Abstract

Significance Statement
We present a patient with a subglottic mass that was initially diagnosed as inflammatory myofibroblastic tumor after extensive workup and expert consultations. With the recognition of the recently-established diagnostic entity of anaplastic lymphoma kinase (ALK)-positive histiocytosis, this case was reevaluated and the diagnosis of ALK-positive histiocytosis was confirmed. Laryngeal ALK-positive histiocytosis is exceedingly rare, and to our knowledge, this is the first reported case in adults. Awareness of this entity will ensure accurate diagnosis and appropriate management.
Case History
A 43-year-old female presented with stridor and increasing shortness of breath over 6 months in 2018. An office tracheoscopy revealed a large subglottic submucosal mass occupying ~80% of the patient’s airway (Figure 1A). Computed tomography revealed a 1.6 cm lobulated subglottic mass arising from the left true vocal cord (Figure 1B). The patient underwent biopsy and subsequent endoscopic laser tumor excision. Gross examination of the surgical excision specimen showed fragments of soft tissue without necrosis or hemorrhage. Histological examination revealed a submucosal mass composed of spindle cells and admixed inflammatory infiltrate (Figure 2A and B). Areas of xanthomatous changes were noted with abundant histiocytes and Touton type giant cells (Figure 2C). No significant cytologic atypia, increased mitotic activity, or necrosis was appreciated. Initial immunohistochemistry studies with CD68 and CD163 (Figure 2D) highlighted abundant histiocytes. Pan cytokeratin, P40, and SOX10 were negative. Ki67 showed a low proliferative index of <5%. Special stains with acid-fast bacilli and Gomori Methenamine-Silver were negative for mycobacterial and fungal microorganisms, respectively. Given the abundance of histiocytes and giant cells, a differential of non-Langerhans histiocytic lesion including xanthogranuloma and Erdheim–Chester disease was considered. Additional studies showed diffuse anaplastic lymphoma kinase 1 (ALK1) staining in both the spindled (Figure 2G, ALK1) and xanthomatous areas, whereas there was no staining for CD1a (Figure 2H), BRAF, or SMA (Figure 2I). Multiplex fusion analysis by targeted next-generation sequencing revealed a PRKAR1A::ALK fusion. This case was consulted with experts in head and neck pathology as well as hematopathology and a diagnosis of inflammatory myofibroblastic tumor (IMT) was rendered given the spindled morphology, positive ALK immunostain, and presence of ALK rearrangement. Subsequent clinical follow-up and endoscopic examination revealed normal subglottic and upper tracheal area with no evidence of disease 6 years after the excision. In the last several years, ALK-positive histiocytosis has become an emerging discrete entity, and the diagnostic criteria were first established and included in the fifth edition of WHO Haematolymphoid Tumours (published August 2024). 1 With the recognition of this new diagnostic entity, this case was reevaluated. Further immunohistochemical studies showed that both the spindle and xanthomatous components of this tumor were diffusely positive for additional histiocytic marker PU.1 (Figure 2E) and a different antibody clone of ALK (D5F3; Figure 2F). Careful examination confirmed that both ALK D5F3 and PU.1 stained the same population of lesional cells, supporting the diagnosis of ALK-positive histiocytosis. This change in diagnosis did not impact clinical management since there were no signs of disease recurrence or systemic involvement.
