Abstract
Background:
Laryngopharyngeal reflux (LPR), allergic rhinitis (AR), and asthma are common airway disorders that often coexist, suggesting shared inflammatory mechanisms. LPR involves gastric reflux into the laryngopharynx, while AR and asthma are linked by the “united airway” hypothesis. Evidence indicates LPR may contribute to AR and asthma exacerbation, yet their interactions remain unclear. Understanding their interaction may enhance clinical outcomes.
Objective:
This systematic review aimed to evaluate the associations between LPR, AR, and asthma by analyzing studies that examined these conditions in various patient populations.
Methodology:
A comprehensive search of electronic databases, including PubMed, Scopus, and Google Scholar, was conducted for studies published up until 2024. Eligible studies were selected based on predefined inclusion criteria, and data on the prevalence, diagnostic methods, and associations between LPR, AR, and asthma were extracted. This systematic review was conducted and registered in PROSPERO (CRD42024588367). Statistical analysis was performed to determine the strength of the associations between these conditions.
Results:
The review identified significant associations between LPR and both AR and asthma. Multiple studies confirmed a positive correlation between LPR and AR, with worse AR symptoms observed in patients with more severe LPR. Additionally, a strong association between LPR and asthma was observed, particularly in patients with poorly-controlled asthma. The analysis also revealed a robust relationship between AR and asthma, consistent with the “united airway” hypothesis, which posits that the upper and lower airways share common inflammatory pathways. These findings suggest that the coexistence of these conditions may exacerbate symptoms and complicate management.
Conclusion:
This systematic review highlights the significant associations between LPR, AR, and asthma, emphasizing the importance of recognizing and addressing these comorbidities in clinical practice. The findings suggest that managing 1 condition may have a beneficial effect on the others, supporting a multidisciplinary approach to diagnosis and treatment.
Introduction
Allergic rhinitis (AR), asthma, and laryngopharyngeal reflux (LPR) disease are prevalent chronic conditions that exhibit considerable overlap in their pathophysiology, clinical manifestations, and management strategies. 1 These disorders contribute significantly to the global burden of respiratory and gastrointestinal diseases, underscoring their widespread impact on public health. 2 Although each condition is distinct, their frequent co-occurrence in patients has prompted increasing interest in exploring potential interconnections and shared mechanisms underlying their development . 2
AR is a chronic inflammatory disorder of the nasal mucosa triggered by allergens, leading to symptoms such as sneezing, nasal congestion, and rhinorrhea.3,4 It is closely associated with asthma, a chronic inflammatory disease of the airways characterized by bronchial hyperresponsiveness, airway inflammation, and episodic airflow obstruction. 5 The relationship between AR and asthma is well-documented, with extensive research indicating that AR serves as a significant risk factor for asthma development, particularly in individuals with inadequately-controlled rhinitis.6,7
Conversely, LPR is characterized by the retrograde movement of gastric contents into the laryngopharyngeal region, resulting in symptoms such as chronic throat clearing, hoarseness, cough, and a sensation of a lump in the throat.8,9 While LPR is classified as a subtype of gastroesophageal reflux disease (GERD), its potential role in the pathogenesis of AR and asthma has garnered increasing scientific attention.1,10 Given the anatomical and physiological proximity of the upper airway, esophagus, and lower respiratory tract, it has been hypothesized that reflux may exacerbate or even contribute to the development of both AR and asthma. 10
Emerging evidence suggests a bidirectional relationship among these conditions. AR and asthma may predispose individuals to LPR due to increased intra-abdominal pressure during episodes of coughing and sneezing, which can facilitate reflux.1,11 -13 Conversely, LPR may exacerbate AR and asthma symptoms by inducing inflammation and hypersensitivity in the upper and lower airways. 4
Considering the intricate interplay between these conditions, a comprehensive systematic review is warranted to clarify the nature of their correlation. The present study aimed to synthesize existing evidence to provide a comprehensive understanding of their interrelationship, which may have significant implications for the diagnosis, treatment, and management of individuals with these comorbid conditions. By examining shared pathophysiological mechanisms and potential therapeutic targets, this review seeks to contribute to a more integrated and effective approach to managing these frequently-coexisting disorders.
Despite the high prevalence of AR, asthma, and LPR, the precise nature of their interconnection remains inadequately understood, posing challenges for diagnosis and treatment.2,3 This knowledge gap impedes the development of optimal, evidence-based therapeutic strategies, potentially leading to suboptimal patient outcomes. Addressing this issue, the current study seeks to systematically review and analyze existing literature to elucidate the correlation between AR, asthma, and LPR, thereby providing insights that may enhance clinical management and patient care.
Methodology
Study Design
The study utilized a systematic approach to investigate the correlation between AR, asthma, and LPR disease. The systematic review adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines to ensure a rigorous and transparent process in identifying, selecting, and analyzing relevant studies. This systematic review was preregistered in PROSPERO (CRD42024588367) to ensure methodological transparency and reduce reporting bias.
Eligibility Criteria
The inclusion criteria comprised studies that were randomized controlled trials (RCTs), cohort studies, case-control studies, cross-sectional studies, or retrospective studies. Eligible participants included adults diagnosed with AR, asthma, and/or LPR, and the outcomes of interest included studies reporting the prevalence of, the incidence of, or the correlation between AR, asthma, and LPR, as well as studies exploring pathophysiological mechanisms linking these conditions. Only studies published in English within the past 20 years were included to capture the most recent and relevant data. Exclusion criteria encompassed non-peer-reviewed articles such as editorials, commentaries, and letters to the editor, animal or in vitro studies, and studies lacking clear diagnostic criteria for AR, asthma, or LPR.
