Abstract
Giant recurrent auricular keloids, particularly those complicated by donor-site involvement, present significant therapeutic challenges due to aggressive fibroblast biology and high recurrence rates. We report a complex case of a 15-year-old female who developed a massive recurrent auricular keloid with associated abdominal donor-site keloid following otoplasty and subsequent skin grafting. Initial otoplasty at age 10 was complicated by keloid formation, requiring excision and abdominal full-thickness grafting. By age 15, she presented with a recurrent auricular keloid causing auricular deformity, tragus distortion, and external auditory canal obstruction, alongside a donor-site keloid. Management involved the radical excision of both keloids. To minimize tension-induced recurrence, the auricular wound underwent deliberate secondary intention healing, while the abdominal site was closed primarily under minimal tension. Adjuvant therapy comprised 6 weekly intralesional interferon-gamma (IFN-γ) injections (1 million units auricle; 0.5 million units abdomen). This multimodal approach leveraged IFN-γ’s antifibrotic properties, including ferroptosis induction in keloid fibroblasts. At 1-year follow-up, both sites exhibited sustained remission with good fibrosis and acceptable cosmesis without recurrence. This case demonstrates that tension-free excision with secondary intention healing, combined with intralesional IFN-γ, offers a promising strategy for managing complex pediatric keloids where conventional therapies carry substantial limitations. The protocol effectively addresses both biological recurrence drivers and psychosocial burdens inherent to disfiguring scars.
Introduction
Keloids represent a challenging fibroproliferative disorder characterized by abnormal wound healing beyond the boundaries of the original injury, with a predilection for anatomical sites such as the auricle following trauma or surgery. 1 It is more common in individuals with darker skin tones (African and Asian), more frequent between the ages 10 and 30. 2 Otoplasty, while effective for correcting prominent ears, carries inherent risks of keloid formation, particularly in genetically susceptible individuals. 3 The retroauricular region is especially vulnerable due to inherent skin tension and mechanical stress, factors exacerbated by external influences such as prolonged mask wear. 2 Despite advances in multimodal therapies—including surgical excision, intralesional steroids, radiation, and pressure devices—keloid recurrence rates remain unacceptably high, ranging from 45% to 100% after surgery alone.1,4 Auricular keloids further complicate reconstruction due to the ear’s complex topography, frequent exposure to friction, and limited tissue reserves for primary closure.4,5 Giant keloids (>5 cm) present additional therapeutic hurdles, often requiring extensive resection and grafting, which paradoxically risk new keloid formation at donor sites. 4 The occurrence of donor-site keloids, though rare, underscores the aggressive biological behavior of keloid fibroblasts and the limitations of conventional interventions. 5
Interferon-gamma (IFN-γ) has emerged as a promising adjuvant therapy due to its antifibrotic properties, including suppression of collagen synthesis and induction of fibroblast apoptosis. 6 However, its application in giant, recurrent auricular keloids—particularly those involving donor sites—remains inadequately documented. This case highlights the formidable clinical challenge posed by such lesions and explores the potential of IFN-γ within a multimodal framework to mitigate recurrence.
Case Presentation
In this case, a 15-year-old girl presented with a huge keloid on the right ear and a keloid on the abdomen, both resulting from previous surgical procedures. She initially underwent otoplasty at age 10 to correct prominent ears, which was successful. However, 2 years later, she developed a medium-sized keloid on the posterior aspect of the right ear. This keloid was surgically excised, and an abdominal full-thickness skin graft was used to cover the resulting defect. By age 15, a huge keloid had formed on the auricle, causing deformity of the ear and tragus, as well as obstructing the external auditory canal (Figure 1). In addition, a keloid was present at the abdominal donor site (Figure 2). In response, the huge auricular keloid was completely excised along with the keloid tissue at the abdominal site. Postoperative management included intralesional injections of interferon gamma 1b—1 million units into the auricle and 0.5 million units into the abdomen—administered weekly at multiple sites within each wound bed for 6 weeks. The abdominal donor site healed by primary intention, whereas the auricular wound was approximated without closure and left open to heal by secondary intention. Over the course of a year, good fibrosis and acceptable cosmetic results were achieved (Figure 1).
