Abstract
Objective:
To investigate comorbidities and severity correlations among inner ear malformations (IEMs) classifications and analyze subtype characteristics to optimize preoperative evaluation and cochlear implantation (CI) strategies.
Methods:
Retrospective multicenter data from Hubei Province were analyzed for 336 children with IEMs who received unilateral CI (2012-2020). IEMs were classified and graded using Sennaroğlu and Adibelli criteria, and data visualization tools were used to assess subtypes, comorbidities, and severity correlations.
Results:
Among pediatric candidates for CI with IEMs, isolated enlarged vestibular aqueduct, isolated incomplete partition type II, and multiple malformations were the 3 most common subtypes, accounting for 80.7% of cases. Vestibular aqueduct malformation (VAM) primarily occurred as an isolated condition, whereas internal auditory canal malformation (IACM), cochlear nerve malformation (CNM), cochlear malformation (CM), and vestibular and semicircular canal malformation (VSCM) tended to coexist, exhibiting significant co-occurrence patterns. Severity analysis revealed a positive correlation between the severity of IACM and CNM (r = .13, P = .015), as well as between CM and VSCM (r = .17, P < .001). Additionally, VAM severity was significantly negatively correlated with the severity of IACM, CNM, CM, and VSCM (r = −.39, −.21, −.38, and −.30, respectively; P < .001).
Conclusion:
This study elucidated a detailed analysis of the subtype characteristics and comorbid relationships of IEMs in pediatric candidates for CI. Furthermore, it identified significant correlations between the severity levels of different malformation types. These insights contribute to refining preoperative evaluation protocols and tailoring treatment strategies for CI candidates.
Introduction
Inner ear malformations (IEMs) are a significant cause of hearing impairment in children and are particularly prevalent among patients with sensorineural hearing loss (SNHL). Studies estimate that 20% to 40% of children with congenital SNHL have some form of IEMs.1,2 In addition to directly affecting hearing, IEMs may profoundly impact the development of language, cognition, and social skills.3-5 Cochlear implantation (CI) and auditory brainstem implantation are the primary surgical treatments for IEMs, with this study focusing specifically on CI candidates. 6
IEMs, due to anatomical abnormalities, significantly influence the implementation of CI, particularly resulting in individual variations in electrode placement. 7 İkiz Bozsoy et al’s recent research further demonstrated that even with proper placement of intracochlear electrodes in patients with IEMs, effective auditory nerve stimulation is not guaranteed, highlighting the unique challenges presented by these anatomical variations during CI surgery. 8 Furthermore, different types of IEMs variably affect postoperative hearing and speech outcomes.9,10 Therefore, accurate classification of IEMs is critical for preoperative evaluation and surgical planning. Currently, mainstream classification methods for IEMs are primarily based on embryological and imaging analyses. Among these, the Sennaroglu classification, after multiple revisions, has become a widely-used clinical standard.6,11-13 However, despite ongoing improvements, approximately 26.25% of patients remain unclassified under the existing Sennaroglu system. 6 To address this, Adibelli proposed a refined system based on Sennaroglu’s classification, categorizing IEMs into 5 anatomical subgroups—internal auditory canal (I), cochlear nerve (N), cochlea (C), vestibular aqueduct (A), and vestibular and semicircular canals (V)—and introduced the INCAV grading system to standardize severity assessment. 14 Nonetheless, the complexity of this grading system limits its broader clinical application.
The complex classification and comorbid relationships of IEMs pose significant challenges in decision-making for CI in children. Understanding the subtype characteristics of this population and the relationships between different classifications is therefore crucial. However, most existing studies are based on case analyses with small sample sizes, and there is a lack of systematic reports on the comorbid relationships and severity correlations between different IEM classifications.15-17 This study significantly expands the sample size through a multicenter cohort to address this gap, utilizing the Sennaroglu classification and the INCAV grading system. Venn diagrams were employed to visualize comorbid relationships between IEM classifications, chord diagrams to represent severity correlations, and sunburst charts to illustrate the characteristics of IEM subtypes.
This study investigated the demographic and clinical characteristics of children with IEMs undergoing CI, elucidating comorbid relationships and severity correlations among IEM classifications. Additionally, a comprehensive analysis of subtype characteristics was conducted. Through this study, we aimed to provide clinicians with valuable insights for preoperative CI evaluation and treatment planning, ultimately advancing the precision treatment of children with IEMs.
