Abstract
Cutaneous mucormycosis, a rare and life-threatening fungal infection caused by Mucorales species, predominantly affects immunocompromised individuals. This report describes a rapidly progressive case in a 6-month-old immunocompetent infant who presented with necrotic facial lesions progressing to extensive eschar over 2 weeks. Despite prompt histopathological confirmation, radical surgical debridement, and immediate liposomal amphotericin B therapy, the infant succumbed to sudden deterioration 3 months after apparent clinical resolution. Initial biopsies confirmed cutaneous mucormycosis with characteristic broad, pauci-septate hyphae, while repeat biopsies at 1 month showed no residual disease. Laboratory and imaging findings were unremarkable, and fungal cultures remained negative. This case highlights the diagnostic and therapeutic challenges of pediatric mucormycosis, particularly in immunocompetent hosts, and underscores the potential for fatal relapse despite adherence to treatment protocols. The paradoxical outcome emphasizes the need for prolonged antifungal therapy, advanced molecular monitoring, and equitable access to diagnostics in resource-limited settings.
Introduction
Mucormycosis is a saprophytic opportunistic fungal infection caused by members of the order of Mucorales, in the class. Zygomycetes, ubiquitous saprophytic fungi that exist widely in nature. It is mainly caused by species of the genera Mucor, Rhizopus, Rhizomucor, and Absidia, although it can also be associated with Cunninghamella, Saksenaea, and Apophysomyces.1,2 The Rhizopus species (44%) and Mucor species (15%) are most commonly identified. 3 This fungal infection is predominately seen in the immunocompromised host. The most frequent form is the rhino-orbito-cerebral mucormycosis, followed by cutaneous mucormycosis, pulmonary mucormycosis, disseminated mucormycosis, gastrointestinal, and other rare forms.4-6 The cutaneous lesions caused by mucormycosis may be caused by an infection at the primary site or secondary to dissemination from another site. Cutaneous mucormycosis has the best outcome with lower mortality. 7 In our report, we present a rare case of primary facial cutaneous mucormycosis in a 6-month-old infant who has not survived.
Case Presentation
A 6-month-old, previously healthy male infant with no significant medical history presented to our emergency department with a 2-week history of progressive blackening involving the right half of his face. The lesion initially manifested as a small, painless boil that developed without preceding major trauma. Despite empirical treatment with topical antibiotics and wound care, the lesion failed to improve and instead rapidly progressed over 2 weeks to extensive necrotic eschar with surrounding erythema, edema, and sharply demarcated borders (Figure 1). The infant appeared otherwise well, with no fever or systemic symptoms.
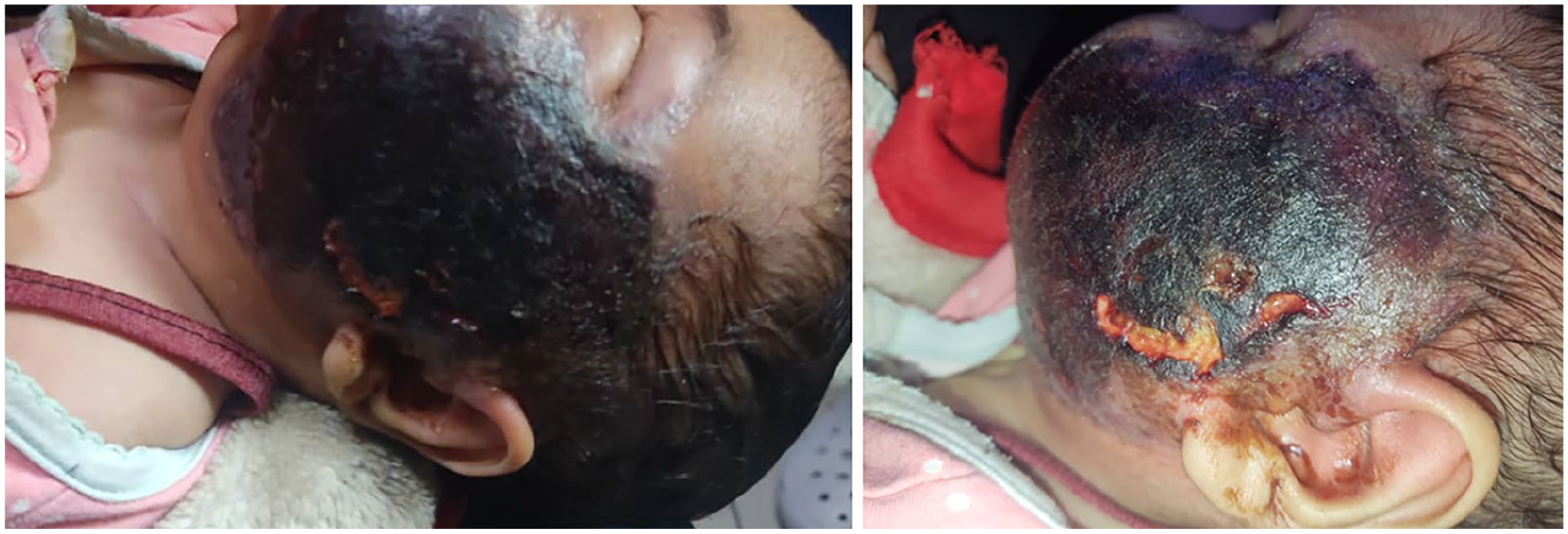
Clinical photographs showing left facial necrosis with well-demarcated eschar and surrounding inflammation.
