Abstract
Purpose of Review:
The purpose of the review is to summarize current literature and evaluate the clinical implications of chronic rhinosinusitis (CRS) endotypes.
Major Findings:
Evolving understanding of CRS endotypes has reshaped the approach to diagnosis and treatment, from a simple phenotypic classification to a more precise molecular and immunologic framework. The differentiation between type 2 and non-type 2 inflammation has led to the development of targeted and precise therapeutic approaches, particularly with the introduction of biologic agents.
Conclusions:
Future advances in biomarker identification and personalized medicine will continue to further refine CRS management by supporting individualized, endotype-specific therapies that improve treatment outcomes.
Keywords
Introduction
Chronic rhinosinusitis (CRS) is a complex, heterogeneous inflammatory condition affecting the nasal and paranasal mucosa. Historically, CRS was classified based on the presence or absence of nasal polyps, dividing it into CRS with nasal polyps (CRSwNP) and CRS without nasal polyps (CRSsNP).1,2 While this dichotomy provided a broad clinical framework, it failed to capture the underlying immunologic and molecular heterogeneity that influences disease presentation and treatment response.3,4 Recent advances in molecular biology and immunopathology have led to a paradigm shift, identifying distinct CRS endotypes based on inflammatory signatures and cytokine profiles rather than solely on phenotypic features.5,6
The molecular biology of CRS endotypes has revealed a diverse range of inflammatory patterns, which are broadly categorized into type 2 (T helper 2 [Th2]-mediated immune response) and non-type 2 inflammation. Type 2 CRS, predominantly seen in CRSwNP, is characterized by eosinophilic infiltration, elevated levels of interleukin-4 (IL-4), IL-5, IL-13, and frequent comorbidities such as asthma and allergic rhinitis.7,8 Non-type 2 CRS, often observed in CRSsNP and some subsets of CRSwNP, is driven by neutrophilic or mixed inflammation, with increased levels of IL-1β, IL-6, tumor necrosis factor-α (TNF-α), and interferon-γ.9 -11 Further subtypes have also been proposed, including type 3 inflammation with fungal or bacterial dysbiosis and an emerging role for epithelial barrier dysfunction.12 -14
These endotypes have significant clinical implications, especially for the development of targeted biologic therapies. While traditional treatments, such as antibiotics and surgery, provide symptomatic relief, they often fail to address the underlying inflammatory mechanisms, leading to high recurrence rates.15,16 By globally blunting the immune system, oral corticosteroids are generally effective in attenuating inflammation in CRS; however, the systemic side effects prevent their use as a maintenance therapy for CRS. The introduction of biologic agents targeting key cytokines in type 2 inflammation, such as IL-4Rα inhibitors (dupilumab), IL-5 antagonists (mepolizumab, reslizumab), and anti-immunoglobulin E (IgE) therapies (omalizumab), has revolutionized CRS management by offering personalized, disease-modifying options.17 -19 Ongoing research continues to refine CRS endotyping to enhance treatment selection and improve patient outcomes.20,21
This review explores the evolution of CRS classification, details the molecular mechanisms characterizing each endotype, and discusses the clinical implications of these findings, particularly in the era of biologic therapy.
Materials and Methods
A comprehensive review of the literature was done using Scopus, PubMed, Cochrane database, and Google Scholar. The databases were searched for the keywords “chronic rhinosinusitis,” “sinusitis endotypes,” “eosinophilic chronic sinusitis,” “non-eosinophilic sinusitis,” “central compartment atopic disease,” “aspirin exacerbated respiratory disease,” “allergic fungal rhinosinusitis,” and “biologic treatments for sinusitis.” In addition, we examined the reference lists of the articles included to identify further relevant articles. Articles written in languages other than English, case reports, and expert opinions were excluded.
An ethical approval was not required for this study, since it is a literature review and does not involve the collection of new data from human or animal subjects.
Discussion
The understanding of CRS endotypes and their associated phenotypes has evolved significantly over the years. In 2016, Tomassen et al 22 proposed 10 distinct clusters based on cytokine affinity and clinical presentation. Clusters 1 to 4 were characterized by a stronger correlation with IL-1β and IL-17, along with lower rates of nasal polyps (11%-47%) and asthma (7%-20%). In contrast, clusters 5 to 10 exhibited elevated levels of IL-5, IgE, and TNF-α, as well as a higher prevalence of nasal polyposis (59%-100%) and asthma (27%-71%).
While this cytokine-based classification successfully integrated molecular and clinical characteristics, it did not fully account for well-recognized subtypes such as aspirin-exacerbated respiratory disease (AERD), central compartment allergic disease (CCAD), and allergic fungal rhinosinusitis (AFRS). Consequently, more recent guidelines have refined CRS classification into 5 primary endotypes: eosinophilic CRS (eCRS), CCAD, AERD, AFRS, and non-eCRS. 1 This classification places a stronger emphasis on the clinical presentation rather than the pathophysiology on the molecular level.