Patients and Methods
Research Institutions and Ethical Approval
This study was based on retrospective case records from 10 rehabilitation institutions across Hubei Province, which covered a broad geographical area. These institutions were managed under a unified designated rehabilitation center, ensuring data diversity and representativeness. The study protocol was reviewed and approved by the Ethics Committee of Wuhan Union Hospital (approval number: [2023]0911). Given the retrospective observational design and the anonymization of patient data in compliance with privacy protection regulations, the Ethics Committee waived the requirement for informed consent.
Participants
This study included 336 patients with IEMs who underwent CI between 2012 and 2020. The inclusion criteria were as follows: bilateral permanent severe to profound SNHL (pure tone average >60 dB HL at 500, 1000, 2000, and 4000 Hz 18 ), speech recognition thresholds below 50% after at least 3 months of hearing aid optimization, unilateral first-time CI, normal tympanic membrane appearance, type A tympanogram, implantation age ≤14 years, and radiological confirmation of unilateral or bilateral IEMs via computed tomography (CT) and/or MRI. Exclusion criteria included conductive components in pure tone or behavioral audiometry, bilateral CI (simultaneous or sequential), cochlear implant reimplantation, severe IEMs (eg, Michel deformity, rudimentary otocyst, or cochlear aplasia), cochlear nerve aplasia or interruption, severe brain abnormalities (eg, leukoencephalopathy, cerebral palsy), other conditions significantly interfering with auditory rehabilitation or language recovery (eg, severe cognitive impairment, bilateral blindness), and incomplete radiological or clinical data.
Imaging Examinations
All patients underwent high-resolution computed tomography of the temporal bone, inner ear magnetic resonance imaging, and brain MRI before surgery. Imaging data were analyzed independently by 2 radiology specialists. IEMs were diagnosed and classified according to the 2017 Sennaroğlu classification criteria (see Table S1). 6 Cases with multiple IEM types were categorized as multiple malformations (MM) to ensure exclusivity and consistency in classification. The severity of IEMs was graded using the 2017 Adibelli grading criteria (see Table S2). 14
Preoperative Auditory and Speech Assessment
All participants underwent preoperative audiological evaluations, including acoustic immittance, behavioral audiometry (for children aged <6 years), pure tone audiometry (for children aged ≥6 years), auditory brainstem response (ABR), and auditory steady-state response (ASSR) testing. ABR and ASSR were performed on all children to objectively assess hearing thresholds. For children who could not remain still or cooperate during the tests, sedation was administered as needed to complete ABR and ASSR recordings. ASSR served as the primary objective method for estimating hearing thresholds due to its frequency specificity and suitability for children with profound hearing loss.
Auditory performance was assessed using the Categories of Auditory Performance (CAP) scale (see Table S3), and speech ability was evaluated using the Speech Intelligibility Rating (SIR) scale (see Table S4). CAP and SIR are standardized tools applicable across all age groups to measure developmental levels of auditory and speech functions.19,20 In this study, infants who had not yet developed spoken language prior to implantation (some of whom exhibited minimal or no voluntary vocalizations) were rated as Level 1 on the SIR scale. This level describes unintelligible or absent speech and is appropriate for preverbal children who primarily rely on gestures or nonverbal means of communication.
Data Analysis
Continuous variables (eg, hearing thresholds, rehabilitation duration, and implantation age) and categorical variables (eg, gender and medical history) were statistically described. Continuous variables were tested for normality and presented as mean ± standard error or median (interquartile range). Ordinal categorical variables were summarized as median (interquartile range), while nominal categorical variables were presented as counts and percentages.
Statistical description and Spearman correlation analyses were performed using SPSS 29.0 (SPSS, Chicago, IL, USA). Sunburst charts were generated using Origin 10.1 (OriginLab Corporation, Northampton, MA, USA). In R 4.3.2 (Foundation for Statistical Computing, Vienna, Austria) Venn diagrams were created with ggVennDiagram 1.5.2 (Gao et al., 2024) 21 , and chord diagrams were generated using circlize 0.4.16 (Gu et al., 2014) 22 . All statistical tests were 2-sided, with a significance threshold of P < .05.