Laboratory investigations, including complete blood count, inflammatory markers (C-reactive protein, Estimated sedimentation rate [CRP, ESR]), and metabolic panels, were unremarkable. On physical examination, there was no crepitus or pus discharge from the lesion.
The blood culture did not grow any fungus. A contrast-enhanced Computed tomography (CT) scan of the face and head revealed soft tissue necrosis limited to the subcutaneous layers, with no evidence of bony erosion or intracranial extension.
An urgent incisional biopsy of the necrotic tissue was performed, demonstrating broad, pauci-septate hyphae with right-angle branching on hematoxylin-eosin staining, confirming a diagnosis of cutaneous mucormycosis. Fungal cultures remained negative.
Within 24 hours of histopathological confirmation, all necrotic skin and subcutaneous tissues were surgically debrided, achieving clear margins. Intraoperative findings confirmed involvement limited to skin and subcutaneous tissue, sparing underlying muscle and bone (Figure 2). Tissue was sent for bacterial and fungal cultures. The open wound was treated with daily topical povidone-iodine dressings alongside strict surveillance of the patient’s fluid and electrolyte status. The raw area remained uncovered by a skin graft and left to heal by second intention. Cutaneous erythema progressively resolved, and the infant exhibited marked clinical improvement with accelerated weight recovery.
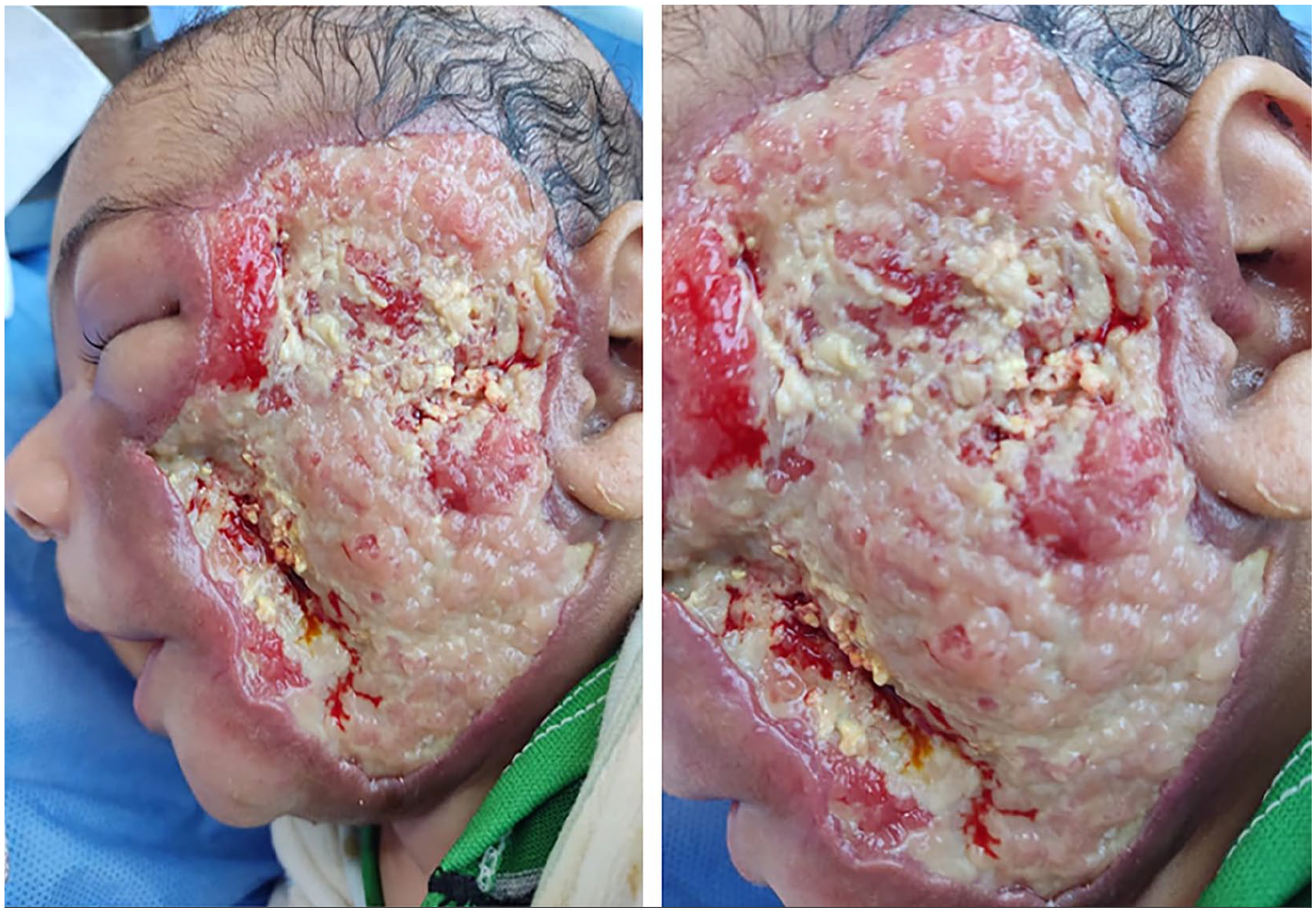
Radical surgical debridement to excise all necrotic tissue.
Liposomal amphotericin B (LAmB) was initiated immediately postoperatively at 5 mg/kg/day, with close monitoring for nephrotoxicity and electrolyte imbalances, which were not observed. Repeat biopsies performed 1 month post-debridement, following clinical improvement and resolution of inflammation (Figure 3), showed no residual fungal elements on histopathology. Antifungal therapy was continued for a total of 6 weeks, guided by sustained clinical resolution. At 3-month follow-up, the infant suddenly deteriorated and died suddenly.