Eosinophilic CRS
eCRS is a subset of CRS marked by a strong Th2-mediated immune response, resulting in chronic inflammation driven by eosinophils. It is commonly associated with severe polyposis, frequent recurrence after surgery, and poor response to corticosteroids.11,23 The hallmark cytokines involved in eCRS pathogenesis include IL-4, IL-5, and IL-13, which collectively drive eosinophilic recruitment, mucus hypersecretion, and epithelial barrier dysfunction.9,24 Elevated IL-5 levels correlate with increased polyp burden and persistent inflammation, making it a key therapeutic target. 2 Patients with eCRS frequently present with nasal obstruction, anosmia, and thick, tenacious mucus, often requiring multiple courses of corticosteroids and revision surgeries due to high recurrence rates.25,26 A subset of patients also exhibits peripheral eosinophilia and increased IgE levels, further reinforcing its atopic and systemic inflammatory nature. 2 Studies indicate that local IgE production in nasal polyps contributes to disease severity, even in patients without systemic atopy. 19 Compared to patients with non-eCRS, those with eCRS are often characterized by a more severe disease burden, including higher polyp scores and increased rates of anosmia. 27 From a surgical perspective, patients with eCRS exhibit more pronounced mucosal inflammation and osteitis than those without eCRS. 28
Histopathologically, eCRS is characterized by dense eosinophilic infiltration in the sinonasal mucosa, elevated periostin levels, and increased expression of eotaxins, which facilitate eosinophil migration.19,24 Recent findings suggest that epithelial barrier dysfunction, mediated by decreased expression of tight junction proteins such as claudin-1 and occludin, contributes to chronic inflammation in eCRS.5,11 In addition, increased expression of thymic stromal lymphopoietin in eCRS suggests that epithelial-derived cytokines play a critical role in promoting Th2 inflammation.8,13
The treatment landscape for eCRS has evolved with the introduction of biologic therapies targeting key inflammatory pathways. Monoclonal antibodies such as dupilumab (IL-4/IL-13 inhibitor), mepolizumab (IL-5 inhibitor), and benralizumab (IL-5 receptor antagonist) have demonstrated efficacy in reducing nasal polyp burden, improving olfaction, and decreasing the need for systemic corticosteroids.23,29 Notably, dupilumab has significantly reduced sinonasal inflammation and improved quality of life for patients with severe eCRS.8,9,13,30 These targeted therapies offer promising alternatives for patients with refractory disease, shifting management toward a more precisely tailored treatment (Table 1).
Landmark and Recent Studies on eCRS.
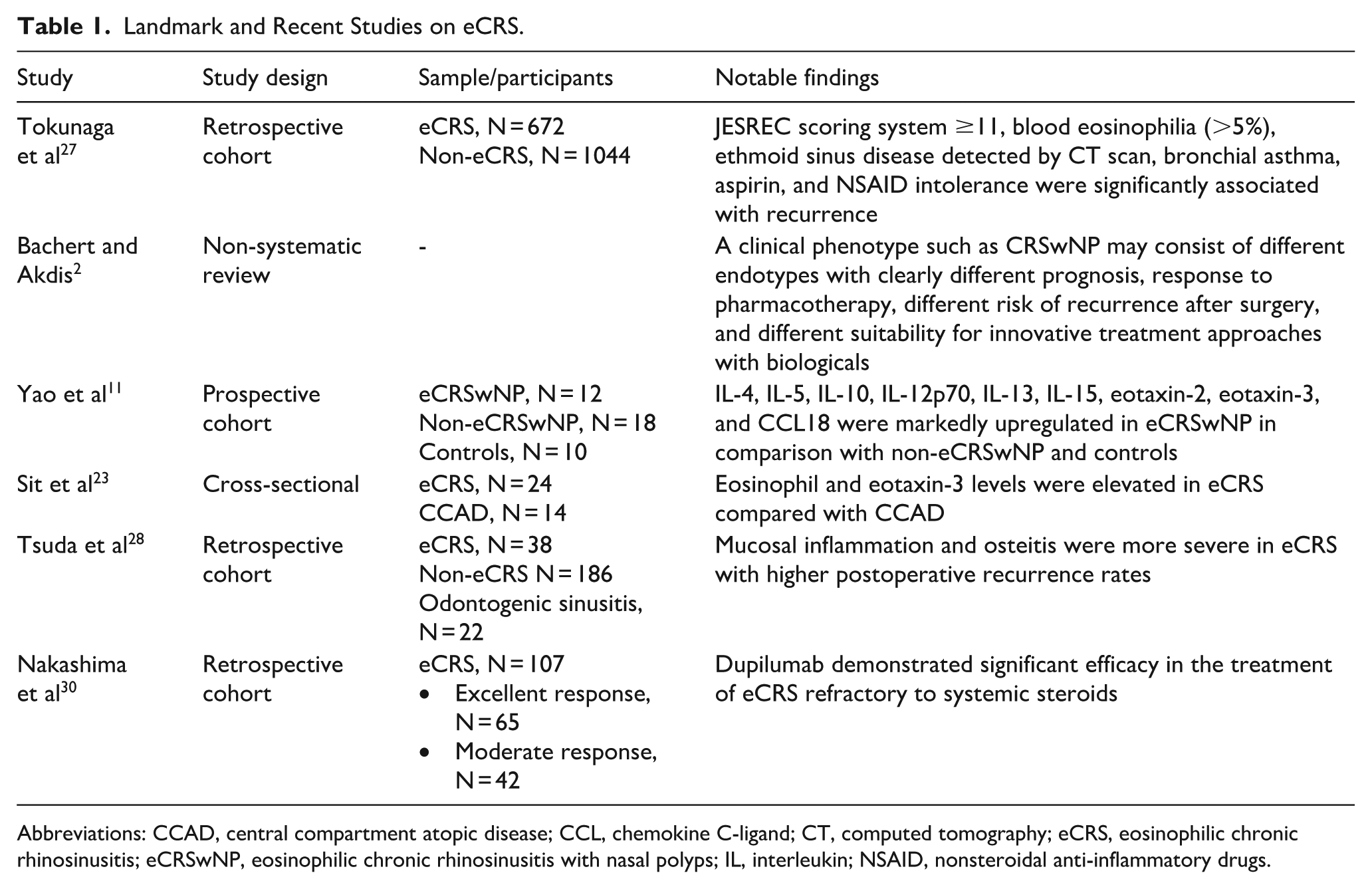
Abbreviations: CCAD, central compartment atopic disease; CCL, chemokine C-ligand; CT, computed tomography; eCRS, eosinophilic chronic rhinosinusitis; eCRSwNP, eosinophilic chronic rhinosinusitis with nasal polyps; IL, interleukin; NSAID, nonsteroidal anti-inflammatory drugs.
