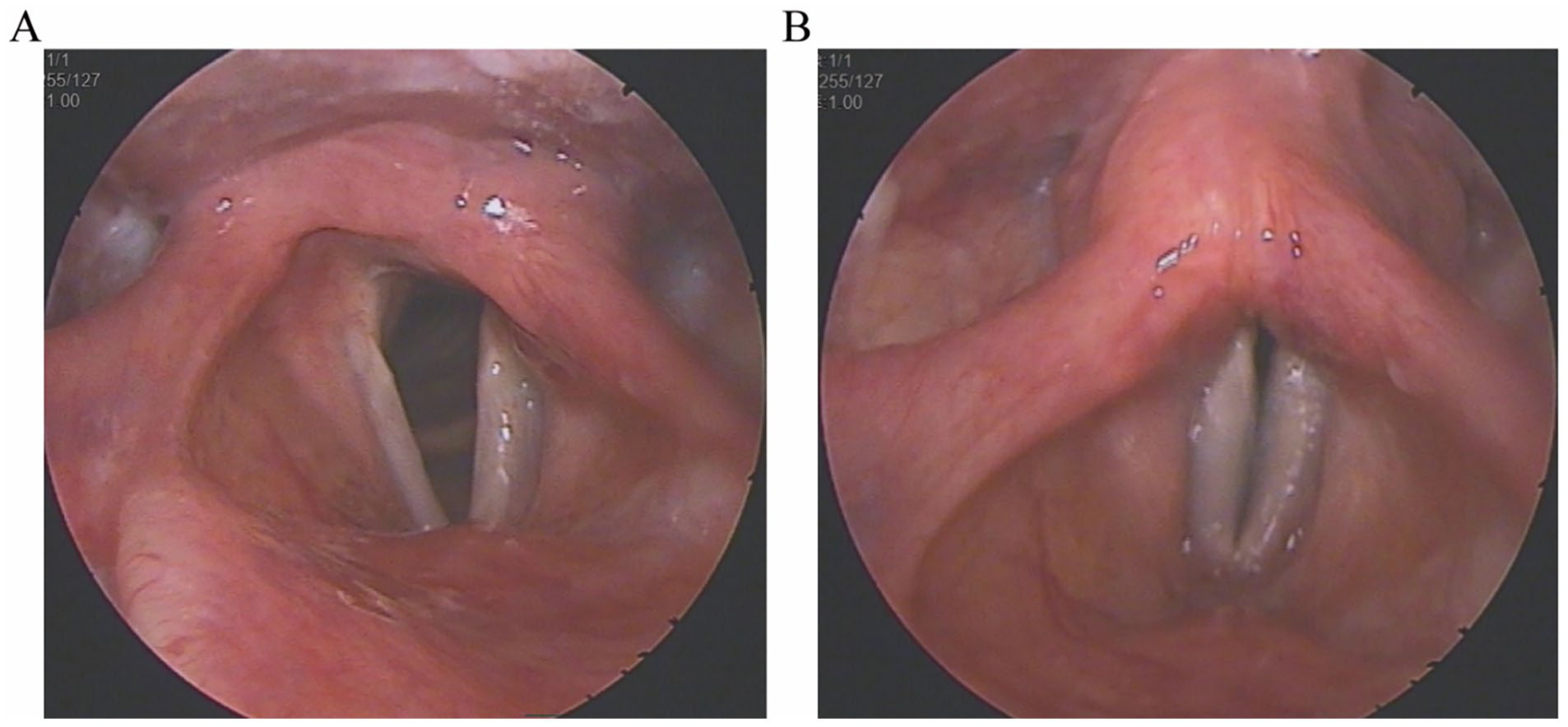
Case 2. Laryngoscopy. During inspiration, the left vocal cord fails to abduct (A); during phonation, vocal cord closure is incomplete (B).
Laboratory tests showed a blood sedimentation rate of 98 mm/hour, CRP of 35.9 mg/L, and serum amyloid A (SAA) >200 mg/L, but no significant abnormalities. Electrocardiogram showed sinus tachycardia and deepening of the Q wave in lead III. Cardiac ultrasound revealed ascending aorta dilation, mild regurgitation of the mitral and pulmonary valves, and impaired left ventricular diastolic function, with an additional hypoechoic lesion in the aortic arch requiring further investigation. Brain CT revealed a left frontal bone osteoma, white matter atrophy, and lacunar infarcts in the bilateral basal ganglia. Abdominal ultrasound showed a possible hepatic hemangioma, liver cysts, and mild dilation of the upper common bile duct, with localized bile duct dilation in the left liver lobe. Additional imaging revealed bilateral carotid and lower limb arterial atherosclerosis.
Coronary CTA revealed calcified plaques in the left coronary artery with mild narrowing, along with non-calcified plaques and mild narrowing in the circumflex artery. Mild atelectasis was seen in the lower lobes of both lungs.