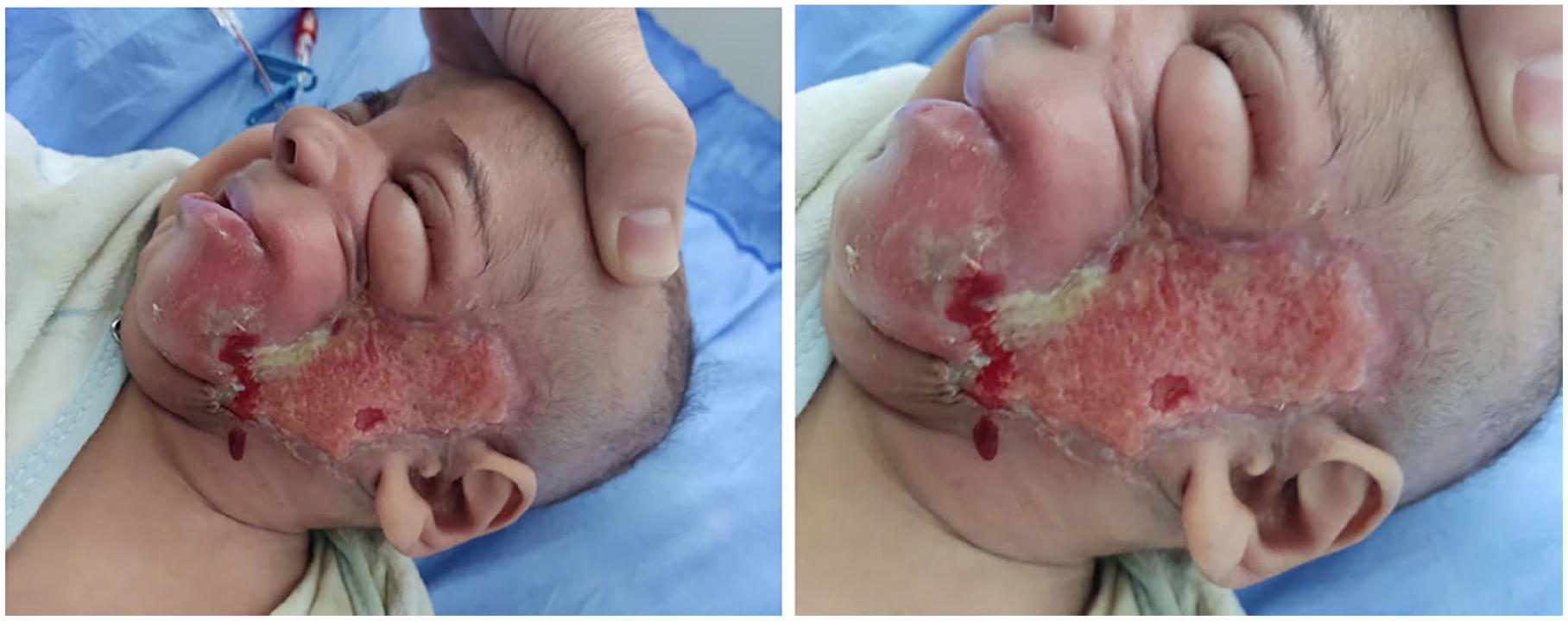
One-month post-debridement showing clinical improvement.
Discussion
Mucormycosis is a serious invasive fungal infection that primarily affects immunocompromised patients. 13 Cutaneous mucormycosis represents the third most common form of the disease, following pulmonary and rhinocerebral manifestations. 18 Rarer forms include gastrointestinal, renal, and palatal mucormycosis, which have been infrequently reported in the literature.20,21 Cutaneous mucormycosis of the face is a rare, life-threatening fungal infection that can occur in both immunocompromised and immunocompetent individuals. 8 The infection may arise as a primary localized presentation in the facial skin or secondary to disseminated or rhinocerebral mucormycosis. 12 The primary type is often seen in infants and neonates. While classically associated with immunocompromising conditions such as organ transplantation, HIV, 10 diabetes mellitus, 11 hematologic malignancies, 11 aplastic anemia, 11 and prolonged corticosteroid use. 11 It has also been documented in trauma patients, burn victims, 8 and infants.9-11 Prematurity has been documented as a risk factor. 3 COVID-19 has recently also emerged as an additional risk factor linked to cases of facial cutaneous mucormycosis, particularly in regions with high infection rates.22,23
Facial cutaneous mucormycosis also occurs in 20% to 30% of immunocompetent individuals. In these cases, infection is often linked to traumatic inoculation 9 or environmental exposure. 18 The absence of overt trauma or comorbidities in this case parallels reports of primary cutaneous mucormycosis in neonates and infants, 10 where minor abrasions or subclinical breaches in skin integrity may serve as portals for fungal entry. 11 Genetic or immunological susceptibilities in immunocompetent hosts are proposed by Walsh et al. 15 Notably, Dave et al 10 report the first documented case of a full-term immunocompetent infant surviving facial cutaneous mucormycosis.