Aspirin-Exacerbated Respiratory Disease
AERD is a distinct endotype of eCRS, characterized by the clinical triad of nasal polyps, asthma, and hypersensitivity to medications that inhibit cyclooxygenase-1 enzymes, typically aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs). This condition is also referred to as NSAID-exacerbated respiratory disease in some contexts. The underlying pathophysiology of AERD is driven by dysregulated arachidonic acid metabolism, resulting in excessive cysteinyl leukotriene production and reduced prostaglandin E2 levels, a key anti-inflammatory mediator.9,31 This imbalance promotes persistent inflammation, increased nasal polyps, and bronchoconstriction following NSAID exposure. Furthermore, AERD is associated with elevated leukotriene C4 synthase expression in sinonasal tissues, which further amplifies the inflammatory response.26,31
Patients with AERD typically experience more aggressive disease progression, requiring multiple sinus surgeries due to rapid polyp recurrence and refractory symptoms.2,32 AERD usually manifests in early adulthood and is associated with severe asthma, often necessitating high-dose inhaled or systemic corticosteroids for symptom control.25,26 AERD is not driven by allergic sensitization, and skin-prick testing for aeroallergens is often negative. 32 Recent research has identified increased mast cell activation in AERD, suggesting a potential role for mast cell stabilizers in treatment.11,31
Management of AERD involves a multimodal approach and can include leukotriene-modifying agents such as montelukast and zileuton, which help mitigate leukotriene-mediated inflammation.9,31 Aspirin desensitization, a specialized therapeutic strategy, has been shown to reduce polyp recurrence and improve asthma control in selected patients; however, chronic use increased the risk of spontaneous hemorrhages.5,26 Recently, biologic therapies targeting IL-4, IL-5, and IgE have demonstrated significant clinical benefits in AERD, offering new therapeutic avenues for patients with severe, refractory disease.17,24 Notably, dupilumab has effectively reduced nasal polyp burden and improved asthma control in AERD patients.8,13,23,33 Emerging evidence suggests that combination therapy with biologics and leukotriene inhibitors may provide even greater symptom control in AERD (Table 2).32,34
Landmark and Recent Studies on AERD.