Results
Demographic and Clinical Characteristics: Pediatric CI Candidates with IEMs
Among 336 pediatric CI candidates with IEMs, 53.6% (180 cases) were male. Perinatal histories included trauma (1 case, 0.3%), noise exposure (3 cases, 0.9%), infection (10 cases, 3.0%), medication history (15 cases, 4.5%), hypoxia (16 cases, 4.8%), cesarean delivery (198 cases, 58.9%), preterm birth (10 cases, 3.0%), and low birth weight (12 cases, 3.6%). The median durations of hearing aid use, rehabilitation, and implantation age were 23, 11, and 51 months, respectively. Preoperative hearing assessment revealed that the median hearing thresholds for the better ear were 110.25 dB HL for unaided hearing, 101.75 dB HL with hearing aids, and 78.75 dB HL for ASSR. The median V-wave threshold of the better ear measured by ABR was 105.00 dB nHL (range: 100.00-105.00). The devices implanted included the following 4 types: Cochlear (Cochlear Ltd, Melbourne, Australia) in 159 cases (47.6%), MED-EL (MED-EL Corp, Innsbruck, Austria) in 101 cases (30.2%), Advanced Bionics (Advanced Bionics LLC, Valencia, CA, USA) in 66 cases (19.8%), and NUROTRON (Zhejiang Nurotron Biotechnology Co Ltd, Hangzhou, China) in 8 cases (2.4%). Preoperative CAP and SIR median scores for all patients were 1 and 1, respectively (Table 1).
Demographic and Clinical Characteristics of Children With IEMs for CI.

Abbreviations: ABR, auditory brainstem response; ASSR, auditory steady-state response; C1-C4, cochlear implantation center pseudonyms; CAP, categories of auditory performance; CI, cochlear implantation; CNY, chinese yuan; IEMs, inner ear malformation; IQR, interquartile range; R1-R10, rehabilitation facility pseudonyms; SIR, speech intelligibility rating.
Comorbid Relationships Among Different Types of IEMs
The prevalence and comorbidity patterns of different types of IEMs among pediatric CI candidates are illustrated in Figure 1. In patients, vestibular aqueduct malformation (VAM) was the most common malformation, accounting for 64.9% (218 cases), followed by cochlear malformations (CM) at 31.5% (106 cases), vestibular and semicircular canal malformations (VSCM) at 22.3% (75 cases), internal auditory canal malformations (IACM) at 13.1% (44 cases), and cochlear nerve malformation (CNM), which were the least common, at 2.3% (8 cases). Comorbidity analysis revealed that VSCM had the highest comorbidity rate (65.3%), frequently involving other inner ear anatomical regions. CNM (62.5%), CM (62.3%), and IACM (52.3%) also exhibited comorbidity rates exceeding 50%. In contrast, VAM had the lowest comorbidity rate at 28.4%, significantly lower than the other 4 types of malformations.