Infection of the facial area can remain localized or rapidly progress as a necrotizing skin lesion to deeper structures, including surrounding bones, muscles, tendons, and parotid gland.8,11 It is less frequently seen as a component of disseminated infection. 14 The rapid progression from a localized boil to extensive necrosis aligns with cases of post-traumatic mucormycosis described by Jundt et al, 9 emphasizing the organism’s propensity for angioinvasion and tissue destruction even in immunocompetent hosts. However, our patient, as we observe when we debride his necrotic lesion, has horizontal spread rather than vertical invasion into surrounding muscles and tendons. Notably, the infant’s normal inflammatory markers and lack of systemic symptoms initially masked the severity of infection, a phenomenon also observed in cases reported by Ben Rejeb et al. 11 Misdiagnosis as pyoderma gangrenosum or bacterial necrotizing fasciitis, as described by Kerr et al, 16 underscores the need for early biopsy in necrotic lesions unresponsive to empirical therapy. The misdiagnosis can be explained by the fact that the lesions in cutaneous mucormycosis start with painful erythema and induration, and progressively become necrotic as they evolve over several days. 13 Tissue histopathological examination remains the diagnostic cornerstone, with broad, pauci-septate hyphae, and right-angle branching confirming infection.13,15,18 Fungal cultures often fail to isolate Mucorales species due to their fastidious growth requirements, as seen here and in prior studies. 19 However, molecular methods like PCR or metagenomic next-generation sequencing could enhance diagnostic accuracy in culture-negative cases,14,19 though these were unavailable in our setting.