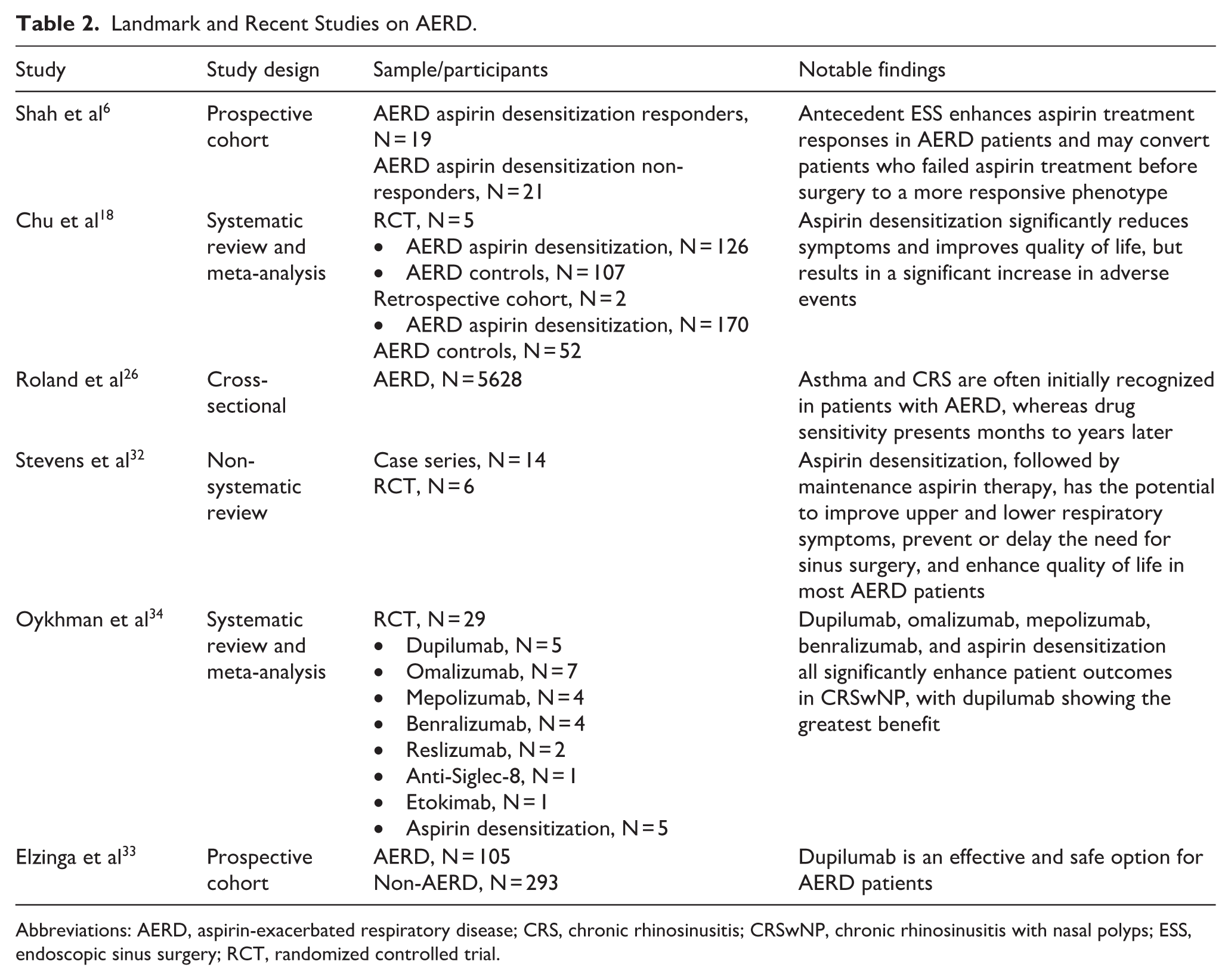
Abbreviations: AERD, aspirin-exacerbated respiratory disease; CRS, chronic rhinosinusitis; CRSwNP, chronic rhinosinusitis with nasal polyps; ESS, endoscopic sinus surgery; RCT, randomized controlled trial.
Central Compartment Allergic Disease
CCAD is an atopic manifestation of CRSwNP. Initially described as central thickening and swelling of inferior and middle turbinates mucosa, 35 CCAD is associated with local allergic reactions specific to allergens, with a 95% specificity for inhalant allergens.36 -38 The main immune mechanism in the development of CCAD is an IgE-dependent reaction to allergen exposure. 38 The typical clinical presentation involves a young adult with a history of allergic rhinitis, presenting with CRS symptoms and polypoid degeneration of the middle turbinates in the anterior and inferior portions.36,37 Although allergies are not a risk factor for CRS, 1 CCAD patients have a strong association with allergy, with studies showing allergy sensitization in 74% to 100% of CCAD patients, including both local and systemic sensitization.39,40 Asthma is only weakly associated with CCAD, affecting ~17% of patients. 41
In imaging studies, the hallmark of CCAD is a central thickening of the turbinates and septum with relatively normal peripheral sinus mucosa. This unique radiographic feature is sometimes referred to as the “black halo” sign. 42 In surgery, a targeted approach to opening the middle meatus obstruction with minimal sinus mucosal disease can often alleviate the patient’s symptoms, 43 with better postoperative control compared to non-CCAD patients.44,45 Monitoring for allergic exacerbation is essential, and long-term symptom improvement is usually maintained, correlating with a low-inflammatory profile subtype of CRS.43,46 Surgery and pharmacotherapy are effective in managing CCAD; however, the addition of allergen immunotherapy improved allergic responses and enabled de-escalation of topical treatment (Table 3). 47
Landmark and Recent Studies on CCAD.