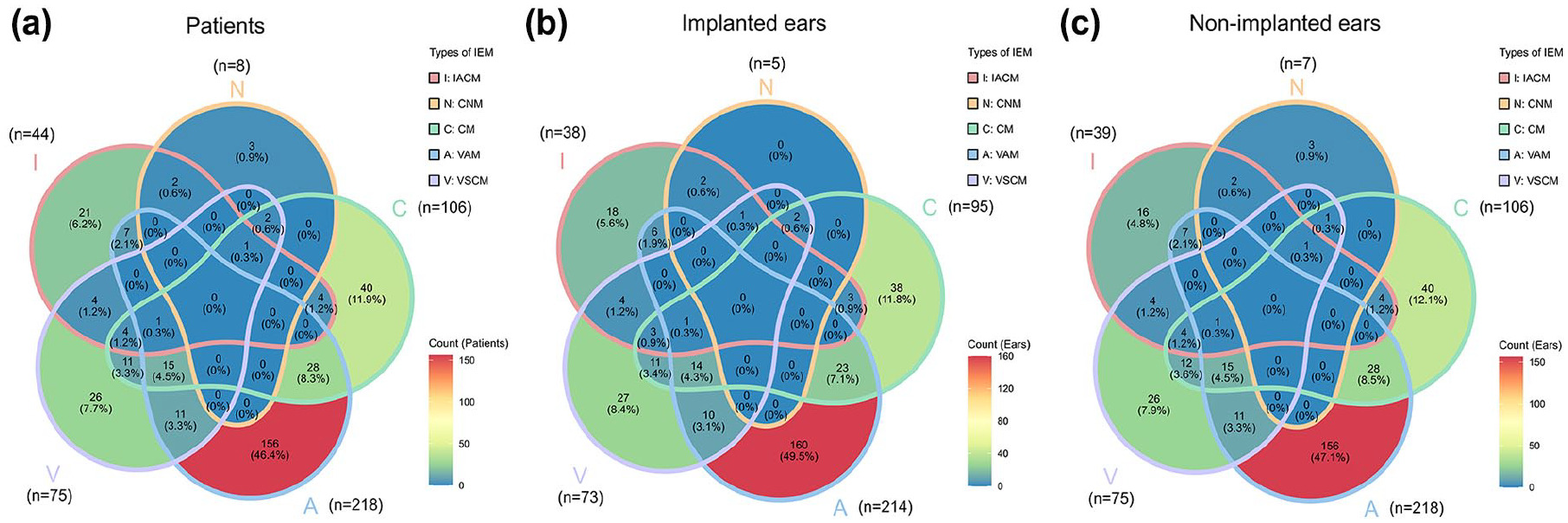
Comorbid relationships among different classifications of malformations in patients (a), implanted ears (b), and non-implanted ears (c) in children with IEMs. VAM was primarily identified as an isolated subtype, while IACM, CNM, CM, and VSCM frequently co-occurred, with over half of these cases affecting multiple inner ear regions. IACM, internal auditory canal malformation; CNM, cochlear nerve malformation; CM, cochlear malformation; VAM, vestibular aqueduct malformation; VSCM, vestibular and semicircular canal malformation; IEM, inner ear malformation.
The distribution of IEMs in the implanted ears is shown in Figure 1b. VAM and VSCM accounted for 66.3% (214 ears) and 22.6% (73 ears), respectively, while CM made up 29.4% (95 ears). The proportions of IACM and CNM were 11.8% (38 ears) and 1.5% (5 ears), respectively. Compared with those in patients, the proportions of IACM, CNM, and CM in the implanted ears showed a slight decrease, while the proportions of VAM and VSCM slightly increased.
The distribution of IEMs in the non-implanted ears is shown in Figure 1c. VAM accounted for 65.9% (218 ears), CM for 32.0% (106 ears), and VSCM for 22.7% (75 ears). The proportions of IACM and CNM were 11.8% (39 ears) and 2.1% (7 ears), respectively. Compared with the implanted ears, non-implanted ears showed a slight decrease in the proportion of VAM, while CM, CNM, and VSCM exhibited slight increases.
Overall, the results revealed significant differences in the distribution of IEMs across patients, implanted ears, and non-implanted ears. VAM had the highest prevalence in all 3 groups, whereas CNM was the least common. The comorbidity rates of VSCM, CNM, CM, and IACM exceeded 50%, with VSCM having the highest comorbidity rate. In contrast, VAM showed a significantly-lower comorbidity rate than the other 4 types of IEMs (Figure 1).
Correlations Between the Severity of Different Types of IEMs
Figure 2 demonstrates the correlations among severity grades of different IEM types at 3 levels: (a) patients, (b) implanted ears, and (c) non-implanted ears in pediatric cochlear implant candidates. In patients (Figure 2a), the severity of IACM was significantly positively correlated with CNM (r = .13, P = .015), and CM was also significantly positively correlated with VSCM (r = .17, P < .001). Conversely, the severity of VAM was significantly negatively correlated with the severity of all 4 other types of malformations, including IACM (r = −.39, P < .001), CNM (r = −.21, P < .001), CM (r = −.38, P < .001), and VSCM (r = −.30, P < .001).