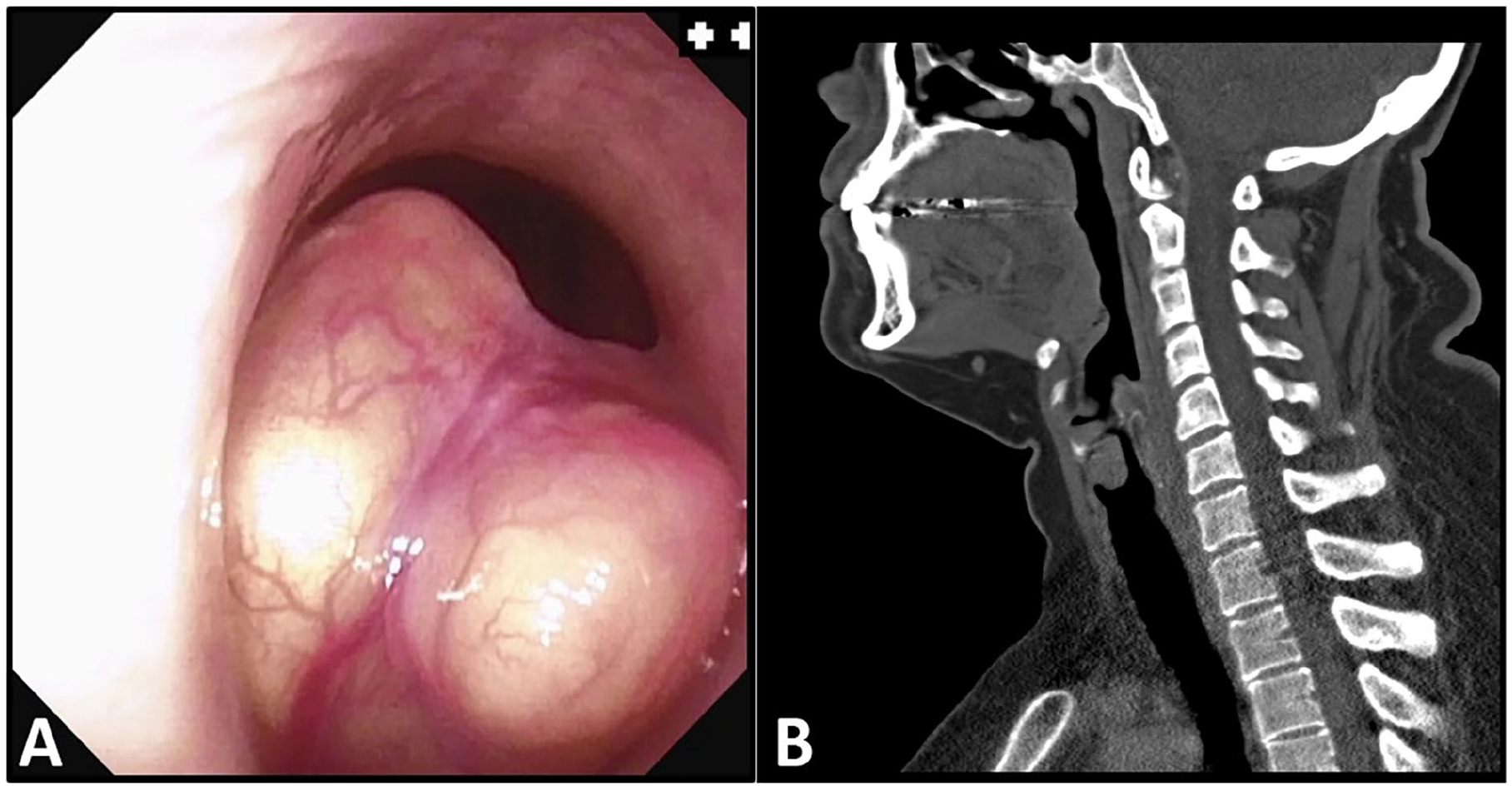
Clinical image studies. (A) Tracheoscopy reveals a large subglottic submucosal mass. (B) Computed tomography reveals a 1.6 cm subglottic soft tissue mass arising from the left true vocal cord.