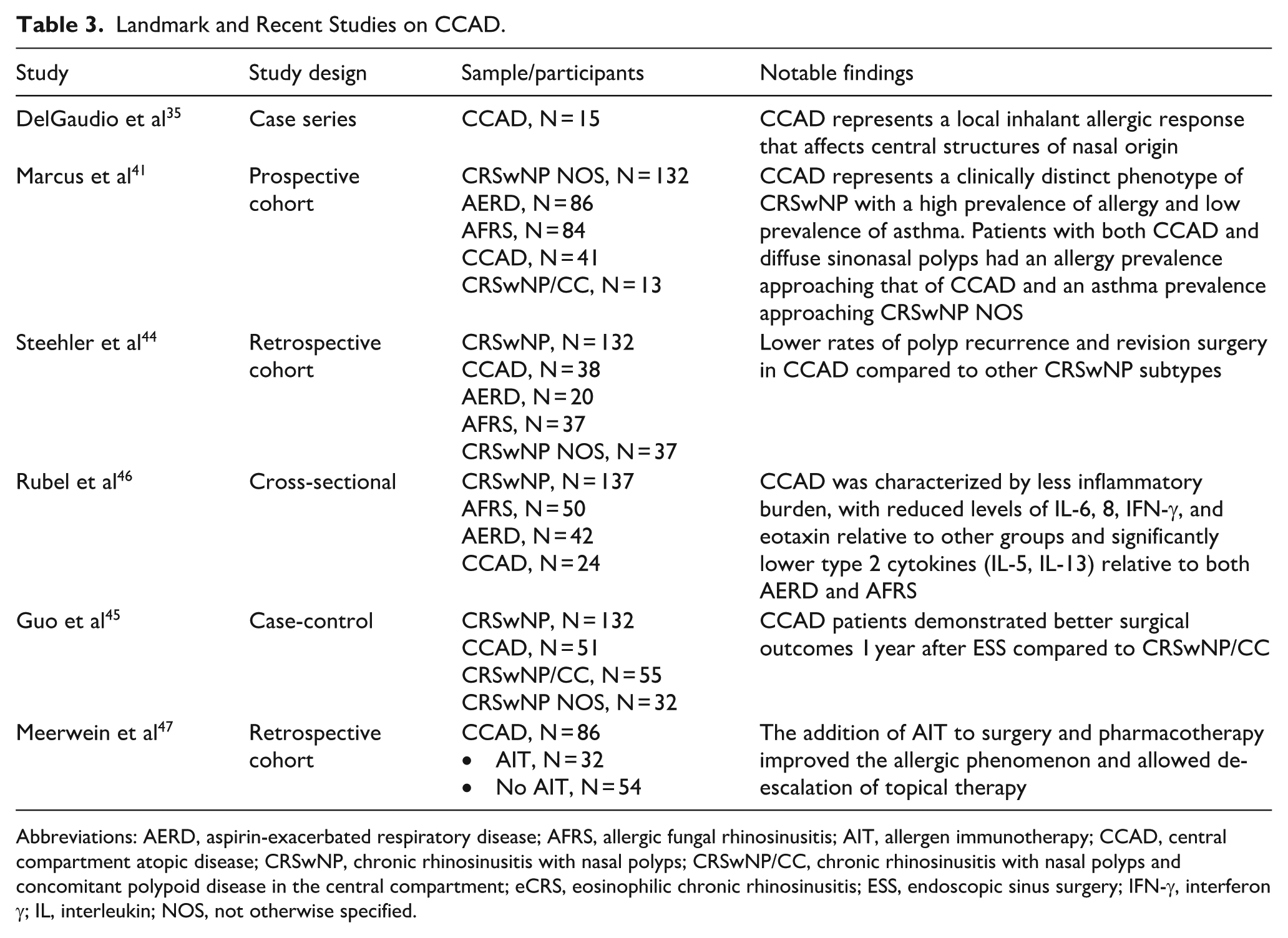
Abbreviations: AERD, aspirin-exacerbated respiratory disease; AFRS, allergic fungal rhinosinusitis; AIT, allergen immunotherapy; CCAD, central compartment atopic disease; CRSwNP, chronic rhinosinusitis with nasal polyps; CRSwNP/CC, chronic rhinosinusitis with nasal polyps and concomitant polypoid disease in the central compartment; eCRS, eosinophilic chronic rhinosinusitis; ESS, endoscopic sinus surgery; IFN-γ, interferon γ; IL, interleukin; NOS, not otherwise specified.
Allergic Fungal Rhinosinusitis
AFRS was first described by Millar et al 48 as an allergic aspergillosis of the maxillary sinuses with features similar to allergic bronchopulmonary aspergillosis. AFRS is a distinct endotype of CRSwNP characterized by type-I hypersensitivity to fungi, leading to the production of thick eosinophilic mucin throughout the nasal cavity and sinuses. 49 While several authors have attempted to update the histopathologic diagnosis criteria,50 -52 the Bent and Kuhn criteria from 1994 53 remain the most widely used. They described 5 major criteria that are mandatory for diagnosis: (1) evidence of type-I IgE-mediated hypersensitivity; (2) nasal polyps; (3) characteristic CT findings; (4) eosinophilic mucin; (5) positive fungi smear. In addition, they mentioned 6 minor criteria that are not mandatory for diagnosis but describe distinct features that support the diagnosis: (1) asthma; (2) unilateral predominance; (3) radiographic bone erosion; (4) positive fungal culture; (5) Charcot-Leyden crystals (products of degraded eosinophils); (6) serum eosinophilia.
Patients with AFRS present with significant nasal polyp burden, thick allergic mucin, and sinus wall expansion, often affecting the orbit and skull base, leading to facial asymmetry and visual changes.54,55 Males, African Americans, and patients from low socioeconomic backgrounds are more likely to develop severe disease, with early disease onset, more severe bony involvement, and higher rates of revision surgery.55 -58
The cornerstone of AFRS treatment combines surgical intervention with medical therapy. Extensive endoscopic sinus surgery (ESS) aims to remove the nasal polyps, clear the allergic mucin, and improve the drainage of the affected sinuses. Understanding the pathogenesis of AFRS as an immunologically mediated type-I hypersensitivity to fungal antigens has led to the development of adjuvant medical therapies that include sinonasal irrigations and topical and oral corticosteroids.50,51,59,60 Rupa et al 61 demonstrated a significant improvement of symptoms and prevention of early recurrence with a postoperative course of oral corticosteroids. Antifungal therapy is generally not recommended due to the noninvasive nature of the fungal involvement.