Search Strategy
A comprehensive literature search was conducted across several electronic databases, including PubMed, Cochrane Library, EMBASE, Scopus, and Web of Science. The search incorporated a combination of MeSH terms and free-text keywords, such as “Allergic Rhinitis,” “Asthma,” “Laryngopharyngeal Reflux,” “Gastroesophageal Reflux Disease,” “correlation,” “association,” “comorbidity,” and appropriate Boolean operators. The initial search results were imported into reference management software to manage citations and remove duplicates. Two independent reviewers screened the titles and abstracts of identified studies to determine eligibility, followed by a full-text review of potentially-eligible studies based on the inclusion and exclusion criteria. Discrepancies between the reviewers were resolved through discussion or consultation with a third reviewer.
Data Extraction
A standardized data extraction form was developed to collect relevant information from the included studies. Extracted data included study characteristics (author(s), publication year, country, study design, sample size, and follow-up duration), participant characteristics (age, gender, and diagnostic criteria for AR, asthma, and LPR), outcomes (prevalence and incidence rates, correlation coefficients, odds ratios [ORs], clinical outcomes, and management strategies), and methodological quality (risk-of-bias assessment). Two independent reviewers performed data extraction, resolving disagreements through discussion or by involving a third reviewer.
Quality Assessment
The methodological quality of the included studies was assessed using appropriate tools, including the Cochrane Risk of Bias tool for RCTs, the Newcastle-Ottawa Scale for cohort and case-control studies, and the Joanna Briggs Institute critical appraisal Tools for cross-sectional studies. Each study was evaluated for selection bias, performance bias, detection bias, attrition bias, and reporting bias. The risk of bias was categorized as low, moderate, or high, and studies with a high risk of bias were included in a sensitivity analysis to evaluate their impact on the overall findings.
Data Synthesis
A qualitative synthesis was conducted to summarize the findings from the included studies, focusing on the strength and direction of the association between AR, asthma, and LPR, as well as potential shared pathophysiological mechanisms. For the quantitative synthesis, statistical analyses were performed using Review Manager (RevMan), Version 5.4; The Cochrane Collaboration, London, UK and STATA software, Version 17; StataCorp LLC, College Station, TX, USA. Pooled ORs with 95% confidence intervals (CIs) quantified the association between AR, asthma, and LPR. A random-effects model was applied to account for heterogeneity, which was assessed using the I2 statistic and Cochran’s Q test. Subgroup analyses were conducted based on factors such as study design, geographic location, and diagnostic criteria. Publication bias was assessed using funnel plots and Egger’s test.
Ethical Considerations
As the study was based on the analysis of previously-published data, it did not involve direct human or animal participation and did not require ethics approval. Nevertheless, ethical guidelines were followed to ensure the integrity and transparency of the research process, including proper citation of sources and adherence to reporting standards.
Results
Study Characteristics
This review included 14 studies that investigated the association between AR, LPR, and asthma (Figure 1).1,14 -26 The studies included in this analysis encompass a variety of designs, populations, and geographical settings, as summarized in Table 1. Most studies were conducted in Asia (eg, Taiwan, China, and Japan), followed by Europe (eg, Belgium, Spain, and Italy) and the Middle East (eg, Saudi Arabia, Lebanon, and Syria). Sample sizes varied significantly, ranging from small-scale studies such as Komatsu et al with 31 participants 26 to large population-based studies such as Feng et al and Kung et al, each analyzing data for over 36 000 and 193 810 individuals, respectively.20,23
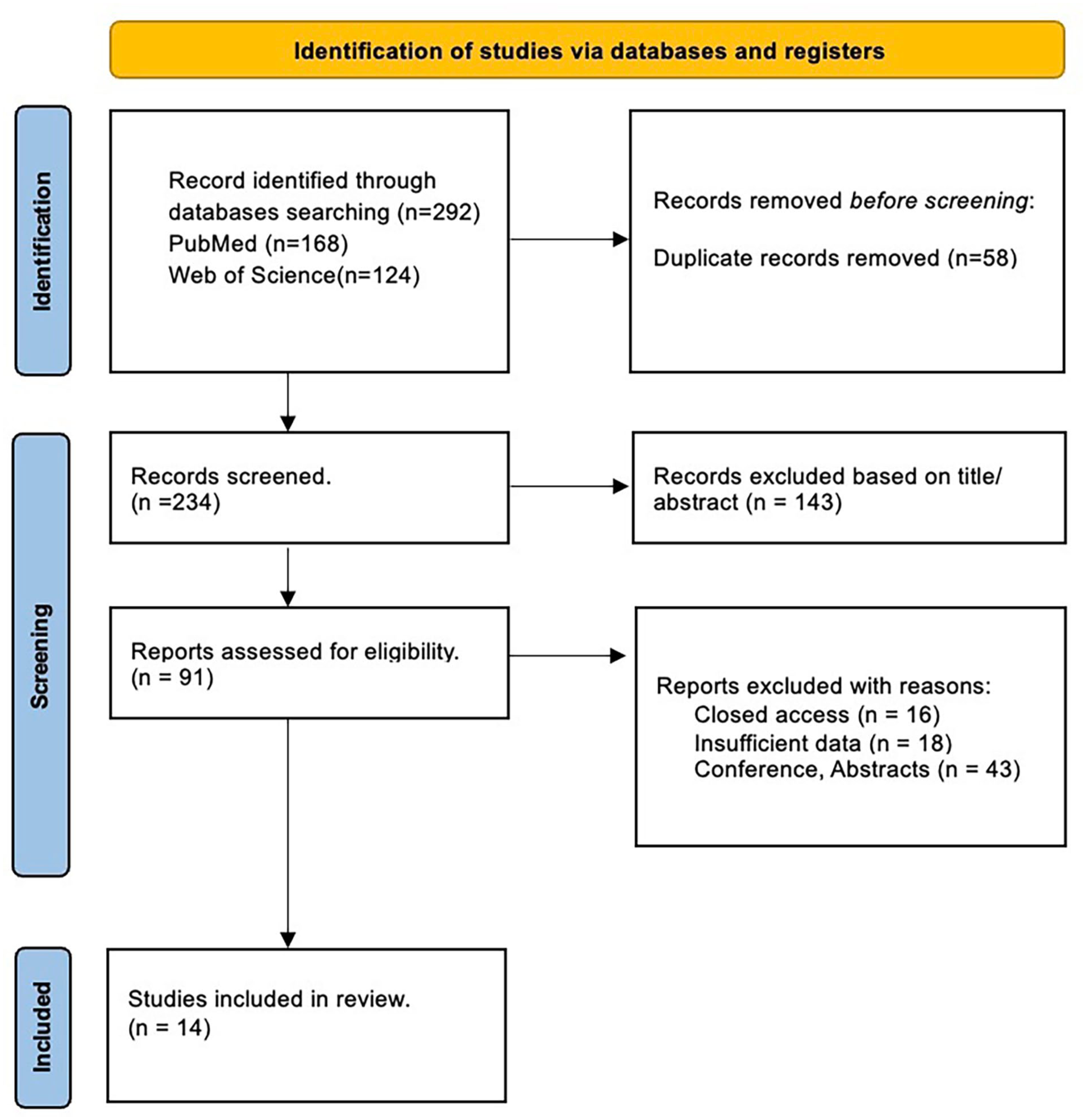
PRISMA figures showing the steps to choose the studies for systematic review. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Study Characteristics.
