Abstract
Objective:
This study described the endoscopic characteristics of recurrent anterior epistaxis (RAE) with and without allergic rhinitis (AR), and the outcomes of treatment with mupirocin ointment.
Material and Methods:
Children with RAE were examined using nasal endoscopy, serum specific immunoglobulin E antibodies, and skin-prick test. The children were divided into AR (RAE with AR) and non-AR (RAE without AR) groups. All children received mupirocin ointment topically twice daily for 4 weeks. Recurrent epistaxis and epistaxis severity scores were compared at 2, 4, and 12 weeks of follow-up.
Results:
A total of 326 patients with 432 nostrils with RAE were included; 76.4% of patients had the bad habit of frequent nose-picking or rubbing. Of the 178 nostrils in the AR group, diffuse telangiectasia was found in 89.3%, scattered telangiectasia in 8.4%, and mucosal ulceration in 2.3%. Of the 254 nostrils in the non-AR group, the endoscopic findings of the anterior septum in the bleeding nostrils included mucosal ulceration in 77.6%, scattered telangiectasia in 17.7%, isolated punctate telangiectasias in 3.1%, and diffuse telangiectasia in 1.6%. In addition, the prevalence of mucosal crusts in the non-AR group was significantly higher than in the AR group (76.8% vs 20.8%), whereas the prevalence of severe hypertrophic inferior turbinates or nasal secretion was significantly higher in the AR group. The prevalence of recurrent epistaxis was significantly higher in the AR group than in the non-AR group at 2 (71.3% vs 57.7%), 4 (65.7% vs 37.8%), and 12 (78.1% vs 43.5%) weeks post-treatment.
Conclusions:
AR was common in children with RAE, with prominent diffuse telangiectasia evident by endoscopy in most, while most non-AR RAE children had mucosal ulceration or scattered telangiectasia. Compared with outcomes in non-AR RAE children, the efficacy of mupirocin ointment in the treatment of RAE with AR was not satisfactory.
Introduction
Recurrent anterior epistaxis (RAE) in the anterior portion of the nasal septum in children is a very common reason for ENT consultation and in pediatric emergencies.1-3 RAE is caused mainly by chronic inflammation and trauma.2,3 Saline nasal spray and certain emollients are typically prescribed for RAE in clinical settings. However, these treatments have been proven effective only for crusting and nasal dryness. Nasal colonization with Staphylococcus aureus is more frequent in children with RAE, potentially contributing to inflammation and neovascularization, which can result in epistaxis. 3 Therefore, in most hospitals, antiseptic cream is a common treatment for RAE in children.1-3 Nevertheless, its effectiveness ranges from 29% to 55%, reflecting a high failure rate.3-5 Mupirocin ointment (2%) is a topical antimicrobial agent that may be beneficial in managing S. aureus infections, particularly those caused by methicillin-resistant S. aureus.6,7
Recent studies have implied that RAE in children is closely related to allergic rhinitis (AR), with an incidence of secondary RAE of AR of 46.5% to 71.25%.8,9 However, most parents and general practitioners are unfamiliar with the symptoms of AR and focus solely on epistaxis, overlooking the presence of AR. Although nasal steroid spray combined with an antiallergic agent has been recommended for AR management,8,9 the resolution rate of epistaxis in patients with AR remains below 60%, and treatment adherence is often poor. 3 Moreover, the reasons for the limited efficacy of antiseptic cream in certain RAE cases remain unclear. In addition, little published data exist on the endoscopic clinical features of RAE with and without AR. Therefore, this study described the endoscopic clinical characteristics of RAE with and without AR, and the outcomes of treatment with mupirocin ointment, to provide a morphological basis for preliminary screening of RAE with and without AR by general practitioners.
Materials and Methods
Ethical Considerations
The study protocol was approved by the Institutional Ethical Review Board of the First People’s Hospital of Longwan District (approval number: K2018-IRB-068). Written informed consent was obtained from the parents of all participants.
Methods
This prospective study included children with RAE who were first brought to the ENT outpatient clinic between January 1, 2019, and September 30, 2023. Inclusion criteria were age more than 3 and <14 years, epistaxis as the primary reason for first visit to the outpatient clinic, no previous use of intranasal corticosteroids or anti-allergy medications, recurrent epistaxis with at least 2 episodes in 1 month, duration of epistaxis exceeded 3 months, and at least 3 months of follow-up. Exclusion criteria were as follows: prior treatment with antiseptic cream or intranasal corticosteroids; chemocautery or electrocautery intervention; obvious nasal anatomic abnormalities, excluding nasal septum deviation; other systemic diseases, including hematological, immune system, hepatic, and renal diseases; and secondary epistaxis induced by a foreign body, tumor, or recent trauma.
All children received a routine ENT examination to evaluate the source of bleeding and the presence of rhinitis, sinusitis, or tumor. A complete blood count, specific immunoglobulin E (sIgE) antibody test, skin-prick test (SPT), blood chemistry (liver and kidney function tests), bleeding time, prothrombin time, and activated partial thromboplastin time were obtained. The participants’ parents were asked to report their children’s nasal symptoms.