Recent studies have investigated the potential role of biologics in treating AFRS. Luong et al 62 proposed that the type-I hypersensitivity response in AFRS drives a type 2 immune response, characterized by elevated IL-4, IL-5, and IL-13 cytokines and high IgE levels, suggesting a possible benefit from biologic immunomodulators. 63 Mostafa et al 64 compared a single postoperative injection of Omalizumab to twice-daily nasal steroid spray, demonstrating significantly greater clinical and endoscopic responses with Omalizumab. Aljubran et al 65 conducted a systematic review and meta-analysis on the therapeutic outcomes of biologics in AFRS, reporting substantial improvements in clinical, laboratory, and radiological parameters. However, further large-scale studies with long-term follow-up are needed to fully assess the efficacy of biologics in AFRS. 66
Current research regarding AFRS pathophysiology could lead to innovative treatments, including antimicrobial peptides for the immune system activation, 67 and prostaglandin E receptor modulator (Table 4).68,69
Landmark and Recent Studies on AFRS.
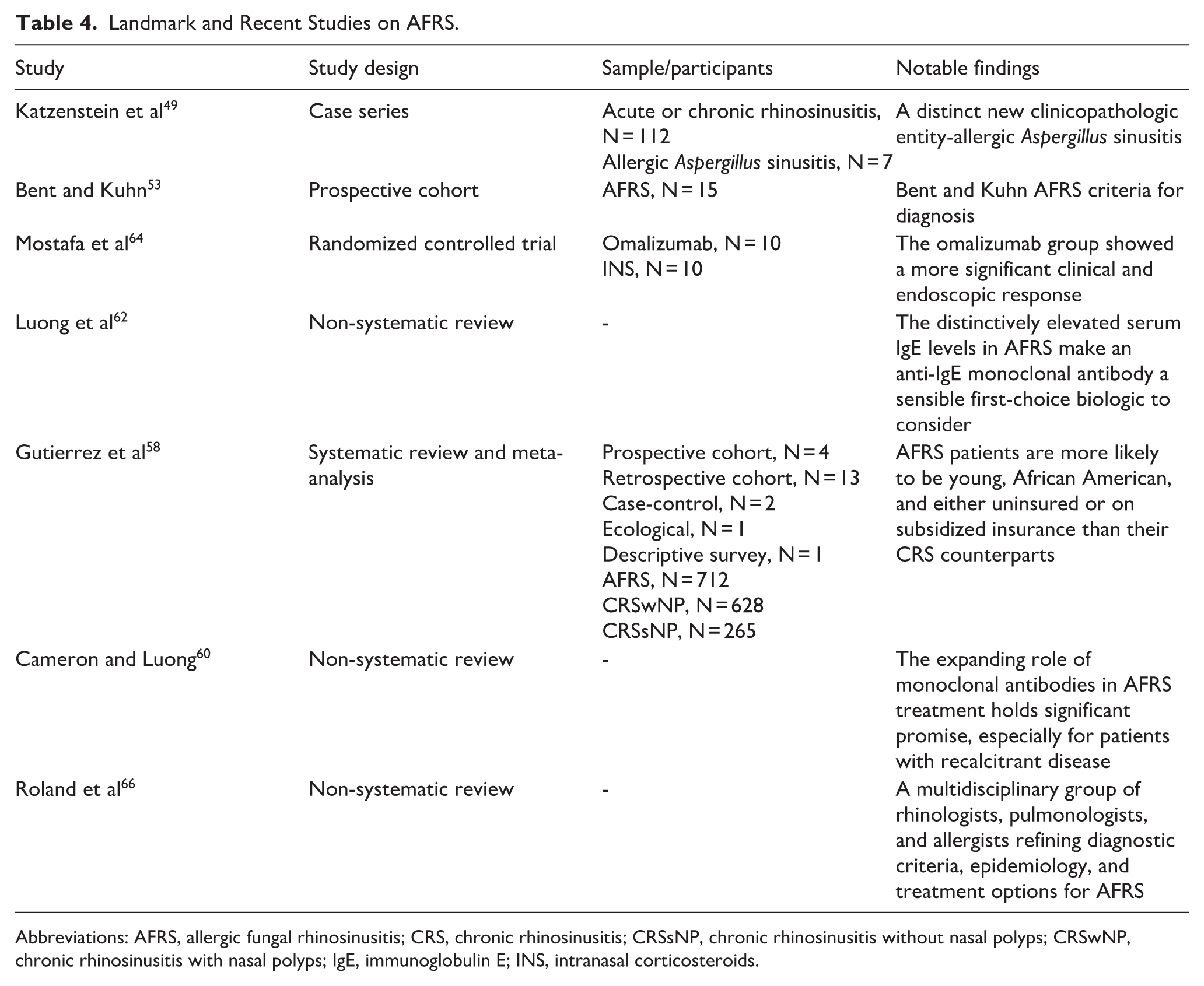
Abbreviations: AFRS, allergic fungal rhinosinusitis; CRS, chronic rhinosinusitis; CRSsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; IgE, immunoglobulin E; INS, intranasal corticosteroids.