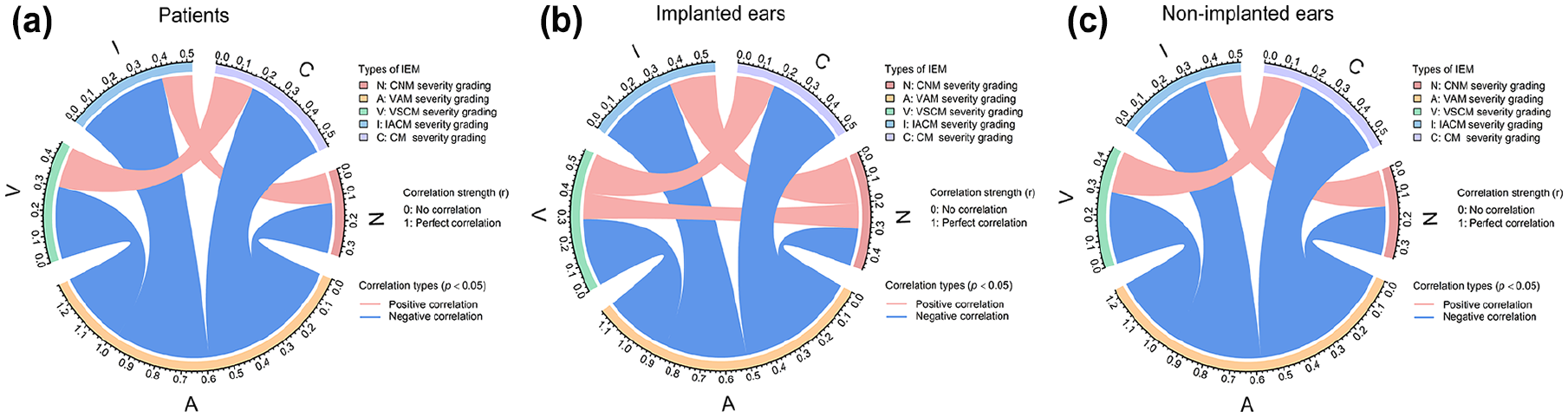
Correlations between the severity grading of different classifications of malformations in patients (a), implanted ears (b), and non-implanted ears (c) in children with IEMs. IACM severity positively correlated with CNM, and CM severity with VSCM. Conversely, EVA severity was negatively correlated with IACM, CNM, CM, and VSCM. IACM, internal auditory canal malformation; CNM, cochlear nerve malformation; CM, cochlear malformation; VAM, vestibular aqueduct malformation; VSCM, vestibular and semicircular canal malformation; IEM, inner ear malformation; EVA, enlarged vestibular aqueduct.
Analysis of the implanted ears (Figure 2b) revealed similar patterns to those in patients, with significant positive correlations between IACM and CNM (r = .20, P < .001), as well as between CM and VSCM (r = .17, P = .001). The severity of VAM remained significantly negatively correlated with all 4 other malformations, including IACM (r = −.35, P < .001), CNM (r = −.16, P = .002), CM (r = −.35, P < .001), and VSCM (r = −.29, P < .001). Additionally, in the implanted ears, a novel positive correlation was observed between CNM and VSCM (r = .11, P = .04).
Results from the non-implanted ears (Figure 2c) mirrored those of the patients, with significant positive correlations between IACM and CNM (r = .15, P = .004) and between CM and VSCM (r = .17, P = .001). The severity of VAM remained significantly negatively correlated with the severity of all other malformations, including IACM (r = −.36, P < .001), CNM (r = −.20, P < .001), CM (r = −.38, P < .001), and VSCM (r = −.30, P < .001).
Overall, the correlations between the severity of different IEM types followed consistent patterns across patients, implanted ears, and non-implanted ears. IACM was positively correlated with CNM, and CM was positively correlated with VSCM. Conversely, VAM was negatively correlated with all other malformations. These correlations were more pronounced in the implanted ears, with the addition of a novel positive correlation between CNM and VSCM.
Subtype Characteristics of IEMs
Figure 3 illustrates the distribution of IEM subtypes across 3 categories: patients (showing the number of children with each malformation subtype), implanted ears (showing the number of implanted ears with each malformation subtype), and non-implanted ears (showing the number of non-implanted ears with each malformation subtype). In patients (Figure 3a), isolated enlarged vestibular aqueduct (EVA) was the most common subtype, accounting for 46.4% (156 cases). Other relatively-common subtypes included isolated incomplete partition type II (IP-II) (17.3%, 58 cases), MM (17.0%, 57 cases), isolated semicircular canal malformations (SCCM) (4.8%, 16 cases), and isolated cochlear hypoplasia type IV (CH-Ⅳ) (4.8%, 16 cases). Isolated enlarged internal auditory canal (EIAC), isolated narrow internal auditory canal (NIAC), and isolated vestibular enlargement (EV) accounted for 3.9% (13 cases), 2.4% (8 cases), and 2.4% (8 cases), respectively. The prevalence of isolated cochlear nerve hypoplasia (CNH) and isolated cochlear hypoplasia type III (CH-Ⅲ) was 0.9% (3 cases) and 0.3% (1 case), respectively. Among these, only one common cavity deformity was identified, which was classified as a multiple malformation.