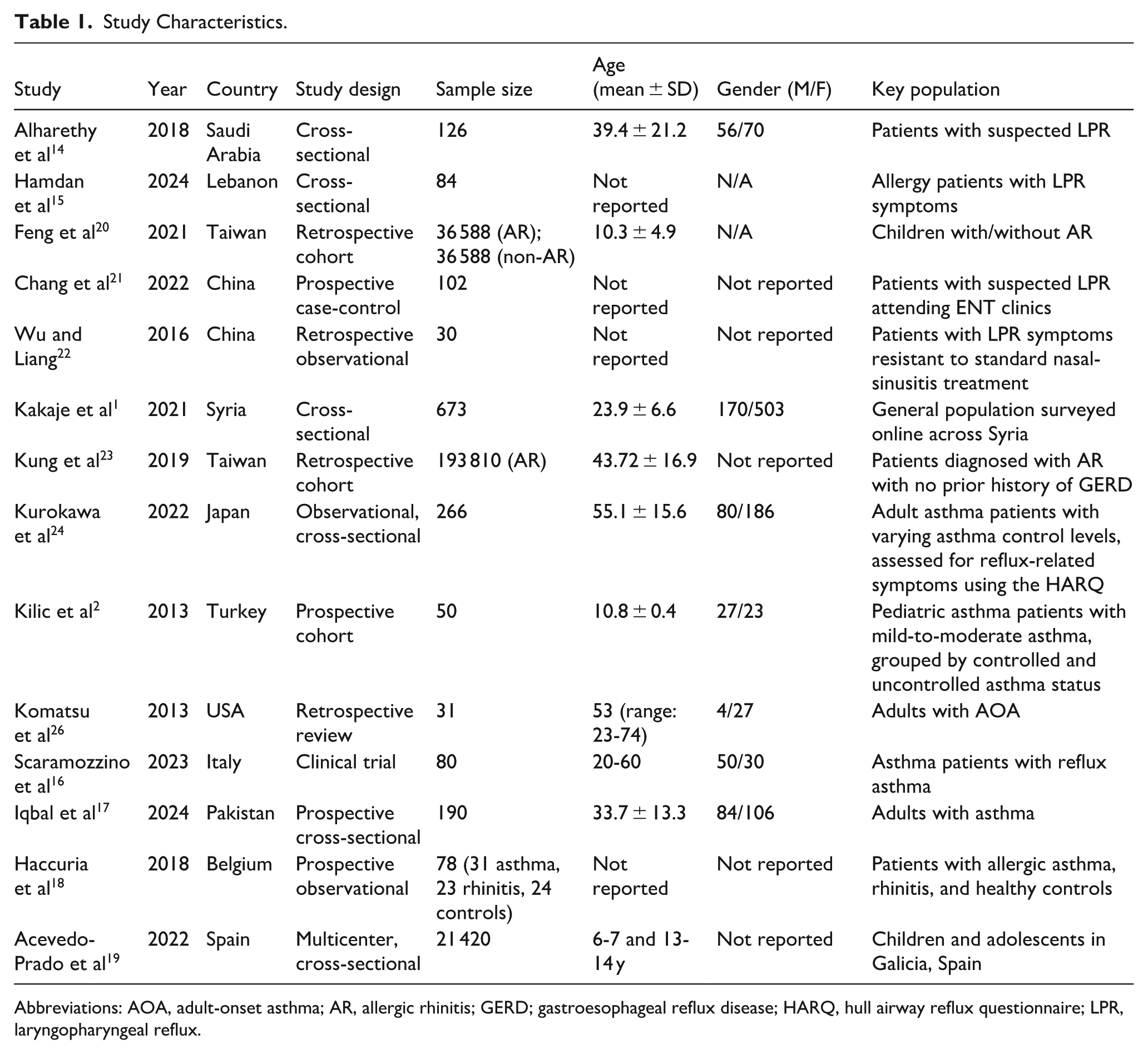
Abbreviations: AOA, adult-onset asthma; AR, allergic rhinitis; GERD; gastroesophageal reflux disease; HARQ, hull airway reflux questionnaire; LPR, laryngopharyngeal reflux.