Non-Eosinophilic CRS
Non-eCRS is characterized by an inflammatory response in which respiratory nasal epithelial cells, when stimulated, drive either a neutrophil-dominated (Th1) or a combined neutrophil- and macrophage-driven (Th17) inflammatory cascade.20,22 This endotype has 2 primary phenotypic presentations: the more common CRSsNP 1 and the less frequent non-eCRSwNP. 19
On nasal endoscopy, non-eCRS is often characterized by polypoid edema in the sinus outflow tracts or frank purulence. 19 Imaging frequently reveals near-total opacification of multiple sinuses or a “foamy” pattern, likely representing obstructing secretions. 43 Oral corticosteroids are not recommended for CRSsNP, as higher tissue neutrophilia is associated with reduced corticosteroid responsiveness. 70 Patients who do not respond to medical therapy often require ESS. 71 Beyond surgery, a subset of non-eCRS patients with low total IgE levels may benefit from long-term macrolide therapy, which exerts immunomodulatory effects by promoting neutrophil degradation and inhibiting neutrophil chemotaxis. 72
Despite increasing research into CRS endotypes, non-type 2 inflammatory diseases, including non-eCRS, remain understudied compared to their type 2 counterparts. As a result, current endotype-driven therapies have shown limited efficacy in non-eCRS, underscoring the need for further investigation into targeted treatment strategies. 70
Czerwaty et al 73 evaluated immune cell infiltration in non-eCRS sinonasal mucosa and identified positive correlations between CD19-positive cell expression and both the Lund-Kennedy Score and the nasal polyp score. A positive correlation was also observed between the number of tissue eosinophils and the expression of CD11b-positive and CD16-positive cells. Assessment of the expression of these cells may serve as a potential biomarker for predicting treatment response, particularly in patients receiving biologic treatments (Table 5).
Landmark and Recent Studies on Non-eCRS.
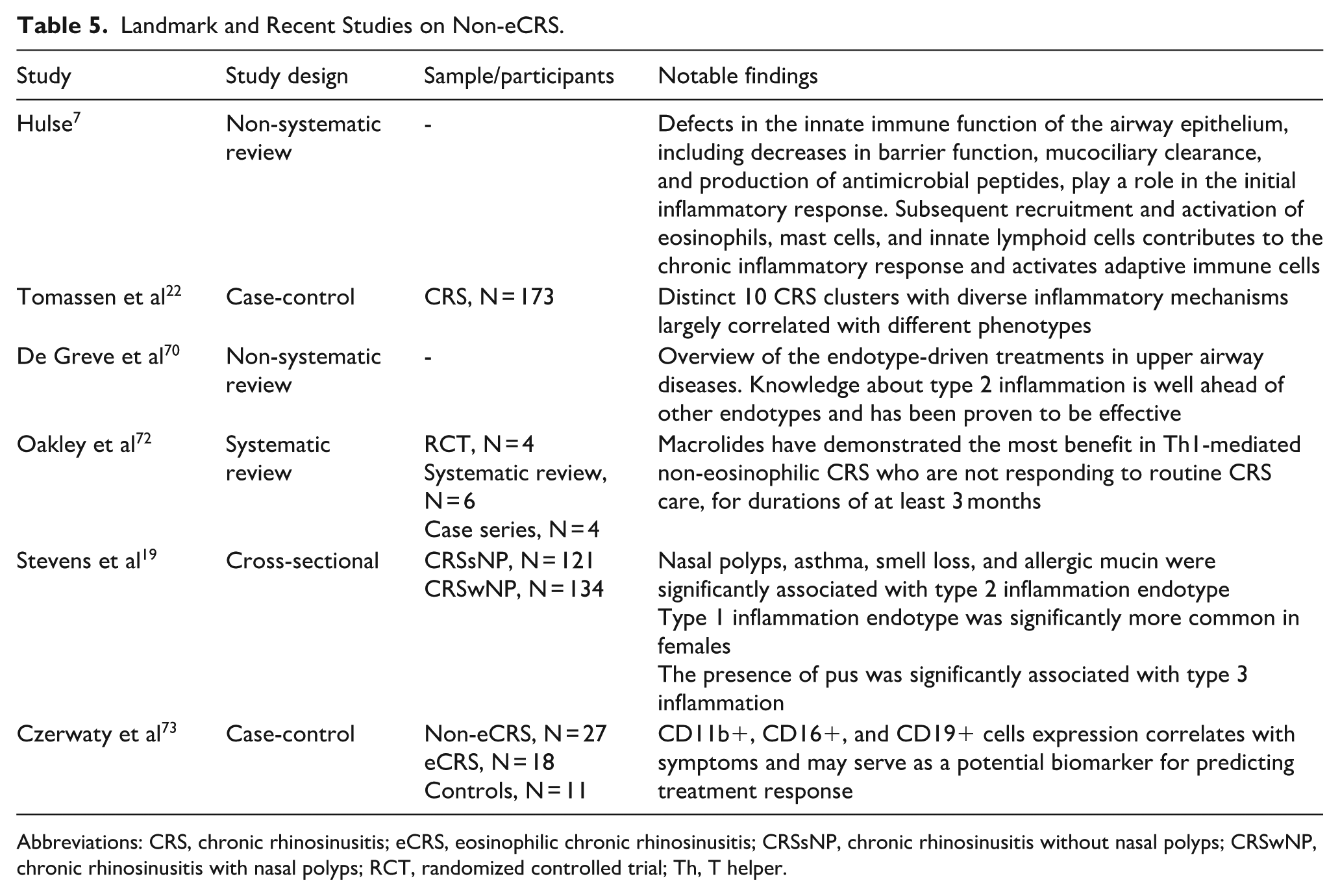
Abbreviations: CRS, chronic rhinosinusitis; eCRS, eosinophilic chronic rhinosinusitis; CRSsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; RCT, randomized controlled trial; Th, T helper.