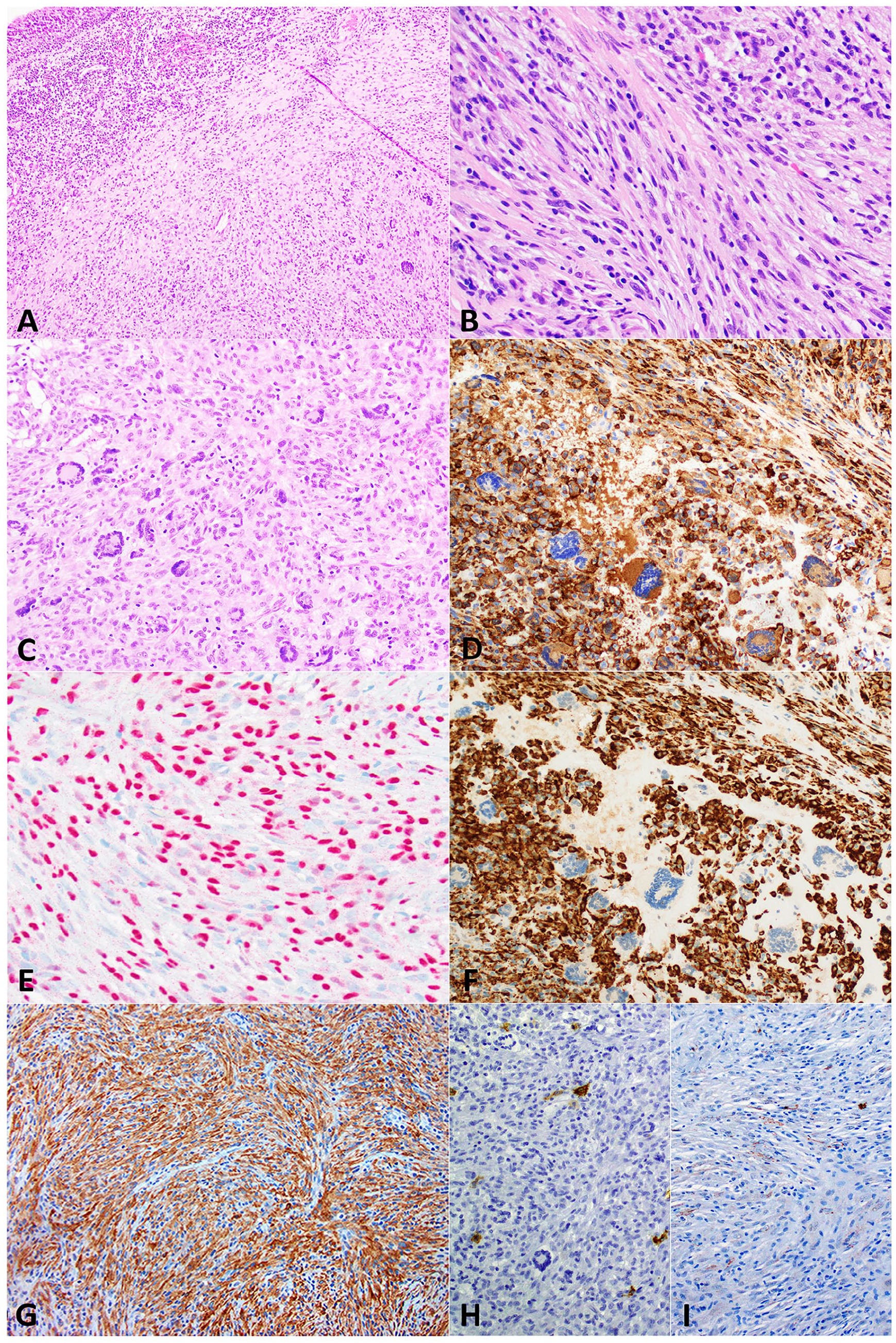
Histomorphology and immunohistochemical stains. (A-C) H&E section (40×, 200×, and 200×, respectively) of the tumor shows a submucosal mass composed mostly of spindle cells with mixed inflammatory cells and scattered giant cells. Mucosa is shown at the upper left corner of (A). (D) CD163 (200×) and (E) PU.1 (200×) show cytoplasmic and nuclear staining of the histiocytes, respectively. (F, G) Two different ALK antibodies, ALK D5F3 (200×) and ALK1 (100×), show cytoplasmic staining in the neoplastic histiocytes with xanthomatous or spindle morphology. (H) CD1a (100×) and (I) SMA (100×) show negative staining. ALK, anaplastic lymphoma kinase.