RAE was divided into AR and non-AR groups. The diagnosis of AR was based on the Chinese Guideline for the Diagnosis and Treatment of Pediatric Allergic Rhinitis (2022 revision), which defines AR as a positive sIgE or positive SPT result along with at least one of the following symptoms: sneezing, runny nose, nasal obstruction, itchy nose, or itchy eyes. 10 The epistaxis severity score (ESS) is used to simply assesses the severity of epistaxis and treatment efficacy, it is a partially validated survey and uses 6 measures to assess the severity of epistaxis: frequency, duration, and intensity of epistaxis episodes, need for medical attention, anemia, and dependence on blood transfusions. The ESS scale is 0 to 10, where 0 is no epistaxis and 10 is the maximum severity of bleeding (0-4 mild, 4-7 moderate, and 7-10 severe).11,12
Endoscopic Assessment and Classification
Based on previously described classifications of vascular patterns,13-15 the endoscopic morphological characteristics of the anterior nasal septum were categorized as follows: diffuse telangiectasia, characterized by multiple visible tortuous or strip-shaped dilated vessels occupying most of the anterior septum (Figure 1); scattered telangiectasia, with a few such vessels distributed across the anterior septum (Figure 2); isolated telangiectasias, either single dilated vessels (Figure 3(A)) or single prominent, punctate hemangiomatoid lesions (Figure 3(B)); mucosal ulceration; mucosal vessels with crusting (Figure 4); hypertrophic inferior turbinates; and mucous secretion in the nasal cavity.
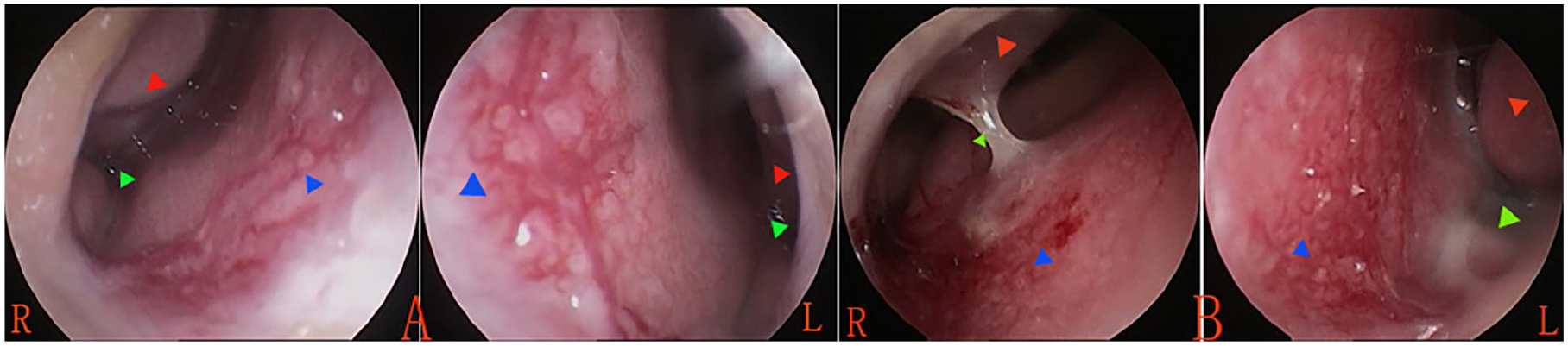
Diffuse telangiectasia. (A) Male, 9 years, SPT++++, hypertrophic inferior turbinate, and mucous secretion; (B) female, 8 years, SPT++, hypertrophic inferior turbinate, and mucous secretion. The red triangle indicates the hypertrophic inferior turbinate, the green triangle indicates mucous secretion from the nasal cavity, and the blue triangle indicates dilated vessels. SPT, skin-prick test.
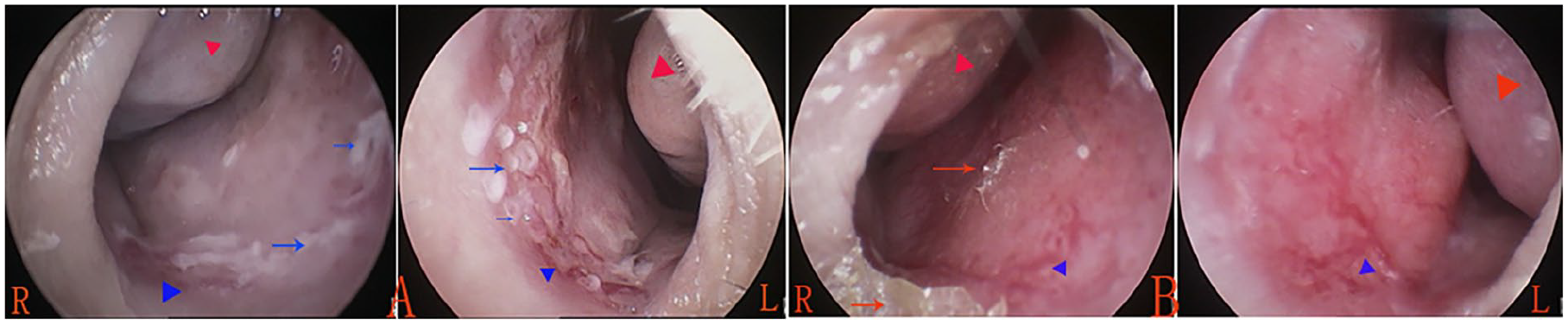
Scattered telangiectasia. (A) Male, 11 years, SPT++++, hypertrophic inferior turbinate, and mucous secretion; (B) male, 8 years, sIgE−, SPT−, nasal dryness, and hypertrophic inferior turbinate. Red triangle indicates the hypertrophic inferior turbinate; blue triangle indicates dilated vessels. Blue arrows indicate mucosal ulceration; red arrows indicate crusts. sIgE, specific immunoglobulin E; SPT, skin-prick test.