After preoperative preparations, the patient underwent partial aortic arch graft replacement, thoracic aortic stent placement, and open heart surgery with cardiopulmonary bypass on June 9, 2025. Intraoperative findings included dilation of the ascending aorta, an intramural hematoma on the posterior wall of the aortic arch, an ulcer, and atherosclerosis. The surgery was successful, and postoperatively, the hoarseness resolved, and the patient no longer had coughing while drinking. The patient was discharged on June 19, 2025.
Discussion
Hoarseness is typically caused by vocal cord fixation, with a wide range of differential diagnoses. Initial clinical evaluations often focus on a thorough history of significant trauma, surgical interventions, and appropriate investigations to rule out malignancies of the skull base, neck, lungs, and esophagus. Viral or bacterial infections causing recurrent laryngeal nerve neuritis are then considered, with symptomatic treatment provided. 6 In rare instances, hoarseness may be attributed to a cardiovascular condition known as Ortner’s syndrome. Given its rarity, Ortner’s syndrome is seldom included in the early differential diagnosis of hoarseness.
Ortner’s syndrome (also referred to as cardiac-vocal cord syndrome) is caused by the enlargement of the aorta and/or pulmonary artery due to various cardiac diseases, resulting in compression of the left recurrent laryngeal nerve as it passes between the pulmonary artery and beneath the aortic arch. This leads to left vocal cord paralysis and hoarseness.5,7 The condition can affect both children and adults of any gender. Aortic pseudoaneurysm is a rare cause of Ortner’s syndrome, with fewer than 10 cases reported in the medical literature to date. Only 1 case has been associated with the ascending aorta. 8
The delay in diagnosing the first patient was due to: (1) A rare form of Ortner’s syndrome caused by an ascending aortic aneurysm associated with a bicuspid aortic valve and moderate aortic stenosis. (2) The patient’s prolonged hoarseness with slow progression and mild accompanying symptoms, which led to a lack of sufficient attention from the treating physician, who did not pursue relevant investigations. (3) The physician’s habitual diagnostic approach, which overlooked a more detailed analysis of the patient’s history. Given the patient’s lack of surgical or traumatic history and the long duration of the condition, malignancy was considered unlikely, and a more thorough physical examination should have been performed to rule out rare causes.
Treatment outcomes for Ortner’s syndrome are dependent on the extent and duration of nerve damage. A review of Ortner’s syndrome patients found that 85.4% experienced improvement in hoarseness following surgical treatment. 5 Another study showed that most cases had improved hoarseness within 6 months after surgery to repair the pseudoaneurysm or implantation of an endovascular stent, consistent with the postoperative recovery observed in this case report. 9 Currently, surgery remains the most common treatment for Ortner’s syndrome. For cases where aneurysm size or severity does not require repair, angioplasty, thyroidoplasty, and vocal cord medialization are emerging alternatives. 5 A review by Hong et al of 3 Ortner’s syndrome cases reported hoarseness improvement in all patients after laryngeal injection laryngoplasty. 10
Ortner’s syndrome as the primary diagnosis in a patient presenting with hoarseness is uncommon in otolaryngology practice, and published cases with complete clinical and diagnostic documentation are rare. The lack of familiarity with this condition among otolaryngologists is evident, as demonstrated by this case, where the patient underwent repeated head and neck CT scans, chest X-rays, and thyroid ultrasounds in different hospitals. This habitual diagnostic thinking hindered the correct identification of the underlying cause. For unexplained vocal cord paralysis, cardiac investigations should be considered as a potential diagnostic pathway.
Conclusions
Ortner’s syndrome due to a bicuspid aortic valve combined with an ascending aortic aneurysm and aortic pseudoaneurysm is rare. This case underscores the complexity of hoarseness in the differential diagnosis and highlights the importance of recognizing rare cardiovascular causes such as Ortner’s syndrome. It is essential for otolaryngologists to be aware of this syndrome, as it is one of the few conditions associated with both hoarseness and swallowing difficulties, albeit infrequently. Symptomatic patients should be referred to a cardiovascular surgeon for further evaluation and treatment, while asymptomatic patients should be regularly monitored. Otorhinolaryngologists must make an early and accurate diagnosis, integrating clinical findings with imaging characteristics.
Footnotes
Acknowledgements
We would like to thank all reviewers and editors for their professional review work, constructive comments, and valuable suggestions on our article.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Both patients provided written informed consent.
Author Contributions
Q.H.: writing – original draft, writing – review and editing. Y.S.: writing – original draft. L.K.: resources, writing – original draft. Y.Z.: resources, writing – original draft. G.S.: resources, writing – original draft. W.S.: resources, writing – original draft. R.C.: resources, writing – original draft. J.D.: supervision, writing – review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant from the Key Support Project of Jiaxing (no: 2023-FC-004).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available upon reasonable request.