The studies targeted diverse populations, including patients with specific conditions such as asthma, AR, or LPR (Table 1). Age distributions spanned from pediatric populations20,25 to adults.17,24 Gender representation varied across studies, with a few failing to report gender data. Key populations included general community samples 1 and specific patient groups, such as those with suspected or confirmed LPR 14 or asthma with or without reflux-related symptoms.
Diagnostic Tools and Measures
The methodologies employed for assessing LPR, AR, and asthma varied across studies, as detailed in Table 2. Tools for LPR assessment included symptom indices like the Reflux Symptom Index (RSI) and reflux-specific scores (eg, Hull airway reflux questionnaire [HARQ]) and objective measures such as 24 hour pH monitoring and hypopharyngeal multichannel intraluminal impedance. AR was often evaluated through questionnaires (eg, Score for Allergic Rhinitis [SFAR]) or clinical testing, while asthma diagnosis relied on standardized measures like the Asthma Control Test and spirometry.
Diagnostic Tools and Measures.
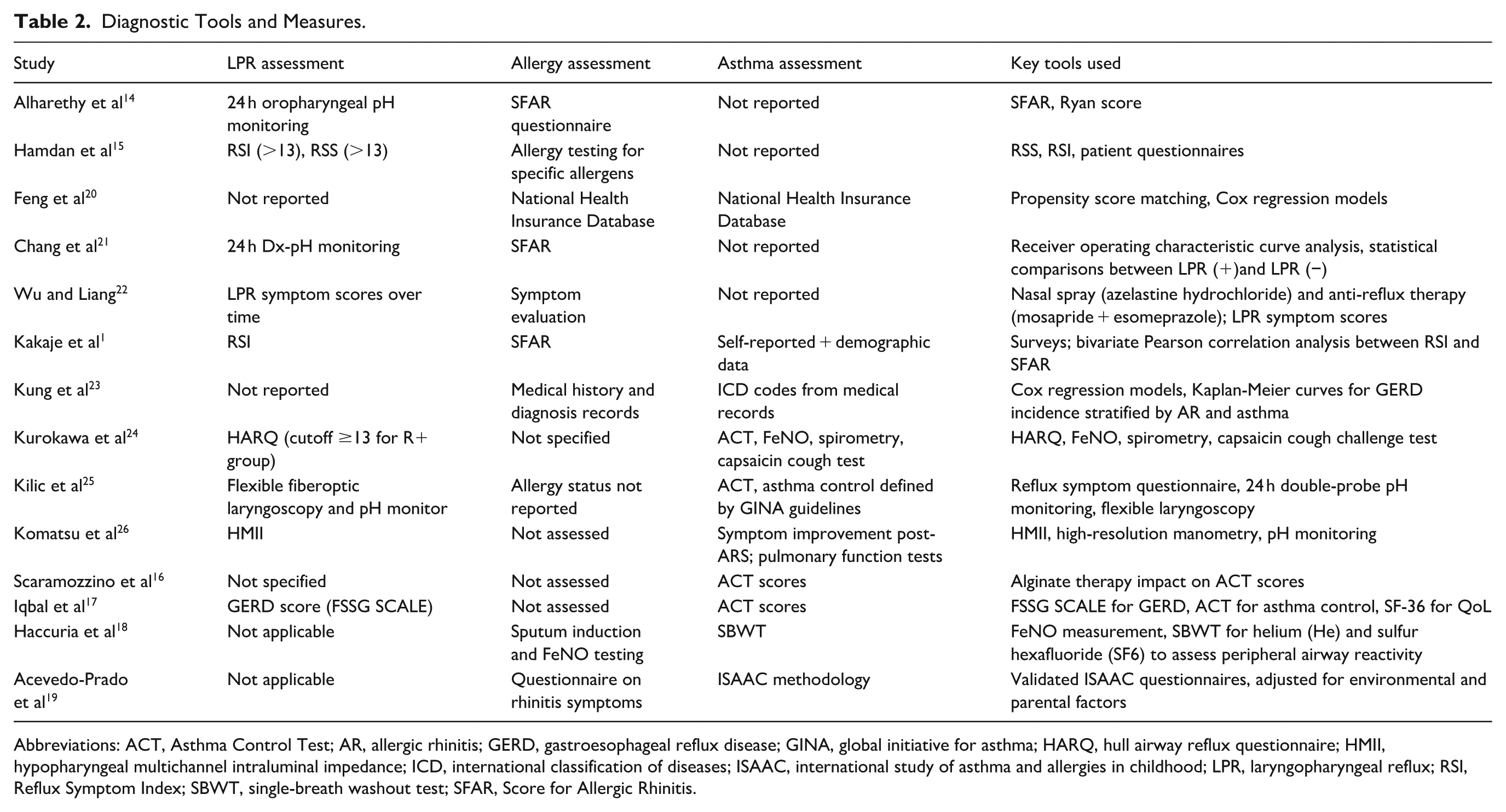
Abbreviations: ACT, Asthma Control Test; AR, allergic rhinitis; GERD, gastroesophageal reflux disease; GINA, global initiative for asthma; HARQ, hull airway reflux questionnaire; HMII, hypopharyngeal multichannel intraluminal impedance; ICD, international classification of diseases; ISAAC, international study of asthma and allergies in childhood; LPR, laryngopharyngeal reflux; RSI, Reflux Symptom Index; SBWT, single-breath washout test; SFAR, Score for Allergic Rhinitis.
