Abstract
Objective:
Adenotonsillectomy is widely recognized as first-line treatment for obstructive sleep disordered breathing (oSDB), especially for older children with severe disease. The best course of treatment for children with milder oSDB is less clear. It has been our practice to offer surgical candidates with mild symptoms (snoring with occasional short, infrequent pauses by parental report) the option of medical treatment with an antibiotic and oral steroid. We examine the immediate and long term-effects of this intervention.
Methods:
We reviewed the records of children presenting to our academic Pediatric Otolaryngology practice in the years 2000 to 2023. Tonsil size before and after treatment, resolution of snoring, and ultimate need for surgery were recorded. Follow-up phone calls were made to patient families when information was incomplete.
Results:
One hundred sixty-nine patients underwent medical treatment for mild oSDB. Among children with acute (≤2 months) symptoms, 38% (20/53) had resolution of snoring and 42% (18/43) did not require surgery. Mean tonsil size (Brodsky grade) decreased from 2.58 to 2.20 after medical treatment in this group. Among children with chronic symptoms, 34% (26/77) had resolution of snoring and 30.0% (18/60) did not require surgery. Their mean tonsil size decreased from 2.58 to 2.13.
Conclusions:
These results suggest that treatment of mild oSDB treatment with short-course amoxicillin-clavulanate and 5 days of prednisolone is safe and can result in resolution of snoring in many children. More than one third of children treated this way avoided surgery with follow-up averaging 5 years. These observations argue for a prospective, randomized trial of this medical treatment as an alternative to immediate surgery.
Level of Evidence:
3 – retrospective review.
Keywords
Introduction
Surgery, usually including tonsillectomy and adenoidectomy, is the standard therapy for obstructive sleep apnea (OSA) and obstructive sleep disordered breathing (oSDB) in children with adenotonsillar hypertrophy. Surgery results in rapid resolution of symptoms for the majority of children with improvement in quality of life compared with observation and symptomatic treatment.1,2 oSDB is common in the pediatric population, with an estimated 1.2% to 5.7% of children affected. 3
There is, however, a group of patients with mild sleep disordered breathing and modest adenotonsillar hypertrophy for whom appropriate therapy is less clear.4,5 In the senior author’s fellowship (G.I.), children with enlarged tonsils and snoring were sometimes treated with a course of oral antibiotics or steroids as a temporizing measure. Using these medications seemed to work in a portion of children, and we continued to offer these medications over 3 decades of clinical practice. The purpose of this report was to review that clinical practice to learn whether the combination of a beta-lactamase stable oral antibiotic and an oral glucocorticoid resulted in the improvement of symptoms and whether this effect was lasting.
Materials and Methods
Clinical Protocol: Children who presented with mild symptoms of oSDB were routinely offered medical treatment as an alternative to immediate surgery. We defined mild oSDB symptoms as snoring with occasional short (<10 seconds), infrequent pauses in breathing according to parental history and/or sleep videotape. Children with polysomnogram-proven OSA or severe symptoms (eg, cyanosis, failure to thrive, cor pulmonale) were not included. Adenoid size was not routinely evaluated in children with mild symptoms of oSDB and large tonsils.
Treatment consisted of a 5 day course of oral prednisolone (1 mg/kg/day in a single daily dose—50 mg maximum dose) and 10 days of oral amoxicillin-clavulanate (45 mg/kg/day (amoxicillin dose) in 2 divided doses, 1750 mg/day maximum). Children allergic to amoxicillin were treated with a second-generation cephalosporin. Patients were reexamined 4 weeks following treatment to monitor response to therapy.
Retrospective Study: This minimal risk retrospective study was approved by the Institutional Review Board of Temple University’s Human Research Protection Program (protocol 31712). A computerized collection of patient office notes and operative reports was queried using the Microsoft® Word “Find” feature to identify children 0 to 18 years of age who were treated for adenotonsillar hypertrophy between 2000 and 2023.
Collected data included age at the initial visit, whether surgical intervention was performed, tonsil size before and after treatment, and resolution of snoring (defined as resolution within 3 months). Tonsil size was graded using the Brodsky system from 0 to 4+ and averaged if between 2 numbers (ie, 2.5 for 2-3+). 6 Families of children who did not undergo surgery by the time of the last office note were contacted by telephone to determine whether surgery was performed elsewhere and whether oSDB symptoms persisted.