Discussion
ALK-positive histiocytosis is a recently-established disease entity of histiocytic neoplasm, which is characterized by ALK immunoreactivity and ALK gene rearrangement. It was first described in a small case series with systemic histiocytic proliferation in 3 infants. 2 Since then, especially in the past 5 years, there have been additional reports including a multi-institutional retrospective review of 39 cases further charactering this disease clinically, radiologically, and pathologically.3-5 Patients’ age range from neonates to 51 years old; however, most cases occur in children (~81.5%) and only a small portion involve adults (~18.5%). ALK-positive histiocytosis can be multisystemic with systemic involvement including liver, spleen, and/or bone marrow, as well as skin and central nervous system. Solitary lesion involving a single organ system has also been reported in central or peripheral nervous system, skin, breast, and soft tissue.6-13 Laryngeal ALK-positive histiocytosis is very rare; so far, there was only 1 reported case in a 3-year-old boy, and our case is the first reported laryngeal ALK-positive histiocytosis in adults to our knowledge.
Histomorphologically, ALK-positive histiocytosis typically shows xanthogranulomatous features with plump foamy histiocytes and variable Touton giant cells (~31% cases). Some cases may show spindle or rarely-epithelioid morphology. Nuclear features include ovoid nuclei with slight to significant indentation. Mild cytological atypia has been seen in a small portion of the reported cases, often with epithelioid appearance, whereas overt high-grade atypia is rarely seen. ALK staining patterns and intensity can be variable; cytoplasmic, membranous, or Golgi dot-like staining patterns have been reported. ALK rearrangement is identified in the majority of ALK-positive histiocytosis cases. The most common ALK fusion partner is KIF5B, and others were identified in single case bases, including CLTC, TPM3, TFG, EML4, DCTN1,5,9,10,13 and PRKAR1A (this report).
Accurate diagnosis of ALK-positive histiocytosis can be challenging. Our case shows heterogeneous histomorphology with predominantly-spindle cell cytology and focal xanthomatous changes. The top differential diagnosis of cases with spindle cell cytology is IMT. By immunohistochemistry, the spindle cells in IMT are positive for ALK and SMA (variable degree). It is also difficult to differentiate between reactive histiocytosis and neoplastic histiocytic proliferation by histomorphology alone. Awareness of the diagnostic entity of ALK-positive histiocytosis and careful selection of immunohistochemical stains are essential to reach an accurate diagnosis, in conjunction with molecular genetic studies.
Surgical resection is usually the first line of management for single system disease, and the prognosis is usually good with a reported response rate of 85%. ALK inhibition therapy has been used as a first-line or second-line therapy in multisystem as well as single system disease, with a reported response rate of 100%.5,14,15 The reported effectiveness of ALK inhibition therapy makes it a promising treatment option for multisystemic disease and disease recurrence after other lines of therapy. Given that these treatment and prognosis data were derived from molecularly-confirmed ALK-positive histiocytosis cases, it is prudent to perform molecular test whenever possible, for both diagnosis confirmation and management purposes, even though molecular study identifying ALK fusion is not required for diagnosis. In conclusion, we reported a rare case of laryngeal ALK-positive histiocytosis, and our case highlights the importance of revisiting prior challenging cases with up-to-date knowledge.
Footnotes
Ethical Considerations
This study was conducted in accordance with the ethical standards of the recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journal formulated by the International Committee of Medical Journal Editors (ICMJE).
Consent to Participate
Written informed consent was obtained from all participant(s).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The literature analyzed during the current study are listed in references and are publically available. All data generated during this study are included in this published article.