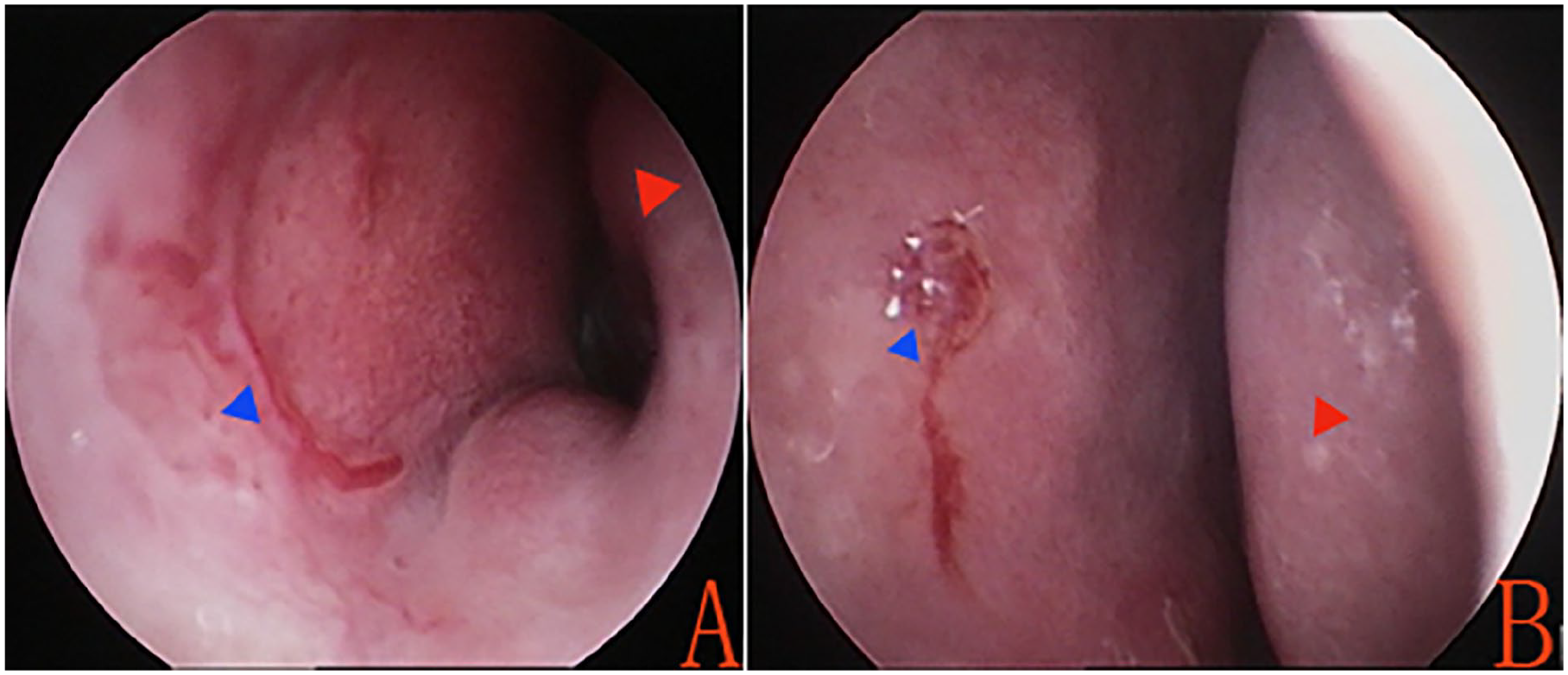
Isolated telangiectasias. (A) Single dilated vessel; (B) single prominent, punctate hemangiomatoid lesion. Red triangle indicates the hypertrophic inferior turbinate; blue triangle indicates dilated vessels.
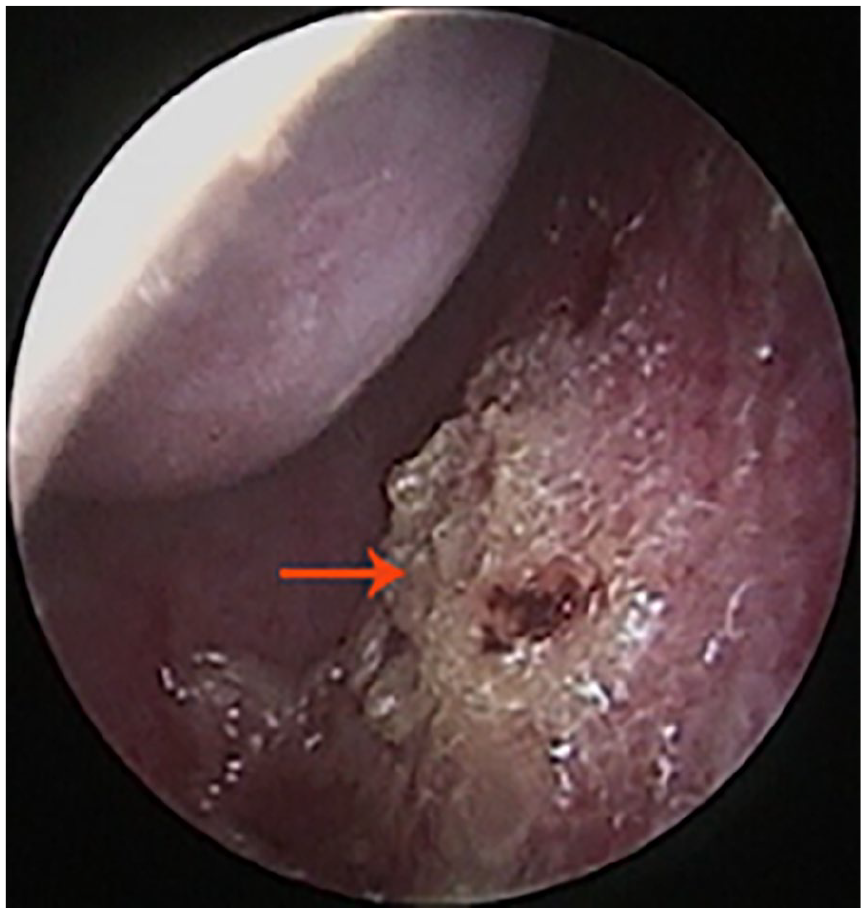
Nasal dryness and mucosal vessels with crusts. Red arrows indicates the crusts.