Several studies employed advanced statistical models for analysis. For example, Feng et al and Kung et al utilized large administrative databases for retrospective cohort analyses,20,23 while others like Chang et al used prospective designs with receiver operating characteristic curve analysis. 21 Tools such as FeNO and capsaicin cough tests were used in specialized studies like Kurokawa et al to assess associations with reflux symptoms in patients with asthma. 24
Key Associations
LPR and AR Association
The relationship between LPR and AR was consistently observed across several studies (Table 3). Alharethy et al found that patients with LPR were more likely to have higher SFAR scores, indicating a positive association between AR and LPR severity (r = 0.35-0.5). 14 Similarly, Chang et al identified a significant correlation between LPR and AR (OR = 2.372, P < .05), with 60% of LPR (+)patients also having AR. 21 Kakaje et al further demonstrated a significant association between LPR symptoms and AR, with an OR of 2.592 (P < .0001). 1
Key Results.
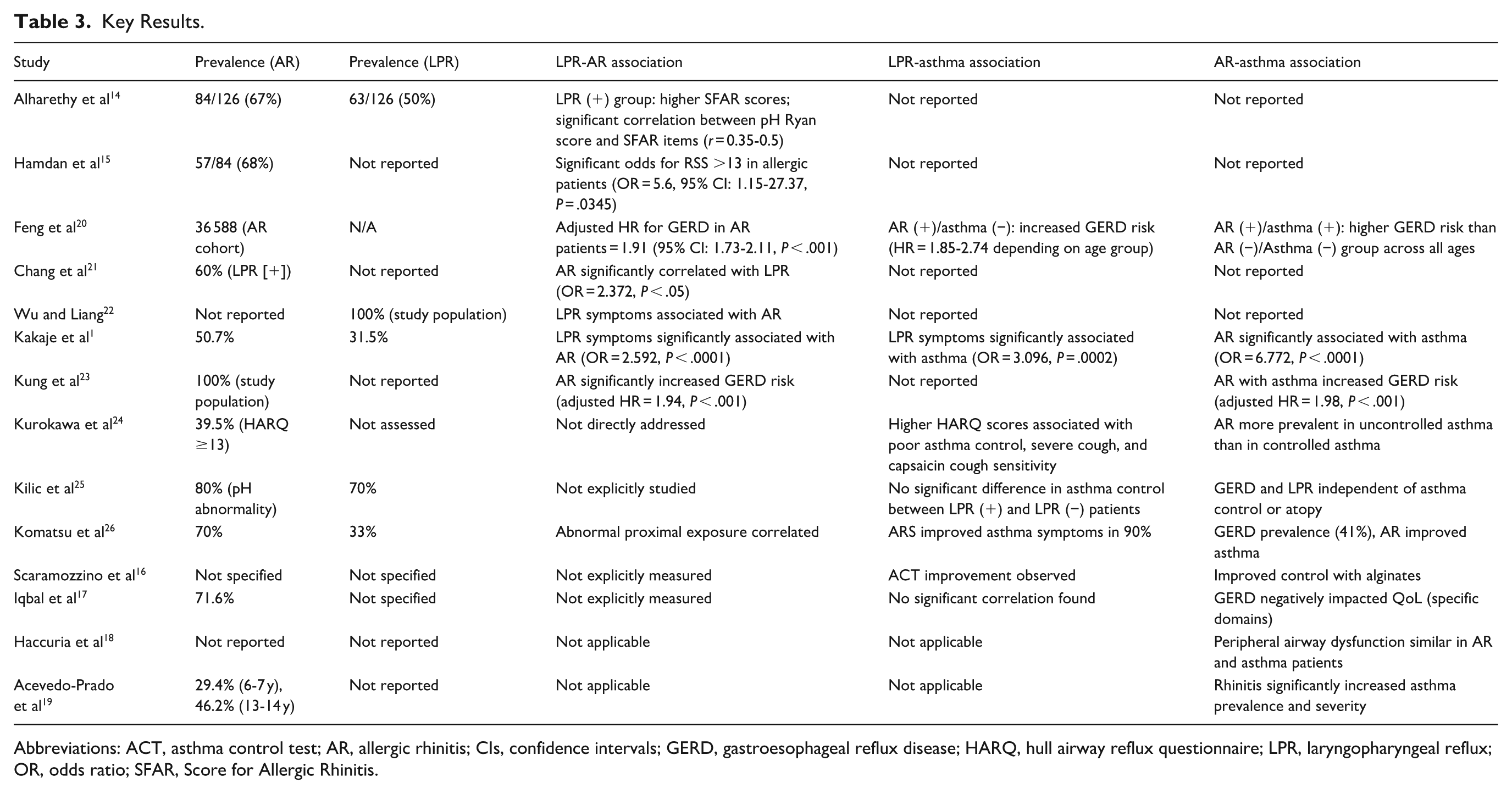
Abbreviations: ACT, asthma control test; AR, allergic rhinitis; CIs, confidence intervals; GERD, gastroesophageal reflux disease; HARQ, hull airway reflux questionnaire; LPR, laryngopharyngeal reflux; OR, odds ratio; SFAR, Score for Allergic Rhinitis.
Studies with larger cohorts also highlighted the connection between AR and LPR. Feng et al observed that AR significantly increased the risk of GERD (adjusted HR = 1.91, 95% CI: 1.73-2.11, P < .001), suggesting a broader association between upper and lower airway inflammatory conditions and reflux disorders. 20
LPR and Asthma Association
The association between LPR and asthma was explored in several studies. Kakaje et al reported a strong correlation between LPR symptoms and asthma (OR = 3.096, P = .0002), underscoring the interplay between reflux and respiratory symptoms. 1 Kurokawa et al identified higher HARQ scores in patients with poorly-controlled asthma, linking LPR symptoms to severe cough and heightened sensitivity to capsaicin cough tests (P < .05). 24
Conversely, Kilic et al found no significant impact of LPR on asthma control in pediatric patients, though abnormal pH monitoring was prevalent in 70% of the sample. 25 This suggests that while LPR is common in asthma patients, its role in asthma severity or control may vary.