Management of mucormycosis involves extensive surgical debridement combined with systemic antifungal therapy.8,9 First-line treatment typically includes amphotericin B administered alongside surgical intervention, while posaconazole and isavuconazole serve as second-line options. 13 The combination of surgical debridement and LAmB remains the gold standard, achieving survival rates of ~68% in pediatric populations.10,14 In the present case, radical excision paired with LAmB (5 mg/kg/day) successfully controlled the infection, with follow-up biopsies confirming eradication at 1 month.
While cutaneous mucormycosis has the lowest mortality (15%-25%) among mucormycosis forms, 13 this case highlights exceptions. Risk factors for poor outcomes include delayed diagnosis, inadequate debridement, and limited access to antifungals.8,14 Notably, our patient’s rapid progression and fatal outcome despite protocol-adherent care suggest unique virulence factors, host-pathogen interactions that lead to an unpredictable outcome. Early diagnosis and treatment are crucial for improving outcomes as antifungal therapy and surgical intervention were associated reduction in mortality risk. 3 In most cases, prevention through management of risk factors is the most effective approach to avoid fatal progression. 11 Despite early histopathological confirmation, radical surgical debridement, and timely antifungal therapy, the patient experienced a fatal outcome after apparent clinical resolution. This paradoxical trajectory highlights critical gaps in understanding the pathogenesis, monitoring, and long-term management of mucormycosis in pediatric populations, particularly in resource-limited settings.
The high mortality rate is linked to factors such as delayed treatment, disseminated infection, and infancy (age <1 year).3,12 In this case, the patient’s young age likely played a critical role in the poor prognosis. Delayed access to antifungals or diagnostics also worsens outcomes. 13 Advocacy for equitable access to antifungals and molecular diagnostics is critical, particularly in regions with high environmental Mucorales exposure.14,17
Conclusion
This case of pediatric cutaneous mucormycosis in an immunocompetent infant illustrates the aggressive nature of Mucorales infections, even in the absence of classic risk factors. Early histopathological diagnosis, aggressive surgical intervention, and timely antifungal therapy are critical to survival, yet fatal relapse remains a devastating possibility. The infant’s sudden deterioration after initial therapeutic success suggests subclinical persistence of fungal elements or undetected dissemination, challenging current monitoring strategies. While cutaneous mucormycosis carries a lower mortality rate than other forms, outcomes depend on rapid diagnosis, resource availability, and prolonged antifungal courses. This report underscores the importance of maintaining a high index of suspicion for mucormycosis in necrotic lesions unresponsive to conventional therapy, regardless of immune status. Multidisciplinary collaboration, equitable access to antifungals, and integration of molecular diagnostics are essential to mitigate disparities in care. Further research into host-pathogen dynamics and immunological vulnerabilities in immunocompetent children is urgently needed to refine management and prevent unexpected mortality.
Footnotes
Acknowledgements
The authors acknowledge the patient and his family for their cooperation.
Consent for Publication
Written informed consent was obtained from the parents of the child for the publication of this case report, including anonymized clinical details and any accompanying data.
Author Contribution
Yasser ALGhabra: conceptualized the case report, managed patient care, collected clinical data, drafted the manuscript, and incorporated revisions. Hossam Alhraki: conducted literature review and analyzed diagnostic data. Vivian Alahmad: contributed to writing the introduction section and collected clinical data. Hisham Alshwaikh: supervised the clinical management, revised the manuscript for intellectual content, and approved the final version. All authors reviewed and approved the manuscript prior to submission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the author or the corresponding author upon reasonable request.