Treatment Method
All of the children were treated twice daily with mupirocin 2% nasal ointment for 4 weeks. They were instructed to apply the ointment in 1 or both nostrils and to then pinch the nostrils together for a few seconds to distribute it on the nasal septum.
Follow-Up
Standard care included oral desloratadine and saline nasal spray administered for 4 weeks in both groups. ESS and nasal endoscopy were conducted at the initial visit and at 2, 4, and 12 weeks post-treatment. A bleeding event at the same site, confirmed by any doctor and requiring intervention during follow-up, was considered recurrent epistaxis. Interventions included applying pressure to the nose for more than 5 minutes, silver nitrate cautery, or additional treatment. Outcome measures included the incidence of recurrent epistaxis, as well as ESS at 2, 4, and 12 weeks of follow-up.
Statistical Analysis
Statistical analyses were performed using SPSS software (v. 22.0; IBM, Inc., Armonk, NY, USA). Continuous variables are presented as mean ± standard deviation, and categorical variables are presented as frequencies or percentages. Fisher’s exact tests and Mann–Whitney U tests were utilized to assess intergroup differences in patient characteristics, surgical parameters, and mean ESS at each follow-up interval, as appropriate. Friedman’s test was used to evaluate significant differences in repeated ESS measures within each treatment group over time. P < .05 was considered statistically significant.
Results
Demographic Data
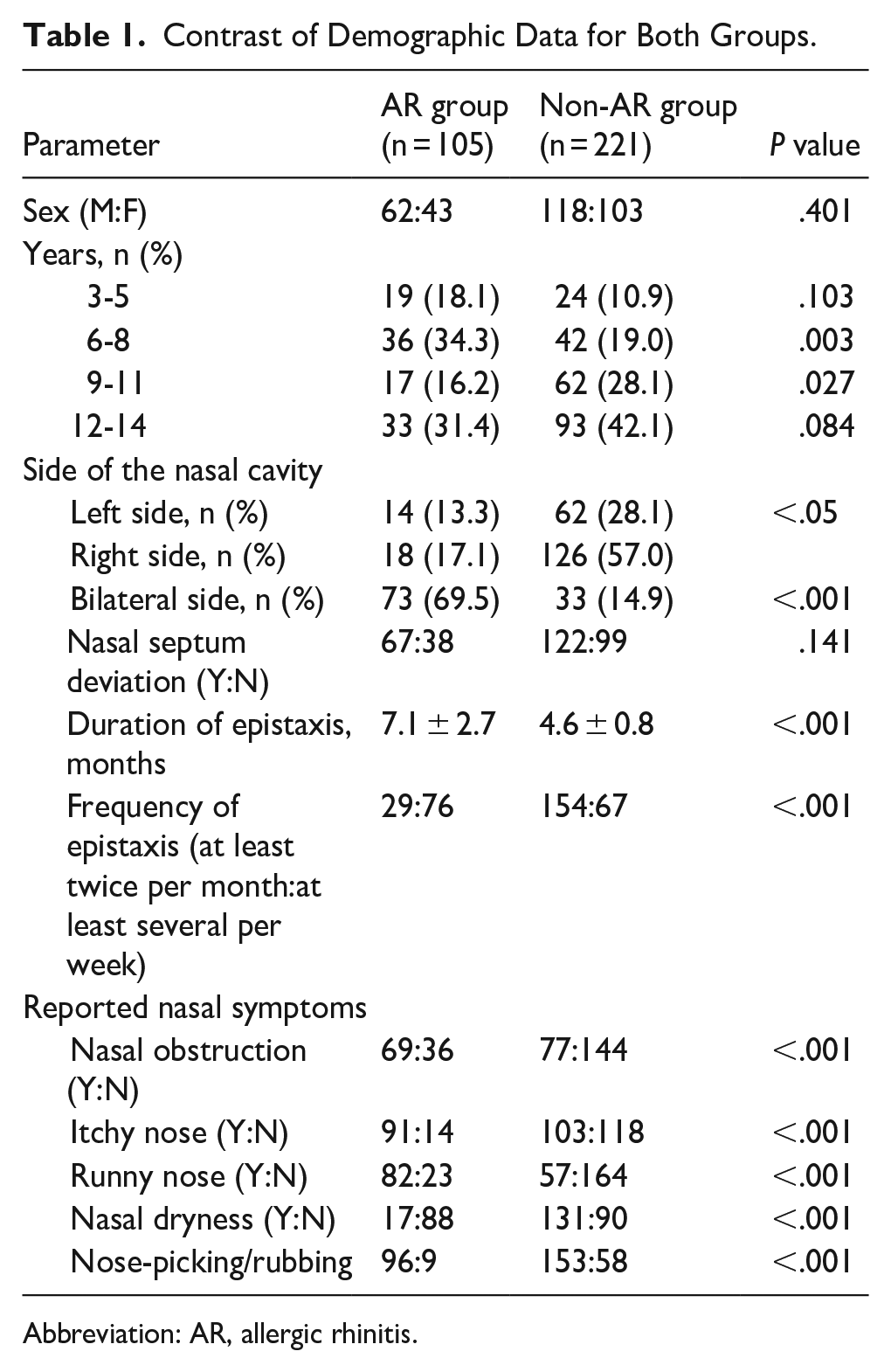
This study included 326 children with RAE: 105 (32.2%) in the AR group and 221 (67.8%) in the non-AR group. Patient sex, mean age, affected side of the nasal cavity, and duration and frequency of epistaxis are summarized in Table 1. Of the 105 patients with AR, 69 (65.7%) were initially misdiagnosed with frequent colds, and 36 (34.3%) with chronic rhinitis.
Contrast of Demographic Data for Both Groups.
Abbreviation: AR, allergic rhinitis.