AR and Asthma Association
The relationship between AR and asthma was one of the strongest associations observed. Feng et al found that AR combined with asthma increased the risk of GERD more than AR or asthma alone, highlighting the interconnection between these conditions. 20 Kakaje et al demonstrated that AR significantly correlated with asthma, with an OR of 6.772 (P < .0001) . 1
In a pediatric context, Acevedo-Prado et al found that rhinitis increased asthma prevalence and severity, with the highest OR observed for exercise-induced asthma in children aged 6 to 7 years (OR = 11.375). 19 These findings emphasize the strong link between allergic conditions and airway hyperresponsiveness.
Statistical Analysis and Interpretation
The statistical methods and limitations for each study are detailed in Table 4. Across studies, ORs, correlation coefficients, and regression models were the primary tools for evaluating associations. Studies with large sample sizes (eg, Feng et al 20 and Kung et al 23 ) provided robust data but relied on administrative databases, limiting insights into symptom severity. Smaller studies (eg, Kilic et al 25 and Komatsu et al 26 ) offered detailed clinical evaluations but were often underpowered to draw definitive conclusions.
Statistical Analysis and Interpretation.
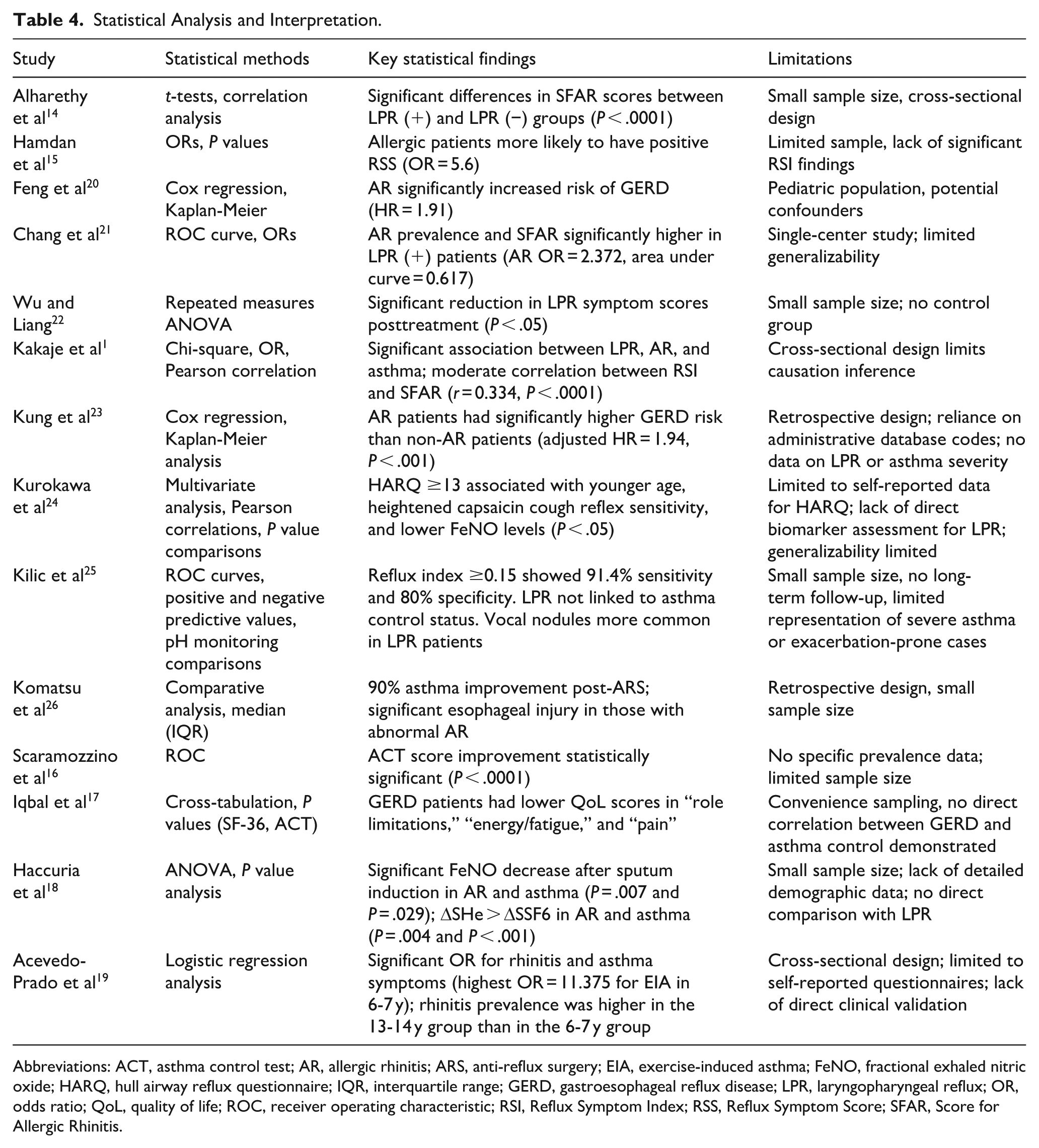
Abbreviations: ACT, asthma control test; AR, allergic rhinitis; ARS, anti-reflux surgery; EIA, exercise-induced asthma; FeNO, fractional exhaled nitric oxide; HARQ, hull airway reflux questionnaire; IQR, interquartile range; GERD, gastroesophageal reflux disease; LPR, laryngopharyngeal reflux; OR, odds ratio; QoL, quality of life; ROC, receiver operating characteristic; RSI, Reflux Symptom Index; RSS, Reflux Symptom Score; SFAR, Score for Allergic Rhinitis.