Clinical Implications of Different CRS Endotypes
The evolving understanding of CRS endotypes has profound clinical implications for its diagnosis and treatment. While type 2 inflammation endotypes, such as eCRS, AERD, and AFRS, will respond better to intranasal and systemic steroids and have a greater need for revision surgeries16,74,75; non-type 2 endotypes, such as non-eCRS, are characterized by low levels of IgE and eosinophils and may respond better to the anti-inflammatory effects of low-dose macrolides.72,76
The introduction of biologic therapies represents a paradigm shift in CRS management, particularly for patients with recalcitrant disease or those at risk of surgical failure. Biologic treatments targeting IL-4, IL-5, and IL-13 have shown promising results in reducing nasal polyp burden, improving olfaction, and decreasing reliance on systemic corticosteroids, offering a much-needed alternative to traditional treatments.17,19 For patients with eCRS, biologic treatments such as dupilumab, 4 mepolizumab, 10 benralizumab, 29 and omalizumab 77 have been shown to significantly improve outcomes, even in the most severe cases.
In the future, identifying the molecular and immunologic profiles of different CRS subtypes will enable more personalized treatment plans by targeting specific disease pathways in each patient. Turner et al 78 described CRS endotyping using mucous specimens, providing a minimally invasive approach that can be performed in-office. Lopez et al 79 and Rebuli et al 80 outlined the use of Leukosorb strips to sample nasal epithelial lining fluid and characterize soluble inflammatory mediators. However, there is currently no commercially available point-of-care diagnostic tool for diagnosing specific endotypes.
De Corso et al 81 extracted calprotectin and IL-5 from patients’ mucus and nasal polyps, finding that elevated levels were significantly associated with uncontrolled disease. These biomarkers may aid in predicting more severe cases, in which earlier initiation of biologic therapy should be considered.
Pelletier et al 82 explored the impact of short-course adjuvant perioperative treatment with dupilumab compared to a placebo, finding it to be associated with improved long-term olfactory outcomes and a sustained reduction in serum IgE levels.
Sialic acid immunoglobulin-like lectins (Siglecs-8 and -9) are surface proteins found on immune cells such as eosinophils, neutrophils, mast cells, and basophils. Upon engagement with antibodies or ligands, eosinophils and neutrophils undergo apoptosis, while mast cells lose their ability to release mediators such as histamine and prostaglandins.
Recent studies have identified Siglec-8 and Siglec-9 ligands in sinonasal submucosal gland cells. This finding suggests that drugs targeting Siglec-8 or Siglec-9 binding could selectively eliminate activated eosinophils or neutrophils. Such targeted therapies may prove effective in treating the AERD endotype, characterized by elevated eosinophil activation, and are currently being evaluated in Phase II trials.83 -85
Ongoing research into the molecular mechanisms underlying CRS will likely further refine classification systems and therapeutic targets, leading to more precise and effective therapeutic interventions.
Conclusions
The evolving understanding of CRS endotypes has reshaped the approach to diagnosis and treatment, moving beyond a simple phenotypic classification to a molecular and immunologic framework. The distinction between type 2 and non-type 2 inflammation has led to the development of more targeted and precise therapeutic strategies, especially with the introduction of biologic agents that target key cytokines involved in conditions such as eCRS, AERD, and AFRS. While traditional treatments, such as corticosteroids and surgery, remain the mainstay of CRS management, the increasing availability of targeted therapies offers new hope for patients with refractory disease, reducing the need for repeated surgical interventions and improving long-term outcomes. However, significant gaps remain in understanding non-type 2 CRS, where treatment options are still limited and require further research. Future advancements in biomarker identification and personalized medicine will continue to refine CRS management, ensuring that each patient receives the most effective and tailored therapy for their specific disease endotype.
Footnotes
Ethical Considerations
An ethical approval was not required for this study, since it is a literature review and does not involve the collection of new data from human or animal subjects.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available in the figshare repository. doi.org/10.6084/m9.figshare.28826750.v1.