Of the 326 patients, the other complaints were nasal obstruction in 44.8% of patients, itchy nose in 59.5%, runny nose in 42.6%, and nasal dryness in 45.4%. In addition, 76.4% of patients had the nose-picking or rubbing behavior. The symptom’s rate in the AR patients was significantly higher than that of the non-AR patients in terms of nasal obstruction, or itchy nose, or runny nose, on the contrary, the nasal dryness rate in the non-AR patients was significantly higher than that of the AR patients (59.3% vs 16.2%). In addition, nose-picking/rubbing behavior rate in the AR patients was significantly higher than that of the non-AR patients (91.4% vs 69.2%). Of the 326 patients, 58.0% of patients had nasal septum deviation, while no significant difference was found between AR and non-AR patients for the number of nasal septum deviations (63.8% vs 55.2%).
No patient had previously received intranasal corticosteroid treatment. Recurrent epistaxis was the primary reason that the patient was first brought to the outpatient clinic. All of the children in the AR group declined allergen-specific immunotherapy. In addition, in the non-AR group, 8 nostrils with isolated punctate telangiectasias underwent bipolar coagulation treatment; these children were not included in the outcome analysis.
Endoscopic Findings
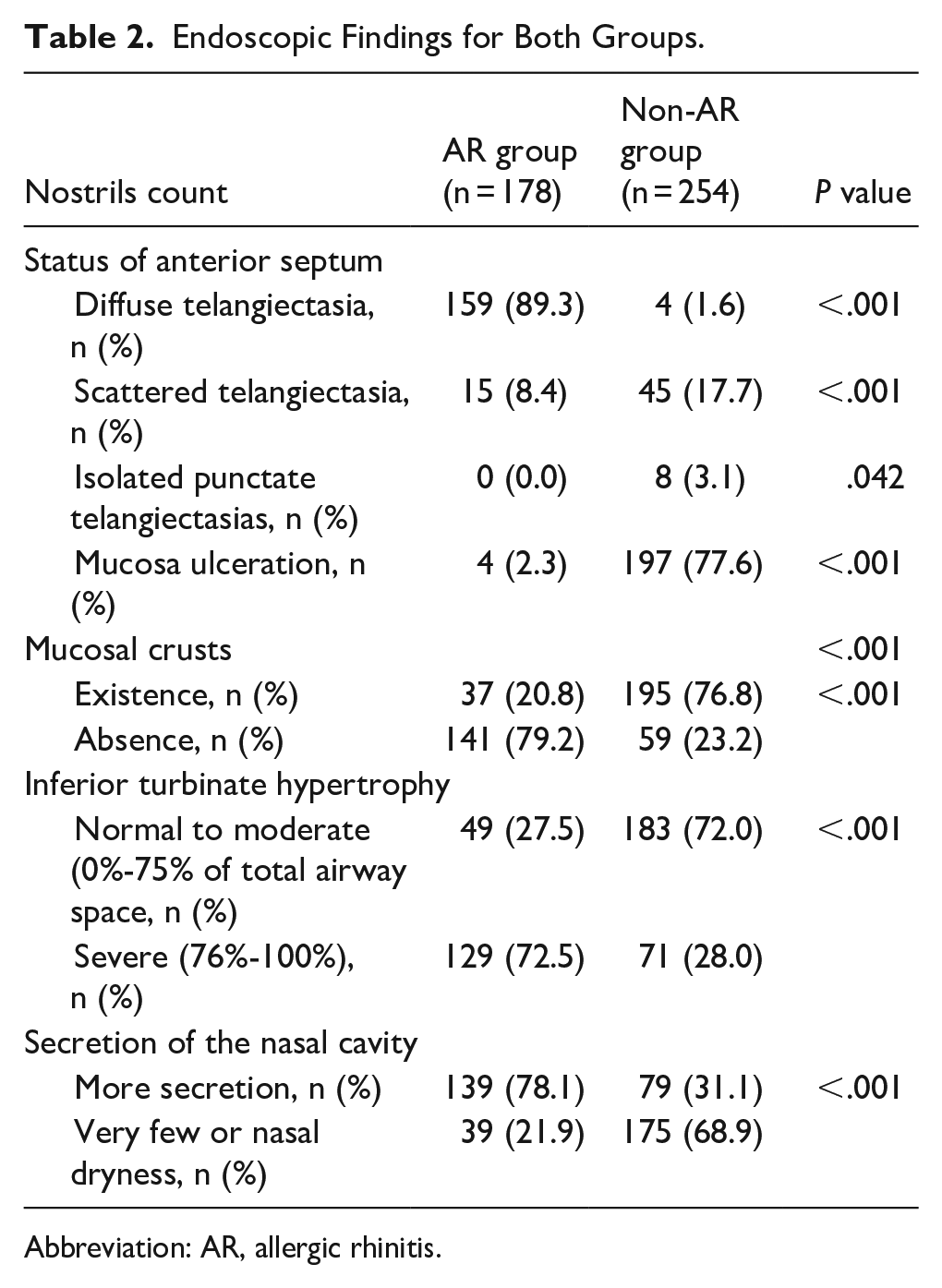
Table 2 summarizes the endoscopic findings. In the AR group, of the 178 nostrils with RAE, the endoscopic findings included diffuse telangiectasia (159, 89.3%), scattered telangiectasia (15, 8.4%), and mucosal ulceration (4, 2.3%). In the non-AR group, of the 254 with RAE, the endoscopic findings included mucosal ulceration (197, 77.6%), scattered telangiectasia (45, 17.7%), isolated punctate telangiectasias (8, 3.1%), and diffuse telangiectasia (4, 1.6%). Most telangiectasias protruded from the nasal mucosa. The prevalence of diffuse telangiectasia was significantly higher in the AR group than in the non-AR group (89.3% vs 1.6%), whereas the prevalence of mucosal ulceration was significantly higher in the non-AR group (77.6% vs 2.3%). The prevalence of mucosal crusts in the non-AR group was significantly higher than in the AR group (76.8% vs 20.8%). In addition, the prevalence of severe hypertrophic inferior turbinates or nasal secretion was significantly higher in the AR group.