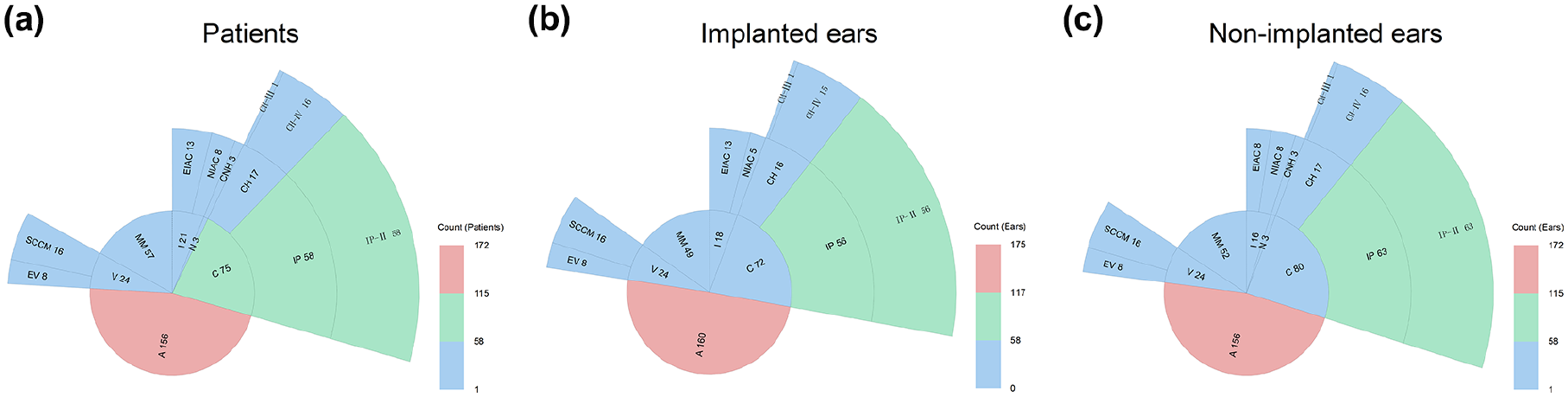
Subtype characteristics of patients (a), implanted ears (b), and non-implanted ears (c) in children with IEMs. The 3 predominant subtypes—isolated EVA, isolated IP-II, and MM—together comprised approximately 80.7% of cases. MM, multiple malformations; EIAC, enlarged internal auditory canal; NIAC, narrow internal auditory canal; CNH, cochlear nerve hypoplasia; CH, cochlear hypoplasia; IP, incomplete partition; EV, enlarged vestibular; EVA, enlarged vestibular aqueduct; SCCM, semicircular canal malformations; CH-Ⅲ, cochlear hypoplasia type III; CH-Ⅳ, cochlear hypoplasia type IV; and IP-Ⅱ, incomplete partition type II; IEM, inner ear malformation.
The subtype characteristics of the implanted ears are shown in Figure 3b. In implanted ears, the proportions of MM (15.2%, 49 ears), isolated CH-Ⅳ (4.6%, 15 ears), isolated NIAC (2.4%, 5 ears), and isolated CNH (0%, 0 ears) were lower than those in patients. Conversely, the proportions of isolated EVA (49.5%, 160 ears), isolated SCCM (5.0%, 16 ears), isolated EIAC (4.0%, 13 ears), and isolated EV (2.5%, 8 ears) were higher than those in patients. The proportions of isolated IP-II (17.3%, 56 ears) and isolated CH-Ⅲ (0.3%, 1 ear) were consistent with those in patients.
The subtype characteristics of the non-implanted ears are shown in Figure 3c. In non-implanted ears, the proportions of MM (15.7%, 52 ears) and isolated EIAC (2.4%, 8 ears) were lower than those in patients. In comparison, the proportions of isolated EVA (47.1%, 156 ears) and isolated IP-II (19.0%, 63 ears) were higher. The proportions of isolated SCCM (4.8%, 16 ears), isolated CH-Ⅳ (4.8%, 16 ears), isolated NIAC (2.4%, 8 ears), isolated EV (2.4%, 8 ears), isolated CNH (0.9%, 3 ears), and isolated CH-Ⅲ (0.3%, 1 ear) were consistent with those in patients.