Data Analysis: Included patients were divided into 2 groups—those with recent onset of symptomatic adenotonsillar hypertrophy (acute – ≤2 months prior to presentation) and those with a duration of symptoms >2 months (designated as chronic). Within each group, the number of patients requiring surgery, the number with complete resolution of snoring, and the mean tonsil size before and after treatment were analyzed. The percentage of patients who avoided surgery long-term and had resolution of snoring was calculated for each group. Fisher’s exact test was used to compare contingency tables with P < .05 indicating statistical significance.
Results
Between January 1, 2000, and June 30, 2023, 169 children who underwent medical treatment for adenotonsillar hypertrophy were identified. Ninety-four/169 (56%) of patients were male. The mean age at the initial presentation was 3 years (range 0.5-14 years). Sixty-six/169 (39%) patients were symptomatic for <2 months and were categorized as acute adenotonsillar hypertrophy. Ninety-five/169 (56%) of patients were symptomatic for more than 2 months and were identified as chronic adenotonsillar hypertrophy. Eight patients had unknown duration of symptoms. Of 169 total patients, 126 patients were identified as needing a telephone interview as they had had neither surgery nor long-term outcomes documented in the office notes. Of 126 patients needing additional information, 63/126 (50%) patients answered a follow-up phone call, with a mean follow-up time since first office visit of 5 years (range 1-16 years).
Need for Surgery/Resolution of Snoring: Of 169 total patients, surgical outcome was known for 105 (62%) children (42 documented in office notes; 63 determined by telephone communication). Out of these 105 patients, 38 (36%) never needed surgery. Of 169 total patients, resolution of snoring was documented for 132 (78%), of whom 48 (36%) had complete resolution of snoring. Of these 48 patients for whom resolution of snoring was known, 8 (17%) had known surgical intervention. Of the 38 patients who never needed surgery, 25 (66%) had complete resolution of snoring.
Of the 66 children medically treated for acute adenotonsillar hypertrophy, the need for surgery was determined for 43 patients, either by office note or phone call. Of these 43 patients, 18 (41.9%) did not undergo surgery. Resolution of snoring was determined for 53 of 66 medically-treated patients, of whom 20 (37.7%) had complete resolution of snoring. Of the 20 patients with complete resolution of snoring, 3 (15%) had known surgical intervention. Of the 18 patients who never needed surgery, 11 (61%) had complete resolution of snoring (Table 1).
Characteristics of Acute and Chronic ATH Patients.
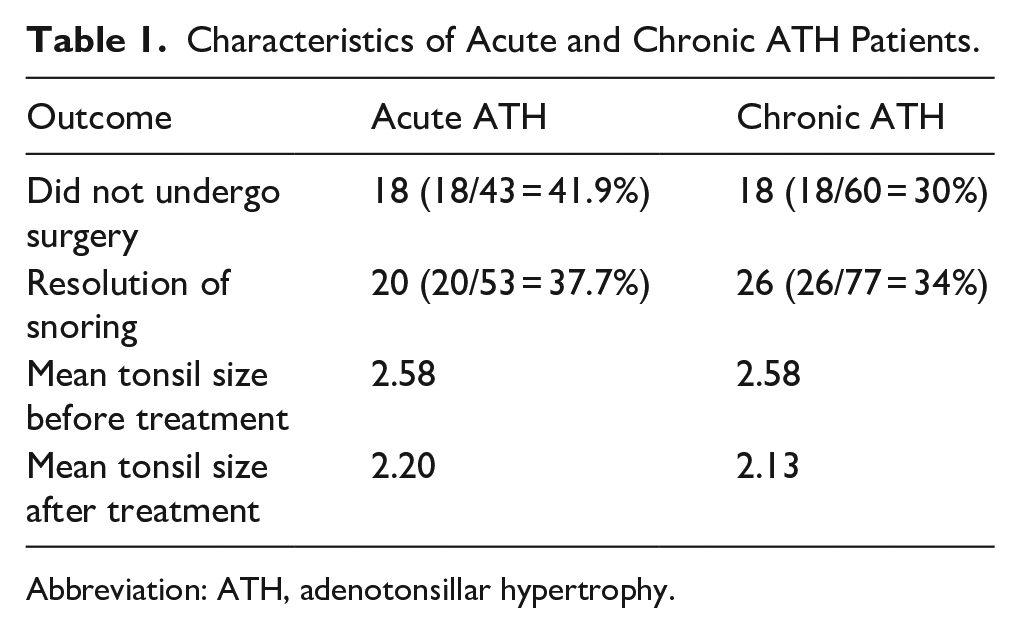
Abbreviation: ATH, adenotonsillar hypertrophy.