Endoscopic Findings for Both Groups.
Abbreviation: AR, allergic rhinitis.
Outcomes
The parents reported that 13 (12.4%) patients in the AR group and 39 (18.2%) in the non-AR group were not compliant with the full course of antiseptic cream; the difference was not significant (P = .243). The antiseptic cream outcomes are shown in Table 3.
Recurrent Epistaxis and ESS at Baseline and 2, 4, and 12 Weeks for Both Groups.
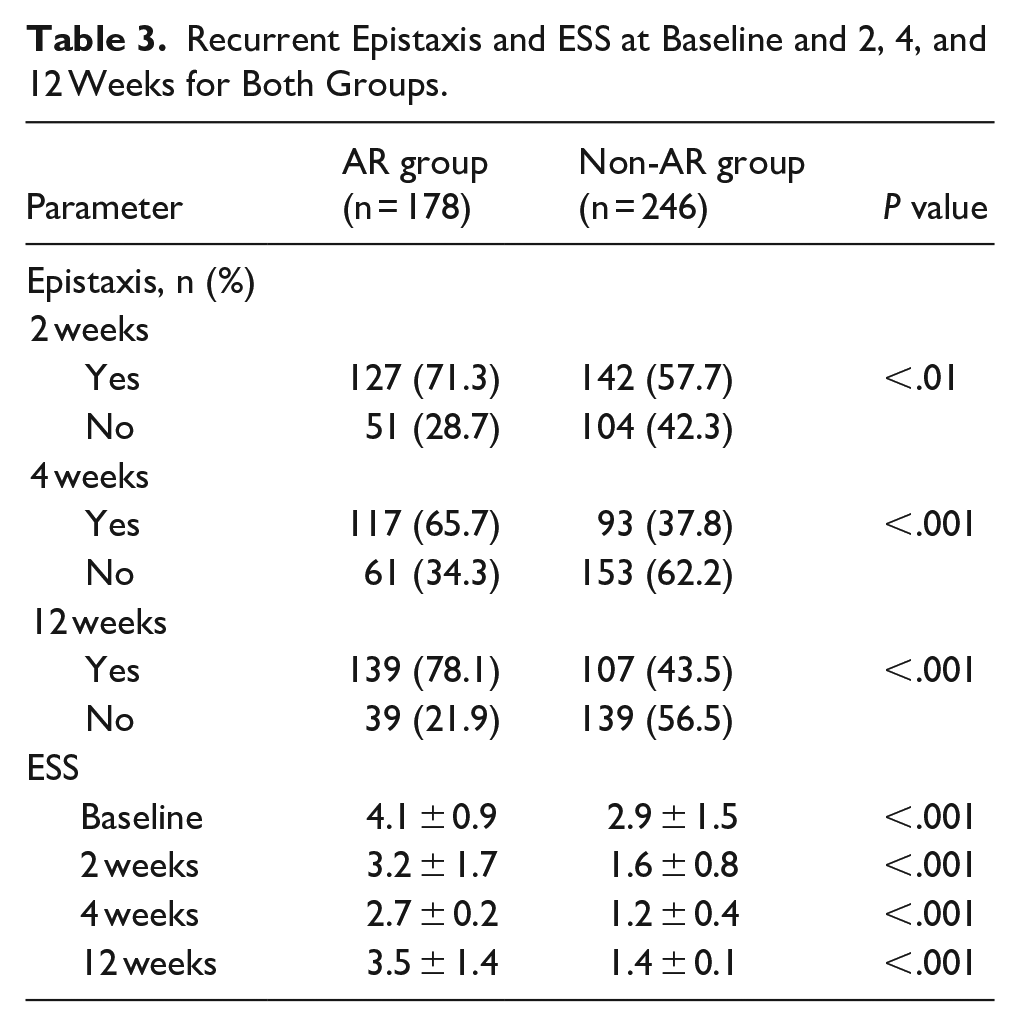
The absence of epistaxis was 28.7% nostrils at 2 weeks, 34.3% nostrils at 4 weeks, and 21.9% nostrils at 12 weeks in the AR group, whereas the absence of epistaxis was 42.3% nostrils at 2 weeks, 62.2% nostrils at 4 weeks, and 56.5% nostrils at 12 weeks in the non-AR group. The prevalence of recurrent epistaxis was significantly higher in the AR group than in the non-AR group at 2 (71.3% vs 57.7%), 4 (65.7% vs 37.8%), and 12 (78.1% vs 43.5%) weeks post-treatment.
The baseline ESSs were significantly higher in the AR group than in the non-AR group. Although ESSs were lower in the AR group at 2 weeks post-treatment, the difference compared with baseline was not significant (P = .085) and did not reach significance at 4 (P = .061) or 12 (P = .113) weeks. By contrast, in the non-AR group, there were significant differences in ESSs from baseline values at 2 (P < .001), 4 (P < .001), and 12 (P < .001) weeks, although there was no significant difference in the mean ESSs at the different follow-up times. In addition, after 2 to 4 weeks of treatment, the crust was reduced and mucosa ulcerations were healed in most patients, whereas the visible prominent vessel did not significantly change (Figure 5).