Key statistical findings include the strong correlation between LPR, AR, and asthma observed in studies like Kakaje et al 1 ), with significant ORs for comorbid conditions. However, limitations such as cross-sectional designs, reliance on self-reported data, and small sample sizes restrict the generalizability of some findings. Overall, the studies highlight the complex interplay between reflux-related disorders, allergic conditions, and asthma.
Risk of Bias Assessment
The risk of bias assessment for the included studies revealed varying levels of concerns across different domains. Several studies, such as those by Alharethy et al and Kung et al, were classified as having “some concerns” overall, primarily due to isolated issues in specific domains. In contrast, studies by Hamdan et al, Feng et al, and Kilic et al were consistently rated with a low risk of bias across most domains, indicating stronger methodological rigor. Notably, domain-specific concerns were observed, such as in D3 and D5, where some studies like Chang et al and Scaramozzino et al demonstrated heightened risks. These findings highlight a mix of high-quality evidence and moderate concerns that necessitate cautious interpretation when integrating findings from these studies into the broader analysis (Figure 2).
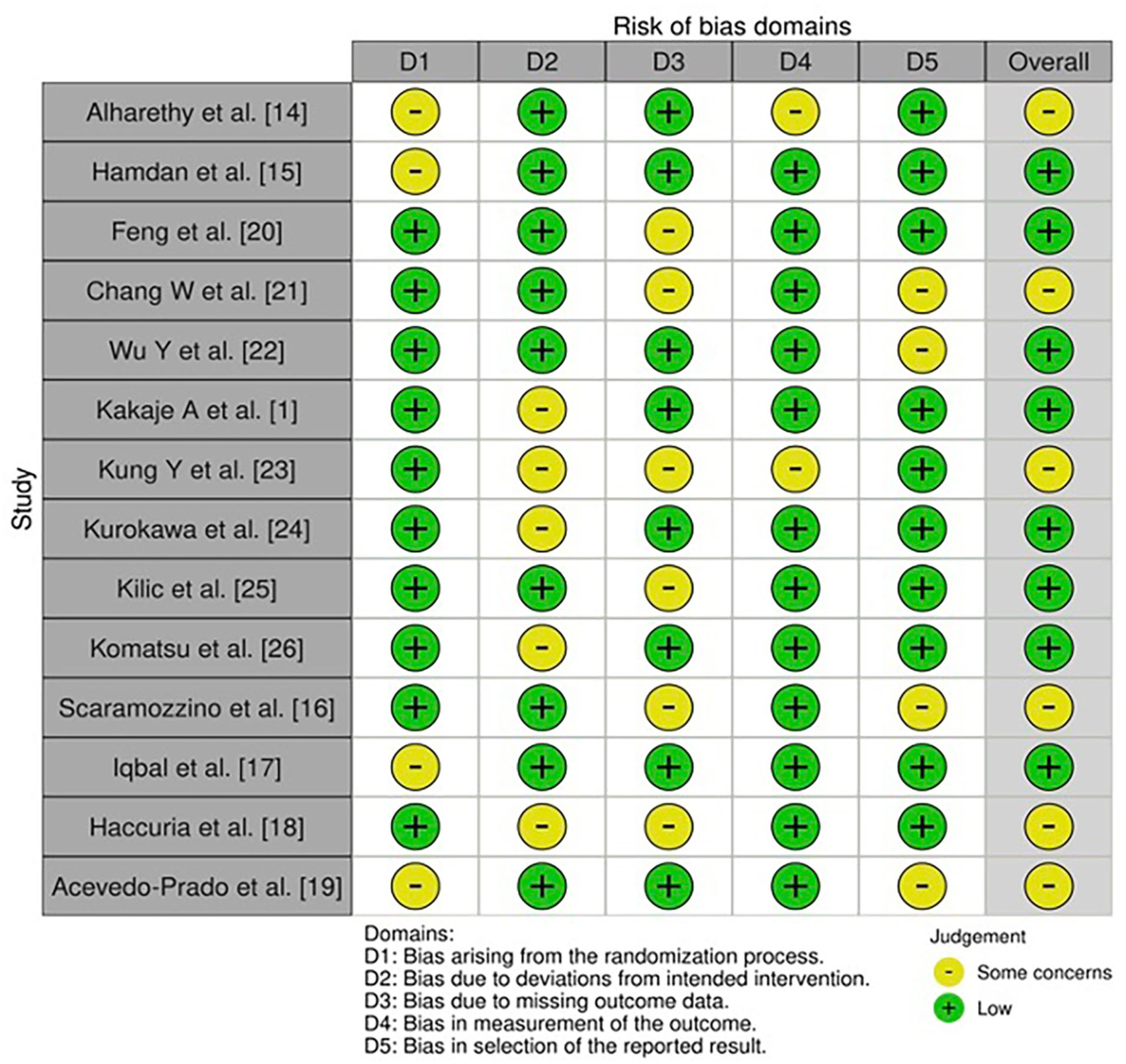
Risk-of-bias assessment among the included studies.
Discussion
The findings of this systematic review highlight significant associations between LPR, AR, and asthma. The results underscore the intricate interplay between reflux-related disorders and respiratory conditions, aligning with previous literature on the shared pathophysiological pathways linking these diseases.27 -30 This discussion synthesizes the observed associations, their potential mechanisms, and their implications for clinical practice and future research.