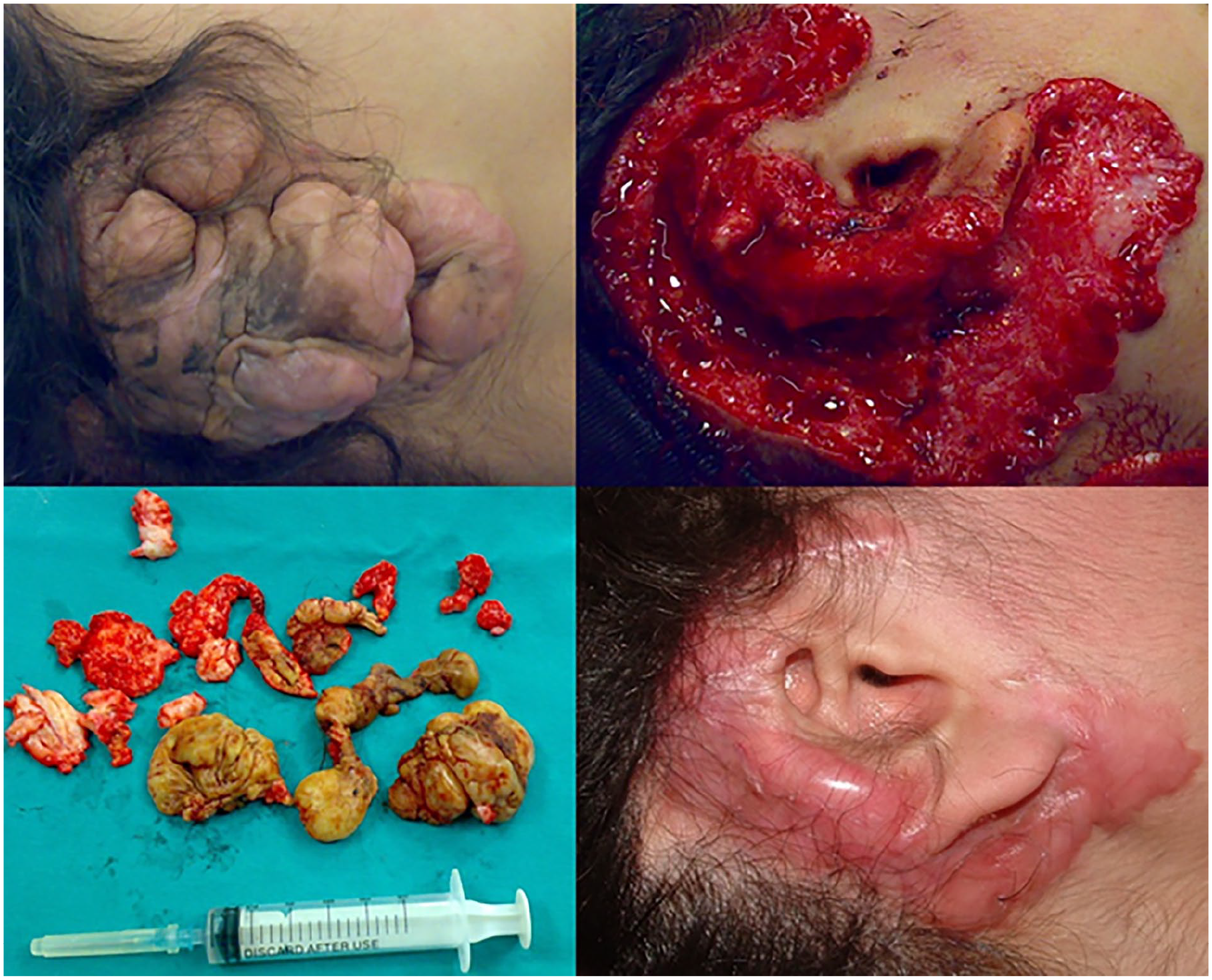
A huge keloid was excised, with good fibrosis and acceptable cosmetic outcomes.