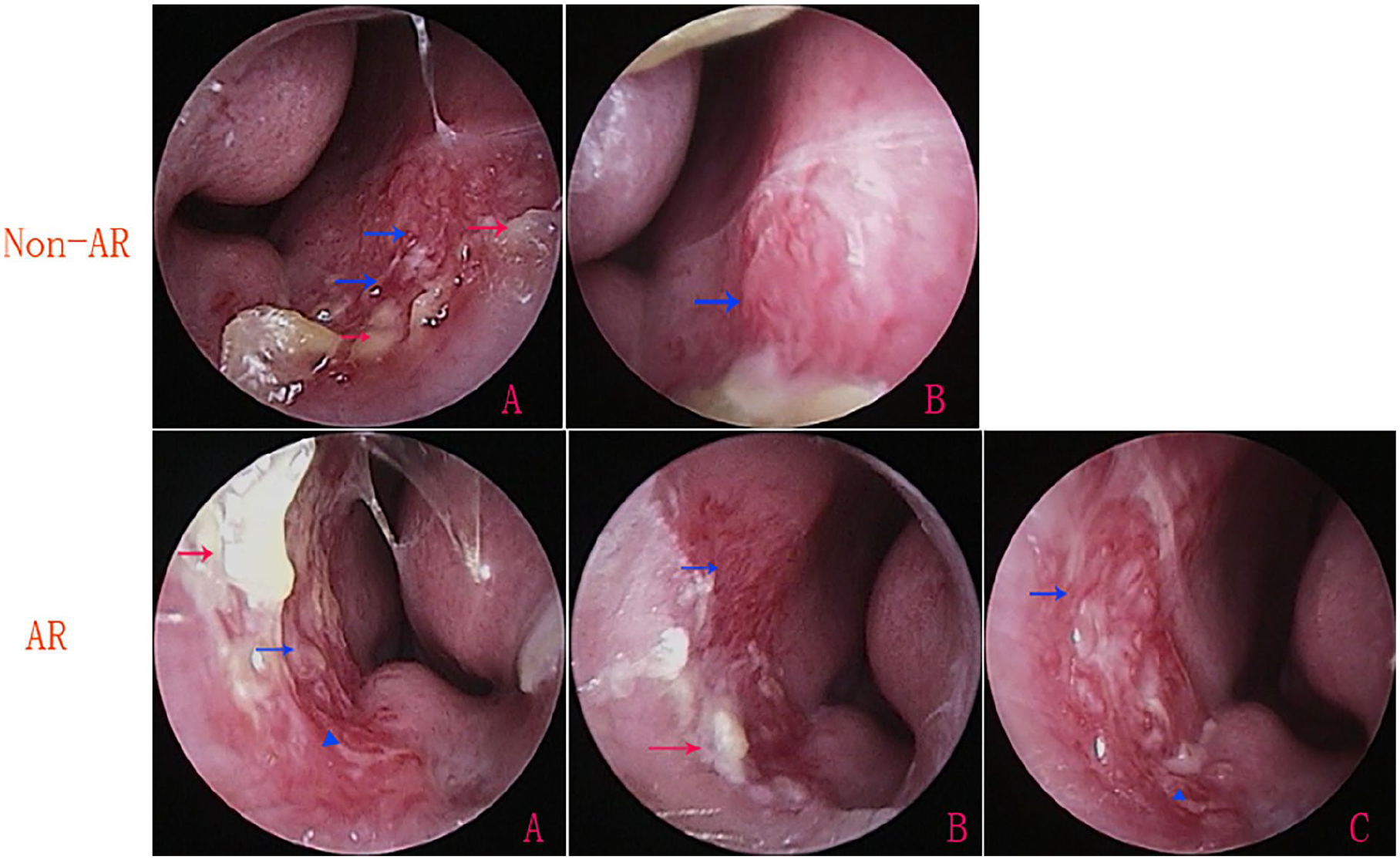
Non-AR patient. (A) Pre-treatment; (B) post-treatment 4 weeks. AR patient. (A) Pre-treatment; (B) post-treatment 2 weeks; and (C) 4 weeks.
In the non-AR group, 7.2% (16/221) patients underwent silver nitrate cautery, and 0.9% (2/221) underwent bilateral electrocoagulation cautery after 12 weeks of ointment treatment; the other patients were not given additional treatment. However, although the AR group had a high failure ointment treatment, only 8.6% (9/105) of patients were willing to receive silver nitrate cautery, and 1.0% (1/105) in bilateral electrocoagulation cautery; other patients refused to accept cautery treatment, who further treated with intranasal corticosteroids combined with oral anti-allergy medication.
Discussion
Several studies have described the morphology of bleeding points in children with RAE. In Montague et al, 13 half of the children had prominent vessels, while Folz et al 14 found that in patients with nasal HHT, the vessels were shaped like spots, loops, or spiders, or resembled raspberries. Mahoney and Shapshay 15 reported that the intranasal vascular patterns included isolated punctate telangiectasias, diffuse interconnecting vasculature, and arteriovenous malformations. Other studies have identified nasal mucosal ulceration.2,4,5
By contrast, the patients in our study had not received prior intranasal corticosteroid treatment, and recurrent RAE was the primary reason for their initial visit. The endoscopic findings in non-AR cases differed from those in patients with AR. The prevalence of diffuse telangiectasia was significantly higher in the AR group than in the non-AR group, whereas mucosal ulceration was significantly more common in the non-AR group. In addition, 57.0% were on the right side and 14.9% were bilateral among the non-AR RAE patients, whereas 69.5% were bilateral in those with RAE with AR. The baseline ESSs were significantly higher in the AR group than in the non-AR group. Furthermore, a hypertrophic inferior turbinate or nasal secretion in most cases of RAE with AR. These results imply that AR should be strongly suspected in patients presenting with frequent bilateral RAE and diffuse telangiectasia; in such cases, further consultation and laboratory testing are warranted.