LPR and AR Association
The positive association between LPR and AR observed in multiple studies supports the hypothesis of a shared inflammatory pathway. Alharethy et al demonstrated a significant correlation between the severity of LPR and AR symptoms, with higher reflux symptom scores associated with worse AR outcomes. 14 This finding aligns with other research suggesting that nasal mucosa inflammation in AR may increase susceptibility to reflux by disrupting the nasopharyngeal barrier.23,31 -33
Moreover, studies have suggested that the acidic content of refluxate in LPR may exacerbate nasal mucosal inflammation, further intensifying AR symptoms.21,34,35 Such bidirectional interactions could explain the high prevalence of AR among LPR patients, as observed in large cohort studies like those by Feng et al. 20 These findings emphasize the need for clinicians to consider upper airway conditions as part of a comprehensive assessment in patients presenting with LPR symptoms.
LPR and Asthma Association
The association between LPR and asthma has been well-documented in the literature,36 -38 and this review corroborates these findings. Kakaje et al reported a significant correlation between LPR and asthma, with an OR indicating a strong association between reflux symptoms and respiratory conditions. 1 The pathophysiological basis for this relationship is thought to involve micro aspiration of gastric contents and vagally-mediated reflexes, both of which can exacerbate bronchial hyperresponsiveness.10,39
Interestingly, Kurokawa et al provided further evidence by showing that LPR symptoms were more severe in patients with poorly-controlled asthma, suggesting that uncontrolled reflux may contribute to asthma exacerbations. 24 However, the heterogeneity in diagnostic methods and patient populations across studies complicates the interpretation of this association. For example, Kilic et al found no significant impact of LPR on asthma control in pediatric patients, highlighting the potential age-dependent differences in the LPR-asthma relationship. 25
AR and Asthma Association
The robust association between AR and asthma is consistent with the “united airway” hypothesis, which posits that the upper and lower airways function as a continuous inflammatory unit.40 -42 Feng et al observed that AR significantly increased the risk of asthma, and the presence of both conditions further heightened the likelihood of GERD. 20 This finding is consistent with previous studies, which reported that AR patients had a higher risk of asthma.43,44
The strong correlation between AR and asthma observed in this review may be attributed to shared immunopathological mechanisms. AR triggers systemic inflammation and the release of cytokines, such as IL-4 and IL-13, which can exacerbate bronchial hyperresponsiveness and lead to asthma symptoms.45,46 Additionally, untreated AR has been shown to impair asthma control, further supporting the need for an integrated approach to managing these comorbid conditions.
Role of Nonacid Components in the Unified Airway
Growing evidence indicates that the nonacid constituents of refluxate particularly pepsin and bile acids may represent a central pathogenic link between upper and lower airway disease, extending even to distal lung pathology such as idiopathic pulmonary fibrosis (IPF). Pepsin has been identified in the nasal cavity, middle ear, lungs, and bronchoalveolar lavage fluid of patients with IPF, despite being synthesized only in the stomach. Its detection in these distal sites provides strong evidence of microaspiration from silent LPR, capable of initiating epithelial injury, sustaining chronic inflammation, and promoting fibrotic remodeling independent of acid exposure.47,48 Bile acids, frequently co-present with pepsin, have shown synergistic epithelial toxicity, amplifying inflammatory cascades and disrupting barrier integrity. 49 These observations align with the unified airway paradigm, suggesting that silent, nonacid LPR may serve as a persistent and underrecognized insult linking LPR, AR, asthma, and potentially IPF. Expanding diagnostic strategies beyond acid detection to identify and address nonacid reflux could therefore have implications not only for upper airway disease control but also for preventing progression of certain chronic lung diseases.
Implications for Clinical Practice
The overlapping symptoms and mechanisms of LPR, AR, and asthma call for a multidisciplinary approach to diagnosis and management. Screening for AR and asthma in patients with LPR, and vice versa, may improve patient outcomes by enabling early identification of comorbidities. Additionally, integrating gastroenterologists, pulmonologists, and otolaryngologists in patient management may optimize therapeutic interventions. Addressing 1 condition such as reducing reflux symptoms may help alleviate symptoms of the other conditions through shared inflammatory pathways. Management strategies should also consider the limitations of current diagnostic tools. For example, the reliance on symptom scores like the RSI or HARQ may underestimate the prevalence of asymptomatic reflux in asthma patients. Future studies should explore more robust biomarkers and objective diagnostic criteria to better delineate the relationships between these conditions.
Limitations and Future Research
Despite the compelling evidence, this review also highlights several limitations in the existing literature. Many studies were cross-sectional or retrospective, limiting causal inferences about the observed associations. Additionally, the variability in diagnostic tools and population characteristics across studies may have introduced heterogeneity into the results.
Future research should prioritize longitudinal studies with standardized diagnostic criteria to elucidate the causal pathways linking LPR, AR, and asthma. Investigating the role of novel biomarkers and genetic predispositions may also provide deeper insights into the mechanisms underlying these associations. Moreover, interventional studies evaluating the impact of treating 1 condition (eg, LPR) on the severity and control of comorbid AR or asthma could help establish evidence-based guidelines for integrated management.
Conclusion
The findings of this review highlight the significant associations between LPR, AR, and asthma, emphasizing the importance of a holistic and interdisciplinary approach to airway disease management. Recognizing and addressing the interplay between these conditions could improve patient outcomes and reduce the burden of respiratory and reflux-related disorders. Future studies should aim to address the limitations of current research and further clarify the mechanisms linking these complex conditions.
Footnotes
Ethical Considerations
This systematic review utilized previously-published data and did not involve human or animal subjects directly.
Author Contributions
L.S.A. conducted the literature search, creating data extracting sheet, introduction writing, and resolved any outstanding disagreements in screening. E.M.A. extracted data, methods writing, and revising the results. H.M.A. screened literatures and discussion writing. N.K.N. screened literatures, wrote the conclusion, and abstract formulation. Y.A.A. both wrote the discussion and references. B.I.A. and M.H. resolving any outstanding disagreements in screening, reviewed, and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