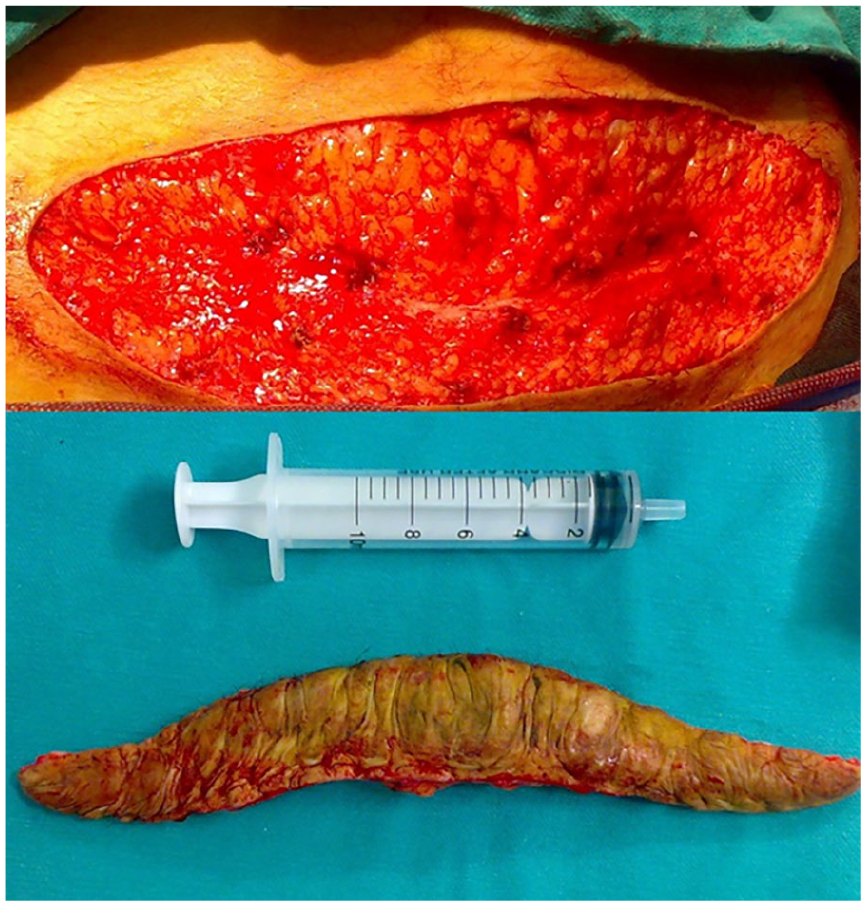
The abdominal donor site keloid.
Discussion
This case illustrates the formidable challenge of managing giant, recurrent auricular keloids, particularly those arising from prior surgical interventions. The development of a massive keloid following otoplasty, further complicated by a donor-site keloid, underscores the aggressive biological behavior of keloid fibroblasts and the limitations of conventional monotherapies.1,3 Our multimodal approach—combining radical excision, secondary intention healing, and adjuvant intralesional IFN-γ—achieved sustained remission without recurrence at 1-year follow-up. This outcome warrants critical analysis of current therapeutic paradigms for complex auricular keloids.
Surgical excision remains the cornerstone for massive keloids where volume precludes noninvasive management.1,4 However, recurrence rates approach 45% to 100% with excision alone, necessitating adjuvant therapies.1,5 In this case, primary closure after initial excision likely increased tension, stimulating fibroblast hyperactivity and recurrence.1,4 For the revision surgery, we avoided primary closure and allowed healing by secondary intention. This reduced mechanical stress on wound margins, a key trigger for collagen overproduction.1,5 While secondary healing risks contour irregularities, the auricle’s convex surfaces often remodel favorably with controlled fibrosis. 4 Donor-site keloids, as seen here abdominally, are rare but highlight keloid fibroblasts’ metastatic potential.4,5 The Excision, Skin grafting, Corticosteroids, Adjuvant radiotherapy, Pressure therapy protocol advocates split-thickness grafts for large defects, but graft failure rates reach 20%, and donor sites themselves may develop keloids. 3 We excised the abdominal keloid but prioritized primary closure under minimal tension, avoiding further grafting. Intralesional corticosteroids (eg, triamcinolone) are first-line adjuvants, inhibiting collagen synthesis via transforming growth factor beta (TGF-β) suppression.1,5 However, their efficacy plateaus in giant keloids, and side effects—skin atrophy, hypopigmentation, pain—limit compliance.1,5 Studies report an 8.1% recurrence rate with postoperative steroids, but outcomes vary widely (3%-60%). 1 IFN-γ offers a mechanistic alternative; it downregulates collagen I/III synthesis, induces fibroblast apoptosis, and, crucially, promotes ferroptosis—a lipid peroxidation-dependent cell death pathway. 6 It has been demonstrated that IFN-γ triggers ferroptosis in keloid fibroblasts by inhibiting the serpine2-SLC7A11/GPX4 axis, depleting glutathione, and elevating reactive oxygen species. 6 A regimen of 1 million units weekly leveraged this antifibrotic activity without corticosteroid side effects. IFN-γ’s recurrence rate (18.7%) rivals corticosteroids but requires further validation in pediatric populations.1,6 Radiotherapy within 24 to 48 hours post-excision reduces recurrence to 2.8% to 15.4% by inhibiting fibroblast proliferation.1,5 However, carcinogenic risks—particularly in pediatric patients—precluded its use here.1,5 Pressure therapy (eg, magnets, silicone earrings) mitigates recurrence by inducing hypoxia and reducing collagen synthesis.1,5 Compliance is challenging long term, but custom devices exerting 1.33 to 3.3 kPa pressure can lower recurrence to 18%. 