Nose-picking and nose-rubbing are considered risk factors for RAE in children.3,16
Teo et al 3 reported that 90% of AR patients had nose-picking/rubbing behavior. In this study, the nose-picking/rubbing habit rate in the AR patients was significantly higher than that of the non-AR patients (91.4% vs 69.2%). The underlying triggers for these bad behaviors may differ between patients with AR and those without AR. Chronic inflammation and nasal dryness may be the primary causes in non-AR patients, whereas rhinorrhea, itchy nose, and nasal obstruction may be the primary causes in AR patients. These associations have been demonstrated by the present study and in previous studies. In this study, the nasal symptoms in the AR patients were significantly higher than those of the non-AR patients in terms of nasal obstruction, itchy nose, or runny nose, whereas the nasal dryness in the non-AR patients was significantly higher than that of the AR patients. Aksakal and Şahin 17 reported the effects of inflammatory factors in the etiology of epistaxis with non-AR. Other researchers have noted that rhinorrhea and itchy nose are significantly associated with RAE in children with AR. 3 In addition, we found that RAE was significantly more common on the right than on the left side in patients with non-AR, further implying right-handed nose-picking behavior. By contrast, RAE was predominantly bilateral in AR patients. It is well known that frequent nose-picking or rubbing leads to mucosal injury, mucosal ulceration, and neovascularization, thereby causing epistaxis.3,13,16
AR is an allergic inflammation in the nasal mucosa after exposure to allergens induced by a Th2-dominated immune response with increased levels of serum-specific IgE, and subsequent crosslinking of the high-affinity IgE receptor on mast cells by IgE-antigen complexes, culminating in mast cell degranulation and releasing the angiogenic factors.18,19 Angiogenesis and microvascular remodeling are considered important events in tissue remodeling in allergic diseases, which include prominent increases in vessel number, vessel size, vascular surface area, and microvascular leakage in the nasal mucosa.20,21 In addition, a disorder of hemostasis is suspected as the contributing factor of recurrent epistaxis in AR patients. 22
The nasal branch of the anterior ethmoidal artery’s septal branch (NbAEA) supplies blood to the anterior nasal septum. NbAEA ligation is recommended for treating persistent nosebleeds and achieving effective hemostasis. 23 However, such ligation is typically unnecessary in pediatric RAE. Nasal colonization by S. aureus is considered an inducing factor for RAE in children,3,13,16 and antibacterial cream is recommended as the first-line treatment.1-3 Reports indicate the absence of epistaxis in 29.8% to 55% of such patients at 4 weeks following antibacterial cream application,4,5,23 and in 23% of patients during 1 year of follow-up. 24 In this study, the absence of epistaxis was 34.3% at 4 weeks and 21.9% at 12 weeks following mupirocin ointment in the AR group, whereas the absence of epistaxis was 62.2% at 4 weeks and 56.5% at 12 weeks in the non-AR group. The possible reasons for this phenomenon were that some allergic and infectious inflammations may recur or persist, leading to nasal mucosal damage or recurrence of mucosal ulceration with the time prolongs and the cessation of treatment. In addition, the differences in reported efficacy may be related to differences in enrollment criteria and follow-up duration; some studies have included both AR and non-AR patients.4,24 Mupirocin ointment has been recommended for the eradication of S. aureus.3,6,7 In this study, mupirocin ointment was used to treat RAE in children. No significant difference in recurrent epistaxis rates was observed between the 2 groups within 2 weeks of treatment, but significant differences were evident at 4 and 12 weeks; the rate was significantly higher in the AR group. Although ESSs were lower in the AR group 2, 4, and 12 weeks after treatment, the reductions did not reach significance compared to baseline. In comparison, in the non-AR group, there were significant differences in ESSs versus baseline at 2, 4, and 12 weeks, although there were no significant differences in the mean ESSs at the different follow-up times.
Next, we address why mupirocin ointment may be more effective for non-AR patients than for AR patients. It may be because non-AR RAE is mainly due to chronic inflammation caused by bacterial infection and nasal dryness. Mupirocin ointment, when combined with nasal irrigation, provides moisturizing and local anti-inflammatory effects that benefit non-AR patients. In a histological analysis, Montague et al 13 observed an inflammatory infiltrate surrounding vessels in non-AR patients. This is further evidence to support the aggressive eradication of nasal Staphylococci with antiseptic creams. However, angiogenesis and microvascular remodeling are long-lasting features of allergic diseases. 21 In this study, the crust was significantly reduced, and mucosa ulcerations were healed in most patients after antiseptic creams, whereas the prominent visible vessels did not change.
The limitation of this study was that the diagnosis of AR was based on serum sIgE and symptoms. Second, there was no histological examination and bacteriological analysis of the nasal mucosa before therapy, and no objective method was used to measure telangiectasia precisely. In addition, although the ESS is a partially validated, standardized tool that is the most frequently used metric for evaluating patient-reported epistaxis severity, it is a subjective test, which is affected by responder description. Third, not all patients were compliant with the full course of antiseptic cream. Certainly, the effect of the ointment could be affected by some factors, including children’s character, age, cooperation degree, and the size of the nostrils. In addition, some patients may apply a saline nasal spray simultaneously with ointment, the effect can also be decreased. Finally, although oral desloratadine and saline nasal spray were administered in both groups, these treatments represent confounding factors that may have affected the assessment of antiseptic cream efficacy.
Conclusions
AR was common in children with RAE, most of whom showed prominent diffuse telangiectasia on endoscopy, while most non-AR RAE showed mucosal ulceration or scattered telangiectasia. Compared with non-AR RAE patients, the efficacy of mupirocin ointment in the treatment of patients with RAE with AR was not satisfactory.
Footnotes
Ethical Considerations
The study protocol was approved by the Institutional Ethical Review Board of the First People’s Hospital of Longwan District (approval number: K2018-IRB-068).
Consent to Participate
Written informed consent was obtained from the parents of all participants.
Author Contributions
Linyan Tao: conception and design of the work, interpretation of data, drafting of the manuscript, agreement to be accountable for all aspects of the work, and final approval of the version to be published. Zhengcai Lou: analysis of data and final approval of the version to be published.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in the published article.