Isolated EVA was the most common subtype of IEMs, with the highest proportion observed in implanted ears (49.5%), compared with that in patients (46.4%) and that in non-implanted ears (47.1%). The proportions of MM and isolated CNH were significantly lower in implanted ears than in patients. In contrast, the proportions of isolated SCCM, isolated EIAC, and isolated EV were slightly higher. In non-implanted ears, the proportion of isolated IP-II (19.0%) was higher than that in patients, while the distribution of other isolated subtypes was generally consistent with patients.
Discussion
This study preliminarily explored the cohort characteristics of pediatric CI candidates with IEMs, focusing on subtype characteristics, comorbidity relationships, and severity correlations. Our findings revealed the following: (1) VAM was predominantly observed as a single subtype, whereas IACM, CNM, CM, and VSCM were frequently comorbid, with more than half of these cases involving other inner ear regions. (2) The severity of IACM was positively correlated with the severity of CNM, and the severity of CM was positively correlated with VSCM. In contrast, the severity of EVA was negatively correlated with the severity of IACM, CNM, CM, and VSCM. (3) Among CI candidates with IEMs, the 3 most common subtypes were isolated EVA, isolated IP-II, and MM, collectively accounting for approximately 80.7% of cases.
Previous studies have reported substantial variations in the subtype characteristics of IEMs among pediatric CI candidates.9,17,23 These discrepancies may stem from factors such as sample size, diagnostic and classification criteria, and regional differences. The findings of this study are similar to those reported by Shi et al, particularly in the ratios of EVA and IP-II. 23 These 2 subtypes accounted for a relatively high proportion of cases in our sample, likely due to 2 reasons. First, these malformations have a high prevalence rate. Second, patients with these subtypes often achieve favorable rehabilitation outcomes following CI surgery, leading to a higher proportion of these patients being selected for CI procedures.24,25
The high prevalence of EVA and IP-II can be explained from a genetic perspective. Both conditions are recognized as autosomal recessive disorders closely associated with mutations in the SLC26A4 gene located on chromosome 7q31.26-28 Studies have identified SLC26A4 mutations as the primary pathogenic factor for EVA and IP-II, with several mutation sites exhibiting mutant allele frequencies exceeding 50% across different ethnic populations.29,30 This mode of inheritance accounts for the high prevalence of these 2 IEMs in the population.
This study is the first to report the proportion of MM in IEMs, accounting for 17.0%. Although the proportion of MM is comparable to that of isolated IP-II, MM involves multiple inner ear structures and may exhibit complex interactions among malformations. Comprehensive consideration of these interactions during clinical evaluation is essential to optimize CI outcomes and postoperative rehabilitation. Enhancing systematic assessment for patients with MM can aid in developing more precise, individualized treatment strategies, ultimately improving postoperative auditory and speech function recovery.
This study revealed a widespread comorbidity of IEMs across various inner ear anatomical structures, which was particularly notable in IACM, CNM, CM, and VSCM. However, this comorbidity was less common in VAM. Due to the typically-small sample sizes and diverse classification standards in previous studies, this phenomenon has not been adequately recognized. Notably, the observed comorbidity spans multiple subtypes, indicating inherent heterogeneity. Addressing this heterogeneity requires larger scale samples to reduce confounding bias, presenting significant challenges for in-depth statistical analyses.
Comorbidity, or co-occurrence, forms the basis for correlation, with widespread comorbidity suggesting potential underlying associations. This study further investigated the correlations between the severity of various IEMs. We found a positive correlation between the severity of IACM and CNM, primarily due to their close anatomical relationship. NIAC can compress microvessels, leading to reduced blood flow and decreased supply of neurotrophic factors to the cochlear nerve. This subsequently affects nerve fiber growth and myelination, impairing auditory signal transmission.31-33 Therefore, during preoperative CI evaluations, special attention should be paid to the integrity and functional status of the cochlear nerve in patients with IACM to more accurately predict postoperative auditory outcomes.
Additionally, this study identified a negative correlation between the severity of VAM and the severity of IACM, CNM, CM, and VSCM. This phenomenon can be attributed to the fact that EVA malformations often occur as isolated EVA, with a much lower comorbidity rate than in the other 4 types of IEMs. As a result, the presence of EVA significantly reduces the likelihood of other types of IEMs, leading to lower severity grades for these malformations.