Of the 95 children medically treated for chronic adenotonsillar hypertrophy, the need for surgery was determined for 60 (63%) patients, either by office note or by telephone call. Of these 60 patients, 18 (30%) did not undergo surgery. Resolution of snoring was determined for 77 (81%) of 95 medically-treated patients, of whom 26 (34%) had complete resolution of snoring. Of the 26 patients with complete resolution of snoring, 5 (19.2%) had known surgical intervention. Of the 18 patients who did not undergo surgery, 12 (67%) had complete resolution of snoring (Table 1).
There was no statistically-significant difference between acute and chronic tonsillar hypertrophy in terms of need for surgery or resolution of snoring (acute vs chronic—surgery vs no surgery, P = 2948, effect size = 0.123; acute vs chronic—snoring vs resolved, P = .7102, effect size = 0.041; Table 2).
Contingency Table Results for Acute and Chronic ATH.
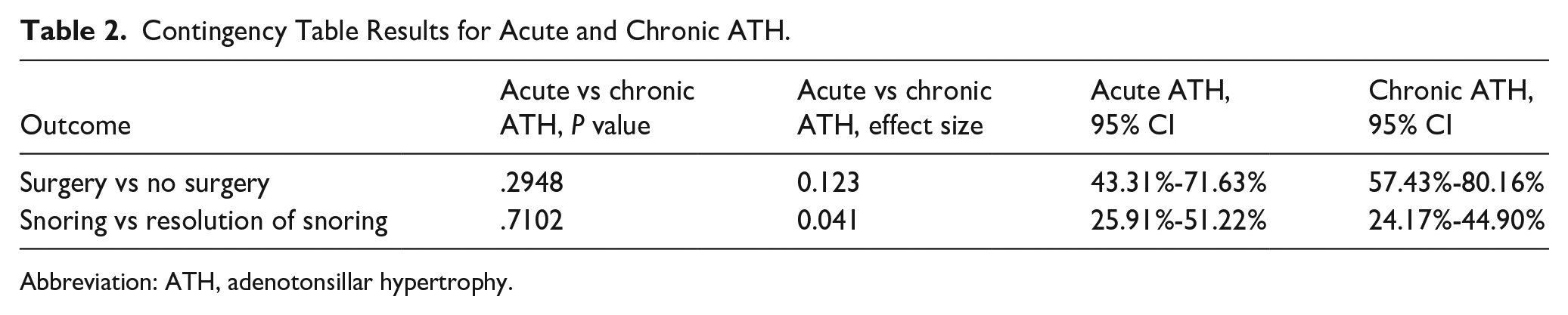
Abbreviation: ATH, adenotonsillar hypertrophy.
Change in Tonsil Size: Tonsil size before medical treatment was documented for 65/66 (98%) of children with acute tonsillar hypertrophy (mean Brodsky scale = 2.58). Tonsil size after medical treatment was known for 40/66 (61%) of children (mean = 2.20). For chronic adenotonsillar hypertrophy patients, tonsil size before treatment was known for 94/95 (99%, mean = 2.58). Posttreatment tonsil size was documented for 64/95 (67%, mean = 2.13). The mean change in tonsil size for acute tonsillar hypertrophy patients (for whom both pre- and posttreatment tonsil size was known, n = 42) was 0.52. The mean change in tonsil size for chronic tonsillar hypertrophy patients (for whom both pre- and posttreatment tonsil size was known, n = 64) was 0.45 (Table 1).
None of the medical treatments was complicated by protracted diarrhea or documented Clostridium difficile infection. Four children did not complete a full 10 day course of antibiotic therapy, complaining of bad taste. Of 169 children treated with medication, 24/63 (38%) contacted by telephone had left our care and underwent surgery by another surgeon.