5 We deferred pressure due to the auricle’s complex geometry, but consider it valuable for earlobe keloids. For pediatric cases, IFN-γ’s safety profile is advantageous over steroids or radiation. However, its cost and need for weekly injections require consideration. Emerging agents show promise in keloid treatment. 5-Fluorouracil inhibits fibroblast proliferation and has demonstrated over 50% flattening in 50% of keloids, although auricle-specific data remain limited; Verapamil enhances collagenase activity; when combined with pressure therapy, it has achieved 55% remission rates in earlobe keloids; The topical immunomodulator imiquimod, which stimulates IFN-γ, has shown efficacy in preventing recurrence, with success reported in 75% of earlobe keloids following excision. 1
Beyond optimizing recurrence control, our adjuvant IFN-γ protocol may synergistically enhance patient-centered outcomes by addressing both fibrotic biology and psychosocial burden. Combining surgical tension reduction with targeted cytokine modulation not only suppresses collagen overproduction but also appears to normalize pathological fibroblast phenotypes more comprehensively than monotherapies, potentially yielding more durable aesthetic results. 7 Critically, longitudinal quality-of-life assessments in auricular keloid patients demonstrate that sustained symptom resolution and scar stability—rather than the absence of recurrence alone—correlate most strongly with psychosocial recovery, particularly regarding self-esteem and social reintegration. 8 This underscores the importance of multimodal regimens that simultaneously target biological recurrence drivers and the functional/emotional sequelae of disfiguring scars, especially in pediatric populations where treatment adherence hinges on tolerability and rapid restoration of normalcy. 9
Giant auricular keloids with donor-site involvement represent an extreme fibroproliferative phenotype. Successful management requires tension-free excision, prioritizing secondary healing over grafting; adjuvant IFN-γ to exploit ferroptosis pathways; and long-term surveillance. Future studies should explore topical IFN-γ inducers (eg, imiquimod) or nanoparticle-delivered siRNA against serpine2 to enhance efficacy while minimizing invasiveness. 6
Conclusion
This case underscores the formidable challenge of giant, recurrent auricular keloids, particularly those complicated by donor-site involvement. By employing radical surgical excision with deliberate secondary intention healing—avoiding tension-inducing closure—combined with adjuvant intralesional IFN-γ, sustained remission was achieved without recurrence at 1 year. IFN-γ’s antifibrotic mechanism, notably ferroptosis induction, offers a targeted biologic adjunct that may circumvent the limitations of corticosteroids or radiotherapy, especially in pediatric populations. While long-term surveillance remains essential, this multimodal strategy demonstrates promise for managing complex keloids where conventional therapies fall short, prioritizing both functional and psychosocial recovery.
Footnotes
Acknowledgements
No acknowledgments were given by any of the authors.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases.
Consent to Participate
Written informed consent was obtained from the patient’s family for the publication of this article.
Author Contributions
All authors contributed to the conception of the study. All authors read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the author or the corresponding author upon reasonable request.