Regarding the positive correlation between the severity of CM and VSCM, both structures originate from the otic vesicle during embryonic development between the 4th and 8th weeks. During this period, the otic vesicle differentiates into the cochlea and vestibular semicircular canals under the regulation of similar signaling pathways, including the Wingless-type (WNT), fibroblast growth factor (FGF), and Sonic Hedgehog (SHH).34-36 Disruption of these signaling pathways during development can simultaneously affect the formation of the cochlea and vestibular semicircular canals, resulting in a positive correlation in their severity. This finding suggests that during preoperative evaluations, the presence of CM should prompt further examination of the vestibular semicircular canals to comprehensively plan surgical strategies.
Limitations and Future Directions
The primary limitation of this study is that the participants were restricted to candidates for CI, rather than all children with IEMs. This restriction may prevent the results from fully reflecting the characteristics of all IEM cases, particularly those not recommended for CI surgery. Additionally, as the data were derived from a multicenter retrospective analysis, variations in evaluation methods and record-keeping among rehabilitation centers may have affected data uniformity, despite the large sample size. Future research should broaden the scope to include children with IEMs who have not undergone CI, enabling a more comprehensive understanding of the impact of IEMs on hearing and language development. Prospective study designs should be adopted to improve data consistency. Furthermore, advancements in imaging technologies and data analysis methods will facilitate deeper exploration of the molecular mechanisms and genetic backgrounds of different malformation types, aiding in the development of more precise, individualized treatment strategies.
Conclusion
This study systematically analyzed pediatric CI candidates with IEMs, revealing significant comorbid relationships among IACM, CNM, CM, and semicircular canal malformations. The findings demonstrated positive correlations between the severity of IACM and CNM, as well as between CM and semicircular canal malformations, suggesting that their anatomical interrelationships may influence surgical planning and outcomes. In contrast, VAM was predominantly observed as an isolated malformation and exhibited negative correlations with the severity of other malformations. By utilizing advanced visualization tools, this study provides new insights into the anatomical complexity of IEMs, which may contribute to improved preoperative assessments and individualized treatment strategies.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251342846 – Supplemental material for Cohort Characteristics of Pediatric Candidates for Cochlear Implantation With Inner Ear Malformations: A Multicenter Retrospective Study
Supplemental material, sj-docx-1-ear-10.1177_01455613251342846 for Cohort Characteristics of Pediatric Candidates for Cochlear Implantation With Inner Ear Malformations: A Multicenter Retrospective Study by Hejie Li, Wenting Yu, Wei Tang, Ting Li, Xuan Yu, Shimin Zong and Hongjun Xiao in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
We would like to express our sincere gratitude to the following institutions in China for their invaluable contributions in providing anonymized data for this study: Department of Rehabilitation for Children with Hearing Impairments, Hubei Disabled Persons’ Federation Rehabilitation Center, Wuhan; Department of Speech Training, Hubei Deaf Infant Healing Center, Wuhan; Wuhan Yimeng Hearing Rehabilitation Center, Wuhan; Huangshi Rehabilitation Center for Persons with Disabilities, Huangshi; Jingzhou Yimeng Hearing and Speech Rehabilitation Center, Jingzhou; Jingmen Menghuan Children’s Rehabilitation Center, Jingmen; Huanggang Qisheng Hearing and Speech Rehabilitation Center, Huanggang; Xianning Hearing and Speech Rehabilitation Center, Xianning; Enshi Yimeng Hearing and Speech Rehabilitation Center, Enshi; and Xiaogan Yide Hearing and Speech Rehabilitation Center, Xiaogan. These institutions were not involved in any other aspect of the study, including its design, analysis, or the reporting of results.
Ethical Considerations
This study was approved by the Research Ethics Board at Wuhan Union Hospital (approval number: [2023]0911).
Author Contributions
Hejie Li, Wenting Yu: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, visualization. Shimin Zong, Hongjun Xiao: Conceptualization, methodology, validation, supervision, project administration, funding acquisition, writing—review and editing. Wei Tang, Ting Li, Xuan Yu: Formal analysis, investigation, data curation, writing—original draft, visualization.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant numbers 82071057, 82101229) and Key Research and Development Program of Hubei Province Project (Grant 2021BCA144).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the 10 rehabilitation institutions (as described previously). Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the corresponding authors with the permission of the 10 rehabilitation institutions (as described previously).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