Discussion
Surgery, usually tonsillectomy and adenoidectomy, provides expedient control of symptoms of oSDB and polysomnogram-proven OSA in the majority of children. In healthy children aged 5 to 9 years diagnosed with mild to moderate OSA by polysomnography, adenotonsillectomy provides benefits in terms of quality of life, symptoms, and behavior as rated by caregivers and polysomnogram scores.7,8
The benefits of surgery for children of younger age, 4 those with milder symptoms, or symptoms of short duration are less clear.9,10 Observation with supportive care (watchful waiting) has been advocated as an alternative to immediate surgery for children with mild OSA.
Several authors have studied the effectiveness of medical treatments to accelerate resolution of oSDB symptoms during watchful waiting. Principal among these are inhibitors of inflammation (glucocorticoids, leukotriene inhibitors) and antibiotics, following the logic that adenotonsillar hypertrophy results from the inflammatory response to bacterial colonization or chronic infection. In 1997, the group from Montreal Children’s Hospital performed a prospective trial of a 5 day course of oral prednisone (1 mg/kg daily) in 9 children with moderate-severe OSA by polysomnography. Tonsil size decreased after treatment in 2/9 children and adenoid size by radiograph decreased in 7/9 children. Mean apnea-hypopnea index decreased from 14 ± 5 to 10 ± 2 after treatment. The authors considered this a failure, despite some dramatic individual improvements, as only 1/9 avoided surgery. 11
In 2008, pulmonologists at the University of Louisville performed a prospective randomized crossover clinical trial of 6 weeks of intranasal budesonide for children with mild OSA (mean apnea/hypoxia index (AHI) = 3) by polysomnography. They found modest but statistically-significant improvement in AHI and sleep architecture in treated children compared to controls. Further, the effects seemed to persist for months after discontinuation of treatment. 12 These results were buttressed by a recent meta-analysis of 17 clinical trials. 13 It confirmed that anti-inflammatory drug therapy significantly decreased AHI with the combination of mometasone furoate nasal spray and oral montelukast sodium showing the most consistent effect.
A few researchers have explored the effects of long-course antibiotics on adenotonsillar hypertrophy. Sclafani et al 14 performed a prospective, randomized, double-blinded, placebo-controlled trial of a 30 day course of amoxicillin-clavulanate (40 mg/kg/day in divided doses). They found antibiotic treatment significantly reduced the need for surgery in children with obstructive adenotonsillar hypertrophy at the 1st month and that the effect persisted at the 3rd and 24th month posttreatment.
Don et al 15 at Los Angeles Children’s Hospital performed a small double-blinded, prospective clinical trial of 30 days of azithromycin (12 mg/kg on days 1-5; the regimen was repeated on days 11-15 and days 21-25) versus placebo. They found a modest decrease in respiratory distress index, but no change in tonsil size in the treated group compared with that in control.
Our protocol, which included both a beta-lactamase stable oral antibiotic and a short-course of an oral steroid, appears to yield superior results compared with these single-agent studies. With the long-term follow-up of more than half of treated patients, one third avoided surgery and one third had complete resolution of snoring.
While our study is strong in terms of patient numbers, consistent treatment protocol, and unusually-long-term follow-up, its retrospective nature and lack of an untreated control group limits interpretation of its results. Still, the prompt symptomatic improvement in a third of children with adenotonsillar hypertrophy of several months duration in our study suggests an acute effect of antibiotic/steroid treatment compared with observation.
We anticipated and encountered few adverse effects from treatment with a 10 day course of oral antibiotics. While we did not specifically monitor the incidence of diarrhea, based on a published study 16 we would expect clinically-significant diarrhea in <10% of children with newer formulations of amoxicillin-clavulanate. There were no documented episodes of C. difficile diarrhea in 169 treated patients. Similarly, a 5 day course of prednisolone at normal dosages is low risk for adrenal suppression, so steroid tapering was not part of our protocol.17,18
Conclusion
These results suggest that treatment of mild symptomatic sleep disordered breathing with a 10 day course of amoxicillin-clavulanate and 5 days of prednisolone is safe, resulted in a decrease or resolution of snoring in many children, and avoided the need for surgery over the long-term for a third of children. A prospective, randomized trial of this medical treatment is justified.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data collected and analyzed during this study are available from the corresponding author upon reasonable request.
